Affiliation:
1Retired Professor of Department of Internal Medicine, Federico II University, 80131 Naples, Italy
Email: fazio0502@gmail.com
ORCID: https://orcid.org/0000-0002-2743-9836
Affiliation:
2Scientific Committee of Foundation “Allineare Sanità e Salute”, 20131 Milan, Italy
ORCID: https://orcid.org/0000-0002-4365-6814
Affiliation:
3Independent Researcher, 37100 Verona, Italy
ORCID: https://orcid.org/0000-0002-5086-3504
Explor Med. 2026;7:1001392 DOI: https://doi.org/10.37349/emed.2026.1001392
Received: December 05, 2025 Accepted: March 05, 2026 Published: March 24, 2026
Academic Editor: Anna Maria Spagnolo, University of Genova, Italy
At the beginning of COVID-19 pandemic, due to the lack of guidance from evidence-based medicine on how to treat the infected patients, the medical class faced significant difficulties, not only with the unknown infection but also with the great number of cases. This led to a great number of hospitalizations and deaths. It would have been necessary to try drugs already available on the market, which might be effective against SARS-CoV-2. This is a short review of studies in the scientific literature dealing with the use of indomethacin as an antiviral drug against SARS-CoVs. We revised studies taken from the scientific literature in PubMed, Science Direct, Scopus, ResearchGate, Google Scholar etc., describing the effects of indomethacin as an antiviral drug against SARS-CoVs. To search for studies, we used the keywords: SARS-CoV, off-label drug, pandemic emergence, repurposed drug, Absence of EBM, Antiviral, Indomethacin, and Mechanisms. Among the studies reviewed, there is an interesting experimental study, published in 2006 by an Italian group, which considered the problem at the time of the previous SARS-CoV epidemic, that clearly demonstrated an antiviral effect of indomethacin, both in vitro and in vivo, against SARS-CoV. Two other studies, both clinical, one retrospective observational, and the other a prospective randomized trial comparing indomethacin with paracetamol, also showed good effects of indomethacin in the treatment of patients with COVID-19. On the basis of these notices, we wonder why, in such an emergency situation, indomethacin was not taken into consideration and was not tried in the treatment of COVID-19.
As everyone knows, since 1992, medical management has shifted from a medicine that relied primarily on the physician’s individual clinical experience and sources deemed reliable to an evidence-based medicine (EBM), that is, a clinical approach that integrates the best available scientific evidence with the patient’s clinical experience and values to make therapeutic decisions [1]. This shift has clearly made medicine safer and more effective because EBM enables informed, more effective, and safer decisions, encourages physicians to actively seek the latest and most relevant information, and ensures that treatment is tailored to the patient’s specific needs [2–4].
However, a conventional and rigid EBM approach, based only on large randomized clinical trials (RCTs), is not always applicable to every disease and to every patient. The question is: What happens if we’re faced with a new disease that lacks the available scientific evidence? Some doctors run into difficulties because they have grown accustomed to relying on RCTs for many years. A clear example of this was the therapeutic management of the SARS-CoV-2 pandemic, COVID-19. Indeed, it is now clear to everyone that, particularly during the first year of the pandemic, physicians ran into significant difficulties and there were numerous hospitalizations and deaths [5]. Some literature suggests that in the absence of clear indications from EBM, for a condition as serious as in a pandemic from a little-known virus, and while waiting for clear evidence from RCTs, we could try to use off-label drugs, among those already in use for other indications and that have a rationale for the use in the treatment of current pandemic [6, 7]. Unfortunately, in a pandemic state, there were specific regulatory, scientific, and clinical decision-making hurdles that hindered this initiative, and only later did some studies, using specific methods (for example, in silico and computational modeling), attempt to identify potential drugs to be repurposed against SARS-CoV-2 [4, 8–10]. In this article, we analyze the chance of using this method to identify a drug that could have been rapidly studied using the EBM methods, to confirm the possibility of its use as an early home treatment of COVID-19. Among the different well-known drugs, we chose indomethacin, an anti-inflammatory drug that has been used for many years and has also shown antiviral activity against various viruses.
