Editor's Picks
Open Access
Review
Exploring s-triazine derivatives as anticancer agents
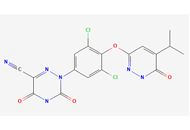
The s-triazine scaffold has emerged as a privileged heterocyclic nucleus/moiety in pharmaceutical discovery and development, owing to its presence in several natural products and clinically relevant therapeutic agents, including enasidenib, gedatolisib, bimiralisib, atrazine, indaziflam, and triaziflam. s-Triazine derivatives are not only economically accessible and synthetically versatile, but they also exhibit a broad spectrum of noteworthy biological activities, encompassing anticancer, anti-inflammatory, antiviral, antidiabetic, anticonvulsant, antitubercular, and antimicrobial properties. Their widespread utility is further supported by the ease of synthesis from inexpensive precursors such as amidines or the readily available 2,4,6-trichloro-1,3,5-triazine (cyanuric chloride), which enables sequential functionalization and the rapid generation of diverse analogues. The heightened reactivity and modularity of the s-triazine core have facilitated the development of structurally rich heterocyclic hybrids with enhanced potency and improved pharmacological profiles. These multitarget-directed systems offer exciting opportunities for addressing various forms of cancer. Considering the increasing pace of innovation in this field, a comprehensive overview of recent advancements in s-triazine-based hybrid molecules is both timely and necessary. This review highlights current progress, key design strategies, and emerging perspectives to inspire continued efforts toward the identification of promising s-triazine-based lead candidates for future drug development as anticancer agents.

Open Access
Review
A review of the effects of pharmaceutical pollutants on humans and aquatic ecosystem
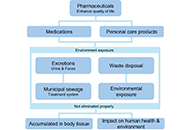
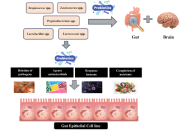
The presence of high-quality water is essential not only for human survival but also for the well-being of plants and animals. This research aimed to examine studies investigating the occurrence of antibiotics, endocrine disruptors, and other pharmaceutical products in water, sediments, and organisms within aquatic ecosystems. These substances have been linked to numerous adverse health effects on both humans and aquatic life, including reproductive issues and neurotoxic effects. The pervasive utilization of antibiotics in medical and agricultural domains has precipitated their ascension as formidable environmental contaminants. Effluents discharged from pharmaceutical industries constitute significant contributors to aquatic ecosystems’ contamination with antibiotics. These pharmacological agents permeate diverse environmental niches, spanning groundwater, surface water, soils, and wastewater treatment facilities, exhibiting concentrations ranging from nanograms to grams per liter. Concurrently, the indiscriminate and excessive application of antibiotics worldwide has engendered escalating apprehensions pertaining to antimicrobial resistance—a formidable global health exigency. This review also delves into the impact of pharmaceutical pollutants on aquatic environments, particularly as endocrine-disrupting compounds. Analysis of surface water in River Taff and River Ely reveals a consistent discharge of approximately 6 kilograms of pharmaceuticals per day. The study examines particular pharmaceuticals, such as diethylstilbestrol (DES), chlorotriazines, chloroquine, and antineoplastic drugs, elucidating their varied effects on reproductive cycles. Pharmaceutical pollutants in aquatic ecosystems, originating from sources like wastewater, agriculture, and improper disposal, persist and adversely affect organisms through bioaccumulation and biomagnification. These contaminants pose significant ecological and health risks, necessitating effective mitigation strategies.
Open Access
Review
Seaweed: a sustainable solution for greening drug manufacturing in the pursuit of sustainable healthcare
The environmental impact of drug manufacturing raises concerns about sustainability in healthcare. To address this, exploring alternative approaches to drug production is crucial. This review focuses on seaweed as a sustainable resource for greening drug manufacturing processes. Seaweed offers advantages such as renewability, abundance, and a positive environmental footprint. The review begins by providing an overview of sustainable drug manufacturing practices and the challenges faced in achieving sustainability. It then discusses seaweed as a sustainable resource, including cultivation techniques and environmental benefits. Seaweed has various applications in drug manufacturing, including extracting and purifying bioactive compounds with potential therapeutic properties. Seaweed’s role in developing green technologies, such as seaweed-based excipients, biodegradable packaging materials, and as a source of sustainable energy for drug manufacturing processes, is highlighted. The environmental and economic implications of incorporating seaweed-based solutions are discussed, emphasizing reduced carbon footprint and cost-effectiveness. Regulatory and industrial perspectives are addressed, examining challenges, and opportunities for implementing seaweed-based drug manufacturing. Collaboration between academia, industry, and regulatory bodies is crucial for successful integration. The review presents future directions and opportunities, including emerging trends and innovations in seaweed-based drug manufacturing, areas for further research, policy development, and industry engagement recommendations. Incorporating seaweed into drug production facilitates a reduction in environmental impact, promotes resource efficiency, and contributes to sustainable healthcare. This review showcases seaweed-based solutions as a means to foster a greener future for drug manufacturing, addressing environmental concerns, and promoting sustainability.

