Editor's Picks
Open Access
Original Article
Application of TELC model to better elucidate neural stimulation by touch
Aim:
This study is to better understand how the transient ion transport activity of touch receptors could change the graded potential to stimulate an action potential firing.
Methods:
The latest transmembrane-electrostatically localized protons/cations charges (TELC) theory is employed for numerical analysis to calculate the neural touch signal transduction responding time required to fire an action potential spike.
Results:
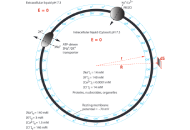
A neural action potential spike was constructed successfully using newly developed time-dependent TELC-based neural transmembrane potential integral equations (Equations 5, 6, and 7). The results explicated that the TELC curve has an inverse relationship with neural transmembrane potential since its curve appears as an inverse mirror image to the action potential spike. Based on the TELC density at resting membrane potential of –70 mV calculated to be 3,900 (excess protons + cations) per μm2 and that at the stimulation threshold level (–55 mV) calculated to be 3,100 (excess protons + cations) per μm2 on extracellular membrane surface, the neural touch signal transduction responding time from PIEZO channel ion conduction to reduce the TELC density to the stimulation level of 3,100 TELC per μm2 has now, for the first time, been calculated for action potential firing.
Conclusions:
The activity of a single or a few PIEZO channels may be sufficient to generate a “graded potential” to trigger an action potential spike firing. With a high number (200–300) of PIEZO channels activated by touch, it can generate the required “graded potential” to reach the stimulation threshold level (–55 mV) within a neural touch signal transduction time as fast as 0.3 ms. The calculated neural touch signal transduction responding time (e.g., 0.3 ms) may have fundamental implications not only for neuroscience but also for other science and technology fields such as bioengineering and sports physiology.
Open Access
Review
Melatonin regulation of phase separation in Neuro-PASC: out-maneuvering Janus-faced amyloids
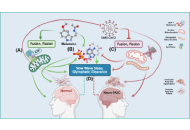
The SAR-CoV-2 virus has evolved to co-exist with human hosts, albeit at a substantial energetic cost resulting in post-infection neurological manifestations [Neuro-post-acute sequelae of SARS-CoV-2 infection (PASC)] that significantly impact public health and economic productivity on a global scale. One of the main molecular mechanisms responsible for the development of Neuro-PASC, in individuals of all ages, is the formation and inadequate proteolysis/clearance of phase-separated amyloid crystalline aggregates—a hallmark feature of aging-related neurodegenerative disorders. Amyloidogenesis during viral infection and persistence is a natural, inevitable, protective defense response that is exacerbated by SARS-CoV-2. Acting as chemical catalyst, SARS-CoV-2 accelerates hydrophobic collapse and the heterogeneous nucleation of amorphous amyloids into stable β-sheet aggregates. The clearance of amyloid aggregates is most effective during slow wave sleep, when high levels of adenosine triphosphate (ATP)—a biphasic modulator of biomolecular condensates—and melatonin are available to solubilize amyloid aggregates for removal. The dysregulation of mitochondrial dynamics by SARS-CoV-2, in particular fusion and fission homeostasis, impairs the proper formation of distinct mitochondrial subpopulations that can remedy challenges created by the diversion of substrates away from oxidative phosphorylation towards glycolysis to support viral replication and maintenance. The subsequent reduction of ATP and inhibition of melatonin synthesis during slow wave sleep results in incomplete brain clearance of amyloid aggregates, leading to the development of neurological manifestations commonly associated with age-related neurodegenerative disorders. Exogenous melatonin not only prevents mitochondrial dysfunction but also elevates ATP production, effectively augmenting the solubilizing effect of the adenosine moiety to ensure the timely, optimal disaggregation and clearance of pathogenic amyloid aggregates in the prevention and attenuation of Neuro-PASC.
Open Access
Original Article
Somatostatin-expressing inhibitory neurons with mTORC1 activation in cortical layers 4/5 are involved in the epileptogenesis of mice
Aim:
Patients with tuberous sclerosis complex (TSC) which is caused by hyperactivation of mechanistic target of rapamycin complex 1 (mTORC1) often show giant cells in the brain. These giant cells are thought to be involved in epileptogenesis, but the underlying mechanisms are unknown. In this study, we focused on mTORC1 activation and γ-amino butyric acid (GABA)ergic signaling in somatostatin-expressing inhibitory neurons (SST-INs) using TSC-related epilepsy model mice.
Methods:
We analyzed the 8-week-old Tsc2 conditional knockout (Tsc2 cKO) mice, which have epileptic seizures that are cured by sirolimus, an mTORC1 inhibitor. After the occurrence of epileptic seizures was confirmed, Tsc2 cKO mice were treated with vehicle or sirolimus. Then, their brains were investigated by hematoxylin and eosin staining, immunohistochemical staining and immunoblotting assay.
Results:
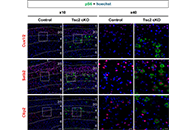
As in TSC patients, giant cells with hyperactivation of mTORC1 were found in the cerebral cortex of Tsc2 cKO mice. These giant cells were mainly SST-INs in the cortical layers 4/5. Giant cells showed decreased expression of GABA type A receptor subunit α1 (GABAAR-α1) compared with normal size cells in control mice and Tsc2 cKO mice. In addition, decreased GABAAR-α1 expression was also confirmed by immunoblotting assay of the whole cerebral cortex. In the cerebral cortex of sirolimus-treated Tsc2 cKO mice, whose epileptic seizures were cured, decreased GABAAR-α1 expression was recovered to the same level as in control mice.
Conclusions:
These results suggest that the epileptic seizures in Tsc2 cKO mice are caused by the deregulation of GABAergic signaling through mTORC1 activation of SST-INs localized in cortical layers 4/5.
Articles
Latest
Most Viewed
Most Downloaded
Most Cited
Open Access
Review
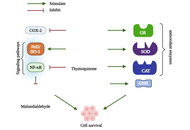
Thymoquinone in Alzheimer’s disease: experimental evidence and neuroprotective mechanisms
Jamil A. Chahrour ... Akram Hijazi
Published: May 14, 2026 Explor Neurosci. 2026;5:1006137
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection (Vol II)
Open Access
Review
Protein aggregation in progressive myoclonus epilepsies and related syndromes
Eva Žerovnik
Published: May 08, 2026 Explor Neurosci. 2026;5:1006136
This article belongs to the special issue Advances in Epilepsy Research

