Affiliation:
Department of Biotechnology, National Institute of Pharmaceutical Education and Research (NIPER), Mohali 160062, Punjab, India
†These authors contributed equally to this work.
Email: apande@niper.ac.in; abbupande@yahoo.co.in
ORCID: https://orcid.org/0000-0002-4145-539X
Affiliation:
Department of Biotechnology, National Institute of Pharmaceutical Education and Research (NIPER), Mohali 160062, Punjab, India
†These authors contributed equally to this work.
ORCID: https://orcid.org/0000-0002-8950-1957
Affiliation:
Department of Biotechnology, National Institute of Pharmaceutical Education and Research (NIPER), Mohali 160062, Punjab, India
ORCID: https://orcid.org/0000-0002-5029-8808
Explor Drug Sci. 2026;4:1008155 DOI: https://doi.org/10.37349/eds.2026.1008155
Received: December 08, 2025 Accepted: February 01, 2026 Published: March 19, 2026
Academic Editor: Fernando Albericio, University of KwaZulu-Natal, South Africa, Universidad de Barcelona, Spain
Antibodies currently represent a leading segment of the biopharmaceutical market and are expected to maintain a significant presence in the therapeutic landscape. Development of therapeutic antibodies represents one of the most transformative advances in the modern medicine field, with hundreds of successful products on the market that have changed the lives of millions and the history of mankind by revolutionizing medical treatment. In its broadest context, antibody-based products consist of full-length antibodies, antibody fragments, polyvalent and polyspecific antibodies, and their conjugates (for targeted delivery of other therapeutic drugs/agents). High target specificity, tailored mechanisms of action, and broad applicability have made antibodies indispensable in a variety of diseases. With advancements in protein engineering as well as growing integration of computational biology, the field of antibody development continues to push boundaries and is expected to drive the next era of breakthroughs. This article charts the journey of therapeutic antibodies from their conceptual roots to their central role in current medicine, and offers a forward-looking perspective on what lies ahead in this dynamic field.
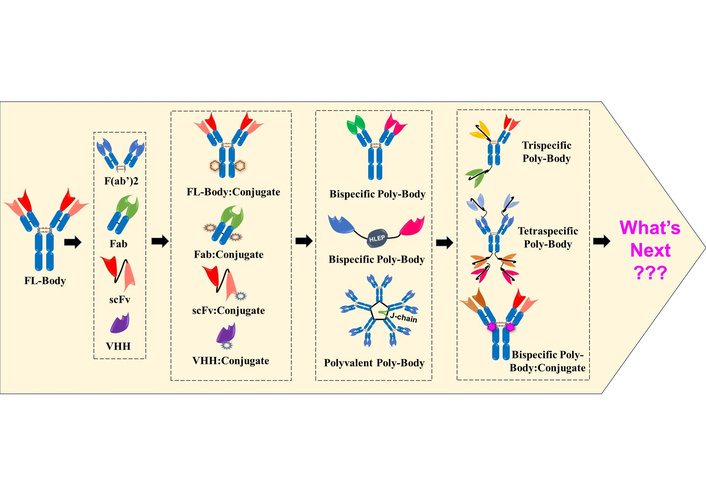
Biopharmaceuticals, the largest segment of the healthcare market, are medicinal products derived from biological sources and/or designed to mimic biological molecules. Based on their biological characteristics, the biopharmaceutical segment can be categorized into seven verticals [1], and antibody-based products constitute the largest vertical, dominating the biopharmaceutical segment [2, 3]. Although antibodies emerged later in the therapeutic landscape, their unparalleled success has allowed them to surpass all other biologic categories in terms of both clinical utility and commercial impact. Since the approval of the first antibody (muromonab in 1986) [4], the field has experienced remarkable growth, underscoring the pivotal role antibodies now play in modern therapeutics. Their high safety, bioavailability, and precise and high-affinity binding to disease-specific targets have made antibody-based products essential in modern therapies, leading to significant improvements in quality of life and life expectancy [5]. By selectively neutralizing or modulating the biological function of their targets, antibodies have become powerful therapeutic agents in the development of innovative treatments across a broad spectrum of diseases [3]. To date, over 200 antibodies and antibody-conjugated products have been approved and are currently in use for the treatment of a wide range of disease conditions, spanning from infectious diseases to cancers [6, 7]. Beyond their therapeutic applications, antibodies and their conjugates are also widely employed in diagnostics, owing to their exceptional target specificity [8–11]. In this article, we explore the evolution of therapeutic antibodies, examining their past and present advancements, while also anticipating future developments in this rapidly advancing field.
The history of antibodies began with the discovery of anti-sera in the 1890s by Emil von Behring and Kitasato [12], which were used as a defensive therapy for invading pathogens and therapeutics for several infections such as diphtheria, influenza, polio, scarlet fever, measles, tetanus, etc. These natural antibodies were polyclonal in nature, i.e., a mixture of antibodies produced from multiple B-cell clones, and were capable of binding to specific and non-specific targets (antigens), resulting in generating undesirable effects. Despite their limitations, these early developments laid the conceptual groundwork for what would become one of the most transformative fields in modern medicine. Later in 1975, with the invention of hybridoma technology, monoclonal antibodies (MAbs) were developed. These full-length antibodies (FL-Bodies) are Y-shaped immunoglobulins, each having two Fab regions joined to an Fc region via a flexible hinge region. The two Fab regions bind to identical epitopes on a single target antigen, making these conventional antibodies bivalent and monospecific in nature [13]. These conventional antibodies transformed the immunotherapy field and were considered as ‘magic bullets’ due to their precise target binding with high specificity. Subsequently, Köhler and Milstein [14] were awarded the Nobel Prize in 1984 for their invention of hybridoma technology and monoclonal monospecific antibodies [15]. This was not merely a scientific breakthrough, but a turning point in the philosophy of medicine, one that shifted the focus from broadly acting treatments to precision-guided therapies rooted in molecular recognition. MAbs generated with hybridoma technology were of murine origin (generated in a mouse), and thus were immunogenic to use as therapeutics for humans. This limitation was addressed by chimeric antibodies, in which murine variable regions were grafted onto constant domains of human origin antibodies. Infliximab antibody is an example of a chimeric antibody. Chimeric antibodies still have a major portion of mice origin, so this was further reduced by CDRs grafting to the human framework, resulting in humanized antibodies. Later on, complete humanized antibodies originated either by producing from transgenic animals or by producing antibodies by phage-display library, which are 100% human in nature. These fully human FL-Bodies become the gold standard with favourable tolerance, optimal pharmacokinetics in humans [7]. The therapeutic success of FL-Bodies led to more targeted and effective therapeutics and a new era of specialized medicine, where target-specific antibodies are used as a therapeutic for a variety of diseases, offering renewed hope and better patient outcomes. Muromonab (which targets the CD3 receptor on T-lymphocytes and blocks their function) became the first antibody to get approval in 1986, and was used as an immunosuppressive therapy in organ transplants [16]. Muromonab was a murine MAb, and thus it was associated with several side effects, such as cytokine release syndrome (CRS) and neurological issues, and thus it was discontinued in 2010–2011. However, the clinical therapeutic efficacy of muromonab laid the foundation for the era of antibody therapeutics. Later on, the surge in the development of new MAbs and their approval for multiple diseases promoted intense research in the antibody field [17]. This rapid progress underscored antibodies’ unmatched versatility and therapeutic relevance across a wide spectrum of clinical conditions. As of today, more than 130 different FL-Bodies have been approved by regulatory agencies in the world for various diseases ranging from infectious diseases to cancer [6]. Frag-Bodies (antibody fragments) are the truncated versions of FL-Bodies (or heavy chain antibodies which are found in Camelidae and Shark families) that retain the capacity to bind to antigen and include F(abʼ)2 (~110 kDa), Fab (~50 kDa), scFv (~25 kDa), and VHH or Nanobody (~12 kDa) [7]. Frag-Bodies are also being developed for therapeutic use. For example, Licartin, a F(abʼ)2-based Frag-Body is an immunoradioconjugate that targets the hepatocellular cancer-associated antigen (HAb18G/CD147) and was approved in 2006 by the NMPA (China) [18]. Similarly, Certolizumab-pegol, a Fab-based Frag-Body, blocks the binding of TNF-alpha to its receptor and is used as an anti-inflammatory therapy [19]. Brolucizumab, a scFv-based Frag-Body, binds to VEGF-A and inhibits neovascularization in macular degeneration disease [20]. Similarly, Caplacizumab is an anti-vWF nanobody approved for the treatment of acquired thrombotic thrombocytopenic purpura [21]. As of today, more than 40 Frag-Bodies products have been approved for various diseases. Their smaller size, improved tissue penetration, and potential for engineering multifunctional formats make Frag-Bodies a highly promising subclass in the expanding antibody landscape.
