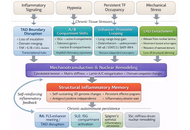

Autoimmune rheumatic diseases arise when the immune system transitions from a flexible, self-regulating network into a metabolically and epigenetically fixed inflammatory attractor state. This review synthesizes emerging evidence that immune tolerance is governed by a coupled epigenetic-metabolic axis integrating mitochondrial fitness, chromatin accessibility, redox balance, and nutrient flux across lymphoid, myeloid, and stromal compartments. We examine how chronic cytokine signaling, hypoxia, and oxidative stress destabilize regulatory programs, imprint glycolytic effector states, and remodel enhancer landscapes, thereby sustaining autoreactive circuits even after inflammatory pathways are pharmacologically suppressed. Multi-omic and spatial analyses reveal that pathogenic chromatin architectures, persistent mitochondrial dysfunction, and intercellular metabolite exchange cooperate to establish self-sustaining inflammatory ecosystems in rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), systemic sclerosis (SSc), and Sjögren’s syndrome. We further highlight therapeutic strategies aimed at tolerance reprogramming, including metabolic correction, chromatin-targeted agents, chimeric antigen receptor regulatory T cells (CAR-Tregs), tolerogenic dendritic cells, and integrative biomarkers that quantify metabolic-epigenetic coherence. By reframing autoimmunity as a disorder of energetic and chromatin desynchronization rather than isolated immune activation, this review outlines a mechanistic path toward durable, drug-free remission through deliberate restoration of the molecular architecture that maintains immune self-recognition.
Autoimmune rheumatic diseases arise when the immune system transitions from a flexible, self-regulating network into a metabolically and epigenetically fixed inflammatory attractor state. This review synthesizes emerging evidence that immune tolerance is governed by a coupled epigenetic-metabolic axis integrating mitochondrial fitness, chromatin accessibility, redox balance, and nutrient flux across lymphoid, myeloid, and stromal compartments. We examine how chronic cytokine signaling, hypoxia, and oxidative stress destabilize regulatory programs, imprint glycolytic effector states, and remodel enhancer landscapes, thereby sustaining autoreactive circuits even after inflammatory pathways are pharmacologically suppressed. Multi-omic and spatial analyses reveal that pathogenic chromatin architectures, persistent mitochondrial dysfunction, and intercellular metabolite exchange cooperate to establish self-sustaining inflammatory ecosystems in rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), systemic sclerosis (SSc), and Sjögren’s syndrome. We further highlight therapeutic strategies aimed at tolerance reprogramming, including metabolic correction, chromatin-targeted agents, chimeric antigen receptor regulatory T cells (CAR-Tregs), tolerogenic dendritic cells, and integrative biomarkers that quantify metabolic-epigenetic coherence. By reframing autoimmunity as a disorder of energetic and chromatin desynchronization rather than isolated immune activation, this review outlines a mechanistic path toward durable, drug-free remission through deliberate restoration of the molecular architecture that maintains immune self-recognition.
DOI: https://doi.org/10.37349/ei.2026.1003255

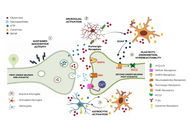
The intricate involvement of glial cells in chronic pain mechanisms represents a paradigm shift in our understanding of pain processing. From microglial-mediated neuroinflammation to astrocytic modulation of synaptic function, glial cells emerge as critical players in the complex neurobiology of chronic pain. As research continues to unravel the multifaceted roles of these cells, novel therapeutic targets and strategies are likely to emerge, potentially revolutionizing the management of chronic pain conditions. Chronic pain is complicated by frequent comorbidities such as fatigue, sleep disturbances, cognitive impairment, and mood disorders. Here, we hypothesize that neuroinflammatory processes are at the root of pain chronification and serve as the common thread linking characteristic hypersensitivity with the cognitive and emotional comorbidities typically associated with chronic pain.
The intricate involvement of glial cells in chronic pain mechanisms represents a paradigm shift in our understanding of pain processing. From microglial-mediated neuroinflammation to astrocytic modulation of synaptic function, glial cells emerge as critical players in the complex neurobiology of chronic pain. As research continues to unravel the multifaceted roles of these cells, novel therapeutic targets and strategies are likely to emerge, potentially revolutionizing the management of chronic pain conditions. Chronic pain is complicated by frequent comorbidities such as fatigue, sleep disturbances, cognitive impairment, and mood disorders. Here, we hypothesize that neuroinflammatory processes are at the root of pain chronification and serve as the common thread linking characteristic hypersensitivity with the cognitive and emotional comorbidities typically associated with chronic pain.
DOI: https://doi.org/10.37349/ei.2026.1003254
This article belongs to the special issue Immunology and Pain

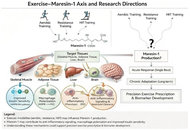
Obesity is a major global health challenge characterized by chronic low-grade inflammation and metabolic dysfunction. Among lipid-derived mediators involved in inflammation resolution, maresin-1 (MaR1)—a specialized pro-resolving mediator derived from docosahexaenoic acid (DHA) and produced mainly by M2 macrophages—has attracted increasing attention due to its potent anti-inflammatory and metabolic regulatory properties. MaR1 promotes the resolution of inflammation by limiting neutrophil infiltration, enhancing macrophage efferocytosis, and shifting cytokine profiles toward an anti-inflammatory phenotype. In addition, it modulates metabolic pathways related to insulin sensitivity and skeletal muscle glucose uptake through signaling mechanisms involving Akt and AMP-activated protein kinase (AMPK). Reduced circulating levels of MaR1 have been consistently associated with metabolic disorders, including obesity, type 2 diabetes, and cardiovascular disease, highlighting its potential as a biomarker of metabolic health. Exercise is a cornerstone non-pharmacological strategy for obesity management and activates molecular pathways—such as AMPK and Sirtuin 1 (SIRT1)—that overlap with those regulated by MaR1. However, human studies examining how different exercise modalities influence MaR1 production remain scarce. This perspective highlights the mechanistic links between exercise and MaR1 biology and proposes a translational research agenda to investigate how aerobic, resistance, and high-intensity interval training modulate MaR1 levels. Understanding this exercise–MaR1 axis may help establish MaR1 as a biomarker of exercise responsiveness and support the development of targeted lifestyle interventions for metabolic disease management.
Obesity is a major global health challenge characterized by chronic low-grade inflammation and metabolic dysfunction. Among lipid-derived mediators involved in inflammation resolution, maresin-1 (MaR1)—a specialized pro-resolving mediator derived from docosahexaenoic acid (DHA) and produced mainly by M2 macrophages—has attracted increasing attention due to its potent anti-inflammatory and metabolic regulatory properties. MaR1 promotes the resolution of inflammation by limiting neutrophil infiltration, enhancing macrophage efferocytosis, and shifting cytokine profiles toward an anti-inflammatory phenotype. In addition, it modulates metabolic pathways related to insulin sensitivity and skeletal muscle glucose uptake through signaling mechanisms involving Akt and AMP-activated protein kinase (AMPK). Reduced circulating levels of MaR1 have been consistently associated with metabolic disorders, including obesity, type 2 diabetes, and cardiovascular disease, highlighting its potential as a biomarker of metabolic health. Exercise is a cornerstone non-pharmacological strategy for obesity management and activates molecular pathways—such as AMPK and Sirtuin 1 (SIRT1)—that overlap with those regulated by MaR1. However, human studies examining how different exercise modalities influence MaR1 production remain scarce. This perspective highlights the mechanistic links between exercise and MaR1 biology and proposes a translational research agenda to investigate how aerobic, resistance, and high-intensity interval training modulate MaR1 levels. Understanding this exercise–MaR1 axis may help establish MaR1 as a biomarker of exercise responsiveness and support the development of targeted lifestyle interventions for metabolic disease management.
DOI: https://doi.org/10.37349/ei.2026.1003253
This article belongs to the special issue Physical Activity and Immune System in Chronic Diseases: Mechanisms and Insights

