Exploring Type 2 Diabetes Mellitus
Guest Editors
Dr. Alessandro Mantovani E-Mail
Assistant Professor, Section of Endocrinology and Metabolism, Department of Medicine, University of Verona, Verona, Italy
Research Keywords: Type 2 diabetes and risk of cardiovascular complications, type 2 diabetes and risk of diabetic foot complications
Dr. Giovanni Targher E-Mail
Associate Professor, Section of Endocrinology and Metabolism, Department of Medicine, University of Verona, Verona, Italy
Research Keywords: Type 2 diabetes and risk of chronic vascular complications of diabetes, role of nonalcoholic fatty liver disease in diabetes
About the Special lssue
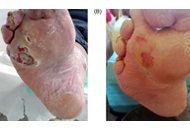
Type 2 diabetes mellitus is a major public health problem that has reached epidemic proportions worldwide. Type 2 diabetes mellitus is also strongly associated with serious chronic complications, including ischemic heart disease, stroke, blindness, kidney disease and lower-extremity amputations leading to disability and early mortality. From 1980 to 2014, the number of people affected by type 2 diabetes mellitus almost quadrupled from 108 million to 422 million worldwide, showing a growth in the global prevalence from 4.7% to 8.5%. This growth trend is not estimated to slow in the near future. Indeed, according to the recent estimates from the International Diabetes Federation, the prevalence of the disease is expected to further increase to 9.9% by the year 2045 with an estimated population of around 640 million people with diabetes worldwide. This undoubtedly represents a potential future healthcare crisis for patients and providers alike. Areas of ongoing research include all aspects of type 2 diabetes mellitus, including epidemiology, physiopathology, risk factors, complications, management and treatment.
Keywords: Type 2 diabetes, chronic complications, epidemiology, management, pharmacologic treatment, risk factors
Published Articles