Thus, the period from the first antibody approval in 1986 to 2000 represents the ‘past era’ of therapeutic antibodies, characterized by the emergence of both FL-Bodies and Frag-Bodies, which laid the foundation for the present-day antibody-based products (Figure 1; 1986–2000). This era of antibody development also witnessed the initiation of a new field: development of FL-Body:Conjugates, a new class of antibody-based products in which FL-Body is conjugated to non-antibody drugs, viz., small chemical drugs, radioisotopes, bacterial toxins, etc., for their targeted (site-specific) delivery. A notable milestone was the approval of Gemtuzumab ozogamicin in 2000, the first FL-Body:Conjugate where an anti-CD33 IgG4 is conjugated to cytotoxic N-acetyl gamma calicheamicin drug, for the treatment of acute myeloid leukemia [22]. Collectively, this foundational era represents not only technical innovation but also marks a paradigm shift in therapeutic science, establishing the core principles of specificity and modularity that now define the era of precision medicine.
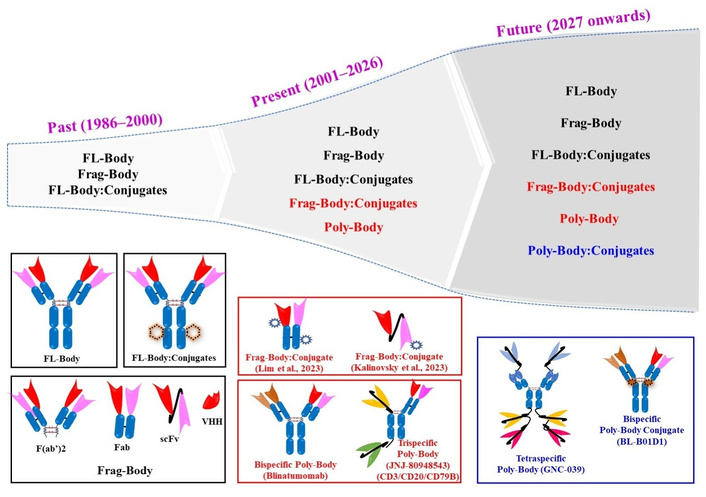
The success of antibodies as therapeutic agents has made them a dominant force in the biopharmaceutical field in the current world. Their market share now represents one of the fastest‑growing segments in pharma, underscoring the economic and clinical indispensability of antibodies [23, 24]. However, treatment regimens using antibodies are challenged by the realization that not only one but many pathways are involved in the pathogenesis of diseases, and there is a need to inhibit/target multiple pathways (antigens) to provide better therapeutic outcomes [25]. This prompted the development of antibodies that can bind to multiple antigens simultaneously [26], i.e., Poly-Bodies, and initiated the present era of therapeutic antibody development. Poly-Bodies are engineered antibodies that can bind to two or more different antigens simultaneously. Thus, Poly-Bodies can be bispecific, trispecific, tetraspecific, and so on. Similarly, Poly-Bodies can be polyvalent, i.e., they can have more than two binding sites for the same epitope (trivalent, tetravalent, and so on). This innovation reflects a deeper shift in therapeutic strategy from one drug-one target to a multifaceted approach that acknowledges the complexity of human diseases. Poly-Bodies have high efficacy due to high affinity and high avidity and are considered superior therapeutic options to conventional antibodies [27]. Thus, Blinatumomab (anti-CD19/CD3) became the first FDA-approved Poly-Body (polyspecific and polyvalent antibody; in 2014) [28]. Similarly, JNJ-80948543 (anti-CD3/CD79b/CD20) is a trispecific Poly-Body developed for the treatment of non-Hodgkin lymphoma patients and is currently in phase I clinical trials (NCT05424822, NCT06139406, NCT06660563) [29]. Since the approval of the first bispecific Poly-Body in 2014, an additional 14 different Poly-Bodies have been granted approval in different countries by March 2025 [7, 27]. In the last couple of years itself (2023–2024), nine different polyspecific Poly-Bodies got approval for the treatment of a variety of diseases and conditions; Glofitamab (anti-CD20/CD3), Epcoritamab (anti-CD20/CD3), Elranatamab (anti-BCMA/CD3), Talquetamab (anti-GPCR5D/CD3), Tarlatamab (anti-DLL3/CD3), Zanidatamab (anti-HER2 epitope 1/HER2 epitope 2), Zenocutuzumab (anti-HER2/HER3), Odronextamab (anti-CD20/CD3), Ivonesclmab (anti-PD-1/VEGF-A) [3, 7, 30]. Thus, the number of Poly-Bodies entering clinical trials and obtaining approval has been increasing at an exponential rate, demonstrating not only scientific advancement but a deepening of antibody utility in treating previously untreatable or refractory diseases.
The present era of antibody development (Figure 1; 2001–2026) has also witnessed the development and approval of numerous other FL-Bodies and Frag-Bodies. The era has also seen a surge in the exploration of new categories of conjugating drugs (nucleic acids, photosensitizers, cytokines, etc.), resulting in the development of newer FL-Body:Conjugates and Frag-Body:Conjugates. For example, in 2020, a photosensitizer-conjugated antibody, Cetuximab Saratolacan (anti-EGFR antibody conjugated to IRDye 700DX), was approved for the treatment of Unresectable Locally Advanced and Recurrent Head & Neck Cancer in Japan [31]. FL-Bodies offer extended half-life and Fc-mediated effector functions but suffer from poor tissue penetration. In contrast, Frag-Bodies provide superior tumor access and rapid clearance, though they lack stability and Fc-mediated immunological responses. Considering the advantages of Frag-Bodies, such as their small size, ease of design and production, and improved tissue penetration, they have become a preferred choice for conjugation. FL-Bodies typically have a half-life of 2–3 weeks, whereas Frag-Bodies, including Fab, scFv, and VHH formats, exhibit half-lives ranging from minutes to hours. This rapid clearance makes Frag-Bodies and their conjugates particularly well-suited for diagnostic applications [32, 33]. Recently, Lim et al. [34] developed a Frag-Body:Conjugate in which an anti-cMET Fab Frag-Body is conjugated to Duo5 cytotoxic drug as a potential treatment for solid tumors. Similarly, Kalinovsky et al. [35] developed anti-GD2 scFv Frag-Body:Conjugate in which anti-GD2 scFv Frag-Body is conjugated to monomethyl auristatin E for the treatment of GD2-positive solid tumors. As of today, more than 15 Frag-Body:Conjugates are in clinical trials, which will be in market in the coming years. This trend highlights not only the therapeutic flexibility of Frag-Bodies but also their increasing value in combination modalities. In contrast to FL-Bodies and Frag-Bodies, Poly-Bodies are multi-targeting, high-avidity, and have superior therapeutic efficacy. However, Poly-Bodies have a challenge of complex manufacturing and potential immunogenicity. Most approved Poly-Bodies are anti-cancer, involving T-cell engager bispecific antibodies, which have a challenge of CRS [36].
