Affiliation:
1Faculty of Nursing, Al-Zaytoonah University of Jordan, Amman 11733, Jordan
Email: omaralqaisi119@gmail.com
ORCID: https://orcid.org/0009-0006-9760-651X
Affiliation:
1Faculty of Nursing, Al-Zaytoonah University of Jordan, Amman 11733, Jordan
ORCID: https://orcid.org/0000-0003-4789-4518
Affiliation:
2Faculty of Medicine, University of Prishtina, 1000 Prishtina, Kosovo
ORCID: https://orcid.org/0009-0000-9309-9959
Affiliation:
2Faculty of Medicine, University of Prishtina, 1000 Prishtina, Kosovo
ORCID: https://orcid.org/0009-0002-4880-1838
Affiliation:
2Faculty of Medicine, University of Prishtina, 1000 Prishtina, Kosovo
ORCID: https://orcid.org/0009-0002-0885-0897
Affiliation:
3Department of Oncology, University of Saskatchewan, Saskatoon, SK S7N 5A2, Canada
ORCID: https://orcid.org/0000-0001-7700-776X
Affiliation:
3Department of Oncology, University of Saskatchewan, Saskatoon, SK S7N 5A2, Canada
ORCID: https://orcid.org/0000-0001-5730-0088
Affiliation:
4Nursing Administration Department, Helwan University, Helwan 11795, Egypt
ORCID: https://orcid.org/0009-0004-8416-5300
Explor Med. 2026;7:1001393 DOI: https://doi.org/10.37349/emed.2026.1001393
Received: October 13, 2025 Accepted: February 05, 2026 Published: March 25, 2026
Academic Editor: Hua Su, University of California, USA
The article belongs to the special issue Practical Tips for Cancer Care: Guidance for Patients, Caregivers, and Healthcare Professionals
Acute paraplegia is a frequent and high-stake presentation in emergency departments. Spinal cord compression (SCC), particularly malignant epidural SCC, is a common oncologic emergency requiring urgent intervention, whereas Guillain-Barré syndrome (GBS) is a rarer but potentially life-threatening autoimmune polyradiculoneuropathy. Early differentiation between these conditions is essential, as delays in diagnosis and treatment are associated with irreversible neurological deficits and increased morbidity. Our objective is to synthesize recent evidence on the pathophysiology, clinical presentation, diagnosis, and management of SCC and GBS, with emphasis on early differentiation and multidisciplinary care strategies in emergency and rehabilitation settings. A scoping review was conducted following the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) guidelines. Electronic databases (PubMed, MEDLINE, Scopus, ScienceDirect, and CINAHL) were searched for English language studies published between January 2020 and June 2025. The review focused on clinical studies and reports addressing early differentiation and multidisciplinary management of SCC and GBS in emergency settings. Nineteen studies met the inclusion criteria. The review found that GBS diagnosis relies heavily on recognizing progressive symmetric weakness and preceding infectious triggers, while SCC requires immediate imaging and prompt corticosteroid administration. The limited number of studies highlights a gap in integrated emergency protocols for distinguishing these conditions. Surgical decompression remains the cornerstone of SCC management, with emerging evidence suggesting potential benefits even beyond the traditional 24-h window. This scoping review reinforces the critical need for early differentiation between SCC and GBS. Although the available literature is limited, it underscores the importance of coordinated multidisciplinary care. Clinicians must remain attentive to evolving diagnostic algorithms, particularly in light of new evidence supporting extended surgical windows for SCC.
Paraplegia is a common presentation in the emergency room worldwide. Differential diagnoses include spinal cord compression (SCC) and Guillain-Barré syndrome (GBS) (Table 1). SCC is an oncologic emergency that all healthcare providers should remain vigilant for. The frequency of mismanagement of SCC, particularly metastatic SCC, varies across healthcare settings, but studies consistently highlight delays in diagnosis and treatment as common and preventable contributors to poor outcomes. Delayed diagnosis and treatment occur in up to 50% of SCC cases, often due to failure to recognize early symptoms such as back pain, weakness, or sensory changes [1]. In fact, oncologists should proactively follow cancer patients who report back pain. A UK-based guideline notes that mismanagement or delayed treatment can lead to irreversible neurological damage and emphasizes that SCC is often under-recognized. SCC is a relatively common oncologic emergency as it affects 5–10% of cancer patients, yet timely magnetic resonance imaging (MRI) and corticosteroid administration are frequently missed or delayed, especially in non-specialist settings.
Comparison between spinal cord compression (SCC) and Guillain-Barré syndrome (GBS).
| Feature | GBS | SCC |
|---|---|---|
| Cause | Autoimmune, often after infection, such as Campylobacter jejuni | Physical compression from a tumor, abscess, fracture, or hematoma |
| Onset | Symptoms develop over a few days | Sudden or progressive, depending on the cause |
| Level of involvement | Peripheral nerves | Spinal cord |
| Type of weakness | Usually symmetrical and starts in the legs according to the type | Often asymmetrical and depends on lesion location |
| Reflexes | Absent or decreased | May be brisk early, reduced if severe damage |
| Sensory symptoms | Numbness and tingling in the limbs | Depends on the sensory level with a defined border below the lesion |
| Sphincter involvement | Rare | Common and often early |
| Pain | Burning or tingling nerve pain | Localized back pain, worse with movement |
| Diagnosis | Clinical signs, lumbar puncture, and nerve conduction studies | MRI of the spine |
| CSF findings | Elevated protein, normal WBC count | Usually, normal |
| Treatment | IVIG or plasmapheresis and supportive care | Early steroids, surgery, or radiotherapy depends on the cause |
| Respiratory failure risk | High—around 25% may require ventilation | Low—unless the cervical cord is affected |
| Recovery | Gradual, usually complete over weeks to months | Varies depending on the cause and timing of treatment |
| Prognosis | Good with early treatment | Worse if the diagnosis is delayed |
MRI: magnetic resonance imaging; CSF: cerebrospinal fluid; WBC: white blood count; IVIG: intravenous immunoglobulin.
All these could be related to a lack of awareness and poor logistics or coordination between referring physicians and tertiary centers [2]. Sandwell and West Birmingham National Health Service Guidelines of the United Kingdom emphasize the need for rapid recognition and management, noting that delays are common and are linked to poor outcomes [3]. The Global Spine Journal emphasizes the importance of early decompression surgery [4]. Any delays beyond 24 h significantly increase the risk of permanent disability. In the emergency room, it is important to distinguish these two treatable causes of paraplegia. Table 1 summarizes key clinical, diagnostic, and management differences between GBS and SCC, highlighting features critical to differential diagnosis in acute paraplegia.
An efficient diagnostic algorithm and unified operational pathway (Figure 1) for paraplegia begins with a clinical assessment focused on the onset and progression of symptoms. This is followed by targeted investigations to differentiate between SCC and GBS. A comprehensive neurological exam that evaluates reflexes, autonomic function, sensory level, and motor weakness is one of the initial steps. The diagnosis of GBS is supported by electrophysiological tests (such as nerve conduction studies and electromyography) and lumbar puncture to detect albuminocytologic dissociation. SCC and other structural causes must be identified or ruled out using MRI of the spine. Prioritizing imaging is supported by recognizing clinical red flags like rapid progression, sensory level, and bowel/bladder involvement. After diagnosis, management includes supportive care—such as respiratory monitoring and physical therapy—and immunotherapies like intravenous immunoglobulin (IVIG) or plasmapheresis, which are the main treatments for GBS. For SCC, management may involve rehabilitation to preserve neurological function, corticosteroids to reduce edema, and immediate spinal decompression surgery.
