Affiliation:
Liver Diseases Research Lab, Department of Biochemistry, Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Pondicherry 605006, India
ORCID: https://orcid.org/0009-0008-6006-8257
Affiliation:
Liver Diseases Research Lab, Department of Biochemistry, Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Pondicherry 605006, India
ORCID: https://orcid.org/0009-0008-9471-1458
Affiliation:
Liver Diseases Research Lab, Department of Biochemistry, Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Pondicherry 605006, India
Email: balasubramaniyan.v@jipmer.edu.in
ORCID: https://orcid.org/0000-0003-1708-4864
Explor Dig Dis. 2025;4:100588 DOI: https://doi.org/10.37349/edd.2025.100588
Received: April 09, 2025 Accepted: July 30, 2025 Published: August 24, 2025
Academic Editor: Jose Carlos Fernandez-Checa, Institute of Biomedical Research of Barcelona (IIBB-CSIC), Spain
Hepatocellular carcinoma (HCC) ranks as the sixth most diagnosed cancer and the third most common cancer-related death globally. The underlying precise molecular mechanisms for its progression remain poorly understood. Interestingly, approximately 90% of HCC-related deaths are not due to the primary tumor itself but rather to its difficult-to-treat metastatic spread. Despite sorafenib being the first-line therapy for HCC, challenges such as drug resistance, frequent recurrence, and metastasis contribute to poor prognosis. In this context, alternative therapeutic strategies are urgently needed. A broad spectrum of phytochemicals, including polyphenolic derivatives, flavonoids, carotenoids, alkaloids, terpenes, lignans, and saponins, has shown considerable promise as potential anti-cancer agents, both in vitro and in vivo. These natural plant-derived compounds exhibit distinct and overlapping mechanisms of action, characterized by their antioxidant, anti-inflammatory, and anti-cancer properties, offering a novel approach to HCC treatment. An extensive literature search was conducted from 2010 to 2024 using reputable electronic databases such as MEDLINE, Embase, Google Scholar, Science Direct, and other reliable sources using different keywords, including HCC, medicinal plants in HCC, HCC metastasis, and mechanism of action of medicinal plants in HCC, among others. This comprehensive review aims to summarize the potential role of plant-based bioactive components in combating HCC through various cellular mechanisms, highlighting their therapeutic potential in the management of both primary and metastatic disease.
Hepatocellular carcinoma (HCC) is the 3rd leading cause of cancer-related death and 6th most common type of cancer globally. According to the Global Cancer Observatory (GLOBOCON 2022, version 1.1), approximately 870,000 new cases of liver cancer were reported globally, with around 760,000 deaths attributed to the disease, reflecting the varying incidence rates worldwide [1]. Notably, the expected incidence of HCC is projected to increase by 55% from 2020 to 2040. The majority of HCC cases are concentrated in East and Southeast Asia, with China accounting for 62.4% of cases, followed by Japan (7.0%), India (5.3%), Thailand (4.2%), and Vietnam (4.0%) [2, 3]. Most HCC cases occur in individuals with cirrhosis, with the highest mortality rate observed in alcoholic liver disease (ALD), followed by MAFLD (metabolic dysfunction-associated fatty liver disease), and HBV (hepatitis B virus) or HCV-related cirrhosis [2, 3].
Moreover, Mongolia has the highest HCC incidence globally, while China reports the majority of cases in Asia, followed by Japan, India, Thailand, and Vietnam [4]. HCC incidents vary by country due to differences in screening practices, etiological factors, and treatment strategies. Hepatic viral diseases, particularly HBV and HCV, were historically the leading cause of HCC, but MAFLD now surpasses them in many countries, except in Africa, where HBV and aflatoxin exposure remain dominant risk factors. In regions like Northern Africa, Western Europe, and the USA, HCV remains a significant contributor to HCC [5]. Furthermore, metabolic disorders such as alpha 1-antitrypsin deficiency, Wilson’s disease, porphyria, and hereditary hemochromatosis are linked to the development of HCC, often in the context of cirrhosis [6]. Autoimmune conditions, including autoimmune hepatitis and primary sclerosing cholangitis, also play a role in HCC pathogenesis [7].