Aim:
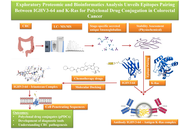
Colorectal cancer (CRC) remains the second leading cause of cancer-related mortality worldwide. While antibody-drug conjugates (ADCs) offer targeted therapeutic options, they are often limited by toxicity, immunogenicity, complex pharmacokinetics, and high production costs. Polyclonal antibodies—capable of recognizing multiple epitopes—present a promising, yet underexplored, alternative for targeted drug delivery. The stage-specific presence of secreted, stable immunoglobulins (IGs) in CRC and their potential utility in drug conjugation strategies remain largely uncharacterized.
Methods:
This study utilized electrospray ionization nano-liquid chromatography tandem mass spectrometry (ESI-nanoLC-MS/MS) proteomic analysis on pre-treatment plasma samples across CRC stages to identify stage-specific IGs. Venn diagram comparisons refined IG candidates, while protein stability was assessed using ProtParam. Molecular docking simulations (via CB-Dock2), epitope mapping (via CABS-dock), and cell-penetrating peptide (CPP) prediction were integrated to explore epitope pairing between IGs and Kirsten rat sarcoma viral oncogene homolog (K-Ras) neoantigen, evaluating their potential for polyclonal drug conjugates (pPDCs).
Results:
A total of 325 secreted IGs were initially identified, with 46 found to be stage-specific. Protein stability analysis shortlisted 5 IGs—3 for early-stage and 2 for advanced-stage CRC. Molecular docking revealed that IG heavy variable 3-64 (IGHV3-64) exhibited high-affinity binding with Irinotecan [binding free energy (ΔG) = −10.0 kcal/mol] and showed epitope-level pairing with K-Ras at residues 2–17 and 106–114. Additional CPP motif analysis supported the potential of IGHV3-64-derived peptides for intracellular delivery, reinforcing their promise in pPDC development.
Conclusions:
IGHV3-64 emerges as a strong candidate biomarker for advanced-stage CRC, demonstrating consistent binding affinity to Irinotecan and epitope pairing with K-Ras. Its inherent CPP features further support its potential for targeted, intracellular delivery in pPDCs design. These findings highlight a novel direction in personalized cancer immunotherapy, warranting further in vitro and in vivo validation to confirm clinical utility.
Aim:
Colorectal cancer (CRC) remains the second leading cause of cancer-related mortality worldwide. While antibody-drug conjugates (ADCs) offer targeted therapeutic options, they are often limited by toxicity, immunogenicity, complex pharmacokinetics, and high production costs. Polyclonal antibodies—capable of recognizing multiple epitopes—present a promising, yet underexplored, alternative for targeted drug delivery. The stage-specific presence of secreted, stable immunoglobulins (IGs) in CRC and their potential utility in drug conjugation strategies remain largely uncharacterized.
Methods:
This study utilized electrospray ionization nano-liquid chromatography tandem mass spectrometry (ESI-nanoLC-MS/MS) proteomic analysis on pre-treatment plasma samples across CRC stages to identify stage-specific IGs. Venn diagram comparisons refined IG candidates, while protein stability was assessed using ProtParam. Molecular docking simulations (via CB-Dock2), epitope mapping (via CABS-dock), and cell-penetrating peptide (CPP) prediction were integrated to explore epitope pairing between IGs and Kirsten rat sarcoma viral oncogene homolog (K-Ras) neoantigen, evaluating their potential for polyclonal drug conjugates (pPDCs).
Results:
A total of 325 secreted IGs were initially identified, with 46 found to be stage-specific. Protein stability analysis shortlisted 5 IGs—3 for early-stage and 2 for advanced-stage CRC. Molecular docking revealed that IG heavy variable 3-64 (IGHV3-64) exhibited high-affinity binding with Irinotecan [binding free energy (ΔG) = −10.0 kcal/mol] and showed epitope-level pairing with K-Ras at residues 2–17 and 106–114. Additional CPP motif analysis supported the potential of IGHV3-64-derived peptides for intracellular delivery, reinforcing their promise in pPDC development.
Conclusions:
IGHV3-64 emerges as a strong candidate biomarker for advanced-stage CRC, demonstrating consistent binding affinity to Irinotecan and epitope pairing with K-Ras. Its inherent CPP features further support its potential for targeted, intracellular delivery in pPDCs design. These findings highlight a novel direction in personalized cancer immunotherapy, warranting further in vitro and in vivo validation to confirm clinical utility.
DOI: https://doi.org/10.37349/ei.2026.1003252

