Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: http://orcid.org/0009-0006-7797-8973
Affiliation:
3Cognitive Neurology and Dementia Clinic, Instituto Nacional de Neurología y Neurocirugía “Dr. Manuel Velasco Suárez,” Mexico City 14269, Mexico
ORCID: https://orcid.org/0009-0002-1799-0277
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
Email: ildefonso.rodriguez@uaslp.mx
ORCID: https://orcid.org/0000-0002-3316-1471
Affiliation:
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0000-7094-5681
Affiliation:
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0007-7921-7008
Affiliation:
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0000-3758-3607
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0002-2933-6539
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0009-7074-245X
Affiliation:
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0000-0002-0436-902X
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0002-2131-8301
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0007-3455-1336
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0009-0009-5888-8771
Affiliation:
1Neurology Service, Hospital Regional de Alta Especialidad “Dr. Ignacio Morones Prieto”, San Luis Potosí 78290, México
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
ORCID: https://orcid.org/0000-0002-4369-540X
Affiliation:
2Faculty of Medicine, Autonomous University of San Luis Potosí, San Luis Potosí 78210, México
4“Dr. Everardo Neumann Peña” Psychiatric Clinic, San Luis Potosí 78430, México
ORCID: https://orcid.org/0009-0002-3071-9094
Explor Neuroprot Ther. 2025;5:1004113 DOI: https://doi.org/10.37349/ent.2025.1004113
Received: March 01, 2025 Accepted: July 14, 2025 Published: August 20, 2025
Academic Editor: Aurel Popa-Wagner, University of Duisburg-Essen, Germany
Aim: Epilepsy is associated with cognitive and neuropsychiatric impairments, affecting attention, memory, executive functions, and emotional well-being. While these impairments are well-documented in general epilepsy populations, limited research focuses on economically active young adults who face unique cognitive demands in competitive academic and professional environments. The objective of this research was to evaluate the cognitive and neuropsychiatric outcomes in economically active young adults with epilepsy compared to matched controls.
Methods: An observational, analytical, case-control study was conducted at a secondary medical center in Mexico. Participants included 25 patients with well-controlled epilepsy and 25 matched controls. Cognitive function was assessed using the Montreal Cognitive Assessment (MoCA) and Addenbrooke’s Cognitive Examination III (ACE-III). Anxiety and depression were evaluated using the Beck Anxiety Inventory and Patient Health Questionnaire-9 (PHQ-9). Statistical analysis included Student’s T-test and Chi-square for cognitive scores and Wilcoxon rank-sum tests for non-parametric data.
Results: Cognitive impairments were significantly more frequent in patients with epilepsy than in controls, particularly in attention (p = 0.041), episodic memory (p = 0.048), clock drawing (p = 0.004), and cube copying (p < 0.001). Verbal (p = 0.011) and semantic fluency (p = 0.027) were also affected. No significant differences were observed in anxiety (p = 0.221) or depression (p = 0.800) between groups.
Conclusions: Economically active young adults with well-controlled epilepsy showed significant cognitive impairments, particularly in attention, memory, visuospatial, and executive functions, independent of anxiety and depression levels. This study underscores the need for comprehensive cognitive and neuropsychiatric evaluations in epilepsy treatment, regardless of seizure control. It advocates for targeted cognitive rehabilitation and a holistic approach to epilepsy care beyond seizure frequency control.
Epilepsy is recognized as a disorder with neurobiological, cognitive, and psychosocial consequences [1]. Cognitive dysfunction, particularly in attention, memory, and executive functions, is observed in up to 70% of patients with epilepsy (PWE) [2], impacting their quality of life [3]. Psychiatric comorbidities such as anxiety and depression are also prevalent, with reported rates 2–3 times higher than in the general population [4]. Meta-analyses have reported a 23.1% prevalence of active depression in individuals with epilepsy, with an increased overall risk of 2.7 compared to the general population [5, 6].
There is an extensive body of research exploring cognitive and psychiatric impairments in epilepsy; existing studies often focus on broad patient populations, including pediatric, elderly, or medically refractory cases [7–11]. However, to the best of our knowledge, no studies have specifically focused on young adults actively participating in the workforce or higher education, a population facing distinct challenges associated with epilepsy. As these individuals must navigate complex cognitive demands in highly competitive academic and professional settings, identifying their specific impairments is essential for developing targeted support systems and effective intervention strategies.
