






Background:
Pain affected gait performance in patients with non-specific chronic low back pain (NSCLBP) due to protective mechanisms in response to pain. Insufficient evidence demonstrated whether pain altered the walking patterns in NSCLBP. Therefore, this review aimed to compare the walking patterns between individuals with and without NSCLBP.
Methods:
The literature was systematically reviewed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The Cochrane, PubMed, Web of Science (WOS), EMBASE, and EBSCOhost were searched for the relevant studies, which were included if they: 1) compared patients with NSCLBP to the healthy controls, 2) evaluated spatiotemporal gait characteristics, 3) were published in English, 4) were peer-reviewed original research articles, and 5) were published since the year of 2000. Two researchers independently assessed the quality of the studies using the modified Downs and Black checklist. Meta-analyses with the random effects model were used, and the standardized mean differences (SMD) and the 95% confidence intervals (CI) were analyzed.
Results:
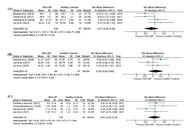
Out of 822 records, thirteen studies were included in the systematic review, and twelve were incorporated into the meta-analysis. The meta-analysis revealed significant differences between NSCLBP patients and healthy controls in walking speed (SMD = –0.67, 95% CI: –1.08 to –0.25, p = 0.002, I2 = 76%) and step length (SMD = –0.55, 95% CI: –0.94 to –0.17, p = 0.004, I2 = 64%).
Discussion:
Within spatiotemporal gait parameters, patients with NSCLBP show moderately consistent reductions in walking speed and step length relative to healthy controls, which may reflect pain-related protective strategies. These alterations underscore clinically meaningful gait impairments that should be considered in the design of targeted rehabilitation programs. PROSPERO registration number is CRD42023418511.
Background:
Pain affected gait performance in patients with non-specific chronic low back pain (NSCLBP) due to protective mechanisms in response to pain. Insufficient evidence demonstrated whether pain altered the walking patterns in NSCLBP. Therefore, this review aimed to compare the walking patterns between individuals with and without NSCLBP.
Methods:
The literature was systematically reviewed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The Cochrane, PubMed, Web of Science (WOS), EMBASE, and EBSCOhost were searched for the relevant studies, which were included if they: 1) compared patients with NSCLBP to the healthy controls, 2) evaluated spatiotemporal gait characteristics, 3) were published in English, 4) were peer-reviewed original research articles, and 5) were published since the year of 2000. Two researchers independently assessed the quality of the studies using the modified Downs and Black checklist. Meta-analyses with the random effects model were used, and the standardized mean differences (SMD) and the 95% confidence intervals (CI) were analyzed.
Results:
Out of 822 records, thirteen studies were included in the systematic review, and twelve were incorporated into the meta-analysis. The meta-analysis revealed significant differences between NSCLBP patients and healthy controls in walking speed (SMD = –0.67, 95% CI: –1.08 to –0.25, p = 0.002, I2 = 76%) and step length (SMD = –0.55, 95% CI: –0.94 to –0.17, p = 0.004, I2 = 64%).
Discussion:
Within spatiotemporal gait parameters, patients with NSCLBP show moderately consistent reductions in walking speed and step length relative to healthy controls, which may reflect pain-related protective strategies. These alterations underscore clinically meaningful gait impairments that should be considered in the design of targeted rehabilitation programs. PROSPERO registration number is CRD42023418511.
DOI: https://doi.org/10.37349/emd.2026.1007124

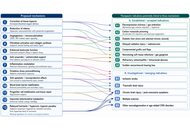
Advanced Therapy Medicinal Products (ATMPs) represent a transformative class of innovative therapies based on genes, cells, or engineered tissues that aim to modify, repair, or replace biological functions at a fundamental level. This review provides a foundational overview of ATMPs, addressing their scientific basis, regulatory classification, clinical translation, and key challenges for future development. The article outlines the four principal categories of ATMPs: gene therapy medicinal products, somatic cell therapy medicinal products, tissue-engineered products, and combined ATMPs, and discusses their distinct mechanisms of action and therapeutic applications. Recent clinical successes, including chimeric antigen receptor T-cell therapies, gene replacement therapies for inherited disorders, and tissue-engineered constructs for regenerative medicine, demonstrate the paradigm shift from symptomatic management towards disease modification or potential cure. However, the clinical implementation of ATMPs presents substantial challenges related to safety monitoring, long-term efficacy, complex manufacturing processes, regulatory evaluation, high treatment costs, and equitable patient access. Ethical considerations, including informed consent, long-term follow-up obligations, and global disparities in availability, further complicate their integration into routine clinical practice. Regulatory agencies have introduced adaptive pathways and accelerated approval mechanisms to facilitate timely patient access while maintaining rigorous standards of quality, safety, and efficacy. Continued technological innovation, coupled with sustainable healthcare policies and international collaboration, will be essential to realise the full therapeutic potential of ATMPs. As evidence accumulates from clinical trials and real-world use, ATMPs are expected to play an increasingly prominent role in modern medicine and the future of personalised healthcare.
Advanced Therapy Medicinal Products (ATMPs) represent a transformative class of innovative therapies based on genes, cells, or engineered tissues that aim to modify, repair, or replace biological functions at a fundamental level. This review provides a foundational overview of ATMPs, addressing their scientific basis, regulatory classification, clinical translation, and key challenges for future development. The article outlines the four principal categories of ATMPs: gene therapy medicinal products, somatic cell therapy medicinal products, tissue-engineered products, and combined ATMPs, and discusses their distinct mechanisms of action and therapeutic applications. Recent clinical successes, including chimeric antigen receptor T-cell therapies, gene replacement therapies for inherited disorders, and tissue-engineered constructs for regenerative medicine, demonstrate the paradigm shift from symptomatic management towards disease modification or potential cure. However, the clinical implementation of ATMPs presents substantial challenges related to safety monitoring, long-term efficacy, complex manufacturing processes, regulatory evaluation, high treatment costs, and equitable patient access. Ethical considerations, including informed consent, long-term follow-up obligations, and global disparities in availability, further complicate their integration into routine clinical practice. Regulatory agencies have introduced adaptive pathways and accelerated approval mechanisms to facilitate timely patient access while maintaining rigorous standards of quality, safety, and efficacy. Continued technological innovation, coupled with sustainable healthcare policies and international collaboration, will be essential to realise the full therapeutic potential of ATMPs. As evidence accumulates from clinical trials and real-world use, ATMPs are expected to play an increasingly prominent role in modern medicine and the future of personalised healthcare.
DOI: https://doi.org/10.37349/eds.2026.1008162