In HCC, invasion typically begins within the liver parenchyma, known as intrahepatic metastasis, where cancer cells circulate and infiltrate adjacent liver tissues. This process is facilitated by various molecular mechanisms, including alterations in cell-cell and cell-extracellular matrix (ECM) interactions, dysregulation of signaling pathways, and remodeling of the ECM. Invasion can be classified as either micro or macro. The majority of invasion occurs in the portal vein (approximately 60%), while only 20% occurs in the hepatic vein [8]. On the other hand, metastasis refers to the spread of cancer cells from the primary malignancy site to distant parts of the body. Radiological reports indicate that metastasis occurs through various routes, including the hematogenous route, lymphatic spread, bile duct invasion, and direct extrahepatic invasion. The hematogenous route is the most common pathway for HCC metastasis, primarily due to the invasion of intrahepatic arterioles and venules that drain into the pulmonary circulation. The lungs (the most common site), bones, and adrenal glands are frequently affected via the hematogenous route, while the diaphragm, abdominal wall, and peritoneum are commonly involved through direct extrahepatic invasion [9, 10].
A key molecular mechanism driving HCC invasion is the deregulation of epithelial-mesenchymal transition (EMT). EMT is a biological process in which epithelial cells lose their polarity and cell-cell adhesion, acquiring mesenchymal traits that enable them to migrate, invade surrounding tissues, and resist apoptosis. Previously, EMT was viewed as a simple binary transition between epithelial and mesenchymal states. However, recent studies reveal that EMT involves varying degrees of phenotypic changes, including partial EMT, intermediate EMT, extreme EMT, and ameboid EMT. In all phases, the ameboid stage shows a high potential for extravasation and metastasis due to squeezing mobility [11]. EMT changes are driven by factors such as disruption of cell-cell adhesion proteins, activation of transcription factors, alterations in cell surface proteins, increased production of degrading enzymes, and activation of key signaling pathways. Molecular markers like E-cadherin and cytokeratin are associated with epithelial cells, while N-cadherin and vimentin are linked to mesenchymal cells with migratory capabilities. Mechanisms altering these protein expressions include upregulation of retinoic acid receptor γ (RARγ), a nuclear receptor that promotes cell migration and invasion by downregulating E-cadherin, and homeodomain-interacting protein kinase 8 (HDGF8), a histone demethylase that also facilitates HCC cell migration [12, 13]. In addition, cytoskeletal remodeling, regulated by RhoGAP (an enzyme that hydrolyzes GTP to GDP), plays a crucial role in cell migration by modulating E-cadherin expression and actin polymerization [14].
Angiogenesis is a key process in HCC invasion and metastasis, involving the activation of endothelial cells to form new blood vessels, which provide nutrients to cancer cells. This process is regulated by various mechanisms; under hypoxic conditions, increased expression of HIF-1α (hypoxia inducible factor-1α) (a hypoxia-induced transcription factor) activates several angiogenic factors in HCC [15]. Pro-angiogenic factors include epidermal growth factor (EGF) [16], platelet-derived growth factor (PDGF) [17], fibroblast growth factor (FGF) [18], endoglin [19], and leptin [20], while anti-angiogenic factors like thrombospondin-1 [21] and endostatin [22] counteract this process. Invasion also involves complex interactions within the tumor microenvironment (TME), which comprises stromal cells such as hepatic stellate cells (HSCs), cancer-associated fibroblasts (CAFs), tumor-associated macrophages (TAMs), and tumor-associated neutrophils (TANs) [23]. HSCs play a critical role in invasion and metastasis by secreting pro-angiogenic factors like vascular endothelial growth factor receptor (VEGFR) and PDGF, along with IL-8, which stimulates new blood vessel formation [24]. Additionally, HSCs modulate immune responses by promoting the transition of macrophages from the M1 to M2 phenotype via the CCL2/CCR2 pathway [25].
