Affiliation:
1Department of Life Sciences, Health and Health Professions, Link Campus University, 00165 Rome, Italy
†These authors share the first authorship.
ORCID: https://orcid.org/0000-0002-9423-4689
Affiliation:
2Department of Health, Life and Environmental Sciences, University of L’Aquila, 67100 L’Aquila, Italy
†These authors share the first authorship.
ORCID: https://orcid.org/0000-0001-5062-0468
Affiliation:
2Department of Health, Life and Environmental Sciences, University of L’Aquila, 67100 L’Aquila, Italy
ORCID: https://orcid.org/0009-0001-7794-9615
Affiliation:
2Department of Health, Life and Environmental Sciences, University of L’Aquila, 67100 L’Aquila, Italy
ORCID: https://orcid.org/0000-0001-9951-8595
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari “Aldo Moro”, 70124 Bari, Italy
ORCID: https://orcid.org/0000-0003-3797-5883
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari “Aldo Moro”, 70124 Bari, Italy
ORCID: https://orcid.org/0009-0007-1494-3519
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari “Aldo Moro”, 70124 Bari, Italy
ORCID: https://orcid.org/0000-0002-6366-1039
Affiliation:
4Department of Experimental Medicine, University of Salento, 73100 Lecce, Italy
ORCID: https://orcid.org/0000-0002-3288-490X
Affiliation:
5Department of General Surgery and Surgical-Medical Specialties, University of Catania, 95124 Catania, Italy
Email: gaetano.isola@unict.it
ORCID: https://orcid.org/0000-0003-4267-6992
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari “Aldo Moro”, 70124 Bari, Italy
#These authors share the last authorship.
ORCID: https://orcid.org/0000-0002-5947-8987
Affiliation:
3Department of Interdisciplinary Medicine, University of Bari “Aldo Moro”, 70124 Bari, Italy
4Department of Experimental Medicine, University of Salento, 73100 Lecce, Italy
5Department of General Surgery and Surgical-Medical Specialties, University of Catania, 95124 Catania, Italy
6Department of Biomedical, Surgical and Dental Sciences, University of Milan, 20122 Milan, Italy
#These authors share the last authorship.
ORCID: https://orcid.org/0000-0003-0104-6337
Explor Med. 2026;7:1001396 DOI: https://doi.org/10.37349/emed.2026.1001396
Received: July 23, 2025 Accepted: March 02, 2026 Published: April 01, 2026
Academic Editor: Marcos Roberto Tovani-Palone, Saveetha Institute of Medical and Technical Sciences (SIMATS), India
Background: Obstructive sleep apnea (OSA) is a syndrome characterized by episodes of complete cessation of breathing (apnea) or inadequate breathing (hypopnea) during sleep.
Methods: A systematic search of PubMed, Cochrane Library, Embase, Scopus, Web of Science, and Lilacs databases was conducted following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We identified 1,532 records; after screening, 9 randomized controlled trials (RCTs), published between 2009 and 2020, met the inclusion criteria. These studies included 698 participants aged 5–75 years.
Results: Nine randomized trials (n = 698; 2009–2020) showed that myofunctional therapy (MFT), alone or as an adjunct, for example, continuous positive airway pressure (CPAP) and nasal washing, reduced apnea-hypopnea index (AHI) versus control in adults and children. Snoring intensity improved in trials that measured it; several studies reported gains in oxygen saturation and mouth-breathing reduction. Protocols targeted the soft palate, tongue, and facial muscles with daily home exercises.
Discussion: MFT appears to be a promising non-invasive treatment for reducing AHI, especially in pediatric patients. Its benefits extend beyond AHI reduction, supporting orofacial function and nasal breathing. However, its clinical integration remains limited due to a lack of standardized protocols and inconsistent reporting of patient adherence. Most studies also have short follow-up periods, which makes it difficult to assess long-term efficacy. To advance evidence-based use of MFT, future research should adopt standardized outcomes, monitor adherence systematically, and include long-term follow-up.
The obstructive sleep apnea syndrome (OSAS) is a sleep-related disorder in which the upper airway repeatedly collapses during sleep, resulting in either incomplete or total interruption of airflow [1]. Although it affects a significant portion of the population, it often goes unrecognized because its clinical presentation is nonspecific and sometimes misleading. OSAS is classified within the spectrum of sleep-related breathing disturbances, commonly referred to as sleep-disordered breathing (SDB) [2]. Typical features include loud snoring, witnessed apneas, nocturnal gasping, and non-restorative sleep, with daytime sleepiness and cognitive complaints; hypertension and autonomic dysregulation further increase cardiovascular risk [3].