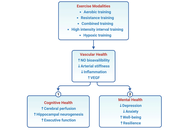
Obesity during the menopausal transition accelerates vascular aging through systemic inflammation, insulin resistance, and estrogen loss. These pathological processes impair endothelial function and arterial compliance, thereby increasing cardiovascular risk while simultaneously disrupting cerebral circulation, neurovascular regulation, and neuroendocrine stability that contribute to cognitive decline and psychological vulnerability. Regular exercise has emerged as an important non-pharmacological strategy to counteract these multidimensional impairments. Particular attention has been given to the modifying role of hormonal status and the differential adaptations observed between premenopausal and postmenopausal states. Evidence indicates that aerobic and multimodal programs enhance nitric oxide bioavailability, vascular elasticity, and cerebral perfusion, whereas resistance training contributes to musculoskeletal strength, metabolic regulation, and psychological resilience. Novel approaches such as interval-based or hypoxic exercise may provide additional benefits for postmenopausal women but require individualized supervision. Importantly, exercise-induced vascular improvements extend beyond cardiovascular protection, restoring cerebral blood flow, promoting hippocampal plasticity, and stabilizing hypothalamic–pituitary–adrenal axis function. These adaptations mediate enhancements in memory, executive performance, mood regulation, and stress resilience. This review synthesizes current findings across aerobic training, resistance training, combined training, high-intensity interval training, and hypoxic conditioning, and proposes an integrative vascular–cognitive–mental health framework that unifies these domains into a coherent model, with vascular function as a central mechanistic pathway linking exercise to cognitive and psychological outcomes, while underscoring the need for precision exercise prescriptions tailored to hormonal status, vascular risk, and functional capacity in obese women.
Obesity during the menopausal transition accelerates vascular aging through systemic inflammation, insulin resistance, and estrogen loss. These pathological processes impair endothelial function and arterial compliance, thereby increasing cardiovascular risk while simultaneously disrupting cerebral circulation, neurovascular regulation, and neuroendocrine stability that contribute to cognitive decline and psychological vulnerability. Regular exercise has emerged as an important non-pharmacological strategy to counteract these multidimensional impairments. Particular attention has been given to the modifying role of hormonal status and the differential adaptations observed between premenopausal and postmenopausal states. Evidence indicates that aerobic and multimodal programs enhance nitric oxide bioavailability, vascular elasticity, and cerebral perfusion, whereas resistance training contributes to musculoskeletal strength, metabolic regulation, and psychological resilience. Novel approaches such as interval-based or hypoxic exercise may provide additional benefits for postmenopausal women but require individualized supervision. Importantly, exercise-induced vascular improvements extend beyond cardiovascular protection, restoring cerebral blood flow, promoting hippocampal plasticity, and stabilizing hypothalamic–pituitary–adrenal axis function. These adaptations mediate enhancements in memory, executive performance, mood regulation, and stress resilience. This review synthesizes current findings across aerobic training, resistance training, combined training, high-intensity interval training, and hypoxic conditioning, and proposes an integrative vascular–cognitive–mental health framework that unifies these domains into a coherent model, with vascular function as a central mechanistic pathway linking exercise to cognitive and psychological outcomes, while underscoring the need for precision exercise prescriptions tailored to hormonal status, vascular risk, and functional capacity in obese women.
DOI: https://doi.org/10.37349/emed.2026.1001409

The increasing survival rates among paediatric and adolescent cancer patients has increased attention on long-term consequences of chemotherapy, particularly male fertility. This review addresses age- and dose-dependent gonadotoxicity and drug type on reproductive potential. It further investigates the damage, including disruption of the hypothalamic-pituitary-gonadal axis and epigenetic alterations that may pose transgenerational risks. A systematic search of PubMed, Scopus, Web of Science, and Google Scholar was conducted for studies from database inception to April 2025. Inclusion criteria included paediatric and adolescent male cancer patients or adult survivors of childhood cancer and reported chemotherapy-related effects on fertility. Preclinical animal models were included to elucidate epigenetic changes. Due to heterogeneity of study designs, a narrative synthesis was performed to categorize findings into hormonal, cellular, and clinical outcomes. Chemotherapy-induced infertility is highly dependent on the developmental stage and specific regimen. Alkylating agents and platinum-based therapies were consistently associated with impaired spermatogenesis, hormonal disruption, and azoospermia or oligospermia. Chemotherapy and cranial irradiation were altered hormonal system that regulates male reproduction and persists epigenetic changes in germ cells. Fertility preservation for postpubertal males is through sperm cryopreservation, while prepubertal boys relied on experimental strategies such as testicular tissue cryopreservation and in vitro spermatogenesis. Chemotherapy induces epigenetic after-effects, including altered DNA methylation patterns that persist even after spermatogenesis recovers. Chemotherapy compromises male fertility through cytotoxic damage and potential long-term genomic instability. The findings highlight that reproductive recovery does not guarantee genomic recovery (epigenetically intact sperm). Consequently, oncofertility care must adapt a reproductive health model that prioritizes early, customized counselling and use of biomarkers to better predict and preserve fertility in young survivors. Although sperm banking remains a standard approach for postpubertal, promising experimental may expand fertility options for prepubertal boys in future.
The increasing survival rates among paediatric and adolescent cancer patients has increased attention on long-term consequences of chemotherapy, particularly male fertility. This review addresses age- and dose-dependent gonadotoxicity and drug type on reproductive potential. It further investigates the damage, including disruption of the hypothalamic-pituitary-gonadal axis and epigenetic alterations that may pose transgenerational risks. A systematic search of PubMed, Scopus, Web of Science, and Google Scholar was conducted for studies from database inception to April 2025. Inclusion criteria included paediatric and adolescent male cancer patients or adult survivors of childhood cancer and reported chemotherapy-related effects on fertility. Preclinical animal models were included to elucidate epigenetic changes. Due to heterogeneity of study designs, a narrative synthesis was performed to categorize findings into hormonal, cellular, and clinical outcomes. Chemotherapy-induced infertility is highly dependent on the developmental stage and specific regimen. Alkylating agents and platinum-based therapies were consistently associated with impaired spermatogenesis, hormonal disruption, and azoospermia or oligospermia. Chemotherapy and cranial irradiation were altered hormonal system that regulates male reproduction and persists epigenetic changes in germ cells. Fertility preservation for postpubertal males is through sperm cryopreservation, while prepubertal boys relied on experimental strategies such as testicular tissue cryopreservation and in vitro spermatogenesis. Chemotherapy induces epigenetic after-effects, including altered DNA methylation patterns that persist even after spermatogenesis recovers. Chemotherapy compromises male fertility through cytotoxic damage and potential long-term genomic instability. The findings highlight that reproductive recovery does not guarantee genomic recovery (epigenetically intact sperm). Consequently, oncofertility care must adapt a reproductive health model that prioritizes early, customized counselling and use of biomarkers to better predict and preserve fertility in young survivors. Although sperm banking remains a standard approach for postpubertal, promising experimental may expand fertility options for prepubertal boys in future.
DOI: https://doi.org/10.37349/emed.2026.1001408

