Affiliation:
1Department of Health and Human Performance, University of Houston, Houston, TX 77204, United States
Affiliation:
2Section of GI Oncology, Houston Methodist Neal Cancer Center, Houston Methodist Hospital, Houston, TX 77030, United States
Affiliation:
2Section of GI Oncology, Houston Methodist Neal Cancer Center, Houston Methodist Hospital, Houston, TX 77030, United States
Email: AEsmail@houstonmethodist.org
Explor Med. 2026;7:1001401 DOI: https://doi.org/10.37349/emed.2026.1001401
Received: November 06, 2025 Accepted: April 20, 2026 Published: May 09, 2026
Academic Editor: Lindsay A. Farrer, Boston University School of Medicine, USA
Hepatocellular carcinoma (HCC) is an aggressive primary liver malignancy with a high propensity for extrahepatic spread, most commonly to the lungs, lymph nodes, and bones. Metastases to atypical sites like the orbital and oral cavity are uncommon and often underrecognized. We report the case of a 74-year-old male with hepatitis C-related cirrhosis who was diagnosed with HCC and initially treated with locoregional therapies. Disease progression was marked by the development of biopsy-confirmed pulmonary metastases, followed by treatment with durvalumab plus tremelimumab immunotherapy and external beam radiation. Despite therapy, further progression occurred with mediastinal lymphadenopathy. The patient subsequently developed visual disturbances, and ophthalmologic evaluation identified a solitary choroidal mass consistent with metastatic HCC. Around the same time, he presented with recurrent oral bleeding, and a biopsy of a maxillary gingival lesion confirmed metastatic HCC. This case highlights the aggressive nature of advanced HCC and its potential for unusual metastatic spread. To our knowledge, this is among the few reported cases of HCC with orbital and oral metastases. Awareness of atypical metastatic presentations and a multidisciplinary approach are essential for timely diagnosis and optimal management.
Liver cancer, predominantly hepatocellular carcinoma (HCC), represents a major global health concern. According to the most global estimates from GLOBOCAN 2022, liver cancer accounted for approximately 866,000 new cases and 758,000 deaths worldwide, ranking among the leading causes of cancer-related mortality globally [1, 2]. In the United States, liver cancer is associated with approximately 43,429 new cases and 30,931 deaths annually, underscoring the aggressive nature of the disease and its high mortality burden [3].
HCC is a highly aggressive cancer, with metastases occurring in approximately 50% to 75% of patients over the course of the disease [4].
Common risk factors for liver cancer include chronic infections with hepatitis B virus (HBV) or hepatitis C virus (HCV), excessive alcohol consumption, and metabolic dysfunction-associated steatotic liver disease, often related to obesity and metabolic syndrome, as well as environmental exposure to aflatoxins. Liver cancer is frequently asymptomatic in its early stages, but as the disease progresses, patients may experience symptoms such as abdominal pain, unintended weight loss, and general malaise [5].
The diagnosis of HCC is typically confirmed by identifying its characteristic imaging features, heterogeneous arterial phase enhancement followed by washout in the portal venous phase on multiphase magnetic resonance imaging (MRI) or computed tomography (CT) scans, particularly in the setting of underlying chronic liver disease [6].
Surgery, orthotopic liver transplant (OLT) or ablation is the standard curative approach to early-stage HCC, depending on liver function and tumor burden [7]. For advanced HCC, systemic therapies are applied, with first-line options including atezolizumab plus bevacizumab (Atezo/Bev), durvalumab plus tremelimumab (Durva/Treme), lenvatinib, and sorafenib. Second-line treatments encompass regorafenib, cabozantinib, and ramucirumab, particularly for patients with elevated alpha-fetoprotein (AFP) levels. Palliative care is essential for patients with end-stage liver disease or poor performance status [8–10].