A distinctive complication of OSA in children, not observed in adults, is its effect on growth. Children with OSA often experience weight gain and a catch-up growth period following adenotonsillectomy (AT) [4].
The treatment approach for OSAS varies depending on the severity of the disorder and the patient’s age. In milder cases without complications, weight loss is recommended as it can significantly improve the apnea-hypopnea index (AHI). For more complex cases, treatment options may include continuous positive airway pressure (CPAP), mandibular advancement devices (MADs), positional therapies, and surgical interventions aimed at correcting anatomical abnormalities or obstructive defects. In children, the most common factor associated with pediatric OSA is the enlargement of tonsils and adenoids, making AT the primary treatment option. Although AT often results in the cure or improvement of OSA in most cases, residual OSA can still occur, particularly in obese children. The prevalence of postoperative residual OSA ranges from 13% to 29% in low-risk populations and can reach levels of up to 73% when obese children are considered. Furthermore, orofacial myofunctional disorders may persist even after successful surgery [5].
While PAP therapy is the mainstay treatment for OSA in adults, its role in pediatric OSA is limited to a subset of children who do not respond to AT or when upper airway obstruction is not primarily caused by adeno-tonsillar hypertrophy, such as in cases of obesity or craniofacial abnormalities [6]. CPAP treatment is well-tolerated and highly effective in infants and older children with OSA. However, practitioners may encounter challenges in finding appropriate equipment and interfaces, particularly for infants and young children [7].
According to another systematic review, rapid maxillary expansion (RME) devices have shown promise as an effective alternative treatment for pediatric OSAS [8]. These devices involve the RME and have been found to be beneficial [9, 10]. In addition to anatomical airway narrowing, neuro-muscular factors contribute to OSA, such as decreased function of the upper airway dilator muscles during sleep or poor muscle activation. Some researchers have explored the relationship between neuromuscular factors and OSA, specifically residual OSA after AT or orthodontic treatment in childhood, supporting the inclusion of orofacial myofunctional therapy (OMT) as an adjunctive approach in pediatric SDB/OSA, including residual OSA after AT [11–13]. The exercises involved in OMT target the soft palate, tongue, and facial muscles, as well as stomatognathic functions. Soft palate exercises include continuous or intermittent pronunciation of oral vocal sounds, while tongue exercises involve various movements along the palatal and buccal surfaces of the teeth. Facial exercises target the lips, buccinators, and jaw muscles. Stomatognathic functions are addressed through nasal inhalation, oral exhalation, blowing and inflating a balloon, as well as specific swallowing and chewing exercises.
The aim of this study was to evaluate the validity of myofunctional therapy (MFT) in the treatment of disordered breathing and OSAS in both children and adults. Globally, it is estimated that approximately 936 million adults aged 30–69 years suffer from mild to severe OSAS, with the majority remaining undiagnosed [14]. The socioeconomic burden of OSAS includes increased healthcare costs, reduced work productivity, and a higher incidence of workplace and traffic accidents, highlighting the need for effective, accessible treatment strategies such as MFT [15, 16].
Compared with prior reviews, this study restricts inclusion to randomized trials only, reports reproducible, exercise-level details of MFT (target muscles, frequency, session length), separates pediatric and adult outcomes, pre-specifies AHI and snoring as primary endpoints, and appraises bias with current RoB 2.0 criteria. This combination provides a tighter estimate of effectiveness and practical parameters for clinical translation.
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement was adhered to as much as possible. In the study section paragraph, there is the PRISMA flowchart.
This review was designed following PICO guidelines to determine the:
population: children and adults diagnosed with OSAS;
intervention: MFT;
comparison: MFT versus another therapy;
outcomes: The primary outcome was to evaluate AHI and snoring.
The secondary outcome was to investigate the efficacy of MFT in the patients with OSAS children and adults.