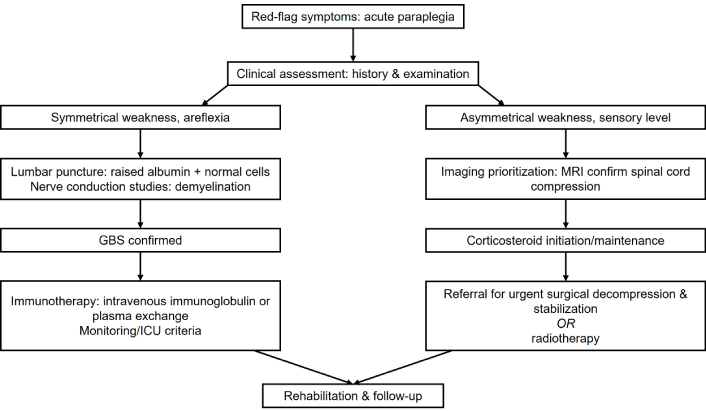
An algorithm for the paraplegia diagnostic approach and unified operational pathway for Guillain-Barré syndrome (GBS) and spinal cord compression. ICU: intensive care unit; MRI: magnetic resonance imaging.
This scoping review aims to map and synthesize existing literature on GBS and SCC from a multidisciplinary emergency perspective, focusing on early recognition, differential diagnosis, and coordinated management in acute care settings. Although not all aspects of the literature on both conditions are covered, the most clinically relevant studies influencing patient management are included and concisely summarized to support bedside decision-making.
A 75-year-old nursing home resident in Canada with pre-existing diffuse metastatic prostate cancer was diagnosed with suspected SCC at 9 am on a Friday in 2023. The on-call neurosurgeon was paged at 10 am but never responded, despite several attempts. Later, the doctor from the rural hospital called the cancer center; somehow, the on-duty radiation oncologist was tied up in a meeting. A busy clerk took the call instead and did not inform the oncologist promptly. The message on a piece of paper was noticed only around 4 pm. The radiation oncologist returned the call and requested the local doctor to administer intravenous dexamethasone 4 mg immediately and also arranged an ambulance to transfer the patient to the cancer center. It took about seven hours to reach the emergency room from the hospital due to inadequate ambulance staff. The radiation oncologist contacted his on-call team of physicists and radiation therapists to provide emergency radiotherapy since the patient was inoperable due to comorbidities and paraplegia likely lasted more than 24 h, which historically benefited less from surgery. He also started subcutaneous degarelix (a gonadotropin antagonist), 240 mg, which is preferred because it does not cause a gonadotropin-releasing hormone flare and reduces prostate-specific antigen and testosterone levels on the same day. The patient suffered from significant complications from his SCC, leading to higher nursing costs over the next four months before his eventual death, such as turning him in bed. The patient became depressed due to his disability and felt like a financial burden to his beloved adult children, as most Canadians do not have substantial savings.
A 40-year-old woman came to the emergency room in London, Ontario, with sudden paraplegia that developed over three days. She had always been healthy and had no family history of cancer. The only relevant history was that she experienced diarrhea two weeks earlier. The emergency room doctor considered the differential diagnoses of GBS vs. SCC. The radiation oncology team was consulted, and an MRI scan was ordered. The MRI results were normal. A lumbar puncture showed albuminocytological dissociation, with elevated cerebrospinal fluid (CSF) protein levels but no pleocytosis. Nerve conduction studies confirmed the GBS diagnosis, likely secondary to Campylobacter jejuni infection. She was treated with IVIG and gradually recovered over three weeks with physiotherapy.
The importance of timely multidisciplinary communication and prompt intervention in both GBS and SCC is emphasized by these cases. Delays can lead to irreversible neurological deficits, especially in SCC. The GBS case illustrates why autoimmune neuropathies should be considered when diagnosing acute paralysis, particularly following infectious prodrome to facilitate early immunotherapy.
This study utilized a scoping review methodology guided by the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) to map the existing literature, clarify key concepts, and identify gaps in research regarding the emergency differentiation of GBS and SCC, particularly given the heterogeneous nature of the available evidence [5]. Prospective registration of the protocol is not required.
The scoping review was conducted to address the complexity of differentiating acute paraplegia in emergency settings, a topic for which recent advances require synthesis. The review aims to provide an overview of existing studies and map the evidence landscape. The PRISMA-ScR methodology was used to ensure transparency and rigor. The literature search was conducted across Scopus, PubMed, MEDLINE, ScienceDirect, and CINAHL databases, covering the period from January 2020 to June 2025. The search was limited to this timeframe to capture the most recent evidence, contemporary diagnostic approaches, and current multidisciplinary management strategies relevant to emergency care. Earlier landmark studies were cited selectively where necessary to provide historical context.
Search terms included ‘Guillain-Barré Syndrome’, ‘autoimmune neuropathy’, ‘molecular mimicry’, and ‘spinal cord compression’, combined with Boolean operators. Two independent reviewers screened titles and abstracts for relevance, followed by full-text evaluation. Inclusion criteria encompassed English-language clinical studies, case reports, and reviews focused on epidemiology, pathophysiology, diagnostics, and management. Discrepancies in selection were resolved through consensus. A PRISMA-ScR flow diagram (Figure 2) illustrates the selection process. Database searches yielded an initial total of 3,420 records. After eliminating 720 duplicate records, 2,700 unique records were screened for titles and abstracts; 2,480 were excluded based on predefined criteria and lack of relevance. Two hundred and twenty full-text articles were then evaluated for eligibility. Two hundred and one of these were excluded for various reasons, including non-empirical study designs (n = 60), non-English language (n = 30), non-relevant populations (n = 71), and pediatric populations (n = 40). Ultimately, 19 studies met the inclusion criteria and were incorporated into the review.
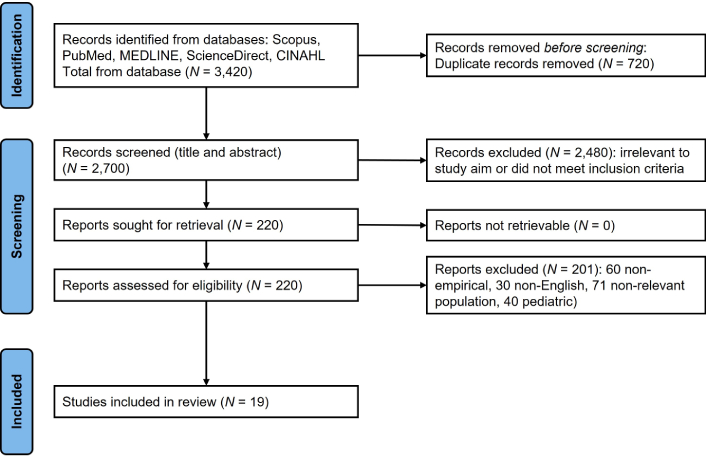
Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) flow diagram. Adapted from “The PRISMA 2020 statement: an updated guideline for reporting systematic reviews” by Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. BMJ. 2021;372:n71 (https://doi.org/10.1136/bmj.n71). © 2021 The Author(s). CC BY 4.0.
Given the scoping nature of this review and the diversity of the 19 included studies (ranging from case reports to retrospective cohorts), a narrative synthesis approach was employed. Quality assessment was not performed, consistent with standard scoping review methodology, as the primary aim was to map the breadth of available evidence and identify research gaps rather than to assess the efficacy of interventions. While a formal quality assessment, such as Mixed Methods Appraisal Tool (MMAT) or Risk of Bias In Systematic reviews (ROBIS), is typically reserved for systematic reviews, this scoping review evaluated sources’ credibility based on study design rigor and peer-review status.