Articles
Latest
Most Viewed
Most Downloaded
Most Cited
Open Access
Review
Advanced Therapy Medicinal Products (ATMPs): fundamental concepts, classification, and translational challenges
Alfi Sophian ... Andhika Harumanto
Published: June 03, 2026 Explor Drug Sci. 2026;4:1008162

Open Access
Review
AI-driven discovery of antimicrobial peptides and derivatives: database and tools
Yunuo Zhou ... Heng Zheng
Published: June 01, 2026 Explor Drug Sci. 2026;4:1008161
This article belongs to the special issue Discovery and development of new antibacterial compounds
Open Access
Original Article
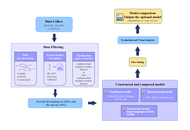
Prediction of absolute bioavailability of medicines in adults and children from oral clearance
Iftekhar Mahmood
Published: May 27, 2026 Explor Drug Sci. 2026;4:1008160

Open Access
Review
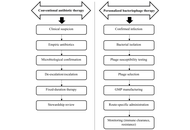
Bacteriophage therapy in the antibiotic resistance era: mechanistic promise, clinical evidence, and system-level barriers
Rana Hussain Abdul Rehman ... Mafaza Ahmar Zia
Published: May 18, 2026 Explor Drug Sci. 2026;4:1008159
Open Access
Original Article
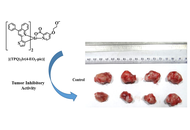
Luminescent iridium(III)-pyridyl based complexes: tumor inhibitory studies in 4T1 mammary carcinoma mice model
Natesan Sundarmurthy Karthikeyan ... Vasanthakumaran Sudarsan
Published: April 28, 2026 Explor Drug Sci. 2026;4:1008158
Open Access
Original Article
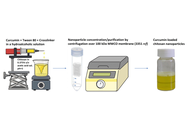
Chitosan-based nanoparticles fabricated by ionotropic gelation using two types of polyphosphates: characterization and in-vitro release modeling of curcumin
Ilya Eydelman ... Amnon C. Sintov
Published: April 15, 2026 Explor Drug Sci. 2026;4:1008157
Open Access
Short Communication
FDA’s approval of resmetirom (Rezdiffra): a breakthrough in MASH management
Muhammad Mazhar Azam ... Bibek Giri
Published: December 03, 2024 Explor Drug Sci. 2024;2:867–874
Open Access
Review
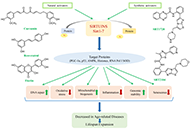
Sirtuin activators as an anti-aging intervention for longevity
Puja Sah ... Donkupar Syiem
Published: January 12, 2025 Explor Drug Sci. 2025;3:100881
Open Access
Review
A review of the effects of pharmaceutical pollutants on humans and aquatic ecosystem
Jaya Vinny Eapen ... Jayesh Antony
Published: August 28, 2024 Explor Drug Sci. 2024;2:484–507

Open Access
Perspective
Clinical studies with drugs and biologics aimed at slowing or reversing normal aging processes—emerging results and future perspectives
Ricardo P. Garay
Published: April 10, 2024 Explor Drug Sci. 2024;2:144–153
Open Access
Review
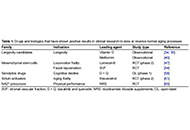
Approved antibacterial drugs in the last 10 years: from the bench to the clinic
Miguel García-Castro ... Juan Manuel López-Romero
Published: June 30, 2023 Explor Drug Sci. 2023;1:180–209
This article belongs to the special issue Exploring Potential Drugs from Natural Products
Open Access
Review
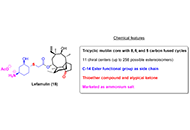
Nature-inspired and medicinally relevant short peptides
Maria G. Ciulla ... Kamal Kumar
Published: June 27, 2023 Explor Drug Sci. 2023;1:140–171
This article belongs to the special issue Exploring Potential Drugs from Natural Products
Open Access
Review
A review of the effects of pharmaceutical pollutants on humans and aquatic ecosystem
Jaya Vinny Eapen ... Jayesh Antony
Published: August 28, 2024 Explor Drug Sci. 2024;2:484–507

Open Access
Review
Approved antibacterial drugs in the last 10 years: from the bench to the clinic
Miguel García-Castro ... Juan Manuel López-Romero
Published: June 30, 2023 Explor Drug Sci. 2023;1:180–209
This article belongs to the special issue Exploring Potential Drugs from Natural Products

Open Access
Review
Sirtuin activators as an anti-aging intervention for longevity
Puja Sah ... Donkupar Syiem
Published: January 12, 2025 Explor Drug Sci. 2025;3:100881

