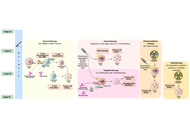

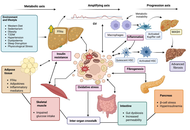


Immunotherapy is a promising treatment strategy for treating colorectal cancer (CRC). Despite significant advances in this field, resistance and low efficacy of immunotherapy remain a principal problem. One of the most important factors affecting the response to immunotherapy is the tumor microenvironment (TME). Among the components of the TME, tumor-associated macrophages (TAMs) are key immune cells involved in cancer progression by stimulating tumor cell proliferation, angiogenesis, epithelial-mesenchymal transition, metastasis, and tumor immune evasion. This review presents currently investigated combination therapy based on the immune checkpoint inhibitors and inhibitors of diverse components of the TME, including TAMs, that can potentially increase the effectiveness of CRC treatment. Therapeutic efficacy, together with the functional activity of TAMs, is estimated in multiple preclinical data obtained with diverse in vitro and in vivo models. Ongoing clinical trials demonstrated the association of treatment effectiveness with TAM phenotypes and functions.
Immunotherapy is a promising treatment strategy for treating colorectal cancer (CRC). Despite significant advances in this field, resistance and low efficacy of immunotherapy remain a principal problem. One of the most important factors affecting the response to immunotherapy is the tumor microenvironment (TME). Among the components of the TME, tumor-associated macrophages (TAMs) are key immune cells involved in cancer progression by stimulating tumor cell proliferation, angiogenesis, epithelial-mesenchymal transition, metastasis, and tumor immune evasion. This review presents currently investigated combination therapy based on the immune checkpoint inhibitors and inhibitors of diverse components of the TME, including TAMs, that can potentially increase the effectiveness of CRC treatment. Therapeutic efficacy, together with the functional activity of TAMs, is estimated in multiple preclinical data obtained with diverse in vitro and in vivo models. Ongoing clinical trials demonstrated the association of treatment effectiveness with TAM phenotypes and functions.
DOI: https://doi.org/10.37349/edd.2026.1005124
This article belongs to the special issue Immunotherapy for Cancer of Digestive System

Aim:
Recent studies suggest an association between sleep patterns and metabolic dysfunction-associated steatotic liver disease (MASLD) among American adults. Despite established sex-specific disparities in MASLD prevalence, the potential influence of sex on the sleep-MASLD relationship is not well defined. Our research aims to elucidate the sex-specific associations of sleep with MASLD by utilizing a nationally representative cohort from the United States.
Methods:
Data from United States adults aged 20 and older in the 2017–2020 NHANES were analyzed in this cross-sectional study. Sleep parameters were assessed via interviewer-administered questionnaires, while MASLD was defined using vibration-controlled transient elastography (VCTE). Sex-specific associations were investigated with sex-stratified multivariable logistic regression models, and their robustness was tested through subgroup and sensitivity analyses.
Results:
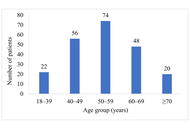
This study included 5,243 participants (51.2% female). A significant association was observed between sleep disorders and a greater likelihood of MASLD among male participants (OR = 1.50; 95% CI: 1.08–2.10). After full adjustment for covariates, the association remained significant in females (OR = 1.51; 95% CI: 1.06–2.16). Conversely, high sleep debt remained significantly associated with MASLD in both sexes (males: OR = 1.64; 95% CI: 1.14–2.37; females: OR = 1.51; 95% CI: 1.06–2.15). Subgroup analyses confirmed that sex did not modify these relationships.
Conclusions:
Sleep disorders and sleep debt were significantly associated with MASLD in both sexes. These findings suggest that sleep health may represent an important modifiable target in MASLD prevention strategies.
Aim:
Recent studies suggest an association between sleep patterns and metabolic dysfunction-associated steatotic liver disease (MASLD) among American adults. Despite established sex-specific disparities in MASLD prevalence, the potential influence of sex on the sleep-MASLD relationship is not well defined. Our research aims to elucidate the sex-specific associations of sleep with MASLD by utilizing a nationally representative cohort from the United States.
Methods:
Data from United States adults aged 20 and older in the 2017–2020 NHANES were analyzed in this cross-sectional study. Sleep parameters were assessed via interviewer-administered questionnaires, while MASLD was defined using vibration-controlled transient elastography (VCTE). Sex-specific associations were investigated with sex-stratified multivariable logistic regression models, and their robustness was tested through subgroup and sensitivity analyses.
Results:
This study included 5,243 participants (51.2% female). A significant association was observed between sleep disorders and a greater likelihood of MASLD among male participants (OR = 1.50; 95% CI: 1.08–2.10). After full adjustment for covariates, the association remained significant in females (OR = 1.51; 95% CI: 1.06–2.16). Conversely, high sleep debt remained significantly associated with MASLD in both sexes (males: OR = 1.64; 95% CI: 1.14–2.37; females: OR = 1.51; 95% CI: 1.06–2.15). Subgroup analyses confirmed that sex did not modify these relationships.
Conclusions:
Sleep disorders and sleep debt were significantly associated with MASLD in both sexes. These findings suggest that sleep health may represent an important modifiable target in MASLD prevention strategies.
DOI: https://doi.org/10.37349/edd.2026.1005123

