Affiliation:
1State Key Laboratory of Bioactive Molecules and Druggability Assessment, Guangdong Basic Research Center of Excellence for Natural Bioactive Molecules and Discovery of Innovative Drugs, College of Pharmacy, Jinan University, Guangzhou 511443, Guangdong, China
ORCID: https://orcid.org/0009-0003-6728-4466
Affiliation:
1State Key Laboratory of Bioactive Molecules and Druggability Assessment, Guangdong Basic Research Center of Excellence for Natural Bioactive Molecules and Discovery of Innovative Drugs, College of Pharmacy, Jinan University, Guangzhou 511443, Guangdong, China
ORCID: https://orcid.org/0009-0002-2242-6857
Affiliation:
1State Key Laboratory of Bioactive Molecules and Druggability Assessment, Guangdong Basic Research Center of Excellence for Natural Bioactive Molecules and Discovery of Innovative Drugs, College of Pharmacy, Jinan University, Guangzhou 511443, Guangdong, China
2Jiangmen Wuyi Hospital of Traditional Chinese Medicine, Affiliated Jiangmen Traditional Chinese Medicine Hospital of Jinan University, Jiangmen 529031, Guangdong, China
ORCID: https://orcid.org/0000-0003-1661-0201
Affiliation:
1State Key Laboratory of Bioactive Molecules and Druggability Assessment, Guangdong Basic Research Center of Excellence for Natural Bioactive Molecules and Discovery of Innovative Drugs, College of Pharmacy, Jinan University, Guangzhou 511443, Guangdong, China
3Henan Key Laboratory of Biomarker Detection and Diagnosis for Neurodegenerative Diseases, School of Chemistry and Chemical Engineering, Shangqiu Normal University, Shangqiu 476000, Henan, China
Email: huangzhengw@jnu.edu.cn
ORCID: https://orcid.org/0000-0003-2351-7347
Explor Med. 2026;7:1001397 DOI: https://doi.org/10.37349/emed.2026.1001397
Received: January 19, 2026 Accepted: March 22, 2026 Published: April 13, 2026
Academic Editor: Chao Wang, Soochow University, China
Idiopathic pulmonary fibrosis (IPF) is a progressive, fatal interstitial lung disease of undefined etiology. In recent years, its global incidence has shown an upward trend, with a median survival of approximately 3–5 years after diagnosis. Currently, clinical treatment outcomes for this disease remain limited. Approved therapeutic agents are nintedanib, pirfenidone, and nerandomilast, all of which are predominantly administered orally. The oral route renders drugs susceptible to degradation in the gastrointestinal tract, leading to reduced drug bioavailability and limited therapeutic efficacy. In this work, we believe that nanodelivery systems (NDSs) represent a promising approach to address the limitations of traditional therapies. Most importantly, we emphasized that inhalation NDSs should be prioritized to enhance the accumulation efficiency of targeted drugs at pulmonary lesion sites. Collectively, this study aims to provide insights into the developmental prospects of NDSs for IPF, paving the way for more efficient and personalized therapeutic approaches to enhance treatment efficacy while minimizing side effects.
Idiopathic pulmonary fibrosis (IPF) is a progressive, fatal interstitial lung disease with an undefined etiology and minimal treatment options [1]. The incidence and severity of the disease increase with age, with the elderly population being the most affected group [2]. Although the precise etiology of IPF remains incompletely understood, current research confirms that multiple risk factors, including genetic predisposition, environmental exposures, gastroesophageal reflux, aspiration of small amounts of fluid, age, gender, smoking, and infections, collectively contribute to its pathogenesis [2]. The typical pathological features of the disease are characterized by abnormal proliferation of fibroblasts and excessive deposition of extracellular matrix, a pathological process leading to destruction of lung tissue architecture and progressive loss of pulmonary function. IPF carries an inferior clinical prognosis, with a median survival of only 3–5 years after diagnosis. It is the most common and challenging disease among interstitial lung diseases [3].
Currently, clinical treatment options for IPF remain extremely limited, with merely three drugs approved worldwide for clinical intervention. These are nintedanib [4], pirfenidone [5], and nerandomilast [6]. Specifically, nintedanib, as a multi-targeted tyrosine kinase inhibitor, blocks the fibrosis process by inhibiting essential signaling pathways mediated by vascular endothelial growth factor receptors (VEGFRs), platelet-derived growth factor receptors (PDGFRs), and fibroblast growth factor receptors (FGFRs) [7]. Pirfenidone achieves the anti-fibrotic effect by inhibiting transforming growth factor-β (TGF-β)-stimulated collagen production, as well as inhibition of fibroblast activation [5, 8]. Nerandomilast, a first-in-class small-molecule agent, targets a fibrotic signaling cascade, offering new hope in this stagnant therapeutic field [9]. The aforementioned drugs can effectively improve lung function and functional capacity in patients with different types of IPF [10–12]. Compared with the placebo group, nintedanib reduced the rate of decline in forced vital capacity (FVC) by 51.0% over 52 weeks [13]. Pirfenidone treatment significantly improved patient prognosis, as the 3-month survival rate was significantly higher in the pirfenidone group than in the control group (55% vs. 34%, P = 0.042) [14]. Furthermore, the probability of FVC decline ≥ 10% or death within 12 months was significantly lower in the pirfenidone group than in the placebo group [15]. Nerandomilast also exerted a significant protective effect on pulmonary function, slowing the rate of FVC decline by approximately 37.5% over 52 weeks [6]. Notably, the combination therapy of pirfenidone plus nintedanib significantly delayed FVC decline, indicating that this combined regimen can effectively inhibit the progression of IPF [16].
Although these drugs can reduce scar formation and delay the progression of IPF, none can achieve a genuine cure for the disease. Moreover, these drugs are primarily administered orally and are susceptible to degradation in the gastrointestinal tract. This significantly reduces their bioavailability and severely limits their therapeutic efficacy [5]. Most critically, nintedanib readily induces severe adverse reactions such as diarrhea and nausea. This is primarily attributed to its extremely low water solubility, merely 0.0309 mg/mL, and unstable gastrointestinal absorption; meanwhile, pirfenidone is constrained by an excessively short half-life of only 2.4 h, leading to rapid renal clearance after absorption [17]. Furthermore, in clinical practice across multiple countries, glucocorticoids have also been used to treat IPF. However, their therapeutic efficacy remains limited, failing to significantly improve patient prognosis [18]. Collectively, the current clinical treatment landscape for IPF remains unsatisfactory.
Since the lungs possess an enormous surface area, they serve as an ideal organ for achieving targeted drug delivery via nanodelivery systems (NDSs). Developing highly efficient, low-toxicity drug delivery strategies using nanocarriers has become a research hotspot in the field of IPF treatment. The core advantage of NDSs lies in their ability to significantly improve the pharmacokinetic properties of drugs. On one hand, encapsulating poorly soluble drugs enhances their water solubility and stability, thereby increasing bioavailability. On the other hand, their suitable particle size and surface modifications enable passive or active targeting, reducing drug accumulation in non-target organs such as the liver and kidneys, and consequently effectively lowering systemic toxic side effects. Currently, widely explored nanocarriers in IPF treatment research include lipid-based nanoparticles, polymeric nanoparticles, nanomicelles, protein nanoparticles, metal nanoparticles, and metal-organic frameworks (MOFs) [19]. Kaur and coworkers [20] developed nintedanib-loaded solid lipid nanoparticles (NIN-SLN) to improve the oral bioavailability and therapeutic potential, and pharmacokinetic studies indicated that NIN-SLN increased the bioavailability of nintedanib by 2.87-fold compared to free nintedanib, while also enhancing nintedanib concentrations in lung tissue. To achieve targeted drug delivery, Zhao et al. [8] constructed polyethylene glycol-modified (PEGylated) bovine serum albumin (BSA) protein nanoparticles for loading pirfenidone and targeting the treatment of IPF. Surface-modified with the fibroblast-targeting peptide FnBAP5, these nanoparticles significantly increased drug accumulation in the lungs by approximately 5-fold. Concurrently, PEGylation enhanced serum pirfenidone concentrations by 4-fold and prolonged drug action time, ultimately leading to improved therapeutic outcomes.
