


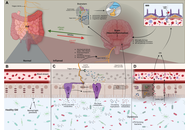
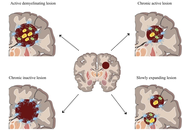

This Perspective explores why hyperbaric oxygen therapy (HBOT) deserves closer clinical and scientific attention as a possible treatment for ischemic stroke and a potential neuroprotective strategy in chronic central nervous system disorders, especially multiple sclerosis and other progressive or age-related neurological conditions. While HBOT is not yet widely accepted for these indications, three factors justify reconsideration: its strong safety profile, a biologically plausible mechanistic rationale, and recurrent signals of benefit from selected neurological studies. We review HBOT’s historical development, current accepted indications, and evidence that adverse effects are generally mild, uncommon, and reversible under modern protocols. In neurology, efficacy remains unproven, yet interest persists due to preliminary findings and repeated patient-reported improvements. A central argument is that the main barrier to progress is not safety, but evidence generation. Conventional randomized controlled trials face major challenges: difficult blinding, wide variation in dosing protocols, and uncertainty about meaningful outcomes in chronic neurological disease. Moreover, HBOT is non-patentable, which limits commercial investment and leaves a potentially valuable intervention underexplored. Mechanistically, we move beyond explanations centred solely on oxygen delivery or oxidative stress. As a working hypothesis, neurological benefits may partly arise from cumulative adaptive responses—including a rebound hormesis following repeated hyperoxic exposure. We conclude with a pragmatic research agenda: continuous, low-cost physiological monitoring in patients already receiving HBOT, coupled with a medium-term goal of adequately powered efficacy trials.
This Perspective explores why hyperbaric oxygen therapy (HBOT) deserves closer clinical and scientific attention as a possible treatment for ischemic stroke and a potential neuroprotective strategy in chronic central nervous system disorders, especially multiple sclerosis and other progressive or age-related neurological conditions. While HBOT is not yet widely accepted for these indications, three factors justify reconsideration: its strong safety profile, a biologically plausible mechanistic rationale, and recurrent signals of benefit from selected neurological studies. We review HBOT’s historical development, current accepted indications, and evidence that adverse effects are generally mild, uncommon, and reversible under modern protocols. In neurology, efficacy remains unproven, yet interest persists due to preliminary findings and repeated patient-reported improvements. A central argument is that the main barrier to progress is not safety, but evidence generation. Conventional randomized controlled trials face major challenges: difficult blinding, wide variation in dosing protocols, and uncertainty about meaningful outcomes in chronic neurological disease. Moreover, HBOT is non-patentable, which limits commercial investment and leaves a potentially valuable intervention underexplored. Mechanistically, we move beyond explanations centred solely on oxygen delivery or oxidative stress. As a working hypothesis, neurological benefits may partly arise from cumulative adaptive responses—including a rebound hormesis following repeated hyperoxic exposure. We conclude with a pragmatic research agenda: continuous, low-cost physiological monitoring in patients already receiving HBOT, coupled with a medium-term goal of adequately powered efficacy trials.
DOI: https://doi.org/10.37349/ent.2026.1004157

Aim:
To assess the effect of a synbiotic supplement composed of Enterococcus faecium and agave inulin on cognitive function in older adults with mild cognitive impairment (MCI).
Methods:
In a triple-blind randomized crossover trial, nineteen adults aged 64–85 years with MCI received either the synbiotic or an isocaloric vehicle for eight weeks, followed by a three-week washout and treatment crossover. Cognitive outcomes were assessed at baseline, after the first intervention, and after crossover using the Modified Mini-Mental State Examination (MMSE), Rey-Osterrieth Complex Figure Test (RCFT; copy and memory), and Abbreviated Instrument for Expectations of Self-Efficacy for Daily Activities in Older Adults (AERAC) self-efficacy scale. Paired and unpaired Student’s t-tests were used for statistical comparisons (p < 0.05).
Results:
The synbiotic group showed significant improvement relative to baseline across all domains: MMSE (p = 0.05), AERAC (p = 0.005), RCFT-copy (p = 0.03), and RCFT-memory (p = 0.03). Post-treatment comparisons between groups also favored the synbiotic, with significant differences in MMSE (p = 0.001), AERAC (p = 0.001), RCFT-copy (p = 0.0095), and RCFT-memory (p = 0.001). After crossover, cognitive gains were sustained and reproduced. MMSE scores reached 17.89 ± 1.45 in the synbiotic-first group versus 18.20 ± 0.63 in the control-first group (p < 0.001). RCFT-copy remained high (29.83 ± 4.18 vs. 29.52 ± 5.60, p = 0.0157), while RCFT-memory scores differed (17.56 ± 6.73 vs. 17.20 ± 3.29, p = 0.0005). AERAC scores continued to improve during crossover (82.60 ± 10.49 vs. 85.46 ± 8.28, p < 0.001). No adverse effects occurred.
Conclusions:
Synbiotic supplementation significantly improved global cognition, visuoconstructive ability, memory, and functional self-efficacy in older adults with MCI. Benefits persisted beyond the initial intervention and were replicated when the control group received the synbiotic, supporting its potential as a safe and effective strategy to mitigate age-related cognitive decline.
Aim:
To assess the effect of a synbiotic supplement composed of Enterococcus faecium and agave inulin on cognitive function in older adults with mild cognitive impairment (MCI).
Methods:
In a triple-blind randomized crossover trial, nineteen adults aged 64–85 years with MCI received either the synbiotic or an isocaloric vehicle for eight weeks, followed by a three-week washout and treatment crossover. Cognitive outcomes were assessed at baseline, after the first intervention, and after crossover using the Modified Mini-Mental State Examination (MMSE), Rey-Osterrieth Complex Figure Test (RCFT; copy and memory), and Abbreviated Instrument for Expectations of Self-Efficacy for Daily Activities in Older Adults (AERAC) self-efficacy scale. Paired and unpaired Student’s t-tests were used for statistical comparisons (p < 0.05).
Results:
The synbiotic group showed significant improvement relative to baseline across all domains: MMSE (p = 0.05), AERAC (p = 0.005), RCFT-copy (p = 0.03), and RCFT-memory (p = 0.03). Post-treatment comparisons between groups also favored the synbiotic, with significant differences in MMSE (p = 0.001), AERAC (p = 0.001), RCFT-copy (p = 0.0095), and RCFT-memory (p = 0.001). After crossover, cognitive gains were sustained and reproduced. MMSE scores reached 17.89 ± 1.45 in the synbiotic-first group versus 18.20 ± 0.63 in the control-first group (p < 0.001). RCFT-copy remained high (29.83 ± 4.18 vs. 29.52 ± 5.60, p = 0.0157), while RCFT-memory scores differed (17.56 ± 6.73 vs. 17.20 ± 3.29, p = 0.0005). AERAC scores continued to improve during crossover (82.60 ± 10.49 vs. 85.46 ± 8.28, p < 0.001). No adverse effects occurred.
Conclusions:
Synbiotic supplementation significantly improved global cognition, visuoconstructive ability, memory, and functional self-efficacy in older adults with MCI. Benefits persisted beyond the initial intervention and were replicated when the control group received the synbiotic, supporting its potential as a safe and effective strategy to mitigate age-related cognitive decline.
DOI: https://doi.org/10.37349/ent.2026.1004156
This article belongs to the special issue Role of Microbiota in Neurological Diseases