Opioids remain central to managing moderate to severe pain, yet they also produce significant and often under-recognized effects on the immune system. In this narrative review, we synthesize evidence from 1994 to 2025 across preclinical, translational, observational, and limited interventional studies in adults to examine how different opioid classes modulate immunity and the clinical relevance of these effects. Opioids act directly on immune cells via mu-opioid receptors (MORs), nociceptin/orphanin FQ receptors (NOR), and Toll-like receptor 4 (TLR4), and indirectly through neuroendocrine, autonomic, neuroinflammatory, and gut microbiota-mediated pathways. Immunologic consequences are drug specific: Morphine, fentanyl, and to a lesser extent methadone exhibit pronounced immunosuppressive profiles; oxycodone appears comparatively less suppressive; and buprenorphine and tramadol generally preserve, and may in some contexts enhance, immune function. Clinically, chronic or intensive opioid exposure is associated with increased risk of infection and sepsis-related mortality, potential facilitation of tumor progression or recurrence, impaired perioperative and transplant outcomes, and contributions to tolerance and opioid-induced hyperalgesia, with convergent data indicating these immune effects are intrinsic to opioid pharmacology. Framing opioid-induced immunomodulation as a clinically meaningful, agent-specific phenomenon argues for incorporating immunologic risk into analgesic selection—prioritizing less immunosuppressive opioids where appropriate, considering rotation and tapering strategies, using peripherally acting antagonists, and implementing multimodal analgesia—while underscoring the need for standardized immunologic endpoints, rigorously controlled clinical studies, and development of next-generation analgesics that maintain effective pain relief while minimizing detrimental immune effects.
Opioids remain central to managing moderate to severe pain, yet they also produce significant and often under-recognized effects on the immune system. In this narrative review, we synthesize evidence from 1994 to 2025 across preclinical, translational, observational, and limited interventional studies in adults to examine how different opioid classes modulate immunity and the clinical relevance of these effects. Opioids act directly on immune cells via mu-opioid receptors (MORs), nociceptin/orphanin FQ receptors (NOR), and Toll-like receptor 4 (TLR4), and indirectly through neuroendocrine, autonomic, neuroinflammatory, and gut microbiota-mediated pathways. Immunologic consequences are drug specific: Morphine, fentanyl, and to a lesser extent methadone exhibit pronounced immunosuppressive profiles; oxycodone appears comparatively less suppressive; and buprenorphine and tramadol generally preserve, and may in some contexts enhance, immune function. Clinically, chronic or intensive opioid exposure is associated with increased risk of infection and sepsis-related mortality, potential facilitation of tumor progression or recurrence, impaired perioperative and transplant outcomes, and contributions to tolerance and opioid-induced hyperalgesia, with convergent data indicating these immune effects are intrinsic to opioid pharmacology. Framing opioid-induced immunomodulation as a clinically meaningful, agent-specific phenomenon argues for incorporating immunologic risk into analgesic selection—prioritizing less immunosuppressive opioids where appropriate, considering rotation and tapering strategies, using peripherally acting antagonists, and implementing multimodal analgesia—while underscoring the need for standardized immunologic endpoints, rigorously controlled clinical studies, and development of next-generation analgesics that maintain effective pain relief while minimizing detrimental immune effects.
DOI: https://doi.org/10.37349/ei.2026.1003251
This article belongs to the special issue Immunology and Pain

Aim:
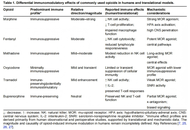
Pneumonia is the leading cause of morbidity and mortality among elderly individuals. This has led to the search for reliable tools such as inflammatory biomarkers, including C-reactive protein (CRP), procalcitonin (PCT), and neutrophil-to-lymphocyte ratio (NLR), to predict disease severity and prognosis. However, the prognostic value of inflammatory biomarkers in elderly women is not fully understood, as the population is often underrepresented in clinical studies.
Methods:
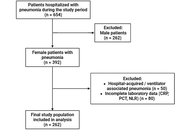
This retrospective study was conducted at Wahidin Sudirohusodo Hospital, Indonesia. The samples used were elderly women aged ≥ 60 years who were hospitalized with community-acquired pneumonia (CAP) between January and December 2023. CRP, PCT, and NLR levels collected within 24 hours of admission were evaluated and correlated with pneumonia severity index (PSI) scores and in-hospital mortality (IHM). Subsequently, receiver operating characteristic (ROC) curve analysis, logistic regression, and Kaplan-Meier survival analysis were performed.
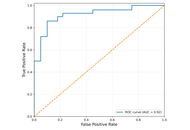
Results:
A total of 262 patients (median age 66 years) were included, of whom 83.2% had mild CAP, and 87.0% survived. Among inflammatory biomarkers, CRP showed the highest, with limited discriminatory ability for mortality [area under the curve (AUC) 0.543], followed by NLR (AUC 0.495), and PCT (AUC 0.466). All markers had high sensitivity (91.2%) but low specificity, and CRP ≥ 14.5 mg/L was significantly associated with reduced survival (p = 0.018).
Conclusions:
CRP shows a modest prognostic value in predicting mortality among elderly women with CAP, while NLR and PCT have limited utility. These results show the need for gender- and age-specific studies to improve risk stratification and outcomes in the vulnerable population.
Aim:
Pneumonia is the leading cause of morbidity and mortality among elderly individuals. This has led to the search for reliable tools such as inflammatory biomarkers, including C-reactive protein (CRP), procalcitonin (PCT), and neutrophil-to-lymphocyte ratio (NLR), to predict disease severity and prognosis. However, the prognostic value of inflammatory biomarkers in elderly women is not fully understood, as the population is often underrepresented in clinical studies.
Methods:
This retrospective study was conducted at Wahidin Sudirohusodo Hospital, Indonesia. The samples used were elderly women aged ≥ 60 years who were hospitalized with community-acquired pneumonia (CAP) between January and December 2023. CRP, PCT, and NLR levels collected within 24 hours of admission were evaluated and correlated with pneumonia severity index (PSI) scores and in-hospital mortality (IHM). Subsequently, receiver operating characteristic (ROC) curve analysis, logistic regression, and Kaplan-Meier survival analysis were performed.
Results:
A total of 262 patients (median age 66 years) were included, of whom 83.2% had mild CAP, and 87.0% survived. Among inflammatory biomarkers, CRP showed the highest, with limited discriminatory ability for mortality [area under the curve (AUC) 0.543], followed by NLR (AUC 0.495), and PCT (AUC 0.466). All markers had high sensitivity (91.2%) but low specificity, and CRP ≥ 14.5 mg/L was significantly associated with reduced survival (p = 0.018).
Conclusions:
CRP shows a modest prognostic value in predicting mortality among elderly women with CAP, while NLR and PCT have limited utility. These results show the need for gender- and age-specific studies to improve risk stratification and outcomes in the vulnerable population.
DOI: https://doi.org/10.37349/ei.2026.1003250