Open Access
Original Article
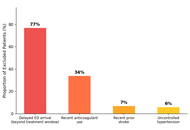
Stroke burden and epidemiological trends in Saudi Arabia: 1990–2021
Ahmed Abdulaziz Almohammadi
Published: April 27, 2026 Explor Neurosci. 2026;5:1006135
This article belongs to the special issue The Science of Ischemic Stroke
Open Access
Original Article
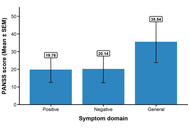
Redox-oxidative stress and micronutrients interplay in the clinical phenotype of schizophrenia in Nigerian subjects
Tolutope Adebimpe Oso ... Oluyemi Akinloye
Published: April 27, 2026 Explor Neurosci. 2026;5:1006134
Open Access
Review
Microbiota–mitochondria crosstalk in the gut–brain axis: a missing link in the etiology and treatment of major depressive disorder
Oluwagbenga Mayowa Adu ... Shukurat Joy Ajenikoko
Published: April 21, 2026 Explor Neurosci. 2026;5:1006133
This article belongs to the special issue Depression: From Pathophysiology to Treatment Innovation
Open Access
Review
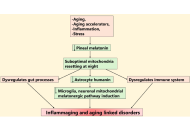
Circadian astrocyte mitochondrial humanin regulates microglia and neuronal mitochondrial melatonergic pathway, with implications for aging and neurodegenerative conditions
George Anderson
Published: April 15, 2026 Explor Neurosci. 2026;5:1006132
Open Access
Review
Current therapeutics for Alzheimer’s disease and clinical trials
Danqing Xiao, Chen Zhang
Published: June 27, 2024 Explor Neurosci. 2024;3:255–271
This article belongs to the special issue Alzheimer’s Disease
Open Access
Review
Effects mediated by melatonin and cortisol of artificial light and noise, alone and in combination, on sleep and health
Nahum M. Gabinet
Published: September 13, 2024 Explor Neurosci. 2024;3:382–417
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Review
Impact of circadian clock dysfunction on human health
Saptadip Samanta, Sk Asif Ali
Published: September 29, 2022 Explor Neurosci. 2022;1:4–30
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Review
Neuropharmacologic modulation of the melatonergic system
Utku Aykan ... Canan Uluoglu
Published: December 22, 2023 Explor Neurosci. 2023;2:287–306
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Review
Negative environmental influences on the developing brain mediated by epigenetic modifications
Maya Komar-Fletcher ... Joanna Michalina Jurek
Published: September 28, 2023 Explor Neurosci. 2023;2:193–211
Open Access
Review
An intricate relationship between circadian rhythm dysfunction and psychiatric diseases
Saptadip Samanta, Debasis Bagchi
Published: August 23, 2024 Explor Neurosci. 2024;3:321–351
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Mini Review
Nutritional treatment with the ketogenic diet in children with refractory epilepsy: a narrative review
Srilaxmi Vityala ... Swathi Nenavath
Published: October 30, 2023 Explor Neurosci. 2023;2:245–250
This article belongs to the special issue Epilepsy

Open Access
Review
Current therapeutics for Alzheimer’s disease and clinical trials
Danqing Xiao, Chen Zhang
Published: June 27, 2024 Explor Neurosci. 2024;3:255–271
This article belongs to the special issue Alzheimer’s Disease

Open Access
Review
Stigma and psychosocial problems in patients with epilepsy
Kubra Yeni
Published: December 06, 2023 Explor Neurosci. 2023;2:251–263
This article belongs to the special issue Epilepsy
Open Access
Review
Impact of circadian clock dysfunction on human health
Saptadip Samanta, Sk Asif Ali
Published: September 29, 2022 Explor Neurosci. 2022;1:4–30
This article belongs to the special issue Circadian Rhythm and Melatonin

Open Access
Review
Negative environmental influences on the developing brain mediated by epigenetic modifications
Maya Komar-Fletcher ... Joanna Michalina Jurek
Published: September 28, 2023 Explor Neurosci. 2023;2:193–211

Open Access
Mini Review
Neuroprotective compounds from three common medicinal plants of West Bengal, India: a mini review
Suvendu Ghosh ... Debosree Ghosh
Published: December 26, 2023 Explor Neurosci. 2023;2:307–317
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection

Open Access
Review
Stigma and psychosocial problems in patients with epilepsy
Kubra Yeni
Published: December 06, 2023 Explor Neurosci. 2023;2:251–263
This article belongs to the special issue Epilepsy

Open Access
Review
Current therapeutics for Alzheimer’s disease and clinical trials
Danqing Xiao, Chen Zhang
Published: June 27, 2024 Explor Neurosci. 2024;3:255–271
This article belongs to the special issue Alzheimer’s Disease

Open Access
Review
Impact of circadian clock dysfunction on human health
Saptadip Samanta, Sk Asif Ali
Published: September 29, 2022 Explor Neurosci. 2022;1:4–30
This article belongs to the special issue Circadian Rhythm and Melatonin

Open Access
Review
Connecting the ends: signaling via receptor tyrosine kinases and cytoskeletal degradation in neurodegeneration
Priyanka Sengupta ... Debashis Mukhopadhyay
Published: February 20, 2024 Explor Neurosci. 2024;3:1–26
This article belongs to the special issue Alzheimer’s Disease

Open Access
Review
Neuropharmacologic modulation of the melatonergic system
Utku Aykan ... Canan Uluoglu
Published: December 22, 2023 Explor Neurosci. 2023;2:287–306
This article belongs to the special issue Circadian Rhythm and Melatonin

Open Access
Review
Neuroprotective prospectives of triterpenoids
Apoorva A. Bankar ... Nazma N. Inamdar
Published: June 26, 2024 Explor Neurosci. 2024;3:231–254
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection
Special Issues
Ongoing Special lssues
Completed Special lssues
Medicinal Plants and Bioactive Phytochemicals in Neuroprotection (Vol II)
Guest Editor: Prof. Marcello Iriti
Submission Deadline: July 31, 2026
Published Articles: 2

Depression: From Pathophysiology to Treatment Innovation
Guest Editors: Prof. Dirk M. Hermann; Dr. Ayan Mohamud Yusuf
Submission Deadline: July 31, 2026
Published Articles: 1

The Science of Ischemic Stroke
Guest Editor: Sheng-Tao Hou
Submission Deadline: July 31, 2026
Published Articles: 1
Advances in Parkinson's Disease Research: From Underlying Mechanisms to Novel Therapeutic Targets and Biodiagnostics
Guest Editor: Prof. Ludmilla A. Morozova-Roche
Submission Deadline: July 31, 2026
Published Articles: 0

Progress in Alzheimer's disease research: etiology, molecular mechanisms involved in disease progression, and advances in therapies aimed at slowing or reversing neurodegeneration
Guest Editor: Prof. Ryszard Pluta
Submission Deadline: December 31, 2026
Published Articles: 3

Advances in Epilepsy Research
Guest Editor: Prof. Jinwei Zhang
Submission Deadline: October 31, 2026
Published Articles: 7

Journal Information
Journal Indexing
Journal Metrics





















 Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys
Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys