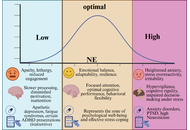
Norepinephrine (NE), a central catecholamine neurotransmitter synthesized primarily in the locus coeruleus (LC), plays a critical role in regulating arousal, attention, emotional processing, and stress responsiveness. While contemporary personality neuroscience has established the role of NE in acute psychological states, its contribution to stable personality traits remains underexplored. This review synthesizes neurobiological, psychological, genetic, and psychopharmacological evidence to propose a NE-personality continuum that links tonic and phasic dynamics of the LC-NE system to enduring individual differences in alertness, anxiety, and adaptability. Alertness is associated with optimal noradrenergic tone and efficient phasic signaling, which enhances the signal-to-noise ratio and attentional focus. Anxiety arises from chronic hyperactivation or dysregulated NE release, particularly involving excessive α1- and β-adrenergic receptor activity and impaired modulation from the prefrontal cortex. Adaptability denotes a harmonious interaction between the limbic system and prefrontal cortex, which facilitates cognitive flexibility and emotional regulation in response to changing environmental demands. The connection between NE activity and personality traits follows an inverted U-shaped pattern. Low tone leads to apathy and less engagement, moderate tone helps with resilience and optimal functioning, and high tone leads to hypervigilance and rigidity. This model combines findings from fundamental neuroscience and clinical research to provide a physiologically based framework for understanding how long-term variations in noradrenergic regulation affect personality traits, as described in established trait theories. The findings underline the feasibility of adding noradrenergic biomarkers and pharmaceutical therapies into clinical practice, as well as the importance of longitudinal and multimodal research to determine trait-level causality. This is especially important for understanding how to use these elements to improve treatment plans for personality disorders.
Norepinephrine (NE), a central catecholamine neurotransmitter synthesized primarily in the locus coeruleus (LC), plays a critical role in regulating arousal, attention, emotional processing, and stress responsiveness. While contemporary personality neuroscience has established the role of NE in acute psychological states, its contribution to stable personality traits remains underexplored. This review synthesizes neurobiological, psychological, genetic, and psychopharmacological evidence to propose a NE-personality continuum that links tonic and phasic dynamics of the LC-NE system to enduring individual differences in alertness, anxiety, and adaptability. Alertness is associated with optimal noradrenergic tone and efficient phasic signaling, which enhances the signal-to-noise ratio and attentional focus. Anxiety arises from chronic hyperactivation or dysregulated NE release, particularly involving excessive α1- and β-adrenergic receptor activity and impaired modulation from the prefrontal cortex. Adaptability denotes a harmonious interaction between the limbic system and prefrontal cortex, which facilitates cognitive flexibility and emotional regulation in response to changing environmental demands. The connection between NE activity and personality traits follows an inverted U-shaped pattern. Low tone leads to apathy and less engagement, moderate tone helps with resilience and optimal functioning, and high tone leads to hypervigilance and rigidity. This model combines findings from fundamental neuroscience and clinical research to provide a physiologically based framework for understanding how long-term variations in noradrenergic regulation affect personality traits, as described in established trait theories. The findings underline the feasibility of adding noradrenergic biomarkers and pharmaceutical therapies into clinical practice, as well as the importance of longitudinal and multimodal research to determine trait-level causality. This is especially important for understanding how to use these elements to improve treatment plans for personality disorders.
DOI: https://doi.org/10.37349/ent.2026.1004155

The NEUROD2 gene encodes a transcription factor essential for neuronal differentiation and cortical development. Pathogenic variants cause a rare autosomal dominant neurodevelopmental disorder with variable expressivity, typically presenting in early infancy with developmental delay, epilepsy, and behavioral abnormalities. We report a newborn girl carrying a de novo heterozygous missense variant NM_006160.4:c.790G>A, p.(Ala264Thr), located outside the canonical basic helix-loop-helix (bHLH) domain. Soon after birth, she presented respiratory depression, hypotonia, feeding difficulties, and electrographic seizures. Magnetic resonance imaging (MRI) showed subcortical white matter hyperintensity, and the electroencephalogram (EEG) revealed abnormal background activity. During follow-up, epilepsy was controlled, but neurodevelopmental delay with autistic features emerged. This case represents the earliest reported clinical onset associated with NEUROD2 variants and expands the phenotypic and mutational spectrum. It highlights that variants outside known hotspots can cause severe disease and supports including NEUROD2 in the differential diagnosis of neonatal neurological impairment.
The NEUROD2 gene encodes a transcription factor essential for neuronal differentiation and cortical development. Pathogenic variants cause a rare autosomal dominant neurodevelopmental disorder with variable expressivity, typically presenting in early infancy with developmental delay, epilepsy, and behavioral abnormalities. We report a newborn girl carrying a de novo heterozygous missense variant NM_006160.4:c.790G>A, p.(Ala264Thr), located outside the canonical basic helix-loop-helix (bHLH) domain. Soon after birth, she presented respiratory depression, hypotonia, feeding difficulties, and electrographic seizures. Magnetic resonance imaging (MRI) showed subcortical white matter hyperintensity, and the electroencephalogram (EEG) revealed abnormal background activity. During follow-up, epilepsy was controlled, but neurodevelopmental delay with autistic features emerged. This case represents the earliest reported clinical onset associated with NEUROD2 variants and expands the phenotypic and mutational spectrum. It highlights that variants outside known hotspots can cause severe disease and supports including NEUROD2 in the differential diagnosis of neonatal neurological impairment.
DOI: https://doi.org/10.37349/ent.2026.1004154
This article belongs to the special issue Neuroprotection in Pediatric Neurological Disorders: from Rare Diseases to Perinatal Brain Injury

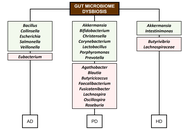
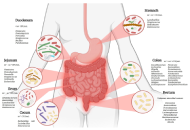
By individual examination, the present review provides an overview of the potential involvement of various human microbiomes, including the gut, oral, skin, and nasal, in the pathophysiology of neurodegenerative diseases. Research has demonstrated that gut microbiome dysbiosis is linked to the pathogenesis of neurodegenerative conditions, including Alzheimer’s, Parkinson’s, and Huntington’s diseases, through mechanisms involving microbial metabolites, neuroinflammation, amyloid aggregation, and altered neurotransmission. Emerging evidence suggests that the oral, skin, and nasal microbiomes may also influence neurodegenerative diseases through mechanisms such as microbial translocation, immune modulation, metabolite production, and interactions with the gut-brain axis. Although the potential of microbiome-based interventions for neurodegenerative diseases has been highlighted, several gaps remain, such as variability between human and animal models, a lack of standardized multi-omics approaches, and a limited understanding of individual microbial roles. Future studies should focus on clarifying the mechanisms by which dysbiosis in human host microbiomes impacts the pathophysiology of neurodegenerative diseases, identifying reliable biomarkers, and developing safe and effective microbiome-based therapies.
By individual examination, the present review provides an overview of the potential involvement of various human microbiomes, including the gut, oral, skin, and nasal, in the pathophysiology of neurodegenerative diseases. Research has demonstrated that gut microbiome dysbiosis is linked to the pathogenesis of neurodegenerative conditions, including Alzheimer’s, Parkinson’s, and Huntington’s diseases, through mechanisms involving microbial metabolites, neuroinflammation, amyloid aggregation, and altered neurotransmission. Emerging evidence suggests that the oral, skin, and nasal microbiomes may also influence neurodegenerative diseases through mechanisms such as microbial translocation, immune modulation, metabolite production, and interactions with the gut-brain axis. Although the potential of microbiome-based interventions for neurodegenerative diseases has been highlighted, several gaps remain, such as variability between human and animal models, a lack of standardized multi-omics approaches, and a limited understanding of individual microbial roles. Future studies should focus on clarifying the mechanisms by which dysbiosis in human host microbiomes impacts the pathophysiology of neurodegenerative diseases, identifying reliable biomarkers, and developing safe and effective microbiome-based therapies.
DOI: https://doi.org/10.37349/ent.2026.1004153
This article belongs to the special issue Role of Microbiota in Neurological Diseases

Short-chain fatty acids (SCFAs) are microbial-derived metabolites produced primarily through the fermentation of dietary fibre by the intestinal microbiota. Current evidence indicates that they play a key role in modulating nociception and pain processing across immune, metabolic, and neural pathways. The prevailing view that SCFAs suppress pain has been challenged by emerging evidence demonstrating that these same metabolites can also drive hyperalgesia. This apparent “SCFA paradox” persists because most studies have examined individual metabolites in isolation rather than considering them within their broader biological context. Here, we propose an integrative framework in which SCFAs function within a competitive receptor triad, and pain outcomes are dictated by the balance among three signalling axes: a pro-inflammatory immune axis driven by acetate acting through G protein-coupled receptor 43 (GPR43), a pro-resolutive metabolic axis mediated by butyrate via histone deacetylase (HDAC) inhibition and activation of GPR109A, and a direct neural sensing axis triggered by propionate through olfactory receptor 78 (OLFR78). Chronic pain, therefore, does not arise simply from the presence or absence of SCFAs, but from the pathological dominance of one of these axes shaped by specific dysbiosis profiles. This framework moves beyond correlation by providing a mechanistic basis for precision interventions designed to rebalance SCFA signalling, offering novel therapeutic opportunities for neuropathic and inflammatory pain conditions.
Short-chain fatty acids (SCFAs) are microbial-derived metabolites produced primarily through the fermentation of dietary fibre by the intestinal microbiota. Current evidence indicates that they play a key role in modulating nociception and pain processing across immune, metabolic, and neural pathways. The prevailing view that SCFAs suppress pain has been challenged by emerging evidence demonstrating that these same metabolites can also drive hyperalgesia. This apparent “SCFA paradox” persists because most studies have examined individual metabolites in isolation rather than considering them within their broader biological context. Here, we propose an integrative framework in which SCFAs function within a competitive receptor triad, and pain outcomes are dictated by the balance among three signalling axes: a pro-inflammatory immune axis driven by acetate acting through G protein-coupled receptor 43 (GPR43), a pro-resolutive metabolic axis mediated by butyrate via histone deacetylase (HDAC) inhibition and activation of GPR109A, and a direct neural sensing axis triggered by propionate through olfactory receptor 78 (OLFR78). Chronic pain, therefore, does not arise simply from the presence or absence of SCFAs, but from the pathological dominance of one of these axes shaped by specific dysbiosis profiles. This framework moves beyond correlation by providing a mechanistic basis for precision interventions designed to rebalance SCFA signalling, offering novel therapeutic opportunities for neuropathic and inflammatory pain conditions.
DOI: https://doi.org/10.37349/ent.2026.1004151

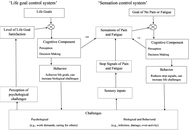
Explanations and treatment of fibromyalgia syndrome (FMS) and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) are controversial, and outcomes are poor. This paper describes sensation-suppression theory, a theory modelled on self-organizing control systems that are capable of adaptation in response to inputs and used in applications of artificial intelligence. The theory shows how the need to suppress inflammatory and other causes of pain or fatigue due to challenging circumstances sensitizes the neurological processing of pain and fatigue, thereby creating the amplified sensations and abnormal cognitions of central sensitivity syndromes. These syndromes are caused by errors in an evolutionarily early behavior-control mechanism of animals that comes into conflict with the later cognitive behavior-control mechanism of humans. Unlike the cognitive and current biological theories, the sensation-suppression theory explains both the personality and biological risk factors for central sensitivity syndromes and why onset is sometimes gradual and sometimes sudden. A specific form of autonomic dysregulation that could act as a new empirical test of the theory is suggested. Recovery is achieved by reversing the biological homeostatic dysregulation through a specific form of pacing where the person changes from one short, non-stressful activity to another, and where activity is calibrated to the level of illness and the patient’s current biological state. Recovery is hampered or prevented by systemic inflammation and lifestyle obligations. The theory provides a sympathetic narrative for the cause and treatment of FMS and ME/CFS and promotes a recovery lifestyle that prioritizes the needs of the patient. Prevention requires hearing what the body is saying.
Explanations and treatment of fibromyalgia syndrome (FMS) and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) are controversial, and outcomes are poor. This paper describes sensation-suppression theory, a theory modelled on self-organizing control systems that are capable of adaptation in response to inputs and used in applications of artificial intelligence. The theory shows how the need to suppress inflammatory and other causes of pain or fatigue due to challenging circumstances sensitizes the neurological processing of pain and fatigue, thereby creating the amplified sensations and abnormal cognitions of central sensitivity syndromes. These syndromes are caused by errors in an evolutionarily early behavior-control mechanism of animals that comes into conflict with the later cognitive behavior-control mechanism of humans. Unlike the cognitive and current biological theories, the sensation-suppression theory explains both the personality and biological risk factors for central sensitivity syndromes and why onset is sometimes gradual and sometimes sudden. A specific form of autonomic dysregulation that could act as a new empirical test of the theory is suggested. Recovery is achieved by reversing the biological homeostatic dysregulation through a specific form of pacing where the person changes from one short, non-stressful activity to another, and where activity is calibrated to the level of illness and the patient’s current biological state. Recovery is hampered or prevented by systemic inflammation and lifestyle obligations. The theory provides a sympathetic narrative for the cause and treatment of FMS and ME/CFS and promotes a recovery lifestyle that prioritizes the needs of the patient. Prevention requires hearing what the body is saying.
DOI: https://doi.org/10.37349/ent.2026.1004152

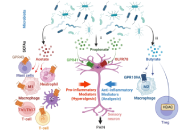
Growing evidence has directly linked the gastrointestinal tract, gut microbiota, and central nervous system, forming the gut-brain axis, a process that has been described as a key mechanism in regulating neurological processes. However, the presence of alterations in the composition of microorganisms in the digestive tract and dysbiosis has been linked to the activation of microglia, increased oxidative stress, alterations in the production of neurotransmitters, and exacerbation of neuroinflammation. These mechanisms have been associated with multiple pathologies and neurological conditions, and regulating them is key to the control of these diseases. In this context, various bacterial species play a neuroprotective role by promoting the integrity of the intestinal barrier, stimulating the synthesis of beneficial metabolites such as short-chain fatty acids (SCFAs), neurotransmitters, and modulating the inflammatory response. In addition, the characterization of these microbial profiles provides a broad perspective on understanding how changes in the microbiota contribute to the progression of neurological diseases. On the other hand, these new updates open up the possibility of designing personalised targeted therapeutic interventions that can regulate the gut microbiota and promote a neuroprotective and neuroregenerative environment. Another key point is that greater emphasis is placed on the need for more controlled clinical studies to validate efficacy and safety in humans, as well as knowledge of the mechanisms of action that make them possible. Finally, the modulation of the gut microbiota using probiotics, prebiotics, and postbiotics represents an innovative and effective opportunity to intervene in neuroimmune processes such as microglial activation, regulation of synaptic pruning, and neuroinflammatory pathways—processes implicated in various neurological diseases. In this context, this review integrates and analyzes the available evidence, highlighting potential interventions as treatments for these pathologies, as well as current limitations, to provide an updated framework to guide future research.
Growing evidence has directly linked the gastrointestinal tract, gut microbiota, and central nervous system, forming the gut-brain axis, a process that has been described as a key mechanism in regulating neurological processes. However, the presence of alterations in the composition of microorganisms in the digestive tract and dysbiosis has been linked to the activation of microglia, increased oxidative stress, alterations in the production of neurotransmitters, and exacerbation of neuroinflammation. These mechanisms have been associated with multiple pathologies and neurological conditions, and regulating them is key to the control of these diseases. In this context, various bacterial species play a neuroprotective role by promoting the integrity of the intestinal barrier, stimulating the synthesis of beneficial metabolites such as short-chain fatty acids (SCFAs), neurotransmitters, and modulating the inflammatory response. In addition, the characterization of these microbial profiles provides a broad perspective on understanding how changes in the microbiota contribute to the progression of neurological diseases. On the other hand, these new updates open up the possibility of designing personalised targeted therapeutic interventions that can regulate the gut microbiota and promote a neuroprotective and neuroregenerative environment. Another key point is that greater emphasis is placed on the need for more controlled clinical studies to validate efficacy and safety in humans, as well as knowledge of the mechanisms of action that make them possible. Finally, the modulation of the gut microbiota using probiotics, prebiotics, and postbiotics represents an innovative and effective opportunity to intervene in neuroimmune processes such as microglial activation, regulation of synaptic pruning, and neuroinflammatory pathways—processes implicated in various neurological diseases. In this context, this review integrates and analyzes the available evidence, highlighting potential interventions as treatments for these pathologies, as well as current limitations, to provide an updated framework to guide future research.
DOI: https://doi.org/10.37349/ent.2026.1004150
This article belongs to the special issue Role of Microbiota in Neurological Diseases