Aim:
Chronic liver disease (CLD) is characterized by progressive impairment of hepatic function and frequent lipid metabolism abnormalities, with reductions in high-density lipoprotein cholesterol (HDL-C) and other lipoprotein fractions shown to parallel worsening liver dysfunction and predict adverse clinical outcomes such as decompensation and mortality. Established prognostic scores like Model for End-Stage Liver Disease (MELD) and Child-Turcotte-Pugh (CTP) capture aspects of disease severity, but composite lipid indices such as the non-HDL/HDL-C ratio (NHHR), which balance atherogenic and protective lipoproteins, have emerged as potentially informative biomarkers in metabolic and liver disorders. This study evaluated the association of NHHR with clinical decompensation in CLD.
Methods:
This cross-sectional study included 220 adults with CLD of mixed etiologies. Baseline demographics, liver disease severity scores, and fasting lipid profiles were obtained. NHHR was calculated, and patients were categorized into tertiles. Spearman correlation coefficients were calculated to examine relationships between NHHR and clinical severity markers. Multivariable logistic regression was used to evaluate the association between NHHR and clinical decompensation. Model performance was compared using receiver operating characteristic curves, net reclassification improvement (NRI), and integrated discrimination improvement (IDI).
Results:
Among 220 patients with CLD (mean age 54.5 ± 11.9 years, 63% male), 96 (43.6%) had decompensated disease. Higher NHHR tertiles were associated with increasing MELD-3.0 scores (P = 0.028) and lower serum albumin (P < 0.001). NHHR correlated positively with MELD-3.0, bilirubin, and international normalized ratio (INR) and inversely with albumin and platelet count. Decompensation prevalence rose across NHHR tertiles (31.1% to 53.4%, P < 0.001). NHHR was independently associated with decompensation (adjusted OR 1.55, 95% CI 1.21–1.98, P < 0.001) and improved model discrimination (AUC 0.79 vs. 0.73).
Conclusions:
NHHR is independently associated with clinical decompensation in CLD and provides incremental prognostic value beyond traditional predictors, suggesting its potential utility in clinical risk assessment and stratification.
Aim:
Chronic liver disease (CLD) is characterized by progressive impairment of hepatic function and frequent lipid metabolism abnormalities, with reductions in high-density lipoprotein cholesterol (HDL-C) and other lipoprotein fractions shown to parallel worsening liver dysfunction and predict adverse clinical outcomes such as decompensation and mortality. Established prognostic scores like Model for End-Stage Liver Disease (MELD) and Child-Turcotte-Pugh (CTP) capture aspects of disease severity, but composite lipid indices such as the non-HDL/HDL-C ratio (NHHR), which balance atherogenic and protective lipoproteins, have emerged as potentially informative biomarkers in metabolic and liver disorders. This study evaluated the association of NHHR with clinical decompensation in CLD.
Methods:
This cross-sectional study included 220 adults with CLD of mixed etiologies. Baseline demographics, liver disease severity scores, and fasting lipid profiles were obtained. NHHR was calculated, and patients were categorized into tertiles. Spearman correlation coefficients were calculated to examine relationships between NHHR and clinical severity markers. Multivariable logistic regression was used to evaluate the association between NHHR and clinical decompensation. Model performance was compared using receiver operating characteristic curves, net reclassification improvement (NRI), and integrated discrimination improvement (IDI).
Results:
Among 220 patients with CLD (mean age 54.5 ± 11.9 years, 63% male), 96 (43.6%) had decompensated disease. Higher NHHR tertiles were associated with increasing MELD-3.0 scores (P = 0.028) and lower serum albumin (P < 0.001). NHHR correlated positively with MELD-3.0, bilirubin, and international normalized ratio (INR) and inversely with albumin and platelet count. Decompensation prevalence rose across NHHR tertiles (31.1% to 53.4%, P < 0.001). NHHR was independently associated with decompensation (adjusted OR 1.55, 95% CI 1.21–1.98, P < 0.001) and improved model discrimination (AUC 0.79 vs. 0.73).
Conclusions:
NHHR is independently associated with clinical decompensation in CLD and provides incremental prognostic value beyond traditional predictors, suggesting its potential utility in clinical risk assessment and stratification.
DOI: https://doi.org/10.37349/edd.2026.1005121
This article belongs to the special issue Gastrointestinal Diseases, Cholesterol, Oxysterols, and Bile Acids

Janus kinase (JAK) inhibitors represent a major advancement in the management of immune-mediated inflammatory diseases. A balanced approach that carefully weighs therapeutic benefits against potential risks is essential. Through appropriate patient selection, close monitoring, and open physician–patient communication, the clinical potential of JAK inhibitors can be optimized while minimizing adverse outcomes. Nine JAK inhibitors have demonstrated utility in hepatogastrointestinal disorders; however, only two have FDA approval. JAK inhibitors are classified into reversible (competitive) and irreversible (covalent) inhibitors according to their chemical binding with amino acids. This review discusses the safety profile, adverse effects, and molecular selectivity of JAK inhibitors, and highlights their therapeutic roles in hepatogastrointestinal diseases, including inflammatory bowel disease, hepatic fibrosis, hepatocellular carcinoma, autoimmune diseases associated with cancer therapy in post-transplant patients, eosinophilic esophagitis, metabolic syndrome, and metabolic dysfunction-associated steatotic liver disease, and acute graft-versus-host disease following liver transplantation.
Janus kinase (JAK) inhibitors represent a major advancement in the management of immune-mediated inflammatory diseases. A balanced approach that carefully weighs therapeutic benefits against potential risks is essential. Through appropriate patient selection, close monitoring, and open physician–patient communication, the clinical potential of JAK inhibitors can be optimized while minimizing adverse outcomes. Nine JAK inhibitors have demonstrated utility in hepatogastrointestinal disorders; however, only two have FDA approval. JAK inhibitors are classified into reversible (competitive) and irreversible (covalent) inhibitors according to their chemical binding with amino acids. This review discusses the safety profile, adverse effects, and molecular selectivity of JAK inhibitors, and highlights their therapeutic roles in hepatogastrointestinal diseases, including inflammatory bowel disease, hepatic fibrosis, hepatocellular carcinoma, autoimmune diseases associated with cancer therapy in post-transplant patients, eosinophilic esophagitis, metabolic syndrome, and metabolic dysfunction-associated steatotic liver disease, and acute graft-versus-host disease following liver transplantation.
DOI: https://doi.org/10.37349/edd.2026.1005122