The treatment of HCC faces significant challenges due to limited therapeutic options. Clinical management is complex and varies depending on tumor stage, liver function, and the patient’s overall health. The Barcelona Clinic Liver Cancer (BCLC) staging system and the Child-Pugh score are widely accepted tools for guiding therapeutic decisions [26, 27]. Patients in the early stages (BCLC 0/A) with preserved liver function and minimal tumor burden are typically eligible for potentially curative treatments, including hepatic resection, local ablation [such as radiofrequency ablation (RFA) or microwave ablation (MWA)], or liver transplantation (LT). The choice among these options depends on factors such as portal hypertension, bilirubin levels, and the presence of multiple nodules [28]. Hepatic resection is best suited for non-cirrhotic or cirrhotic patients with a single nodule (BCLC stage 0 or A), Child-Pugh A liver function, and no significant portal hypertension. Surgical resection offers a 5-year survival rate of ~ 70% though tumor recurrence is common, occurring in 75–80% of patients within 5 years. Recurrence is especially prevalent in cases related to viral hepatitis, particularly HCV [29, 30]. For patients who are not suitable for surgery, LT is a viable alternative. Eligibility typically follows the Milan criteria (a single lesion ≤ 5 cm or 2–3 nodules ≤ 3 cm without vascular invasion). LT provides favourable long-term outcomes, with low recurrence rates and a 10-year survival rate of around 70% [28]. In regions with limited deceased donor organs, living donor LT is practiced. Post-transplant immunosuppressants like tacrolimus are essential to prevent graft rejection, which is closely linked to recurrence risk. Sirolimus, however, has shown potential in reducing tumor progression [31, 32]. For patients ineligible for surgery or transplantation, non-surgical locoregional therapies such as RFA, MWA, percutaneous ethanol injection (PEI), and transarterial chemoembolization (TACE) are preferred. RFA is especially effective for nodules smaller than 2 cm and serves as an alternate to surgery for early-stage HCC, with a median overall survival of about 60 months and 5-year recurrence rates of 50–70% [33]. TACE is the first-line therapy for intermediate-stage HCC (BCLC-B), particularly in patients with preserved liver function (Child-Pugh A) and no vascular invasion or metastasis. More recently, selective internal radiation therapy (SIRT) with yttrium-90 microspheres has been used palliatively in BCLC-B patients; however, phase 3 trials have not demonstrated superior survival compared to sorafenib, alone or combined [34, 35].
Advanced HCC with BCLC-D patients are treated with first-line or second-line systemic therapies. Current systemic therapies include targeted therapy and immunotherapy. Sorafenib, an oral multi-kinase inhibitor, targets serine/threonine kinases RAF-1 and RAF, as well as tyrosine kinases such as VEGFR-1, -2, -3, and PDGFR-β, which are crucial in cancer development. Approved in 2007 as the first-line treatment for advanced HCC, sorafenib has been associated with adverse effects including hand-foot syndrome (7.0%), asthenia (7.4%), and diarrhea (13.1%), which can impact quality of life and lead to treatment discontinuation [36, 37]. Another option, regorafenib also a multi-kinase inhibitor targets VEGFRs 1–3, TIE2, RET, and RAF-1. It inhibits tumor cell proliferation, induces apoptosis, and exerts anti-angiogenic effects by blocking multiple pathways [38]. However, it is also linked to adverse reactions such as fatigue, hypertension, hand-foot skin reactions, and gastrointestinal symptoms, including diarrhea, nausea, and anorexia. Hepatotoxicity is a particular concern in HCC patients, who often have compromised liver function [39].
Another treatment option for HCC is the combination of atezolizumab and bevacizumab. Atezolizumab is a monoclonal antibody that inhibits PD-L1 (programmed death-ligand 1), while bevacizumab targets VEGF, a key mediator of angiogenesis [40]. In 2020, this combination was approved as a new standard treatment for patients with unresectable HCC. However, atezolizumab may cause immune-related adverse events such as hepatitis, pneumonitis, and colitis, while bevacizumab is associated with hypertension, bleeding, thromboembolism, and proteinuria. The combination therapy can increase the overall risk of these side effects [41]. A comparative overview of these therapies, including their mechanisms, clinical outcomes, and toxicity profiles, is presented in Table 1.