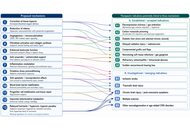
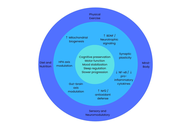
Neurodegenerative disorders represent a major and growing global health challenge, characterized by progressive neuronal loss, multifactorial pathophysiology, and limited disease-modifying pharmacological options. Increasing attention has therefore been directed toward non-pharmacological and integrative interventions as complementary strategies for neuroprotection and symptom management. These approaches target key mechanisms implicated in neurodegeneration, including oxidative stress, neuroinflammation, mitochondrial dysfunction, synaptic impairment, and dysregulated neuroplasticity. This narrative integrative review synthesizes current preclinical and clinical evidence on non-pharmacological interventions with demonstrated or emerging neuroprotective potential across major neurodegenerative disorders, including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and related conditions. The review focuses on four principal domains: physical activity and exercise, nutritional and dietary strategies, mind–body and psychosocial interventions, and sensory or neuromodulatory therapies. Collectively, these interventions influence convergent neurobiological pathways, including neurotrophic signaling, immune modulation, autonomic regulation, and gut–brain communication. Studies indicate that structured physical exercise enhances neurotrophic factor expression and mitochondrial resilience; dietary patterns rich in antioxidants and anti-inflammatory components mitigate oxidative damage and neuroinflammation; mind–body practices modulate stress-related neuroendocrine pathways and promote functional connectivity; and sensory or neuromodulatory interventions engage limbic and cortical networks relevant to cognition, mood, and motor control. Importantly, multimodal and integrative approaches appear to exert synergistic effects, aligning with the complex and systemic nature of neurodegenerative processes. Despite promising findings, challenges related to methodological heterogeneity, biomarker validation, and translational implementation persist. Future research should prioritize standardized protocols, objective neuroprotective endpoints, and personalized intervention frameworks supported by digital health technologies. Overall, non-pharmacological and integrative therapies represent a valuable, increasingly evidence-based component of comprehensive neuroprotective strategies, with significant potential to enhance quality of life and complement pharmacological treatments in the care of neurodegenerative diseases.
Neurodegenerative disorders represent a major and growing global health challenge, characterized by progressive neuronal loss, multifactorial pathophysiology, and limited disease-modifying pharmacological options. Increasing attention has therefore been directed toward non-pharmacological and integrative interventions as complementary strategies for neuroprotection and symptom management. These approaches target key mechanisms implicated in neurodegeneration, including oxidative stress, neuroinflammation, mitochondrial dysfunction, synaptic impairment, and dysregulated neuroplasticity. This narrative integrative review synthesizes current preclinical and clinical evidence on non-pharmacological interventions with demonstrated or emerging neuroprotective potential across major neurodegenerative disorders, including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and related conditions. The review focuses on four principal domains: physical activity and exercise, nutritional and dietary strategies, mind–body and psychosocial interventions, and sensory or neuromodulatory therapies. Collectively, these interventions influence convergent neurobiological pathways, including neurotrophic signaling, immune modulation, autonomic regulation, and gut–brain communication. Studies indicate that structured physical exercise enhances neurotrophic factor expression and mitochondrial resilience; dietary patterns rich in antioxidants and anti-inflammatory components mitigate oxidative damage and neuroinflammation; mind–body practices modulate stress-related neuroendocrine pathways and promote functional connectivity; and sensory or neuromodulatory interventions engage limbic and cortical networks relevant to cognition, mood, and motor control. Importantly, multimodal and integrative approaches appear to exert synergistic effects, aligning with the complex and systemic nature of neurodegenerative processes. Despite promising findings, challenges related to methodological heterogeneity, biomarker validation, and translational implementation persist. Future research should prioritize standardized protocols, objective neuroprotective endpoints, and personalized intervention frameworks supported by digital health technologies. Overall, non-pharmacological and integrative therapies represent a valuable, increasingly evidence-based component of comprehensive neuroprotective strategies, with significant potential to enhance quality of life and complement pharmacological treatments in the care of neurodegenerative diseases.
DOI: https://doi.org/10.37349/ent.2026.1004149
This article belongs to the special issue Breakthroughs in Mechanisms and Treatments for Neurodegenerative Diseases

Background:
Atrial fibrillation (AF) substantially increases the risk of ischemic stroke (IS), underscoring the need for effective anticoagulation strategies. Direct oral anticoagulants (DOACs) have largely supplanted vitamin K antagonists (VKAs) due to their favorable safety profile and ease of use. Factor XI (FXI) inhibitors, which target the intrinsic coagulation pathway, are emerging as potential alternatives that may offer reduced bleeding risk. This systematic review evaluates the efficacy and safety of FXI inhibitors compared with DOACs for stroke prevention in AF.
Methods:
A total of 20 studies fulfilled the inclusion criteria, comprising 11 randomized controlled trials (RCTs), five systematic reviews or meta-analyses, and four narrative, cohort, or modeling studies. Eligible investigations compared FXI inhibitors with DOACs in patients diagnosed with AF. The primary outcomes assessed were stroke or systemic embolism, major bleeding, and all-cause mortality. Methodological quality was evaluated according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) framework, the revised Cochrane Risk of Bias 2 (RoB 2) tool for RCTs, and the Newcastle-Ottawa Scale (NOS).
Results:
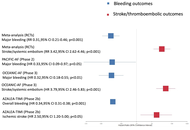
FXI inhibitors were associated with a significant reduction in major bleeding [relative risk (RR) 0.31; 95% confidence interval (CI) 0.21–0.46] and clinically relevant non-major bleeding (RR 0.66; 95% CI 0.47–0.93) compared with DOACs. Conversely, FXI inhibitors demonstrated an increased risk of stroke or systemic embolism (RR 3.17; 95% CI 2.18–4.62), as observed in the OCEANIC-AF trial [hazard ratio (HR) 3.79; 95% CI 2.46–5.83]. No significant difference was noted in all-cause mortality (RR 0.85; 95% CI 0.67–1.08). Limited evidence suggests that FXI inhibitors may also reduce bleeding-related hospitalizations.
Discussion:
FXI inhibitors provide a favorable bleeding profile but are less effective than DOACs for stroke prevention in patients with AF. Further long-term RCTs are warranted to delineate their role, particularly in populations at high risk of bleeding.
Background:
Atrial fibrillation (AF) substantially increases the risk of ischemic stroke (IS), underscoring the need for effective anticoagulation strategies. Direct oral anticoagulants (DOACs) have largely supplanted vitamin K antagonists (VKAs) due to their favorable safety profile and ease of use. Factor XI (FXI) inhibitors, which target the intrinsic coagulation pathway, are emerging as potential alternatives that may offer reduced bleeding risk. This systematic review evaluates the efficacy and safety of FXI inhibitors compared with DOACs for stroke prevention in AF.
Methods:
A total of 20 studies fulfilled the inclusion criteria, comprising 11 randomized controlled trials (RCTs), five systematic reviews or meta-analyses, and four narrative, cohort, or modeling studies. Eligible investigations compared FXI inhibitors with DOACs in patients diagnosed with AF. The primary outcomes assessed were stroke or systemic embolism, major bleeding, and all-cause mortality. Methodological quality was evaluated according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) framework, the revised Cochrane Risk of Bias 2 (RoB 2) tool for RCTs, and the Newcastle-Ottawa Scale (NOS).
Results:
FXI inhibitors were associated with a significant reduction in major bleeding [relative risk (RR) 0.31; 95% confidence interval (CI) 0.21–0.46] and clinically relevant non-major bleeding (RR 0.66; 95% CI 0.47–0.93) compared with DOACs. Conversely, FXI inhibitors demonstrated an increased risk of stroke or systemic embolism (RR 3.17; 95% CI 2.18–4.62), as observed in the OCEANIC-AF trial [hazard ratio (HR) 3.79; 95% CI 2.46–5.83]. No significant difference was noted in all-cause mortality (RR 0.85; 95% CI 0.67–1.08). Limited evidence suggests that FXI inhibitors may also reduce bleeding-related hospitalizations.
Discussion:
FXI inhibitors provide a favorable bleeding profile but are less effective than DOACs for stroke prevention in patients with AF. Further long-term RCTs are warranted to delineate their role, particularly in populations at high risk of bleeding.
DOI: https://doi.org/10.37349/ent.2026.1004148
This article belongs to the special issue Interdisciplinary Approach to Therapeutic Strategies of Neuroprotection in Present and Future