Five main areas of clinical management were identified through thematic analysis using iterative coding cycles. The data was collected and analyzed using a thematic approach. The studies were categorized based on key themes: etiology, clinical manifestations, diagnostic methods, therapeutic interventions, and prognosis. Qualitative thematic coding was performed manually by the authors. Dedicated qualitative data analysis software (such as NVivo or ATLAS.ti) was not used, as the scope and number of included studies allowed for structured manual coding. This enabled a thorough comparison of the conditions and facilitated the identification of parallels and differences pertinent to acute paraplegia presentations. Thematic synthesis was used to integrate data from multiple studies to draw insightful conclusions about clinical management and outcomes.
As illustrated in Figure 2, the exclusion of a significant number of articles (n = 201) was primarily due to the lack of specific focus on the emergency differentiation or multidisciplinary management of these conditions, or the use of non-clinical study designs. This rigorous selection process ensures that the included studies (n = 19) provide the most relevant evidence for acute clinical decision-making (Table 2).
Database search results: 19 studies.
| No. | Author/reference | Design | Country | Results |
|---|---|---|---|---|
| 1 | Breville et al. [6] | Review | Switzerland | Detecting patients at risk of poor outcome may improve management. Clinical poor prognostic factors: higher age, presence of diarrhea within 4 weeks of symptom onset, rapid and severe weakness progression, dysautonomia, decreased vital capacity, and facial, bulbar, and neck weakness. Biological, neurophysiological and imaging measures of unfavorable outcome: multiple anti-ganglioside antibodies elevation, increased serum and CSF neurofilaments light (NfL) and heavy chain, decreased NfL CSF/serum ratio, hypoalbuminemia, nerve conduction study with early signs of demyelination or axonal loss, and enlargement of nerve cross-sectional area on ultrasound. Depicting prognostic biomarkers aims at predicting short-term mortality and need for cardio-pulmonary support and monitoring therapeutic responses in future clinical trials. |
| 2 | Lee et al. [7] | Review | South Korea | Although ophthalmoplegia is a hallmark of anti-GQ1b antibody syndrome, recent studies have identified this antibody in patients with acute vestibular syndrome, optic neuropathy with disc swelling, and acute sensory ataxic neuropathy of atypical presentation. Ophthalmoplegia associated with anti-GQ1b antibody positivity is complete in > 50% of the patients but may be monocular or concomitant. The prognosis is mostly favorable; however, approximately 14% of patients experience relapse. |
| 3 | Vargas et al. [8] | Retrospective cohort | USA | 90 patients met the inclusion criteria (45 SBRT and 45 EBRT matched cases). Balance of the covariates, SINS, age, follow-up time, and primary tumor histology after the matching process was confirmed between groups (p = 0.062, p = 0.174, and 0.991, respectively, along with matched tumor histology). The SBRT group had a higher 5-year rate of vertebral body fracture at 22.22% (n = 10) compared with 6.67% (n = 3) in the EBRT group (p = 0.044). Survival analysis was used to adjust for uneven follow-up time and showed a significant difference in fracture rates between the two groups (p = 0.044). SBRT also was associated with a higher rate of local control (86.67% vs 77.78%). |
| 4 | Inan et al. [9] | Review | Turkey | Electrophysiological studies, US, and immunological markers have been explored for discrimination; however, definitive criteria for differentiation remain elusive. Recent follow-up studies have further blurred the boundaries between recurrent GBS and A-CIDP, suggesting the persistence of underlying immune processes even in GBS patients without clinical deterioration. This emphasizes the necessity of reevaluating diagnostic criteria and treatment strategies. In conclusion, distinguishing recurrent GBS from A-CIDP remains an ongoing challenge. Existing evidence questions the categorization of recurrent GBS as a distinct entity, challenging its very existence. Continued research is necessary to refine diagnostic criteria and deepen our understanding of these conditions, ultimately advancing patient care. |
| 5 | Antoine [10] | Review | France | About 20 antibodies have been identified in immune-mediated neuropathies, recognizing membrane or intracellular proteins or glycolipids of neuron and Schwann cells. This article reviews the different methods used for their detection, what we know about their pathogenic role, how they have helped identify several disorders, and how they are essential for diagnosis. Despite sustained efforts, some immune-mediated disorders still lack identified autoantibodies, notably the classical form of GBS & CIDP. The article also tries to determine potential future developments in antibody research, particularly the use of “omic” approaches and the search for other types of biomarkers beyond diagnostic ones, e.g., those that can identify patients who will respond to a given treatment. |
| 6 | Finsterer [11] | Review | Austria | The most common subtypes of GBS are acute inflammatory demyelinating polyneuropathy (AIDP) and acute motor axonal neuropathy (AMAN). The most common triggers of GBS, in 3/4 of cases, are previous infections, most commonly Campylobacter jejuni (C. jejuni), Mycoplasma pneumoniae, and cytomegalovirus. C. jejuni is responsible for about a third of GBS cases. GBS due to C. jejuni is usually more severe than that due to other causes. Clinical presentation of GBS is highly dependent on the structure of pathogenic lipo-oligosaccharides (LOS) that trigger the innate immune system via Toll-like-receptor (TLR)-4 signaling. AIDP is due to demyelination, whereas in AMAN, structures of the axolemma are affected in the nodal or inter-nodal space. |
| 7 | Bragazzi et al. [12] | Modeling study | Iran | In 2019, there were 150,095 [95% uncertainty intervals (UI) 119,924 to 188,309] total cases of GBS worldwide, which resulted in 44,407 (95% UI 28,016 to 64,777) years lived with disability (YLDs). Globally, there was a 6.4% (95% UI 3.6 to 9.5) increase in the age-standardized prevalence of GBS per 100,000 population between 1990 and 2019. High-income Asia Pacific [1.9 (95% UI: 1.5 to 2.4)] and East Asia [0.8 (95% UI: 0.6 to 1.0)] had the highest and lowest age-standardized prevalence rates (per 100,000), respectively, in 2019. Nationally, Japan [6.4 (95% UI: 5.3 to 7.7)] and China [0.8 (95% UI: 0.6 to 1.0)] had the highest and lowest age-standardized prevalence rates (per 100,000). The age-standardized burden of GBS increased with increasing age and was higher in males in all age groups. Furthermore, the age-standardized prevalence of GBS (per 100,000) had a positive association with the level of development, as measured by socio-demographic index, although this association was not strong. Upper respiratory infections and unknown causes accounted for the highest proportions of underlying causes. Data from the Global Burden of Diseases Study (GBD) 2019 were used. GBD 2019 modelled the prevalence of GBS using hospital and claims data. YLDs were estimated as the product of the GBS prevalence and the disability weight. This article also reported proportions in the age-standardized prevalence rate that were due to 6 underlying causes of GBS. |
| 8 | Connors et al. [13] | Case report | USA | A 61-year-old male was admitted to an LTACH for the rehabilitation of GBS following COVID-19 infection and IVIG. Rehabilitation in the LTACH setting uses a variety of skilled treatment interventions to meet patient-driven goals and maximize their function to the highest level possible in preparation of their discharge to a subacute or homecare setting. In this case, this was accomplished through individual OT and PT sessions, OT/PT cotreatment sessions, and targeted group therapy sessions focused on leg, arm, and fine motor coordination exercises. |
