Editor's Picks
Open Access
Review
The influence of sex and gender differences in shaping the immune response to influenza infection and vaccination
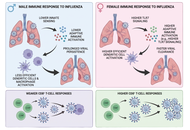
This review highlights the significant sex-based differences in immune responses to influenza infection and vaccination. Men are generally more susceptible to severe influenza outcomes, while women often mount stronger immune responses but experience more adverse effects. These disparities are influenced by biological factors, including sex hormones and genes, as well as gender-related social and environmental conditions. Evidence from both human and animal studies reveals sex-specific variations in antibody production, vaccine effectiveness, and clinical outcomes. Age, hormonal status, and stress further modulate these differences. Understanding these complex interactions is essential for developing tailored and equitable vaccination and treatment strategies.
Open Access
Review
Immuno-materials: exploring the translational aspects among immunology, biomaterials and biomedicine
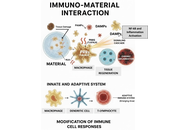
Immuno-materials, which represent a confluence of immunology and materials science, are dramatically transforming the fields of healthcare. Through the targeted modulation of immune responses, these innovative materials offer promising avenues for advancements in vaccine development, immunotherapy, tissue engineering, and diagnostics. This review examines the fundamental principles of immuno-materials, elucidating key concepts and methodologies pertinent to their development. We investigate strategies for engineering materials that can elicit customized immune responses, capable of both stimulation and suppression, and we discuss their potential roles as adjuvants, carriers, or scaffolds. Furthermore, we address the challenges and opportunities associated with the clinical translation of these materials, emphasizing the critical importance of biocompatibility, safety, and efficacy. This review aims to critically evaluate and contextualize the transformative potential of immuno-materials, mapping their journey from laboratory design to proven clinical applications in enhancing human health.
Open Access
Editorial
The neutrophil-to-lymphocyte ratio in aging and immunosenescence
In recent years the number of publications reporting the use of the neutrophil/lymphocyte ratio (NLR) in disparate fields of human and veterinary medicine has swelled in the medical literature from less than 50/year in 2010 to 892 in 2019, and this figure has more than doubled up to 1,857 in 2024 [1].
Articles
Latest
Most Viewed
Most Downloaded
Most Cited
Open Access
Review
Epigenetic and metabolic reprogramming in autoimmune rheumatology: toward immune tolerance reprogramming
Ola A Al-Ewaidat, Moawiah M Naffaa
Published: May 29, 2026 Explor Immunol. 2026;6:1003255
Open Access
Review
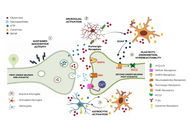
Is neuroinflammation the common thread in chronic pain? Recent advances and implications of the role of glial cells
Silvia Natoli ... Maurizio Marchesini
Published: May 27, 2026 Explor Immunol. 2026;6:1003254
This article belongs to the special issue Immunology and Pain
Open Access
Perspective
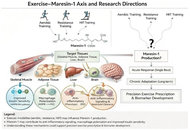
Does exercise enhance maresin-1 production? A new direction in metabolic anti-inflammatory research
Mehran Alipour ... Hassane Zouhal
Published: May 09, 2026 Explor Immunol. 2026;6:1003253
This article belongs to the special issue Physical Activity and Immune System in Chronic Diseases: Mechanisms and Insights
Open Access
Original Article
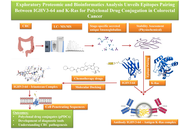
Exploratory proteomic and bioinformatics analysis unveils epitope pairing between IGHV3-64 and K-Ras for polyclonal drug conjugation in colorectal cancer
Raajesh Anand ... Ravikumar Sambandam
Published: May 07, 2026 Explor Immunol. 2026;6:1003252
Open Access
Review
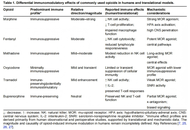
Balancing analgesia and immunity: revisiting the immune consequences of opioid therapy
Lucia Daiana Voiculescu ... Sanjay Menghani
Published: April 28, 2026 Explor Immunol. 2026;6:1003251
This article belongs to the special issue Immunology and Pain
Open Access
Original Article
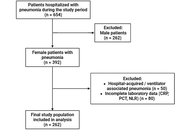
C-reactive protein, procalcitonin, and neutrophil-to-lymphocyte ratio as mortality predictors in elderly women with pneumonia
Irawaty Djaharuddin ... Demak Lumban Tobing
Published: April 23, 2026 Explor Immunol. 2026;6:1003250
Open Access
Review
Exploring the possible link between the spike protein immunoglobulin G4 antibodies and cancer progression
Mikolaj Raszek ... Alberto Rubio-Casillas
Published: April 25, 2024 Explor Immunol. 2024;4:267–284
This article belongs to the special issue Old and New Paradigms in Viral Vaccinology
Open Access
Review
Rheumatoid arthritis: a complex tale of autoimmune hypersensitivity
Jihye Heo ... Jea-Hyun Baek
Published: June 20, 2024 Explor Immunol. 2024;4:358–375
This article belongs to the special issue Chronic Inflammation and Autoimmunity
Open Access
Review
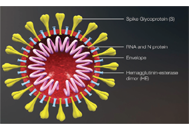
COVID-19 vaccine and immune response
Sevilay Hintistan, Hatice Demirağ
Published: December 31, 2021 Explor Immunol. 2021;1:341–355
This article belongs to the special issue Vaccine-induced Immune Responses Against SARS-CoV-2 Infections
Open Access
Review
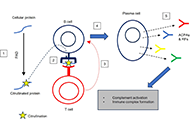
The future of complement therapeutics
Martin Kolev ... Pascal Deschatelets
Published: October 18, 2024 Explor Immunol. 2024;4:577–615
Open Access
Review
Critical role of keratinocytes in cutaneous immune responses
Rakesh Kumar Gupta ... Dhananjay Shukla
Published: August 27, 2024 Explor Immunol. 2024;4:502–522
Open Access
Review
Neoantigen vaccines: advancing personalized cancer immunotherapy
Alaa A. A. Aljabali ... Lorca Alzoubi
Published: April 08, 2025 Explor Immunol. 2025;5:1003190
Open Access
Review
Exploring the possible link between the spike protein immunoglobulin G4 antibodies and cancer progression
Mikolaj Raszek ... Alberto Rubio-Casillas
Published: April 25, 2024 Explor Immunol. 2024;4:267–284
This article belongs to the special issue Old and New Paradigms in Viral Vaccinology

Open Access
Review
The role of cytokines in wound healing: from mechanistic insights to therapeutic applications
Rachel Si-Yin Wong ... Dinesh Kumar Srinivasan
Published: February 11, 2025 Explor Immunol. 2025;5:1003183
Open Access
Review
The future of complement therapeutics
Martin Kolev ... Pascal Deschatelets
Published: October 18, 2024 Explor Immunol. 2024;4:577–615

Open Access
Review
Entrance to the multifaceted world of CD4+ T cell subsets
Murilo Porfírio de Aguiar, Julia Hailer Vieira
Published: March 05, 2024 Explor Immunol. 2024;4:152–168
Open Access
Review
Chemokines and nanomaterials: interaction for useful immune-applications
Giuseppe Bardi
Published: August 31, 2022 Explor Immunol. 2022;2:637–647
Open Access
Review
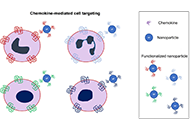
Immunology of biodegradable nanoparticles: a brief overview on a wide growing field
Anissa Pisani, Giuseppe Bardi
Published: April 30, 2021 Explor Immunol. 2021;1:48–60
Open Access
Review
The role of cytokines in wound healing: from mechanistic insights to therapeutic applications
Rachel Si-Yin Wong ... Dinesh Kumar Srinivasan
Published: February 11, 2025 Explor Immunol. 2025;5:1003183