Recurrent infections in children often prompt evaluation for primary immunodeficiency diseases, particularly those affecting humoral immunity. Assessment of memory B cell subsets and vaccine-specific antibody responses provides critical insight into long-term protective immunity. While peripheral blood mononuclear cell (PBMC) isolation is common, direct whole blood staining offers faster processing, reduced cell loss, and better preservation of fragile B cell populations. This comprehensive protocol describes a standardised whole blood flow cytometry method for memory B cell phenotyping, alongside ELISA-based measurement of vaccine-specific antibody titres as part of assessing children with recurrent infections.
Recurrent infections in children often prompt evaluation for primary immunodeficiency diseases, particularly those affecting humoral immunity. Assessment of memory B cell subsets and vaccine-specific antibody responses provides critical insight into long-term protective immunity. While peripheral blood mononuclear cell (PBMC) isolation is common, direct whole blood staining offers faster processing, reduced cell loss, and better preservation of fragile B cell populations. This comprehensive protocol describes a standardised whole blood flow cytometry method for memory B cell phenotyping, alongside ELISA-based measurement of vaccine-specific antibody titres as part of assessing children with recurrent infections.
DOI: https://doi.org/10.37349/ei.2026.1003248

Aim:
To evaluate the relationship between serum calprotectin and serum amyloid A with musculoskeletal ultrasonographic findings in rheumatoid arthritis (RA) patients, as RA is the most common chronic inflammatory joint disease in which the infiltration and activation of inflammatory cells are important. Calprotectin and serum amyloid A protein are over-secreted in response to acute and chronic inflammation. Musculoskeletal ultrasound is more sensitive than physical examination for the evaluation of synovitis.
Methods:
A control group of 30 healthy individuals, 30 patients with active RA, and 30 patients with inactive RA participated in this cross-sectional study. Utilizing the RA disease activity (Disease Activity Score 28, DAS28) score was evaluated. Serum amyloid A and serum calprotectin were measured in all participants, and musculoskeletal ultrasound on the hands and wrists were done for all subjects.
Results:
A significant difference was observed among the studied groups with respect to serum calprotectin and serum amyloid A levels (P < 0.001). A significant positive correlation was observed between serum amyloid A and several inflammatory and clinical parameters, including C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), DAS28 score, serum calprotectin, and synovitis. Similarly, serum calprotectin levels demonstrated a significant positive correlation with ESR, DAS28 score, serum amyloid A, and synovitis. These findings highlight the potential value of both serum amyloid A and serum calprotectin as biomarkers reflecting disease activity and inflammatory burden in RA.
Conclusions:
Serum amyloid A and serum calprotectin can be used as markers of RA activity.
Aim:
To evaluate the relationship between serum calprotectin and serum amyloid A with musculoskeletal ultrasonographic findings in rheumatoid arthritis (RA) patients, as RA is the most common chronic inflammatory joint disease in which the infiltration and activation of inflammatory cells are important. Calprotectin and serum amyloid A protein are over-secreted in response to acute and chronic inflammation. Musculoskeletal ultrasound is more sensitive than physical examination for the evaluation of synovitis.
Methods:
A control group of 30 healthy individuals, 30 patients with active RA, and 30 patients with inactive RA participated in this cross-sectional study. Utilizing the RA disease activity (Disease Activity Score 28, DAS28) score was evaluated. Serum amyloid A and serum calprotectin were measured in all participants, and musculoskeletal ultrasound on the hands and wrists were done for all subjects.
Results:
A significant difference was observed among the studied groups with respect to serum calprotectin and serum amyloid A levels (P < 0.001). A significant positive correlation was observed between serum amyloid A and several inflammatory and clinical parameters, including C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), DAS28 score, serum calprotectin, and synovitis. Similarly, serum calprotectin levels demonstrated a significant positive correlation with ESR, DAS28 score, serum amyloid A, and synovitis. These findings highlight the potential value of both serum amyloid A and serum calprotectin as biomarkers reflecting disease activity and inflammatory burden in RA.
Conclusions:
Serum amyloid A and serum calprotectin can be used as markers of RA activity.
DOI: https://doi.org/10.37349/ei.2026.1003249

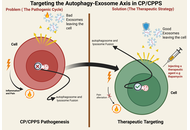
Chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS) is a debilitating condition of the urogenital system, with an elusive and multifactorial pathogenesis. Recent data show that there is a potential interplay between dysregulated autophagy and altered exosomal communication that may contribute to the persistent inflammation and pain characteristic of CP/CPPS. This review synthesizes recent advances to propose a hypothetical model: cellular stress in the prostate may trigger dysfunctional autophagy, which could reprogram secreted exosomes biogenesis and cargo in a series of lipid-raft microdomain-involved mechanisms and secretory autophagy. The outcomes of this process include the release of pro-inflammatory cytokines (e.g., IL-1β, TNF-α) enriched exosomes, damage-associated molecular patterns (DAMPs), microRNAs (e.g., miR-155), and fibrotic mediators (e.g., TGF-β1). These signalosomes are hypothesized to transmit the inflammatory and nociceptive signals and may contribute to coordinating the dysregulation of immune cells (such as the M1 polarization of macrophages), sensitization of neurons, and tissue fibrosis, thereby potentially perpetuating the presence of a chronic disease. We critically assess the available evidence based on human studies, animal models, and in vitro systems, but recognize that there is a present requirement for additional CP/CPPS-specific mechanistic evidence. Furthermore, we explore the translational implications of this axis, discussing its promise for yielding novel exosome-based diagnostic biomarkers and its potential as a therapeutic target, while also highlighting the significant preclinical challenges and risks that must be overcome. Ultimately, the autophagy-exosome axis presents a new, integrative concept of CP/CPPS, which shifts the paradigm to the mechanisms of intercellular communication and provides new possibilities to carry out mechanism-selective research and future treatment options.
Chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS) is a debilitating condition of the urogenital system, with an elusive and multifactorial pathogenesis. Recent data show that there is a potential interplay between dysregulated autophagy and altered exosomal communication that may contribute to the persistent inflammation and pain characteristic of CP/CPPS. This review synthesizes recent advances to propose a hypothetical model: cellular stress in the prostate may trigger dysfunctional autophagy, which could reprogram secreted exosomes biogenesis and cargo in a series of lipid-raft microdomain-involved mechanisms and secretory autophagy. The outcomes of this process include the release of pro-inflammatory cytokines (e.g., IL-1β, TNF-α) enriched exosomes, damage-associated molecular patterns (DAMPs), microRNAs (e.g., miR-155), and fibrotic mediators (e.g., TGF-β1). These signalosomes are hypothesized to transmit the inflammatory and nociceptive signals and may contribute to coordinating the dysregulation of immune cells (such as the M1 polarization of macrophages), sensitization of neurons, and tissue fibrosis, thereby potentially perpetuating the presence of a chronic disease. We critically assess the available evidence based on human studies, animal models, and in vitro systems, but recognize that there is a present requirement for additional CP/CPPS-specific mechanistic evidence. Furthermore, we explore the translational implications of this axis, discussing its promise for yielding novel exosome-based diagnostic biomarkers and its potential as a therapeutic target, while also highlighting the significant preclinical challenges and risks that must be overcome. Ultimately, the autophagy-exosome axis presents a new, integrative concept of CP/CPPS, which shifts the paradigm to the mechanisms of intercellular communication and provides new possibilities to carry out mechanism-selective research and future treatment options.
DOI: https://doi.org/10.37349/ei.2026.1003247

