Affiliation:
1Department of Biotechnology, School of Applied and Life Sciences, Uttaranchal University, Dehradun 248007, Uttarakhand, India
†These authors contributed equally to this work.
ORCID: https://orcid.org/0000-0002-5688-9860
Affiliation:
1Department of Biotechnology, School of Applied and Life Sciences, Uttaranchal University, Dehradun 248007, Uttarakhand, India
2Department of Zoology, School of Applied and Life Sciences, Uttaranchal University, Dehradun 248007, Uttarakhand, India
3Uttaranchal Institute of Pharmaceutical Sciences, Uttaranchal University, Dehradun 248007, Uttarakhand, India
†These authors contributed equally to this work.
Email: par.yadav2011@gmail.com
ORCID: https://orcid.org/0000-0002-5971-8258
Affiliation:
4Department of Zoology, Kalindi College, University of Delhi, New Delhi 110008, Delhi, India
5Department of Biotechnology Engineering and Food Technology, Chandigarh University, Mohali 140413, Punjab, India
ORCID: https://orcid.org/0000-0002-7437-0755
Affiliation:
6Institute of Pharmaceutical Research, GLA University, Mathura 281406, Uttar Pradesh, India
ORCID: https://orcid.org/0000-0002-6980-6761
Affiliation:
7Department of Nuclear Medicine, School of Medicine, Kyungpook National University, Daegu 41944, Republic of Korea
8Cardiovascular Research Institute, Kyungpook National University, Daegu 41944, Republic of Korea
Email: prakashg@knu.ac.kr
ORCID: https://orcid.org/0000-0002-0658-4604
Explor Med. 2026;7:1001399 DOI: https://doi.org/10.37349/emed.2026.1001399
Received: November 11, 2025 Accepted: April 03, 2026 Published: April 20, 2026
Academic Editor: Xiaofeng Wang, Cleveland Clinic Lerner College of Medicine of Case Western Reserve University, USA
The involvement of the Internet of Things (IoT) technology and artificial intelligence (AI) in the matter of maternal healthcare has allowed monitoring pregnant women in real-time and predicting poor pregnancy outcomes, including stillbirth and premature birth. Nevertheless, to diminish the risks of devices, the introduction of such technologies has to be accompanied by harsh safety monitoring programs. Materiovigilance, as the systematic sensing and monitoring of adverse events associated with medical instruments, is also crucial to patient safety in high-risk obstetric environments. Wearable sensors, e.g., fetal Dopplers, smart fabrics, and adhesive patches, have enhanced the prediction of stillbirth by offering continuous acquisition of physiological data but presents a hazard of ill effects if not controlled adequately. The AI introduction into the sphere of Materiovigilance enhances regulatory conformity, real-time decision-making, and raises the possibility of risk identification. Despite the massive potential, issues such as inaccurate data, poor infrastructure, and underreporting persist, particularly in low- and middle-income countries. In such circumstances, the Materiovigilance Programme of India is another staged effort to enhance the device safety supervision and reporting systems. Engagement of different stakeholders such as clinicians, engineers, regulatory agencies, and technology developers provides an opportunity to secure the safety and efficacy of AI-equipped medical devices. Enhancement of Materiovigilance systems is required to preserve proper maternal-fetal health and sustain clinics in digital obstetrics, as any error in maternal-fetal monitoring may lead to preventable death.
Maternal, neonatal, and child health have always been a top priority in global public health programs. The maternal and child mortality reduction rates have been assigned the utmost to any of the movements such as the sustainable development goals (SDGs) and the Millennium Development Goals (MDGs) that establishes the vital criteria of efficiency and equality of the health system. Nevertheless, despite this, stillbirth, an excruciating and largely preventable outcome, has long been a significantly understudied phenomenon both among researchers and policymakers, despite carrying with them a colossal emotional, social, and financial burden on the families of affected families [1]. The two differences in the definition of stillbirth by the World Health Organization (WHO) and the International Classification of Diseases (ICD) are that they both use the state that the fetus has no signs of life once it has been completely expelled/extracted by the mother, even at any stage of gestation. Stillbirths have been commonly defined as deaths of the fetus at or after 20–28 weeks of gestation, whereas an earlier loss is referred to as miscarriage [2]. It is estimated that more than 1.9 million stillbirths are taking place annually across the globe, with most of them being in the region of South Asia and sub-Saharan Africa [3]. The stillbirth rates are a sensitive indicator of the quality of healthcare at large and availability of the healthcare to implement interventions in time, labor conditions as well as prenatal care.
The developments in artificial intelligence (AI) and the Internet of Things (IoT) have led to the emergence of new trends in forecasting the risk of stillbirth through data-based modelling and continuous physiological monitoring. Machine learning (ML) methods such as ensemble, random forest, neural network have great accuracy in predictions when applied on large obstetric data sources [4–6]. An extensive evaluation conducted by Li et al. [7], based on data regarding over 14 million pregnancies, demonstrates that ML algorithms can be an accurate method of predicting stillbirth and, therefore, help in the early identification of risks and intervention. Moreover, combining maternal characteristics and biomarkers has proved to have an excellent predictive power with an area under the curve (AUC) reaching as high as 0.984, especially in cases of mean arterial pressure (MAP), uterine artery pulsatility index (UtA-PI), and placental growth factor (PlGF). This is more so when the models are applied in the first trimester, which is the most essential period in terms of preventive care [8]. Similarly, a study has shown that incorporation of mothers, newborns, and obstetrics risk factors may lead to accuracy in predicting up to 0.99 AUC, in distinguishing live births and stillbirths [9]. Although those predictive frameworks have enormous potential, they require high-level monitoring to prove their safety, reliability, and clinical validation of the implemented IoT devices and algorithms used in maternal health. At that, it is necessary to introduce a planned course of surveillance and risk management encapsulating medical devices, or Materiovigilance. The ability to detect and fix any adverse episodes or failures in real-time is increasingly becoming crucial due to the growing reliance on wearable sensors on the body, intelligent wearable patches, and long-distance monitoring systems to monitor pregnancy. In the medical field of obstetric care, poor functioning of devices, faulty interpretation, or late notification may be disastrous and potentially fatal to both the mother and the fetus.
Enhancing the regulatory system of medical device safety is the objective of the Materiovigilance Programme of India (MvPI) and other countrywide programs, in particular, in resource-scarce settings [10]. Healthcare systems can ensure that obstetric technology innovation is aligned with patient safety and legal and ethical accountability in combination with Materiovigilance and predictive care based on AI.
Finally, cutting the stillbirth rates through early detection and intervention occurs with a revolutionary opportunity due to the combination of AI, IoT, and Materiovigilance. But this should go hand in hand with intensive device monitoring, interdisciplinary collaboration, and a robust regulatory framework to provide assurance of safe, equitable, and successful care of the fetus and mother.