Systemic treatments for advanced HCC: a comparative analysis of efficacy, survival outcomes, and safety profiles
| Drug | Target | Benefits | Median OS and hazard ratio | Common adverse effects | Reference |
|---|---|---|---|---|---|
| Sorafenib | TKI—VEGFR, PDGFR, RAF | First-line for advanced HCC, delays HCC progression | 6.5 months (5.56–7.56) and 0.68 | HFSR, diarrhea, fatigue, hypertension | [36] |
| Donafenib | TKI—RAF/MEK/ERK | Modified structure of sorafenib | 12.1 months and 0.831 | HFSR, diarrhea, and elevated LFT | [42] |
| Lenvatinib | TKI—VEGFR, FGFR, PDGFR, RET, KIT | Non-inferior to sorafenib, higher response rate (24%) | 13.6 months (range from 12.1–14.9) and 0.92 | Palmar-plantar erythrodysesthesia, hypertension, weight loss, proteinuria, diarrhea | [43] |
| Regorafenib | TKI—VEGFR, PDGFR, FGFR | Second-line after sorafenib, OS benefit (~ 3 months) | 10.6 months (range from 9.1–12.1) and 0.63 | Fatigue, diarrhea, hypertension, HFSR | [38] |
| Cabozantinib | TKI—MET, VEGFR, AXL | Second/Third-line, OS and PFS benefit, activity in bone/lung metastases | 10.2 months and 0.76 | Diarrhea, HFSR, fatigue, increased LFTs | [44] |
| Ramucirumab | Anti-VEGFR-2 monoclonal antibody | Second-line if AFP > 400 ng/mL, improves OS in AFP-high HCC | 8.5 months (range from 7.0–10.6) and 0.710 | Hypertension, proteinuria, bleeding, fatigue | [45] |
| Apatinib | TKI of VEGFR-2 | Second-line treatment after chemotherapy failure | 8.7 months (range from 7.5–9.8) and 0.785 | Hypertension, HFSR | [46] |
| FOLFOX (5-FU + leucovorin + oxaliplatin) | Cytotoxic chemotherapy | Some benefit in Asia, used in advanced/metastatic HCC | 5.9 months and 0.75 | Hematological symptoms, diarrhea, neuropathy | [47] |
| Nivolumab + ipilimumab | Checkpoint inhibitors (PD-1 + CTLA-4) | Active in metastatic HCC | 23.7 months (range from 18.8–29.4) and 0.76 | Hepatitis-colitis, endocrinopathies, skin rash | [48] |
| Atezolizumab + bevacizumab | PD-L1 + VEGF inhibition | First-line standard in advanced HCC | 19.2 months (range from 17.0–23.7) and 0.66 | Hypertension, GI bleed, fatigue, immune hepatitis | [40] |
5-FU: 5-fluorouracil; AFP: alpha-fetoprotein; GI: gastrointestinal; HCC: hepatocellular carcinoma; HFSR: hand-foot skin reaction; LFT: liver function test; OS: overall survival; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; PFS: progression-free survival; TKI: tyrosine kinase inhibitor; VEGF: vascular endothelial growth factor; VEGFR: VEGF receptor
Another immunotherapy target is CTLA-4, a CD28 homolog that inhibits T-cell activation by preventing CD28 from binding to CD80/CD86. Monoclonal antibodies against CTLA-4 reduce helper T-cell activity and enhance regulatory T-cell (Treg) function, thereby suppressing immune responses. A phase I/II study (NCT02519348) evaluated tremelimumab (anti-CTLA-4 antibody) combined with durvalumab (T300 + D) versus monotherapy with either agent. The combination showed a higher overall response but also a greater incidence of adverse events compared to targeted therapies [49].
HCC remains a major clinical challenge due to limited treatment options and significant side effects associated with current therapies. Plant-derived phytochemicals offer a promising alternative, as they can reduce cancer burden without causing severe toxicity to normal tissues.
These compounds act through various mechanisms, including antioxidant, anti-inflammatory, and pro-apoptotic pathways (Figure 1). Phytochemicals are classified into groups such as polyphenols, alkaloids, and flavonoids, all of which have demonstrated anticancer properties. Compared to conventional chemotherapy, phytochemicals generally exhibit better metabolism and biotransformation, making them both safer and potentially more effective. Plant-based therapies not only enhance treatment efficacy but also help mitigate treatment-related side effects.
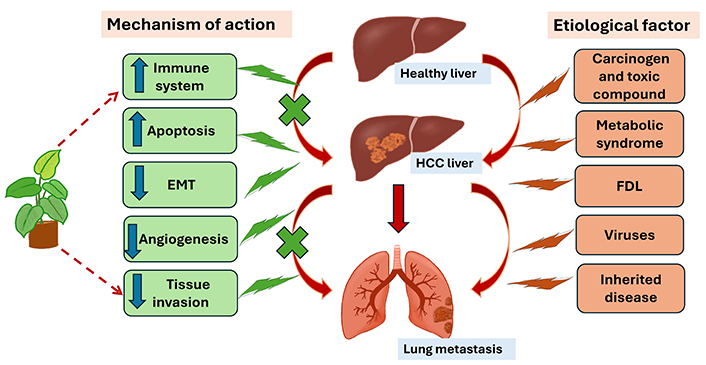
Overview of the etiological factors contributing to HCC. The figure also highlights the mechanisms of medicinal plants in suppressing tumor growth and metastasis. EMT: epithelial-mesenchymal transition; FDL: fatty liver disease; HCC: hepatocellular carcinoma
Numerous phytochemicals have significantly advanced cancer research and show promising therapeutic potential in HCC, primarily by modulating key molecular pathways involved in tumor growth, survival, and metastasis.