Few healthcare centers in Mexico comprehensively assess epilepsy patients, particularly regarding cognitive and neuropsychiatric aspects [12]. Neuropsychological evaluations are primarily reserved for candidates for epilepsy surgery, excluding a significant subset of patients with relatively well-controlled seizures who remain functionally active in society. These individuals, including students, employees, and caregivers, are often overlooked for broader cognitive assessments despite their participation in key social and economic roles.
This study aims to address this gap by evaluating the cognitive and neuropsychiatric profile of economically active young adults with epilepsy in comparison to matched controls. By identifying potential subclinical cognitive impairments, we aim to highlight the need for more inclusive screening protocols and targeted interventions to improve quality of life. Additionally, given that psychiatric comorbidities such as anxiety and depression occur at higher rates in epilepsy patients, we underscore their relevance in comprehensive patient management. Neglecting these factors may lead to suboptimal epilepsy treatment and negatively impact patients’ overall well-being.
An observational, analytical, case-control study was conducted. All participants provided written informed consent, as per a protocol approved by the Research Ethics Committee (Approval No. 17–24) at the “Dr. Ignacio Morones Prieto” Regional High Specialty Hospital in San Luis Potosí, Mexico.
The study included patients aged 15 to 50 years with a well-documented epilepsy etiology confirmed through electroencephalography (EEG), brain magnetic resonance imaging (MRI), and comprehensive clinical history. Notably, only economically and socially active individuals were included, such as students and employed adults who demonstrated high levels of independence and functionality, leading everyday lives despite their epilepsy diagnosis. This group is often overlooked for neuropsychological evaluations in Mexico due to the assumption that their cognitive performance is unimpaired by their condition.
To minimize the possibility of bias, we excluded the following: all the patients with developmental epileptic encephalopathies, a prior history of anxiety or depression, uncontrolled diabetes, uncontrolled hypertension, hypothyroidism, cerebrovascular disease (ischemic or hemorrhagic), or any central nervous system (CNS) oncological pathology.
The patient group was compared to a control group, which was matched for sociocultural and economic characteristics. Both groups were assessed using validated screening tools for the Mexican population to evaluate not only cognitive performance but also the presence of affective disorders. This comprehensive approach aimed to determine the extent to which cognitive impairments could be attributed to epilepsy rather than underlying anxiety or depression.
For this purpose, the Beck Anxiety Inventory (BAI) was used to screen for anxiety [13], and the Patient Health Questionnaire-9 (PHQ-9) was employed to assess depressive symptoms [14]. These tools helped differentiate cognitive deficits related to epilepsy from those potentially influenced by affective comorbidities.
Regarding cognitive screening, the Montreal Cognitive Assessment (MoCA) version 8.2 was the primary tool used for detecting cognitive impairments [15, 16]. To provide a more detailed analysis, particularly in the domains of memory, language, visuospatial abilities, and executive function, the Addenbrooke’s Cognitive Examination III (ACE-III) was administered [17].
This layered approach allowed for a nuanced evaluation of cognitive performance while accounting for the potential confounding effects of anxiety and depression.
Statistical analyses were performed using IBM SPSS Statistics v.25 for Mac. The arithmetic mean, median (measures of central tendency), standard deviation, minimum, and maximum (measures of dispersion) were used for quantitative variables. For qualitative variables, relative frequency (percentages) and absolute frequency were used. The Chi-square (χ²) test was used for qualitative variables. For quantitative variables, the Student’s T-test was utilized in cases of normal distribution, and the Wilcoxon signed-rank test was used in cases of abnormal distribution. A 95% confidence interval was determined, with a p-value < 0.05 in two directions for statistically significant values.
The sample size was calculated using a formula for comparing two proportions, considering 95% confidence and 80% statistical power. The expected prevalence of cognitive impairment in PWE was set at 70% (p1 = 0.7). Based on a meta-analysis of 29,891 patients, a 23.1% prevalence of active depression in PWE (p2 = 0.231) was included to account for potential confounding by depression [5].
Using the standard formula [18]:
Where Zα/2 = 1.96, Zβ = 0.84, and p = 0.4655, the minimum required sample size was 17 participants per group. A final sample size of 25 participants per group was selected to ensure statistical robustness and account for potential variability.
A total of 25 patients (P) and 25 controls (C) were evaluated, with both groups matched appropriately for age (p = 0.65), gender (p = 0.765), and education level (p = 0.846) (see Table 1).