The global stillbirth rate, assessed in 2021, showed 1.9 million babies who died after 28 weeks of pregnancy, reaching 13.9 per 1,000 total births. The differences in stillbirth risk rates between nations with low and high prevalence are significantly substantial, as risk is exposed twenty times more in countries with higher rates. India experienced the maximum stillbirths while Pakistan and Nigeria, along with the Democratic Republic of the Congo, Ethiopia, and Bangladesh followed. The distribution of stillbirths appears uneven throughout the regions of particular countries since areas outside urban centers have documented a higher incidence. Sub-Saharan Africa and Southern Asia account for three-quarters of all stillbirths, with a stillbirth rate seven times higher in sub-Saharan Africa than in Europe, North America, Australia, and New Zealand [11]. A global analysis by Hug et al. [12] from 2019 estimated that 2.0 million newborns experienced stillbirth at ages 28 weeks or above, thus producing a global measurement of 13.9 stillbirths per 1,000 total births. The rates of stillbirth presented different figures between regions and eastern and southern Africa, along with South Asia, recorded the second and third highest numbers. The global stillbirth reduction rate declined at an annual average of 2.3 per 1,000 births between 2000 and 2019, although these numbers remained lower than the annual reduction rates of the neonatal mortality rate (29%) and mortality rate among 1–5 month old children (43%) [12]. The experience of stillbirth negatively impacts parental mental well-being and the overall emotional state of families at large. Studies reveal that after perinatal loss, including stillbirth, women from low- and middle-income nations present depressive symptoms to the extent of 41% on average. The importance emerges to provide complete grief care and mental health services to families who have been affected [13].
Stillbirth, defined as the intrauterine death of a foetus at or after 20–28 weeks of gestation or later, remains a significant global health concern with multifactorial etiologies. Recent studies have identified several clinical risk factors associated with an increased likelihood of stillbirth.
The risk of antepartum stillbirth proves independently significant toward maternal age because women between 35–39 years face higher probabilities of this occurrence. Risk factors that significantly increase antepartum stillbirth include smoking along with having never given birth and hypertension and diabetes prior to pregnancy and non-smoking. The 40+ age group among pregnant women faces antepartum stillbirth risks that amount to 1 in 455 continuing pregnancies after 40 weeks of gestation, but this number stands at 1 in 1,177 for women under 40 years old. The most prevalent type of unexplained stillbirth occurs while perinatal infections cause more stillbirths in women who surpass age 40 [14].
Although the study by Boo et al. [15] indicate that incidence of stillbirth is associated threefold more with severe anaemia during pregnancy (aRR: 3.37; 95% CI: 1.97–6.11), according to the Indian cohort research; yet post multivariate analysis stillbirth in non-significantly associated with BPL and parity.
Stillbirth is significantly influenced by conditions like gestational hypertension and pre-eclampsia. Chronic hypertension and a history of pre-eclampsia were linked to an increased risk of stillbirth, according to a research done in Jakarta, Indonesia [8]. Two important risk factors include low birth weight (LBW) and preterm birth. According to multivariate analysis, stillbirth was strongly correlated with both preterm birth (aOR: 0.35; 95% CI: 0.16–0.74) and LBW (aOR: 0.09; 95% CI: 0.04–0.23) [16].
A stillbirth can occur because of both congenital birth defects combined with genetic health conditions. Congenital abnormalities served as the cause of 16.1% of early new-born fatalities and stillbirths, according to case-control data from tertiary institutions in Addis Ababa, Ethiopia. Prenatal screening together with genetic counselling demonstrates their worth in identifying pregnancies that face high risk through these research results [17]. Other risk factors include environmental and behavioral risk factors.
In a second effort to clarify the genetic landmarks linked to stillbirth, an organized discovery of reported maternal, paternal, placental, and fetal genes was conducted through the GeneCards database. The retrieval of the genes involved was done through pertinent keywords connected to the complications of pregnancy, fetal development, placental dysfunction and paternal genetic contribution. Duplicates were eliminated and genes were sorted according to biological origin and reportedly useful functions in pregnancy outcomes. The four sets of genes have been comparatively analyzed to identify distinctive and shared genetic factors. Figure 1 shows the distribution of genes as maternal, paternal, placental and fetal. It is also important to note that a significant percentage of the genes were only related to fetal expression, indicating that genetic control of the fetus plays a dominant role in pregnancy maintenance. Nevertheless, some considerable overlap of genes was found in all four groups, which may indicate common molecular pathways that can mediate these maternal-fetal interactions and placental development. The overlapping genes could be important regulatory centres in angiogenesis, immunomodulation, oxidative stress response, mitochondrial activity and cellular apoptosis—processes influencing the adverse pregnancy outcome and stillbirth significantly. The complete list of identified genes is found in Table S1 (Maternal), Table S2 (Paternal), Table S3 (Fetal), & Table S4 (Placental), containing the list of the gene symbols, the description of the gene, and the categories of the type. The applicability of this integrative genetic profiling is that it provides a framework that establishes a baseline to help the researcher identify a candidate biomarker and a molecular target that can support the methods of predictive modeling and risk stratification in stillbirth research.
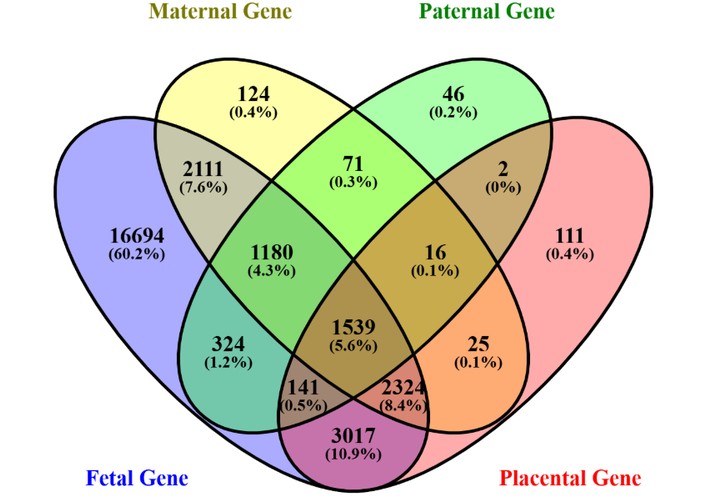
Venn diagram representing the distribution and overlap of reported maternal, paternal, placental, and fetal genes extracted from the GeneCards database.
A comparative analysis of different AI models for the prediction of stillbirth is shown in Table 1 and Figure 2. Models random forest (RF), gradient boosting (GB) and extreme GB (XGBoost) showed the highest accuracy, sensitivity, and specificity in prediction.
List of models used for the prediction of stillbirth.
| Model name | Accuracy | Sensitivity (recall) | Specificity | References |
|---|---|---|---|---|
| Blender model | 95.90% | 91.6% | NS | [18] |
| XGBoost | 96.70% | 25% | 90% | [7] |
| GB classifier | 95.24% | 91% | 88% | [19] |
| RF | 85.0% | 92.2% | 77.9% | [9] |
| 96.8% | 96.3% | 97.2% | [20] | |
| RF, LR, GB, XGBoost | 99.72% | 96.67% | 99.6% | [21] |
| Stacking ensemble (SE) model | 90% | 91% | 88% | [19] |
NS: not specified; GB: gradient boosting; RF: models random forest; XGBoost: extreme GB.
The high incidence of stillbirth throughout the worldwide population, particularly in regions with limited financial means, underlines the pressing need for continuous risk assessment solutions that monitor pregnant women persistently [12]. Standard prenatal treatment based on routine check-ups fails to detect brief yet evolving foetal problems between testing sessions before stillbirth occurs. RT stillbirth prediction and prevention at a new level can be enabled through the combination of AI technology with the IoT network [22].
These systems establish feedback processes that transform AI analysis outcomes into clinical warning signals for medical staff use. When built into digital healthcare platforms, warnings help decrease the interval between physiological deterioration and the clinical reaction to these changes [23]. The system can start both automated treatment suggestions with remote consultations or backup in-person patient interventions. Pilot studies demonstrate that this technological integration leads to improved diagnosis of foetal growth restriction and preeclampsia since these pathologies create significant stillbirth risks [24].
Edge computing implements localized decision-making and reduces latency, which proves vital for situations that have limited resources or occur in remote areas. Full-headed prenatal care ecosystems will require a mobile health platform and electronic health record interoperability as more systems become operational [25]. In conclusion, the combination of IoT and AI technologies offers a strong, scalable, and predictive foundation for preventing stillbirths. Table 2 summarizes existing AI-IoT integration frameworks applied to stillbirth monitoring and Materiovigilance, highlighting their architectures, data acquisition methods, analytical capabilities, and clinical relevance. These solutions pave the path for a new age of predictive, preventative, and precision-oriented prenatal care by improving clinical attentiveness and providing timely information to pregnant moms and caregivers.
AI-IoT integration frameworks for stillbirth monitoring and Materiovigilance.
| Framework layer | AI-IoT components | Role in stillbirth monitoring | Role in Materiovigilance & risk reduction | Representative references |
|---|---|---|---|---|
| Sensing layer | Foetal movement, pressure, phonocardiography, ECG/PPG wearables. | Continuous acquisition of foetal activity, foetal heart sounds, and maternal physiological messages. | Early detection of material breakdown, over-contact pressure and skin-device contacts. | [26, 27] |
| Data acquisition & IoT connectivity | BLE, Wi-Fi, IoMT gateways, wearables connected to the cloud. | Provides the transmission of maternal-foetal information in real-time to facilitate round-the-clock monitoring. | The remote device performance tracking, fault detection, and adverse event reporting are supported. | [22] |
| Edge intelligence layer | ML-based artefact removal algorithms, edge AI. | Reduces motion artifacts and improves fetal signal reliability. | Detects abnormal sensor behavior, drift, or hardware malfunction in early stages. | [28] |
| Cloud analytics layer | Time-series analysis, predictive models, and deep learning. | Determines abnormal foetal movements and early foetal distress. | Anticipates failure of devices, false positives, and material fatigue. | [29, 30] |
| Clinical decision support systems (CDSS) | Risk scoring, alerts, dashboards: AI. | Helps clinicians make decisions that can help the foetus and intervene. | Flags suspicious activity of devices and assists in reporting of regulations. | [31] |
| User interface layer | M-health applications, clinician portals, visualisations. | Enhances the comprehension of foetal health indicators to users. | Cuts psychological stress levels by cutting false alarms and enhancing transparency. | [32] |
| Materiovigilance & feedback loop | Adverse event detection under AI control, automated reporting. | Correlates maternal births to device performance statistics. | Facilitates active after-market monitoring, design efficiency, and safety assurance. | [33] |
| Security & privacy layer | AI in cybersecurity and anomaly detection, encrypted IoT protocols. | Secures confidential maternal/foetal health information. | Eliminates manipulation of data, unauthorized access, and safety hazards. | [34] |
AI: artificial intelligence; ECG: electrocardiography; IoT: Internet of Things; ML: machine learning.
The use of IoT devices has significantly enhanced the pregnancy monitoring system because today it is possible to constantly monitor key health parameters in real time. Wearable devices like smartwatches, foetal Dopplers, and patches stick onto a mother make easy data collection on her heart rate, blood pressure, the foetus’s movement, and activity in the uterus possible. Besides making monitoring of the homes easier, this innovation provides the medical staff with prompt data to manage potential health issues. Focusing on physiological indicators, the transmission of data, pre-processing, and the application of algorithms to the analysis of data, the paper discusses the existing research on wearable sensors, processing of physiological data, and AI during pregnancy monitoring. AI turns out to be a potent instrument for smart diagnosis, early detection, and long-term health in pregnancy monitoring [35]. A randomized controlled experiment with 200 pregnant women who needed to be monitored for disorders, including pre-eclampsia and fetal growth restriction, was carried out by Bekker et al. [36] (2023) as a practical application. Devices for blood pressure and cardiotocography (CTG) at home were given to the participants. In order to minimize the need for frequent hospital visits, the study showed that home-based tele monitoring is practical and capable of accurately tracking fetal heart rate (FHR) and uterine contractions [37].
Acceptance of wearable IoT devices in prenatal care revolutionized maternal health tracking through continuous remote and unobtrusive parameter monitoring (Figure 3). Through smartwatches alongside foetal Dopplers and adhesive biopatches medical staff obtain increased frequencies of vital health data including maternal heart rate, along with blood pressure readings, uterine activity measurements, and foetal movement observations. Wearables serve women with high-risk pregnancies especially well, since early detection of fetal abnormalities substantially reduces the risk of stillbirth.