| 9 | Galieri et al. [14] | Systematic review | Italy | A total of 32 studies met the inclusion criteria. Robotic-assisted surgery demonstrated high accuracy in pedicle screw placement (~92–94%) and reduced intraoperative blood loss and radiation exposure, although long-term clinical outcomes were comparable to conventional techniques. Intraoperative navigation improved instrumentation precision, while augmented reality enhanced ergonomic workflow and reduced surgeon distraction. AI tools showed promise in surgical planning, guidance, and outcome prediction but lacked definitive evidence of clinical superiority. Minimally invasive surgery (MIS) techniques—including endoscopic discectomy and MIS-transforaminal lumbar interbody fusion—offered reduced blood loss, shorter hospital stays, and faster recovery, with equivalent pain relief, fusion rates, and complication profiles compared to open procedures. Lateral and oblique approaches [extreme lateral interbody fusion or oblique lateral interbody fusion (OLIF)] further optimized alignment and indirect decompression, with favorable perioperative metrics. This systematic review evaluates the literature from Feb 2020 to Feb 2025 on technological and procedural innovations in lumbar spinal stenosis. Eligible studies focused on degenerative lumbar pathologies, advanced surgical technologies, and reported clinical or perioperative outcomes, including RCTs, comparative studies, meta-analyses, and large case series. |
| 10 | Palacio Giraldo et al. [15] | Systematic review | Austria | The main treatment goals for spinal metastases include local control, pain relief, improvement or prevention of neurologic symptoms, and maintenance of stability. MRI is considered the gold standard for diagnosing spinal metastases, although alternative imaging modalities are also described. Treatment options include systemic therapies, radiotherapy, and surgical techniques for metastatic lesions. The combination of SBRT with surgical spinal decompression of the spine is an evolving treatment approach with promising results. Several treatment management algorithms, including the NOMS (neurology, oncology, mechanical stability, and systemic disease), and LMNOP (localization, mechanical instability, neurology, oncology) algorithms, have been proposed to aid in the decision-making process regarding individual treatment modalities. The primary factors influencing treatment decisions are primary tumor histology and life expectancy. This review focuses on the symptoms, diagnosis & treatment of spinal metastases, which represent a late complication of the primary tumor. |
| 11 | Khan et al. [16] | Review | Bangladesh | It comprehensively examines the life-threatening nature and intensity of GBS by assessing its etiology, progression, and prevalence in low- and middle-income nations while also considering global trends. It proposed the implementation of standard efficacious treatment & diagnostic resources that are readily accessible and successful in affluent nations and should also be readily accessible in impoverished nations without any unnecessary delay. It emphasized the epidemiological data with molecular epidemiological analysis and the utilization of AI technology in low- and middle-income nations. The goal was to decrease the incidence of GBS cases and facilitate early detection. |
| 12 | Kim et al. [17] | Cohort study | Korea | The Korean National Health Insurance Service claims data from 2010 to 2016 were used to identify incident cases as newly hospitalized patients with a primary diagnosis of GBS (the 10th revision of the International Classification Disease code of G61.0). New cases were defined as patients not having claim records for GBS within one year before the hospital admission for GBS. |
| 13 | Leitner et al. [18] | Retrospective cohort study (propensity score analysis) | Austria | Single-center propensity score analyses with inverse-probability-of-treatment-weights (IPTW) of OS and short-term neurologic outcomes in MSCC patients treated with or without surgery between 2007 and 2020. Among 398 patients with MSCC, 194 (49%) underwent surgery. During a median follow-up of 5.8 years, 355 patients (89%) died. MBs was the most important predictor for spine surgery (p < 0.0001) and the strongest predictor of favorable OS (p < 0.0001). Surgery was associated with improved OS after accounting for selection bias with the IPTW method (p = 0.021) and emerged as the strongest determinant of short-term neurological improvement (p < 0.0001). Exploratory analyses delineated a subgroup of patients with an MBs of 1 point who still benefited from surgery, and surgery did not result in a higher risk of short-term oncologic disease progression. This was a single-center, open-label, RCT performed on 2 parallel groups: 50 intensive care unit adults admitted for GBS and at risk for acute respiratory failure were randomized to early mechanical ventilation via face mask or endotracheal intubation owing to the presence or absence of impaired swallowing (experimental arm), or to conventional care (control arm). The primary outcome was the incidence of pneumonia up to intensive care unit discharge (or 90 days, whichever occurred first). |
| 14 | Li et al. [19] | Retrospective cohort | China | This study hypothesizes that, unlike in cases of trauma and degenerative disorders, where delayed decompression surgery often leads to poor outcomes, delayed decompression surgery for malignant epidural spinal cord compression-induced paralysis yields relatively favorable results. It included patients who had been paralyzed for more than 48 hours and underwent decompressive surgery between Jan 2012 and Dec 2020. Data collected mainly included patient demographics, tumor pathologies, neurological function (Frankel grades), ambulatory status, and imaging. Primary outcome measure: neurological recovery. |
| 15 | Melone et al. [20] | RCT | France | 25 patients were randomized in each group: no significant difference between groups for the incidence of pneumonia (10/25 (40%) vs 9/25 (36%), P = 1). There was no significant difference between groups for the time to onset of pneumonia (P = 0.50, Gray test). During follow-up, there were 16/25 (64%) mechanically ventilated patients in the control group, and 25/25 (100%) in the experimental arm (P < 000.1). The time on ventilator was non-significantly shorter in the experimental arm (14 [7; 29] versus 21.5 [17.3; 35.5], P = 0.10). There were no significant differences between groups for length of hospital stay, neurological scores, the proportion of patients who needed tracheostomy, in-hospital death, or any serious adverse events. |
| 16 | Oshomoji et al. [21] | Systematic review | Nigeria | Key findings indicate that specific autoantibodies, e.g., anti-GM1 and anti-GQ1b, are associated with distinct subtypes of GBS, contributing to its heterogeneity. Understanding these autoimmune mechanisms is crucial for improving diagnostic accuracy, therapeutic strategies, and prognostic indicators in GBS. This review highlights significant gaps in current research, emphasizing the need for further studies to explore the genetic and environmental factors that influence GBS susceptibility and the role of vaccinations in triggering autoimmune responses. It aims to elucidate the autoimmune mechanisms underlying the various subtypes of GBS, including AIDP, AMAN, acute motor and sensory axonal neuropathy (AMSAN), and Miller Fisher syndrome (MFS). A thorough literature search identified 71 studies published between 2010 and 2024. |
| 17 | Shea and Kwan [22] | Narrative review | Hong Kong, China | This review presents a comprehensive approach to the management of spinal metastases. The wide spectrum of clinical presentation in spinal metastases necessitates a personalized approach to treatment planning. This includes a comprehensive diagnostic workup, oncological management, palliation of symptoms, and surgical intervention if appropriate. A systematic and multidisciplinary approach allows optimal shared decision making to reach an evidence-informed and value-congruent treatment plan for the patient. We highlight how advances in stereotactic body radiotherapy (SBRT) and separation surgery may be incorporated into clinical management from a spine surgeon’s perspective. |