Beyond delivering traditional small-molecule drugs, NDSs demonstrate unique advantages in the targeted delivery of nucleic acid therapeutics, offering novel pathways for molecular-level intervention in IPF. Commonly used nucleic acid drugs include antisense oligonucleotides, mRNA, miRNA, siRNA, and saRNA [19]. The pathogenesis of IPF involves multiple complex pathways, including fibroblast activation, excessive extracellular matrix deposition, dysregulated inflammatory responses, and oxidative stress imbalance. These pathological processes are all regulated by the transcription and expression of key genes. By utilizing nanocarriers to deliver nucleic acid therapeutics, precise intervention at the genetic level can be achieved to target these pathogenic pathways. Studies demonstrate that targeted delivery of nucleic acid therapeutics against pivotal regulatory molecules like heat shock protein 47 (HSP47), Plekhf1, erythrocyte 2-associated factor-2 (pNrf2), or interleukin‑11 (IL-11) effectively suppresses collagen synthesis, mitigates inflammatory responses, and restores redox homeostasis, thereby delaying or even reversing the progression of IPF [19, 21]. Among various delivery systems, lipid nanoparticles, particularly cationic lipid delivery systems, stand out. Their positively charged lipid components efficiently assemble with negatively charged RNA molecules through electrostatic interactions, forming stable complexes. This significantly enhances nucleic acid drug loading efficiency and serum stability, ensuring effective gene silencing or expression regulation within target cells. For example, the cationic lipid nanoparticles constructed by Chen et al. [22] enabled efficient loading and stable assembly of siIL11 through electrostatic interactions. Following effective delivery of siIL11 to target cells, these NDSs specifically silenced IL-11 expression. By inhibiting the IL-11RA/gp130 signaling axis and its downstream ERK and AKT pathways, it exerted a significant anti-fibrotic effect. Furthermore, cationic polymers are frequently employed for nucleic acid delivery systems in non-lipid-based delivery systems. Ding et al. [23] reported a siRNA delivery system based on the cationic polymer, which was modified with perfluorooctylbromide (PFOB) to form a cationic polymer/siRNA/PFOB nanoemulsion (Figure 1). It efficiently downregulated signal transducer and activator of transcription 3 (STAT3) expression, significantly inhibited α‑smooth muscle actin (α-SMA) production and the migration of human fibroblasts, thereby effectively delaying IPF progression.
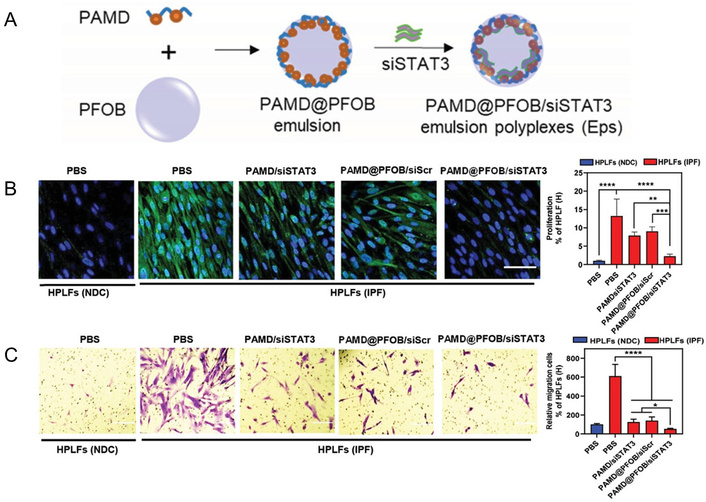
Schematic illustration of the components and preparation of cationic polymer/siRNA/PFOB nanoemulsion in inhibiting IPF. (A) Preparation of PAMD@PFOB/siSTAT3 Eps. (B) Immunofluorescence staining of α-SMA (green). Cell nuclei stained with DAPI (blue) (scale bar = 100 µm, n = 6). (C) Representative images of fibroblast migration with different treatments (n = 3). Adapted from [23]. © 2022 The Authors. Distributed under a license of CC BY. IPF: idiopathic pulmonary fibrosis; PFOB: perfluorooctylbromide.
Therefore, NDSs demonstrate significant potential for the efficient delivery of therapeutic agents targeting IPF. Such systems can substantially enhance bioavailability and pulmonary targeting efficiency while reducing systemic toxicity. Together, these unique advantages position NDSs as highly promising therapeutic tools for advancing IPF treatment.