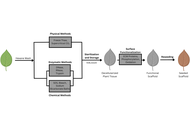
The development of biomaterials capable of supporting complex tissue growth remains a central challenge in regenerative medicine and tissue engineering, particularly in replicating the structural, mechanical, and transport functions of native extracellular matrices. While decellularized animal tissues have demonstrated significant success as scaffolds for tissue engineering, they are still constrained by cost, immunogenicity, and ethical concerns. In recent years, decellularized plant tissues have emerged as a compelling alternative scaffold platform due to their inherent vascular architectures, ethical sourcing, tunable mechanical properties, cytocompatibility, and sustainability. This review summarizes current strategies for the decellularization of plant tissues, including chemical, enzymatic, and physical approaches, and discusses how these methods preserve plant cell wall structure while removing immunogenic components. Advances in surface loading and functionalization, including protein coatings, oxidation, nanoparticle incorporation, peptide conjugation, and bioactive molecule loading, have further enhanced cell adhesion, differentiation, biodegradability, and immunomodulation. Recent applications of decellularized plant scaffolds in cardiac, skeletal muscle, bone, nerve, and wound healing contexts are reviewed, highlighting proof-of-concept successes and remaining challenges. Beyond therapeutic applications, plant-derived scaffolds have also enabled physiologically relevant in vitro models for vascular biology, mechanotransduction, cancer, metabolic tissues, and drug response studies. Collectively, these advances position decellularized plant tissues as versatile, low-cost, and ethically favorable biomaterials with growing relevance for both regenerative medicine and tissue modeling.
The development of biomaterials capable of supporting complex tissue growth remains a central challenge in regenerative medicine and tissue engineering, particularly in replicating the structural, mechanical, and transport functions of native extracellular matrices. While decellularized animal tissues have demonstrated significant success as scaffolds for tissue engineering, they are still constrained by cost, immunogenicity, and ethical concerns. In recent years, decellularized plant tissues have emerged as a compelling alternative scaffold platform due to their inherent vascular architectures, ethical sourcing, tunable mechanical properties, cytocompatibility, and sustainability. This review summarizes current strategies for the decellularization of plant tissues, including chemical, enzymatic, and physical approaches, and discusses how these methods preserve plant cell wall structure while removing immunogenic components. Advances in surface loading and functionalization, including protein coatings, oxidation, nanoparticle incorporation, peptide conjugation, and bioactive molecule loading, have further enhanced cell adhesion, differentiation, biodegradability, and immunomodulation. Recent applications of decellularized plant scaffolds in cardiac, skeletal muscle, bone, nerve, and wound healing contexts are reviewed, highlighting proof-of-concept successes and remaining challenges. Beyond therapeutic applications, plant-derived scaffolds have also enabled physiologically relevant in vitro models for vascular biology, mechanotransduction, cancer, metabolic tissues, and drug response studies. Collectively, these advances position decellularized plant tissues as versatile, low-cost, and ethically favorable biomaterials with growing relevance for both regenerative medicine and tissue modeling.
DOI: https://doi.org/10.37349/ebmx.2026.101367
This article belongs to the special issue Nature-Based Biomaterials for Biomedical Applications

The global rise of antimicrobial resistance (AMR) has emerged as one of the most pressing threats to public health. This crisis calls for the urgent development of alternative therapeutic agents against antibiotic-resistant pathogens. Antimicrobial peptides (AMPs) are widely present in nature, with a broad range of effects and a low risk of causing drug resistance. Therefore, they are an ideal choice for the development of the next generation of antimicrobial drugs. To overcome the inefficiencies of traditional AMP discovery, artificial intelligence (AI) and machine learning (ML) technologies have been increasingly used to predict and design AMPs. Multiple AMP databases were used to train ML models for predicting the activity of AMPs or generating AMP sequences. This review briefly provides a comprehensive overview of AMP databases and computational tools, highlighting their capabilities and challenges. Future work should integrate larger datasets and experimental validation to accelerate clinical translation.
The global rise of antimicrobial resistance (AMR) has emerged as one of the most pressing threats to public health. This crisis calls for the urgent development of alternative therapeutic agents against antibiotic-resistant pathogens. Antimicrobial peptides (AMPs) are widely present in nature, with a broad range of effects and a low risk of causing drug resistance. Therefore, they are an ideal choice for the development of the next generation of antimicrobial drugs. To overcome the inefficiencies of traditional AMP discovery, artificial intelligence (AI) and machine learning (ML) technologies have been increasingly used to predict and design AMPs. Multiple AMP databases were used to train ML models for predicting the activity of AMPs or generating AMP sequences. This review briefly provides a comprehensive overview of AMP databases and computational tools, highlighting their capabilities and challenges. Future work should integrate larger datasets and experimental validation to accelerate clinical translation.
DOI: https://doi.org/10.37349/eds.2026.1008161
This article belongs to the special issue Discovery and development of new antibacterial compounds

The global rise in metabolic and non-communicable diseases (NCDs) has prompted an urgent search for preventive and complementary therapeutic approaches beyond conventional pharmacotherapy. Functional foods and nutraceuticals—dietary components and bioactive compounds with proven physiological benefits—represent a growing field of research within foodomics and nutritional sciences. These substances modulate oxidative stress, inflammation, lipid metabolism, glucose metabolism, and gut microbiota composition, offering potential in preventing and managing diseases such as obesity, type 2 diabetes, cardiovascular disorders, non-alcoholic fatty liver disease, and cancer. This review explores the mechanisms of action, and clinical implications of functional foods and nutraceuticals, providing a comprehensive overview of their bioactive constituents, molecular pathways, and translational potential. Challenges related to bioavailability, standardization, and regulatory recognition are also discussed, alongside perspectives for future development in foodomics-driven precision nutrition.
The global rise in metabolic and non-communicable diseases (NCDs) has prompted an urgent search for preventive and complementary therapeutic approaches beyond conventional pharmacotherapy. Functional foods and nutraceuticals—dietary components and bioactive compounds with proven physiological benefits—represent a growing field of research within foodomics and nutritional sciences. These substances modulate oxidative stress, inflammation, lipid metabolism, glucose metabolism, and gut microbiota composition, offering potential in preventing and managing diseases such as obesity, type 2 diabetes, cardiovascular disorders, non-alcoholic fatty liver disease, and cancer. This review explores the mechanisms of action, and clinical implications of functional foods and nutraceuticals, providing a comprehensive overview of their bioactive constituents, molecular pathways, and translational potential. Challenges related to bioavailability, standardization, and regulatory recognition are also discussed, alongside perspectives for future development in foodomics-driven precision nutrition.
DOI: https://doi.org/10.37349/eff.2026.1010157