Alkaloids are cyclic compounds containing one or more basic nitrogen atoms, making them unique and valuable in medicine. They are classified based on their chemical structure and the plant source from which they are derived. For instance, vinblastine, extracted from Vinca rosea (Apocynaceae family), is classified as an indole alkaloid. It, along with its analogue vincristine, exhibits anticancer activity by inhibiting microtubule polymerization [50]. Another class, proto-alkaloids, consists of plant metabolites derived from aromatic amino acids such as tryptophan, tyrosine, or phenylalanine. An example is colchicine, which is derived from Colchicum autumnale and inhibits cancer progression by disrupting the cytoskeleton of tumor cells, thereby reducing tumor burden in HCC [51]. Camptothecin, a pyrroloquinoline alkaloid extracted from the plant Camptotheca acuminata, exhibits antioxidant properties by decreasing the expression of the Nrf2 protein in an HCC mouse model [52]. Moreover, some alkaloids can modulate immune responses, enhancing the immune system’s ability to recognize and eliminate malignant cells.
Flavonoids are secondary metabolites classified as polyphenolic compounds, abundantly present in fruits, vegetables, tea, and medicinal plants. They are characterized by aromatic rings with specific degrees of hydroxylation and various substituents that distinguish each flavonoid [53]. These compounds have attracted significant attention due to their ability to modulate signaling pathways involved in tumor development, progression, angiogenesis, and metastasis. Quercetin, found in high concentrations in various berries, onions, apples, and red wine, inhibits the proliferation of liver cancer cells by reducing inflammation and inducing cell cycle arrest [54]. Apigenin, extracted from Petroselinum crispum (parsley), demonstrates antimetastatic potential in an in vivo HCC model by altering miRNA expression and inducing apoptosis [55]. Luteolin, a flavonoid mainly found in broccoli, pepper, thyme, and celery, exhibits anticancer activity in vitro HCC models by modulating the expression of the p53 protein [56]. Genistein, derived from dyer’s broom (Genista tinctoria), reverses the EMT markers and inhibits cancer cells’ migration and invasion [57].
Terpenoids are among the most diverse groups of phytochemicals, derived from terpenes composed of repeating isoprene units that form characteristic carbon skeletons. Based on the number of isoprene units, terpenes are classified into several groups, including monoterpenoids, sesquiterpenoids, diterpenoids, and triterpenoids [58]. Tanshinone I, a diterpenoid found in lavender, peppermint, cherries, celery seeds, and lemongrass, exhibits anticancer activity by inducing apoptosis and inhibiting p53-mediated autophagy in the HepG2 cell line model [59]. Parthenolide, a sesquiterpene lactone derived from Tanacetum parthenium (feverfew), exerts anticancer effects by modulating the immune system, reducing inflammation, and interfering with signaling pathways involved in the inflammatory response [60]. Ursolic acid, a triterpenoid found in apple peel, rosemary, thyme, oregano, and lavender, functions as an antioxidant and anti-inflammatory agent, and also reverses sorafenib resistance in HCC cells in an in vitro model [61]. Furthermore, saponins, a class of triterpenoids, exhibit anticancer properties by inducing apoptosis and blocking the β-catenin signaling pathway in the HCC mouse model [62]. Additionally, various terpenoids have been shown to inhibit angiogenesis, migration, and invasion of cancer cells, further highlighting their potential in cancer therapy.
Polyphenols have shown strong potential to inhibit cancer progression in both in vitro and in vivo models. These compounds are classified based on their structure and function. For example, flavonoids, found in the root bark of plant species such as Ramulus mori (Moraceae family) and Sophora flavescens (Leguminosae family), act as antioxidants and anti-inflammatory agents, and inhibit the proliferation of HCC cells in vitro by modulating the ERK signaling pathway [63]. Stilbenes, another class of polyphenols found in red grapes and peanuts, exhibit anticancer and antimetastatic properties in vitro HCC models by inducing autophagy [64]. Phenolic acids, primarily present in fruits (especially berries), vegetables, cereals, legumes, and beverages such as coffee and wine, demonstrate anticancer activity by inducing apoptosis and modulating the TME in both in vivo and in vitro [65]. Lignans combat HCC through multiple mechanisms, including the induction of apoptosis and the inhibition of angiogenesis. Sesame and flax seeds are among the most concentrated dietary sources of lignans [66].