The present era of antibodies marks success for conjugated antibodies, especially in diagnostic and therapeutic treatment of cancer and suggests an exciting era of targeted, highly effective, and safer cancer treatment that promises to substitute conventional chemotherapies across various cancers [37]. Building on this progress, advances in computer-based biology, high-throughput screening technologies, and artificial intelligence have greatly accelerated the pace of antibody discovery and development, firmly establishing antibodies as a vital element of precision medicine. It is notable that, despite these remarkable advances, antibody development still faces several critical obstacles that can limit therapeutic potential. Immunogenicity remains a primary concern, as anti-drug antibody (ADA) responses can reduce therapeutic efficacy, alter pharmacokinetic properties, and, in some cases, trigger severe adverse immune reactions. The challenge is compounded by the fact that ADA development varies among patients, influenced not only by individual factors but also by geographical and racial differences. However, the major reason for ADA development is the non-human nature of antibodies for murine, chimeric, and humanized antibodies. This results in poor pharmacokinetics (fast clearance) and immunogenicity, and thus limits clinical efficacy. These interpatient variations highlight a complex obstacle in antibody therapy that still needs to be effectively addressed [38, 39]. Beyond immunogenicity and pharmacokinetic concerns, antibody therapeutics face major challenges in large-scale production, cost management, and supply sustainability. Despite their clinical success, high manufacturing costs continue to restrict accessibility, particularly in low- and middle-income countries. A substantial share of these costs arises from drug substance (DS) manufacturing, including both upstream and downstream processes. Meeting the growing and diversifying demand for antibodies also requires adequate and flexible biomanufacturing capacity. Most MAbs are produced in mammalian systems, mainly Chinese hamster ovary (CHO) cells, using large-scale bioreactors. While global capacity has expanded, imbalances persist, with some firms facing shortages and others excess capacity and financial strain. To address this, the industry increasingly relies on contract manufacturing organizations (CMOs) and single-use bioreactor technologies, which provide flexibility, rapid scalability, and lower capital investment. Moreover, emerging DS manufacturing technologies and formats are being explored to reduce costs, increase supply flexibility, and enhance development efficiency. Incorporating process economic analysis from the preclinical stage through late-stage development is also critical to evaluate feasibility, optimize manufacturing strategies, and select the most suitable production approach for each antibody program [40].
The clinical success of antibodies initiated a competitive environment among pharma companies and research groups. This positive competition is driving innovations in making highly efficacious and precision antibody-based products. The development of Poly-Bodies and conjugated antibodies is an example of such innovations (Figure 1; 2027 onwards). Hundreds of these next-generation products are in advanced stages of clinical trials which are going to be marketed in the coming years [41, 42]. The clinical success of Poly-Bodies and conjugated antibodies is expanding the boundaries of therapy, especially in complex diseases such as cancer and multifaceted inflammatory disorders. As clinical approvals for antibody-based products increase each year, the field is expected to grow further, providing more opportunities for diverse clinical uses.
Specificity is a crucial deciding factor for the therapeutic efficacy in the case of antibodies. The future of antibody-based products looks incredibly promising with the ongoing development of trispecific, tetraspecific, and pentaspecific antibodies. Sonelokimab (anti-interleukin (IL)-17A/IL-17F/albumin) is a trispecific antibody for hidradenitis suppurativa disease that is currently in phase 3 clinical trial (NCT07007637) and is expected to be approved in the near future [43]. Umizortamig (GNC039, anti-CD3/4-1BB/EGFRVIII/PD-L1) is a tetraspecific Poly-Body for glioma and solid tumor and is currently in a phase I clinical study (CN110831973A) [44]. Researchers are also pushing boundaries in engineering multivalent antibodies or Poly-Bodies with 10, 20, or even more binding sites which were once thought impossible. Thanks to accelerating technological advances, these ultra-multivalent antibodies may soon become a reality. The development of Poly-Body:Conjugates is also significantly advancing the antibody-based product pipeline. BL-B01D1 is a first-in-class Poly-Body:Conjugate in which an anti-EGFR/HER3 bispecific Poly-Body is conjugated to topoisomerase I inhibitor (Ed-04) and is currently in phase 3 clinical trial (NCT06118333) for recurrent or metastatic nasopharyngeal carcinoma [45]. Thus, in the future, we will see more Poly-Bodies and their immunoconjugates in the market.
Drug payload in antibody:conjugates is a crucial factor for deciding their efficacy. Rapid progress in conjugation chemistry, therapeutic payload engineering, and nanotechnology is expanding the spectrum of payloads that can be effectively conjugated to antibodies. In the future, there will be a variety of payloads such as carbon-dots [46], nanorobots [47], PROTACs (proteolysis targeting chimeras) [48], etc. This could offer new mechanisms, improved efficacy with a multi-payload strategy that further enhances the versatility and clinical potential of antibody conjugates. Moreover, the future of antibodies is being transformed by advances in genetic and protein engineering technologies [49] that enable the precise design of Poly-Bodies, their optimization with enhanced affinity and specificity, stability, pharmacokinetics, and especially the design of complex Poly-Body formats and their conjugates. With every engineering milestone, the vision of truly personalized treatments is coming closer to reality, inspiring optimism across the scientific community. Along with that, artificial intelligence and machine learning (AI/ML) are also accelerating antibody development, helping in predictive analytics, high-throughput screening of lead candidates, and ultimately shortening the antibody development timeline and enhancing the success rate [50]. AI’s role in the design of de novo antibodies and predicting their molecular behavior will reshape antibody development [51, 52]. This convergence of biology and computation is already producing candidates that would have taken years to generate by traditional means. A notable example is AU-007, a human IgG1 MAb designed using AI to bind IL-2 at its CD25 binding epitope. Developed by Aulos Bioscience, AU-007 has advanced to the Phase 2 portion of a Phase 1/2 clinical trial evaluating its efficacy in treating solid tumors. Interim results presented at the 2024 ASCO Annual Meeting demonstrated AU-007ʼs ability to harness IL-2 to reduce solid tumors, supporting ongoing and planned Phase 2 expansion cohorts in combination with low-dose aldesleukin in melanoma, renal cell carcinoma, and non-small cell lung cancer [53] (Aulos https://aulosbio.com/). While specific wet-lab validation hit rates for AU-007 are not publicly disclosed, the development of AI-designed antibodies like AU-007 exemplifies the potential of AI/ML to enhance the efficiency and success rates of antibody discovery, offering a promising alternative to traditional methods such as hybridoma technology and phage display. While AI/ML approaches have significantly accelerated antibody discovery and optimization, their application still faces important limitations. Current models rely heavily on the availability and quality of training data, which are often biased toward well-studied targets and antibody formats. As a result, predictions may not generalize reliably to novel antigens or complex multispecific architectures [54, 55]. In addition, AI-based designs frequently require extensive experimental validation, as in silico predictions do not fully capture protein folding, developability issues, immunogenicity, or in vivo behavior. Limited interpretability of some models and integration with downstream manufacturing and regulatory requirements further constrain widespread adoption [56]. Addressing these challenges will be essential for realizing the full potential of AI/ML in antibody design. The approved therapeutic antibodies, along with their format, target, clinical indication, and approval status, are summarized in Table 1.