Given the unique open-airway structure of the lungs, they serve as an ideal target organ for direct drug delivery via inhalation. Compared to traditional administration methods like oral administration or injection, inhalation delivery utilizes aerosol atomization technology to precisely deliver drug-loaded nanoparticles to pathological sites of IPF, such as alveolar spaces and pulmonary interstitium [24]. This localized delivery strategy significantly enhances drug bioavailability at the lesion site, improving therapeutic efficacy while minimizing systemic drug exposure and associated toxic side effects. Furthermore, inhalation administration markedly improves patient treatment compliance. However, inhalation delivery imposes stringent physicochemical requirements on nanoparticle delivery systems. Research indicates that nanoparticles for inhalation therapy typically require a size range of 100–500 nm to evade macrophage phagocytosis and achieve effective deposition within the alveoli. Furthermore, the surface charge of nanoparticles plays a crucial role in regulating their pulmonary behavior [25]. Charged particles, particularly negatively charged nanoparticles, exhibit higher alveolar deposition efficiency, stronger mucus penetration capabilities, and superior compatibility with pulmonary epithelial cells [25]. Therefore, in the design of inhalation-based NDSs, particle size regulation and surface charge optimization are critical considerations for ensuring delivery efficiency and biosafety. Given that most NDSs can meet the aforementioned requirements through optimized design of particle size and charge, inhalable NDSs show highly promising application prospects in the treatment of IPF. It should be noticed that inhalable NDSs (e.g., polymeric nanoparticles, MOFs, and nanomicelles) have been exploited in the area of therapy of many other respiratory diseases, like asthma, pulmonary infection, and lung cancers [26–28]. For instance, Guo et al. [28] developed an inhalable nanomicelle for the efficient delivery of dihydroartemisinin (DHA) and sorafenib (SRF) in lung cancer treatment, featuring a surface potential of –22.5 mV and a hydrated particle size of 251.8 nm. Following nebulized administration, its pulmonary deposition exceeded intravenous delivery by 3.3-fold, with in vivo experiments demonstrating superior antitumor efficacy. Thence, the feasibility of IPF management can be foreseeable.
The pathological microenvironment of IPF patients exhibits distinct characteristics, with abnormal increases in airway mucus secretion being one of its hallmark pathological features [29]. Under this pathological state, the thickness of the mucus layer in affected lungs significantly increases while viscosity substantially rises, forming a dense mucus barrier. This barrier directly impedes drug penetration to target lesions within the lungs and may also destabilize inhalation-based NDSs, reducing controlled-release efficiency and ultimately severely compromising therapeutic outcomes. Research indicates that NDSs modified with hydrophilic materials such as PEG can form a hydration layer through hydrogen bonding, effectively resisting non-specific protein adsorption and thereby achieving efficient penetration of the mucus layer [30]. Simultaneously, zwitterionic polymers leverage their charge-balanced structure to construct stable anti-mucus layers through ionic solvation, demonstrating superior mucus penetration capabilities. Furthermore, diverse carrier designs, including natural polysaccharide carriers and environmentally responsive smart carriers, significantly enhance the mucus penetration of NDSs, thereby increasing the effective drug concentration in lung tissue [31].
Overall, NDSs offer novel technical approaches for treating IPF. These NDSs have demonstrated promising efficacy in treating IPF and delaying disease progression, offering novel supplements to existing clinical therapies. However, numerous challenges persist in the clinical translation process, such as precise control over nanoparticle size, uniformity, stability, and batch-to-batch consistency under industrial manufacturing conditions [32]. In vivo safety evaluation involves complex issues such as long-term biocompatibility, potential organ accumulation, degradation behavior, and immune responses, necessitating the establishment of a systematic and standardized assessment framework [19]. Addressing these translational barriers effectively is crucial for advancing nanomedicines from laboratory research to practical clinical application, ultimately benefiting a large number of IPF patients.
FVC: forced vital capacity
IL-11: interleukin‑11
IPF: idiopathic pulmonary fibrosis
MOFs: metal-organic frameworks
NDSs: nanodelivery systems
NIN-SLN: nintedanib-loaded solid lipid nanoparticles
PEGylated: polyethylene glycol-modified
PFOB: perfluorooctylbromide
MZ: Conceptualization, Investigation, Writing—original draft. YJ: Investigation. CW: Conceptualization, Validation. ZH: Writing—review & editing, Supervision, Funding acquisition. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
We thank the support from the Science and Technology Projects in Guangzhou (Grant No. 202102070001) and the Open Funding of Henan Key Laboratory of Biomarker Detection and Diagnosis for Neurodegenerative Diseases (Grant No. HKLBDDND-202504). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 537
Download: 15
Times Cited: 0