Population characteristics
| Characteristics | Patientsn = 25 | Controlsn = 25 | p |
|---|---|---|---|
| Age, expressed as median (IQR) | 22 (18.5–34.5) | 23 (19–36.5) | 0.65 |
| Gender | |||
| Male | 9 (36%) | 8 (32%) | 0.765 |
| Female | 16 (64%) | 17 (68%) | |
| Education level | |||
| Elementary school (6th) | 2 (8%) | 3 (12%) | 0.846 |
| Secondary (9th) | 13 (52%) | 10 (40%) | |
| High school | 8 (32%) | 10 (40%) | |
| Bachelor’s degree | 2 (8%) | 2 (8%) | |
Age is presented as median and interquartile ranges due to the non-normal age distribution in both groups. Categorical variables are displayed as frequency and percentage
The student’s T-test was used to compare age, and Chi-square tests were applied to categorical variables. No statistically significant differences were indicated, suggesting adequate matching between the patient and control groups.
Among the patients, 68% had focal epilepsy, 28% had generalized epilepsy, and 4% had epilepsy of unknown onset. The most common diagnosis was mesial temporal sclerosis (44%), followed by juvenile myoclonic epilepsy (20%) (see Figure 1).
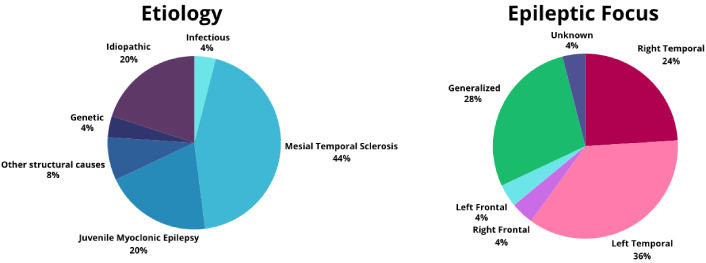
Epilepsy etiology and epileptic focus. A pie chart on the left shows the percentage distribution of the different epilepsy etiologies. On the right side, the percentages for specific epileptic foci are displayed
Regarding the use of anti-seizure medications (ASMs), the usage pattern was as follows: Most patients (68%) were on monotherapy, with levetiracetam being the most prescribed medication (68%), followed by carbamazepine (24%) and valproate (20%) (see Figure 2).
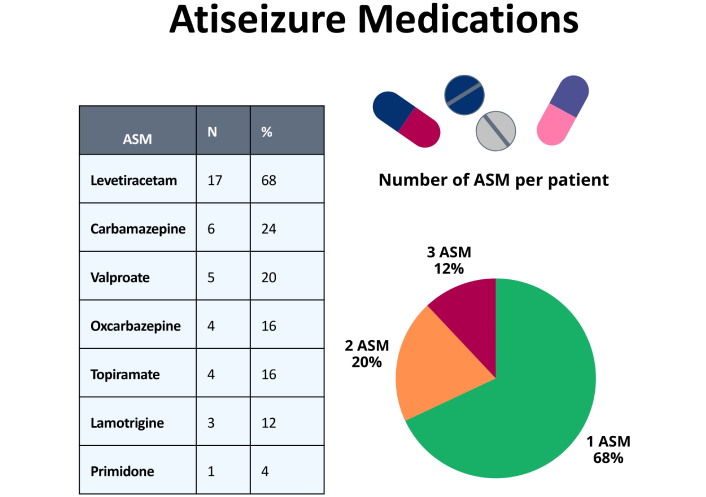
Use of anti-seizure medications (ASMs). The box on the left shows the percentage of use for each ASM. The pie chart on the right displays the number of ASMs used per patient
Seizure control among patients was generally favorable. Eight percent were seizure-free for over a year, and 12 percent were seizure-free for 6 to 12 months. Additionally, 16 percent reported no seizures in the last three months, while 44 percent experienced 1 to 3 seizures. Only 20 percent had four or more seizures in the previous three months. This distribution reflects a predominance of well-controlled epilepsy in the study population (see Figure 3).