Background:
Multiple sclerosis (MS) is a chronic, autoimmune, inflammatory disease that affects the central nervous system. Although the true etiology of MS remains unknown, recent research suggests that it arises from a combination of genetic vulnerability and environmental factors. The human leukocyte antigen (HLA) region is a highly polymorphic locus on chromosome 6 encoding antigen-presenting molecules central to adaptive immunity. MS exhibits significant genetic and geographic heterogeneity, reflecting complex interactions between HLA polymorphisms and environmental influences. Risk and protective alleles differ across populations, reinforcing the importance of studying such variations to better understand the disease’s pathogenesis and guide therapeutic strategies.
Methods:
This systematic review followed the “Preferred Reporting Items for Systematic reviews and Meta-Analyses” (PRISMA) guidelines, and a bibliographic search was conducted in the Medline (PubMed) and Web of Science databases using the keywords “Multiple Sclerosis”, “Genetic Polymorphisms”, “SNPs”, and “Human Leukocyte Antigen”.
Results:
Twenty-one studies were included, comprising a total of over 50,000 participants across diverse populations. The reviewed studies demonstrate that the alleles DRB1*15:01, DQB1*06:02, DRB1*03:01, DRB1*04:01, DRB1*15:03, DPB1*03:01, as well as the haplotypes DRB1*15:01~DQB1*06:02 and DRB1*15:01~DQA1*01:02~DQB1*06:02, show high expression and are strongly associated with MS susceptibility. In contrast, the alleles A*02:01 and DRB1*01:01 have shown a protective role.
Discussion:
The evidence confirms a central role of HLA class II alleles and conserved extended haplotypes, particularly DRB1*15:01-containing haplotypes, in MS susceptibility, while highlighting protective alleles and marked variability across ancestral backgrounds. These findings underscore the importance of high-resolution HLA typing, standardized haplotype definitions and inclusion of diverse populations to refine MS risk estimates.
Background:
Multiple sclerosis (MS) is a chronic, autoimmune, inflammatory disease that affects the central nervous system. Although the true etiology of MS remains unknown, recent research suggests that it arises from a combination of genetic vulnerability and environmental factors. The human leukocyte antigen (HLA) region is a highly polymorphic locus on chromosome 6 encoding antigen-presenting molecules central to adaptive immunity. MS exhibits significant genetic and geographic heterogeneity, reflecting complex interactions between HLA polymorphisms and environmental influences. Risk and protective alleles differ across populations, reinforcing the importance of studying such variations to better understand the disease’s pathogenesis and guide therapeutic strategies.
Methods:
This systematic review followed the “Preferred Reporting Items for Systematic reviews and Meta-Analyses” (PRISMA) guidelines, and a bibliographic search was conducted in the Medline (PubMed) and Web of Science databases using the keywords “Multiple Sclerosis”, “Genetic Polymorphisms”, “SNPs”, and “Human Leukocyte Antigen”.
Results:
Twenty-one studies were included, comprising a total of over 50,000 participants across diverse populations. The reviewed studies demonstrate that the alleles DRB1*15:01, DQB1*06:02, DRB1*03:01, DRB1*04:01, DRB1*15:03, DPB1*03:01, as well as the haplotypes DRB1*15:01~DQB1*06:02 and DRB1*15:01~DQA1*01:02~DQB1*06:02, show high expression and are strongly associated with MS susceptibility. In contrast, the alleles A*02:01 and DRB1*01:01 have shown a protective role.
Discussion:
The evidence confirms a central role of HLA class II alleles and conserved extended haplotypes, particularly DRB1*15:01-containing haplotypes, in MS susceptibility, while highlighting protective alleles and marked variability across ancestral backgrounds. These findings underscore the importance of high-resolution HLA typing, standardized haplotype definitions and inclusion of diverse populations to refine MS risk estimates.
DOI: https://doi.org/10.37349/ent.2026.1004147

The relationship between the gut microbiota and the central nervous system has gained attention as a biological axis that may influence the development of several neurological disorders. Recent evidence integrating genomic, neurobiological, and microbiota research shows how bacterial composition, host genetic variability, and the mechanisms of the microbiota-gut-brain axis interact in conditions such as autism spectrum disorder, epilepsy, and schizophrenia. These interactions function through neural, metabolic, and immunological related pathways involving intestinal and blood-brain barrier permeability. Genome-wide association studies (GWAS) and Mendelian randomization analyses highlight shared immunogenetic pathways that may shape both microbial profiles and neurological susceptibility. Consistent patterns of dysbiosis and alterations in neuroactive metabolites have also been reported, linking microbiota changes to neuroinflammation and disrupted neuronal signaling. This review synthesizes the current evidence supporting the integration of the microbiota-gut-brain axis and its underlying communication pathways. It also outlines the present therapeutic strategies for neurological disorders such as autism spectrum disorder, epilepsy, and schizophrenia, highlighting their potential to modulate neurological function. Additionally, it discusses the existing limitations in the field and offers insights into future research directions within this rapidly evolving area.
The relationship between the gut microbiota and the central nervous system has gained attention as a biological axis that may influence the development of several neurological disorders. Recent evidence integrating genomic, neurobiological, and microbiota research shows how bacterial composition, host genetic variability, and the mechanisms of the microbiota-gut-brain axis interact in conditions such as autism spectrum disorder, epilepsy, and schizophrenia. These interactions function through neural, metabolic, and immunological related pathways involving intestinal and blood-brain barrier permeability. Genome-wide association studies (GWAS) and Mendelian randomization analyses highlight shared immunogenetic pathways that may shape both microbial profiles and neurological susceptibility. Consistent patterns of dysbiosis and alterations in neuroactive metabolites have also been reported, linking microbiota changes to neuroinflammation and disrupted neuronal signaling. This review synthesizes the current evidence supporting the integration of the microbiota-gut-brain axis and its underlying communication pathways. It also outlines the present therapeutic strategies for neurological disorders such as autism spectrum disorder, epilepsy, and schizophrenia, highlighting their potential to modulate neurological function. Additionally, it discusses the existing limitations in the field and offers insights into future research directions within this rapidly evolving area.
DOI: https://doi.org/10.37349/ent.2026.1004146
This article belongs to the special issue Role of Microbiota in Neurological Diseases