We registered the protocol on international prospective register of systematic reviews (PROSPERO) (CRD42021237554) and followed PRISMA. Searches were run in PubMed, Scopus, Embase, Web of Science, Cochrane Library, and Lilacs from 1 Jan 2009 to 31 Dec 2020; English language; humans. The core string (adapted per database) was: (myofunctional therapy OR orofacial myotherapy OR oropharyngeal exercise OR tongue exercise OR swallowing exercise OR breathing exercise) AND (apnea OR OSA OR OSAS OR snoring OR sleep-disordered breathing). Two reviewers (SS and CV), independently, deduplicated (EndNote X9), screened titles/abstracts (Rayyan), and assessed full texts; disagreements were resolved by a third reviewer (MP). Pre-specified outcomes: primary AHI and snoring; secondary oxygen saturation, daytime symptoms/sleep quality, mouth breathing, and adherence. Risk of bias was assessed with Cochrane RoB 2.0. Owing to heterogeneity of protocols and populations, we performed a structured narrative synthesis and reported effect directions; no meta-analysis was attempted. Searches were conducted up to 31 Dec 2020 according to the original protocol; therefore, studies published after this date were not included.
We included randomized controlled trials (RCTs) that focused on the efficacy of respiratory muscle therapy compared to control therapy or no treatment. Studies involving participants of any age group, ranging from 5 to 75 years old, were considered.
We excluded articles published prior to 2009, abstracts, letters to the editor, pilot studies, commentaries, literature reviews, systematic reviews, case reports, case series, animal studies, and clinical guidelines. However, literature reviews, systematic reviews, and clinical guidelines were screened to identify relevant RCTs.
The selection process for this study was made in two phases. In the first stage, studies were considered according to the following inclusion (A): MFT and SDB.
Language restrictions: English.
Studies after 2009.
The initial electronic search strategy yielded a total of 1,532 articles. After eliminating 1,305 duplicate records, 227 studies proceeded to title and abstract screening. At this stage, 203 papers were excluded (157 reviews and 46 unrelated to the topic). Consequently, 24 articles were retrieved for full-text assessment, of which 15 were further excluded for being off-topic. In the end, 9 studies fulfilled all the inclusion criteria and were incorporated into the systematic review. The selection process is visually presented in a PRISMA flowchart (Figure 1).
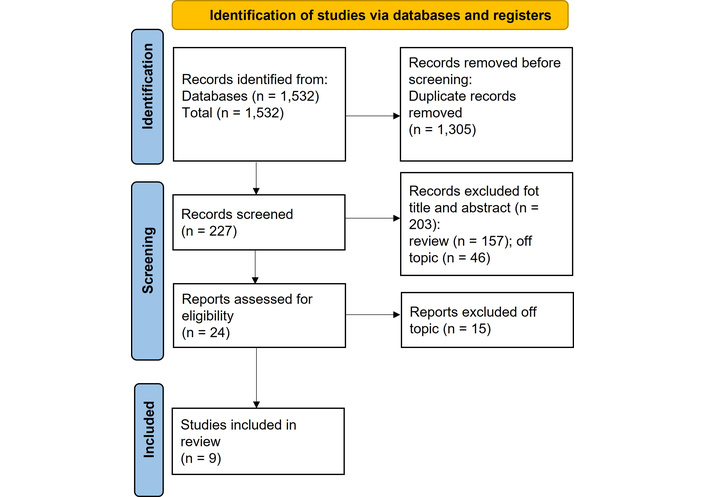
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart. Adapted from [17]. Accessed Mar 1, 2026. © 2024–2026 the PRISMA Executive. Distributed under a Creative Commons CC BY 4.0 license.
Duplicate records were removed using EndNote X9 software. Two independent reviewers (SS and CV) then performed title and abstract screening in a blinded fashion using Rayyan QCRI. Discrepancies were resolved by consensus or by consulting a third reviewer (MP). Studies were selected based on methodological rigor, including clarity of outcome definitions, completeness of follow-up, and use of intention-to-treat analysis. All the authors separately did initial screenings. At this stage, the title and abstract, if available, were analyzed by all the authors. The articles were selected if considered interesting by all the reviewers. A detailed full-text analysis was carried out, and three reviewers (SS, CV, and MP) extracted data from each study. There was no dissent related to data screening and extraction.
This systematic review was carried out by using original studies, comparative studies, research journal articles and, once analyzed, we collected data regarding six areas (country- design- sample size, intervention, inclusion criteria, sex and age, time, outcomes) as summary in Table 1 and other five areas (intervention therapy, details, duration, frequency, control therapy) as summary in Table 2. We also extracted protocol fidelity/adherence (sessions/day, minutes/session, completion rate) when available, to contextualize AHI changes.
Main characteristics of randomized controlled trials (RCTs) included in the systematic review, reporting study design, sample size, intervention type, inclusion criteria, demographics, follow-up time, and evaluated outcomes.
| Study | Country/Disctrict, design, sample size | Intervention | Inclusion criteria | Sex and age | Time | Outcomes |
|---|---|---|---|---|---|---|
| Villa et al. (2015) [11] | Italy, single-blinded RCT, n: 27 | OE + nasal washing, n: 14Controls (nasal washing), n: 13 | Children with AHI > 5 events/hChildren with residual OSAS AHI > 1 event/h after AT persistence of respiratory symptoms | 4.82 ± 1.36 years24 males3 females | 2 months of treatment | AHI, oxygen saturation, SCR, IOPI measurements |
| Diaferia et al. (2013) [18] | Brazil, RCT, n.d. | MFT + CPAP vs. sham | OSAS patients (criteria not detailed) | n.d. | 3 months | AHI, snoring, oxygen saturation |
| Diaféria et al. (2017) [19] | Brazil, RCT, n.d. | MFT + CPAP vs. sham | OSAS patients (criteria not detailed) | n.d. | 3 months | AHI, PSG, functional outcomes |
| Erturk et al. (2020) [20] | Turkey, non-blinded RCT, n: 41 | IMT, n:15OE, n: 14Control, n: 12 | Patients with mild, moderate, or severe OSAS, were not using CPAP | Patients aged 19 to 75 years | 3 months of treatment | AHI, PSG, MIP and MEP values, inspiratory muscle work and expiratory muscle work outcomes |
| Guimarães et al. (2009) [21] | Brazil, single-blinded RCT, n: 31 | OE, n: 16Controls (deep breathing), n: 15 | Patients between 25 and 65 years of age with a recent diagnosis of moderate OSAS | Intervention group: 51.5 ± 6.8 yearsControls: 47.7 ± 9.8 years21 males10 females | 3 months of treatment | AHI, PSG, snoring outcomes Epworth daytime sleepiness scale, and Pittsburgh sleep quality index questionnaires |
| Villa et al. (2017) [22] | Italy, single-blinded RCT, n.d. | OE vs. nasal washing | Children with OSAS | n.d. | 2 months | AHI, sleep quality |
| Ieto et al. (2015) [23] | Brazil, RCT, n.d. | OE + nasal washing vs. control | Adults with primary snoring or mild OSAS | n.d. | 3 months | Snoring intensity, AHI |
| Kim et al. (2020) [24] | Korea, RCT, n.d. | MFT vs. no treatment | Adults with OSAS | n.d. | 12 weeks | AHI, sleep parameters |
| Lin et al. (2020) [25] | Taiwan, China, RCT, n.d. | Nasal washing + MFT + endurance training vs. control | Adults with OSAS | n.d. | 12 weeks | AHI, oxygen saturation, functional outcomes |
n.d.: no data. AHI: apnea-hypopnea index; AT: adenotonsillectomy; CPAP: continuous positive airway pressure; IMT: inspiratory muscle training; IOPI: Iowa Oral Performance Instrument; MEP: maximal expiratory pressure; MFT: myofunctional therapy; MIP: maximal inspiratory pressure; OE: oropharyngeal exercises; OSAS: obstructive sleep apnea syndrome; PSG: polysomnography; SCR: sleep clinical record.
Summary of intervention protocols across the included studies, detailing the type of therapy, specific exercises, duration, frequency, and control conditions.
| Study | Intervention therapy | Details | Duration | Frequency | Control therapy |
|---|---|---|---|---|---|
| Villa et al. (2015) [11] | OE | Isometric and isotonic exercises, nasal breathing rehabilitation, labial seal and lip tone exercises, and tongue posture exercises | 2 months | Three times/day, 10–20 repetitions each time | Nasal washing |
| Diaferia et al. (2013) [18] | Myofunctional therapy + CPAP therapy | Soft palate, pharyngeal constrictor muscles, suprahyoid muscles, tip and root of the tongue, cheeks, and lips | 3 months | 20 min, 3 times/day | Head movements without any therapeutic function |
| Diaféria et al. (2017) [19] | Myofunctional therapy + CPAP therapy | 1) Soft palate exercises2) Tongue brushing3) Tongue pushing and sucking4) Tongue rotation5) Facial exercises | 3 months | 1) 3 min, 3 times/day2) 5 min, 3 times/day3) 20 min, 3 times/day4) 10 min on each side, 3 times/day | Head movements without any therapeutic function |
| Erturk et al. (2020) [20] | 2 groups:1) Passive myofunctional therapy with oral device2) Active myofunctional therapy | 1) IMT with a threshold IMT device2) OE included soft palate, tongue, and facial muscle exercises | 12 weeks | 7 days/week5 days/week | Control group not specified |
| Guimarães et al. (2009) [21] | OE | Exercises for the soft palate, tongue, and facial muscles | 3 months | Repeated exercises for a total of about 20 min per day | Deep breathing and nasal washing |
| Villa et al. (2017) [22] | OE | 1) Nasal breathing rehabilitation2) Labial seal and lip tone exercises3) Tongue posture exercises | 2 months | Three times a day, 10–20 repetitions each time | Nasal washing |
| Ieto et al. (2015) [23] | Nasal washing and OE | Exercises for the soft palate, tongue, and stomatognathic functions | 3 months | 8 min set of exercises (20 times), 3 times/day | Deep breathing and nasal washing |
| Kim et al. (2020) [24] | Myofunctional therapy | Tongue and lips exercises | 12 weeks | Perform 10 exercises with 10 repetitions each, and perform at least 10 sets every day | Control group that did not receive any treatment |
| Lin et al. (2020) [25] | Daily nasal washing + myofunctional therapy and general endurance training | Retropalatal, retroglossal, hypopharyngeal, facial, and TMJ exercises | 12 weeks | 24 sessions, daily nasal washing + upper airway muscle strengthening for 20 min, respiratory muscle strengthening for 15 min, and general endurance training for 45 min | Control group that did not receive any treatment |
CPAP: continuous positive airway pressure; IMT: inspiratory muscle training; OE: oropharyngeal exercises; TMJ: temporomandibular joint.
We considered studies evaluating MFT delivered as a stand-alone program or in combination with other therapies (e.g., CPAP, adjunctive breathing/aerobic training, nasal washing), as well as trials comparing active vs. passive MFT approaches or sham protocols [11, 19–21, 24]. For each study, we extracted the MFT protocol (exercise set, frequency, duration), the comparator(s) (e.g., usual care, CPAP, sham), and follow-up. This information was prespecified in the extraction form and did not contribute to effect estimation beyond descriptive synthesis.
Three reviewers (SS, CV, and MP) independently evaluated the risk of bias. This evaluation was carried out following the Cochrane-recommended approach for assessing the risk of bias in randomized controlled clinical studies, including four quality parameters: sequence generation, consideration of incomplete outcome data, freedom of selective outcome reporting, and other sources of bias.
The primary outcome was to evaluate AHI and snoring.
The secondary outcome was to investigate the efficacy of MFT in patients with OSAS children and adults.
Levels of evidence and grade of recommendation followed the grading recommendations of the GRADE Working Group [26].
Our 9 final studies, published between 2009 and 2020, in Table 3, were all level 2B since we excluded reviews and meta-analyses, which would have been level 1A.
Summary of the risk of bias for each article included in the systematic review.
| Study | Incomplete outcome data | Selective reporting | Other bias |
|---|---|---|---|
| Villa et al. (2015) [11] |  |  |  |
| Diaferia et al. (2013) [18] |  |  |  |
| Diaféria et al. (2017) [19] |  |  |  |
| Erturk et al. (2020) [20] |  |  |  |
| Guimarães et al. (2009) [21] |  |  |  |
| Villa et al. (2017) [22] |  |  |  |
| Ieto et al. (2015) [23] |  |  |  |
| Kim et al. (2020) [24] |  |  |  |
| Lin et al. (2020) [25] |  |  |  |
+: low risk of bias; –: high risk of bias; ?: unclear risk of bias.