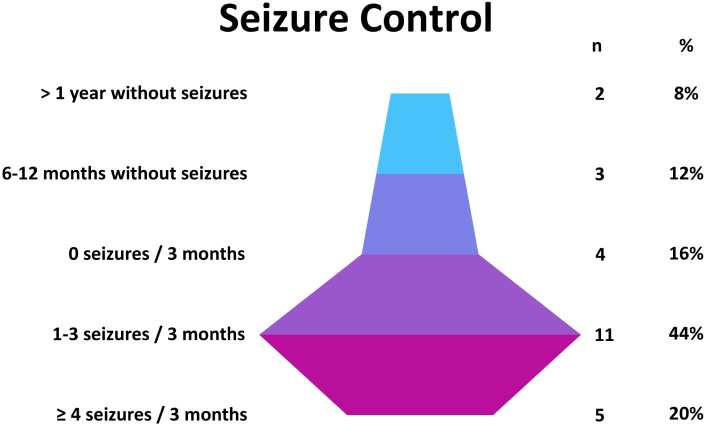
Illustrates the distribution of seizure control among patients over the last year. This distribution reflects a predominantly well-controlled seizure pattern, highlighting that most patients experienced infrequent seizures or were seizure-free for extended periods
There was no significant difference in the anxiety levels between the groups (see Table 2) or in depression levels (see Figure 4).
Beck Anxiety Inventory (BAI)
| BAI | Patientsn = 25 | Controls n = 25 | p |
|---|---|---|---|
| Mean | 22.24 | 17.28 | 0.221 |
| Standard deviation | 12.541 | 10.151 | |
| Standard error mean | 2.508 | 2.030 |
This table presents the mean, standard deviation, and standard error of the mean scores for the BAI in patients with epilepsy and controls. Statistical analysis was performed using the Student’s T-test. No significant differences were found between the groups (p = 0.221)
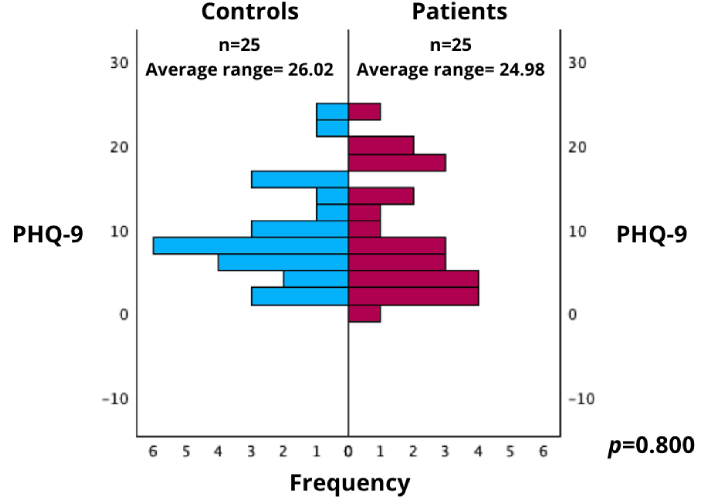
PHQ-9 scores in patients and controls. This figure shows the distribution of PHQ-9 scores comparing patients with epilepsy and controls, evaluated using the Wilcoxon rank-sum test. PHQ-9: Patient Health Questionnaire-9
The average rank for the control group was 26.02, while the average rank for the patient group was 24.98.
No significant differences were found between the groups (p = 0.800), indicating that both groups exhibited similar levels of depressive symptoms.
These results suggest that depressive symptoms were not a differentiating factor between PWE and the control group in this study.