List of approved therapeutic antibodies.
| Name | Format | Target | Indication | Approval |
|---|---|---|---|---|
| Muromonab* | FL-Body | CD3 | Organ transplant rejection (heart, liver, kidney) | FDA (1986);EMA (1986);CECMED (1989);NMPA (1999) |
| IOR-T3 | FL-Body | CD3 | Organ transplant rejection | CECMED (1989);NMPA (1999) |
| Nebacumab* | Poly-Body | CD3 | Gram-negative sepsis | EMA (1991) |
| (99m Tc) Fanolesomab | Poly-Body:Conjugate | CD15 | Scintigraphic imaging of patients with equivocal signs and symptoms of appendicitis | FDA (2004) |
| Edrecolomab* | FL-Body | EpCAM | Colorectal cancer | EMA (1995) |
| Abciximab | Frag-Body | GPIIb/IIIa | Blood clotting | FDA (1994);EMA (1995) |
| Daclizumab* | FL-Body | IL-2R (CD25) | Organ transplant rejection, multiple sclerosis | FDA (1997);EMA (1999) |
| Rituximab | FL-Body | CD20 | Rheumatoid arthritis, lymphocytic leukemia, follicular lymphoma, non-Hodgkin’s lymphoma, systemic scleroderma | FDA (1997);EMA (1998);PMDA (2001);NMPA (2000);CDSCO (2002) |
| Infliximab | FL-Body | TNF-alpha | Crohnʼs disease, psoriatic arthritis, ankylosing spondylitis, rheumatoid arthritis, Behcet’s disease, ulcerative colitis (UC), plaque psoriasis, Kawasaki disease | FDA (1998);EMA (1998);NMPA(2006);PMDA (2002);CDSCO (2013) |
| Trastuzumab | FL-Body | HER2 | Breast and gastric cancer | FDA (1998);EMA (1998);CDSCO (2012) |
| Palivizumab | FL-Body | RSV-F | Prevention of respiratory syncytial viral infection | FDA (1998);EMA (1999) |
| Basiliximab | FL-Body | IL-2R (CD25) | Organ (kidney, liver) transplant rejection | FDA (1998);EMA (1998);PMDA (2002);NMPA (2004);CDSCO (1999) |
| Etanercept | Frag-Body | TNF-alpha | Psoriasis, psoriatic arthritis, ankylosing spondylitis, rheumatoid arthritis, plaque psoriasis | FDA (1998);EMA (2000);PMDA (2005);NMPA (2010);CDSCO (2012) |
| (99m Tc) Arcitumomab | Frag-Body:Conjugate | Carcinoembryonic antigen (CEA) | Diagnosis of CEA expression in cancer | FDA (1999) |
| Gemtuzumab ozogamicin | FL-Body:Conjugate | CD33 | Acute myelogenous leukemia | FDA (2017);PMDA (2005);EMA (2018); |
| Wut3 | FL-Body | CD3 | Organ transplant rejection | NMPA (2010) |
| Alemtuzumab | FL-Body | CD52 | Lymphocytic leukemia, multiple sclerosis | FDA (2001);EMA (2001);PMDA (2014) |
| Adalimumab | FL-Body | TNF-alpha | Ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis, IBD | FDA (2002);EMA (2003);PMDA (2008);NMPA (2010);CDSCO (2014) |
| Ibritumomab tiuxetan | FL-Body:Conjugate | CD20 | Non-Hodgkin’s lymphoma, relapsed or refractory, low-grade or follicular NHL | FDA (2002);EMA (2004);CDSCO (2017) |
| Tositumomab-I131* | FL-Body:Conjugate | CD20 | Non-Hodgkin lymphoma | FDA (2003) |
| Omalizumab | FL-Body | IgE | Allergic asthma, urticaria | FDA (2003);EMA (2005);PMDA (2006);CDSCO (2015);NMPA (2017) |
| Efalizumab* | FL-Body | Integrin subunit alpha L (ITGAL) or CD11a | Psoriasis | FDA (2003);EMA (2004) |
| Anti-IL-8 monoclonal antibody (ENBOKE) | FL-Body | IL-8 | Atopic dermatitis, pustular psoriasis | NMPA (2003) |
| Alefacept* | Frag-Body | CD2 | Psoriasis | FDA (2003) |
| Cetuximab | FL-Body | EGFR | Colorectal, head and neck cancer | FDA (2004);EMA (2004);PMDA (2008);NMPA (2020);CDSCO (2006) |
| Bevacizumab | FL-Body | VEGF-A | Colorectal, breast, cervical, liver, ovarian, renal cancer | FDA (2004);EMA (2005);CDSCO (2005);PMDA (2007);NMPA (2010) |
| Natalizumab | FL-Body | Integrin subunit alpha 4 (ITGA4) | Multiple sclerosis, Crohn’s disease | FDA (2004);EMA (2006);PMDA (2014);CDSCO (2018) |
| Tocilizumab | FL-Body | IL-6R | Rheumatoid arthritis, Castleman’s disease, vasculitis, drug hypersensitivity, SARS-CoV-2, scleroderma | FDA (2010);EMA (2009);PMDA (2005);CDSCO (2009);NMPA (2013) |
| Abatacept | Frag-Body | CD80, CD86 | Psoriatic arthritis, rheumatoid arthritis, graft versus host disease | FDA (2005);EMA (2007);PMDA (2010);NMPA (2020) |
| Panitumumab | FL-Body | EGFR | Colorectal cancer | FDA (2006);EMA (2007);PMDA (2010);CDSCO (2017) |
| I131 derlotuximab biotin | FL-Body:Conjugate | DNA/Histone H1 | Lung cancer | NMPA (2006) |
| I131 metuximab | Frag-Body:Conjugate | CD147 | Liver cancer | NMPA (2006) |
| Nimotuzumab | FL-Body | EGFR | Head & neck cancer, anaplastic astrocytoma, brain & esophageal cancer, glioma | CECMED (2006);CDSCO (2006);NMPA (2012) |
| Ranibizumab | Frag-Body | VEGF-A | Macular degeneration, choroidal neovascularization, diabetic macular edema, diabetic retinopathy | FDA (2006);EMA (2007);PMDA (2009);NMPA (2010);CDSCO (2007) |
| Eculizumab | FL-Body | C5 | Paroxysmal nocturnal hemoglobinuria, myasthenia gravis, neuromyelitis optica, hemolytic uremic syndrome | FDA (2007);EMA (2007);PMDA (2010);NMPA (2018) |
| Rilonacept | Frag-Body | IL-1 | Cryopyrin-associated periodic syndrome, an inborn genetic disorder, pericarditis | FDA (2008) |
| Certolizumab pegol | Frag-Body:Conjugate | TNF-alpha | Psoriatic arthritis, rheumatoid arthritis, Crohn’s disease, and ankylosing spondylitis | FDA (2008);EMA (2009);PMDA (2012);NMPA (2019) |
| Romiplostim | Frag-Body | Thrombopoietin receptor (TPOR) | Aplastic anemia, acute radiation syndrome, idiopathic thrombocytopenic purpura | FDA (2008);EMA (2009);PMDA (2011);NMPA (2022) |
| Canakinumab | FL-Body | IL-1β | Muckle-wells syndrome, adult-onset still’s disease, gouty arthritis, rheumatoid arthritis | FDA (2009);EMA (2009);PMDA (2011);CDSCO (2011) |
| Catumaxomab* | Poly-Body | CD3/EpCAM | Malignant ascites | EMA (2009) |
| Ustekinumab | FL-Body | IL-12/23 | Psoriatic, IBD | FDA (2009);EMA (2009);PMDA (2011);NMPA (2017) |
| Ofatumumab | FL-Body | CD20 | Chronic lymphocytic leukemia, multiple sclerosis | FDA (2009);EMA (2010);PMDA (2013);NMPA (2021);CDSCO (2016) |
| Golimumab | FL-Body | TNF-alpha | Rheumatoid and psoriatic arthritis, ankylosing spondylitis, UC, axial spondyloarthritis | FDA (2009);EMA (2009);PMDA (2011);CDSCO (2014);NMPA (2017) |
| Denosumab | FL-Body | RANKL | Bone loss, rheumatoid arthritis, bone metastases, bone cancer, malignant hypercalcaemia, osteoporosis | FDA (2010);EMA (2010);PMDA (2012);CDSCO (2017);NMPA (2019) |