Open Access
Review
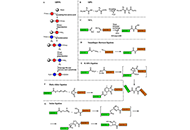
Mechanisms of action and health benefits of probiotics: a comprehensive review
Seemi Tasnim Alam ... Md. Aftab Uddin
Published: September 17, 2025 Explor Drug Sci. 2025;3:1008129
Open Access
Review
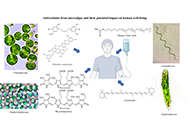
Antioxidants from microalgae and their potential impact on human well-being
Leonel Pereira ... Ana Valado
Published: May 31, 2024 Explor Drug Sci. 2024;2:292–321
Open Access
Review
Nature-inspired and medicinally relevant short peptides
Maria G. Ciulla ... Kamal Kumar
Published: June 27, 2023 Explor Drug Sci. 2023;1:140–171
This article belongs to the special issue Exploring Potential Drugs from Natural Products

Open Access
Review
A review of the effects of pharmaceutical pollutants on humans and aquatic ecosystem
Jaya Vinny Eapen ... Jayesh Antony
Published: August 28, 2024 Explor Drug Sci. 2024;2:484–507

Open Access
Review
Antioxidants from microalgae and their potential impact on human well-being
Leonel Pereira ... Ana Valado
Published: May 31, 2024 Explor Drug Sci. 2024;2:292–321

Open Access
Review
Seaweed: a sustainable solution for greening drug manufacturing in the pursuit of sustainable healthcare
Leonel Pereira, João Cotas
Published: February 27, 2024 Explor Drug Sci. 2024;2:50–84

Open Access
Editorial
Machine learning for drug science
Walter F. de Azevedo Jr.
Published: April 16, 2023 Explor Drug Sci. 2023;1:77–80
This article belongs to the special issue Machine Learning for Drug Science
Open Access
Review
Nature-inspired and medicinally relevant short peptides
Maria G. Ciulla ... Kamal Kumar
Published: June 27, 2023 Explor Drug Sci. 2023;1:140–171
This article belongs to the special issue Exploring Potential Drugs from Natural Products

Open Access
Review
Approved antibacterial drugs in the last 10 years: from the bench to the clinic
Miguel García-Castro ... Juan Manuel López-Romero
Published: June 30, 2023 Explor Drug Sci. 2023;1:180–209
This article belongs to the special issue Exploring Potential Drugs from Natural Products

Special Issues
Ongoing Special lssues
Completed Special lssues
Reactive Future: Molecular Architectures and Nanoformulations Targeting Oxidative Stress
Guest Editor: Barbara De Filippis
Submission Deadline: September 30, 2026
Published Articles: 0

Nanoformulations for Non-Intravenous Drug Delivery
Guest Editor: Prof. Zhengwei Huang
Submission Deadline: October 31, 2026
Published Articles: 1

Precision Dosing: From Therapeutic Drug Monitoring to AI-Driven Personalization
Guest Editor: Jinwei Zhang
Submission Deadline: August 31, 2026
Published Articles: 0

Target-Driven Anticancer Drug Discovery for the 21st Century
Guest Editor: Prof. Michio Kurosu
Submission Deadline: October 31, 2026
Published Articles: 0

Peptide Science Without Borders: Novel Insights for Drug Discovery
Guest Editors: Prof. Fernando Albericio; Prof. Fanny Guzmán Quimbayo
Submission Deadline: October 31, 2026
Published Articles: 1

Innovations in Hepatology: Bridging Drug Development and Biomarker Discovery
Guest Editor: Prof. Ralf Weiskirchen
Submission Deadline: October 31, 2026
Published Articles: 0

Investigating the Therapeutic Potential of the Chromone Scaffold in the Treatment of Respiratory Infections
Guest Editor: Prof. Diana Pinto
Submission Deadline: June 30, 2026
Published Articles: 0
The Role of Triazine Scaffolds in Modern Drug Development
Guest Editors: Prof. Fernando Albericio; Prof. Ayman El-Faham
Submission Deadline: October 31, 2026
Published Articles: 1

Advances and Innovations in Gene-Based Medicine
Guest Editor: Prof. Juergen Reichardt
Submission Deadline: October 31, 2026
Published Articles: 1

Drug Repurposing: Accelerating Cancer Therapeutic Discoveries
Guest Editors: Prof. Wei-Lin Jin; Dr. Jin-Min Ma
Submission Deadline: June 30, 2026
Published Articles: 0

The Rise of Targeted Covalent Inhibitors in Drug Discovery
Guest Editors: Prof. F. Javier Luque; Prof. Jerônimo Lameira
Submission Deadline: October 31, 2026
Published Articles: 1
Discovery and development of new antibacterial compounds
Guest Editor: Kamal Kumar
Submission Deadline: December 31, 2025
Published Articles: 7

Journal Information
Journal Indexing
Journal Metrics




















 Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys
Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys