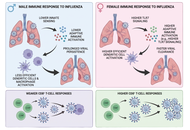
This review highlights the significant sex-based differences in immune responses to influenza infection and vaccination. Men are generally more susceptible to severe influenza outcomes, while women often mount stronger immune responses but experience more adverse effects. These disparities are influenced by biological factors, including sex hormones and genes, as well as gender-related social and environmental conditions. Evidence from both human and animal studies reveals sex-specific variations in antibody production, vaccine effectiveness, and clinical outcomes. Age, hormonal status, and stress further modulate these differences. Understanding these complex interactions is essential for developing tailored and equitable vaccination and treatment strategies.
This review highlights the significant sex-based differences in immune responses to influenza infection and vaccination. Men are generally more susceptible to severe influenza outcomes, while women often mount stronger immune responses but experience more adverse effects. These disparities are influenced by biological factors, including sex hormones and genes, as well as gender-related social and environmental conditions. Evidence from both human and animal studies reveals sex-specific variations in antibody production, vaccine effectiveness, and clinical outcomes. Age, hormonal status, and stress further modulate these differences. Understanding these complex interactions is essential for developing tailored and equitable vaccination and treatment strategies.
DOI: https://doi.org/10.37349/ei.2026.1003244

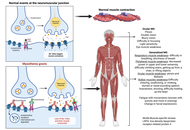
Myasthenia gravis (MG) is an autoimmune disorder of the neuromuscular junction marked by fluctuating muscle weakness, driven by pathogenic antibodies targeting acetylcholine receptors, muscle-specific kinase, or lipoprotein receptor-related protein 4. Traditional treatments such as corticosteroids and immunosuppressants provide symptomatic relief but are limited by systemic toxicity and variable efficacy. Advances in immunology have led to targeted biologics that address specific disease mechanisms, such as B-cell depleting antibodies like rituximab and inebilizumab, complement inhibitors such as eculizumab, ravulizumab, and zilucoplan, and neonatal Fc receptor antagonists like efgartigimod and rozanolixizumab that lower pathogenic IgG levels. Emerging molecular and cellular therapies, including T-regulatory cell therapy, RNA-based modulation, and chimeric antigen receptor T-cell therapy approaches, seek to achieve durable immune tolerance rather than transient suppression. However, therapeutic response varies due to immunologic heterogeneity, long-term safety concerns, and limited global access. Future directions focus on precision medicine, biomarker-guided treatment, and AI-assisted disease monitoring, alongside next-generation therapeutics such as long-acting FcRn inhibitors (IMVT-1402), bispecific antibodies (gefurulimab), and oral small molecules (remibrutinib, iptacopan). Collectively, these developments mark a shift from generalized immunosuppression toward mechanism-based, patient-centered care aimed at restoring lasting immune balance and improving quality of life in MG. This narrative review discusses these aspects to comprehend the existing understanding and challenges.
Myasthenia gravis (MG) is an autoimmune disorder of the neuromuscular junction marked by fluctuating muscle weakness, driven by pathogenic antibodies targeting acetylcholine receptors, muscle-specific kinase, or lipoprotein receptor-related protein 4. Traditional treatments such as corticosteroids and immunosuppressants provide symptomatic relief but are limited by systemic toxicity and variable efficacy. Advances in immunology have led to targeted biologics that address specific disease mechanisms, such as B-cell depleting antibodies like rituximab and inebilizumab, complement inhibitors such as eculizumab, ravulizumab, and zilucoplan, and neonatal Fc receptor antagonists like efgartigimod and rozanolixizumab that lower pathogenic IgG levels. Emerging molecular and cellular therapies, including T-regulatory cell therapy, RNA-based modulation, and chimeric antigen receptor T-cell therapy approaches, seek to achieve durable immune tolerance rather than transient suppression. However, therapeutic response varies due to immunologic heterogeneity, long-term safety concerns, and limited global access. Future directions focus on precision medicine, biomarker-guided treatment, and AI-assisted disease monitoring, alongside next-generation therapeutics such as long-acting FcRn inhibitors (IMVT-1402), bispecific antibodies (gefurulimab), and oral small molecules (remibrutinib, iptacopan). Collectively, these developments mark a shift from generalized immunosuppression toward mechanism-based, patient-centered care aimed at restoring lasting immune balance and improving quality of life in MG. This narrative review discusses these aspects to comprehend the existing understanding and challenges.
DOI: https://doi.org/10.37349/ei.2026.1003246
This article belongs to the special issue Advances in Cellular and Molecular Treatment of Autoimmune Diseases

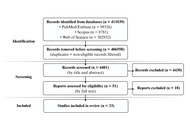
Breast cancer exhibits substantial clinical and molecular heterogeneity, partly shaped by interactions between tumor biology and the host immune system. Germline variants in immune-related genes may influence inflammatory tone, immune regulation, and tumor-immune interactions. However, evidence linking inherited immune genetic variation to breast cancer risk and clinical behavior remains fragmented and heterogeneous across studies. We conducted a structured integrative review of 33 human genetic associations evaluating germline variants in immune-related genes and their associations with breast cancer risk, prognosis, and clinicopathological features. Data were synthesized using a comparative, pathway-oriented analytic framework. Variants in cytokine genes, particularly TGFB1, IL6, IL1B, and IL10, were the most frequently associated with susceptibility, although effect directions varied across populations and genetic models. In contrast, variants in chemokine pathways (CXCL12) and immune checkpoint regulator genes (B7-H4/VTCN1, PD-1/PDCD1) showed consistent associations with tumor progression, immune evasion, and subtype-specific clinical features, including human epidermal growth factor receptor 2 (HER2)-positive disease and metastatic presentation. Across studies, substantial heterogeneity was observed, driven by differences in ethnic composition, sample size, methodological design, and deviations from the Hardy-Weinberg equilibrium. The findings support a pathway-oriented interpretation in which germline immune variation differentially influences immune regulation and tumor progression rather than uniformly determining disease risk. While inflammatory and immunoregulatory pathways appear to shape basal immune tone, immune effector function is less consistently associated with germline variation. Further progress will require extensive, multiethnic studies integrating genetic, transcriptomic, and functional data to clarify how inherited immune variation contributes to breast cancer biology.
Breast cancer exhibits substantial clinical and molecular heterogeneity, partly shaped by interactions between tumor biology and the host immune system. Germline variants in immune-related genes may influence inflammatory tone, immune regulation, and tumor-immune interactions. However, evidence linking inherited immune genetic variation to breast cancer risk and clinical behavior remains fragmented and heterogeneous across studies. We conducted a structured integrative review of 33 human genetic associations evaluating germline variants in immune-related genes and their associations with breast cancer risk, prognosis, and clinicopathological features. Data were synthesized using a comparative, pathway-oriented analytic framework. Variants in cytokine genes, particularly TGFB1, IL6, IL1B, and IL10, were the most frequently associated with susceptibility, although effect directions varied across populations and genetic models. In contrast, variants in chemokine pathways (CXCL12) and immune checkpoint regulator genes (B7-H4/VTCN1, PD-1/PDCD1) showed consistent associations with tumor progression, immune evasion, and subtype-specific clinical features, including human epidermal growth factor receptor 2 (HER2)-positive disease and metastatic presentation. Across studies, substantial heterogeneity was observed, driven by differences in ethnic composition, sample size, methodological design, and deviations from the Hardy-Weinberg equilibrium. The findings support a pathway-oriented interpretation in which germline immune variation differentially influences immune regulation and tumor progression rather than uniformly determining disease risk. While inflammatory and immunoregulatory pathways appear to shape basal immune tone, immune effector function is less consistently associated with germline variation. Further progress will require extensive, multiethnic studies integrating genetic, transcriptomic, and functional data to clarify how inherited immune variation contributes to breast cancer biology.
DOI: https://doi.org/10.37349/ei.2026.1003245