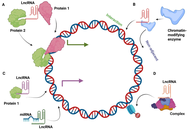
Hepatocellular carcinoma (HCC) functions as a major cancer-related death factor around the world. Research indicates that long non-coding RNAs (lncRNAs) play essential roles during HCC onset and development because they belong to the novel RNA subclass that extends beyond 200 nucleotides without protein-coding capability. LncRNAs regulate the expression of downstream target genes and cancer-related signaling pathways, thereby promoting the proliferation, migration, invasion, autophagy, and apoptosis of tumor cells. The study of lncRNA function has been substantially facilitated by the emergence of lncRNA-specific microarrays and the increased accessibility of next-generation sequencing technologies. The function of lncRNAs can be predicted using computational and molecular methodologies. LncRNAs have the potential to function as repressors, scaffolds, regulators of super-enhancers, or molecular decoys. Proliferation, invasion, survival, DNA damage response (DDR), and chromatin dynamics can all be influenced by lncRNAs. Additionally, they can affect stemness/differentiation. The recurrence of tumors may be facilitated by the aberrant expression of these transcripts, which may result in therapy resistance. LncRNAs have the potential to function as innovative prognostic or theranostic biomarkers in HCC and other malignancies. In addition, RNA-based therapeutics may be implemented to target lncRNAs as a novel treatment approach for primary or recurrent HCC. In this review, we investigate the functions of lncRNAs in the pathophysiology of HCC and suggest their potential for novel therapeutic application in the treatment of HCC.
Hepatocellular carcinoma (HCC) functions as a major cancer-related death factor around the world. Research indicates that long non-coding RNAs (lncRNAs) play essential roles during HCC onset and development because they belong to the novel RNA subclass that extends beyond 200 nucleotides without protein-coding capability. LncRNAs regulate the expression of downstream target genes and cancer-related signaling pathways, thereby promoting the proliferation, migration, invasion, autophagy, and apoptosis of tumor cells. The study of lncRNA function has been substantially facilitated by the emergence of lncRNA-specific microarrays and the increased accessibility of next-generation sequencing technologies. The function of lncRNAs can be predicted using computational and molecular methodologies. LncRNAs have the potential to function as repressors, scaffolds, regulators of super-enhancers, or molecular decoys. Proliferation, invasion, survival, DNA damage response (DDR), and chromatin dynamics can all be influenced by lncRNAs. Additionally, they can affect stemness/differentiation. The recurrence of tumors may be facilitated by the aberrant expression of these transcripts, which may result in therapy resistance. LncRNAs have the potential to function as innovative prognostic or theranostic biomarkers in HCC and other malignancies. In addition, RNA-based therapeutics may be implemented to target lncRNAs as a novel treatment approach for primary or recurrent HCC. In this review, we investigate the functions of lncRNAs in the pathophysiology of HCC and suggest their potential for novel therapeutic application in the treatment of HCC.
DOI: https://doi.org/10.37349/edd.2026.1005120
This article belongs to the special issue Prevention, Screening and Diagnosis for Primary Liver Cancer

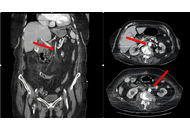
Acute mesenteric ischemia (AMI) is a rare but highly lethal vascular emergency resulting from the abrupt interruption of intestinal blood flow. In advanced stages, extensive bowel necrosis may require near total enterectomy, leading to short bowel syndrome and permanent dependence on parenteral nutrition. An 86-year-old woman with atrial fibrillation and multiple cardiometabolic comorbidities presented with acute abdominal pain, nausea, and vomiting. Initial laboratory findings revealed marked leukocytosis and severe systemic inflammation (CRP: 21.0 mg/L). Computed tomography (CT) angiography demonstrated impaired perfusion of the superior mesenteric artery. Emergency laparotomy confirmed extensive jejunoileal ischemic necrosis, necessitating near total enterectomy with stapled jejunoileal anastomosis. Second-look surgery revealed progressive ischemia of the ileocecal region and right colon, requiring extended right hemicolectomy. Postoperatively, the patient was managed with total parenteral nutrition and intensive supportive care. Despite temporary stabilization and discharge on home parenteral nutrition, she died six months later. AMI complicated by short bowel syndrome is associated with poor prognosis in elderly patients. Persistent systemic inflammation, progressive organ dysfunction, and intestinal failure remain major determinants of adverse long-term outcomes, highlighting the critical importance of early diagnosis, prompt surgical intervention, and multidisciplinary postoperative management.
Acute mesenteric ischemia (AMI) is a rare but highly lethal vascular emergency resulting from the abrupt interruption of intestinal blood flow. In advanced stages, extensive bowel necrosis may require near total enterectomy, leading to short bowel syndrome and permanent dependence on parenteral nutrition. An 86-year-old woman with atrial fibrillation and multiple cardiometabolic comorbidities presented with acute abdominal pain, nausea, and vomiting. Initial laboratory findings revealed marked leukocytosis and severe systemic inflammation (CRP: 21.0 mg/L). Computed tomography (CT) angiography demonstrated impaired perfusion of the superior mesenteric artery. Emergency laparotomy confirmed extensive jejunoileal ischemic necrosis, necessitating near total enterectomy with stapled jejunoileal anastomosis. Second-look surgery revealed progressive ischemia of the ileocecal region and right colon, requiring extended right hemicolectomy. Postoperatively, the patient was managed with total parenteral nutrition and intensive supportive care. Despite temporary stabilization and discharge on home parenteral nutrition, she died six months later. AMI complicated by short bowel syndrome is associated with poor prognosis in elderly patients. Persistent systemic inflammation, progressive organ dysfunction, and intestinal failure remain major determinants of adverse long-term outcomes, highlighting the critical importance of early diagnosis, prompt surgical intervention, and multidisciplinary postoperative management.
DOI: https://doi.org/10.37349/edd.2026.1005119

Exploring long non-coding RNAs (lncRNAs) in liver diseases, particularly liver fibrosis, presents significant opportunities for augmenting our understanding and treatment of these conditions. The rapid advancement of high-throughput sequencing technologies has revealed the complex networks of lncRNAs, highlighting their crucial functions in liver fibrosis. Identifying dysregulated lncRNAs offers promising diagnostic and prognostic biomarkers, as well as potential therapeutic targets. Extracellular vesicles contribute to the relevance of lncRNAs by protecting them from degradation and maintaining their activity in circulation, as exemplified by the role of lncRNA H19 in liver fibrosis. LncRNAs are vital in liver pathology, influencing fibrosis and cirrhosis by modulating responses to liver injury from ethanol. They affect inflammation, oxidative stress, and apoptosis through interactions with pathways like NF-κB and microRNA networks. LncRNAs also control hepatic stellate cells, the production of extracellular matrix, and the activation of stem cells, which opens up new ways to treat fibrosis. Ethanol modulates lncRNA expression, impacting liver fibrosis and cirrhosis development. LncRNAs also influence hepatocellular carcinoma progression by affecting cell proliferation, immune response, and tumor growth. Despite these insights, the regulatory networks and molecular mechanisms of lncRNAs in liver disorders are not entirely understood. In this review, we focus on unraveling these complexities and identifying effective lncRNAs that could revolutionize liver disease treatment, offer novel diagnostic and therapeutic avenues, and improve patient outcomes.
Exploring long non-coding RNAs (lncRNAs) in liver diseases, particularly liver fibrosis, presents significant opportunities for augmenting our understanding and treatment of these conditions. The rapid advancement of high-throughput sequencing technologies has revealed the complex networks of lncRNAs, highlighting their crucial functions in liver fibrosis. Identifying dysregulated lncRNAs offers promising diagnostic and prognostic biomarkers, as well as potential therapeutic targets. Extracellular vesicles contribute to the relevance of lncRNAs by protecting them from degradation and maintaining their activity in circulation, as exemplified by the role of lncRNA H19 in liver fibrosis. LncRNAs are vital in liver pathology, influencing fibrosis and cirrhosis by modulating responses to liver injury from ethanol. They affect inflammation, oxidative stress, and apoptosis through interactions with pathways like NF-κB and microRNA networks. LncRNAs also control hepatic stellate cells, the production of extracellular matrix, and the activation of stem cells, which opens up new ways to treat fibrosis. Ethanol modulates lncRNA expression, impacting liver fibrosis and cirrhosis development. LncRNAs also influence hepatocellular carcinoma progression by affecting cell proliferation, immune response, and tumor growth. Despite these insights, the regulatory networks and molecular mechanisms of lncRNAs in liver disorders are not entirely understood. In this review, we focus on unraveling these complexities and identifying effective lncRNAs that could revolutionize liver disease treatment, offer novel diagnostic and therapeutic avenues, and improve patient outcomes.
DOI: https://doi.org/10.37349/edd.2026.1005118
This article belongs to the special issue Cirrhosis and Its Complications