The incidence of extrahepatic metastases in HCC is estimated to range from 15% to 50%, with the lungs (47–55%). Lymph nodes (45–53%), bones (28–37%), and adrenal glands (11–12%) are the most commonly affected sites [11]. Less frequently, HCC may metastasize to the peripheral peritoneal cavity, brain, spleen, pancreas, gallbladder, diaphragm, skin, and gastrointestinal tract [11]. The presence of extrahepatic spread typically advances the disease of Barcelona Clinic Liver Cancer (BCLC) stage C or D and is associated with reduced survival [11]. Nevertheless, prognosis is more strongly influenced by intrahepatic tumor burden and patient performance status than by the presence of extrahepatic metastases [11].
In this case, we present a rare case of HCC with an unusual pattern of metastatic spread, including sequential involvement of the choroid and oral cavity. To our knowledge, this is one of the few reported cases of HCC with choroidal and oral metastases, highlighting the importance of recognizing atypical metastatic presentations and integrating multidisciplinary evaluation into ongoing management.
A detailed timeline summarizing the patient’s clinical progression, treatment interventions, and metastatic evolution is presented in Figure 1.
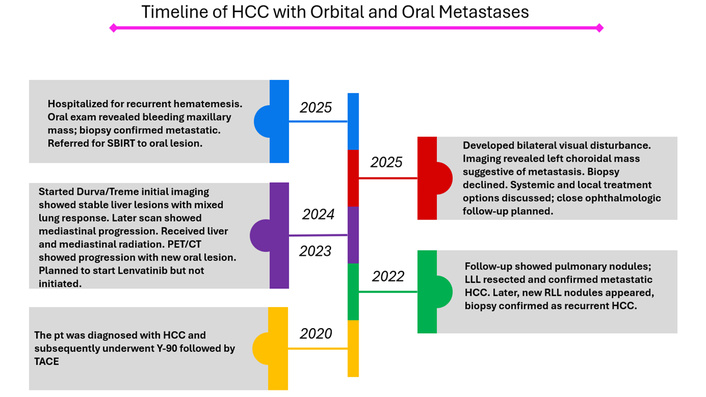
Chronological time of disease progression and management. The diagram illustrates the sequence of key clinical events, including initial diagnosis of hepatocellular carcinoma, locoregional therapies with Y-90 radioembolization and TACE, and development of pulmonary metastases, initiation of systemic immunotherapy with Durva/Treme, radiotherapy, and subsequent disease progression with orbital and oral metastases. Planned systemic therapy with lenvatinib is also indicated. Durva/Treme: durvalumab plus tremelimumab; HCC: hepatocellular carcinoma; LLL: left lower lobe; PET/CT: positron emission tomography/computed tomography; pt: patient; RLL: right lower lobe; SBIRT: stereotactic body image-guided radiotherapy; TACE: transarterial chemoembolization; Y-90: yttrium-90.
A 74-year-old Caucasian male with a history of HCV-related cirrhosis achieved sustained virologic response (SVR) after antiviral therapy and was first diagnosed with HCC in May 2020. At the time of diagnosis, he had preserved liver function with Child-Pugh class A, an Eastern Cooperative Oncology Group (ECOG) performance status of 0–1, and BCLC stage B disease, with no previous episodes of hepatic decompensation such as ascites, encephalopathy, jaundice, or variceal bleeding. He had a prior history of moderate alcohol use that he had since discontinued. The initial liver lesion was discovered incidentally during routine surveillance imaging, as the patient was asymptomatic. He underwent transarterial radioembolization with yttrium-90 (Y-90) in October 2020 and achieved a partial response. Follow-up imaging in June 2021 revealed a residual viable tumor measuring 2.4 cm, prompting transarterial chemoembolization (TACE) in August 2021. Surveillance chest imaging in December 2021 identified two small pulmonary nodules, and interval growth of a left lower lobe nodule was noted by March 2022. The patient then underwent surgical wedge resection of the left lower lobe lesion in April 2022, with pathology confirming metastatic moderately differentiated HCC, which upstaged the disease to stage IV and led to his removal from the liver transplant waitlist. Combination immune checkpoint inhibitor therapy with Durva/Treme was initiated in April 2023, and he completed a total of 25 treatment cycles. Restaging with CT and MRI in September 2023 during immunotherapy demonstrated stable treated hepatic lesions with possible pulmonary progression. Subsequent imaging in December 2023 showed a mixed response pattern in the lungs, with partial response in some nodules and stability in others. Serial imaging through mid-2024 continued to show a decrease in the size of treated liver lesions, although progressive mediastinal lymph node involvement eventually developed. A positron emission tomography (PET)/CT scan performed in December 2024 confirmed disease progression with newly fluorodeoxyglucose (FDG)-avid mediastinal and hilar lymphadenopathy, while a bone scan showed no evidence of osseous metastases. He subsequently received external beam radiation therapy to the liver and mediastinum in February 2025. Follow-up PET/CT demonstrated further progression, including persistent hypermetabolic mediastinal lymphadenopathy and a new FDG-avid oral cavity lesion. Given the evidence of systemic and symptomatic progression, the patient was transitioned to lenvatinib therapy in May 2025, although treatment had not yet been initiated at the time of this report. Throughout this period, he remained under close multidisciplinary oncologic care, with consideration given to localized treatments such as stereotactic body radiotherapy (SBRT) to the oral lesion.