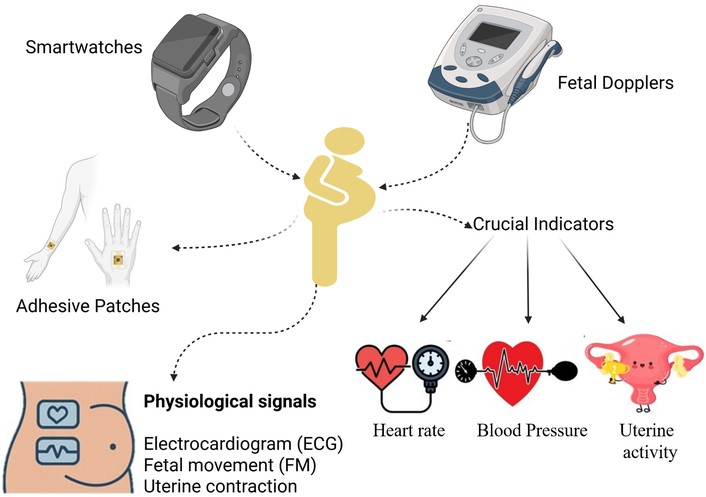
Illustration of sensor placement on a pregnant woman for maternal and fetal monitoring. Brief history of non-invasive and wearable devices used to monitor maternal and fetal health. Constant recording of physiological signals such as electrocardiography (ECG), fetal movement (FM), uterine contractions, heart rate, and blood pressure are done using smartwatches, adhesive biosensor patches, and fetal Dopplers. These critical signs allow tracking and prioritizing risks in the course of pregnancy.
Lei et al. [38] used a bioinspired mineral hydrogel to create a mechanically flexible, self-healing ionic skin sensor that detects small pressure changes. The development of technologies that mimic the mechanical flexibility and multifunctionality of human skin for accurate pressure detection was symbolized by this sensor, which might be very useful for ongoing pregnancy monitoring. Similarly, utilizing convex microarrays, Xiong et al. [39] developed a capacitive pressure sensor that is highly sensitive and flexible. This breakthrough demonstrated the sensor’s potential for monitoring UC throughout pregnancy as well as its use in health surveillance and human-robot interactions by tracking physiological signals and robotic hand motions. By creating an ultrasensitive textile pressure sensor for e-textiles based on conductive fibers, Lee et al. [40] made another noteworthy contribution to this field. The ability to integrate fabric and sensors without sacrificing sensitivity or flexibility opens the door for its use in wearable technology, including health monitoring devices. In their assessment of the development of contractive fiber-based flexible and stretchy physical sensors, Trung and Lee [41] emphasized the applications of these sensors in wearable human activity monitoring and personal healthcare. The importance of these sensors in developing sensor-integrated systems for RT health status monitoring was highlighted by their study, which also offers a potential path for pregnancy monitoring applications [41]. In 2018, the skin-like hierarchical polymer structure architecture was introduced to the triboelectric sensor, which increased the sensitivity and the capabilities to detect the pressures and movements. This structure resembles a human being in its tactical efficiency of sensing. Soft and stretchable electronics using liquid metals were reviewed to make a highly deformable and still conducting material for flexible electronics. With these materials, it is possible to generate sensors that can monitor the maternal and foetal health parameters during pregnancy [35, 42].
Inertial sensors are used to recognize body movement and poses, whereas the pressure sensors are used in the accurate recognition of the uterine contractions, determining labor progress and evaluating the possibility of its occurrence of complications. Such recent advancements as self-healing ionic skin sensors and textile sensors, made of conductive fibers, further improve the performance of such devices and their compatibility with wearable health-monitoring systems. Acoustic sensors used in fetal phonocardiography (fPCG) have developed into critical devices for contact-less pregnancy health evaluation. The device serves as a low-cost passive tool which detects acoustic heart vibrations made by the foetus to assess foetal health. The advancement of FHR tracking mainly focuses on sensor invention and signal processing technologies because it improves both measurement precision and signal dependability [35]. A breakthrough emerged through Charlier et al.’s [43] development of AcCorps which represents a low-cost 3D-printed acoustic sensor specifically designed for fPCG. The device improved acoustic amplification optimization in low-frequency bands because this capability is fundamental to detecting foetal heart sounds. The potential benefits of FHR monitoring accuracy improvement were confirmed after testing with pregnant volunteers and test bench setups and silico-based research [43]. Khandoker et al. [44] designed a low-cost, non-invasive fPCG device that uses four piezoelectric sensors to record the fetal heart’s noises. It offered a promising substitute for conventional fetal-monitoring techniques and showed a strong correlation (r = 0.96) with fetal echocardiogram (fECG) readings. The study highlighted how fPCG might enhance prenatal care by providing a dependable and easy way to track FHR, especially in environments with limited resources. Researchers have investigated multiple technologies for expanding pregnant health monitoring areas by integrating sound sensors [43]. Uterine contractions and FHR are measured via the abdomen sensor. This sensor is positioned directly beneath the umbilicus [45]. Foetal movement is a key marker of foetal activity, and research has detected fetal movement (FM) signals from a pregnant woman’s belly using a multipoint IMU. With an IMU sensor at each corner, a circuit for signal processing, a circuit for a rechargeable battery, and a secure digital memory card, the device had a triangular form. The device’s softness was increased by using a flexible flat wire as the signal transmission line and a thermoplastic elastomer enclosure. By analyzing signal energy and determining the signal interval, the study discovered that FM transmissions could be identified instantly [45, 46]. Qin et al. [47] developed a wearable device with three-axis acceleration sensors to detect FM along with acoustic sensing components. The system adopted a total solution for FM surveillance, which aimed at creating uninterrupted, precise evaluation tools while eliminating conventional monitoring barriers. Aravindan et al. [48] presented more instances of fPCG applications in remote monitoring devices. Electrohysterography served as their monitoring modality, yet they centered their work on building a wireless wearable device that included fPCG detection through acoustic sensing technology. These advancements highlight how sensor technology is changing, particularly in the area of pregnancy monitoring. Researchers are opening the door to more accurate, complete, and easily available healthcare solutions by utilizing advancements in sensor design and integration. These developments hold promise for improving maternal and foetal health monitoring, facilitating the early identification of any problems, and enhancing overall healthcare results.
In spite of the fact that wearable pressure sensing, motion, and acoustic sensing technology has shown a lot of potential in continuous health and maternal-foetal monitoring, there are several physiological, technological and psychological risks associated with their extensive and sustained use that must be taken into account (Figure 4) [49]. The phenomenon of long-term skin contact with flexible substrates, conductive fibres and polymer-based micro structured materials, which are commonly used in capacitive, triboelectric, and textile based sensors, can lead to the undesired dermatological outcomes such as discomfort, allergic contact dermatitis or skin irritation, particularly in wet conditions or in the context of long-term monitoring [50]. Pregnant women can be more susceptible to these dangers because of their heightened skin sensitivity and straining of the abdomen. Moreover, unless sensor thickness, stiffness gradients, or fixation pressure are properly adjusted, repetitive mechanical loading of very sensitive pressure sensors or tight fixation may result in local tissue compression, pain, or disturbed microcirculation [51].