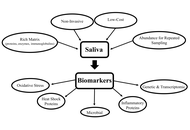
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), is a chronic immune-mediated condition typically requiring invasive endoscopy for monitoring. The bidirectional oral-gut axis suggests that saliva may serve as a non-invasive diagnostic fluid for studying gut inflammations. Studies of salivary biomarkers have shown varying results. Inflammatory biomarkers such as IL-6 are the most robust salivary biomarker, consistently correlating with endoscopic activity. In contrast, salivary calprotectin lacks the reliability of its fecal counterpart, showing diagnostic value primarily in pediatric cases with oral manifestations. Microbial analysis indicates reduced salivary diversity, specifically an enrichment of Prevotella and Veillonella alongside a depletion of core commensals like Streptococcus. While oxidative stress markers such as advanced oxidation protein products (AOPPs) can distinguish disease severity, they lack long-term prognostic utility. Conversely, recent shifts toward exosome-based transcriptomic analysis have improved the stability of salivary microRNAs, offering high precision in differentiating IBD phenotypes. Despite these advancements, clinical integration is currently hindered by small cohort sizes, the confounding effects of local oral health, and a lack of standardized collection protocols. To establish saliva as a reliable tool in the IBD clinical toolkit, future research must prioritize multi-panel biomarker approaches and longitudinal studies to validate diagnostic accuracy across diverse patient populations.
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), is a chronic immune-mediated condition typically requiring invasive endoscopy for monitoring. The bidirectional oral-gut axis suggests that saliva may serve as a non-invasive diagnostic fluid for studying gut inflammations. Studies of salivary biomarkers have shown varying results. Inflammatory biomarkers such as IL-6 are the most robust salivary biomarker, consistently correlating with endoscopic activity. In contrast, salivary calprotectin lacks the reliability of its fecal counterpart, showing diagnostic value primarily in pediatric cases with oral manifestations. Microbial analysis indicates reduced salivary diversity, specifically an enrichment of Prevotella and Veillonella alongside a depletion of core commensals like Streptococcus. While oxidative stress markers such as advanced oxidation protein products (AOPPs) can distinguish disease severity, they lack long-term prognostic utility. Conversely, recent shifts toward exosome-based transcriptomic analysis have improved the stability of salivary microRNAs, offering high precision in differentiating IBD phenotypes. Despite these advancements, clinical integration is currently hindered by small cohort sizes, the confounding effects of local oral health, and a lack of standardized collection protocols. To establish saliva as a reliable tool in the IBD clinical toolkit, future research must prioritize multi-panel biomarker approaches and longitudinal studies to validate diagnostic accuracy across diverse patient populations.
DOI: https://doi.org/10.37349/edd.2026.1005117

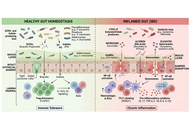
Microbial metabolites are now recognized as central mediators of host–microbe communication that shape intestinal immune homeostasis and influence the development of inflammatory gastrointestinal diseases. The objective of this review is to synthesize current mechanistic evidence on how microbiota-derived metabolites regulate epithelial and immune functions in the gut, with a focus on metabolite-driven inflammatory pathways. In the healthy intestine, short-chain fatty acids (SCFAs), indole derivatives, secondary bile acids, and polyamines support epithelial integrity, regulate mucosal immunity, and maintain metabolic balance. SCFAs, particularly butyrate, attenuate inflammation by serving as an energy source for colonocytes, inhibiting histone deacetylases, activating G protein-coupled receptors (GPCRs; GPR41, GPR43, GPR109A), and reinforcing epithelial barrier function. In parallel, microbial tryptophan metabolites such as indole-3-propionic acid and indole-3-aldehyde activate aryl hydrocarbon receptor signaling, promoting IL-22 production, antimicrobial peptide expression, and Th17–Treg balance. In inflammatory bowel disease, dysbiosis disrupts these protective pathways, leading to depletion of SCFA- and indole-producing taxa and accumulation of pro-inflammatory metabolites such as succinate. These metabolic shifts impair epithelial-immune crosstalk, amplify NF-κB-dependent inflammation, and compromise mucosal repair. Therapeutic strategies targeting microbial metabolites, including precision prebiotics, next-generation probiotics, engineered microbial consortia, and postbiotics, show translational promise. However, their clinical application remains constrained by interindividual variability, incomplete causal resolution, and challenges in targeted delivery. Integrative multi-omics approaches and mechanistically informed models are therefore essential to advance metabolite-based diagnostics and therapies for gut inflammation.
Microbial metabolites are now recognized as central mediators of host–microbe communication that shape intestinal immune homeostasis and influence the development of inflammatory gastrointestinal diseases. The objective of this review is to synthesize current mechanistic evidence on how microbiota-derived metabolites regulate epithelial and immune functions in the gut, with a focus on metabolite-driven inflammatory pathways. In the healthy intestine, short-chain fatty acids (SCFAs), indole derivatives, secondary bile acids, and polyamines support epithelial integrity, regulate mucosal immunity, and maintain metabolic balance. SCFAs, particularly butyrate, attenuate inflammation by serving as an energy source for colonocytes, inhibiting histone deacetylases, activating G protein-coupled receptors (GPCRs; GPR41, GPR43, GPR109A), and reinforcing epithelial barrier function. In parallel, microbial tryptophan metabolites such as indole-3-propionic acid and indole-3-aldehyde activate aryl hydrocarbon receptor signaling, promoting IL-22 production, antimicrobial peptide expression, and Th17–Treg balance. In inflammatory bowel disease, dysbiosis disrupts these protective pathways, leading to depletion of SCFA- and indole-producing taxa and accumulation of pro-inflammatory metabolites such as succinate. These metabolic shifts impair epithelial-immune crosstalk, amplify NF-κB-dependent inflammation, and compromise mucosal repair. Therapeutic strategies targeting microbial metabolites, including precision prebiotics, next-generation probiotics, engineered microbial consortia, and postbiotics, show translational promise. However, their clinical application remains constrained by interindividual variability, incomplete causal resolution, and challenges in targeted delivery. Integrative multi-omics approaches and mechanistically informed models are therefore essential to advance metabolite-based diagnostics and therapies for gut inflammation.
DOI: https://doi.org/10.37349/edd.2026.1005116
This article belongs to the special issue Inflammatory Diseases of the Gastrointestinal Tract