| 18 | Tewedaj et al. [23] | Retrospective cross-sectional study | Ethiopia | 60 GBS patients admitted to TASH from Jan 2018 to Dec 2022. Data on demographics, clinical features, treatments, complications, and outcomes were extracted from medical records. Bivariate and multivariate logistic regression analyses identified factors associated with mortality and poor hospital outcomes. Ascending paralysis (76.7%) was the predominant presentation. Absent or reduced reflexes were seen in 91.7% of patients. The most common antecedent event was gastroenteritis (26.7%), followed by upper respiratory tract infection (URTI) (15%) and vaccination (11.7%). The mean interval from symptom onset to hospital presentation was 8.77 days, and the peak symptom severity was 4.47 days. The axonal variant (75.5%) was the most common subtype, followed by the demyelinating variant (24.5%). IVIG (intravenous immunoglobulin) was administered to 41.7% of patients. Respiratory failure requiring invasive mechanical ventilator (MV) support occurred in 26.7% of cases. The mortality rate was 10%, with mechanical ventilation being the only factor significantly associated with mortality (95% CI 2.067–184.858; P < 0.010). At discharge, 55% had a good outcome, and 45% had a poor outcome, according to the Hughes Functional Disability Scale (HFDS). MV (AOR 0.024, 95% CI 0.001–0.607) and a GBS disability score > 3 (AOR 0.106, 95% CI 0.024–0.467) were factors significantly associated with poor hospital outcomes. GBS in this cohort primarily affected individuals of young age, commonly preceded by gastroenteritis and characterized by a high frequency of the axonal variant. Mechanical ventilation was found to be significantly linked to mortality. Alongside mechanical ventilation requirements, severe disability upon presentation emerged as a crucial determinant of poor outcomes upon discharge. |
| 19 | Xu et al. [24] | Population-based study + systematic review and meta-analysis | China | A total of 1.44 billion person-years in insurance data was covered, with 3,534 GBS cases identified. The annual incidences of GBS in urban China between 2013 and 2017 ranged from 0.41 (95% CI: 0.27 to 0.58) to 0.58 (95% CI: 0.38 to 0.82) per 100,000 person-years. The incidence was the highest in Northwest China and the lowest in Northeast China. The meta-analysis included 122 articles. The quality assessment showed that the quality scores of 43.3% of studies were ≥ 0.75 (the total score is 1). The global incidence of GBS was 1.12 (95% CI: 0.98 to 1.27) per 100,000 person-years. The incidences in West Europe, South Asia, and North Europe were higher, while the incidences in Australia and New Zealand, Southeast Asia, and North Africa were lower. The incidence of enteric infections was positively associated with the incidence of GBS (coefficient = 0.0000185, P = 0.007). The incidence in Europe, Australia, and America rose significantly from 1960 to 2020 (coefficient = 0.01, t = 2.52, P = 0.015). A population-based study to calculate the incidence of GBS in urban China based on the National Urban Medical Insurance database from 2013 to 2017. To identify GBS cases, natural language processing was used first for handling the lengthy and unstructured diagnostic information and then checked by prestigious neurologists. Secondly, a systematic review and meta-analysis were performed to analyze the incidence of GBS worldwide. Up to July 4, 2022, Medline, Embase, and Web of Science were retrieved to identify the population-based studies regarding the incidence of GBS. The basic information and the statistics regarding incidence were extracted. Quality assessment considered sample representativeness, condition assessment, and statistical methods. |
A-CIDP: acute onset chronic inflammatory demyelinating polyneuropathy; AI: artificial intelligence; CSF: cerebrospinal fluid; EBRT: external beam radiation therapy; GBS: Guillain-Barré syndrome; GM1: ganglioside M1; GQ1b: ganglioside Q1b; LTACH: long-term acute care hospital; MBs: modified Bauer score (likely a spinal metastasis grading scale variant); MRI: magnetic resonance imaging; MSCC: malignant spinal cord compression; TASH: Tikur Anbessa Specialized Hospital (Ethiopia’s main tertiary hospital in Addis Ababa); OS: overall survival; OT/PT: occupational therapy/physiotherapy; RCTs: randomized controlled trials; SINS: Spinal Instability Neoplastic Score; US: ultrasonography.
Recurring delays and missed diagnoses of SCC have been consistently reported in clinical practice, highlighting the need for an updated review of current management strategies. SCC, especially malignant epidural SCC (MESCC), happens when metastatic tumors invade the vertebral column and press on the spinal cord. This can cause pain, motor problems, and paralysis. Quick action is essential to protect neurological function and improve quality of life. Recent surgical studies highlight the changing role of decompressed surgery in MESCC treatment as follows.
A 2024 study found that decompressed surgery after > 48 h of paralysis still resulted in meaningful neurological recovery: > 80% improved by at least one Frankel grade, and nearly 60% regained ambulatory ability [19]. The 1969 landmark study introduced the Frankel classification (Grades A–E), widely used to assess neurological impairment following spinal cord injury [25].
A 2023 study found that surgery significantly improves overall survival (OS) and neurological outcomes, even in patients with poor prognostic scores [18]. Using propensity score analysis, researchers showed that decompression surgery followed by radiotherapy led to better functional recovery compared to radiotherapy alone.
Innovative endoscopic spine surgeries and robot-guided decompressions are increasingly used. These ultra-minimally invasive approaches reduce tissue disruption, accelerate recovery, and preserve spinal stability. Bone cement augmentation is also used to reinforce vertebral bodies post-tumor resection [14].
Artificial intelligence (AI) is increasingly used in radiotherapy treatment planning, dose optimization, and dosimetry [26]. Other areas of radiotherapy advances include the following.
Radiotherapy remains a cornerstone of MESCC treatment, especially when surgery is contraindicated. The most notable recent research on optimizing dose, timing, and technique is the SCORAD randomized trial, which compared single-fraction (8 Gy) vs. multi-fraction (20 Gy in 5 sessions) radiotherapy [27]. The full name of the SCORAD randomized trial is: “Effect of Single-Fraction vs Multifraction Radiotherapy on Ambulatory Status Among Patients With Spinal Canal Compression From Metastatic Cancer: The SCORAD Randomized Clinical Trial”. While single-fraction did not meet strict non-inferiority criteria for ambulatory status at 8 weeks, it showed comparable outcomes at 1, 4, and 12 weeks, suggesting it may be suitable for patients with limited life expectancy.
SBRT delivers high-dose radiation with precision, sparing surrounding tissues. It is increasingly used in postoperative settings and for recurrent MESCC, especially in radio-resistant tumors, as it is believed that the larger dose per fraction may be able to overcome radio-resistance. SBRT has shown promising results in local tumor control and pain relief [28, 29]. However, vertebral body collapse may occur after SBRT.
Two key peer-reviewed papers authored by Dr. Arjun Sahgal investigated vertebral body fractures following spine SBRT. All these studies have shaped current guidelines and underscore the need for careful patient selection and risk stratification when using SBRT for spinal metastases: (a) The 2013 landmark multi-institutional analysis reported on 410 spinal segments treated with SBRT [30]. It found a 14% incidence of vertebral compression fractures (VCFs), with risk factors including high radiation dose ≥ 20 Gy/fraction, baseline VCF, and lytic tumors. (b) Another multi-institutional study was published in 2016, which expanded on earlier findings with a larger cohort and confirmed that SBRT is associated with a significant risk of VCF, especially in patients with spinal instability [31]. (c) A summary of clinical implications of VCF risk post-SBRT and emphasizes the importance of Spinal Instability Neoplastic Score (SINS) in treatment planning [8]. Table 3 summarizes the foundational paper of the SINS system, which helps clinicians assess spinal stability in patients with metastatic spine disease and guides surgical decision-making [32]. This score guides decisions on surgical referral and urgency.
A concise summary table of the Spinal Instability Neoplastic Score (SINS), which is used to assess spinal stability in patients with spinal metastases.
| Category | Criteria | Score |
|---|---|---|
| Location | Junctional (occiput–C2, C7–T2, T11–L1, L5–S1) | 3 |
| Mobile spine (C3–C6, L2–L4) | 2 | |
| Semi-rigid (T3–T10) | 1 | |
| Rigid (S2–S5) | 0 | |
| Pain | Yes, mechanical (movement-related) | 3 |
| Yes, but not mechanical | 1 | |
| No pain | 0 | |
| Bone lesion quality | Lytic | 2 |
| Mixed (lytic/blastic) | 1 | |
| Blastic | 0 | |
| Radiographic alignment | Subluxation or translation | 4 |
| Deformity (kyphosis/scoliosis) | 2 | |
| Normal alignment | 0 | |
| Vertebral body collapse | > 50% collapse | 3 |
| < 50% collapse | 2 | |
| No collapse, but > 50% vertebral body involved | 1 | |
| No collapse | 0 | |
| Posterolateral involvement | Bilateral pedicle/lamina involvement | 3 |
| Unilateral involvement | 1 | |
| None | 0 |
Interpretation: 0–6 points: stable; 7–12 points: indeterminate (possibly unstable); 13–18 points: unstable.