Regarding cognitive screening tools, a significant difference was observed in MoCA scores using the T-test (patients: 20.76 vs. controls: 24.28, p = 0.044). Although the mean ACE-III score was lower in patients, it did not reach statistical significance (patients: 69.08 vs. controls: 81.32, p = 0.206) (see Table 3a)
Cognitive performance
| Cognitive screening tools | Patientsn = 25 | Controls n = 25 | P-value | |
|---|---|---|---|---|
| 3a. MoCA and ACE-II total scores | ||||
| MoCA | Mean | 20.76 | 24.28 | 0.044 |
| Standard deviation | 3.491 | 4.505 | ||
| Standard error mean | 0.698 | 0.901 | ||
| ACE-III | Mean | 69.08 | 81.32 | 0.206 |
| Standard deviation | 11.979 | 9.4919 | ||
| Standard error mean | 2.396 | 1.882 | ||
| 3b. MoCA and ACE-III screening results for cognitive impairment | ||||
| MoCA | No cognitive impairment | 4 (16%) | 12 (48%) | 0.015 |
| Cognitive impairment | 21 (84%) | 13 (52%) | ||
| ACE-III | No cognitive impairment | 4 (16%) | 16 (64%) | < 0.001 |
| Cognitive impairment | 21 (84%) | 9 (36%) | ||
| 3c. Verbal and semantic fluency performance | ||||
| Verbal fluency | < 11 per minute | 16 (64%) | 7 (28%) | 0.011 |
| ≥ 11 per minute | 9 (36%) | 18 (72%) | ||
| Semantic fluency | < 17 per minute | 19 (76%) | 11 (44%) | 0.027 |
| ≥ 17 per minute | 6 (24%) | 14 (56%) | ||
3a. MoCA and ACE-III total scores: Displays the total scores for Montreal Cognitive Assessment (MoCA) and Addenbrooke’s Cognitive Examination (ACE-III). Statistical analysis was performed using the Student’s T-test. 3b. MoCA and ACE-III screening results for cognitive impairment: The dichotomous analysis of cognitive impairment using MoCA and ACE-III is presented as a 2 × 2 Chi-square table. 3c. Verbal and semantic fluency performance: This section displays verbal and semantic fluency results, analyzed as a 2 × 2 Chi-square table. P-values are reported to indicate statistical significance
A dichotomous analysis was performed using the Chi-square test, considering the cut-off points of both tests. The comparison revealed that the number of subjects reaching MoCA scores of 26 or higher, indicating an average score, was 84%, compared to 52% of the controls (p = 0.015). Additionally, the analysis revealed that the number of patients reaching ACE-III scores ≥ 82, the cut-off point for this test, was 84% compared to 36% in controls (p < 0.001) (see Table 3b).
A secondary analysis was conducted to evaluate subjects’ performance in different areas. The MoCA attention score was assessed, which allocates 2 points for digit span (1 point for a series of 5 numbers in forward order and 1 for a series of 3 numbers in backward order), 1 point for a vigilance test (where the subject taps each time they hear the letter “A” in a sequence of letters), and 3 points for a serial subtraction task, subtracting seven from 100. The total expected score in this section is 6 points. A statistically significant difference was found in favor of the controls (p = 0.041) (see Figure 5a).
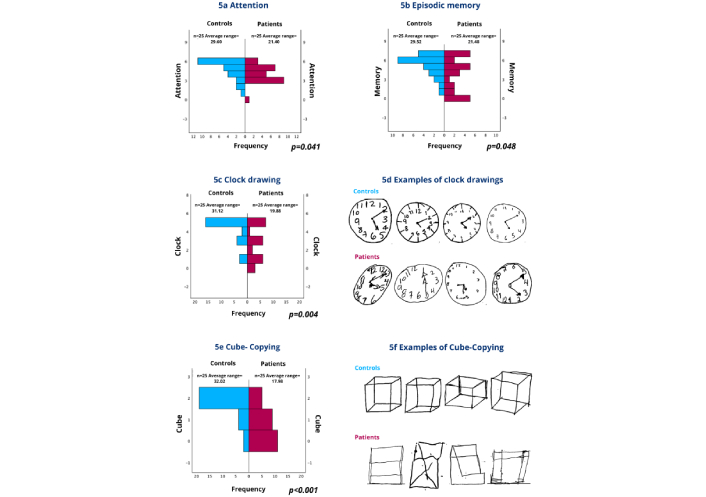
Performance in attention, episodic memory, the clock-drawing test, and the cube-copying test. 5a. Attention: Displays performance on the attention section of the MoCA, which includes the letter A test, forward and backward digit span, and serial subtraction of 7 from 70. Results are presented using the Wilcoxon rank-sum test. 5b. Episodic memory: This section presents performance on episodic memory, which was assessed using the delayed recall test from the ACE-III. Participants were asked to recall seven details after 5 minutes, and results were shown using the Wilcoxon rank-sum test. 5c. Clock drawing: Compares performance on the Clock-drawing test with a scoring range of 0 to 5 points, using the Wilcoxon rank-sum test. 5d. Examples of clock drawings: Representative examples of clock-drawing test performance between controls and patients with epilepsy. Controls demonstrated better spatial organization, accurate number placement, and correct hand positioning for the requested time (11:10). Patients with epilepsy showed various degrees of impairment, including poor circle formation, scattered number placement, and incorrect hand positioning, resulting in significantly lower scores (p = 0.004). 5e. Cube-copying test: Displays results of the cube-copying test with a scoring range of 0 to 2 points, analyzed using the Wilcoxon rank-sum test. 5f. Examples of cube-copying: Representative examples of cube-copying test performance between controls and patients with epilepsy. Successfully controls the reproduction of three-dimensional cubes with proper perspective, depth, and spatial relationships. Patients with epilepsy demonstrated significant visuospatial difficulties, often producing two-dimensional squares or poorly executed three-dimensional attempts, resulting in significantly lower scores (p < 0.001). MoCA: Montreal Cognitive Assessment; ACE-III: Addenbrooke’s Cognitive Examination III
Episodic memory was also evaluated using the delayed recall test from ACE-III, where subjects were asked to recall the name and address of a person after 5 minutes, remembering seven details (1. Luis; 2. Gonzalez; 3. Street; 4. Alameda; 5. Number 12; 6. Zapopan; 7. Jalisco). Controls performed better (p = 0.048) (see Figure 5b).