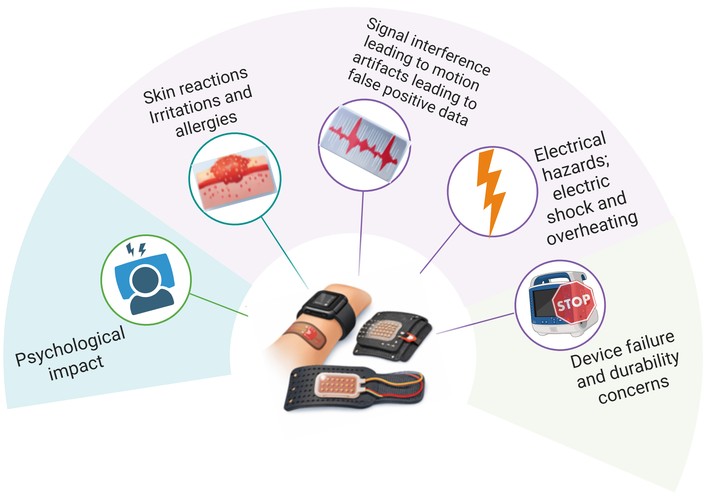
Schematic illustration summarizing the major risks related to prolonged use of wearable sensors. These involve skin responsiveness such as irritation and allergic response caused by sustained skin contact; signal interference caused by environmental noise and body movement, leading to motion artefacts and false-positive results; electrical hazards, e.g., the risk of electric shock and overheating during prolonged use; component failure and wear problems, e.g., mechanical stress, material wear or repetitive use; psychological effects, e.g., increased anxiety caused by a false alarm or a non-linear feedback. The main character displays a typical set of wearable devices, whereas the background items reveal the main types of risk that are essential in the safety assessment and Materiovigilance of wearable healthcare systems.
Motion artifacts, sensor motion, and environmental interference of wearable sensors are especially susceptible to signal integrity in terms of compromising data quality and reliability [52]. False positives or failure to detect foetal heart sounds or movements in foetal monitoring systems will be caused by inaccurate detection of foetal cycles or movements by maternal motion, maternal respiration, or improper placement of sensors [53]. This may cause maternal anxiety or wrong clinical interpretation. There are also more concerns regarding the use of automated signal processing and ML algorithms in relation to the problem of algorithmic bias, overfitting, and insufficient validation on a variety of demographics, which could limit generalizability and clinical reliability [54]. Also, material and electrical safety remain a significant concern, particularly when it comes to wearable devices that can be used over an extended period or at home [55]. The issues of electrical leakage, overheating of the device, or the reduction of sensing work can be caused by uneven fabrication quality, material degradation, or insulation breakdown of the low-cost textile-based, fiber-integrated, or 3D-printed sensors over time [56]. Moreover, it is hard to assure the safety, reliability and reproducibility among users and situations of use since numerous wearable foetal monitoring systems are not thoroughly longitudinally clinically validated and not all of them are regulated in a uniform manner.
Finally, one should pay attention to behavioral and psychological hazards. Continuous self-monitoring, facilitated by wearable technology, can be a source of dependence among its users or anxiety associated with health, especially when the results of the devices are ambiguous or conflicting with those of the clinician. False alarm or inaccurate feedback of foetal movement in maternal health monitoring may result in improper consultation with the doctor and unnecessary stress [57]. To minimize the risks and provide the safe implementation of wearable technologies in healthcare practice, each of these possible adverse effects highlights the user-centered design, strict biocompatibility testing, consistent signal processing, and comprehensive clinical validation.
The AI-IoT-enabled clinical trials and studies table underscores a rapidly growing field of IoT sensing technology and AI analytics convergence in enhancing the ability to predict stillbirth and maternal-foetal health through the use of AI analytics and IoT sensing technology. To enhance real-time surveillance and Materiovigilance of monitoring hardware performance, which is required to effectively train AI models to identify and understand physiological patterns associated with adverse outcomes, such IoT devices will be able to gather comprehensive vital indicators, including foetal heart rate, foetal motion, and factors of the mother. A number of trials (Table 3), including ones evaluating wearable foetal electrocardiography (ECG) patches and in-home non-stress foetal monitor devices, have proven this. Recent studies confirm that wearable sensors in combination with AI-based algorithms can achieve a considerable advancement in early detection and classification of foetal health conditions to address the issues of signal noise and artefacts of maternal movement with the help of advanced feature extraction and ML techniques [58, 59]. Concerning predictive analytics, the articles presented in the table approximate the likelihood of stillbirth and other related problems based on multimodal data streams and ML algorithms and computational systems, including deep learning and hybrid optimization approaches. Even though systematic reviews highlight the need for additional prospective, externally validated models that are integrated into clinical workflows to create trust and clinical utility, these AI models that are trained on large datasets have performed positively in classifying high-risk pregnancies. The convergence of IoT and AI is also accompanied by wider attempts to deploy edge-AI models offering real-time analysis and decision support, especially in the resource-constrained settings where conventional healthcare resources are constrained. As an example, predictive and reconstruction techniques that rely on transformers to predict and recreate foetal heart rate signals can enable strong AI capabilities to be placed directly in wearable technology to provide early alerts and intervention. Taken together, this literature does not only underscore the technical feasibility of AI-IoT systems in managing maternal-foetal health, but also demonstrates that the system still exhibits gaps that should be addressed, including standardisation, explainability, and massive validation [60].
Integration of clinical trial data within an AI-IoT architecture for predictive stillbirth risk and Materiovigilance.
| Study/Trial name | Clinical trial ID | Study type | IoT/Sensing technology | AI/Predictive modelling approach | Gestational age range | Primary outcomes | Relevance to Materiovigilance & stillbirth risk | References |
|---|---|---|---|---|---|---|---|---|
| Pregnancy, Preterm Birth & Stillbirth Outcomes Study | NCT02738892 | Observational cohort | Hospital EHR, fetal monitoring systems | Statistical risk modelling, multivariate analysis | All trimesters | Stillbirth, preterm birth | Foundational dataset for AI-based stillbirth risk models | [61, 62] |
| Modified Biophysical Profile | NCT03729089 | Interventional | Ultrasound, CTG sensors | Threshold-based + predictive analytics | ≥ 28 weeks | Adverse fetal outcomes | Enhances sensor-driven risk stratification | [63] |
| In-Home Non-Stress Fetal Monitoring Device | NCT07223996 | Interventional (device) | Home-based IoT FHR monitor | Signal processing, trend analysis | 28–40 weeks | Device safety, fetal well-being | Core Materiovigilance study for remote IoT devices | [64] |
| AI-Powered CURA™ Risk Screening App | NCT06974188 | Interventional (AI) | Mobile health data, EHR integration | Machine learning risk scoring | All trimesters | Identification of high-risk pregnancy | Demonstrates clinical deployment of AI decision support | [65] |
| Remote Telemedicine Fetal Monitoring Study | NCT06366711 | Feasibility | Wearable IoT sensors, cloud platform | Time-series analytics | Late second–third trimester | Monitoring feasibility | Supports continuous surveillance and early warning systems | [66] |
| Monica Novii Wireless Patch System | NCT03223324 | Interventional (device) | Wearable fetal ECG IoT patch | Signal quality algorithms | ≥ 32 weeks | FHR accuracy and safety | Materiovigilance of wireless fetal monitoring devices | [67] |
| Umbilical Cord Abnormalities Study | NCT05901688 | Observational | Ultrasound imaging systems | Regression-based risk modeling | Mid–late pregnancy | Adverse fetal outcomes | Contributes anatomical predictors for AI models | [68] |
| PRISMA Maternal & Newborn Health Study | NCT05904145 | Observational | Integrated clinical data platforms | Predictive analytics | All trimesters | Fetal and maternal mortality | Population-level surveillance for stillbirth trends | [69] |
AI: artificial intelligence; CTG: cardiotocography; ECG: electrocardiography; IoT: Internet of Things; FHR: fetal heart rate.