| Brentuximab vedotin | FL-Body:Conjugate | CD30 | Hodgkin lymphoma, large T-cell lymphoma, Sezary syndrome, Mycosis fungoides, | FDA (2011);EMA (2012);PMDA (2014);NMPA (2020) |
| Belatacept | Frag-Body | CD80, CD86 | Renal transplant rejection | FDA (2011);EMA (2011);CDSCO (2012) |
| Aflibercept | Frag-Body | VEGF | Colorectal cancer, retinal vein occlusion, diabetic macular edema, diabetic retinopathy, macular edema | FDA (2011);EMA (2012);PMDA (2012);NMPA (2018) |
| Belimumab | FL-Body | BAFF | Systemic lupus erythematosus, lupus nephritis | FDA (2011);EMA (2011);PMDA (2017);NMPA (2022) |
| Ipilimumab | FL-Body | CTLA-4 | Metastatic melanoma, liver cancer, renal cell carcinoma, mesothelioma, non-small cell lung cancer | FDA (2011);EMA (2011);PMDA (2015);NMPA (2021) |
| Pertuzumab | FL-Body | HER2 | Breast cancer | FDA (2012);EMA (2013);PMDA (2013);NMPA (2018) |
| Raxibacumab | FL-Body | Bacillus anthracis protective antigen | Anthrax infection | FDA (2012) |
| Mogamulizumab | FL-Body | CCR4 | Mycosis fungoides, T-cell lymphoma | FDA (2018);PMDA (2012);EMA (2018) |
| Trastuzumab emtansine | FL-Body:Conjugate | HER2 | Breast cancer | FDA (2013);EMA (2013);NMPA (2020);PMDA (2013) |
| Obinutuzumab | FL-Body | CD20 | Chronic lymphocytic leukemia, follicular lymphoma, non-Hodgkin’s lymphoma | FDA (2013);EMA (2014);PMDA (2018);NMPA (2021) |
| Conbercept | Frag-Body | VEGF | Macular degeneration | NMPA (2013) |
| Itolizumab | FL-Body | CD6 | CRS (cytokine release syndrome), plaque psoriasis, SARS-CoV 2 | CDSCO (2013) |
| Blinatumomab | Poly-Body | CD19/CD3 | Acute lymphoblastic leukemia | FDA (2014);EMA (2015);PMDA (2018);NMPA (2020) |
| Pembrolizumab | FL-Body | PD-1 | Breast, cervical, colorectal, esophageal, gastric, liver, head & neck, lung, pancreatic, urogenital, cancer, and B-cell lymphoma, Hodgkin’s lymphoma, malignant lymphoma | FDA (2014);EMA (2015);PMDA (2016);CDSCO (2016);NMPA (2018) |
| Eftrenonacog alfa | Frag-Body | Factor IX | Hemophilia B | FDA (2014);PMDA (2014);EMA (2016);NMPA (2021) |
| Vedolizumab | FL-Body | α4β7 integrin | Inflammatory bowel disease | FDA (2014);EMA (2014);CDSCO (2016);PMDA (2018) |
| Secukinumab | FL-Body | IL-17A | Psoriasis, psoriatic arthritis, ankylosing spondylitis, plaque psoriasis | FDA (2015);EMA (2015);PMDA (2014);CDSCO (2015);NMPA (2019) |
| Ramucirumab | FL-Body | VEGFR-2 | Liver, gastric, colorectal, and non-small cell lung cancer | FDA (2014);EMA (2014);PMDA (2015);CDSCO (2016);NMPA (2022) |
| Efmoroctocog alfa | Frag-Body | Factor VIII (FVIII) | Hemophilia A | FDA (2014);EMA (2015);PMDA (2014) |
| Siltuximab | FL-Body | IL-6 | Multicentric Castleman’s disease | FDA (2014);EMA (2014);NMPA (2021);CDSCO (2016) |
| Nivolumab | FL-Body | PD-1 | Melanoma, non-small cell lung cancer, colorectal cancer, esophageal cancer, gastric cancer, Hodgkin’s lymphoma, renal carcinoma, head and neck cancer, mesothelioma | FDA (2014);EMA (2015);PMDA (2014);CDSCO (2016);NMPA (2019) |
| Elotuzumab | FL-Body | SLAMF7 (SLAM family member 7) | Multiple myeloma | FDA (2015);EMA (2016);PMDA (2016) |
| Alirocumab | FL-Body | PCSK9 (Proprotein convertase subtilisin/kexin type 9) | High cholesterol, hyperlipoproteinemia type IIa | FDA (2015);EMA (2015);PMDA (2016) |
| Axicabtagene ciloleucel | Frag-Body | CD19 | Large B-cell lymphoma (LBCL) | FDA (2022);EMA (2015) |
| Mepolizumab | FL-Body | IL-5 | Asthma | FDA (2015);EMA (2015);PMDA (2016);CDSCO (2018);NMPA (2021) |
| Necitumumab | FL-Body | EGFR | Non-small cell lung cancer | FDA (2015);EMA (2016);PMDA (2019) |
| Idarucizumab | Frag-Body | Dabigatran | Reversal of dabigatran-induced anticoagulation | FDA (2015);EMA (2015);PMDA (2016);NMPA (2018);CDSCO (2017) |
| Dinutuximab | Frag-Body | GD2 | Neuroblastoma | FDA (2015);EMA (2015);PMDA (2021);NMPA (2021) |
| Daratumumab | FL-Body | CD38 | Multiple myeloma | FDA (2015);EMA (2016);PMDA (2017);CDSCO (2017);NMPA (2019) |
| Evolocumab | FL-Body | PCSK9 | Hypercholesterolemia, hyperlipoproteinemia, coronary artery disease, stroke | FDA (2015);EMA (2015);PMDA (2016);CDSCO (2017);NMPA (2018) |
| Atezolizumab | FL-Body | PD-L1 | Bladder, liver, lung, urogenital cancer | FDA (2016);EMA (2017);CDSCO (2017);PMDA (2018);NMPA (2020) |
| Olaratumab* | FL-Body | PDGFRA | Soft tissue sarcoma | FDA (2016);EMA (2016) |
| Bezlotoxumab | FL-Body | Clostridium difficile Toxin B | Prevention of C. difficile infection recurrence | FDA (2016);EMA (2017);PMDA (2017) |
| Ixekizumab | FL-Body | IL-17A | Plaque psoriasis, psoriatic arthritis, pustular psoriasis, non-radiographic axial spondyloarthritis | FDA (2016);EMA (2016);PMDA (2016);NMPA (2019) |
| Brodalumab | FL-Body | IL-17R | Plaque psoriasis, psoriatic arthritis, pustular psoriasis, axial spondylarthritis, erythrodermic psoriasis | FDA (2017);EMA (2017);PMDA (2016);NMPA (2020) |
| SII rmab | FL-Body | Rabies virus GP | Post-exposure prophylaxis of rabies | CDSCO (2016) |
| Reslizumab | FL-Body | IL-5 | Asthma | FDA (2016);EMA (2016) |
| Obiltoxaximab | FL-Body | B. anthracis PA | Anthrax infection | FDA (2016);EMA (2020) |
| Inotuzumab ozogamicin | FL-Body:Conjugate | CD22 | Hematological malignancy | FDA (2017);EMA (2017);PMDA (2018);CDSCO (2018);NMPA (2021) |
| Sarilumab | FL-Body | IL-6R | Rheumatoid arthritis | FDA (2017);EMA (2017);PMDA (2017);HC (2017) |
| Dupilumab | FL-Body | IL-4R alpha | Atopic dermatitis, eosinophilic esophagitis, asthma, nasal polyps | FDA (2017);EMA (2017);PMDA (2018);NMPA (2020) |
| Durvalumab | FL-Body | PD-L1 | Bladder cancer, lung cancer | FDA (2017);EMA (2018);PMDA (2018);NMPA (2019) |
| Idecabtagene vicleucel | Frag-Body | BCMA | Refractory multiple myeloma | FDA (2021) |
| Avelumab | FL-Body | PD-L1 | Merkel cell carcinoma, renal cell carcinoma, urogenital cancer | FDA (2017);EMA (2017);PMDA (2017) |
| Emicizumab | Poly-Body | Factor IXa/X | Hemophilia A | FDA (2017);CDSCO (2018);EMA (2018);PMDA (2018);NMPA (2018) |
| Benralizumab | FL-Body | IL-5R alpha subunit | Asthma | FDA (2017);EMA (2018);PMDA (2018) |
| Ocrelizumab | FL-Body | CD20 | Multiple sclerosis | FDA (2017);EMA (2018) |