Several medicinal plants have shown promising therapeutic potential against HCC through mechanisms including the induction of apoptosis, inhibition of metastasis, anti-inflammatory effects, and modulation of key oncogenic signaling pathways (Table 2). Below is an overview of key phytochemicals derived from selected medicinal plants that have shown efficacy in preclinical HCC metastasis models, highlighting their molecular targets and proposed modes of action.
Key medicinal plants and phytochemicals with proven efficacy against HCC metastasis
| S. No. | Scientific name | Common name | Active compound | Mechanism of action | Therapeutic potential | Reference |
|---|---|---|---|---|---|---|
| Apoptosis and cell cycle arrest | ||||||
| 1 | Ferula assa-foetida | Asafoetida-devil’s dung | Farnesiferol C | ↑ caspase activation results in increased apoptosis | Induction of apoptosis | [67] |
| 2 | Petroselinum crispum | Parsley | Apigenin | Induction of cell cycle arrest and apoptosis, inhibiting the PI3K/Akt signaling pathways by overexpression of miR-199a/b-5p | Cell cycle arrest, apoptosis | [55] |
| Activates anti-angiogenic factor | ||||||
| 3 | Berberis vulgaris | Barberry | Berberine | Block the HIF-1α/VEGF axis, reduced expression of Id-1 by inactivation of p16INK4a/RB pathway | Anti-angiogenic | [68] |
| 4 | Solanum lycopersicum | Tomato | Lycopene | ↓ expression of HIF-1α, VEGF, CD31, MMP-2, and MMP-9 | Suppression of neovascularization | [69] |
| Metabolic reprogram | ||||||
| 5 | Picrorhiza kurroa | Kutki | Picroside II | ↓ glycolytic enzyme expression activity | Metabolic reprogramming | [70] |
| 6 | Silybum marianum | Milk thistle | Silybin | Activating the AMPK-DR5 pathway inhibits intracellular ATP levels and glycolysis | ↓ ATP production and glycolysis | [71] |
| Reduces tissue invasion | ||||||
| 7 | Arctium lappa | Greater burdock | Arctigenin | Wnt/β-catenin signaling, ↑ expression of E-cadherin, ↓ N-cadherin, and vimentin, prevent EMT | EMT suppression | [72] |
| 8 | Camptotheca acuminata | Happy tree | Camptothecin | ↓ expression of Nrf2, ↑ expression of E-cadherin, and ↓ N-cadherin | EMT inhibition | [73] |
| 9 | Curcuma longa | Turmeric | Curcumin | ↓ expression of MMPs-2/9, VEGF,inhibit the PI3K/Akt/mTOR/NF-κB signaling | EMT suppression, angiogenesis inhibition | [74] |
| 10 | Dioscorea zingiberensis | Zingiber yam | Diosgenin | Inhibit platelet activation, inhibit P2Y2 receptor activity, and ↑ E-cadherin expression | EMT inhibition and reducing metastatic potential | [75] |
| 11 | Glycyrrhiza inflata | Chinese licorice | Licochalcone A | Downregulation of the MKK4/JNK signaling pathway and NF-κB transcriptional activation | EMT inhibition | [76] |
| 12 | Glycine max | Soybean | Genistein | Inhibiting the EIF5A2/PI3K/Akt pathway, ↑ miR-1275, attenuate the EMT and stemness | EMT & stemness inhibition | [77] |
| 13 | Panax quinquefolius | Ginseng | Ginsenoside | Inhibit the HIF-1α and NF-κB signaling pathway, preventing EMT by ↓ vimentin and ↑ E-cadherin | EMT suppression | [78] |
| 14 | Plumbago zeylanica | Chitrak | Plumbagin | ↑ expression of E-cadherin and ↓ N-cadherin, vimentin, and snail, preventing EMT | EMT suppression | [79] |
| 15 | Rosmarinus officinalis | Rosemary | Rosmarinic acid | Inhibition of PI3K/Akt/mTOR signal pathway | Anti-invasion, anti-survival | [80] |
| 16 | Salvia miltiorrhiza | Danshen | Tanshinone IIA | ↓ expression of Rho GTPases regulates the cytoskeleton remodeling | EMT suppression | [81] |
| 17 | Scutellaria baicalensis | Chinese skullcap | Baicalein | ↓ expression of MMP-2, MMP-9, ↑ expression of TIMP-1 and 2 | Anti-invasion | [82] |
| 18 | Thymus vulgaris | Thyme | Carvacrol | Reduced the activity of argyrophilic nucleolar organizing regions, proliferating cell nuclear antigen, and MMPs-2/9 | Suppresses proliferation & metastasis | [83] |