Wearable sensing technologies have tremendous possibilities in continuing maternal-fetal monitoring but they require special risk-reduction methods that consider the technological, psychological and physiological challenges. Physiological issues relating to prolonged contact with the skin can also be prevented by working with biocompatible, breathable, and hypo-allergenic materials, such as medical grade silicones, hydrogel and moisture-wicking fabrics [70]. The complications of skin irritation, local tissue compression and poor microcirculation can be minimized through the design of sensors with ultra-thin profiles, graded stiffness and flexible mechanical structures. Periodic wear-time notices and adjustable fixation systems are also useful in avoidance of discomfort, particularly to pregnant users who have sensitive skin [71]. The wearable system must include advanced motion-artifact-removal methods, sensor fusion algorithms and adaptive filtering algorithms to deal with signal integrity and data reliability. Mistakes related to the maternal movement or improper attachment can be reduced by real-time signal quality indicators and guidelines on ergonomic positioning of sensors [72]. ML models should also be trained and tested on diverse populations, and performance should be reported publicly to reduce the algorithmic bias and enhance clinical generalization. Material and electrical safety can be minimised by strict quality control processes and by means of long-term encapsulation of the elements, and additional layers of insulation to prevent electrical leakage and overheating [73]. Standardized clinical evaluation systems and regulatory oversight can also help to improve device repeatability and dependability by performing long-term performance tests in the real world and following international medical device safety standards.
Lastly, the behavioral and psychological risks of continuous self-observation may be addressed in the design of user-friendly interfaces, which focus on meaningful feedback displayed in visual, context-specific terms instead of raw data. Clinician-guided thresholds, alert verification systems, and instructional support help to avoid false alarms, anxiety, and over-reliance. To ensure safe adoption, the wearable technologies need to be incorporated into the hybrid care models, in which automated monitoring supplements but does not substitute professional judgement. All of these strategies combined underline the importance of interdisciplinary design, comprehensive validation, and responsible execution in an attempt to extract the maximum benefits of wearable maternal-foetal monitoring systems as well as minimize any risk.
Materialovigilance denotes the technology, which utilizes AI and ML to monitor and control patient safety, has caused patient safety to be a far more desirable matter, such as recognizing patterns and anomalies in extensive amounts of data produced by medical devices, including social media and electronic health records. With the assistance of AI and learning, machines are able to analyze real-time records of medical equipment and identify errors and safety risks at the earliest. This is a proactive care that safeguards the patients, but also makes certain that there is an immediate reaction in the event of any negative occurrence, which in this instance is stillbirth. The algorithms and predictive analytics help find patterns and patient characteristics. The processes of using data and being very analytical have the potential to transform a healthcare process significantly, as they provide the possibility of improved diagnostic accuracy, signal identification, personalized treatment, patient outcome, and the decision-making process of Materiovigilance [74].
Patient safety is one of the major considerations in the utilization of medical devices and it requires effective risk management systems and continuous monitoring, such as Materiovigilance. Materiovigilance, statistical detection, evaluation, and prevention of unfavourable incidents involving medical equipment, has advanced significantly through global cooperation and technological development. The past few years have witnessed a significant enhancement of the Materiovigilance systems due to the implementation of AI and ML that can investigate the available data in real-time, identify the signs of safety at an early stage, and regulate the safety targets more efficiently, eventually leading to the enhancement of patient outcomes and adherence to safety standards. Programmes such as MvPI aim to enhance the Indian medical device monitoring network in developing countries such as India. Nonetheless, problems such as low quality of data, underreporting, and infrastructure constraints still persist. The utilization of AI and ML-based solutions has a high potential of overcoming these limitations by enhancing the reliability of data, predictive risk analytics, and decision-making efficiency. The use of AI-based clinical decision support systems and more complex predictive algorithms in the future, along with improvements in real-time patient monitoring, data traceability, and information security, upcoming technologies such as wearables and blockchain are continuing to develop the sphere of Materiovigilance. Also, allowing to reveal and fix issues related to devices as soon as possible, AI- and ML-enabled predictive analytics improve patient safety and encourage proactive adherence to regulations. In order to ensure the successful introduction of such innovative systems and in order to ensure the safety of the population in terms of technology-enabled methods of Materiovigilance, it will be essential to continue the collaboration between medical workers, engineers, regulatory organizations, and technology developers [75]. Materiovigilance is a critical component of healthcare policy that oversees and addresses safety issues associated with medical devices to ensure patient safety. Timely reporting, consistency, and adverse event identification, however, are challenging for traditional systems. AI can transform the concept of Materiovigilance, as it can improve data processing, real-time monitoring, and predictive analytics. The current paper reviews AI-IoT integration and how it can offer more safety to medical devices in the context to materialovigilance, focusing on how this technology can enhance patient safety, individualize medical devices, and reduce regulatory reporting. Ultimately, AI-based solutions have the potential to enhance patient outcomes by detecting adverse events, predicting patient decline, and providing personalized treatment regimens. It is also feasible to process big and complex datasets using AI and make proactive decisions and early alerts about new risks associated with medical equipment. AI will have a significant reduction of the administrative workload of healthcare professionals by computerizing routine processes and enhancing the accuracy of these processes. Moreover, the AI can enhance the post-market surveillance by identifying trends and abnormalities in real time, thus accelerating remedial measures. In order to ensure the proper development and use of AI in Materiovigilance, though, drawbacks such as algorithmic biases, data protection, and responsibility in ethics and law should be considered. To address these challenges and maximize the use of AI in healthcare, it is essential to have effective regulatory frameworks, transparency, and interdisciplinary collaboration [76].