| Guselkumab | FL-Body | IL-23p19 subunit | Plaque psoriasis, psoriatic arthritis, palmoplantar pustulosis, erythrodermic psoriasis, generalized pustular psoriasis | FDA (2017);EMA (2017);PMDA (2018);NMPA (2019) |
| Erenumab | FL-Body | CGRP-R (Calcitonin gene-related peptide receptor) | Migraine prevention | FDA (2018);EMA (2018);PMDA (2021) |
| Ravulizumab | FL-Body | C5 | Paroxysmal nocturnal hemoglobinuria, myasthenia gravis, haemolytic | FDA (2018);EMA (2019);PMDA (2019) |
| Moxetumomab pasudotox* | FL-Body:Conjugate | CD22 | Hairy cell leukemia | FDA (2018);EMA (2021) |
| Fremanezumab | FL-Body | CGRP | Migraine prevention | FDA (2018);EMA (2019);PMDA (2021) |
| Sintilimab | FL-Body | PD-1 | Non-small cell lung cancer, liver cancer, gastric cancer, esophageal cancer, Hodgkin’s lymphoma | NMPA (2018) |
| Ibalizumab | FL-Body | CD4 | HIV infection | FDA (2018);EMA (2019) |
| Galcanezumab | FL-Body | CGRP | Migraine prevention, cluster headache | FDA (2018);EMA (2018);PMDA (2021) |
| Tildrakizumab | FL-Body | IL-23p19 | Plaque psoriasis | FDA (2018);EMA (2018);PMDA (2020) |
| Emapalumab | FL-Body | IFN-γ | Primary hemophagocytic lymphohistocytosis | FDA (2018);NMPA (2022) |
| Cemiplimab | FL-Body | PD-1 | Cutaneous squamous cell carcinoma, basal cell carcinoma, and non-small cell lung cancer | FDA (2018);EMA (2019) |
| Toripalimab | FL-Body | PD-1 | Nasopharyngeal carcinoma, esophageal squamous cell carcinoma, urogenital cancer, malignant melanoma | NMPA (2018);FDA (2023);EMA (2024) |
| Caplacizumab | Frag-Body | vWF (von Willebrand Factor) | Thrombotic thrombocytopenic purpura | FDA (2019);EMA (2018) |
| Lanadelumab | FL-Body | Plasma kallikrein | Hereditary angioedema attacks | FDA (2018);EMA (2018);PMDA (2022);NMPA (2020) |
| Burosumab | FL-Body | FGF23 | X-linked hypophosphatemia, osteomalacia | FDA (2018);EMA (2018);PMDA (2019);NMPA (2021) |
| Tislelizumab | FL-Body | PD-1 | Esophageal squamous cell carcinoma, liver cancer, urogenital cancer, non-small cell lung cancer, Hodgkin’s lymphoma | FDA (2024);EMA (2023);NMPA (2019) |
| Risankizumab | FL-Body | IL-23p19 | Plaque psoriasis, psoriatic arthritis, Crohn’s disease | FDA (2019);EMA (2019);PMDA (2019) |
| Trastuzumab deruxtecan | FL-Body:Conjugate | HER2 | Breast and gastric cancer | FDA (2019);EMA (2021);PMDA (2020) |
| Brolucizumab | Frag-Body | VEGF-A | Macular degeneration | FDA (2019);EMA (2020);PMDA (2020) |
| Crizanlizumab | FL-Body | P-selectin | Vaso-occlusive crisis | FDA (2019) |
| Enfortumab vedotin (PADCEV) | FL-Body:Conjugate | Nectin-4 | Urogenital cancer | FDA (2019);EMA (2022);PMDA (2021) |
| Romosozumab | FL-Body | Sclerostin | Osteoporosis in postmenopausal women at risk of fracture, male osteoporosis | FDA (2019);EMA (2019);PMDA (2019) |
| Efgartigimod alfa | Frag-Body | FcRn | Myasthenia gravis | FDA (2021);EMA (2022);PMDA (2022) |
| Camrelizumab | FL-Body | PD-1 | Advanced renal cell carcinoma, esophageal cancer, liver cancer, nasopharyngeal cancer, non-small cell lung cancer | NMPA (2019) |
| Polatuzumab vedotin | FL-Body:Conjugate | CD79b | Diffuse large B-cell lymphoma | FDA (2019);EMA (2020);PMDA (2021) |
| Luspatercept | Frag-Body | TGF-β | Beta-thalassemia, myelodysplastic syndrome | FDA (2019);EMA (2020);NMPA (2022) |
| Rabimabs | Poly-Body | Rabies virus GP (glycoprotein) | Rabies virus infection | CDSCO (2019) |
| Netakimab | FL-Body | IL-17A | Plaque psoriasis, psoriatic arthritis, and ankylosing spondylitis | MoH RF (2019) |
| Racotumomab | FL-Body | NeuGcGM3 | Recurrent or advanced non-small cell lung cancer | CECMED (2013);ANMAT (2013) |
| Pabinafusp alfa | FL-Body:Conjugate | Transferrin receptor | Mucopolysaccharidosis II | PMDA (2021) |
| Inebilizumab | FL-Body | CD19 | Neuromyelitis optica and neuromyelitis optica spectrum disorders | FDA (2020);NMPA (2022);PMDA (2021) |
| Teprotumumab | FL-Body | IGF-1R (insulin-like growth factor-1 receptor) | Thyroid eye disease (Graves’ orbitopathy) | FDA (2020) |
| Sacituzumab govitecan | FL-Body:Conjugate | TROP-2 | Urogenital and breast cancer | FDA (2020);EMA (2021);NMPA (2022) |
| Atoltivimab + Odesivimab + Maftivimab (INMAZEB) | Poly-Body | Ebola virus glycoprotein | Ebola virus infection | FDA (2020) |
| Cetuximab sarotalocan | FL-Body:Conjugate | EGFR | Head and neck cancer | PMDA (2020) |
| Isatuximab | FL-Body | CD38 | Multiple myeloma | FDA (2020);EMA (2020);PMDA (2020) |
| Belantamab mafodotin | FL-Body:Conjugate | BCMA | Multiple myeloma | FDA (2025);EMA (2025) |
| Levilimab | FL-Body | IL-6R | COVID-19, rheumatoid arthritis | Russia (2020) |
| Margetuximab | FL-Body | HER2 | Breast cancer | FDA (2020) |
| Satralizumab | FL-Body | IL-6R | Neuromyelitis optica and neuromyelitis optica spectrum disorders | FDA (2020);EMA (2021);PMDA (2020);HC (2020);NMPA (2021) |
| Tafasitamab | FL-Body | CD19 | Diffuse large B-cell lymphoma | FDA (2020);EMA (2021) |
| Naxitamab | FL-Body | GD2 | High-risk neuroblastoma and refractory osteomedullary disease | FDA (2020) |
| Ansuvimab | FL-Body | Ebola virus glycoprotein | Ebola infection | FDA (2020) |
| Eptinezumab | FL-Body | CGRP | Migraine prevention | FDA (2020);EMA (2022) |
| Prolgolimab | FL-Body | PD-1 | Melanoma | Russia (2020) |
| Olokizumab | FL-Body | IL-6 | Rheumatoid arthritis, COVID-19 | Russia (2020) |
| Penpulimab | FL-Body | PD-1 | Metastatic nasopharyngeal carcinoma, non-small cell lung cancer, Hodgkin’s lymphoma | FDA (2025);NMPA (2021) |
| Dostarlimab | FL-Body | PD-1 | Endometrial cancer is a solid cancer | FDA (2021);EMA (2021) |
| Evinacumab | FL-Body | ANGPTL3 | Homozygous familial hypercholesterolemia | FDA (2021);EMA (2021) |
| Sugemalimab | FL-Body | PD-L1 | Non-small cell lung cancer | NMPA (2021) |
| Envafolimab | Frag-Body | PD-L1 | Solid tumors | NMPA (2021) |
| Telitacicept | Frag-Body | BAFF | Systemic lupus erythematosus | NMPA (2021) |
| Regdanvimab* | FL-Body | SARS-CoV-2 | COVID-19 infection | EMA (2021);ANVISA (2021) |
| Amubarvimab + Romlusevimab | Poly-Body | SARS-CoV-2 | COVID-19 infection | NMPA (2021) |
| Brexucabtagene autoleucel | Frag-Body | CD19 | Relapsed or refractory mantle cell lymphoma, acute lymphoblastic leukaemia | FDA (2020);EMA (2020) |