Metabolic dysfunction-associated steatotic liver disease (MASLD) is a systemic metabolic disorder closely related to insulin resistance, obesity, and type 2 diabetes mellitus. While sustained hyperglycemia and insulin resistance are central to the pathogenesis of MASLD, there is growing evidence suggesting that glucose dysregulation in this condition is heterogeneous and dynamic. Glycemic variability (GV), which reflects fluctuations in glucose levels over time, has emerged as a complementary dimension of glucose homeostasis and has been increasingly explored in relation to hepatic steatosis, fibrosis, and cardiometabolic risk. Experimental data indicate that recurrent glucose oscillations promote oxidative stress, inflammatory signaling, and fibrogenic activation, while observational studies have reported associations between short- and long-term GV and MASLD-related phenotypes in diverse populations. The aim of this review is to describe the mechanistic links between insulin resistance, GV, and MASLD, summarize the current clinical and population evidence evaluating GV in this context, and identify key methodological and conceptual gaps that should inform future research.
Metabolic dysfunction-associated steatotic liver disease (MASLD) is a systemic metabolic disorder closely related to insulin resistance, obesity, and type 2 diabetes mellitus. While sustained hyperglycemia and insulin resistance are central to the pathogenesis of MASLD, there is growing evidence suggesting that glucose dysregulation in this condition is heterogeneous and dynamic. Glycemic variability (GV), which reflects fluctuations in glucose levels over time, has emerged as a complementary dimension of glucose homeostasis and has been increasingly explored in relation to hepatic steatosis, fibrosis, and cardiometabolic risk. Experimental data indicate that recurrent glucose oscillations promote oxidative stress, inflammatory signaling, and fibrogenic activation, while observational studies have reported associations between short- and long-term GV and MASLD-related phenotypes in diverse populations. The aim of this review is to describe the mechanistic links between insulin resistance, GV, and MASLD, summarize the current clinical and population evidence evaluating GV in this context, and identify key methodological and conceptual gaps that should inform future research.
DOI: https://doi.org/10.37349/edd.2026.1005115

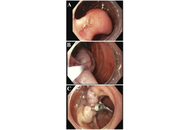
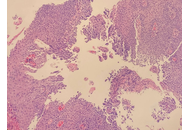
Gangliocytic paraganglioma (GP) is a rare tumor that involves the gastrointestinal system and often occurs in the second portion of the duodenum. This is the case of a 73-year-old female presenting for an unintentional 10-pound weight loss over the previous year and a computed tomography (CT) scan of the abdomen and pelvis showing a mass in the second portion of the duodenum with omental nodules. She had no other symptoms. An upper endoscopy revealed a pedunculated 5 cm polypoid mass that was endoscopically resected and found to be a GP on pathology. GP is the third most frequent histopathologic type of gastrointestinal neuroendocrine tumors (NET) after gastrinomas and somatostatinomas. Clinical presentations vary from asymptomatic patients to those having gastrointestinal bleeding, melena, anemia, and abdominal pain. GP presents as a single solid well-demarcated mass that is polypoid, pedunculated, or sessile. Histological features of GP show three cell types: spindle cells, ganglion or ganglion-like cells, and epithelioid cells. Biopsy specimens might not contain all 3 characteristic cell types; thus, definitive diagnosis could be challenging. Periampullary GP should be considered as a tumor with malignant potential. The optimal treatment has not been clarified, but endoscopic resection remains the most common treatment of choice. En bloc resection with negative margins for a pedunculated lesion is possible. This is a rare case of GP in a female patient presenting with weight loss only. It highlights the importance of considering GP in the differential diagnosis for duodenal masses, while keeping a broad differential due to the low rates of diagnostic biopsies. More research is needed to establish standardized management protocols for GP and improve patient care.
Gangliocytic paraganglioma (GP) is a rare tumor that involves the gastrointestinal system and often occurs in the second portion of the duodenum. This is the case of a 73-year-old female presenting for an unintentional 10-pound weight loss over the previous year and a computed tomography (CT) scan of the abdomen and pelvis showing a mass in the second portion of the duodenum with omental nodules. She had no other symptoms. An upper endoscopy revealed a pedunculated 5 cm polypoid mass that was endoscopically resected and found to be a GP on pathology. GP is the third most frequent histopathologic type of gastrointestinal neuroendocrine tumors (NET) after gastrinomas and somatostatinomas. Clinical presentations vary from asymptomatic patients to those having gastrointestinal bleeding, melena, anemia, and abdominal pain. GP presents as a single solid well-demarcated mass that is polypoid, pedunculated, or sessile. Histological features of GP show three cell types: spindle cells, ganglion or ganglion-like cells, and epithelioid cells. Biopsy specimens might not contain all 3 characteristic cell types; thus, definitive diagnosis could be challenging. Periampullary GP should be considered as a tumor with malignant potential. The optimal treatment has not been clarified, but endoscopic resection remains the most common treatment of choice. En bloc resection with negative margins for a pedunculated lesion is possible. This is a rare case of GP in a female patient presenting with weight loss only. It highlights the importance of considering GP in the differential diagnosis for duodenal masses, while keeping a broad differential due to the low rates of diagnostic biopsies. More research is needed to establish standardized management protocols for GP and improve patient care.
DOI: https://doi.org/10.37349/edd.2025.1005113

