Therapeutic Targets for Neuroprotection in Ischemic Stroke
Guest Editor
Dr. Silvia Fischer E-Mail
Institute of Biochemistry, medicine, Justus-Liebig-University, Giessen, Germany
Research Keywords: Endothelial cells; angiogenesis; endothelium; innate immunity; inflammatory responses
About the Special lssue
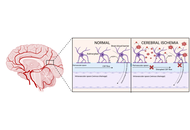
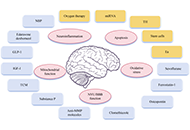
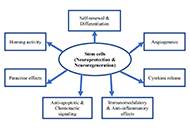
Stroke still remains the leading causes of death worldwide, accounting for 7.08 million deaths in 2020. Ischemic stroke is responsible for approximately 87% of all strokes, while hemorrhagic stroke accounts for 13%. Ischemic stroke, caused by, for example, thrombosis, embolism or systemic hypoperfusion, results in impaired blood flow and cell death, especially of neurons. Clinically, only intravenous thrombolysis with recombinant tissue plasminogen activator (rt-PA) and mechanical thrombectomy are administered for acute ischemic stroke to restore blood flow. Unfortunately, both treatments are limited because of a very short therapeutic window and numerous contraindications. Therefore, more knowledge about pathological cellular and molecular mechanisms in ischemic stroke is necessary to develop new strategies for neuroprotection. Some of the molecular events that can be targeted by neuroprotectants include glutamate release, glutamate receptor activation, intracellular Ca2+ increase, mitochondrial dysfunction, activation of several intracellular enzymes, generation of free radicals, apoptosis, blood-brain barrier disruption, and inflammation. Furthermore, ischemic stroke results in the release of danger-associated molecular patterns (DAMPs) from dying cells such as heat shock proteins, histones, thioredoxin, adenosine triphosphate, high mobility group box protein 1, and numerous self-nucleic acids. These DAMPs can orchestrate innate and adaptive immune responses in the injured regions of the brain by glial cell activation, leading to the release of cytokines and chemokines, and the recruitment of leukocytes. Activated neutrophils further release neutrophil extracellular traps (NETs) which have been identified in infarcted lesions in specimens from ischemic stroke patients. Thus, inhibitory targeting of these events might provide promising therapeutic interventions to treat ischemic stroke.
This special issue summarizes the new knowledge about cellular and molecular mechanisms in ischemic stroke and treatment modalities.
Keywords: ischemic stroke; neuroprotection; neurons; glial cells; danger-associated molecular patterns; inflammation
Published Articles