On this basis and for the in-depth knowledge of indomethacin acquired over the years through its frequent use to treat serious diseases such as myocarditis and pericarditis, we have revised the scientific literature which supports the off-label use of this well-known drug, used since 1960 as an anti-inflammatory drug, but that also has showed in vitro and in vivo (in dogs) antiviral activity against SARS-CoVs [11]. To write this review article, we also relied on our direct experience gained during the COVID-19 pandemic as members of the COVID-19 home therapy Facebook group (https://terapiadomiciliarecovid19.it/, now Foundation TDC19 ETS), directly treating hundreds of patients at home via telemedicine. This experience has also influenced, to some extent, the selection and interpretation of the reviewed studies. Indeed, at the time, that home therapy group also believed that anti-inflammatory drugs could play a role even in the early treatment of SARS-CoV-2 infection. We also reviewed the literature on the topic, available in some of the most popular search engines, such as PubMed, Science Direct, Scopus, Google Scholar, ResearchGate, etc., using the keywords: SARS-CoV, off-label drug, pandemic emergence, repurposed drug, absence of EBM, antiviral, indomethacin, and mechanisms. There is not much literature on this topic, given the particularity of treating COVID-19 with indomethacin, so in addition to reviewing mainly studies published in English in impacted, peer-reviewed journals, some impressions published online, not subjected to peer review, were also reported. The three authors examined the articles separately, and then they discussed each article while writing the manuscript.
Following the previous SARS-CoV epidemic, a group of Italian virologists published a study clearly demonstrating indomethacin’s antiviral efficacy against SARS-CoV-1, both in vitro and in vivo in animals (dogs) [11]. In this study, SARS-CoV-1 was cultured in monkey Vero cells and A459 lung epithelial cells, while canine coronavirus (CCoV) was cultured in dog A72 cells. Antiviral activity was analyzed by determining the median infectious dose of the cell culture (50% tissue culture infectious dose-TICD50), viral RNA synthesis by Northern blot analysis and real-time polymerase chain reaction (RT-PCR), and analysis of viral protein synthesis by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDSPAGE) after 35S-methionine labeling. In vivo antiviral efficacy was determined by evaluating virus titers in dogs infected with CCoV treated orally with a dose of indomethacin of 1 mg/kg body weight. Indomethacin does not affect coronavirus binding or entry into host cells, but it acts by blocking viral RNA synthesis at cytoprotective doses. This effect is independent of cyclooxygenase (COX) inhibition. The antiviral activity of indomethacin was very potent (> 1,000-fold reduction in virus yield) and confirmed in vivo in CCoV-infected dogs [11]. The study design was very scientifically sound; however, these experimental results should be confirmed as clinical efficacy in large clinical RCTs in humans. The genomic sequence of SARS-CoV-1 is highly homologous to that of SARS-CoV-2, and both viruses use the same type of ACE2 receptor to enter the cells of the infected organism. Furthermore, the immune system responds to these viruses in very similar ways [12]. Furthermore, in a 2020 preprint, Xu et al. [13] showed that indomethacin also possesses direct and potent antiviral activity against SARS-CoV-2, both in vitro and in vivo.
Some doctors around the world, based on scientific literature reporting antiviral action of indomethacin against SARS-CoVs, began treating COVID-19 with this drug. The first report of clinical use of indomethacin for the treatment of COVID-19 was published online in May 2020 by Clark C. Unfortunately, it has many limitations, because it was not a peer-reviewed publication, but only the report of the experience of three doctors who treated about 60 COVID-19 patients, apparently with good results (Table 1). Dr. Jonathan Leibowitz, a primary care physician in Brooklyn, New York, came up with the idea of using indomethacin after his colleague, Dr. Robert Rothstein, pointed him to a 2006 study [11] demonstrating the drug’s effectiveness against human SARS-CoVs and CCoV. So, when Dr. Aline Benjamin, also a primary care physician in Brooklyn, was suffering from COVID-19, he suggested her to take indomethacin. She recalls, “I had muscle aches, a terrible cough, I couldn’t say three words without coughing”. After taking the first doses of indomethacin, and by the next morning, her cough was “a million times better”. Based on her positive experience, she began prescribing indomethacin to patients with COVID-19 symptoms [14].