Similarly, performance in the clock-drawing test was compared, with a scoring range of 0 to 5 points, showing better results in the controls (p = 0.004) (see Figures 5c and 5d). In the cube-copying test, with a range of 0 to 2 points, the controls also performed better (p < 0.001) (see Figures 5e and 5f).
Verbal fluency was evaluated by giving subjects one minute to name as many words as possible, starting with the letter “P”. While 72% of controls achieved a rate of ≥ 11 words per minute, only 36% of patients met the expected minimum (p = 0.011) (see Table 3c). Finally, semantic fluency was assessed by asking subjects to name as many animals as possible within a one-minute time frame. Notably, 56% of controls listed ≥ 17 animals, compared to 24% of patients (p = 0.027) (see Table 3c).
A sub-analysis was conducted to compare each of the points analyzed between the different etiologies of epilepsy, but no statistically significant difference was found. The type of medication or seizure control was also analyzed to determine if it made a difference, but no significant differences were found.
This study demonstrates that cognitive impairments persist as a significant challenge even in well-controlled and functionally active epilepsy patients. The specific pattern of deficits observed—particularly in attention, episodic memory, visuospatial processing, and executive functions—suggests the involvement of distributed neural networks extending beyond the primary epileptogenic focus, consistent with previous research emphasizing the multifactorial nature of cognitive dysfunction in epilepsy [3].
Approximately 70% of patients achieve optimal seizure control with medical therapy alone [19]. However, focusing solely on seizure frequency provides an incomplete view of patient management [20]. Our cohort exemplifies this complexity: despite 68% being on monotherapy with reasonable seizure control, significant cognitive impairments were evident across multiple domains [21–23].
The observed attention deficits align with the disruption of frontal-parietal circuits that comprise the “attention network” [24]. These circuits are vulnerable to interictal epileptiform discharges occurring even in well-controlled patients [25, 26]. Impairments in serial subtraction and vigilance tasks indicate a disruption of both sustained and selective attention mechanisms, which are essential for academic and occupational success.
Episodic memory deficits, as demonstrated through delayed recall testing, correlate with the high prevalence of mesial temporal sclerosis (44%) in our cohort. However, memory dysfunction across different epilepsy etiologies suggests the involvement of broader limbic-cortical networks beyond hippocampal pathology, with significant implications for understanding memory consolidation processes in epilepsy.
The striking differences in clock-drawing and cube-copying performance (p = 0.004 and p < 0.001, respectively) highlight significant visuospatial processing deficits. These tasks require the integration of spatial perception, motor planning, and executive control, suggesting dysfunction in parietal-occipital networks affected by epileptiform activity, which occurs through direct involvement and secondary network disruption [27].
Deficits in verbal and semantic fluency (p = 0.011 and p = 0.027, respectively) reflect language network dysfunction, which requires efficient retrieval of semantic memory, phonological processing, and executive control. Given that 68% of patients had focal epilepsy with common temporal lobe involvement, these impairments may reflect both direct effects on language-processing regions and indirect effects through pathway disruption [28].
Four key mechanisms likely contribute to cognitive impairments in well-controlled epilepsy: a) Interictal Epileptiform Activity: Subclinical discharges disrupt normal neural processing, causing transient cognitive interference [29]. b) Network Reorganization: Chronic epilepsy leads to maladaptive brain network reorganization, affecting cognitive efficiency in areas distant from the primary focus [30]. c) Medication Effects: ASMs can have subtle cognitive effects, particularly on processing speed and attention [31, 32]. d) Chronic Neuroinflammation: Epilepsy-associated neuroinflammation may contribute to progressive cognitive decline [33].