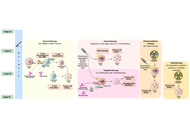
Autoimmune rheumatic diseases arise when the immune system transitions from a flexible, self-regulating network into a metabolically and epigenetically fixed inflammatory attractor state. This review synthesizes emerging evidence that immune tolerance is governed by a coupled epigenetic-metabolic axis integrating mitochondrial fitness, chromatin accessibility, redox balance, and nutrient flux across lymphoid, myeloid, and stromal compartments. We examine how chronic cytokine signaling, hypoxia, and oxidative stress destabilize regulatory programs, imprint glycolytic effector states, and remodel enhancer landscapes, thereby sustaining autoreactive circuits even after inflammatory pathways are pharmacologically suppressed. Multi-omic and spatial analyses reveal that pathogenic chromatin architectures, persistent mitochondrial dysfunction, and intercellular metabolite exchange cooperate to establish self-sustaining inflammatory ecosystems in rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), systemic sclerosis (SSc), and Sjögren’s syndrome. We further highlight therapeutic strategies aimed at tolerance reprogramming, including metabolic correction, chromatin-targeted agents, chimeric antigen receptor regulatory T cells (CAR-Tregs), tolerogenic dendritic cells, and integrative biomarkers that quantify metabolic-epigenetic coherence. By reframing autoimmunity as a disorder of energetic and chromatin desynchronization rather than isolated immune activation, this review outlines a mechanistic path toward durable, drug-free remission through deliberate restoration of the molecular architecture that maintains immune self-recognition.
Autoimmune rheumatic diseases arise when the immune system transitions from a flexible, self-regulating network into a metabolically and epigenetically fixed inflammatory attractor state. This review synthesizes emerging evidence that immune tolerance is governed by a coupled epigenetic-metabolic axis integrating mitochondrial fitness, chromatin accessibility, redox balance, and nutrient flux across lymphoid, myeloid, and stromal compartments. We examine how chronic cytokine signaling, hypoxia, and oxidative stress destabilize regulatory programs, imprint glycolytic effector states, and remodel enhancer landscapes, thereby sustaining autoreactive circuits even after inflammatory pathways are pharmacologically suppressed. Multi-omic and spatial analyses reveal that pathogenic chromatin architectures, persistent mitochondrial dysfunction, and intercellular metabolite exchange cooperate to establish self-sustaining inflammatory ecosystems in rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), systemic sclerosis (SSc), and Sjögren’s syndrome. We further highlight therapeutic strategies aimed at tolerance reprogramming, including metabolic correction, chromatin-targeted agents, chimeric antigen receptor regulatory T cells (CAR-Tregs), tolerogenic dendritic cells, and integrative biomarkers that quantify metabolic-epigenetic coherence. By reframing autoimmunity as a disorder of energetic and chromatin desynchronization rather than isolated immune activation, this review outlines a mechanistic path toward durable, drug-free remission through deliberate restoration of the molecular architecture that maintains immune self-recognition.
DOI: https://doi.org/10.37349/ei.2026.1003255

Aim:
Adult-onset Still’s disease (AOSD) is a rare systemic inflammatory disorder marked by fever, rash, joint pain, and hyperferritinemia. While immune dysregulation is implicated in AOSD, the exact causal mechanisms remain unclear. This study aimed to investigate the genetic causal relationship between 731 immune cell phenotypes and AOSD, and to identify protective or risk-associated profiles.
Methods:
Using a two-sample Mendelian randomization (TSMR) approach, we applied inverse variance weighted (IVW) as the primary method, supplemented by MR-Egger, weighted median, simple mode, and weighted mode methods for robustness. Genetic instrumental variables for immune traits were sourced from recent genome-wide association studies (GWAS), and AOSD genetic predispositions were derived from the finn-b-STILL_ADULT cohort, comprising 201,947 individuals of European ancestry (3,403 AOSD cases and 198,544 controls).
Results:
We identified 49 immune cell-related traits showing nominally significant associations with AOSD (all adjusted P > 0.05 after FDR correction). Among these, 34 traits showed nominally protective trends, while 15 showed nominally risk-associated trends. Reciprocally, AOSD showed nominally suggestive effects on 40 immune cell traits, with 25 exhibiting a trend toward decreased levels and 15 toward increased levels. Additionally, we conducted multiple sensitivity analyses to explore potential heterogeneity and pleiotropy, though the primary findings did not survive FDR correction.
Conclusions:
These nominally significant associations between immune cell traits and AOSD, though not surviving FDR correction, may offer hypothesis-generating insights for future therapeutic research. The observed directional trends—with certain traits showing nominally protective or risk-associated patterns—suggest potential avenues for further exploration in the development of targeted treatment approaches for AOSD.
Aim:
Adult-onset Still’s disease (AOSD) is a rare systemic inflammatory disorder marked by fever, rash, joint pain, and hyperferritinemia. While immune dysregulation is implicated in AOSD, the exact causal mechanisms remain unclear. This study aimed to investigate the genetic causal relationship between 731 immune cell phenotypes and AOSD, and to identify protective or risk-associated profiles.
Methods:
Using a two-sample Mendelian randomization (TSMR) approach, we applied inverse variance weighted (IVW) as the primary method, supplemented by MR-Egger, weighted median, simple mode, and weighted mode methods for robustness. Genetic instrumental variables for immune traits were sourced from recent genome-wide association studies (GWAS), and AOSD genetic predispositions were derived from the finn-b-STILL_ADULT cohort, comprising 201,947 individuals of European ancestry (3,403 AOSD cases and 198,544 controls).
Results:
We identified 49 immune cell-related traits showing nominally significant associations with AOSD (all adjusted P > 0.05 after FDR correction). Among these, 34 traits showed nominally protective trends, while 15 showed nominally risk-associated trends. Reciprocally, AOSD showed nominally suggestive effects on 40 immune cell traits, with 25 exhibiting a trend toward decreased levels and 15 toward increased levels. Additionally, we conducted multiple sensitivity analyses to explore potential heterogeneity and pleiotropy, though the primary findings did not survive FDR correction.
Conclusions:
These nominally significant associations between immune cell traits and AOSD, though not surviving FDR correction, may offer hypothesis-generating insights for future therapeutic research. The observed directional trends—with certain traits showing nominally protective or risk-associated patterns—suggest potential avenues for further exploration in the development of targeted treatment approaches for AOSD.
DOI: https://doi.org/10.37349/emed.2026.1001406

Tricuspid valve endocarditis has potential associations with various conditions, but it is commonly related to intravenous drug abuse. Often, its eradication can become very challenging due to high post-operative mortality and high rate of recurrence due to persistence of drug abuse habits. The bidirectional Glenn shunt (BGS), typically employed in congenital heart surgery, combined with tricuspid valvectomy, has occasionally been used for recurrent endocarditis. Herein, we present a 31-year-old woman with drug addiction scheduled for her fourth reintervention due to the early degeneration and infection of a previous pulmonary homograft used for recurring tricuspid valve endocarditis. The final surgical strategy was valvectomy and BGS, aimed at eliminating all potential sources of infection and providing more time for the patient to overcome drug addiction, enabling further treatment if necessary.
Tricuspid valve endocarditis has potential associations with various conditions, but it is commonly related to intravenous drug abuse. Often, its eradication can become very challenging due to high post-operative mortality and high rate of recurrence due to persistence of drug abuse habits. The bidirectional Glenn shunt (BGS), typically employed in congenital heart surgery, combined with tricuspid valvectomy, has occasionally been used for recurrent endocarditis. Herein, we present a 31-year-old woman with drug addiction scheduled for her fourth reintervention due to the early degeneration and infection of a previous pulmonary homograft used for recurring tricuspid valve endocarditis. The final surgical strategy was valvectomy and BGS, aimed at eliminating all potential sources of infection and providing more time for the patient to overcome drug addiction, enabling further treatment if necessary.
DOI: https://doi.org/10.37349/emed.2026.1001407