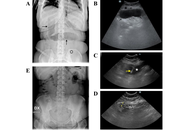
In the context of the various management options available for obesity, intragastric balloons (IGBs) represent a solution for patients at high surgical risk. Swallowable IGBs are devices that can be ingested orally and then inflated within the stomach to aid in weight loss. Although generally well tolerated, these devices may migrate and cause gastrointestinal obstructive symptoms. Here we report on two cases where these obstructive complications of swallowable IGBs were promptly and non-invasively diagnosed with ultrasonography (US), which also permitted conservative management via US-guided percutaneous aspiration of the impacted balloons. These cases demonstrate that the US may provide a rapid and effective tool for managing IGBs migration, potentially reducing the need for surgical intervention.
In the context of the various management options available for obesity, intragastric balloons (IGBs) represent a solution for patients at high surgical risk. Swallowable IGBs are devices that can be ingested orally and then inflated within the stomach to aid in weight loss. Although generally well tolerated, these devices may migrate and cause gastrointestinal obstructive symptoms. Here we report on two cases where these obstructive complications of swallowable IGBs were promptly and non-invasively diagnosed with ultrasonography (US), which also permitted conservative management via US-guided percutaneous aspiration of the impacted balloons. These cases demonstrate that the US may provide a rapid and effective tool for managing IGBs migration, potentially reducing the need for surgical intervention.
DOI: https://doi.org/10.37349/edd.2026.1005114
This article belongs to the special issue Advances in Hepato-gastroenterology: Diagnosis, Prognostication, and Disease Stratification

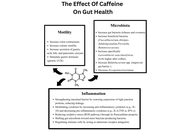
This review goes over the impact of caffeine consumption in inflammatory bowel disease (IBD), examining epidemiology, clinical outcomes, mechanistic studies, and translational research. Caffeine, a widely consumed methylxanthine, exerts diverse physiological effects on the gastrointestinal tract. Mechanistic and preclinical data offer plausible biological pathways by which caffeine could influence the IBD course. Caffeine’s antagonism of adenosine receptors may modulate immune cell activation and cytokine release; its effects on gut motility and secretion can alter symptom perception, and caffeine-mediated changes in intestinal epithelial barrier function, oxidative stress, and the gut microbiome have been demonstrated. These effects make it a lucrative investigational option, and various studies have demonstrated that caffeine intake may reduce the incidence of IBD and may even have disease-modifying effects in regular consumers. However, differences in caffeine source (coffee, tea, soda), dose, concurrent dietary patterns, and disease subtype (Crohn’s disease versus ulcerative colitis) limit definitive causal inference. Clinical implications remain cautious: while moderate caffeine intake may be tolerable and even helpful for many patients, individualized assessment is advisable, particularly for those with symptom-triggering sensitivity or overlapping functional bowel disorders. Future research should target mechanistic links and clinically meaningful outcomes to inform evidence-based dietary guidance for people with IBD.
This review goes over the impact of caffeine consumption in inflammatory bowel disease (IBD), examining epidemiology, clinical outcomes, mechanistic studies, and translational research. Caffeine, a widely consumed methylxanthine, exerts diverse physiological effects on the gastrointestinal tract. Mechanistic and preclinical data offer plausible biological pathways by which caffeine could influence the IBD course. Caffeine’s antagonism of adenosine receptors may modulate immune cell activation and cytokine release; its effects on gut motility and secretion can alter symptom perception, and caffeine-mediated changes in intestinal epithelial barrier function, oxidative stress, and the gut microbiome have been demonstrated. These effects make it a lucrative investigational option, and various studies have demonstrated that caffeine intake may reduce the incidence of IBD and may even have disease-modifying effects in regular consumers. However, differences in caffeine source (coffee, tea, soda), dose, concurrent dietary patterns, and disease subtype (Crohn’s disease versus ulcerative colitis) limit definitive causal inference. Clinical implications remain cautious: while moderate caffeine intake may be tolerable and even helpful for many patients, individualized assessment is advisable, particularly for those with symptom-triggering sensitivity or overlapping functional bowel disorders. Future research should target mechanistic links and clinically meaningful outcomes to inform evidence-based dietary guidance for people with IBD.
DOI: https://doi.org/10.37349/edd.2026.1005112
This article belongs to the special issue Inflammatory Diseases of the Gastrointestinal Tract

Herpes simplex esophagitis (HSE) is a viral infection of the esophagus caused by the herpes simplex virus (HSV), most commonly HSV-1. It predominantly presents among immunosuppressed individuals. Eosinophilic esophagitis (EoE) is a chronic, inflammatory, immune-mediated disease characterized by significant eosinophilic infiltration in the esophageal mucosa. It is often associated with atopic diseases, including asthma, food allergies, and eczema. Coexistence of HSE and EoE is rare and may be underdiagnosed due to challenges in diagnosing both conditions simultaneously. A major diagnostic dilemma can be traced to their histopathological similarities and differences. HSE is typically characterized by multinuclear giant cells containing intranuclear inclusions, while EoE involves eosinophilic infiltration in the esophageal epithelium. This report highlights the rare but remarkable coexistence between HSE and EoE secondary to a unique patient case. Although each condition may cause esophagitis individually, together—particularly in immunocompetent individuals—they do present a different diagnostic and therapeutic challenge.
Herpes simplex esophagitis (HSE) is a viral infection of the esophagus caused by the herpes simplex virus (HSV), most commonly HSV-1. It predominantly presents among immunosuppressed individuals. Eosinophilic esophagitis (EoE) is a chronic, inflammatory, immune-mediated disease characterized by significant eosinophilic infiltration in the esophageal mucosa. It is often associated with atopic diseases, including asthma, food allergies, and eczema. Coexistence of HSE and EoE is rare and may be underdiagnosed due to challenges in diagnosing both conditions simultaneously. A major diagnostic dilemma can be traced to their histopathological similarities and differences. HSE is typically characterized by multinuclear giant cells containing intranuclear inclusions, while EoE involves eosinophilic infiltration in the esophageal epithelium. This report highlights the rare but remarkable coexistence between HSE and EoE secondary to a unique patient case. Although each condition may cause esophagitis individually, together—particularly in immunocompetent individuals—they do present a different diagnostic and therapeutic challenge.
DOI: https://doi.org/10.37349/edd.2026.1005111