A union between the AI-IoT-mediated medical technology and Materiovigilance has led to a paradigm shift in enhancing patient safety, particularly in maternity-fetal care. The increasing interest of obstetrics in wearable technologies and predictive algorithms to ensure the reliability, efficacy, and safety of the procedures requires complex monitoring systems. Materiovigilance plays an important role in this regard since it allows detecting and alleviating any issue related to the device at the initial stages of product development. Incorporating maternal demographic, clinical and biometric streams of wearable data, a variety of algorithms, such as RF, XGBoost, and GB classifier, have demonstrated accuracy approaching 100 percent or higher [20, 77] some hybrid models demonstrate up to 99.7 percent accuracy. With the application of such AI models within the structure of IoT, it may be possible to monitor maternal and fetal indicators in real-time, such as maternal blood pressure, uterine contractions, and FHR [78]. In this way, the emergence of the problems that are likely to lead to stillbirth can be detected in time. However, digital health systems create new risks, including delayed warnings, algorithm bias, data transmission failures, and sensor malfunctions, all of which can pose a risk to patient safety should they go unrestrained. This underlines the requirements of an efficient Materiovigilance system which evaluates the clinical effectiveness of combined AI-IoT systems in multiple real-life situations beyond the surveillance of adverse events. Nevertheless, the use of such digital health systems also introduces novel risks, including delayed warning, bias in the algorithm, transmission errors, and sensor failure, among others, and these risks to patient safety cannot be ignored. This indicates the importance of an effective Materiovigilance system to evaluate the clinical quality of combined AI-IoT systems and varied real-life situations besides tracking the undesirable events. To address such challenges, MvPI has expanded its scope to include the use of medical devices based on digital and AI. It is essential to offer these programs in a low-resource setting because wearable device technology is increasingly being used to offset the inability to attend prenatal care in person [79]. International regulations have evolved with these technological advancements. The European Union Medical Device Regulation (MDR 2017/745) mandates the continuous post-market clinical review and management of risk of software-integrated medical devices. In the U.S., algorithm transparency and performance monitoring in the real world also receive considerable focus in the FDA Digital Health Software Precertification Program [80, 81]. Edge computing, which enables low-latency, real-time decision-making in remote or underserved locations, is increasingly included in developing architectures to provide successful surveillance [6]. When paired with AI, these technologies can provide early warnings and dynamically adjust to physiological changes, improving outcomes for both the mother and the fetus. Furthermore, by helping physicians comprehend algorithm decisions, explainable AI frameworks included in Materiovigilance processes might boost clinician confidence in these systems in critical care scenarios [25].
The recent advances in the sphere of wearable maternal-foetal monitoring have made it possible to create a multi-modal sensor design that includes chest, limb, and abdomen wearables to allow simultaneous tracking of the key maternal and foetal physiological signals. According to the evidence provided by recent systematic reviews, such integrated systems can record maternal ECG, fECG, oxygen saturation (SpO2), body movement, and uterine activity (Liu et al. [35], 2024). This yields multi-parameter data that is more clinically informative and richer than the single-modality devices. At the same time, the first guidelines to acquire data have been developed and compared to well-known methods of research, which proved that it is possible to monitor the heart rate and physical activity of a mother with the help of wearable accelerators and ECG sensors full-time during her pregnancy and during the different activities she does in everyday life [82]. The fact that it is possible to monitor the heart rate of a mother and her physical activity during her pregnancy and during the different activities she performs in her daily life is also justified by survey data provided by more than 500 pregnant volunteers who state that they are willing to use a wearable. Almost 12,000 pregnant users were exposed to large field validation studies conducted in diverse geographic settings and socioeconomic settings [83]. Recent advances in deep learning-based signal processing have also markedly improved the reliability of wearable sensor measurements, with new foetal signal separation algorithms demonstrating substantial error rates compared to old ones, even in low sampling and motion artefact conditions. Collectively, these findings provide strong evidence of the feasibility, accuracy, and scalability of the integrated wearable systems to continue to monitor maternal and foetal health [84].
The proposed wearable maternal foetal monitoring system would be instigated in a systematic manner based on a step-by-step, practically feasible roadmap. The initial phase focuses on optimization of hardware by selecting biocompatible and flexible sensor materials to enhance user comfort and minimize skin irritation due to prolonged use and miniaturization of multi-sensor modules with good wireless connectivity and long battery life, so that it can be used continuously even in non-clinical environments [82]. To counteract the motion, respiration, and environmental noise, the second phase dwells on signal processing and AI incorporation. It also uses real-time artefact reduction and signal segmentation algorithms, which are assisted with ML-based feature extraction and classification algorithms, which proved to be superior compared to the traditional threshold-based strategies of abnormal physiological signal detection. To make early intervention possible, predictive AI models are also added to the set of algorithms. Measures obtained by wearables are compared to clinical reference instruments such as Doppler ultrasound and hospital-grade ECG systems in the third phase, with pilot studies on diverse cohorts at different stages of gestation [49]. Algorithms and hardware are continuously enhanced to ensure long-term safety and reproducibility and user experience, and to ensure health system integration and compliance with regulations. The final phase uses AI-based clinical decision support tools in daily care procedures and in line with Materiovigilance and regulation, such as international medical device standards and national programs, such as the MvPI [85]. Having measurable performance criteria within the framework of the latest research in the wearable maternal-foetal monitoring and AI-based healthcare analytics, this stepwise framework demonstrates that the proposed plan is feasible in real-life implementation and makes sense conceptually.
The use of AI and IoT technology in the precise prediction of stillbirth is still understudied, despite the growing awareness of unfavorable pregnancy outcomes worldwide. There is a dearth of research specifically focused on stillbirth prediction, despite the fact that several AI-driven models have been created for related obstetric outcomes such preterm delivery, foetal growth restriction, and preeclampsia [86]. The majority of current research either uses small, retrospective datasets with low generalizability or treats stillbirth as a secondary goal. A significant gap in the translational pipeline is that there is currently no extensively verified or clinically used AI model created specifically for stillbirth risk prediction. Experts have a scarce understanding of how AI with IoT systems can precisely predict stillbirth, even though awareness of adverse pregnancy outcomes continues to increase globally [87, 88]. The medical field lacks proper research on stillbirth prediction, even though several AI-based models predict related obstetric outcomes, including preterm delivery, foetal growth restriction, and preeclampsia [89]. Most existing studies base their research on limited retrospective datasets that offer restricted possibilities for generalization, and stillbirth remains their secondary focus. No broadly accepted or clinically deployed model exists for stillbirth risk prediction because the translational pipeline lacks an extensively verified solution designed specifically for this purpose. This shortage of AI applications targeting specific risk assessment delays the progress toward essential predictive models that serve the precision medicine framework. The combination of AI methods renders the possibility to unite data types, including electronic medical records, together with continuous monitoring data and molecular biomarkers, including sFlt-1 and PlGF, for detecting concealed patterns leading to stillbirth occurrences [90]. Organizations should initiate AI-based prospective research on stillbirth, which focuses on risk profiling, biomarker development, and longitudinal statistical analysis. A successful reduction of research gaps in neonatal care requires joint work between data scientists, physicians, and biological researchers across multiple disciplines [89]. Achieving explainable AI frameworks through future research should focus on developing tools that physicians can rely upon, together with multi-cultural verification that enables simple digital health platform integration.