Initial MRI of the liver in May 2020 revealed a 4.8 cm arterially enhancing lesion with washout in the liver dome involving segments 4A and 8, consistent with HCC by Liver Imaging Reporting and Data System (LI-RADS) criteria. Surveillance chest imaging beginning in December 2021 identified two small pulmonary nodules, with subsequent follow-up in March 2022 demonstrating interval growth of one nodule in the left lower lobe, which was resected and pathologically confirmed as metastatic moderately differentiated HCC. Multiple CT and MRI scans performed during immunotherapy from 2023 to 2024 showed stable or mixed responses in the treated hepatic lesions and variable responses among the pulmonary nodules, eventually giving way to progressive mediastinal lymphadenopathy. PET/CT in December 2024 confirmed FDG-avid mediastinal and hilar lymphadenopathy with no osseous involvement on bone scan. Later follow-up PET/CT revealed ongoing progression with hypermetabolic mediastinal lymphadenopathy and a new FDG-avid oral cavity lesion (Figure 2). Ophthalmologic evaluation disclosed bilateral posterior vitreous detachment (PVD) on dilated funduscopic examination. Multimodal imaging of the left eye identified a solitary choroidal mass in the superotemporal macula, with spectral-domain optical coherence tomography showing dome-shaped choroidal elevation, overlying retinal distortion, and associated subretinal fluid. Fundus autofluorescence demonstrated hyperautofluorescence with central heterogeneity, and indocyanine green (ICG) angiography revealed no additional choroidal lesions in either eye. These ocular findings were highly suggestive of choroidal metastasis (Figure 3). MRI of the face noted a small elliptical nodule in the left posterolateral lobe along the retina, potentially representing a retinal metastatic lesion, along with asymmetric enhancement in the right maxilla centered around the first molar and extending into the oral cavity, although visualization of the oral cavity was limited by metal artifacts from dental hardware. Recurrent oral bleeding prompted esophagogastroduodenoscopy (EGD), which initially identified a Mallory-Weiss tear, although a repeat examination during the same admission showed no active bleeding or stigmata of recent hemorrhage. Physical examination revealed a polypoid mass on the maxillary gingiva, and biopsy of this lesion confirmed metastatic HCC, with microscopic findings of ulcerated squamous mucosa, florid inflamed granulation tissue, and infiltrating nests, cords, and trabeculae of epithelial cells exhibiting abundant eosinophilic granular cytoplasm, round nuclei with prominent nucleoli, and increased mitotic activity. Immunohistochemistry demonstrated positivity for pancytokeratin, HepPar-1, and glypican-3 (focally), while staining was negative for CK7 (Figure 4).