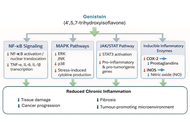
Genistein, a soy-derived isoflavone, has attracted considerable interest for its anti-inflammatory and anticancer properties. Chronic inflammation is a major driver of tumour initiation, progression, metastasis, and therapeutic resistance, creating a need for multi-target agents capable of modulating both inflammatory and oncogenic pathways. This review critically evaluates the molecular mechanisms, pharmacological basis, and translational prospects of genistein in inflammation-related cancers, incorporating recent advances in omics technologies and nanotechnology-based delivery systems. A comprehensive survey of the literature from major scientific databases was conducted, and evidence was assessed based on mechanistic depth, strength of data, and relevance to clinical translation. Genistein modulates multiple interconnected signalling pathways, including NF-κB, MAPK, PI3K/Akt, JAK/STAT, and apoptotic cascades, leading to reduced pro-inflammatory cytokine production, oxidative stress, and tumour cell proliferation. Epigenetic regulation, growth-factor pathway inhibition, and non-coding RNA modulation further contribute to its pleiotropic effects. Nanotechnology-based delivery platforms are being explored to improve their poor oral bioavailability and therapeutic performance in preclinical models. However, clinical translation remains limited by low bioavailability, context-dependent estrogenic activity, inter-individual metabolic variability, and the lack of large-scale, well-controlled clinical trials, standardized dosing, and validated biomarkers. Genistein represents a promising multi-target lead compound at the intersection of inflammation, immunology, and oncology, with future progress dependent on integrating multi-omics data and advanced delivery technologies for personalised clinical applications.
Genistein, a soy-derived isoflavone, has attracted considerable interest for its anti-inflammatory and anticancer properties. Chronic inflammation is a major driver of tumour initiation, progression, metastasis, and therapeutic resistance, creating a need for multi-target agents capable of modulating both inflammatory and oncogenic pathways. This review critically evaluates the molecular mechanisms, pharmacological basis, and translational prospects of genistein in inflammation-related cancers, incorporating recent advances in omics technologies and nanotechnology-based delivery systems. A comprehensive survey of the literature from major scientific databases was conducted, and evidence was assessed based on mechanistic depth, strength of data, and relevance to clinical translation. Genistein modulates multiple interconnected signalling pathways, including NF-κB, MAPK, PI3K/Akt, JAK/STAT, and apoptotic cascades, leading to reduced pro-inflammatory cytokine production, oxidative stress, and tumour cell proliferation. Epigenetic regulation, growth-factor pathway inhibition, and non-coding RNA modulation further contribute to its pleiotropic effects. Nanotechnology-based delivery platforms are being explored to improve their poor oral bioavailability and therapeutic performance in preclinical models. However, clinical translation remains limited by low bioavailability, context-dependent estrogenic activity, inter-individual metabolic variability, and the lack of large-scale, well-controlled clinical trials, standardized dosing, and validated biomarkers. Genistein represents a promising multi-target lead compound at the intersection of inflammation, immunology, and oncology, with future progress dependent on integrating multi-omics data and advanced delivery technologies for personalised clinical applications.
DOI: https://doi.org/10.37349/ei.2026.1003243

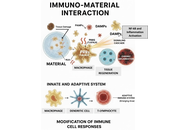
Immuno-materials, which represent a confluence of immunology and materials science, are dramatically transforming the fields of healthcare. Through the targeted modulation of immune responses, these innovative materials offer promising avenues for advancements in vaccine development, immunotherapy, tissue engineering, and diagnostics. This review examines the fundamental principles of immuno-materials, elucidating key concepts and methodologies pertinent to their development. We investigate strategies for engineering materials that can elicit customized immune responses, capable of both stimulation and suppression, and we discuss their potential roles as adjuvants, carriers, or scaffolds. Furthermore, we address the challenges and opportunities associated with the clinical translation of these materials, emphasizing the critical importance of biocompatibility, safety, and efficacy. This review aims to critically evaluate and contextualize the transformative potential of immuno-materials, mapping their journey from laboratory design to proven clinical applications in enhancing human health.
Immuno-materials, which represent a confluence of immunology and materials science, are dramatically transforming the fields of healthcare. Through the targeted modulation of immune responses, these innovative materials offer promising avenues for advancements in vaccine development, immunotherapy, tissue engineering, and diagnostics. This review examines the fundamental principles of immuno-materials, elucidating key concepts and methodologies pertinent to their development. We investigate strategies for engineering materials that can elicit customized immune responses, capable of both stimulation and suppression, and we discuss their potential roles as adjuvants, carriers, or scaffolds. Furthermore, we address the challenges and opportunities associated with the clinical translation of these materials, emphasizing the critical importance of biocompatibility, safety, and efficacy. This review aims to critically evaluate and contextualize the transformative potential of immuno-materials, mapping their journey from laboratory design to proven clinical applications in enhancing human health.
DOI: https://doi.org/10.37349/ei.2026.1003242
This article belongs to the special issue Immuno-Materials: at the interdisciplinary of immunology and materials