List of clinical studies using indomethacin for COVID-19.
| Studies | Design | Sample size | Comparator | Outcomes | Limitations |
|---|---|---|---|---|---|
| Clark C.2020 [14] | Open | 60 | No one | Cough | Not peer review |
| Rajah R.2020 [15] | Prospective | 17 | No one | Number of recovered patients | Not randomized |
| Fazio S.2021 [28] | Observational retrospective | 85 | No one | Symptom duration | Not randomized |
| Krymchantowski A.V.2022 [16] | Observational retrospective | 37 | No one | Headache | Not randomized |
| Ravichandran R.2022 [29] | Open-label RCT | 103 vs 107 | Paracetamol | Patients requesting retrospective support | Open label |
RCT: randomized controlled trial. The study by Fazio et al. [28] used indomethacin in the context of a therapeutic regimen with other substances.
In a nephrology department in India, 17 symptomatic patients who tested positive for SARS-CoV-2 were treated with indomethacin. All patients were administered indomethacin at a dose of 25 mg twice daily. In 14 patients, fever, cough, and musculoskeletal pain resolved after the first two doses. One patient required an increase in the dose to 75 mg daily. Two patients developed hypoxemia and required intravenous methylprednisolone. All patients had significant comorbidities (Table 1) [15].
One observational, retrospective study used indomethacin in 37 patients with COVID-19 presenting with refractoriness headache to the usual symptomatic medications. All of these patients except one reported greater than 50% headache relief since the third day of treatment (Table 1) [16].
In December 2021, our group published the results of a retrospective observational study of the outcomes and hospitalization rates of Italian patients with a confirmed diagnosis of COVID-19 and treated remotely at home within 3 days or 3 days after the onset of symptoms with a combination of drugs, the main one being indomethacin, due to its known antiviral activity (Table 1). We were able to analyze complete data for 158 patients. Treatment consisted of indomethacin 75 mg daily for patients up to 70 kg, 100 mg daily for patients over 70 kg, cardioaspirin 100 mg daily, 200 mg daily of both hesperidin and quercetin, and one 20 mg tablet daily of omeprazole. This association treatment was studied to potentiate the action of indomethacin while reducing its adverse effects. In fact, hesperidin and quercetin reinforced the antiviral and anti-inflammatory actions of indomethacin, protecting the organism from the damage caused by reactive oxygen species (ROS), cardioaspirin enhanced the anti-platelet effect already present in indomethacin, reducing the possibility of thromboembolic phenomena, and omeprazole protected the gastroduodenal mucosa from the possible adverse effects of indomethacin at the gastrointestinal level [17–25]. Furthermore, it has been demonstrated in murine models that quercetin administration protects the gastrointestinal mucosa of rats treated with indomethacin, preventing the expression of adhesion molecules, the production of IL-8, and the increase of myeloperoxidase [26] and barrier dysfunction [27].