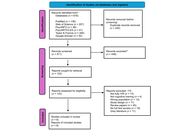
Background:
Acquired brain injury (ABI) often causes long-lasting impairments in written language and handwriting that limit autonomy and daily functioning. Despite their relevance, these deficits have received limited research attention compared with spoken language disorders. The present work aims to systematically review interventions designed to improve reading, writing, and handwriting abilities in individuals with ABI.
Methods:
Following PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines, PubMed, American Psychological Association (APA) PsycINFO, Cochrane Library, Web of Science, and Google Scholar were searched from inception to 1 November 2025. Eligible studies were randomized or non-randomized clinical trials (non-RCTs) involving adults or adolescents with ABI and documented written language impairments. Risk of bias was assessed using the Risk of Bias 2 tool (RCTs) and Risk of Bias in Non-randomized Studies—of Interventions tool (non-RCTs).
Results:
Twelve studies met inclusion criteria (2 RCTs, 10 non-RCTs), all conducted in post-stroke populations, highlighting the absence of evidence from other ABI aetiologies. Three main intervention categories emerged: (1) Behavioral treatments, which consistently improved trained spelling and functional writing, with some advantages for errorless learning in maintaining gains. (2) Technology-assisted approaches, including assistive software, digital spelling aids, and handwriting-focused programs, which showed feasibility, high usability, and improvements in accuracy, legibility, and motor fluency. (3) Neuromodulation, with one RCT showing that dual-site transcranial direct current stimulation can modestly enhance behavioral writing therapy. Most non-RCTs showed serious or critical risk of bias, and sample sizes were small, limiting generalizability.
Discussion:
Current evidence—although preliminary and restricted to post-stroke ABI—indicates that behavioral, technological, and neuromodulatory interventions can improve aspects of written language and handwriting after ABI. However, the available literature is characterized by small samples, substantial methodological variability, and a paucity of standardized and ecologically valid outcome measures. High-quality, adequately powered trials with standardized, functional outcomes are urgently needed, particularly in non-stroke ABI populations.
Background:
Acquired brain injury (ABI) often causes long-lasting impairments in written language and handwriting that limit autonomy and daily functioning. Despite their relevance, these deficits have received limited research attention compared with spoken language disorders. The present work aims to systematically review interventions designed to improve reading, writing, and handwriting abilities in individuals with ABI.
Methods:
Following PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines, PubMed, American Psychological Association (APA) PsycINFO, Cochrane Library, Web of Science, and Google Scholar were searched from inception to 1 November 2025. Eligible studies were randomized or non-randomized clinical trials (non-RCTs) involving adults or adolescents with ABI and documented written language impairments. Risk of bias was assessed using the Risk of Bias 2 tool (RCTs) and Risk of Bias in Non-randomized Studies—of Interventions tool (non-RCTs).
Results:
Twelve studies met inclusion criteria (2 RCTs, 10 non-RCTs), all conducted in post-stroke populations, highlighting the absence of evidence from other ABI aetiologies. Three main intervention categories emerged: (1) Behavioral treatments, which consistently improved trained spelling and functional writing, with some advantages for errorless learning in maintaining gains. (2) Technology-assisted approaches, including assistive software, digital spelling aids, and handwriting-focused programs, which showed feasibility, high usability, and improvements in accuracy, legibility, and motor fluency. (3) Neuromodulation, with one RCT showing that dual-site transcranial direct current stimulation can modestly enhance behavioral writing therapy. Most non-RCTs showed serious or critical risk of bias, and sample sizes were small, limiting generalizability.
Discussion:
Current evidence—although preliminary and restricted to post-stroke ABI—indicates that behavioral, technological, and neuromodulatory interventions can improve aspects of written language and handwriting after ABI. However, the available literature is characterized by small samples, substantial methodological variability, and a paucity of standardized and ecologically valid outcome measures. High-quality, adequately powered trials with standardized, functional outcomes are urgently needed, particularly in non-stroke ABI populations.
DOI: https://doi.org/10.37349/ent.2026.1004145
This article belongs to the special issue Neuropsychology of Handwriting

Background:
Fully immersive virtual reality (IVR) is an emerging technology approach for cognitive training in individuals with mild cognitive impairment (MCI) and dementia. While interest in fully IVR continues to grow, it remains unclear the extent of effectiveness and the key components that contribute to successful implementation. This study aimed to explore the effectiveness of fully IVR cognitive training for individuals with MCI or dementia from previous research literature.
Methods:
A scoping review was conducted using a systematic search strategy based on the population, concept, and context framework.
Results:
Out of the 816 records identified, 123 full texts were screened, and eight studies were included in the review. The included studies all involved participants completing a cognitive training intervention using fully IVR headsets, with cognitive outcomes measured before and after the intervention. The most consistent improvements across the included studies were executive function, memory, and visuospatial abilities. Only two studies explicitly referenced a theoretical model.
Discussion:
Fully IVR cognitive training demonstrates promise for improving specific cognitive domains in individuals living with MCI or dementia. However, inconsistencies in outcomes and limited theoretical grounding highlight the need for further exploration. Broader considerations are discussed in the discussion section.
Background:
Fully immersive virtual reality (IVR) is an emerging technology approach for cognitive training in individuals with mild cognitive impairment (MCI) and dementia. While interest in fully IVR continues to grow, it remains unclear the extent of effectiveness and the key components that contribute to successful implementation. This study aimed to explore the effectiveness of fully IVR cognitive training for individuals with MCI or dementia from previous research literature.
Methods:
A scoping review was conducted using a systematic search strategy based on the population, concept, and context framework.
Results:
Out of the 816 records identified, 123 full texts were screened, and eight studies were included in the review. The included studies all involved participants completing a cognitive training intervention using fully IVR headsets, with cognitive outcomes measured before and after the intervention. The most consistent improvements across the included studies were executive function, memory, and visuospatial abilities. Only two studies explicitly referenced a theoretical model.
Discussion:
Fully IVR cognitive training demonstrates promise for improving specific cognitive domains in individuals living with MCI or dementia. However, inconsistencies in outcomes and limited theoretical grounding highlight the need for further exploration. Broader considerations are discussed in the discussion section.
DOI: https://doi.org/10.37349/ent.2026.1004144
This article belongs to the special issue Innovations in Neurotechnology: Integrating AI, Neuroimaging, and VR for the Assessment and Treatment of Neurological Conditions

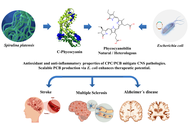
Phycocyanobilin (PCB), the covalently bound chromophore of the cyanobacterial protein C-phycocyanin (CPC), is recognized as a bioactive molecule with neuroprotective and anti-inflammatory properties. PCB and CPC, frequently coexisting in Spirulina extracts or experimental formulations, have demonstrated beneficial effects in preclinical models of multiple sclerosis, ischemic stroke, and Alzheimer’s disease. Reported mechanisms include attenuation of oxidative stress, reduction of neuroinflammation, and preservation of mitochondrial function, thereby contributing to a reparative microenvironment within the central nervous system. PCB can be obtained through two complementary approaches: Extraction from cyanobacterial biomass, where it remains covalently bound to CPC, and heterologous biosynthesis in Escherichia coli (E. coli), which enables production of free PCB as a high-purity, scalable linear tetrapyrrole suitable for translational applications. This mini-review summarizes current evidence on the neuroprotective actions of PCB and CPC, highlights their molecular targets, and discusses biotechnological advances that support their potential role in remyelination. By bridging natural pigment pharmacology with recombinant production strategies, PCB is positioned as a multitarget candidate of growing interest for the development of future neuroprotective and neurorepair therapies.
Phycocyanobilin (PCB), the covalently bound chromophore of the cyanobacterial protein C-phycocyanin (CPC), is recognized as a bioactive molecule with neuroprotective and anti-inflammatory properties. PCB and CPC, frequently coexisting in Spirulina extracts or experimental formulations, have demonstrated beneficial effects in preclinical models of multiple sclerosis, ischemic stroke, and Alzheimer’s disease. Reported mechanisms include attenuation of oxidative stress, reduction of neuroinflammation, and preservation of mitochondrial function, thereby contributing to a reparative microenvironment within the central nervous system. PCB can be obtained through two complementary approaches: Extraction from cyanobacterial biomass, where it remains covalently bound to CPC, and heterologous biosynthesis in Escherichia coli (E. coli), which enables production of free PCB as a high-purity, scalable linear tetrapyrrole suitable for translational applications. This mini-review summarizes current evidence on the neuroprotective actions of PCB and CPC, highlights their molecular targets, and discusses biotechnological advances that support their potential role in remyelination. By bridging natural pigment pharmacology with recombinant production strategies, PCB is positioned as a multitarget candidate of growing interest for the development of future neuroprotective and neurorepair therapies.
DOI: https://doi.org/10.37349/ent.2026.1004143
This article belongs to the special issue Natural Products in Neurotherapeutic Applications