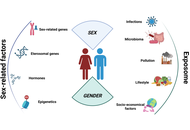
Autoimmune diseases (ADs) comprise a heterogeneous group of disorders characterised by a breakdown of immune tolerance and chronic immune-mediated tissue damage. Their onset and progression result from a complex interplay between genetic susceptibility, hormonal influences, immune regulatory mechanisms, and environmental exposures collectively referred to as the exposome. A striking feature of ADs is their pronounced sexual dimorphism, with a markedly higher prevalence in women for most conditions, alongside sex-specific differences in clinical presentation, severity, and prognosis. This review provides a comprehensive overview of the genetic and environmental determinants contributing to autoimmunity, with particular emphasis on how sex- and gender-related factors shape immune tolerance and disease susceptibility. We discuss central and peripheral tolerance mechanisms, the role of key regulators such as AIRE and FOXP3, sex chromosome-linked effects including X-chromosome inactivation, and the immunomodulatory impact of sex hormones across different life stages. Furthermore, we examine how environmental exposures, infections, and geographic variability interact with genetic background and sex-specific immune regulation to influence autoimmune disease risk. By integrating biological sex and gender-related factors within the framework of immune tolerance and the exposome, this review highlights the need for a sex-aware and personalised approach to understanding, diagnosing, and treating ADs.
Autoimmune diseases (ADs) comprise a heterogeneous group of disorders characterised by a breakdown of immune tolerance and chronic immune-mediated tissue damage. Their onset and progression result from a complex interplay between genetic susceptibility, hormonal influences, immune regulatory mechanisms, and environmental exposures collectively referred to as the exposome. A striking feature of ADs is their pronounced sexual dimorphism, with a markedly higher prevalence in women for most conditions, alongside sex-specific differences in clinical presentation, severity, and prognosis. This review provides a comprehensive overview of the genetic and environmental determinants contributing to autoimmunity, with particular emphasis on how sex- and gender-related factors shape immune tolerance and disease susceptibility. We discuss central and peripheral tolerance mechanisms, the role of key regulators such as AIRE and FOXP3, sex chromosome-linked effects including X-chromosome inactivation, and the immunomodulatory impact of sex hormones across different life stages. Furthermore, we examine how environmental exposures, infections, and geographic variability interact with genetic background and sex-specific immune regulation to influence autoimmune disease risk. By integrating biological sex and gender-related factors within the framework of immune tolerance and the exposome, this review highlights the need for a sex-aware and personalised approach to understanding, diagnosing, and treating ADs.
DOI: https://doi.org/10.37349/ei.2026.1003241

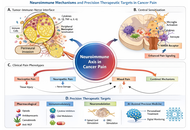
Pain is among the most prevalent and debilitating symptoms experienced by patients with cancer, significantly impairing functional status and overall quality of life. Its origin is multifactorial, involving direct tumor infiltration, treatment-induced tissue and neural injury, and immune-mediated inflammatory processes. This section examines the epidemiology of cancer-related pain, delineates its complex pathophysiology with particular focus on neuroimmune interactions, and reviews current pharmacological and interventional approaches for managing both acute and chronic pain in oncological settings. Advancing knowledge of immune-driven pain mechanisms holds promise for the development of more targeted, effective, and personalized analgesic strategies in cancer care.
Pain is among the most prevalent and debilitating symptoms experienced by patients with cancer, significantly impairing functional status and overall quality of life. Its origin is multifactorial, involving direct tumor infiltration, treatment-induced tissue and neural injury, and immune-mediated inflammatory processes. This section examines the epidemiology of cancer-related pain, delineates its complex pathophysiology with particular focus on neuroimmune interactions, and reviews current pharmacological and interventional approaches for managing both acute and chronic pain in oncological settings. Advancing knowledge of immune-driven pain mechanisms holds promise for the development of more targeted, effective, and personalized analgesic strategies in cancer care.
DOI: https://doi.org/10.37349/ei.2026.1003240

Atopic dermatitis is a chronic inflammatory skin condition characterized by immune dysregulation and dysfunction of the epidermal barrier. Although occludin, a vital tight junction protein, is essential for preserving barrier integrity, very little is known about its potential as a serum biomarker in atopic dermatitis. This pilot case-control study measured serum occludin levels in 30 adult atopic dermatitis patients and 30 age- and sex-matched healthy controls between May and August 2025, using a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kit. Findings showed serum occludin levels were significantly higher in atopic dermatitis patients (mean: 2.50 ± 1.10 ng/mL) compared to healthy controls (mean: 0.68 ± 0.69 ng/mL; p < 0.001), with levels showing a decreasing trend with age. Serum occludin exhibited high diagnostic performance [area under the curve (AUC) = 0.92] and represents a sensitive and specific, non-invasive biomarker for atopic dermatitis. These preliminary findings lay the groundwork for future studies exploring serum occludin as a potential biomarker for atopic dermatitis assessment.
Atopic dermatitis is a chronic inflammatory skin condition characterized by immune dysregulation and dysfunction of the epidermal barrier. Although occludin, a vital tight junction protein, is essential for preserving barrier integrity, very little is known about its potential as a serum biomarker in atopic dermatitis. This pilot case-control study measured serum occludin levels in 30 adult atopic dermatitis patients and 30 age- and sex-matched healthy controls between May and August 2025, using a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kit. Findings showed serum occludin levels were significantly higher in atopic dermatitis patients (mean: 2.50 ± 1.10 ng/mL) compared to healthy controls (mean: 0.68 ± 0.69 ng/mL; p < 0.001), with levels showing a decreasing trend with age. Serum occludin exhibited high diagnostic performance [area under the curve (AUC) = 0.92] and represents a sensitive and specific, non-invasive biomarker for atopic dermatitis. These preliminary findings lay the groundwork for future studies exploring serum occludin as a potential biomarker for atopic dermatitis assessment.
DOI: https://doi.org/10.37349/ei.2026.1003239