Of the 158 patients enrolled, 85 began treatment within 3 days of symptom onset (group 1), while 73 patients began treatment after the third day of symptom onset (group 2). The two groups were comparable for associated comorbidities, co-therapies and baseline risk. The distinction into 2 groups was made based on the previous impression that patients who began treatment earlier had better outcomes, and the idea that indomethacin, intended as an antiviral, should be administered as soon as possible. Treatment was initiated in group 1 on average 2.05 ± 0.68 days after the onset of symptoms, and in group 2, 5.85 ± 1.36 days after the onset of symptoms. Following treatment, symptoms resolved in a median of 6 days in group 1, while in group 2, they persisted for a median of 13 days. No patient in group 1 required hospitalization, while 14 patients in group 2 required hospitalization, two of whom died. Furthermore, a chest X-ray and blood tests were performed a median of 24 and 35 days after the first positive swab in groups 1 and 2, respectively. Only 1 patient in group 1 showed signs of interstitial lung disease, and 2 patients had an abnormally elevated D-dimer level, which returned to normal within 3 weeks. In contrast, in group 2, 42% of patients showed signs of interstitial lung disease, and over 30% of patients had elevated D-dimer levels. Early treatment with the therapeutic scheme containing indomethacin also appeared to produce a lower average mortality than that reported in Italy in the same period (2020–2021) in patients treated with the suggested standard treatments. However, to be certain of this, it would have been necessary to carry out a direct comparison study which, unfortunately, was not possible. It is possible that the results of this treatment regimen are not attributable to indomethacin alone. It is more likely that the combination of substances determined its good effects, although it is plausible that indomethacin may have played a decisive role. This observational retrospective study should have been confirmed by a prospective, randomized controlled trial, but unfortunately this was not possible in that particular period. However, with all the limitations of its design, our study showed that a therapeutic intervention with a combination of drugs, including indomethacin, resulted in a significantly better prognosis when the therapeutic intervention was performed within the first 3 days. The risk of an unfavorable outcome increased fourfold for each day of delay [28] This study has many limitations: the first is that it is only observational, the second is its small sample size, the third is the existence of comorbidities and other confounding factors. Moreover, the complex therapeutic scheme does not allow for full understanding of whether the results were due to indomethacin, the interference of the therapy they had undergone before coming to our observation, and those of natural disease variability, reassurance, and placebo effects.
Subsequently, in April 2022, an open-label, randomized trial compared indomethacin with paracetamol in 210 patients hospitalized for mild-to-moderate COVID-19 (Table 1). Of these, 103 patients were randomized to treatment with indomethacin 75 mg daily (150 if BMI > 30) in addition to standard hospital care, and the remaining 107 patients were randomized to treatment with paracetamol 650 mg four times daily. The study results clearly demonstrated that treatment with indomethacin was associated with significant symptomatic benefits and improved oxygen saturation levels. In fact, 20 of 107 patients (18.7%) in the paracetamol arm desaturated and required respiratory support, compared to no patients in the indomethacin group. Furthermore, the median number of days to become afebrile was three days in the indomethacin group, while in the paracetamol group it was seven days. The median number of days to resolution of cough and myalgia was four days and seven days in the indomethacin and paracetamol groups, respectively. The results of this study demonstrate the superiority of indomethacin over paracetamol against the worsening of COVID-19 towards desaturation due, in general, to interstitial pneumonia or pulmonary thromboembolism [29]. This open label trial of indomethacin vs paracetamol provides the strongest human evidence to date. However, we acknowledge the design limits of this RCT. The small sample size, the lack of blinding, unclear selection bias, and the choice of a poor comparator may affect the outcome interpretation.
A real-time meta-analysis of four studies, published online, reports very interesting results, though not definitive; this is probably due to an insufficient number of cases in the studies. This meta-analysis reports that two studies, although both from the same team, reported better outcomes in patients treated with indomethacin. The meta-analysis, conducted using the most serious outcomes reported, shows a 74% lower risk, although this value did not reach statistical significance, probably due to the small sample size of the studies. Currently, the data are limited, with only 605 patients in the various trials, and the studies come from only three different groups [30]. Many other experimental studies on the antiviral efficacy of indomethacin against coronaviruses have been published subsequently, confirming the potential efficacy of this drug against SARS-CoVs [31–37].
It must be acknowledged that the limitations of the early, non-peer-reviewed clinical experiences and the small, open-label randomized trial can explain why the above data failed to catalyze larger-scale research. The cited meta-analysis, mentioning a 74% risk reduction that lacked statistical significance, encapsulates the frustrating yet inconclusive state of evidence, which explains a residual clinical hesitation.
Although we do not have a large prospective randomized controlled trial that demonstrates the efficacy of indomethacin in the treatment of COVID-19, we have some evidence:
1. Indomethacin has clearly shown in experiments in vitro and in animals a strong antiviral action against different viruses, in particular against SARS-CoVs [11];
2. A retrospective, observational study shows that a therapeutic regimen containing indomethacin, initiated within the first 3 days of symptom onset in patients with confirmed COVID-19, resulted in a clear improvement in prognosis compared to the same treatment initiated later. The two groups had comparable comorbidities and therapies for concomitant diseases [28];
3. An open label, randomized controlled trial demonstrates that, in patients with COVID-19 with mild-to-moderate disease, treatment with indomethacin significantly reduced the number of patients who desaturate and required respiratory support versus a comparable group treated with paracetamol [29].
At the beginning of the COVID-19 pandemic, although being in extreme therapeutic difficulties with this new disease, unfortunately, no attempt was made to identify drugs already on the market that could be repurposed for the treatment of COVID-19, nor was a dialogue encouraged among scientists with different opinions and knowledge. The suggestions for managing COVID-19 in Italy were rather lacking and useless, if not harmful in some cases. In fact, it was recommended to wait and watch, and symptomatic treatment with paracetamol (acetaminophen) or non-steroidal anti-inflammatory drugs (NSAIDs), unless blood oxygen saturation, measured by pulse oximetry, dropped below 92% or complications requiring hospitalization arose [38] (Figure 1), discouraging other types of therapeutic initiatives.
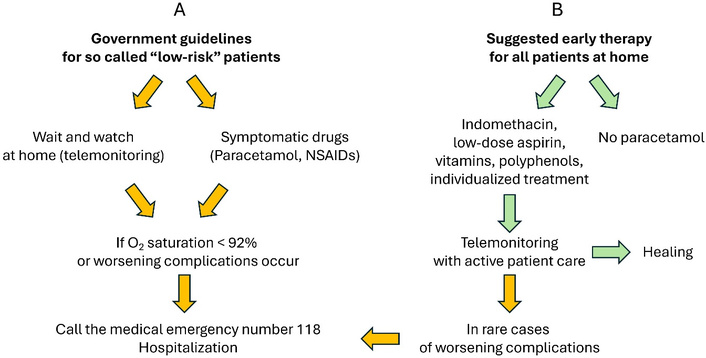
Strategies for COVID-19 treatment. (A) Government suggestions, (B) active early treatment described in the text. At the beginning of COVID-19 pandemic, the Italian Government suggestions were to consider patients with O2 saturation > 92% as they were at “low risk”, even if they had temperature > 38°C. Guidelines of “telemonitoring” were principally to wait for 72 hours, watching the patient, measuring temperature and O2 saturation by a finger pulse oximeter, and the symptomatic use of paracetamol or NSAIDs. In the case of O2 desaturation < 92% or worsening or appearance of complications, hospitalization of the patient. NSAIDs: non-steroidal anti-inflammatory drugs.
The interaction between patients and primary care physicians was rather lacking, resulting in an excessive number of hospitalizations. The Emergency Departments were overwhelmed with people in critical conditions and there were not enough resources to attend to everyone, leading to an excessive number of deaths. However, some doctors, organized in interacting groups, began to use under their own responsibility some well-known drugs, presumably useful in controlling the disease, based on the known mechanism of action of the drug on viral pathology. Among these drugs, some doctors chose indomethacin.
Indomethacin is a very inexpensive and well-known drug. It is an inhibitor of the enzymes COX-1 and 2, with a powerful anti-inflammatory effect. It has been on the market for over 60 years and is primarily used to reduce inflammation in the joints and muscles [39]. It has been used and is in the guidelines for treatment of some forms of pericarditis [40]. Like other NSAIDs, Indomethacin can cause serious side effects, particularly with prolonged use, affecting the gastrointestinal tract, kidneys, cardiovascular system, and brain. These safety concerns likely contributed to the disinclination to a wide adoption of indomethacin, especially for home use. However, these adverse effects are infrequent in the short term (treatment of COVID-19 with indomethacin lasts on average a week), gastric protection can be used during treatment, and it should be used with caution in patients with comorbidities affecting the listed organs [41]. Indomethacin, along with ibuprofen, is effective for treating patent ductus arteriosus (PDA) in infants with respiratory distress syndrome [42]. Rectal indomethacin is effective in treating endoscopic retrograde cholangiography (ERCP) induced pancreatitis [43]. Indomethacin also inhibits platelet aggregation by blocking COX-1 enzyme. By inhibiting COX-1, indomethacin reduces the production of thromboxane A2, thus decreasing platelet aggregability [44]. Finally, more interesting and important regarding the topic we are dealing with, indomethacin has been shown to have a powerful antiviral action against some viruses [45–47] also in humans [38, 48–50] and, in particular, against SARS-CoVs [7, 48, 50–55]. Its antiviral activity is independent of its COX inhibitory action. It selectively activates protein kinase R (PKR), which, in turn, induces rapid phosphorylation of eukaryotic initiation factor-2α (eIF2α), inhibiting viral replication [56, 57] (Figure 2). However, the human relevance of this mechanism remains indirect. Therefore, the link between antiviral signaling and potential clinical outcomes needs restraint.
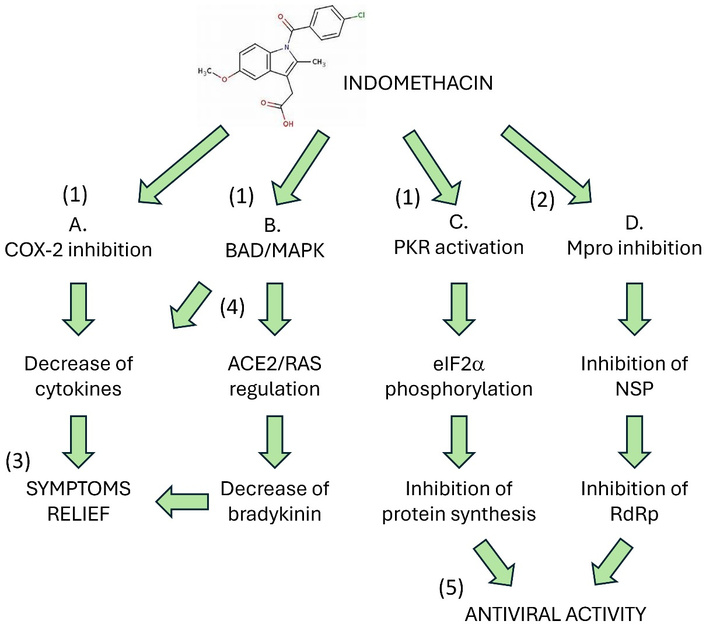
Action mechanisms of Indomethacin in COVID-19 therapy. (A) Since the predominant pharmacological activity is the inhibition of COX-2 overexpression, this drug represents a treatment option for the control of the symptoms of excess inflammation and cytokine storm [58]. (B) Indomethacin blocks the pro-apoptotic protein “BAD”, which in turn is linked to the synthesis of the cytokines IL-6, IL-8, IL-23, and to the activation of MAPK8 and MAPK10, which are key mediators of inflammation, vasoconstriction, and thrombosis [59]. Furthermore, the RAS system, deranged in viral infection due to excess angiotensin II and a defect in ACE2, leads to an increase in bradykinin [60–64], which is counteracted by indomethacin [62, 63]. (C) Indomethacin activates double-stranded RNA (dsRNA)-dependent protein kinase R [45], causing rapid phosphorylation of the alpha subunit of eukaryotic initiation factor 2 and blocking viral replication. (D) A molecular docking study suggested that indomethacin is a potential antagonist of the SARS-CoV-2 main protease [65]. eIF2α: eukaryotic initiation factor-2α; Mpro: major protease of coronavirus; NSP: non-structural proteins; PKR: protein kinase R; RAS: renin-angiotensin system; RdRp: RNA-dependent RNA polymerase. Number in parentheses: 1: consistent biochemical evidence; 2: in silico evidence (molecular docking); 3: consistent clinical evidence; 4: theoretical speculations; 5: in vitro and animal studies.