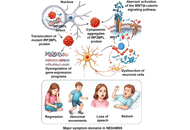
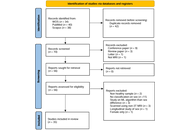
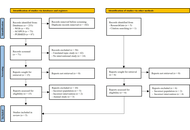
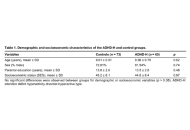
Neurodevelopmental Disorder with Regression, Abnormal Movements, Loss of Speech, and Seizures (NEDAMSS) is an ultra-rare, progressive neurological disorder, with more than 60 individuals described in the medical literature. It is caused by de novo mutations in the interferon regulatory factor 2 binding protein-like (IRF2BPL) gene, leading to early-onset symptoms including seizures, developmental delays, intellectual disability, and other severe neurological impairments, typically beginning in infancy or early childhood. This review aims to consolidate and refine current knowledge on NEDAMSS, focusing on the molecular functions of IRF2BPL, the spectrum of clinical features, and underlying disease mechanisms. A comprehensive understanding of NEDAMSS is essential for guiding the development of targeted interventions and therapeutic strategies to improve patient outcomes. By integrating current findings, we focus both on the progress made and the gaps that remain in research, providing a foundation for future studies to advance diagnosis, treatment, and overall patient care. We reviewed the published literature through studies available up to 2025 to synthesize current knowledge on clinical features, genetics, and proposed disease mechanisms. Reported phenotypes show substantial heterogeneity, and current genotype-phenotype correlations remain limited by small cohorts and inconsistent reporting. Key next steps include standardized phenotyping, natural history studies, and biomarker development to enable trial-ready outcome measures and accelerate targeted therapy development.
Neurodevelopmental Disorder with Regression, Abnormal Movements, Loss of Speech, and Seizures (NEDAMSS) is an ultra-rare, progressive neurological disorder, with more than 60 individuals described in the medical literature. It is caused by de novo mutations in the interferon regulatory factor 2 binding protein-like (IRF2BPL) gene, leading to early-onset symptoms including seizures, developmental delays, intellectual disability, and other severe neurological impairments, typically beginning in infancy or early childhood. This review aims to consolidate and refine current knowledge on NEDAMSS, focusing on the molecular functions of IRF2BPL, the spectrum of clinical features, and underlying disease mechanisms. A comprehensive understanding of NEDAMSS is essential for guiding the development of targeted interventions and therapeutic strategies to improve patient outcomes. By integrating current findings, we focus both on the progress made and the gaps that remain in research, providing a foundation for future studies to advance diagnosis, treatment, and overall patient care. We reviewed the published literature through studies available up to 2025 to synthesize current knowledge on clinical features, genetics, and proposed disease mechanisms. Reported phenotypes show substantial heterogeneity, and current genotype-phenotype correlations remain limited by small cohorts and inconsistent reporting. Key next steps include standardized phenotyping, natural history studies, and biomarker development to enable trial-ready outcome measures and accelerate targeted therapy development.
DOI: https://doi.org/10.37349/ent.2026.1004142

Background:
This systematic review critically evaluates whether machine learning (ML) identifies biologically meaningful sex-related brain architecture or merely exploits methodological artifacts and allometric scaling. While ML models achieve high classification accuracies, it remains unclear if these reflect stable, mechanistically informative dimorphism or are driven by confounds such as total intracranial volume (TIV) and site-specific noise. We examine how imaging modalities, algorithms, and population strata influence both classification outcomes and biological interpretability.
Methods:
Following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, we searched Web of Science, PubMed, and Scopus through January 2024. Included studies [healthy humans, 3T magnetic resonance imaging (MRI), ML-based sex classification] were assessed for risk of bias, focusing on data leakage, validation strategies, and confound management.
Results:
Thirty-five studies (n > 110,000) were included. While reported accuracies reached 98.06% for T1-weighted MRI, 96.0% for diffusion MRI (dMRI), and 94.72% for functional MRI (fMRI), performance was highly dependent on population characterization and age. Deep learning consistently outperformed traditional ML (TML) but showed high sensitivity to methodological artifacts. Notably, studies failing to correct for TIV reported potentially inflated accuracies, suggesting that many models identify physical scale rather than intrinsic neuroanatomical dimorphism.
Discussion:
High classification accuracies are often bolstered by methodological confounds and a lack of cross-site validation. There is a significant discrepancy between ML-driven predictive power and biological inference validity. Current pipelines do not yet allow for robust, generalizable inference about brain sex. To move beyond statistical separation toward mechanistic understanding, the field must prioritize TIV-corrected benchmarks and diverse non-WEIRD (Western, Educated, Industrialized, Rich, Democratic) datasets. We conclude that while ML is a powerful pattern detector, its results must be interpreted with caution regarding biological dimorphism.
Background:
This systematic review critically evaluates whether machine learning (ML) identifies biologically meaningful sex-related brain architecture or merely exploits methodological artifacts and allometric scaling. While ML models achieve high classification accuracies, it remains unclear if these reflect stable, mechanistically informative dimorphism or are driven by confounds such as total intracranial volume (TIV) and site-specific noise. We examine how imaging modalities, algorithms, and population strata influence both classification outcomes and biological interpretability.
Methods:
Following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, we searched Web of Science, PubMed, and Scopus through January 2024. Included studies [healthy humans, 3T magnetic resonance imaging (MRI), ML-based sex classification] were assessed for risk of bias, focusing on data leakage, validation strategies, and confound management.
Results:
Thirty-five studies (n > 110,000) were included. While reported accuracies reached 98.06% for T1-weighted MRI, 96.0% for diffusion MRI (dMRI), and 94.72% for functional MRI (fMRI), performance was highly dependent on population characterization and age. Deep learning consistently outperformed traditional ML (TML) but showed high sensitivity to methodological artifacts. Notably, studies failing to correct for TIV reported potentially inflated accuracies, suggesting that many models identify physical scale rather than intrinsic neuroanatomical dimorphism.
Discussion:
High classification accuracies are often bolstered by methodological confounds and a lack of cross-site validation. There is a significant discrepancy between ML-driven predictive power and biological inference validity. Current pipelines do not yet allow for robust, generalizable inference about brain sex. To move beyond statistical separation toward mechanistic understanding, the field must prioritize TIV-corrected benchmarks and diverse non-WEIRD (Western, Educated, Industrialized, Rich, Democratic) datasets. We conclude that while ML is a powerful pattern detector, its results must be interpreted with caution regarding biological dimorphism.
DOI: https://doi.org/10.37349/ent.2026.1004141

Background:
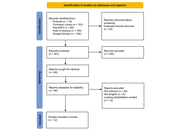
Alzheimer’s disease (AD) is a prevalent neurodegenerative disorder in older adults, and growing evidence suggests that alterations in the gut-brain axis may contribute to its pathophysiology. Probiotics have been proposed as a potential adjunctive strategy to modulate gut microbiota and related systemic pathways; however, clinical evidence in AD remains limited and heterogeneous. This systematic review evaluated current evidence on the effects of probiotic supplementation on cognitive outcomes and health-related biomarkers in patients with AD.
Methods:
Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, PubMed, Web of Science, and Scopus were systematically searched for double-blind randomized controlled trials published within the last 10 years (up to November 30, 2025). Studies assessing probiotic supplementation in clinically diagnosed AD patients were included. Methodological quality and risk of bias were evaluated using the Physiotherapy Evidence Database (PEDro) scale, the McMaster assessment tool, and the Cochrane Risk of Bias 2.0 tool.
Results:
Of 253 records identified, five randomized controlled trials met the inclusion criteria, comprising 328 participants. The studies evaluated different probiotic formulations, mainly involving Lactobacillus and Bifidobacterium species, administered over short-term interventions (12 weeks). Some trials reported statistically significant improvements in selected cognitive outcomes, inflammatory and oxidative stress markers, metabolic parameters, brain-derived neurotrophic factor levels, anxiety, and instrumental activities of daily living. However, results were heterogeneous and several outcomes were non-significant. No serious adverse events related to probiotic supplementation were reported.
Discussion:
Probiotic supplementation may be associated with strain-specific and short-term effects on selected cognitive and biological outcomes in patients with AD. Nevertheless, limited trial numbers, small sample sizes, and substantial heterogeneity preclude definitive conclusions. Further well-designed randomized controlled trials with standardized probiotic formulations and longer follow-up periods are required.
Background:
Alzheimer’s disease (AD) is a prevalent neurodegenerative disorder in older adults, and growing evidence suggests that alterations in the gut-brain axis may contribute to its pathophysiology. Probiotics have been proposed as a potential adjunctive strategy to modulate gut microbiota and related systemic pathways; however, clinical evidence in AD remains limited and heterogeneous. This systematic review evaluated current evidence on the effects of probiotic supplementation on cognitive outcomes and health-related biomarkers in patients with AD.
Methods:
Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, PubMed, Web of Science, and Scopus were systematically searched for double-blind randomized controlled trials published within the last 10 years (up to November 30, 2025). Studies assessing probiotic supplementation in clinically diagnosed AD patients were included. Methodological quality and risk of bias were evaluated using the Physiotherapy Evidence Database (PEDro) scale, the McMaster assessment tool, and the Cochrane Risk of Bias 2.0 tool.
Results:
Of 253 records identified, five randomized controlled trials met the inclusion criteria, comprising 328 participants. The studies evaluated different probiotic formulations, mainly involving Lactobacillus and Bifidobacterium species, administered over short-term interventions (12 weeks). Some trials reported statistically significant improvements in selected cognitive outcomes, inflammatory and oxidative stress markers, metabolic parameters, brain-derived neurotrophic factor levels, anxiety, and instrumental activities of daily living. However, results were heterogeneous and several outcomes were non-significant. No serious adverse events related to probiotic supplementation were reported.
Discussion:
Probiotic supplementation may be associated with strain-specific and short-term effects on selected cognitive and biological outcomes in patients with AD. Nevertheless, limited trial numbers, small sample sizes, and substantial heterogeneity preclude definitive conclusions. Further well-designed randomized controlled trials with standardized probiotic formulations and longer follow-up periods are required.
DOI: https://doi.org/10.37349/ent.2026.1004140
This article belongs to the special issue Role of Microbiota in Neurological Diseases