| Amivantamab | Poly-Body | EGFR/c-Met | Non-small cell lung cancer | FDA (2021);EMA (2021) |
| Zimberelimab | FL-Body | PD-1 | Hodgkin’s lymphoma | NMPA (2021) |
| Bimekizumab | FL-Body | IL-17A/IL-17F | Plaque psoriasis, pustular psoriasis, erythrodermic psoriasis | FDA (2023);EMA (2021);PMDA (2022) |
| Loncastuximab tesirine | FL-Body:Conjugate | CD19 | Diffuse large B-cell lymphoma | FDA (2021);EMA (2022) |
| Tisotumab vedotin | FL-Body:Conjugate | TF (tissue factor) | Cervical cancer | FDA (2021) |
| Tralokinumab | FL-Body | IL-13 | Atopic dermatitis | FDA (2021);EMA (2021) |
| Tezepelumab | FL-Body | TSLP (thymic stromal lymphopoietin) | Severe asthma | FDA (2021);EMA (2022) |
| Aducanumab | FL-Body | Amyloid beta | Alzheimer’s disease | FDA (2021) |
| Disitamab vedotin | FL-Body:Conjugate | HER2 | Gastric and urothelial cancer | NMPA (2021) |
| Sotrovimab* | FL-Body | SARS-CoV-2 | COVID-19 infection | EMA (2021);PMDA (2021) |
| Casirivimab + Imdevimab* | Poly-Body | SARS-CoV-2 | COVID-19 infection | EMA (2021) |
| Anifrolumab | FL-Body | IFNAR-1(Interferon alpha receptor-1) | Systemic lupus erythematosus | FDA (2021);EMA (2022);PMDA (2021) |
| Faricimab | Poly-Body | VEGF-A/ANG-2 | Diabetic macular edema and wet age-related macular degeneration | FDA (2022);EMA (2022);PMDA (2022) |
| Teplizumab | FL-Body | CD3 | Delayed onset of type 1 diabetes | FDA (2022);EMA (2026) |
| Cadonilimab | Poly-Body | CTLA-4/PD-1 | Cervical cancer | NMPA (2022) |
| Ublituximab | FL-Body | CD20 | Multiple sclerosis | FDA (2022);EMA (in review) |
| Ormutivimab | FL-Body | Rabies virus GP (glycoprotein) | Rabies virus prophylaxis | NMPA (2022) |
| Tisagenlecleucel (KYMRIAH) | Frag-Body | CD19 | Relapsed or refractory follicular lymphoma, ALL, DLBCL, FL | FDA (2022);EMA (2018) |
| Tremelimumab | FL-Body | CTLA-4 | Liver cancer | FDA (2022);EMA (2023);PMDA (2022) |
| Lisocabtagene maraleucel | Frag-Body | CD19 | DLBCL, PMBCL, FL3B | FDA (2021);EMA (2022) |
| Mirvetuximab soravtansine | FL-Body:Conjugate | Folate receptor alpha | Ovarian cancer | FDA (2022) |
| Spesolimab | FL-Body | IL-36R | Generalized pustular psoriasis | FDA (2022);EMA (2022) |
| Sutimlimab | FL-Body | C1s | Cold agglutinin disease and autoimmune hemolytic anemia | FDA (2022);EMA (2022) |
| Mosunetuzumab-axgb | Poly-Body | CD20/CD3 | Follicular lymphoma | FDA (2022);EMA (2022) |
| Nirsevimab | FL-Body | RSV | RSV infection | FDA (2023);EMA (2022) |
| Nemolizumab | FL-Body | IL-31RA | Atopic dermatitis | PMDA (2022);FDA (2024);EMA (2025) |
| Relatlimab + Nivolumab | Poly-Body | LAG-3/PD-1 | Melanoma | FDA (2022);EMA (2022) |
| Tixagevimab + Cilgavimab* | FL-Body | SARS-CoV-2 spike protein | Covid infection | EMA (2022) |
| Serplulimab | FL-Body | PD-1 | Solid cancer, lung cancer | NMPA (2022) |
| Teclistamab | Poly-Body | BCMA/CD3 | Multiple myeloma | FDA (2022);EMA (2022) |
| Ozoralizumab | Poly-Body | TNF-alpha/albumin | Rheumatoid arthritis | FDA (in review);PMDA (2022) |
| Tebentafusp | Poly-Body | CD3 and GP100 | Metastatic uveal melanoma | FDA (2022);EMA (2022) |
| Retifanlimab | FL-Body | PD-1 | Metastatic or recurrent locally advanced Merkel cell carcinoma | FDA (2023) |
| Lecanemab | FL-Body | Amyloid beta protofibrils | Alzheimer’s disease | FDA (2023);EMA (in review) |
| Mirikizumab | FL-Body | IL-23p19 | Inflammatory bowel disease | PMDA (2023) |
| Rozanolixizumab | FL-Body | FcRn | Myasthenia gravis | FDA (2023);EMA (2024);PMDA (2023) |
| Talquetamab | Poly-Body | GPCR- 5D and CD3 | Multiple myeloma | FDA (2023);EMA (2023) |
| Epcoritamab | Poly-Body | CD20 and CD3 | B-cell lymphoma | FDA (2023);EMA (2023) |
| Lebrikizumab | FL-Body | IL-13 | Atopic dermatitis | FDA (2024);EMA (2023) |
| Glofitamab | Poly-Body | CD20 and CD3 | B-cell lymphoma | FDA (2023);EMA (2023) |
| Elranatamab | Poly-Body | BCMA and CD3 | Multiple myeloma | FDA (2023);EMA (2023) |
| Pozelimab | FL-Body | C5 | CD55-deficient protein-losing enteropathy (CHAPLE) disease | FDA (2023) |
| Narsoplimab | FL-Body | MASP-2 | Hematopoietic stem cell transplant-associated thrombotic microangiopathy (TA-TMA) | FDA (2025) |
| Donanemab | FL-Body | Amyloid beta | Alzheimer’s disease | FDA (2024);EMA (2025) |
| Cosibelimab | FL-Body | PD-L1 | Squamous cell carcinoma | FDA (2024) |
| Concizumab | FL-Body | Tissue factor pathway inhibitor (TFPI) | Hemophilia A or B | FDA (2024);EMA (2024);PMDA (2023) |
| Crovalimab | FL-Body | C5 | Paroxysmal nocturnal hemoglobinuria | FDA (2024);EMA (2024) |
| Odronextamab | Poly-Body | CD20, CD3 | Follicular lymphoma | EMA (2024) |
| Zolbetuximab | FL-Body | Claudin 18.2 | Gastroesophageal junction adenocarcinoma | FDA (2024);EMA (2024) |
| Vilobelimab | FL-Body | C5a | SARS-CoV-2-induced septic acute respiratory distress syndrome | FDA (2025) |
| Garadacimab | FL-Body | Factor XIIa | Hereditary angioedema | FDA (2025);EMA (2025) |
| Marstacimab | FL-Body | TFPI | Hemophilia | FDA (2024);EMA (2024) |
| Tarlatamab | Poly-Body | DLL3 and CD3 | Small-cell lung cancer | FDA (2025) |
| Axatilimab | FL-Body | CSF-1R | Graft-versus-host disease | FDA (2024) |
| Linvoseltamab | Poly-Body | BCMA and CD3 | Multiple myeloma | FDA (2025);EMA (2025) |
| Zanidatamab | Poly-Body | HER2 | Biliary tract cancers | FDA (2024);EMA (2025) |
| Zenocutuzumab | Poly-Body | HER2 and HER3 | NRG1+ non-small cell lung and pancreatic cancer | FDA (2024) |
| Datopotamab deruxtecan | FL-Body:Conjugate | TROP-2 | Breast cancer | FDA (2025);EMA (2025) |
| Nipocalimab | FL-Body | FcRn | Generalized myasthenia gravis | FDA (2025);EMA (2025) |
| Clesrovimab | FL-Body | RSV | RSV infection | FDA (2025) |
| Sibeprenlimab | FL-Body | APRIL | Nephropathy proteinuria | FDA (2025) |
| Telisotuzumab vedotin | FL-Body:Conjugate | cMET | Non-small cell lung cancer | FDA (2025) |
| Depemokimab | FL-Body | IL-5 | Asthma with type 2 inflammation and chronic rhinosinusitis with nasal polyps | FDA (2025) |