| 19 | Withania somnifera | Ashwagandha | Withaferin A | ↓ expression of CD44, CD90, and EpCAM, inhibition of PI3K/Akt signaling pathway by ↑ miR-200c | EMT suppression | [84] |
| Induces autophagy | ||||||
| 20 | Allium sativum L. | Garlic | Allicin | Induced autophagy | Cell survival regulation | [85] |
| 21 | Rheum palmatum | Turkish rhubarb | Emodin | Induced S and G2/M phase cell cycle arrest, induced autophagy, suppressed Wnt/β-catenin pathways | Inhibits proliferation and metastasis | [86] |
| Activation of the immune system | ||||||
| 22 | Andrographis paniculata | King of bitters | Andrographolide | ↑ miR-22-3p expression level and ↓ HMGB1 and MMP-9 expression levels | Immunomodulation | [87] |
| 23 | Crocus sativus | Saffron crocus | Crocetin | ↑ expression of SHP-1, reduced activation STAT3, ↓ expression of MMP-9 | Inhibition of inflammation and invasion | [88] |
| 24 | Gentiana macrophylla | Qinjiao | Luteoloside | ↓ ROS level, ↓ expression of NLRP3, ↓ secretion of IL-1β | Anti-inflammatory response | [89] |
| 25 | Salvia miltiorrhiza | Danshen | Cryptotanshinone | ↓ IDO1 enzyme activity modulates immunoregulation | T cell modulation, immunoregulation | [90] |
| 26 | Terminalia bellirica | Bahera | Tannins | Modulating tumor immune microenvironment, restoration of CD8+ T cell infiltration and function | Reversal of immune suppression in the tumor microenvironment | [91] |
EMT: epithelial-mesenchymal transition; HCC: hepatocellular carcinoma; HIF-1α: hypoxia inducible factor-1α; Id-1: inhibitor of differentiation-1; IDO1: indoleamine 2,3-dioxygenase 1; MMP-2: matrix metalloproteinase-2; RB: retinoblastoma; VEGF: vascular endothelial growth factor
Curcumin, a potent polyphenol extracted from Curcuma longa of the Zingiberaceae family, is widely found in the southeastern and southern regions of tropical Asia. It has been a key component of Ayurvedic and traditional Chinese medicine for centuries [92]. It has gained attention for its anti-proliferative, antimetastatic, and anti-inflammatory properties, as well as its ability to regulate multiple signaling pathways, making it a promising anticancer agent. A study reported that curcumin modulates the expression of cell adhesion markers—E-cadherin, N-cadherin, vimentin, and fibronectin, thereby regulating EMT through the TGF-β1 (transforming growth factor-β1) pathway and inhibiting HIF-1α in HepG2 cells, ultimately reducing tumor invasion and migration [93].
Curcumin acts on multiple molecular targets, including TGF-β, toll-like receptors (TLRs), and matrix metalloproteinases (MMPs), and also inhibits HCV replication, highlighting its antiviral potential [94, 95]. Curcumin inhibits cancer cell proliferation by mechanisms such as suppressing CDK2 activity in colon cancer [96]. In vitro studies have further demonstrated that curcumin induces cell cycle arrest at the S phase by downregulating cyclin A1 and promotes apoptosis by upregulating pro-apoptotic proteins, including Bax and caspase-3 [97]. It further suppresses cancer progression by blocking key signaling pathways such as NF-κB and Wnt, which are involved in cell proliferation and migration [98]. The combination of curcumin with metformin enhances therapeutic efficacy compared to curcumin alone by targeting the PI3K/Akt/mTOR/NF-κB and EGFR/STAT3 pathways, thereby suppressing angiogenesis and metastasis [74]. However, curcumin faces limitations such as poor absorption, rapid metabolism, and high clearance, with most of it excreted in unmetabolized sulfated or glucuronidated forms. A study using pH-sensitive nanoparticles co-loaded with doxorubicin and curcumin demonstrated improved drug delivery and enhanced inhibition of HCC proliferation [99].