Furthermore, this study opens the door for the development of new research agendas centered on stillbirth by using knowledge from related domains, like telemedicine, preeclampsia prediction, and foetal monitoring. It promotes the use of wearable IoT technology to provide continuous monitoring outside of clinical settings and multi-modal AI systems that can combine clinical, physiological, and molecular data for RT risk assessment. The ultimate goals of this program are to promote clinical acceptance of predictive technology, stimulate multidisciplinary innovation, and significantly lessen the worldwide incidence of stillbirth by means of early intervention and individualized treatment.
The review offers valuable information on the essential medical issue of maternal-foetal concern, which is the use of AI and IoT technology to predict and prevent stillbirth. AI has worked well in enhancing the forecasting abilities of preeclampsia, preterm labor, and foetal distress, but very little is done regarding stillbirth as concerns clinical and computer prediction algorithms. The research gaps in high-risk pregnancies remain a serious issue since it hinders the establishment of new preventive care methods in situations with weak fetuses. The question is among the first attempts to explore the potential of AI and IoT in the detection of the threat of stillbirth, as it argues both the methods and the challenges of implementation. This study discusses biosensors and remote monitoring devices in conjunction with ML algorithms to attain such a system of quick, targeted risk assessments in pregnant women. The fact that AI models are using placental or inflammatory marker data will enhance prediction accuracy in applying precision medicine in obstetrics, since it encourages scientists to build AI systems with tested models using repository-based stillbirth data. The paper reiterates the importance of conducting future prospective cohort studies in concurrently harnessing effective data presented by IoT devices when addressing the ethical issues of the implementation of the digital health systems. The findings of the research provided a significant background to the technological development that may come after, which will decrease the incidence of stillbirths as it will contribute to providing timely medical assistance, tailored medical care, and more evidence-based clinical decisions.
The data provided in Table 1 demonstrates that AI and ML models are highly predictive when it comes to identifying and preventing maternal issues. The models that are based on AI prove to be highly correct, sensitive (recall), and specific in many trials, proving their powerful ability to detect high-risk pregnancies and minimize false alarms. High accuracy (between 85.0% and 99.72% has been achieved with the help of the advanced ensemble and tree-based models such as XGBoost, GB, RF, and stacking ensemble (SE) models. It is important to note that the results of Innab et al. [21] (2024) were the best ones, with a combined RF, logistic regression, GB, and XGBoost model demonstrating an impressive reliability rate of 99.72%, a sensitivity of 96.67%, and a specificity of 99.6%. Similarly, Gunenc et al. [20] (2025) have shown that AI is effective in the early detection of risks with 96.8% accuracy, 96.3% sensitivity, and 97.2% specificities. The GB classifier and SE model also performed well and provided over 90% sensitivity. High sensitivity ensures that most of the potential cases are well identified, and this is important in minimizing maternal issues.
RF models showed high sensitivity of more than 92%, which validated their usefulness in clinical decision support, but specific models, such as the Blender model, had high recall (91.6%) and high accuracy (95.9%). XGBoost is highly specific (90%) and therefore capable of reliably identifying low-risk cases, although the sensitivity is reduced in one study (25%). This underscores the importance of model selection and the features of the data set. In a nutshell, the findings of Table 1 indicate that compared to traditional risk assessment methods, AI-driven predictive models can enhance the process of detecting maternal complications at the earliest possible stage by at least 10–30%. Based on such findings, AI has enormous prospects in enhancing maternal healthcare prevention, enabling timely clinical interventions, and ultimately reducing maternal morbidity and mortality.
The AI and IoT technology for stillbirth prediction face serious practical challenges despite the ongoing expansion of the sensor and maternal-foetal monitoring industries. A significant weakness in the literature is the lack of pertinent data sets that could play a significant role and could be well annotated to give the essential criteria of still births, and this is the greatest barrier that hinders the development of reliable prediction systems. The research has significant issues of model repeatability and generalization because of data heterogeneity due to sensor differences, maternal conditions, and gestational stages. The use of physiological, clinical, and biochemical data relates to complex technical solutions and necessitates complex network designs and interpretation tactics when using wearable technology in constrained resource environments or serving underprivileged clientele. Patient consent, along with algorithm prejudice and privacy of information, emerge as priorities when wearable technology is applied in restricted resource environments or when attending to poorer groups. The discipline needs to promote multidisciplinary collaboration between obstetricians, biomedical engineers, and data scientists, as well as specialists in the field of public health, to provide comprehensive and fair responses.
The research of the future must focus on creating explicable and clear AI frameworks, which are trustworthy in giving verdicts in RT and which doctors can easily understand. Furthermore, to test AI algorithms and guarantee scalability, longitudinal, prospective cohort studies involving a variety of populations are crucial. Improved IoT integration will lessen the need for in-hospital monitoring by enabling continuous, RT maternal and foetal health surveillance, especially with cloud and edge computing platforms. Combining AI prediction models with biomarker-based diagnostics has the potential to develop individualized treatment pathways and further the precision obstetrics’ aim of preventing stillbirths.
AI: artificial intelligence
AUC: area under the curve
CTG: cardiotocography
ECG: electrocardiography
fECG: fetal ecocardiogram
FHR: fetal heart rate
FM: fetal movement
fPCG: fetal phonocardiography
GB: gradient boosting
ICD: International Classification of Diseases
IoT: Internet of Things
LBW: low birth weight
MDGs: Millennium Development Goals
ML: machine learning
MvPI: Materiovigilance Programme of India
PlGF: placental growth factor
RF: models random forest
SDGs: sustainable development goals
SE: stacking ensemble
WHO: World Health Organization
XGBoost: extreme gradient boosting
The supplementary tables for this article are available at: https://www.explorationpub.com/uploads/Article/file/1001399_sup_1.xlsx.
Authors (Atreyi Pramanik and Pardeep Yadav) are thankful to Ms. Shivani Singh, Ph.D. Scholar of the Uttaranchal University C/o Dr. Pardeep Yadav, for her assistance during the extraction of the gene’s information, presented in the supplementary files. During the preparation of this work, the author(s) used ChatGPT in order to improve the grammar and syntax. After using the tool/service, author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
AP: Writing—original draft, Formal analysis, Data curation. PY: Conceptualization, Methodology, Writing—review & editing, Supervision. SKJ: Investigation, Visualization, Validation. SPP: Resources, Software, Data curation. PG: Conceptualization, Supervision, Project administration, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The supplementary datasets and figures can be found in the supplementary files.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 480
Download: 18
Times Cited: 0