Although anxiety and depression levels were comparable between groups, this finding requires cautious interpretation. Literature reports prevalence rates 2 to 3 times higher in epilepsy patients [5, 22]. The absence of differences may reflect our sample size or the exclusion of patients with prior psychiatric diagnoses, potentially underestimating the comorbidity burden.
Importantly, matching both groups for mood symptoms demonstrates that observed cognitive deficits are attributable to epilepsy itself rather than untreated affective disorders, strengthening the validity of our cognitive findings.
Cognitive impairments in our economically active population have profound implications for quality of life and social integration. Attention deficits impact workplace productivity and academic performance, while memory impairments affect skill acquisition and information retention. Executive dysfunction impairs problem-solving and adaptive behavior in complex environments.
These findings challenge traditional epilepsy management focused primarily on seizure frequency. Our results indicate that seizure freedom alone is insufficient for optimal cognitive functioning and quality of life, necessitating a shift in paradigm toward comprehensive care [16].
Our findings support several clinical practice changes: a) Routine cognitive screening, regardless of seizure control status. b) Multidisciplinary care teams, including neuropsychologists and rehabilitation specialists. c) Cognitive profile consideration in ASM selection and treatment planning. d) Educational and workplace accommodations, even with well-controlled seizures.
Our findings align with international studies demonstrating persistent cognitive deficits in patients with reasonable seizure control. However, our focus on economically active young adults provides unique insights into an often-overlooked population that faces significant cognitive demands [34].
The 84% prevalence of cognitive impairment (ACE-III criteria) exceeds some international rates, potentially reflecting cultural and educational factors specific to the Mexican population or methodological differences.
Critical research priorities include a) conducting longitudinal studies examining cognitive trajectory patterns. b) To use advanced neuroimaging to identify structural and functional correlates. c) To search for randomized controlled trials of targeted cognitive rehabilitation programs. d) Biomarker development for cognitive risk prediction and early intervention.
Our study has several limitations, particularly methodological constraints. Our small sample size (n = 25 per group), although statistically justified, limits generalizability and the capabilities for subgroup analysis. The inherent variability in neuropsychological assessments requires larger cohorts for robust findings. Future studies should aim for at least 100 participants per group to facilitate meaningful subgroup analyses.
Reliance on screening tools (MoCA and ACE-III), though validated and practical, represents a significant constraint. These instruments detect impairment but may miss subtle deficits or fail to provide detailed cognitive profiles. Comprehensive neuropsychological batteries would yield more nuanced, clinically relevant information.
Selection bias resulting from focusing on economically active young adults limits the generalizability of the findings to the broader population. Excluding patients with psychiatric history may have selected higher-functioning individuals, potentially underestimating the actual cognitive burden. The single-center design and cross-sectional methodology further limit the interpretability of the results.
We lacked a systematic assessment of ASM dosing, plasma levels, medication burden, epilepsy duration, age of onset, and detailed seizure patterns—all potentially influencing cognitive outcomes. Educational quality, socioeconomic status, and occupational complexity were not assessed despite their potential confounding effects.
Cognitive assessments may be influenced by cultural and linguistic factors not fully captured by validated instruments. As a cross-sectional study, we cannot determine whether impairments are stable, progressive, or reversible.
This pilot study—the first in Mexico focusing on economically active young adults with epilepsy—demonstrates significant cognitive impairments in attention, memory, visuospatial, and executive functions despite well-controlled seizures and comparable mood symptoms to controls. These findings indicate that cognitive deficits are attributable to epilepsy itself rather than neuropsychiatric comorbidities, challenging the traditional seizure-focused approach to epilepsy management.
Our results establish that epilepsy evaluation requires a comprehensive assessment integrating neuropsychological and neuropsychiatric components regardless of seizure control status. Cognitive screening and individualized rehabilitation should become standard care components, recognizing that epilepsy encompasses far more than seizure frequency. These findings provide crucial evidence for the need to redefine treatment success in epilepsy beyond seizure control to include cognitive and functional outcomes.
While this study reveals significant cognitive impairments, definitive conclusions regarding the underlying mechanisms or relationships between epilepsy subtypes require larger sample sizes and targeted study designs. These findings establish a foundation for future research addressing the complex interplay between epilepsy, cognition, and functional outcomes in this important but underexplored population.
Based on our findings, fundamental changes in epilepsy care delivery are warranted to address the comprehensive needs of patients with cognitive impairments.
