Liquid Biopsy in Thoracic Cancer
Guest Editors
Dr. Floriana Morgillo E-Mail
Medical Oncology, University of Campania “Luigi Vanvitelli”, Naples, Italy
Research Keywords: lung cancer; targeted therapy; immunotherapy; translational research; resistance mechnisms
Dr. Carminia Maria Della Corte E-Mail
Medical Oncology, University of Campania “Luigi Vanvitelli”, Naples, Italy
About the Special lssue
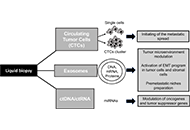
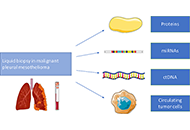
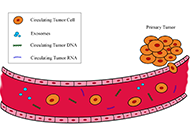
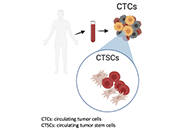
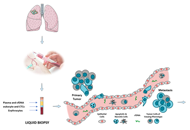
“Liquid biopsy” of biological fluids (e.g., plasma and serum, urine, saliva, cerebrospinal fluid, pleural fluid, ascites, stool) is emerging as a powerful tool for non-invasive diagnosis, screening, prognosis, and stratification of cancer patients. This approach is based on the fact that tumor cells release molecules (proteins, DNA, RNA), circulating tumor cells (CTC), and extra cellular vesicles (EV) that can be used as biomarkers.
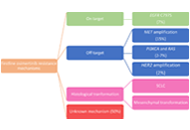
Considerable attention has been devoted to the analysis of EGFR mutations in patients with NSCLC, as it is often challenging to obtain tissue biopsies from these patients to help to inform treatment. At present, the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA) approve the use of information from ctDNA analysis to help to select patients with EGFR-mutant NSCLC for anti-EGFR therapy in the event that a tumour sample is not evaluable. This could offer a pragmatic solution that provides molecular profiling information for patients while avoiding repeat biopsies for some individuals.
Further studies are ongoing to evaluate the role of this approach for small cell lung cancer and malignant mesothelioma.
Liquid biopsies may be repeated frequently to provide a more detailed picture of the natural history of the disease with a longitudinal assessment of tumor burden and clues about clonal evolution and emergence of drug- resistant clones leading to clinical relapse, thus implementing our understanding of disease evolution and management.
Keywords: CTC; early diagnosis; tumor heterogeneity; biomarkers; therapeutic resistance
Published Articles