* Indicates antibodies withdrawn from the market due to one or more reasons such as clinical failure, loss of efficacy, severe side effects, or commercial reasons. FDA: Food and Drug Administration; EMA: European Medicines Agency; PMDA: Pharmaceuticals and Medical Devices Agency; NMPA: National Medical Products Administration; CDSCO: Central Drugs Standard Control Organization; CECMED: Centro para el Control Estatal de Medicamentos, Equipos y Dispositivos Médicos; HC: Health Canada; MoH RF: Ministry of Health of the Russian Federation; ANMAT: National Administration of Drugs, Food and Medical Devices. FL-Bodies: full-length antibodies; Frag-Bodies: antibody fragments; Poly-Body: polyspecific and polyvalent antibody; ALL: acute lymphoblastic leukemia; DLBCL: diffuse large B-cell lymphoma; FL: follicular lymphoma; FL3B: follicular lymphoma grade 3B; NHL: non-Hodgkin lymphoma; PMBCL: primary mediastinal large B-cell lymphoma.
Currently, antibody production primarily relies on mammalian cell culture platforms, particularly CHO cells, which remain the industry standard for achieving proper folding and post-translational modifications [57, 58]. However, considering the rapid progress in biotechnology, materials science, and biofabrication, we hypothesize that bioprinting may, in the distant future, emerge as an alternative approach for antibody production. Although no experimental evidence currently supports this concept, protein-based 3D printing materials have already demonstrated success in creating complex biological structures, providing a conceptual foundation for such a possibility [59]. If such technological convergence were achieved, it might allow decentralized, on-demand antibody manufacturing, where hospitals or clinical facilities could potentially fabricate personalized antibodies at the point of care, thereby reducing logistical and supply-chain challenges. Building on this perspective, we also propose the theoretical concept of ‘reversible antibodies’ or ‘RoboBodies’, a highly speculative idea in which engineered antibodies could transiently bind one target and then sequentially interact with others with precise control. While there is currently no experimental evidence to support this idea, advances in protein engineering, nanotechnology, and synthetic biology may eventually make such innovations scientifically conceivable.
It is important to note that the implementation of such next-generation antibody therapies would face considerable regulatory, ethical, and economic challenges. Regulatory approval processes would need to adapt to account for AI-designed molecules and on-demand bioprinted therapeutics, ensuring safety and efficacy. Ethical considerations, including equitable access and potential misuse, would require careful evaluation. Additionally, the economic feasibility of decentralized antibody production and advanced AI-driven design platforms remains uncertain and would likely influence the pace of adoption. At present, these ideas remain our theoretical hypothesis, proposed to highlight the long-term potential of merging advanced manufacturing technologies with antibody therapeutics. Rigorous experimental exploration and technological validation will be required before this concept can move toward practical application. Rapid technological progress is bringing these innovations closer to practical application, as demonstrated by the successful development of Poly-Bodies, which were previously considered a significant engineering challenge. The future of the field holds substantial promise, and antibodies are expected to remain a central component of therapeutic strategies, with ongoing research focused on improving their efficacy and broadening their clinical applications.
Antibody-based therapeutics have established themselves as an important player in modern medicine, with proven effectiveness in infectious diseases and a rapidly expanding role in oncology, autoimmune, cardiovascular, and neurodegenerative disorders. Their strong clinical performance, combined with sustained commercial success, continues to drive intensive research and development efforts, resulting in the emergence of next-generation antibody formats such as multispecific Poly-Bodies, antibody–drug conjugates, and innovative delivery platforms. Moreover, advances in manufacturing technologies, including optimized mammalian and alternative expression systems, single-use bioprocessing, and emerging production strategies, are expected to improve scalability, flexibility, and cost efficiency. At the same time, progress in genetic and protein engineering, conjugation chemistry, and computational approaches like AI-driven discovery and optimization is accelerating antibody design, enhancing specificity, potency, and multi-pathway targeting capabilities.
Despite these advances, several challenges remain, including immunogenicity, manufacturing complexity, supply sustainability, and global accessibility. Addressing these limitations will require continued innovation across discovery, development, and regulatory frameworks. Thus, antibody therapeutics are expected to remain a central component of future treatment strategies, playing a critical role in advancing precision and personalized medicine and improving patient outcomes across a wide range of diseases.
ADA: anti-drug antibody
AI/ML: artificial intelligence and machine learning
CHO: Chinese hamster ovary
CRS: cytokine release syndrome
DS: drug substance
FL-Bodies: full-length antibodies
Frag-Bodies: antibody fragments
IL: interleukin
MAbs: monoclonal antibodies
Poly-Body: polyspecific and polyvalent antibody
The authors thank the Indian Council of Medical Research (ICMR), India for providing Sandeep with a senior research fellowship (ICMR-SRFship, 45/08/2022-DDI/BMS).
AHP: Writing—original draft, Data curation, Writing—review & editing, Funding acquisition. Sandeep: Writing—original draft, Data curation, Writing—review & editing, Funding acquisition. JA: Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
This study was funded by the Biotechnology Industry Research Assistance Council (BIRAC) under the Department of Biotechnology (DBT), Government of India (grant number—BT/PR/62235/MRNAR/169/15/2025). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 2758
Download: 57
Times Cited: 0