Aim:
Attention is a core cognitive function that supports higher-order processes such as reasoning, problem solving, and intelligence. In children with attention deficit hyperactivity disorder (ADHD), particularly the hyperactive subtype, impairments in attentional control may interfere with the development and expression of cognitive abilities. This study examined the relationship between visuospatial attention and both verbal and nonverbal intelligence in children with ADHD-hyperactive type (ADHD-H).
Methods:
A sample of 65 children with ADHD-H and 73 typically developing controls (aged 8–10 years) completed three tasks: the Benton Visual Form Discrimination Test (VFDT), assessing complex visuospatial attention; the Raven’s Colored Progressive Matrices (RCPMs), measuring nonverbal fluid intelligence; and the Verbal Abstraction Test (Comprehension and Verbal Absurdities subtests), assessing verbal reasoning. Independent-samples t-tests and mixed-design ANOVAs were conducted to compare group performance and examine within-task variability.
Results:
Children with ADHD-H performed significantly worse than controls on both the VFDT and the RCPMs total scores. Qualitative analysis revealed a marked decline in performance across VFDT item sets, more frequent peripheral errors in later trials. Group differences in RCPMs emerged in gestalt and analogy subcomponents but not in perceptual similarity items. Conversely, verbal abstraction scores did not differ significantly between groups.
Conclusions:
Findings suggest that attentional deficits, rather than global intellectual impairment, primarily account for lower nonverbal reasoning performance in children with ADHD-H. Verbal reasoning abilities appear relatively preserved. These results underscore the need for differential diagnostic assessment and targeted interventions to strengthen visuospatial attention and cognitive control in ADHD-H.
Aim:
Attention is a core cognitive function that supports higher-order processes such as reasoning, problem solving, and intelligence. In children with attention deficit hyperactivity disorder (ADHD), particularly the hyperactive subtype, impairments in attentional control may interfere with the development and expression of cognitive abilities. This study examined the relationship between visuospatial attention and both verbal and nonverbal intelligence in children with ADHD-hyperactive type (ADHD-H).
Methods:
A sample of 65 children with ADHD-H and 73 typically developing controls (aged 8–10 years) completed three tasks: the Benton Visual Form Discrimination Test (VFDT), assessing complex visuospatial attention; the Raven’s Colored Progressive Matrices (RCPMs), measuring nonverbal fluid intelligence; and the Verbal Abstraction Test (Comprehension and Verbal Absurdities subtests), assessing verbal reasoning. Independent-samples t-tests and mixed-design ANOVAs were conducted to compare group performance and examine within-task variability.
Results:
Children with ADHD-H performed significantly worse than controls on both the VFDT and the RCPMs total scores. Qualitative analysis revealed a marked decline in performance across VFDT item sets, more frequent peripheral errors in later trials. Group differences in RCPMs emerged in gestalt and analogy subcomponents but not in perceptual similarity items. Conversely, verbal abstraction scores did not differ significantly between groups.
Conclusions:
Findings suggest that attentional deficits, rather than global intellectual impairment, primarily account for lower nonverbal reasoning performance in children with ADHD-H. Verbal reasoning abilities appear relatively preserved. These results underscore the need for differential diagnostic assessment and targeted interventions to strengthen visuospatial attention and cognitive control in ADHD-H.
DOI: https://doi.org/10.37349/ent.2026.1004139
This article belongs to the special issue Advances in the Pathogenesis, Diagnosis and Treatment of Attention Deficit Hyperactivity Disorder

Background:
Multiple sclerosis (MS) is a chronic inflammatory and neurodegenerative disease affecting the central nervous system, the cause of which remains unknown. Environmental, genetic, and immunological factors are considered risk factors. MS has no cure; therefore, therapy focuses on reducing the number of outbreaks, controlling symptoms, and therapies aimed at modifying the course of the disease. Innovative strategies that promote remyelination and repair of damaged brain tissue are under investigation. This review aims to compile and systematize the available knowledge on the multifactorial nature of MS, highlighting the main risk factors. It also discusses the mechanisms underlying the pathogenesis of the disease, current therapies, and prospects, presenting a comprehensive overview of the effect of various drugs on remyelination and repair of central nervous system damage.
Methods:
A comprehensive literature search, guided by Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standards, was conducted across PubMed, Cochrane Library, Web of Science, and ClinicalTrials.gov to identify relevant clinical trials. Of the studies retrieved, 13 were selected for this review. These trials specifically explored integrated therapeutic approaches, combining pharmacological and non-pharmacological interventions, for managing MS.
Results:
The results reflect the multifactorial nature of MS and the existence of several promising therapies to combat inflammation and demyelination, as well as to promote remyelination. Reducing inflammation remains the main target, but new approaches such as clemastine, liothyronine, interleukin (IL)-2, N-acetylglucosamine, and intracranial transplantation of fetal human neural precursor cells have shown promising results.
Discussion:
Currently, the therapies available for MS target the peripheral immune system. Therefore, more studies are needed on treatment therapies that combine immunomodulation of the peripheral and central nervous systems to reduce the neurological disability of patients. It is also concluded that the therapies were safe and were well tolerated, given the occurrence of a small number of adverse events.
Background:
Multiple sclerosis (MS) is a chronic inflammatory and neurodegenerative disease affecting the central nervous system, the cause of which remains unknown. Environmental, genetic, and immunological factors are considered risk factors. MS has no cure; therefore, therapy focuses on reducing the number of outbreaks, controlling symptoms, and therapies aimed at modifying the course of the disease. Innovative strategies that promote remyelination and repair of damaged brain tissue are under investigation. This review aims to compile and systematize the available knowledge on the multifactorial nature of MS, highlighting the main risk factors. It also discusses the mechanisms underlying the pathogenesis of the disease, current therapies, and prospects, presenting a comprehensive overview of the effect of various drugs on remyelination and repair of central nervous system damage.
Methods:
A comprehensive literature search, guided by Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standards, was conducted across PubMed, Cochrane Library, Web of Science, and ClinicalTrials.gov to identify relevant clinical trials. Of the studies retrieved, 13 were selected for this review. These trials specifically explored integrated therapeutic approaches, combining pharmacological and non-pharmacological interventions, for managing MS.
Results:
The results reflect the multifactorial nature of MS and the existence of several promising therapies to combat inflammation and demyelination, as well as to promote remyelination. Reducing inflammation remains the main target, but new approaches such as clemastine, liothyronine, interleukin (IL)-2, N-acetylglucosamine, and intracranial transplantation of fetal human neural precursor cells have shown promising results.
Discussion:
Currently, the therapies available for MS target the peripheral immune system. Therefore, more studies are needed on treatment therapies that combine immunomodulation of the peripheral and central nervous systems to reduce the neurological disability of patients. It is also concluded that the therapies were safe and were well tolerated, given the occurrence of a small number of adverse events.
DOI: https://doi.org/10.37349/ent.2026.1004138
 Previous
Previous