Healthcare systems should implement evidence-based policies requiring routine cognitive evaluation for all epilepsy patients, establishing cognitive screening as an essential component of epilepsy care standards. This paradigm shift recognizes cognitive function as a critical outcome measure alongside seizure control.
Epilepsy management must evolve to include mandatory access to neuropsychological services, occupational therapy, and cognitive rehabilitation programs. Insurance policies should provide comprehensive coverage for neuropsychological assessments and cognitive interventions, recognizing these services as medically necessary rather than optional.
Healthcare systems should establish standardized protocols to ensure that all newly diagnosed epilepsy patients receive comprehensive cognitive and neuropsychiatric evaluations within six months of diagnosis. Annual cognitive monitoring using validated screening tools should become an integral part of routine epilepsy follow-up, similar to current seizure documentation practices.
Clear referral pathways must connect neurology services with neuropsychology, psychiatry, and rehabilitation specialists to ensure seamless care coordination. Treatment decision-making should systematically integrate cognitive assessment results into the selection of antiseizure medications and intervention planning, moving beyond seizure control as the sole therapeutic target.
Educational institutions and employers require standardized cognitive accommodation programs specifically designed for individuals with epilepsy. These programs should address the unique pattern of deficits identified in our study—attention, memory, visuospatial, and executive dysfunction—rather than generic disability accommodations.
Healthcare systems should invest in training programs for educators, employers, and healthcare providers about cognitive challenges faced by individuals with epilepsy. This education is essential for reducing stigma and improving functional outcomes in academic and workplace settings.
The establishment of structured peer support programs can connect young adults with epilepsy who face similar cognitive and psychosocial challenges, providing both emotional support and practical strategies for managing cognitive difficulties in competitive environments.
Future research should prioritize longitudinal studies examining cognitive trajectories, neuroimaging investigations of structural and functional correlates, and randomized controlled trials of targeted cognitive rehabilitation programs. Implementation science approaches are needed to evaluate the effectiveness and cost-effectiveness of proposed policy changes.
This study’s findings extend beyond Mexico’s healthcare context, providing evidence relevant to epilepsy care globally, particularly in resource-limited settings where comprehensive cognitive assessment is often unavailable. The focus on economically active young adults addresses a critical gap in epilepsy research and highlights the need for age-specific, functionally oriented care approaches.
By demonstrating significant cognitive impairments in well-controlled epilepsy patients, this research supports a fundamental reconceptualization of epilepsy as a complex neurological condition requiring multidimensional therapeutic approaches. The proposed policy and clinical recommendations provide a roadmap for healthcare systems to implement evidence-based, comprehensive epilepsy care that addresses the full spectrum of patient needs.
The ultimate goal is to ensure that individuals with epilepsy receive holistic care, promoting not only seizure control but also optimal cognitive functioning, quality of life, and successful social, educational, and occupational integration. This comprehensive approach represents the next evolution in epilepsy care, moving from a disease-focused to a person-centered model that recognizes and addresses the complex challenges faced by young adults with epilepsy in contemporary society.
ACE-III: Addenbrooke’s Cognitive Examination III
ASMs: anti-seizure medications
MoCA: Montreal Cognitive Assessment
PHQ-9: Patient Health Questionnaire-9
PWE: patients with epilepsy
The authors thank all participants who volunteered for this study and the staff at the Central Hospital “Dr. Ignacio Morones Prieto” for their support during data collection. The resources for this study were provided by the authors and participating institutions.
RGRM: Conceptualization, Methodology, Investigation, Formal analysis, Writing—original draft, Writing—review & editing. RMR: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. IRL: Conceptualization, Methodology, Supervision, Writing—review & editing, Project administration. CMT, LMMC, JLRC, LDMF, DMG, DGMR, MOHB, SFG, MGG, GAMS, and ISDH: Investigation, Data curation. HGHR: Validation, Formal analysis
The authors declare that there are no conflicts of interest.
The study was approved by the Research Ethics Committee (Approval No. 17–24) in the Central Hospital “Dr. Ignacio Morones Prieto”, in San Luis Potosí, Mexico, which complies with the ethical standards of the Declaration of Helsinki (Geneva, 2024 revision).
Informed consent to participate in the study was obtained and signed by all participants.
Informed consent to publication was obtained from participants. The paper does not involve privacy issues, such as displaying individual details (images or videos).
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
Not applicable.
© The Author(s) 2025.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2025. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
