Calcium Signaling Apparatus in Cancers
Guest Editor
Dr. Zui Pan E-Mail
College of Nursing and Health Innovation, The University of Texas at Arlington, TX, USA
Research Keywords: Esophageal cancer; calcium signaling; store-operated calcium entry; live cell imaging; chemotherapy
About the Special lssue
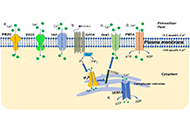
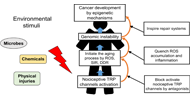
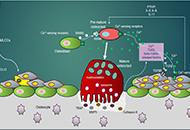
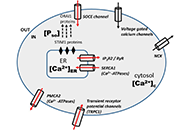
Dysregulated
intracellular Ca2+ signaling is associated with the development of
malignant phenotypes. Cancer cells remodel their Ca2+ signaling
apparatus to enhance proliferation, to increase cell motility and
invasion, to resist apoptosis, to escape from immune-attack, or to have
neovascularization. There has been an increasing awareness that
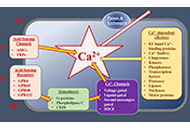
tumorigenic pathways are associated with altered expression or abnormal
activation of Ca2+ channels, transporters or Ca2+-pumps. Targeting the
altered Ca2+ signaling apparatus to develop new anti-cancer drugs is
emerging, yet many challenges remains. Intracellular Ca2+ homeostasis
plays important role in normal physiology and cell functions; thus, it
is important to identify the cancer specific properties of the Ca2+ apparatus. Different cancers or different stages during tumor
development may present distinct different alteration in Ca2+ signaling
apparatus. More specific, more potent and less off-target compounds
targeting Ca2+ channels/transporters/pumps are urgently needed.
This
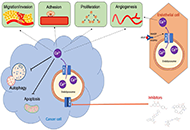
special issue will highlight the current state of diverse molecular
mechanisms of intracellular Ca2+ involved in cell proliferation,
apoptosis, migration, invasion, or angiogenesis in different cancers. It
will also discuss developing effective novel cancer treatment targeting
Ca2+ channels, transporters or Ca2+-pumps.
Keywords: Calcium channels; cell proliferation; apoptosis; migration; metastasis; transcription factors; chemotherapy
Published Articles