409 results in Exploration of Medicine
Latest
Sort by :
- Latest
- Most Viewed
- Most Downloaded
- Most Cited
Open Access
Review
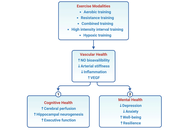
Integrating vascular, cognitive, and mental health benefits of exercise in obese women across menopause: a review
Jiwoong Jang, Hun-Young Park
Published: June 03, 2026 Explor Med. 2026;7:1001409
Open Access
Review
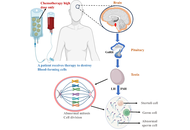
Beyond survival: how paediatric and adolescent chemotherapy shapes male fertility in adulthood
Vickram Agaram Sundaram ... Hitesh Chopra
Published: June 03, 2026 Explor Med. 2026;7:1001408
Open Access
Case Report
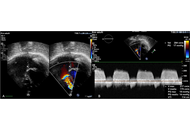
Life-saving alternatives: bidirectional Glenn shunt after pulmonary homograft for relapsing tricuspid endocarditis
Vincenzo Boschetti ... Giovanni Battista Luciani
Published: May 28, 2026 Explor Med. 2026;7:1001407
Open Access
Original Article
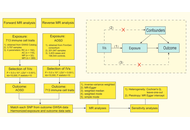
Bidirectional Mendelian randomization exploring the causal relationship between immune cell dynamics and adult-onset Still’s disease
Jing-Lei Xie ... Sheng-Xiao Zhang
Published: May 28, 2026 Explor Med. 2026;7:1001406
Open Access
Review
USE of ultrasounds as diagnostic tool in periodontics: a scoping review
Davide Gerardi ... Sara Bernardi
Published: May 25, 2026 Explor Med. 2026;7:1001405
Open Access
Review
Artificial intelligence strategies for emotion recognition in cancer pain research
Marco Cascella ... Giuseppe Polese
Published: May 19, 2026 Explor Med. 2026;7:1001404
This article belongs to the special issue Innovative Approaches to Chronic Pain Management: from Multidisciplinary Strategies to Artificial Intelligence Perspectives

Open Access
Original Article
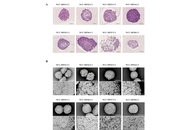
Spheroid versus monolayer culture: proteomic insights into divergent pathways in myxofibrosarcoma
Yomogi Shiota ... Yuki Yoshimatsu
Published: May 17, 2026 Explor Med. 2026;7:1001403
Open Access
Original Article
Associations between self-reported adherence to antihypertensive medications and potential mechanisms of behavior change in a general outpatient sample
Mark J. Butler ... Ashley M. Goodwin
Published: May 11, 2026 Explor Med. 2026;7:1001402
This article belongs to the special issue Drug Adherence in Hypertension

Open Access
Case Report
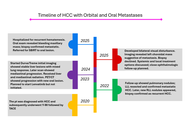
Unusual dual metastasis of hepatocellular carcinoma to the orbit and oral cavity: a case report and literature review
Saifudeen Abdelrahim ... Abdullah Esmail
Published: May 09, 2026 Explor Med. 2026;7:1001401
Open Access
Systematic Review
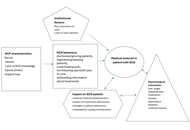
Medical mistrust and sickle cell disease: a systematic review
Meghna Shukla ... Ramona Benkert
Published: April 20, 2026 Explor Med. 2026;7:1001400
Open Access
Review
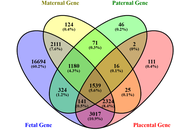
Integration of biomedical imaging and sensing technologies with AI-IoT for Materiovigilance and predictive modeling of stillbirth risk in maternal-fetal health monitoring
Atreyi Pramanik ... Prakash Gangadaran
Published: April 20, 2026 Explor Med. 2026;7:1001399
Open Access
Original Article
Associations of polypharmacy with cognitive impairment and functional status among older adults
Mi-Kyoung Jun, Hye-Min Ku
Published: April 13, 2026 Explor Med. 2026;7:1001398

Open Access
Perspective
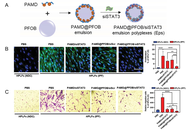
Nanodelivery systems-empowered therapies of idiopathic pulmonary fibrosis
Meihong Zhang ... Zhengwei Huang
Published: April 13, 2026 Explor Med. 2026;7:1001397
Open Access
Systematic Review
Myofunctional therapy in sleep disorders: a systematic review
Sabina Saccomanno ... Angelo Michele Inchingolo
Published: April 01, 2026 Explor Med. 2026;7:1001396

Open Access
Perspective
The objective imperative: advancing chronic orofacial pain research with the brain-heart axis
Takahiko Nagamine
Published: March 27, 2026 Explor Med. 2026;7:1001395

Open Access
Original Article
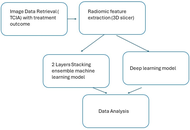
Prognostic prediction of head and neck cancer through radiomics: a stacking ensemble approach with machine learning and deep learning models
Heylie YT Wong ... Sarah WY Lee
Published: March 26, 2026 Explor Med. 2026;7:1001394
This article belongs to the special issue Artificial Intelligence in Precision Imaging: Innovations Shaping the Future of Clinical Diagnostics
Open Access
Review
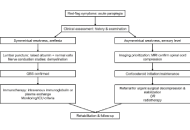
Paraplegia in the emergency room: spinal cord compression and Guillain-Barré syndrome—a 2025 scoping review from a multidisciplinary emergency perspective
Omar Alqaisi ... Aliaa Ezz Eldin Abd Elmoaty
Published: March 25, 2026 Explor Med. 2026;7:1001393
This article belongs to the special issue Practical Tips for Cancer Care: Guidance for Patients, Caregivers, and Healthcare Professionals
Open Access
Review
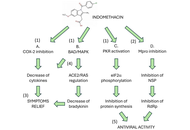
Indomethacin, a drug with known antiviral activity against SARS-CoVs neglected during the COVID-19 pandemic
Serafino Fazio ... Paolo Bellavite
Published: March 24, 2026 Explor Med. 2026;7:1001392
Open Access
Retraction
Retraction: An overview of current strategies and future prospects in drug repurposing in tuberculosis
Editorial Office
Published: March 24, 2026 Explor Med. 2026;7:1001391

Open Access
Perspective
Treatment of hypertension in Norway in relation to new European hypertension guidelines
Sverre E. Kjeldsen ... Tor Ole Klemsdal
Published: March 17, 2026 Explor Med. 2026;7:1001390
This article belongs to the special issue Drug Adherence in Hypertension

 Previous
Previous