The combination treatment offers synergistic benefits. Radiotherapy is most effective when administered within 24 h of diagnosis. Postoperative radiotherapy enhances tumor control and reduces the risk of recurrence [33]. In terms of patient selection, prognostic tools like the modified Bauer score and Tokuhashi score help identify candidates who will benefit most from surgery and/or radiotherapy diagnosis [34, 35]. The Tokuhashi score, developed initially and later revised by Tokuhashi et al. [36], is a validated prognostic scoring system designed to estimate survival and guide treatment decisions in patients with spinal metastases. In clinical practice, the Tokuhashi score is widely used to guide treatment decisions by estimating survival and identifying patients who may benefit from surgery, radiotherapy, or conservative treatment (Tables 4 and 5). Unnecessary aggressive treatment will tire out the patient and caregiver, as well as wasting scarce healthcare resources. A multidisciplinary approach can achieve optimal outcomes, but it requires significant effort and collaboration among neurosurgeons, radiation oncologists, and palliative care teams. Personalized treatment plans based on tumor type, neurological status, and life expectancy are essential [15, 22]. Both papers discuss modern strategies for spinal metastases, highlighting the role of multidisciplinary, personalized approaches based on clinical and oncologic parameters, as well as the estimation of survival.
The Tokuhashi score, a prognostic tool used to estimate survival and guide treatment decisions for patients with spinal metastases, based on survival estimates and tumor characteristics.
| Parameter | Score options |
|---|---|
| General condition (Karnofsky Performance Status) | Poor (10–40%) = 0Moderate (50–70%) = 1Good (80–100%) = 2 |
| Number of extraspinal bone metastases | ≥ 3 = 01–2 = 10 = 2 |
| Number of metastases in the vertebral body | ≥ 3 = 02 = 11 = 2 |
| Metastases to major internal organs | Non-removable = 0Removable = 1None = 2 |
| Primary site of cancer | Lung, osteosarcoma, stomach, bladder, esophagus, pancreas = 0Liver, gallbladder, unidentified = 1Other cancers = 2Kidney, uterus = 3Rectum = 4Thyroid, prostate, breast, carcinoid tumor = 5 |
| Neurological status (Frankel classification) | Frankel A, B = 0Frankel C, D = 1Frankel E = 2 |
Interpretation: total score range: 0–15. Prognostic guidance: 0–8 points: predicted survival is generally less than 6 months, and conservative or palliative management is usually considered; 9–11 points: predicted survival is approximately 6–12 months, and selected patients may be considered for surgical intervention; 12–15 points: predicted survival is more than 1 year, and surgical treatment may be appropriate in suitable candidates.
Comparison of two widely used scoring systems for spinal metastases.
| Feature | Tokuhashi score | Spinal Instability Neoplastic Score (SINS) |
|---|---|---|
| Purpose | Predicts life expectancy in patients with spinal metastases | Assesses spinal stability in neoplastic disease |
| Clinical use | Guides surgical vs. conservative treatment decisions | Determines need for surgical stabilization or referral |
| Scoring range | 0–15 points | 0–18 points |
| Key parameters | - General condition- Number of extraspinal bone metastases- Number of vertebral metastases- Metastases to major organs- Primary tumor type- Neurological status | - Location of lesion- Pain characteristics- Bone lesion type- Spinal alignment- Vertebral body collapse- Posterolateral involvement |
| Interpretation | 0–8: predicted survival generally < 6 months (conservative or palliative management usually considered)9–11: predicted survival approximately 6–12 months (selected patients may be considered for surgery)12–15: predicted survival > 1 year (surgical treatment may be appropriate in suitable candidates) | 0–6: stable7–12: indeterminate13–18: unstable |
| Strengths | Prognostic clarity for survival-based decisions | Objective assessment of mechanical instability |
| Limitations | Less emphasis on mechanical factors | Does not predict survival |
| Developed by | Tokuhashi et al., Japan | Spine Oncology Study Group |
| Year introduced | 1990 (updated 2005) | 2010 |
Digital implants and neurostimulation, with experimental technologies like brain-spine interface implants and stem cell research, can restore mobility in spinal cord injury patients [37]. Use of AI in image analysis, real-world data from retrospective analyses from academic centers are helping refine treatment protocols and identify gaps in care [26, 38].
GBS is less well-known. It is an acute autoimmune demyelinating polyneuropathy [13]. It is a severe inflammatory polyneuropathy with a clinically rapid onset of muscle weakness and sensory disturbances due to immune-mediated damage of the peripheral nervous system [12]. The global incidence of GBS ranges from 0.89 to 1.89 cases per 100,000 population per year, with regional variations. The incidence in 2019 was approximately 150,095 cases worldwide, or 1.9 per 100,000 age-standardized point prevalence [16]. The incidence is higher among individuals over 50 years of age than among those under 50, increasing from 1.7 per 100,000 in those under 50 to 3.3 per 100,000 in those 50 or older. The relative risk is 1.78 for men and is affected more frequently than among women [12]. Prevalence data for GBS are shown in Table 6 [39–41]. Sejvar et al. [40] reported a comprehensive meta-analysis that remains one of the most cited sources for global and regional GBS incidence rates. Xu et al. [24] reported the most recent data in 2024.
Epidemiological prevalence of Guillain-Barré syndrome (GBS).
| Region | Prevalence rate | Source/note |
|---|---|---|
| Global | 0.84–1.91 per 100,000 annually | [39] (McGrogan et al., 2009), [40] (Sejvar et al., 2011) |
| North America | 1.65–1.79 per 100,000 annually | [41] (Alshekhlee et al., 2008) |
| Europe | 1.2–1.9 per 100,000 annually | [40] (Sejvar et al., 2011) |
| Asia | 0.9–1.8 per 100,000 annually | [40] (Sejvar et al., 2011) |
| Africa | Limited data, estimated ≈ 1 per 100,000 | [40] (Sejvar et al., 2011) |
| Jordan (Salt outbreak) | 19 cases in the 1976 outbreak | [40] (Sejvar et al., 2011) |
| Age < 50 years (global) | 1.7 per 100,000 | [40] (Sejvar et al., 2011) |
| Male vs. female (global) | Relative risk: 1.78 (higher in males) | [40] (Sejvar et al., 2011) |
Given its potential for morbidity, especially respiratory failure and long-term disability, understanding GBS is essential for all healthcare workers. Early recognition and intervention are crucial because approximately 25% of patients will need mechanical ventilation due to respiratory muscle weakness [20]. The authors also pointed out that the death rate in GBS ranges from 3% to 7%. This indicates that while many patients recover, some may experience persistent neurological deficits or fatigue that impact their quality of life. Furthermore, GBS places a considerable economic burden. The direct medical costs per patient can be high in high-income countries, covering expenses for hospitalization, intensive care, and rehabilitation services. Indirect costs, like lost productivity due to disability and long-term disability, also contribute to the overall economic impact.
In summary, GBS is a rare but serious neurological disorder that occurs globally. The incidence depends on region, age, and sex. It constitutes a serious health and economic challenge. Recent national data from South Korea also highlighted that GBS imposes a considerable economic and healthcare burden, emphasizing the need for improved surveillance and management strategies [17]. This publication presents a comprehensive analysis of GBS burden across 204 countries, highlighting disparities in incidence and data availability, especially in low-resource regions. Another publication also explores differences in GBS presentation and outcomes across regions, emphasizing the need for improved surveillance and diagnostic infrastructure in underrepresented areas [17].
The financial impact of Campylobacter infections in the USA is significant, with costs associated with GBS estimated at $1.7 billion each year [42]. Most of this cost stems from indirect factors like premature death, while direct medical expenses account for approximately $0.2 billion. When factoring in Campylobacter-associated GBS and its related costs, the total economic burden may reach $8 billion annually. This highlights the urgent need to address the public health implications of Campylobacter infections and their connection to GBS to mitigate these substantial financial strains.