Open Access
Review
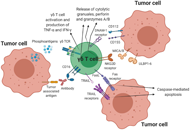
Allogeneic gamma delta T cells as adoptive cellular therapy for hematologic malignancies
Navdeep Jhita, Sunil S. Raikar
Published: June 07, 2022 Explor Immunol. 2022;2:334–350
This article belongs to the special issue Interplay of γδ T cells and Tumor Cells
Open Access
Review
Crosstalk between keratinocytes and immune cells in inflammatory skin diseases
Xinhui Ni, Yuping Lai
Published: December 31, 2021 Explor Immunol. 2021;1:418–431
This article belongs to the special issue Cross Talk Among Skin Cells and Immune Cells
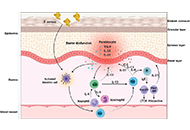
Open Access
Review
High avidity of vaccine-induced immunoglobulin G against SARS-CoV-2: potential relevance for protective humoral immunity
Georg Bauer
Published: March 16, 2022 Explor Immunol. 2022;2:133–156
This article belongs to the special issue Vaccine-induced Immune Responses Against SARS-CoV-2 Infections
Open Access
Review
Critical role of keratinocytes in cutaneous immune responses
Rakesh Kumar Gupta ... Dhananjay Shukla
Published: August 27, 2024 Explor Immunol. 2024;4:502–522

Open Access
Review
Neoantigen vaccines: advancing personalized cancer immunotherapy
Alaa A. A. Aljabali ... Lorca Alzoubi
Published: April 08, 2025 Explor Immunol. 2025;5:1003190

Special Issues
Ongoing Special lssues
Completed Special lssues
Old and New Paradigms in Cancer Immune Response and Immunotherapy
Guest Editor: Dr. Ángel Miguel García Lora
Submission Deadline: August 31, 2026
Published Articles: 0

Immunogenetics in Neurological and Neuropsychiatric Disorders
Guest Editors: Dr. Franca Rosa Guerini; Dr. Elisabetta Bolognesi
Submission Deadline: July 31, 2026
Published Articles: 0

Obesity, Inflammation, and Immuno-Oncology: The Adipose Tissue Perspective
Guest Editors: Dr. Federico Greco; Prof. Bruno Beomonte Zobel
Submission Deadline: July 31, 2026
Published Articles: 0

The Microbiome-Autoimmunity Axis: Mechanisms and Therapeutic Implications
Guest Editor: Dr. Jixin Zhong
Submission Deadline: July 31, 2026
Published Articles: 0
Metabolic Control of Acute Immune Response
Guest Editor: Dr. Didier Payen
Submission Deadline: July 31, 2026
Published Articles: 0

Immunogenetics of Chronic Illnesses
Guest Editors: Dr. Apostolos P. Georgopoulos; Dr. Lisa M. James
Submission Deadline: August 31, 2026
Published Articles: 1

Immuno-Materials: at the interdisciplinary of immunology and materials
Guest Editor: Dr. Wantong Song
Submission Deadline: August 31, 2026
Published Articles: 1

Immunology of Transplantation
Guest Editor: Prof. Andrzej Lange
Submission Deadline: July 31, 2026
Published Articles: 1
Novel Vaccines development for Emerging, Acute, and Re-emerging Infectious Diseases
Guest Editors: Dr. Wenping Gong; Dr. Ashok Aspatwar
Submission Deadline: August 31, 2026
Published Articles: 4

Advances in Cellular and Molecular Treatment of Autoimmune Diseases
Guest Editor: Prof. Sofia Kossida
Submission Deadline: November 30, 2026
Published Articles: 4

The Nutritional Influence on Immune Functionality
Guest Editor: Dr. Lucia Malaguarnera
Submission Deadline: July 31, 2026
Published Articles: 3

Hypersensitivity Syndrome Reactions versus Allergy and Drug
Guest Editor: Dr. Manuela Neuman
Submission Deadline: June 30, 2026
Published Articles: 2

Immunology and Pain
Guest Editors: Dr. Giustino Varrassi; Dr. Matteo Leoni; Dr. Martina Rekatsina
Submission Deadline: May 01, 2026
Published Articles: 9
Physical Activity and Immune System in Chronic Diseases: Mechanisms and Insights
Guest Editors: Prof. Hassane Zouhal; Prof. Ismail Laher
Submission Deadline: June 30, 2026
Published Articles: 3

Autoantibodies Associated to Thrombosis and Hemostasis
Guest Editor: Dr. Jean Amiral
Submission Deadline: August 31, 2026
Published Articles: 6
Old and New Paradigms in Viral Vaccinology
Guest Editors: Dr. Marc H.V. Van Regenmortel; Dr. Vladimir N. Uversky
Submission Deadline: July 16, 2025
Published Articles: 12

Journal Information
Journal Indexing
Journal Metrics

























 Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys
Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys