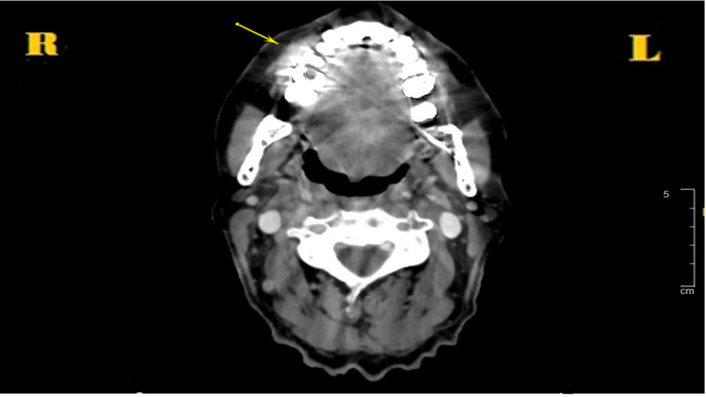
Axial contrast-enhanced computed tomography (CT) image of the neck (soft-tissue window). It shows a hyperdense lesion involving the right maxillary alveolar ridge, consistent with metastatic involvement of the oral cavity.
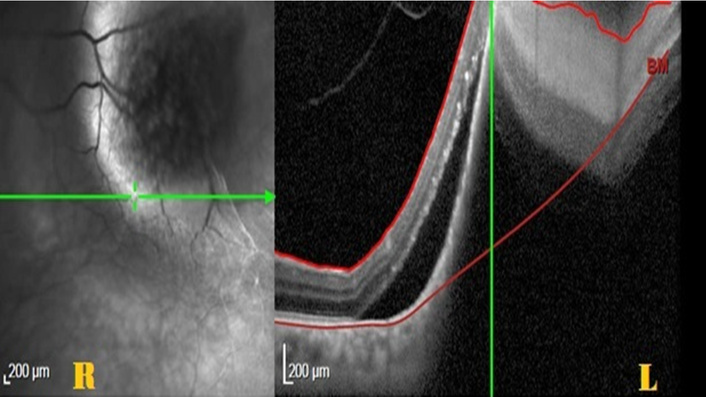
Multimodal imaging of the left eye demonstrating choroidal metastasis. (L) Fundus near-infrared reflectance imaging shows a well-defined, dome-shaped lesion involving the macula; (R) spectral-domain optical coherence tomography (SD-OCT) reveals a dome-shaped choroidal mass with overlying retinal elevation and associated subretinal fluid, consistent with a solitary choroidal metastasis.
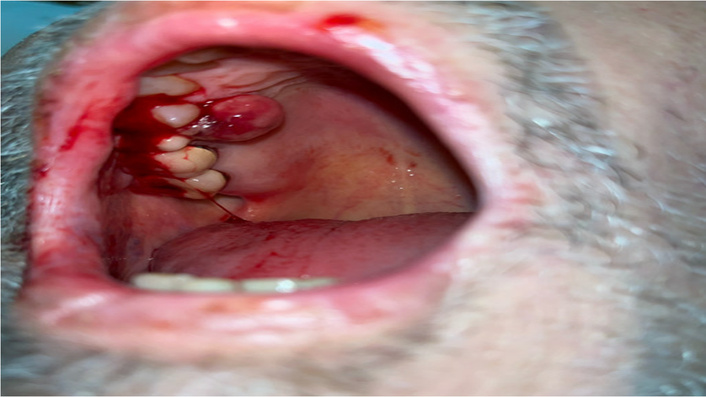
Intraoral clinical photograph. It shows a polypoid mass involving the right maxillary gingiva, identified as the source of recurrent oral bleeding. Histopathologic examination of the biopsy specimen confirmed metastatic hepatocellular carcinoma (HCC).
The patient was initially asymptomatic at the time of HCC diagnosis, with the liver lesion detected only through routine surveillance imaging. He tolerated locoregional therapies including Y-90 radioembolization and subsequent TACE, without major reported difficulties and completed 25 cycles of Durva/Treme combination immunotherapy with no prominent complaints documented during that phase of treatment. As the disease progressed to involve extrahepatic sites, new symptoms emerged. He developed gradually worsening visual disturbances over several weeks, describing small translucent mobile floaters in both eyes, wispy cloud-like opacities in the right eye, multiple round floaters of varying size in the left eye that remained stable, and blurred vision bilaterally. He specifically denied photopsia, flashes of light, eye trauma, or any history of diabetes. He also reported a persistent left-sided headache. Around March 2025, he began experiencing recurrent oral bleeding, which had been ongoing for approximately one month by the time of ophthalmologic evaluation. This bleeding included episodes of hematemesis that led to hospitalization. The patient noted that the polypoid mass on the maxillary gingiva, later confirmed as metastatic HCC and believed to be the source of the bleeding, had been present for about two months according to his recollection. When a biopsy of the choroidal lesion was offered to confirm the suspected ocular metastasis, the patient declined the procedure. Despite the advancing disease and the appearance of symptomatic involvement at both ocular and oral sites, he continues to be followed closely with multidisciplinary oncologic care and is planned to begin systemic lenvatinib therapy along with potential localized radiation to the oral lesion.
In HCC, early detection and awareness of common metastatic sites are essential for optimizing treatment strategies and improving prognosis. Accurate risk stratification plays a key role in minimizing unnecessary expenses, treatment delays, and disease-related complications [12]. A national study conducted in 2014 analyzed data from over 50,000 HCC patients using the University Health Consortium (UHC) and Nationwide Inpatient Sample database [13]. It found that extrahepatic metastases occur in approximately 13–36% of cases, the most frequent sites being the lungs (6%), bones (3%), peritoneum (3%), regional lymph nodes (2%), spleen (1%), and adrenal glands (1%) [12, 13]. Similarly, a 2011 study assessing outcomes in HCC patients with distant metastases reported that the lungs (39.5%), lymph nodes (34.2%), bones (25.4%), and adrenal glands (8.8%) were the most commonly affected sites, with less frequent involvement of the brain (1.2%), spleen (0.6%), and breast (0.3%), totaling 376 metastatic events in 342 patients [13]. In contrast, metastases to the orbital and oral cavity remain exceedingly rare and often underrecognized in clinical practice.
Orbital metastases account for only 3–7% of all orbital masses, usually presenting unilaterally 92%, while only 8% of patients have bilateral involvement, while oral cavity involvement is estimated in just 1–3% of cases [14]. These metastases typically occur in advanced stages of cancer and may, in rare cases, precede the diagnosis of the primary tumor [15]. Among primary tumors, lung, prostate, and breast cancer are the most frequently associated with orbital metastasis [15]. Oral cavity metastases are more commonly linked to cancer of the thyroid, kidney, and lung. Within the orofacial area, the mandible is the most frequently involved site (81%), and similar to primary HCC, metastatic disease shows a predominance in males [16].
In this report, we present a unique case of HCC with metastases to both the oral cavity and the orbit. HCC-related metastases to either site are extraordinarily uncommon, and simultaneous involvement of both anatomical regions has not been previously described. This case highlights the rare and aggressive nature of disease dissemination and represents an exceptional presentation in the literature.
Metastatic spread in HCC primarily occurs via the hematogenous route, most commonly to the lungs, and is often associated with pulmonary involvement. Another possible route was described by Vitale et al. [17]. They reported a case of metastatic HCC involving the parotid gland in the absence of pulmonary metastases [17]. The author explained that via Batson’s plexus (BPP), a network of valveless veins is located in the epidural space between the spinal column and dura mater. Malignant cells entering this plexus can bypass the pulmonary, caval, and portal venous systems by lodging directly in the venous and sinusoidal networks of bone connected to the BPP [16–19]. Another possible route of orbital involvement is through initial infiltration of the orbital bone, which subsequently extends into adjacent orbital soft tissue [19]. In the present case, the patient had biopsy-confirmed pulmonary metastases prior to the development of choroidal and oral lesions, supporting hematogenous dissemination as the primary route of spread. BPP may have contributed as a secondary pathway, facilitating tumor seeding to uncommon sites, such as the orbit and maxillofacial region, particularly in the setting of advanced disease and mediastinal lymphadenopathy. This combined mechanism underscores the aggressive biology and extensive dissemination potential of advanced HCC observed in our patient.
In addition to anatomical pathways such as BPP, the seed and soil hypothesis may provide a complementary explanation for the metastatic pattern observed in this case. This concept suggests that tumor cells (seeds) preferentially colonize distant sites (soil) with a microenvironment, including stromal components, cytokine signaling, and vascular characteristics, which play a critical role in supporting tumor progression and metastatic spread. Although orbital and oral metastases from HCC are rare, local microenvironmental factors may create conditions that support tumor cell implantation and proliferation at these sites. While this remains speculative, it may help explain the occurrence of metastases in these uncommon locations [20].
The proposed pathways relevant to this case are summarized schematically in Figure 5.
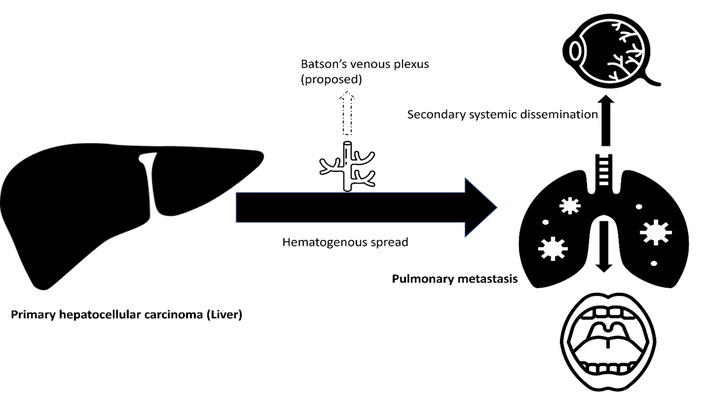
Schematic illustration of metastatic dissemination in advanced hepatocellular carcinoma (HCC). Tumor spread occurs primarily via hematogenous dissemination to the lungs, followed by secondary systemic dissemination to the choroid and oral cavity. A potential contributory role of Batson’s venous plexus is shown schematically.
Orbital metastases are typically classified into four distinct types based on their clinical presentation: infiltrative (restricted eye movement and occasionally enophthalmos), mass effect (proptosis or displacement of the globe), inflammatory (reduced vision, eye pain, redness), and functional (cranial nerve dysfunction, visual symptoms without prominent external signs) [4, 19]. Our patient’s symptoms—progressive floaters, blurred vision, and lack of pain or redness—were consistent with the functional category. Ophthalmologic imaging identified a solitary choroidal mass, and although biopsy was not performed, clinical and radiographic findings strongly supported the diagnosis of choroidal metastasis.
In clinical practice, the decision to pursue a biopsy for suspected orbital metastasis depends on the level of diagnostic certainty and its potential impact on management. Biopsy is generally recommended in cases where the primary is unknown, imaging findings are atypical, or when histopathological confirmation would influence treatment decisions. The potential benefit of biopsy lies in providing a definitive histopathological diagnosis, particularly in cases of diagnostic uncertainty or when it may alter therapeutic management. However, in patients with a known primary malignancy and characteristic imaging features, a presumptive diagnosis may be sufficient. Previous case reports have emphasized that the decision to perform orbital biopsy should be guided by a careful assessment of whether the results would alter clinical management, given the associated procedural risk, including visual loss, hemorrhage, and diplopia. In addition, other reports highlighted that the orbital metastases may be misdiagnosed due to atypical presentation, leading to delays in appropriate oncologic management, and have recommended comprehensive staging and multidisciplinary evaluation prior to biopsy [21]. In the present case, the diagnosis of choroidal metastasis was supported by clinical context and multimodal imaging, and the patient declined biopsy, making non-invasive evaluation the preferred approach.
Similarly, oral metastases can present with a variety of symptoms, including localized pain, inflammation, or paresthesia, which often complicates and delays diagnosis [14]. In this case, the patient experienced recurrent oral bleeding initially attributed to a Mallory-Weiss tear. However, subsequent examination revealed a polypoid mass on the maxillary gingiva, which was confirmed by biopsy to be metastatic HCC. MRI showed asymmetric enhancement of the right maxilla with extension into the oral cavity, consistent with local invasion.
Histologically, oral metastatic HCC lesions may exhibit strands or duct-like structures composed of hepatocyte-like cells within a richly vascular stroma. These features can mimic adenocarcinoma due to a pseudo-glandular pattern or appear poorly differentiated, making it challenging to determine the tumor’s original type. Therefore, immunohistochemical (IHC) staining is essential for diagnosis [16]. In our case, IHC demonstrated strong positivity for pancytokeratin, HepPar-1, and glypican-3, confirming the diagnosis of metastatic HCC.
For orbital metastases, therapeutic strategies are primarily palliative due to typically advanced stages at presentation and limited survival, often less than 12 months [22]. Patients with HCC that has metastasized to the orofacial region generally face a poor prognosis, with an average overall survival (OS) of just 21 weeks and only 15% surviving beyond one year after diagnosis [23]. The apparent difference in survival between oral and orbital metastases may reflect underlying differences in overall disease burden and pattern of dissemination. Oral metastases are often associated with more widespread systemic disease, which may contribute to a more advanced clinical stage at presentation. In contrast, orbital metastases may be detected earlier due to prominent visual symptoms, potentially allowing for earlier clinical evaluation. Additionally, variability in reported outcomes may reflect differences in treatment approaches, as well as limitations related to the small number of cases available in the literature. Therefore, these survival estimates should be interpreted with caution [20, 21].
This outlook remains unfavorable even with treatment directed at the primary tumor, such as surgery, radiotherapy, TACE, or systemic therapy [23]. The decision to pursue systemic versus local therapy depends on disease extent, patient performance status, and symptom burden. In cases with isolated or limited involvement, local treatments may provide temporary symptom control; however, systemic progression often limits long-term outcomes. Therefore, early multidisciplinary evaluation is crucial to tailor treatment plans and optimize both quality of life and disease management [22].
Clinically diagnosed cases of HCC metastasizing to the orbit are exceedingly rare. Over the past 39 years, only 33 cases have been reported in the literature, with the earliest reported in 1980 [15, 24].
The first documented case of HCC metastasizing to the mandible was reported by Dick et al. in 1957 [25]. Since that initial report, approximately 80 cases of oral metastasis from HCC have been documented, with the most comprehensive literature review to date summarizing 77 of them [23]. A comprehensive review by Pesis et al. [26] published in 2014 analyzed 41 cases of HCC metastasis to the jaws reported prior to 2012, highlighting the rarity of oral cavity involvement and its frequent presentation as an initial manifestation of disease. These historical cases are summarized as a single entry in Table 1, while the present review emphasizes more recently reported individual cases to provide updated clinical context.
Reported cases of HCC metastasis to the orbital and oral cavity.
| Number | Author | Year | Number of cases | Sex | Metastatic site | Age (years) | Treatment |
|---|---|---|---|---|---|---|---|
| 1 | Eldesouky et al. [27] | 2014 | 6 | M | Orbital | 47–70 | N/A |
| 2 | Geske et al. [28] | 2017 | 3 | 2 M, 1 F | Orbital | 56–63 | RT, surgery |
| 3 | Protopapa et al. [15] | 2020 | 1 | M | Orbital | 53 | RT |
| 4 | Baldovin et al. [24] | 2021 | 1 | M | Orbital | 62 | Surgery |
| 5 | Filippini et al. [21] | 2021 | 1 | M | Orbital | 71 | N/A |
| 6 | Wonglhow et al. [19] | 2024 | 1 | M | Orbital | 65 | Systemic treatment |
| 7 | Iwai et al. [29] | 2024 | 1 | M | Orbital | 65 | BSC |
| 8 | Shaikh et al. [13] | 2025 | 1 | M | Orbital | 73 | BSC |
| 9 | Lim et al. [22] | 2025 | 2 | 2 M | Orbital | 59–62 | RT, systemic treatment |
| 10 | Pesis et al. [26] | 2014 | 41 | 35 M, 6 F | Oral cavity | 15–82 | RT, surgery |
| 11 | Misra et al. [16] | 2015 | 1 | F | Oral cavity | 50 | N/A |
| 12 | Alrumaih et al. [30] | 2015 | 1 | M | Oral cavity | 73 | N/A |
| 13 | Jiang et al. [31] | 2016 | 1 | M | Oral cavity | 65 | N/A |
| 14 | Kwon et al. [32] | 2016 | 1 | M | Oral cavity | 50 | N/A |
| 15 | Xue et al. [33] | 2017 | 1 | M | Gingival | 60 | RT, systemic treatment |
| 16 | Md Radzi et al. [34] | 2018 | 1 | M | Mandible | 61 | N/A |
| 17 | Adnot et al. [35] | 2018 | 1 | M | Maxillary | 56 | RT |
| 18 | Liu et al. [36] | 2019 | 1 | M | Mandibular ramus | 73 | RT |
| 19 | Hou et al. [37] | 2019 | 1 | M | Gingival | 78 | RT |
| 20 | Nadkarni et al. [38] | 2020 | 1 | M | Buccal mucosa | 49 | N/A |
| 21 | Lyon et al. [39] | 2022 | 1 | F | Hard palate | 37 | RT |
| 22 | Huang et al. [40] | 2022 | 1 | M | Gingival | 68 | N/A |
| 23 | Fernández-Ferreira et al. [41] | 2023 | 1 | M | Mandibular | 74 | N/A |
| 24 | Reesha et al. [42] | 2023 | 2 | 2 M | Mandibular | 60–67 | N/A |
| 25 | Mašulović et al. [23] | 2023 | 1 | F | Mandibular | 65 | RT |
| 26 | Chaudhry et al. [43] | 2023 | 1 | M | Buccal mucosa | 70 | RT |
| 27 | Zhan et al. [44] | 2025 | 1 | M | Gingival | 53 | Systemic treatment |
| 28 | Present case* | 2025 | 1 | M | Orbital + oral cavity | 74 | RT, systemic treatment |
*: The final entry (“present case”) represents the patient described in this manuscript. BSC: best supportive care; F: female; M: male; N/A: not available or not reported; RT: radiotherapy.
We conducted a systematic search of PubMed for reports of orbital and oral metastases from HCC published over the past decade. This review identified a total of 76 cases, comprising 17 orbital and 59 oral cavity metastases. The clinical details of these patients are summarized in Table 1. Notably, our case is the only documented instance of HCC presenting simultaneous metastases to both the orbital and the oral cavity. Although prior reports have described the involvement of each site separately, no previous publication has reported concurrent metastases to both regions. This underscores the rarity of our case and highlights the aggressive and widespread nature of disease dissemination. To the best of our knowledge, this represents one of the very few reported cases of HCC with synchronous metastases to both the choroid and oral cavity.
In advanced HCC, rare metastatic sites such as the orbit and oral cavity can emerge, often presenting with subtle or misleading symptoms that may be easily overlooked. Because these atypical presentations, such as persistent visual disturbances or unexplained oral bleeding, can signal disease progression, they warrant prompt and targeted clinical evaluation. Managing such complex cases effectively requires a dedicated multidisciplinary approach to ensure that these unusual symptoms are addressed within a comprehensive care plan.
This case highlights the rare occurrence of choroidal and oral metastases from HCC, reflecting aggressive disease behavior. Atypical metastatic sites should be considered in patients with new symptoms and known HCC. Early recognition and multidisciplinary management are essential for timely diagnosis, symptom control, and improving the patient’s quality of life.
BCLC: Barcelona Clinic Liver Cancer
BPP: Batson’s plexus
CT: computed tomography
Durva/Treme: durvalumab plus tremelimumab
FDG: fluorodeoxyglucose
HCC: hepatocellular carcinoma
HCV: hepatitis C virus
IHC: immunohistochemical
MRI: magnetic resonance imaging
PET: positron emission tomography
TACE: transarterial chemoembolization
Y-90: yttrium-90
SA, EAN, and AE: Conceptualization, Investigation, Investigation, Writing—original draft, Writing—review & editing, Validation. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work.
The authors declare that they have no conflicts of interest.
Institutional Review Board (IRB) approval was waived for this single patient case report in accordance with institutional policy. The study was conducted in accordance with the guidelines of the Declaration of Helsinki.
Written informed consent was obtained from the patient to participate in this study.
Written informed consent was obtained from the patient for publication of this case report.
The data of this study that supports our results are available upon request from the corresponding author, Abdullah Esmail (AEsmail@houstonmethodist.org).
This study received no external funding.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 302
Download: 6
Times Cited: 0