Aim:
This study aimed to develop a rice and sautéed mung bean meal using freeze-drying to preserve its safety, nutritional quality, and sensory attributes, providing a nutrient-dense meal suitable for disaster response and emergency feeding.
Methods:
White rice, mung beans, smoked herring, horseradish leaves, and seasonings were prepared, cooked, and freeze-dried. The freeze-dried product was vacuum-packed and evaluated for microbiological safety, physicochemical and proximate composition, micronutrient content (iron and vitamin A), and sensory acceptability by 50 Filipino panelists using a nine-point hedonic scale. Statistical comparisons with the traditional cooked meal were performed using paired t-tests.
Results:
The freeze-dried meal exhibited low microbial counts water activity, and moisture content, confirming its safety. Proximate analysis showed high protein (21.69 g/100 g), moderate carbohydrates (65.53 g/100 g), low fat (7.03 g/100 g), and total energy of 412.15 kcal/100 g. Micronutrient content per 117 g serving was 0.35 mg iron and 10.56 μg retinol equivalents vitamin A. Sensory evaluation revealed high acceptability for aroma, taste, and texture, while appearance and color showed minor reductions compared to the control, with statistically significant differences (p < 0.05) in some attributes.
Conclusions:
Freeze-drying effectively produced a safe, nutrient-rich, and sensorially acceptable instant rice and mung bean meal. The product demonstrates strong potential for long-shelf-life and is a convenient option for disaster response and emergency feeding, though further optimization may improve visual appeal.
Aim:
This study aimed to develop a rice and sautéed mung bean meal using freeze-drying to preserve its safety, nutritional quality, and sensory attributes, providing a nutrient-dense meal suitable for disaster response and emergency feeding.
Methods:
White rice, mung beans, smoked herring, horseradish leaves, and seasonings were prepared, cooked, and freeze-dried. The freeze-dried product was vacuum-packed and evaluated for microbiological safety, physicochemical and proximate composition, micronutrient content (iron and vitamin A), and sensory acceptability by 50 Filipino panelists using a nine-point hedonic scale. Statistical comparisons with the traditional cooked meal were performed using paired t-tests.
Results:
The freeze-dried meal exhibited low microbial counts water activity, and moisture content, confirming its safety. Proximate analysis showed high protein (21.69 g/100 g), moderate carbohydrates (65.53 g/100 g), low fat (7.03 g/100 g), and total energy of 412.15 kcal/100 g. Micronutrient content per 117 g serving was 0.35 mg iron and 10.56 μg retinol equivalents vitamin A. Sensory evaluation revealed high acceptability for aroma, taste, and texture, while appearance and color showed minor reductions compared to the control, with statistically significant differences (p < 0.05) in some attributes.
Conclusions:
Freeze-drying effectively produced a safe, nutrient-rich, and sensorially acceptable instant rice and mung bean meal. The product demonstrates strong potential for long-shelf-life and is a convenient option for disaster response and emergency feeding, though further optimization may improve visual appeal.
DOI: https://doi.org/10.37349/eff.2026.1010156

This Perspective explores why hyperbaric oxygen therapy (HBOT) deserves closer clinical and scientific attention as a possible treatment for ischemic stroke and a potential neuroprotective strategy in chronic central nervous system disorders, especially multiple sclerosis and other progressive or age-related neurological conditions. While HBOT is not yet widely accepted for these indications, three factors justify reconsideration: its strong safety profile, a biologically plausible mechanistic rationale, and recurrent signals of benefit from selected neurological studies. We review HBOT’s historical development, current accepted indications, and evidence that adverse effects are generally mild, uncommon, and reversible under modern protocols. In neurology, efficacy remains unproven, yet interest persists due to preliminary findings and repeated patient-reported improvements. A central argument is that the main barrier to progress is not safety, but evidence generation. Conventional randomized controlled trials face major challenges: difficult blinding, wide variation in dosing protocols, and uncertainty about meaningful outcomes in chronic neurological disease. Moreover, HBOT is non-patentable, which limits commercial investment and leaves a potentially valuable intervention underexplored. Mechanistically, we move beyond explanations centred solely on oxygen delivery or oxidative stress. As a working hypothesis, neurological benefits may partly arise from cumulative adaptive responses—including a rebound hormesis following repeated hyperoxic exposure. We conclude with a pragmatic research agenda: continuous, low-cost physiological monitoring in patients already receiving HBOT, coupled with a medium-term goal of adequately powered efficacy trials.
This Perspective explores why hyperbaric oxygen therapy (HBOT) deserves closer clinical and scientific attention as a possible treatment for ischemic stroke and a potential neuroprotective strategy in chronic central nervous system disorders, especially multiple sclerosis and other progressive or age-related neurological conditions. While HBOT is not yet widely accepted for these indications, three factors justify reconsideration: its strong safety profile, a biologically plausible mechanistic rationale, and recurrent signals of benefit from selected neurological studies. We review HBOT’s historical development, current accepted indications, and evidence that adverse effects are generally mild, uncommon, and reversible under modern protocols. In neurology, efficacy remains unproven, yet interest persists due to preliminary findings and repeated patient-reported improvements. A central argument is that the main barrier to progress is not safety, but evidence generation. Conventional randomized controlled trials face major challenges: difficult blinding, wide variation in dosing protocols, and uncertainty about meaningful outcomes in chronic neurological disease. Moreover, HBOT is non-patentable, which limits commercial investment and leaves a potentially valuable intervention underexplored. Mechanistically, we move beyond explanations centred solely on oxygen delivery or oxidative stress. As a working hypothesis, neurological benefits may partly arise from cumulative adaptive responses—including a rebound hormesis following repeated hyperoxic exposure. We conclude with a pragmatic research agenda: continuous, low-cost physiological monitoring in patients already receiving HBOT, coupled with a medium-term goal of adequately powered efficacy trials.
DOI: https://doi.org/10.37349/ent.2026.1004157

Aim:
This study focused on evaluating the bioaccessibility and antioxidant potential of the protein and phenolic compounds present in a beverage (
Methods:
The
Results:
After SGID, protein digestibility of
Conclusions:
This product demonstrated potential as an antioxidant functional beverage, which should be further confirmed by additional in vitro and in vivo assays. It also remains pending sensory and microbiological safety evaluations to obtain a final product that is both organoleptically acceptable and safe for consumption.
Aim:
This study focused on evaluating the bioaccessibility and antioxidant potential of the protein and phenolic compounds present in a beverage (
Methods:
The
Results:
After SGID, protein digestibility of
Conclusions:
This product demonstrated potential as an antioxidant functional beverage, which should be further confirmed by additional in vitro and in vivo assays. It also remains pending sensory and microbiological safety evaluations to obtain a final product that is both organoleptically acceptable and safe for consumption.
DOI: https://doi.org/10.37349/eff.2026.1010155
This article belongs to the special issue Natural Bioactive Compounds in Functional Foods: From Foodomics Insights to Clinical Relevance

Background:
Diabetes mellitus (DM) refers to a group of metabolic diseases characterized by reduced insulin synthesis and release, leading to hyperglycemia, oxidative stress, and inflammation. The prevalence of DM continues to increase worldwide, primarily driven by type 2 DM (T2DM). While numerous prescription drugs are widely available for the management of T2DM, these treatments must be coupled with significant changes in diet and lifestyle to have long-term effects. This comparative review aims to summarize and critically evaluate some of the dietary antioxidant and anti-inflammatory functional foods (FFs), including bioactive compounds on glycemic regulation, oxidative stress, inflammatory markers, and other outcomes in T2DM patients.
Methods:
A comprehensive literature search was performed from June 2014 to December 2025 in Cochrane Library, Embase, Google Scholar, PubMed/Medline, and Scopus databases.
Results:
Thirty-one meta-analyses describing the effects of specific FFs on glycemic outcomes, including fasting blood glucose, HbA1c, insulin levels, insulin resistance, oxidative or inflammatory stress, and/or the doses needed to achieve these endpoints, in T2DM patients were included. Comparative analyses of the data for green tea in T2DM showed significant antioxidant effects, but conflicting data for glycemic regulation, and no significant effect on inflammatory markers. Flaxseed products significantly reduced oxidative stress and HbA1c but had no anti-inflammatory effects. Both anthocyanins and resveratrol from fruits and vegetables significantly reduced fasting blood glucose, HbA1c, as well as oxidative and inflammatory markers. Turmeric and curcumin significantly reduced fasting blood glucose, but the data for ginger were conflicted. In T2DM patients, turmeric and ginger significantly reduced markers of oxidation and inflammation.
Discussion:
Overall, improvements in diet, including the incorporation of specific FFs and exercise, along with standard treatments, may be cost-effective methods to reduce the public health impact of T2DM. Limitations included the significant heterogeneity, high risk of bias, and low to moderate quality of the randomized controlled trial (RCT).
Background:
Diabetes mellitus (DM) refers to a group of metabolic diseases characterized by reduced insulin synthesis and release, leading to hyperglycemia, oxidative stress, and inflammation. The prevalence of DM continues to increase worldwide, primarily driven by type 2 DM (T2DM). While numerous prescription drugs are widely available for the management of T2DM, these treatments must be coupled with significant changes in diet and lifestyle to have long-term effects. This comparative review aims to summarize and critically evaluate some of the dietary antioxidant and anti-inflammatory functional foods (FFs), including bioactive compounds on glycemic regulation, oxidative stress, inflammatory markers, and other outcomes in T2DM patients.
Methods:
A comprehensive literature search was performed from June 2014 to December 2025 in Cochrane Library, Embase, Google Scholar, PubMed/Medline, and Scopus databases.
Results:
Thirty-one meta-analyses describing the effects of specific FFs on glycemic outcomes, including fasting blood glucose, HbA1c, insulin levels, insulin resistance, oxidative or inflammatory stress, and/or the doses needed to achieve these endpoints, in T2DM patients were included. Comparative analyses of the data for green tea in T2DM showed significant antioxidant effects, but conflicting data for glycemic regulation, and no significant effect on inflammatory markers. Flaxseed products significantly reduced oxidative stress and HbA1c but had no anti-inflammatory effects. Both anthocyanins and resveratrol from fruits and vegetables significantly reduced fasting blood glucose, HbA1c, as well as oxidative and inflammatory markers. Turmeric and curcumin significantly reduced fasting blood glucose, but the data for ginger were conflicted. In T2DM patients, turmeric and ginger significantly reduced markers of oxidation and inflammation.
Discussion:
Overall, improvements in diet, including the incorporation of specific FFs and exercise, along with standard treatments, may be cost-effective methods to reduce the public health impact of T2DM. Limitations included the significant heterogeneity, high risk of bias, and low to moderate quality of the randomized controlled trial (RCT).
DOI: https://doi.org/10.37349/eemd.2026.101474

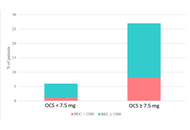
Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare systemic vasculitis with heterogeneous clinical manifestations. Identifying reliable biomarkers is crucial to predicting disease evolution and guiding therapy. We analyzed clinical, biological, and functional data from 33 patients with EGPA in the vasculitic phase. Blood eosinophil count (BEC), eosinophilic cationic protein (ECP), antineutrophil cytoplasmic antibodies (ANCAs), specific IgE to staphylococcal enterotoxins (SE-IgE), and serum free light chains (FLCs) were evaluated. Severe eosinophilic asthma (SEA) and chronic rhinosinusitis with nasal polyps (CRSwNPs) were present in 100% and 88.5% of patients, respectively. Median BEC was 1950 cells/mm3, with elevated ECP (60.0 µg/L). SE-IgE was detected in 54.2% of patients. A significant negative correlation emerged between λ FLCs and oral corticosteroid (OCS) dose (r = –0.58, p = 0.009). Forced expiratory volume in 1 second (FEV1) was significantly lower in C-ANCA+ patients (p = 0.006). ECP and SE-IgE may serve as markers of eosinophilic activity and epithelial barrier damage in EGPA. λ FLCs might be a useful indicator of OCS exposure and treatment response. These biomarkers could support the evaluation of disease evolution and treatment response.
Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare systemic vasculitis with heterogeneous clinical manifestations. Identifying reliable biomarkers is crucial to predicting disease evolution and guiding therapy. We analyzed clinical, biological, and functional data from 33 patients with EGPA in the vasculitic phase. Blood eosinophil count (BEC), eosinophilic cationic protein (ECP), antineutrophil cytoplasmic antibodies (ANCAs), specific IgE to staphylococcal enterotoxins (SE-IgE), and serum free light chains (FLCs) were evaluated. Severe eosinophilic asthma (SEA) and chronic rhinosinusitis with nasal polyps (CRSwNPs) were present in 100% and 88.5% of patients, respectively. Median BEC was 1950 cells/mm3, with elevated ECP (60.0 µg/L). SE-IgE was detected in 54.2% of patients. A significant negative correlation emerged between λ FLCs and oral corticosteroid (OCS) dose (r = –0.58, p = 0.009). Forced expiratory volume in 1 second (FEV1) was significantly lower in C-ANCA+ patients (p = 0.006). ECP and SE-IgE may serve as markers of eosinophilic activity and epithelial barrier damage in EGPA. λ FLCs might be a useful indicator of OCS exposure and treatment response. These biomarkers could support the evaluation of disease evolution and treatment response.
DOI: https://doi.org/10.37349/eaa.2026.1009127
This article belongs to the special issue Environment, Infectious Diseases, and Allergy