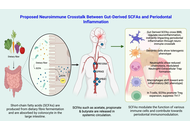
Short-chain fatty acids (SCFAs), primarily acetate, propionate, and butyrate, are microbial metabolites generated through the fermentation of dietary fibers by the gut microbiota and are increasingly recognized as critical regulators of host immune homeostasis. Beyond their metabolic roles, SCFAs exert potent immunomodulatory effects across mucosal tissues, including the periodontium, by shaping both innate and adaptive immune responses. This review synthesizes current evidence on the dual roles of SCFAs in periodontal health and disease, with particular emphasis on the contrasting effects of systemically derived versus locally accumulated SCFAs within periodontal tissues. Gut-derived SCFAs absorbed into the circulation modulate immune function through activation of G protein-coupled receptors (GPR41, GPR43, and GPR109A) and inhibition of histone deacetylases. These pathways promote regulatory T cell differentiation, skew macrophage polarization toward anti-inflammatory M2 phenotypes, and regulate neutrophil and dendritic cell activity. These effects support immune tolerance, epithelial barrier integrity, and resolution of inflammation, thereby contributing to periodontal homeostasis. In contrast, SCFAs produced locally within periodontal pockets can reach millimolar concentrations that disrupt epithelial and fibroblast function, induce oxidative stress, and amplify inflammatory signalling, ultimately driving tissue destruction and disease progression. Emerging evidence links gut microbial composition and systemic SCFA availability to periodontal inflammation through immune and neuroimmune crosstalk, supporting a biologically plausible gut-oral axis. Translational strategies aimed at harnessing the immunoregulatory properties of SCFAs are critically evaluated in this review. While these approaches are promising, challenges related to dosing, delivery, inter-individual variability, and limited human interventional data remain, underscoring the need for rigorously designed translational studies.
Short-chain fatty acids (SCFAs), primarily acetate, propionate, and butyrate, are microbial metabolites generated through the fermentation of dietary fibers by the gut microbiota and are increasingly recognized as critical regulators of host immune homeostasis. Beyond their metabolic roles, SCFAs exert potent immunomodulatory effects across mucosal tissues, including the periodontium, by shaping both innate and adaptive immune responses. This review synthesizes current evidence on the dual roles of SCFAs in periodontal health and disease, with particular emphasis on the contrasting effects of systemically derived versus locally accumulated SCFAs within periodontal tissues. Gut-derived SCFAs absorbed into the circulation modulate immune function through activation of G protein-coupled receptors (GPR41, GPR43, and GPR109A) and inhibition of histone deacetylases. These pathways promote regulatory T cell differentiation, skew macrophage polarization toward anti-inflammatory M2 phenotypes, and regulate neutrophil and dendritic cell activity. These effects support immune tolerance, epithelial barrier integrity, and resolution of inflammation, thereby contributing to periodontal homeostasis. In contrast, SCFAs produced locally within periodontal pockets can reach millimolar concentrations that disrupt epithelial and fibroblast function, induce oxidative stress, and amplify inflammatory signalling, ultimately driving tissue destruction and disease progression. Emerging evidence links gut microbial composition and systemic SCFA availability to periodontal inflammation through immune and neuroimmune crosstalk, supporting a biologically plausible gut-oral axis. Translational strategies aimed at harnessing the immunoregulatory properties of SCFAs are critically evaluated in this review. While these approaches are promising, challenges related to dosing, delivery, inter-individual variability, and limited human interventional data remain, underscoring the need for rigorously designed translational studies.
DOI: https://doi.org/10.37349/ei.2026.1003238

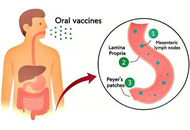
For decades, vaccines have been a key tool against microbial infections. However, the high cost of production and purification renders vaccines largely inaccessible to many developing countries. The limitations of conventional vaccines can be overcome by edible vaccines. To produce an oral vaccine, favourable vectors, such as plants and probiotics, are used. Recent studies have revealed the immunomodulatory effects of probiotics. To improve the efficacy of these vaccines, several adjuvant approaches have been employed. Postbiotics can be used as promising therapy for preventing infections and enhancing the host immune system due to their unique biochemical and microbial-derived properties. In this review, we discuss the feasibility of postbiotics as adjuvants for oral vaccines, highlighting their mechanisms of action, safety profile, and potential to enhance both mucosal and systemic immune responses.
For decades, vaccines have been a key tool against microbial infections. However, the high cost of production and purification renders vaccines largely inaccessible to many developing countries. The limitations of conventional vaccines can be overcome by edible vaccines. To produce an oral vaccine, favourable vectors, such as plants and probiotics, are used. Recent studies have revealed the immunomodulatory effects of probiotics. To improve the efficacy of these vaccines, several adjuvant approaches have been employed. Postbiotics can be used as promising therapy for preventing infections and enhancing the host immune system due to their unique biochemical and microbial-derived properties. In this review, we discuss the feasibility of postbiotics as adjuvants for oral vaccines, highlighting their mechanisms of action, safety profile, and potential to enhance both mucosal and systemic immune responses.
DOI: https://doi.org/10.37349/ei.2026.1003237

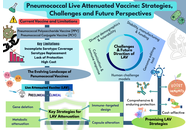
Pneumococcal disease remains a major global health challenge despite the availability of polysaccharide and conjugate vaccines. Although these platforms have reduced invasive disease, their limitations, such as poor immunogenicity in infants, lack of durable protection, and restricted coverage, highlight the need to explore innovative preventive strategies. Next-generation vaccines that provide comprehensive protection, sustained immunity, and cost-effectiveness are urgently required. Live attenuated vaccines (LAVs) represent a promising frontier in this effort, with recent advances focused on overcoming developmental and safety challenges. This review highlights the evolving pneumococcal vaccine landscape, with emphasis on LAV strategies. We summarize the strengths and shortcomings of current vaccines, examine recent advances in LAV development, including key aspects of attenuation, immune-protective mechanisms, and delivery approaches. LAVs demonstrate potential to induce balanced mucosal, humoral, and cellular immunity, addressing critical gaps left by existing platforms. Key challenges related to genetic stability, safety, and translational applicability are also discussed. By synthesizing established knowledge and highlighting advancements, this review underscores the promise of LAVs as next-generation candidates that can provide broader, longer-lasting protection against pneumococcal disease.
Pneumococcal disease remains a major global health challenge despite the availability of polysaccharide and conjugate vaccines. Although these platforms have reduced invasive disease, their limitations, such as poor immunogenicity in infants, lack of durable protection, and restricted coverage, highlight the need to explore innovative preventive strategies. Next-generation vaccines that provide comprehensive protection, sustained immunity, and cost-effectiveness are urgently required. Live attenuated vaccines (LAVs) represent a promising frontier in this effort, with recent advances focused on overcoming developmental and safety challenges. This review highlights the evolving pneumococcal vaccine landscape, with emphasis on LAV strategies. We summarize the strengths and shortcomings of current vaccines, examine recent advances in LAV development, including key aspects of attenuation, immune-protective mechanisms, and delivery approaches. LAVs demonstrate potential to induce balanced mucosal, humoral, and cellular immunity, addressing critical gaps left by existing platforms. Key challenges related to genetic stability, safety, and translational applicability are also discussed. By synthesizing established knowledge and highlighting advancements, this review underscores the promise of LAVs as next-generation candidates that can provide broader, longer-lasting protection against pneumococcal disease.
DOI: https://doi.org/10.37349/ei.2026.1003236
This article belongs to the special issue Novel Vaccines development for Emerging, Acute, and Re-emerging Infectious Diseases
 Previous
Previous