Important considerations in GBS involve differential diagnoses that also present with acute weakness. SCC requires immediate intervention with corticosteroids, surgical decompression, and/or radiotherapy. Other urgent differentials include stroke, acute encephalitis, botulism, and toxin exposure—each necessitating specific emergency treatments, as delays can lead to irreversible neurological damage. Another important consideration is the public concern over vaccination, which stems from the rare association of GBS with certain vaccines [43, 44], such as those for influenza [45] and coronavirus disease 2019 (COVID-19) [46, 47]. However, a 2022 study supports the long-term immunological safety of multiple vaccinations, showing no evidence of autoimmune or lymphoproliferative complications over a 5-year follow-up [48].
Another notable point is the limited specific epidemiological data on GBS. Nevertheless, a notable outbreak in early January 1976 occurred in Salt, Jordan, following contamination of the water supply, resulting in gastroenteritis. This study involved 19 cases of GBS, suggesting a possible localized increase in incidence due to environmental factors [46].
GBS is an acute autoimmune neuropathy in which the immune system mistakenly attacks parts of the peripheral nervous system, leading to muscle weakness and often paralysis [49]. Because of the complexity and variety of the underlying pathophysiological mechanisms, GBS involves both humoral and cellular immune responses. Vaccines can rarely be linked to GBS. The pathogenesis of GBS also involves cellular mechanisms in addition to humoral mechanisms. Within peripheral nerves, inflammation and demyelination occur because of infiltration of activated T lymphocytes and macrophages. The characteristic muscle weakness results from this infiltration, leading to demyelination and axonal degeneration that disrupts nerve conduction. Molecular mimicry is a well-described principal mechanism by which GBS is induced. In this instance, structural similarities between microbial antigens and components of host nerve tissue trigger an autoreactive immune response. In the context of GBS, infections with pathogens such as Campylobacter jejuni can induce the production of antibodies that cross-react with gangliosides. These sialic acid-containing glycosphingolipids are highly expressed in the peripheral nervous system. The immune-mediated damage to nerve tissues is due to this cross-reactivity. GBS is the most commonly unrecognized sequela of Campylobacter jejuni infection. The bacterium’s lipo-oligosaccharides (LOS) exhibit molecular mimicry with human gangliosides ganglioside M1 (GM1) and ganglioside D1a (GD1a). This mimicry triggers the production of antiganglioside antibodies, which bind to nerve tissues and activate complement pathways, resulting in nerve damage [49].
Test results in specific GBS subtypes show the presence of antiganglioside antibodies. These antibodies result in complement activation and subsequent injury of the nerve via the target of specific gangliosides present on neuronal membranes [11]. In particular, anti-GM1 antibodies are common in the acute motor axonal neuropathy (AMAN) variant of GBS, and anti-ganglioside Q1b (GQ1b) antibodies in the Miller Fisher syndrome variant, which manifests as ophthalmoplegia, ataxia, and areflexia [7, 11].
GBS is a spectrum of pathological subtypes, mainly distinguished by the degree of nerve injury from relatively benign to severe weakness and respiratory distress [50]. The acute inflammatory demyelinating polyneuropathy (AIDP) subtype is an immune-mediated demyelination wherein the peripheral nerve myelin sheath is damaged, thus causing slow motor nerve conduction velocities. However, the AMAN subtype is defined by direct degeneration of the axons without prominent demyelination, but with frequent anti-GM1 antibodies and preceding Campylobacter jejuni infections [51]. Antiganglioside antibody binding to neuronal membranes starts a cascade of reactions resulting in membrane attack complex (MAC) formation [10]. For its part, the MAC disrupts the neuronal membrane integrity and causes cellular damage with subsequent clinical manifestations of GBS [10, 21]. Recent studies have demonstrated how gut dysbiosis and increased intestinal permeability influence immune responses in GBS, opening up new possibilities for microbiome-targeted therapeutic interventions and diagnostic biomarkers. More research is required to confirm these associations. Comprehending these intricate relationships holds potential for personalized medicine strategies in autoimmune neuropathies [10].
The demyelination process impairs nerve signal transmission—a key pathophysiological hallmark of GBS. Early, common symptoms, such as limb weakness and difficulties with swallowing and breathing emphasizes the importance of early detection and treatment. This highlights the severity of untreated GBS, which can progress to flaccid paralysis [6]. The hallmark of GBS is progressive, symmetric, symptomatic muscle weakness, beginning in the legs and progressing proximally [6]. Patients frequently complain of paresthesia and numbness preceding or accompanying the weakness [52]. The most common cranial nerve involvement includes facial weakness and also bulbar symptoms of dysphagia and dysarthria [52]. In some instances, autonomic dysfunction can occur as fluctuations in blood pressure, cardiac arrhythmias, and failure of sphincters [53]. In severe cases, respiratory muscle weakness necessitates ventilatory support [20, 23].
Since not all GBS patients have antecedent infections, and the definition of molecular mimicry for autoimmune disease is controversial, additional contributory factors are involved. There may be a genetic predisposition associated with increased susceptibility, and some human leukocyte antigen (HLA) types may predispose a person to increased risk. Although the onset of this disease is unpredictable, it may also be influenced by environmental factors, such as the strain of the infecting pathogen or the host’s immune status [48].
GBS is primarily driven by immune dysregulation following infections. From a functional perspective, several underlying triggers may contribute to this immune dysfunction, including gut dysbiosis and increased intestinal permeability, chronic viral persistence, heavy metal or environmental toxin exposure, stress-induced cortisol imbalance, nutrient deficiencies (e.g., vitamin D, B12, omega-3s), and, in rare cases, vaccinations [11, 54]. These factors collectively disrupt immune tolerance and regulatory pathways, increasing the risk of autoimmune reactions targeting the nervous system [55, 56]. Identifying and addressing these root causes through targeted diagnostics and integrative therapies can support immune balance and neurological recovery [57].
In the two weeks preceding GBS, the virus usually causes an infectious illness [11, 53]. The most commonly documented antecedent is Campylobacter jejuni infection, but cytomegalovirus, Epstein-Barr virus, influenza, and Mycoplasma pneumoniae have also been reported [11, 53]. Recent studies have begun to explore a potential association with SARS-CoV-2 infection; however, insufficient evidence exists to clarify the mechanisms by which these diseases may be causally linked. Other risk factors, aside from recent vaccination or surgical procedures, are scarce [11, 58].
Specific investigations support the diagnosis of GBS, which is primarily clinical. Albuminocytological dissociation is often identified through lumbar puncture, characterized by elevated CSF protein levels without pleocytosis [58, 59]. Nerve conduction studies help confirm the diagnosis and distinguish subtypes [58, 59]. Other potential causes of acute flaccid paralysis can be ruled out with MRI [6].
Although nerve conduction studies and lumbar puncture with albuminocytologic dissociation are the gold standards for diagnosing GBS, early testing can yield false-negative results, requiring repeat evaluations. Although MRI requires substantial resources and may not always be available, it is essential for ruling out SCC and other mimics. To prevent misdiagnosis, clinicians should integrate clinical findings with diagnostic tests and consider alternative diagnoses early in the presentation, such as SCC when interpreting MRI images [58–60].
GBS is often self-limiting with appropriate treatment, but it may lead to serious neurological, respiratory, autonomic, and psychological complications, particularly in severe or untreated cases. In approximately a quarter of the patients, severe complications occur, including respiratory failure due to diaphragmatic weakness, which may require mechanical ventilation [33]. Consequently, autonomic dysfunction may lead to life-threatening cases of cardiac arrhythmias and blood pressure instability [60]. Another risk of prolonged immobility is deep vein thrombosis and pulmonary embolism. However, chronic pain and fatigue can last and negatively impact long-term quality of life [58].