Aim:
Two empirical methods were used to predict the absolute bioavailability (F) of medicines in adults and children following oral administration in the absence of intravenous (IV) dosing. This study systematically evaluates the predictive performance of Equation 1 to predict F in adults and children.
Methods:
Equation 3 [F = Q/(Q + CLoral)] was used for the prediction of F in adults and children. In Equation 3, clearance is the observed oral clearance following oral administration of a medicine and Q is either liver blood or plasma flow rate. The predictive performance of Equation 3 was evaluated in adults and children for three categories of medicines; medicines which are mainly metabolized in the liver, medicines which are metabolized both in the liver and the gut, and medicines which are mainly renally excreted. From the literature, oral clearance and F values for adults and children were obtained. The predictive performance of these two methods (blood or plasma flow rate) was assessed by comparing the predicted F of the medicines used in this study with the observed F (obtained from clinical studies).
Results:
More than 90% predicted F values were within 0.5–2-fold prediction error in adults and children by both methods for all three categories of medicines. Plasma flow rate provided slightly better results than the blood flow rate.
Conclusions:
The proposed methods indicate that the estimation of F of medicines in adults and children is possible with reasonable accuracy (within 0.5–2-fold prediction error). The method is useful to estimate F, especially in children, because it is not ethical to administer medicines by both IV and oral routes to children just for the sake of estimating F.
Aim:
Two empirical methods were used to predict the absolute bioavailability (F) of medicines in adults and children following oral administration in the absence of intravenous (IV) dosing. This study systematically evaluates the predictive performance of Equation 1 to predict F in adults and children.
Methods:
Equation 3 [F = Q/(Q + CLoral)] was used for the prediction of F in adults and children. In Equation 3, clearance is the observed oral clearance following oral administration of a medicine and Q is either liver blood or plasma flow rate. The predictive performance of Equation 3 was evaluated in adults and children for three categories of medicines; medicines which are mainly metabolized in the liver, medicines which are metabolized both in the liver and the gut, and medicines which are mainly renally excreted. From the literature, oral clearance and F values for adults and children were obtained. The predictive performance of these two methods (blood or plasma flow rate) was assessed by comparing the predicted F of the medicines used in this study with the observed F (obtained from clinical studies).
Results:
More than 90% predicted F values were within 0.5–2-fold prediction error in adults and children by both methods for all three categories of medicines. Plasma flow rate provided slightly better results than the blood flow rate.
Conclusions:
The proposed methods indicate that the estimation of F of medicines in adults and children is possible with reasonable accuracy (within 0.5–2-fold prediction error). The method is useful to estimate F, especially in children, because it is not ethical to administer medicines by both IV and oral routes to children just for the sake of estimating F.
DOI: https://doi.org/10.37349/eds.2026.1008160

Immunotherapy is a promising treatment strategy for treating colorectal cancer (CRC). Despite significant advances in this field, resistance and low efficacy of immunotherapy remain a principal problem. One of the most important factors affecting the response to immunotherapy is the tumor microenvironment (TME). Among the components of the TME, tumor-associated macrophages (TAMs) are key immune cells involved in cancer progression by stimulating tumor cell proliferation, angiogenesis, epithelial-mesenchymal transition, metastasis, and tumor immune evasion. This review presents currently investigated combination therapy based on the immune checkpoint inhibitors and inhibitors of diverse components of the TME, including TAMs, that can potentially increase the effectiveness of CRC treatment. Therapeutic efficacy, together with the functional activity of TAMs, is estimated in multiple preclinical data obtained with diverse in vitro and in vivo models. Ongoing clinical trials demonstrated the association of treatment effectiveness with TAM phenotypes and functions.
Immunotherapy is a promising treatment strategy for treating colorectal cancer (CRC). Despite significant advances in this field, resistance and low efficacy of immunotherapy remain a principal problem. One of the most important factors affecting the response to immunotherapy is the tumor microenvironment (TME). Among the components of the TME, tumor-associated macrophages (TAMs) are key immune cells involved in cancer progression by stimulating tumor cell proliferation, angiogenesis, epithelial-mesenchymal transition, metastasis, and tumor immune evasion. This review presents currently investigated combination therapy based on the immune checkpoint inhibitors and inhibitors of diverse components of the TME, including TAMs, that can potentially increase the effectiveness of CRC treatment. Therapeutic efficacy, together with the functional activity of TAMs, is estimated in multiple preclinical data obtained with diverse in vitro and in vivo models. Ongoing clinical trials demonstrated the association of treatment effectiveness with TAM phenotypes and functions.
DOI: https://doi.org/10.37349/edd.2026.1005124
This article belongs to the special issue Immunotherapy for Cancer of Digestive System

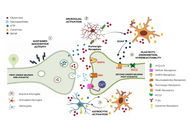
The intricate involvement of glial cells in chronic pain mechanisms represents a paradigm shift in our understanding of pain processing. From microglial-mediated neuroinflammation to astrocytic modulation of synaptic function, glial cells emerge as critical players in the complex neurobiology of chronic pain. As research continues to unravel the multifaceted roles of these cells, novel therapeutic targets and strategies are likely to emerge, potentially revolutionizing the management of chronic pain conditions. Chronic pain is complicated by frequent comorbidities such as fatigue, sleep disturbances, cognitive impairment, and mood disorders. Here, we hypothesize that neuroinflammatory processes are at the root of pain chronification and serve as the common thread linking characteristic hypersensitivity with the cognitive and emotional comorbidities typically associated with chronic pain.
The intricate involvement of glial cells in chronic pain mechanisms represents a paradigm shift in our understanding of pain processing. From microglial-mediated neuroinflammation to astrocytic modulation of synaptic function, glial cells emerge as critical players in the complex neurobiology of chronic pain. As research continues to unravel the multifaceted roles of these cells, novel therapeutic targets and strategies are likely to emerge, potentially revolutionizing the management of chronic pain conditions. Chronic pain is complicated by frequent comorbidities such as fatigue, sleep disturbances, cognitive impairment, and mood disorders. Here, we hypothesize that neuroinflammatory processes are at the root of pain chronification and serve as the common thread linking characteristic hypersensitivity with the cognitive and emotional comorbidities typically associated with chronic pain.
DOI: https://doi.org/10.37349/ei.2026.1003254
This article belongs to the special issue Immunology and Pain