Based on the data from the literature and the spread of the disease, we believe that a large trial with a prospective, randomized, controlled design should have been done to verify whether the early administration of indomethacin had real efficacy in humans to treat COVID-19, instead of the applied “wait and watch” strategy. Unfortunately, despite our and some physicians’ requests, this was not done. Probably, due to the pandemic situation in Italy and in the whole world, it was very arduous to organize a high-quality RCT, and/or there were regulatory obstacles, perhaps even due to the need to develop effective vaccines that could be approved conditionally by the various health agencies due to the emergency. There was a competitive landscape of pandemic research, with most of the available energy and funds directed towards research into vaccines and new (expensive) antivirals. This, unfortunately, may have de facto sidelined repurposing candidates like indomethacin, lacking strong commercial advocacy. Therefore, for a rapid response, we should have rationally attempted to use off-label drugs already available that might have had some effect on this disease, without causing damage to health [66].
Anti-inflammatory drugs were not considered for this purpose at the beginning because such drugs (corticosteroids or NSAIDs) could interfere with the immune response to the virus, excessively attenuating it [67]. These types of drugs would have been considered only in the second phase of the disease, to attenuate an excessive inflammatory response. The use of anticoagulant or antiplatelet drugs was also considered too late, namely when, after the first autopsies, initially discouraged, it was discovered that the disease was complicated by numerous thromboembolic events, primarily in the pulmonary circulation [68]. This could explain the finding of particularly high D-dimer levels in patients with a more severe course of the disease.
To best address the COVID-19 pandemic in the absence of EBM guidelines, there should have been greater dialogue between Healthcare Institutions and physicians working in the field, including those working remotely, who had gained experience treating COVID patients at home. COVID-19 was treated as a disease requiring hospital-based treatment only when patients’ conditions worsened. This led to severe overload of hospital departments, resulting in serious disfunctions, enormous health care costs, and an excessive number of deaths. Based on what is reported here, we believe that it would still be useful to conduct a large RCT to verify the anti-COVID-19 efficacy of indomethacin in humans, with a non-inferiority design compared to other more expensive antivirals for COVID-19 already on the market. From what happened during the SARS-CoV-2 pandemic, we must draw a key lesson for future pandemics: the need for a pre-established, agile framework to prioritize and rapidly test repurposed drug candidates with strong mechanistic rationale, even amid the drive for novel therapies. The case of indomethacin should be used as a paradigm for improving future preparedness.
CCoV: canine coronavirus
COX: cyclooxygenase
EBM: evidence-based medicine
NSAIDs: non-steroidal anti-inflammatory drugs
RCTs: randomized clinical trials
SF: Conceptualization, Data curation, Writing—original draft, Writing—review & editing. AD: Data curation, Writing—original draft, Writing—review & editing. PB: Data curation, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
SF and AD declare no conflict of interest. PB consulted with Vanda Omeopatici s.r.l. (Roma, Frascati), a company producing food supplements, which had no role in this manuscript.
Not applicable.
Not applicable.
Not applicable.
No new data were created or analyzed in this study. Data sharing does not apply to this article.
This research received no external funding.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1311
Download: 46
Times Cited: 0