Digestive diseases comprise a diverse range of illnesses, which are prevalent worldwide and represent an important health issue. This is particularly relevant for the impact of metabolic dysfunction-associated steatotic liver disease (MASLD) due to its close association with the obesity pandemic, contributing to the escalation of MASLD as the most common form of chronic liver disease, and the main cause of liver cancer. Not only does MASLD reflect the deterioration of liver health, but it also has far-reaching consequences for the development of extrahepatic digestive diseases. Along with the progression of liver and digestive diseases to liver, colorectal and pancreatic cancer, the onset of inflammation in diseases of the digestive tract, drug-induced liver injury, and cholestasis, drives and contributes to the rise of these diseases in the future, which merit the attention of clinical and translational research to increase our understanding of the pathogenic mechanisms underlying these disorders in order to improve the diagnosis, management, and treatment. With this goal in mind, the current collaborative review gathers experts in a wide range of liver and digestive diseases to provide an up-to-date overview of the mechanisms of disease and identify novel strategies for the improvement of these important health issues.
Digestive diseases comprise a diverse range of illnesses, which are prevalent worldwide and represent an important health issue. This is particularly relevant for the impact of metabolic dysfunction-associated steatotic liver disease (MASLD) due to its close association with the obesity pandemic, contributing to the escalation of MASLD as the most common form of chronic liver disease, and the main cause of liver cancer. Not only does MASLD reflect the deterioration of liver health, but it also has far-reaching consequences for the development of extrahepatic digestive diseases. Along with the progression of liver and digestive diseases to liver, colorectal and pancreatic cancer, the onset of inflammation in diseases of the digestive tract, drug-induced liver injury, and cholestasis, drives and contributes to the rise of these diseases in the future, which merit the attention of clinical and translational research to increase our understanding of the pathogenic mechanisms underlying these disorders in order to improve the diagnosis, management, and treatment. With this goal in mind, the current collaborative review gathers experts in a wide range of liver and digestive diseases to provide an up-to-date overview of the mechanisms of disease and identify novel strategies for the improvement of these important health issues.
DOI: https://doi.org/10.37349/edd.2026.1005110

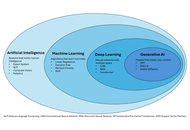
Interventional radiology (IR) is an ideal domain for artificial intelligence (AI) due to its data-intensive nature. This review provides a targeted guide for clinicians on AI applications in liver interventions, specifically focusing on hepatocellular carcinoma and portal hypertension. Key findings from recent literature demonstrate that AI models achieve high accuracy in predicting the response to transarterial chemoembolization and in non-invasively estimating the hepatic venous pressure gradient. Furthermore, emerging deep learning architectures, such as Swin Transformers, are outperforming traditional mRECIST criteria in longitudinal treatment monitoring. Despite these technical successes, the transition from “code to bedside” is hindered by limited external validation and the “black box” nature of complex algorithms. We conclude that the future of IR lies in the “AI-augmented” interventional radiologist paradigm, in which AI serves as a precision tool for patient selection and procedural safety rather than as a replacement for clinical judgment.
Interventional radiology (IR) is an ideal domain for artificial intelligence (AI) due to its data-intensive nature. This review provides a targeted guide for clinicians on AI applications in liver interventions, specifically focusing on hepatocellular carcinoma and portal hypertension. Key findings from recent literature demonstrate that AI models achieve high accuracy in predicting the response to transarterial chemoembolization and in non-invasively estimating the hepatic venous pressure gradient. Furthermore, emerging deep learning architectures, such as Swin Transformers, are outperforming traditional mRECIST criteria in longitudinal treatment monitoring. Despite these technical successes, the transition from “code to bedside” is hindered by limited external validation and the “black box” nature of complex algorithms. We conclude that the future of IR lies in the “AI-augmented” interventional radiologist paradigm, in which AI serves as a precision tool for patient selection and procedural safety rather than as a replacement for clinical judgment.
DOI: https://doi.org/10.37349/edd.2026.1005109
This article belongs to the special issue Advances in Hepato-gastroenterology: Diagnosis, Prognostication, and Disease Stratification

Aim:
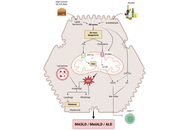
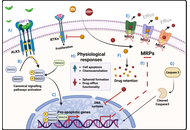
Hepatocellular carcinoma (HCC) accounts for 90% of liver tumors and is the fourth leading cause of cancer-related deaths worldwide. Current treatments have poor outcomes for HCC, highlighting the urgent need for new and effective therapies. Growth differentiation factor 11 (GDF11), a member of the TGF-β superfamily, regulates differentiation, proliferation, and migration processes, effects observed in cancer, including HCC. In this study, we aimed to investigate the chemosensitizing effects on human liver cancer cells.
Methods:
We pre-treated Huh7 and Hep3B cells with GDF11 50 ng/mL for 72 h in the presence of sorafenib (Sfb) or cisplatin (CDDP) and evaluated cellular response.
Results:
Pre-treatment with GDF11 lowered the IC50 of CDDP and Sfb in Huh7 cells. Similar effects were observed in Hep3B cells. Additionally, combining GDF11 with CDDP or Sfb significantly reduced cell viability and decreased the size and number of spheroids. Furthermore, we found that the chemosensitizing effect is initiated by GDF11 binding to the type I receptor ALK5. Inhibition of ALK5 abolished SMAD2 activation, impacting the chemosensitizing effects. Finally, GDF11, combined with Sfb or CDDP, reduced the activity of drug transporters MRP2, MRP3, and MRP4, which explains its chemosensitizing properties.
Conclusions:
GDF11 increases the sensitivity of HCC-derived cell lines to Sfb and CDDP by modulating the drug-efflux transporters MRP2, MRP3, and MRP4.
Aim:
Hepatocellular carcinoma (HCC) accounts for 90% of liver tumors and is the fourth leading cause of cancer-related deaths worldwide. Current treatments have poor outcomes for HCC, highlighting the urgent need for new and effective therapies. Growth differentiation factor 11 (GDF11), a member of the TGF-β superfamily, regulates differentiation, proliferation, and migration processes, effects observed in cancer, including HCC. In this study, we aimed to investigate the chemosensitizing effects on human liver cancer cells.
Methods:
We pre-treated Huh7 and Hep3B cells with GDF11 50 ng/mL for 72 h in the presence of sorafenib (Sfb) or cisplatin (CDDP) and evaluated cellular response.
Results:
Pre-treatment with GDF11 lowered the IC50 of CDDP and Sfb in Huh7 cells. Similar effects were observed in Hep3B cells. Additionally, combining GDF11 with CDDP or Sfb significantly reduced cell viability and decreased the size and number of spheroids. Furthermore, we found that the chemosensitizing effect is initiated by GDF11 binding to the type I receptor ALK5. Inhibition of ALK5 abolished SMAD2 activation, impacting the chemosensitizing effects. Finally, GDF11, combined with Sfb or CDDP, reduced the activity of drug transporters MRP2, MRP3, and MRP4, which explains its chemosensitizing properties.
Conclusions:
GDF11 increases the sensitivity of HCC-derived cell lines to Sfb and CDDP by modulating the drug-efflux transporters MRP2, MRP3, and MRP4.
DOI: https://doi.org/10.37349/edd.2025.1005108