Aim:
To assess the effect of a synbiotic supplement composed of Enterococcus faecium and agave inulin on cognitive function in older adults with mild cognitive impairment (MCI).
Methods:
In a triple-blind randomized crossover trial, nineteen adults aged 64–85 years with MCI received either the synbiotic or an isocaloric vehicle for eight weeks, followed by a three-week washout and treatment crossover. Cognitive outcomes were assessed at baseline, after the first intervention, and after crossover using the Modified Mini-Mental State Examination (MMSE), Rey-Osterrieth Complex Figure Test (RCFT; copy and memory), and Abbreviated Instrument for Expectations of Self-Efficacy for Daily Activities in Older Adults (AERAC) self-efficacy scale. Paired and unpaired Student’s t-tests were used for statistical comparisons (p < 0.05).
Results:
The synbiotic group showed significant improvement relative to baseline across all domains: MMSE (p = 0.05), AERAC (p = 0.005), RCFT-copy (p = 0.03), and RCFT-memory (p = 0.03). Post-treatment comparisons between groups also favored the synbiotic, with significant differences in MMSE (p = 0.001), AERAC (p = 0.001), RCFT-copy (p = 0.0095), and RCFT-memory (p = 0.001). After crossover, cognitive gains were sustained and reproduced. MMSE scores reached 17.89 ± 1.45 in the synbiotic-first group versus 18.20 ± 0.63 in the control-first group (p < 0.001). RCFT-copy remained high (29.83 ± 4.18 vs. 29.52 ± 5.60, p = 0.0157), while RCFT-memory scores differed (17.56 ± 6.73 vs. 17.20 ± 3.29, p = 0.0005). AERAC scores continued to improve during crossover (82.60 ± 10.49 vs. 85.46 ± 8.28, p < 0.001). No adverse effects occurred.
Conclusions:
Synbiotic supplementation significantly improved global cognition, visuoconstructive ability, memory, and functional self-efficacy in older adults with MCI. Benefits persisted beyond the initial intervention and were replicated when the control group received the synbiotic, supporting its potential as a safe and effective strategy to mitigate age-related cognitive decline.
Aim:
To assess the effect of a synbiotic supplement composed of Enterococcus faecium and agave inulin on cognitive function in older adults with mild cognitive impairment (MCI).
Methods:
In a triple-blind randomized crossover trial, nineteen adults aged 64–85 years with MCI received either the synbiotic or an isocaloric vehicle for eight weeks, followed by a three-week washout and treatment crossover. Cognitive outcomes were assessed at baseline, after the first intervention, and after crossover using the Modified Mini-Mental State Examination (MMSE), Rey-Osterrieth Complex Figure Test (RCFT; copy and memory), and Abbreviated Instrument for Expectations of Self-Efficacy for Daily Activities in Older Adults (AERAC) self-efficacy scale. Paired and unpaired Student’s t-tests were used for statistical comparisons (p < 0.05).
Results:
The synbiotic group showed significant improvement relative to baseline across all domains: MMSE (p = 0.05), AERAC (p = 0.005), RCFT-copy (p = 0.03), and RCFT-memory (p = 0.03). Post-treatment comparisons between groups also favored the synbiotic, with significant differences in MMSE (p = 0.001), AERAC (p = 0.001), RCFT-copy (p = 0.0095), and RCFT-memory (p = 0.001). After crossover, cognitive gains were sustained and reproduced. MMSE scores reached 17.89 ± 1.45 in the synbiotic-first group versus 18.20 ± 0.63 in the control-first group (p < 0.001). RCFT-copy remained high (29.83 ± 4.18 vs. 29.52 ± 5.60, p = 0.0157), while RCFT-memory scores differed (17.56 ± 6.73 vs. 17.20 ± 3.29, p = 0.0005). AERAC scores continued to improve during crossover (82.60 ± 10.49 vs. 85.46 ± 8.28, p < 0.001). No adverse effects occurred.
Conclusions:
Synbiotic supplementation significantly improved global cognition, visuoconstructive ability, memory, and functional self-efficacy in older adults with MCI. Benefits persisted beyond the initial intervention and were replicated when the control group received the synbiotic, supporting its potential as a safe and effective strategy to mitigate age-related cognitive decline.
DOI: https://doi.org/10.37349/ent.2026.1004156
This article belongs to the special issue Role of Microbiota in Neurological Diseases

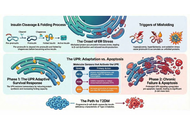
Endocrine resistance in estrogen receptor-positive (ER+) breast cancer has undergone a fundamental reconceptualization over the past decade. The discovery that activating mutations in the ESR1 gene encoding ERα emerge under aromatase inhibitor (AI) selection pressure and drive ligand-independent receptor activation established a shift from empirical treatment sequencing to molecularly guided intervention. This review provides a synopsis of the structural biology underlying constitutive ER activation, the evolutionary dynamics of ESR1-mutant clones detectable through circulating tumor DNA (ctDNA), and the clinical evidence demonstrating that early molecular detection can trigger therapeutic switches that alter disease trajectory. The regulatory approval of elacestrant for ESR1-mutant disease and randomized trial data showing progression-free survival (PFS) benefit from ctDNA-guided endocrine switching (PADA-1, SERENA-6) position ESR1 genotyping as a dynamic biomarker with direct therapeutic implications. We examine the integration of oral selective ER degraders (SERDs) into treatment algorithms, the role of co-occurring alterations in the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway, and emerging directions, including machine learning approaches to ctDNA kinetics and adaptive trial designs that treat clonal evolution as an actionable target. The convergence of structural mechanisms, liquid biopsy technology, and biomarker-driven drug development provides a framework for precision oncology in endocrine-resistant breast cancer. While these advances are substantial, important challenges remain, including the lack of mature overall survival (OS) data from interception trials, cost and accessibility barriers to serial ctDNA monitoring in diverse global healthcare settings, the unresolved question of optimal therapeutic sequencing in patients with concurrent ESR1 and PI3K pathway alterations, and the need to distinguish clinically actionable low-variant allele frequency (VAF) ESR1 calls from background noise in liquid biopsies.
Endocrine resistance in estrogen receptor-positive (ER+) breast cancer has undergone a fundamental reconceptualization over the past decade. The discovery that activating mutations in the ESR1 gene encoding ERα emerge under aromatase inhibitor (AI) selection pressure and drive ligand-independent receptor activation established a shift from empirical treatment sequencing to molecularly guided intervention. This review provides a synopsis of the structural biology underlying constitutive ER activation, the evolutionary dynamics of ESR1-mutant clones detectable through circulating tumor DNA (ctDNA), and the clinical evidence demonstrating that early molecular detection can trigger therapeutic switches that alter disease trajectory. The regulatory approval of elacestrant for ESR1-mutant disease and randomized trial data showing progression-free survival (PFS) benefit from ctDNA-guided endocrine switching (PADA-1, SERENA-6) position ESR1 genotyping as a dynamic biomarker with direct therapeutic implications. We examine the integration of oral selective ER degraders (SERDs) into treatment algorithms, the role of co-occurring alterations in the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway, and emerging directions, including machine learning approaches to ctDNA kinetics and adaptive trial designs that treat clonal evolution as an actionable target. The convergence of structural mechanisms, liquid biopsy technology, and biomarker-driven drug development provides a framework for precision oncology in endocrine-resistant breast cancer. While these advances are substantial, important challenges remain, including the lack of mature overall survival (OS) data from interception trials, cost and accessibility barriers to serial ctDNA monitoring in diverse global healthcare settings, the unresolved question of optimal therapeutic sequencing in patients with concurrent ESR1 and PI3K pathway alterations, and the need to distinguish clinically actionable low-variant allele frequency (VAF) ESR1 calls from background noise in liquid biopsies.
DOI: https://doi.org/10.37349/etat.2026.1002375
This article belongs to the special issue Breaking Boundaries in Breast Cancer Care: Emerging Controversies and Innovation in Surgical and Medical Approaches
 Previous
Previous