Neuromuscular complications include persistent weakness or paralysis, and many patients experience chronic neuropathic pain [58]. Additional issues such as bladder dysfunction and significant psychological distress, including anxiety, depression, or post-traumatic stress, may occur [58]. Furthermore, about 5–10% of patients may progress to chronic inflammatory demyelinating polyneuropathy (CIDP), necessitating long-term immunotherapy [9]. Early recognition and management of these complications are essential to optimize outcomes and prevent long-term disability (Table 7).
| Test name | Description | Purpose |
|---|---|---|
| Spinal tap (lumbar puncture) | Needle aspiration of cerebrospinal fluid (CSF) from the lower back | To detect elevated protein levels in CSF, often without increased white blood cells |
| Electromyography (EMG) | Thin needle electrodes are inserted into the muscle to measure electrical activity | To evaluate electrical activity and confirm nerve involvement |
| Nerve conduction studies | Electrodes were placed on the skin to stimulate nerves with small electrical impulses | To assess the speed and strength of signals traveling through the nerve |
| Blood test | To detect infections and inflammatory markers | To rule out other causes and detect recent infection, like Campylobacter jejuni |
| Magnetic resonance imaging | Imaging of the spinal cord and nerves | To exclude other conditions that mimic GBS |
Early immunomodulatory therapy with IVIG or plasmapheresis is crucial for reducing disease severity and improving outcomes [61, 62] (Table 8). In Table 8, * indicates supportive care, which is the main responsibility of the nurses and includes continuous monitoring of respiratory function with early intervention for respiratory failure, managing autonomic dysfunction, and preventing deep vein thrombosis caused by physical inactivity [63, 64]. Other nursing interventions involve maintaining the airway, ensuring proper nutrition, and preventing pressure ulcers. Both nurses and doctors should provide psychological support to patients and families during the recovery process [59]. Therefore, this table highlights the specialized services offered only by nurses. Nurses are in a privileged position to help patients and their families go through this healing journey.
Medical and nursing management of Guillain-Barré syndrome (GBS), which can broadly be categorized as primary disease and supportive treatments [1, 3, 5, 6].
| Medical management | Nursing management |
|---|---|
|
|
* indicates supportive care, which is the primary responsibility of the nurses.
This scoping review identified 19 key studies that collectively underscore the complexity of diagnosing acute paraplegia. While the number of included studies is relatively small compared to broader neurological reviews, this specific subset represents the highest quality evidence currently available for the emergency context. The scarcity of integrated protocols for differentiating GBS from SCC in the first 24 h is a significant finding in itself. It highlights a critical gap in the current literature and supports the urgent need for the development of unified, multidisciplinary diagnostic pathways.
A multidisciplinary team approach, including doctors, nurses, physiotherapists, and psychologists, is essential to provide GBS and SCC patients with optimal care. Nurses play a vital role in continuously monitoring skin integrity, nutrition, psychological support, respiratory status, and autonomic function. While psychological counseling addresses the anxiety and depression common in these conditions, early rehabilitation interventions led by physiotherapists support functional recovery. Standardized nursing care procedures and educational initiatives help reduce complications and improve patient outcomes.
In summary, the first-line immunotherapy includes either IVIG or plasmapheresis (plasma exchange), both of which are equally effective when initiated early in the disease course. IVIG is typically administered at a dose of 0.4 g/kg/day over five consecutive days and works by neutralizing pathogenic autoantibodies and modulating immune responses [61, 62, 65]. Alternatively, plasmapheresis involves 4 to 6 plasma exchanges over 1 to 2 weeks to physically remove circulating autoantibodies and immune complexes responsible for nerve damage [65, 66]. Clinical trials and meta-analyses have confirmed the efficacy of both therapies in shortening the time to recovery and reducing disease severity, though combining them offers no additional benefit [67].
It is crucial to identify and address root causes such as infections, gut dysbiosis, toxic exposures, and nutrient deficiencies through targeted testing. Natural immune-modulating agents like omega-3 fatty acids, curcumin, vitamin D, and glutathione [67] are used to reduce inflammation and oxidative stress. In contrast, B vitamins, magnesium, and coenzyme Q10 (CoQ10) support nerve repair and mitochondrial function. Gut healing with probiotics, L-glutamine, and dietary interventions helps to restore immune tolerance [67]. Integrative medicine approaches, particularly yoga combined with Ayurvedic therapies, have shown promising results in enhancing motor and sensory recovery in GBS, as they significantly improve muscle strength, sensory function, balance, daily living activities, sleep quality, and psychological well-being [68, 69]. Neurodegenerative supplements such as alpha-lipoic acid and lion’s mane mushroom may support neurodegeneration and enhance functional recovery [70].
The limitation of this review to English-language studies and the potential for publication bias are its main limitations. The variety of study designs, the small sample sizes in case reports, and the absence of randomized trials in some fields hamper evidence synthesis. Notably, there is a dearth of high-quality information regarding the effects of novel therapies on quality of life and long-term rehabilitation outcomes. To close these gaps, longitudinal and interventional studies should be given top priority in future research. A key finding of this scoping review is the paucity of literature directly addressing the emergency differentiation between GBS and SCC. Only 19 studies met the inclusion criteria, suggesting that while these conditions are well-studied individually, there is a lack of integrated research focusing on their simultaneous consideration in acute care settings. This highlights the urgent need for future prospective studies to develop unified diagnostic protocols for acute paraplegia in the emergency department.
These include evaluating minimally invasive surgical techniques in SCC, optimizing immunotherapy protocols, investigating integrative treatment approaches, and validating early diagnostic biomarkers for GBS. It’s also crucial to conduct extensive epidemiological research to inform clinical recommendations tailored to a given area.
SCC is an oncologic emergency with potentially irreversible consequences if treatment is delayed. This overview of updated research in SCC aims to support patient management, especially in light of ongoing controversies. It is essential to recognize that both surgery and radiotherapy should be considered as treatment options, and the conventional 24–48-h therapeutic window may not be absolute. Clinicians should prioritize treatment options and give patients the benefit of the doubt. Multidisciplinary collaboration is crucial for the rehabilitation of these patients.
GBS is a medical emergency that requires early diagnosis and often hospitalization, especially to monitor for respiratory failure and autonomic instability. Treatment aims to halt the immune attack, support vital functions, and promote neurological recovery.
AI: artificial intelligence
AMAN: acute motor axonal neuropathy
CSF: cerebrospinal fluid
GBS: Guillain-Barré syndrome
GM1: ganglioside M1
IVIG: intravenous immunoglobulin
MAC: membrane attack complex
MESCC: malignant epidural spinal cord compression
MRI: magnetic resonance imaging
PRISMA-ScR: Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews
SBRT: stereotactic body radiotherapy
SCC: spinal cord compression
SINS: Spinal Instability Neoplastic Score
VCFs: vertebral compression fractures
OA: Conceptualization, Investigation, Formal analysis, Writing—original draft, Data curation, Writing—review & editing. M Subih: Conceptualization, Investigation, Formal analysis, Writing—original draft, Data curation, Writing—review & editing. LS: Conceptualization, Investigation, Formal analysis, Writing—original draft. M Stublla: Conceptualization, Investigation, Formal analysis, Writing—original draft, Data curation, Writing—review & editing. DB: Writing—review & editing. PT: Conceptualization, Investigation, Formal analysis, Writing—original draft, Data curation, Writing—review & editing. OS: Writing—review & editing. AEEAE: Writing—review & editing. All authors read and approved the submitted version.
Patricia Tai, who is the Editorial Board Member and Guest Editor of Exploration of Medicine, and Omar Alqaisi, who is the Guest Editor of Exploration of Medicine, had no involvement in the decision-making or the review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
All relevant data is contained within the manuscript.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1503
Download: 14
Times Cited: 0