The studies were evaluated as having high, unclear, or low risk, based on selection bias, as can be observed in Table 3. The evaluation of the methodological quality of the included studies revealed variable levels of bias across different domains. Regarding incomplete outcome data, three studies [18, 19, 22] were judged at low risk, while the majority of the others presented a high or unclear risk. For selective reporting, consistent low risk was observed in the earlier trials by the other two studies [18, 22], whereas other studies showed either high or unclear risk, suggesting that outcome reporting was not always sufficiently transparent. With respect to other potential sources of bias, most trials were rated as unclear, reflecting insufficient information to permit a firm judgment, while a few were assessed as high risk. Overall, older and more methodologically rigorous studies tended to report lower risks of bias, while more recent investigations often suffered from limitations such as incomplete reporting, small sample sizes, or insufficient methodological details. This heterogeneity in study quality highlights the need for standardized protocols and more robust trial designs in future research on MFT for OSAS.
Across the included trials, MFT was delivered in several configurations:
stand-alone exercise programs targeting tongue, soft palate, and orofacial muscles;
randomized comparisons allocating patients to MFT, CPAP, combined MFT + CPAP, or sham/speech therapy;
head-to-head comparisons of active vs. passive MFT approaches;
adjunctive combinations, such as MFT with nasal washing or with aerobic training sessions.
This heterogeneity in delivery and co-interventions is summarized in Table 1 (study characteristics) and is considered in the interpretation of effects below.
The primary outcome was to evaluate AHI and snoring. The results of this systematic review indicate that with MFT, there is a reduction in the index, but the reduction in snoring is not evaluated in all articles, but Ieto et al. [23] demonstrated that oropharyngeal exercises are effective in reducing objectively measured snoring and are possible treatment of population suffering from snoring [27].
The secondary outcome was to investigate the efficacy of MFT in the patients with OSAS children and adults and the results demonstrated that use both alone and in combination with other therapies is useful in both adult and children’s patients. While AHI reduction was consistently reported and remains the main outcome, other clinically relevant endpoints such as snoring intensity, sleep quality, daytime sleepiness, and patient-reported quality of life were not systematically assessed.
Notably, several studies reported a more pronounced reduction in AHI among pediatric patients compared to adults, possibly due to greater tissue plasticity and neuromuscular responsiveness in younger subjects. Additionally, improvements in peripheral oxygen saturation and reductions in mouth breathing were consistently reported, particularly in interventions combining MFT with nasal breathing exercises.
Across 9 RCTs, MFT reduced AHI versus control or usual care; snoring reduction was demonstrated in two trials that measured it; oxygen saturation and mouth breathing improved in several pediatric cohorts. These outcomes are highly consistent with emerging evidence that early functional rehabilitation can enhance upper airway tone, reduce obstruction, and improve overall sleep quality.
While the Results section objectively summarizes study outcomes, here we discuss mechanisms, patient adherence, and the clinical significance of these findings. OSAS is defined as a pathology that recognizes multiple risk factors for its development, often combined with each other [20].
The mechanism of action underlying this correlation could be based on structural or functional alterations. The therapeutic approach must also be aimed at acting on various factors. Even some craniofacial factors, such as a reduced size of the skull and jaw or the reduced width of the upper respiratory tract, or a short lingual frenulum, are currently considered risk factors in the development of OSAS, together with particular malocclusions [11, 18, 23]. In the diagnosis and therapeutic choice, we must pay attention to the age of the patient, particularly in adolescents and children; therefore, the treatment of malocclusions through orthodontic or functional therapy could represent a fundamentally important aid in the treatment of OSAS in pediatric individuals [28–30]. CPAP remains the standard treatment for the most severe forms of OSAS. MADs are also highly effective in patients with moderate or severe diseases who are unable to tolerate CPAP. However, their use can be more challenging in partially or completely edentulous patients, although in such cases, integrated osteo-implants may represent a possible solution [21].
The MFT approach: a set of exercises that involve the soft palate, tongue, and facial muscles closely involved in the apnea characteristic of the syndrome; this can, in fact, determine a notable improvement in the parameters commonly used to monitor obstructive apnea syndromes, up to reducing the AHI by 50%. The physiological rationale behind these improvements lies in the enhancement of upper airway muscle tone and coordination. By strengthening the tongue, soft palate, and oropharyngeal muscles, MFT reduces airway collapsibility during sleep and promotes more stable breathing patterns. Improved nasal breathing and swallowing function further contribute to maintaining airway patency and reducing mouth breathing, mechanisms that explain the observed clinical benefits [31].
However, regarding many of these cofactors and the effect of these therapies, considerable confusion remains in the scientific world, and it is of fundamental importance to clarify their action in order to intercept and promptly treat this pathology. The study characteristics of the selected articles demonstrate the rigorous approach taken to assess the effectiveness of MFT in the treatment of OSAS [20]. Among the 9 chosen studies, considered the gold standard in clinical research for evaluating treatment efficacy. The inclusion of a minimum sample size of 15 patients in all studies ensures that the results are based on a sufficiently representative group, enhancing the reliability and generalizability of the findings. Furthermore, the minimum follow-up period of 2 months in each study allows for the evaluation of the sustained impact of MFT on OSAS over a reasonable timeframe [25].
Regarding qualitative characteristics, the consistent reporting of primary outcome measurements across all studies, such as the AHI, Apnea Index, oxygen saturation levels, snoring intensity, and neck circumference, highlights the standardized and comprehensive approach taken in assessing the effectiveness of MFT. These outcome measures are crucial in capturing different aspects of OSAS severity and the treatment response, ensuring a comprehensive evaluation of the therapy’s impact [22]. A major factor influencing the efficacy of MFT is patient compliance. Unlike CPAP, which offers a passive form of treatment, MFT requires active daily participation. Studies have shown that higher adherence to exercise frequency is directly associated with greater reductions in AHI and symptom severity. However, adherence metrics were rarely quantified in the included studies, making it difficult to assess dropout reasons or long-term feasibility. Strategies such as structured follow-up, caregiver involvement for pediatric patients, and the integration of digital tools (e.g., mobile applications, telemonitoring) could improve compliance and should be considered in future trials. However, variability in exercise protocols across studies poses a challenge to reproducibility [19]. There is currently no universally accepted standardized regimen, and this heterogeneity limits the ability to draw definitive conclusions on the most effective therapeutic approach [32, 33].
Adherence to MFT represents a critical factor for its effectiveness. Unlike CPAP, which provides a passive treatment modality, MFT requires active, daily patient engagement. Reported adherence rates varied across studies and were not always explicitly quantified, but several authors noted difficulties related to motivation, time commitment, and integration of exercises into daily routines. Potential barriers included a lack of supervision, limited awareness of the long-term benefits, and the repetitive nature of the exercises. Dropouts were often associated with poor motivation or perceived limited benefits. These findings underscore the importance of structured follow-up, patient education, and possibly the use of digital tools (e.g., mobile applications or telemonitoring) to enhance compliance. Future trials should systematically measure and report adherence data, since feasibility is essential for translating MFT into routine clinical practice.
In addition to behavioral interventions such as MFT, recent advances in neurotechnology have opened novel therapeutic perspectives. Temporal interference (TI) stimulation, a non-invasive technique using intersecting high-frequency electrical fields to selectively activate deep neural targets, has shown preliminary promise in modulating hypoglossal nerve activity and oropharyngeal muscles involved in OSA pathophysiology. Early studies suggest that TI stimulation may achieve functional benefits similar to MFT while overcoming the challenge of patient adherence, since it does not require daily active participation. Although still experimental, these innovations highlight the potential future integration of neuromodulation strategies with conventional therapies to broaden the management options for OSA.
Although a formal meta-analysis could not be conducted because of the high variability in study designs, populations, and intervention protocols, the findings highlight the urgent need for standardization. A preliminary framework for future MFT protocols could include:
clearly defined sets of oropharyngeal and tongue exercises;
standardized daily frequency and session duration;
minimum treatment length of 8–12 weeks;
objective outcome measures such as AHI reduction and polysomnography;
systematic assessment of secondary outcomes, including snoring, oxygen saturation, and quality of life.
Such a framework may guide future clinical trials and support the development of reproducible, evidence-based guidelines. To enhance clinical applicability, we also provide in Table 4 a preliminary summary of key elements that could form the basis for future standardized MFT protocols.
Proposed framework for future MFT protocols.
| Component | Recommendation |
|---|---|
| Exercise types | Oropharyngeal and tongue exercises |
| Frequency | 2–3 sessions/day |
| Duration per session | 15–20 min |
| Total treatment length | 8–12 weeks minimum |
| Primary outcomes | AHI reduction, PSG |
| Secondary outcomes | Snoring, oxygen saturation, QOL |
AHI: apnea-hypopnea index; MFT: myofunctional therapy; PSG: polysomnography, QOL: quality of life.
The inclusion of a total population of 698 patients with OSAS, derived from the nine included studies, strengthens the reliability and relevance of the findings [13]. The large number of participants allows for a more comprehensive assessment of the effectiveness of various MFT interventions in managing OSAS. The confirmation of the OSAS diagnosis through clinical examinations, particularly polysomnography, ensures the accuracy and validity of the patient selection, enhancing the overall robustness of the review [23]. As far as MFT is concerned, it still has many limitations; there are not many publications that can validate protocols that make MFT fundamental for the treatment of OSA [11].
There is a clear lack of cooperation among the various medical professionals involved in the treatment of OSAS.
This systematic review has several limitations that should be acknowledged. First, the number of RCTs available on MFT in OSAS is still limited, and the sample sizes of most included studies were relatively small, which may reduce the generalizability of the findings. Second, the included trials were highly heterogeneous in terms of study design, patient populations (adults versus children, varying severity of OSAS), and especially in the type, frequency, and duration of MFT exercises. The absence of a standardized protocol makes it difficult to establish optimal treatment parameters and reduces comparability across studies. Third, although AHI reduction was consistently reported and remains the main outcome, other clinically relevant endpoints such as snoring intensity, sleep quality, daytime sleepiness, and patient-reported quality of life were inconsistently measured across the trials. These outcomes were therefore not omitted from our analysis but were often unavailable due to study design limitations. Moreover, long-term follow-up data are largely lacking, since most included trials assessed outcomes only over short periods (2–6 months). As a result, the durability of MFT effects beyond the intervention phase remains uncertain. Finally, some studies presented methodological weaknesses, including unclear allocation procedures, incomplete reporting of outcomes, or potential sources of bias, as highlighted in our risk of bias assessment. Publication bias cannot be ruled out, since only English-language articles were included. Taken together, these limitations suggest that while current evidence supports a potential role for MFT in OSAS management, further large-scale, well-designed, and standardized trials with longer follow-up are required before firm conclusions can be drawn.
In conclusion, this systematic review indicates that MFT can reduce the AHI and may improve snoring, oxygen saturation, and mouth breathing, with particularly encouraging signals in pediatric populations. To establish its role in routine care, future multicenter trials should adopt standardized exercise protocols (clearly defined sets, frequency, and duration), monitor adherence systematically, include core outcome sets encompassing both objective sleep metrics and patient-relevant measures, and ensure longer follow-up to assess durability. Comparative designs against, or in combination with, CPAP and MADs will help position MFT within integrated OSA pathways.
AHI: apnea-hypopnea index
AT: adenotonsillectomy
CPAP: continuous positive airway pressure
IMT: inspiratory muscle training
IOPI: Iowa Oral Performance Instrument
MADs: mandibular advancement devices
MEP: maximal expiratory pressure
MFT: myofunctional therapy
MIP: maximal inspiratory pressure
OE: oropharyngeal exercises
OMT: orofacial myofunctional therapy
OSA: obstructive sleep apnea
OSAS: obstructive sleep apnea syndrome
PICO: population, intervention, comparison, outcome
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
PSG: polysomnography
QOL: quality of life
RCTs: randomized controlled trials
RME: rapid maxillary expansion
SCR: sleep clinical record
SDB: sleep-disordered breathing
TI: temporal interference
TMJ: temporomandibular joint
SS: Resources, Data curation, Formal analysis, Software. CV: Resources, Data curation, Formal analysis, Software. MP: Conceptualization, Formal analysis, Data curation. SES: Investigation, Visualization. FI: Formal analysis, Supervision. LF: Validation, Formal analysis, Writing—review & editing. ADI: Methodology, Investigation. AP: Software, Formal analysis. GI: Methodology, Software. GD: Conceptualization, Formal analysis. AMI: Validation, Investigation. All authors read and approved the submitted version.
Gaetano Isola, who is the Associate Editor of Exploration of Medicine, had no involvement in the decision-making or the review process of this manuscript. The other authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
This study does not involve original data; all data analyzed are publicly available and have been appropriately cited.
This research received no external funding.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 4193
Download: 31
Times Cited: 0