Diverticulitis is one of the most common gastrointestinal causes of hospitalization in Western society. While previously characterized as a disease of older patients, new literature highlights an increasing incidence among the younger population. Over the past few decades, the understanding of etiology and management of diverticulitis has changed drastically. New data refute past beliefs while promoting other novel recommendations to mitigate incidence and subsequent complications. Data now confirms the safety and possible protective benefit of particulate food, while highlighting evidence-based approaches for the use of diagnostic imaging and antibiotics. We recognize modifiable and non-modifiable risk factors that are commonly seen throughout the literature and play a significant role in the management and prevention of diverticulitis. Emerging evidence also links chronic inflammation with subsequent microbial dysbiosis and alterations in the neuroendocrine system, leading to visceral hypersensitivity and perturbation of the gut-brain axis. This review provides a comprehensive update on acute uncomplicated diverticulitis according to the most recent evidence-based literature, encompassing the risks, diagnostic modalities, and management treatment regimens.
Diverticulitis is one of the most common gastrointestinal causes of hospitalization in Western society. While previously characterized as a disease of older patients, new literature highlights an increasing incidence among the younger population. Over the past few decades, the understanding of etiology and management of diverticulitis has changed drastically. New data refute past beliefs while promoting other novel recommendations to mitigate incidence and subsequent complications. Data now confirms the safety and possible protective benefit of particulate food, while highlighting evidence-based approaches for the use of diagnostic imaging and antibiotics. We recognize modifiable and non-modifiable risk factors that are commonly seen throughout the literature and play a significant role in the management and prevention of diverticulitis. Emerging evidence also links chronic inflammation with subsequent microbial dysbiosis and alterations in the neuroendocrine system, leading to visceral hypersensitivity and perturbation of the gut-brain axis. This review provides a comprehensive update on acute uncomplicated diverticulitis according to the most recent evidence-based literature, encompassing the risks, diagnostic modalities, and management treatment regimens.
DOI: https://doi.org/10.37349/edd.2025.1005107
This article belongs to the special issue Diverticulitis: Pathomechanism, Diagnosis and Treatment

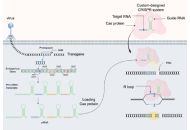
Gastric cancer (GC) is one of the most common malignant tumors, ranking fifth in incidence and third in mortality worldwide. China also bears a high burden of GC, second only to lung cancer. With the advancement of clustered regularly interspaced short palindromic repeats (CRISPR) technology, the mechanisms underlying the development and progression of various tumor types have been elucidated. This article summarizes the application of CRISPR technology in the functional genomics identification and target screening of GC genes, explores the use of chimeric antigen receptor T (CAR-T) cell therapy for solid gastric tumors, and discusses the progress and significance of CRISPR technology in constructing GC models using organoids.
Gastric cancer (GC) is one of the most common malignant tumors, ranking fifth in incidence and third in mortality worldwide. China also bears a high burden of GC, second only to lung cancer. With the advancement of clustered regularly interspaced short palindromic repeats (CRISPR) technology, the mechanisms underlying the development and progression of various tumor types have been elucidated. This article summarizes the application of CRISPR technology in the functional genomics identification and target screening of GC genes, explores the use of chimeric antigen receptor T (CAR-T) cell therapy for solid gastric tumors, and discusses the progress and significance of CRISPR technology in constructing GC models using organoids.
DOI: https://doi.org/10.37349/edd.2025.1005106

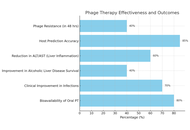
Liver cirrhosis is a condition characterized by scarring of liver tissue resulting from impaired liver function and systemic complications. It shows symptoms like jaundice, ascites, and hepatic encephalopathy. It has significant mortality and morbidity worldwide. As the study of microbial dysbiosis grows, it investigates how an imbalance in gut bacteria can speed up the progression of liver cirrhosis by spreading bacteria, endotoxins, and inflammation all over the body. Dysbiosis damages the gut–liver axis and eventually the liver. The study aims to analyze the therapeutic potential of bacteriophage therapy in liver cirrhosis. Bacteriophage treatment is a new focused method for treating microbial dysbiosis. Bacteriophages are viruses that target and attack harmful pathogens without affecting the helpful ones or causing an imbalance in the gut microbiota’s equilibrium. Since broad-spectrum antibiotics can affect the gut microbiota and lead to antibiotic resistance, phages are a better alternative due to their selectivity. According to preclinical research conducted in animal models, bacteriophage therapy can lower the bacterial load, enhance liver function tests, and decrease the systemic inflammatory indicators. Bacteriophage safety, as well as potential effectiveness in balancing gut microbiota, reducing systemic inflammation, and relieving symptoms such as hepatic encephalopathy, has been shown by preliminary clinical trials and case reports. However, issues like phage-resistant bacteria, patient-specific gut microbiota variation, and lack of clinical trials continue to prevent general use. Additional research is required to determine if it can be used in clinical practice, including large clinical trials and individualized strategies. Bacteriophage therapy is a promising and new technique for improving liver cirrhosis outcomes.
Liver cirrhosis is a condition characterized by scarring of liver tissue resulting from impaired liver function and systemic complications. It shows symptoms like jaundice, ascites, and hepatic encephalopathy. It has significant mortality and morbidity worldwide. As the study of microbial dysbiosis grows, it investigates how an imbalance in gut bacteria can speed up the progression of liver cirrhosis by spreading bacteria, endotoxins, and inflammation all over the body. Dysbiosis damages the gut–liver axis and eventually the liver. The study aims to analyze the therapeutic potential of bacteriophage therapy in liver cirrhosis. Bacteriophage treatment is a new focused method for treating microbial dysbiosis. Bacteriophages are viruses that target and attack harmful pathogens without affecting the helpful ones or causing an imbalance in the gut microbiota’s equilibrium. Since broad-spectrum antibiotics can affect the gut microbiota and lead to antibiotic resistance, phages are a better alternative due to their selectivity. According to preclinical research conducted in animal models, bacteriophage therapy can lower the bacterial load, enhance liver function tests, and decrease the systemic inflammatory indicators. Bacteriophage safety, as well as potential effectiveness in balancing gut microbiota, reducing systemic inflammation, and relieving symptoms such as hepatic encephalopathy, has been shown by preliminary clinical trials and case reports. However, issues like phage-resistant bacteria, patient-specific gut microbiota variation, and lack of clinical trials continue to prevent general use. Additional research is required to determine if it can be used in clinical practice, including large clinical trials and individualized strategies. Bacteriophage therapy is a promising and new technique for improving liver cirrhosis outcomes.
DOI: https://doi.org/10.37349/edd.2025.1005105
This article belongs to the special issue Gut Microbiota towards Personalized Medicine in Metabolic Disease
 Previous
Previous