The Araliaceae family, particularly the Panax genus, produces ginsenosides—also known as gintonin—a group of triterpenoid saponins derived from ginseng with notable anti-HCC properties [100]. Ginseng, primarily found in China and India, contains various ginsenoside subtypes, including ocotillol-type pseudo-ginsenosides, oleanane, protopanaxatriol (PPT), and protopanaxadiol (PPD). Among these, PPD significantly inhibits HCC cell migration and invasion. A recent study reported that PPD reduces STAT3 phosphorylation, thereby preventing its dimerization and nuclear translocation, leading to downregulation of TWIST1 expression. This effect reverses EMT in HCC by increasing E-cadherin levels and decreasing N-cadherin levels [101]. Another PPD derivative, ginsenoside Rh2, inhibits autophagy and reduces β-catenin levels in HCC cells in a dose-dependent manner [102]. A modified form, 20(S)-PPD, induces apoptosis by suppressing the PI3K/Akt signaling pathway, further inhibiting HCC proliferation [100, 103]. Additionally, ginsenoside Rg3 has been shown to reduce long non-coding RNA (lncRNA) expression and inhibit the PI3K/Akt pathway in vitro studies, resulting in reduced HCC cell migration [104].
Glycyrrhiza glabra (liquorice), a medicinal plant from the Leguminosae family, is widely used in Ayurvedic medicine and primarily found in Central Asia and China [105]. Its root extract contains glycyrrhizin, a sweet-tasting tetracyclic triterpenoid saponin, commonly used as a flavouring agent in food and medicine. Liquorice is rich in bioactive compounds, including glycyrrhizin, glycyrrhetinic acid (GA), liquiritigenin, isoliquiritigenin, licochalcone A, licopyrano-coumarin, and glabrocoumarin. Among these, glycyrrhizin and GA have shown significant anticancer and antimetastatic potential. Glycyrrhizin is metabolized into GA by gut microbiota, which is responsible for its therapeutic effects [106]. GA (also known as enoxolone) exhibits anti-inflammatory, anticancer, and pro-apoptotic activities. It suppresses HCC proliferation by inhibiting the JNK1 pathway, which is associated with malignant transformation, and reduces the self-renewal capacity of HSCs [107]. In combination with sorafenib, GA enhances anticancer efficacy. It also inhibits cell migration and metastasis by targeting the EGFR, ERK, and Akt signaling pathways [108]. GA binds specifically to a receptor on the sinusoidal surface of hepatocytes (GA receptor), enabling its use in targeted drug delivery. For instance, GA has been conjugated with 5-fluorouracil (5-FU) via an alkyl side chain, resulting in increased ROS (reactive oxygen species) production and enhanced apoptosis [109]. Additionally, licochalcone A, a compound from Glycyrrhiza inflata, shows synergistic effects with sorafenib by blocking the MKK-4/JNK signaling pathway, thereby inhibiting HCC metastasis [76].
Picrorhiza kurroa (kutki) is a medicinal plant native to high-altitude regions (3,000–5,500 meters) of the Himalayas, India, Pakistan, Nepal, and Tibet. Belonging to the Plantaginaceae family, it includes two species: P. kurroa Royle ex Benth and P. scrophulariiflora Pennell [110, 111]. Valued in Ayurvedic medicine, the plant’s roots and rhizomes produce two primary crystalline compounds—picroline and kutkin—which contain various bioactive phytochemicals, including glycosides, iridoids, alkaloids, and terpenes. Among these, iridoids are the major class, featuring compounds such as picroside I–V, verminoside, catalpol, veronicoside, specioside, 6-feruloylcatalpol, pikuroside, and aucubin [112]. Picroside II has demonstrated antimetastatic and anti-angiogenic effects in both cellular and animal models [113]. It reduces inflammation by lowering TNF-α, IL-1β, and IL-6 levels and inhibiting NF-κB signaling in rat lung tissue [114]. In HCC models, picroside II targets glycosylphosphatidylinositol (GPI)-anchored signaling, suppressing cell proliferation and migration. However, further studies are needed to fully understand its role in inhibiting tumor cell migration [70].
Silymarin is a bioactive extract derived from Milk thistle (Silybum marianum), a medicinal plant known for its distinctive milky-white-veined leaves. It remains stable in acidic environments but degrades in alkaline conditions, making it effective in the acidic microenvironment of tumors. Silymarin comprises several active compounds, including silybin (or silibinin), isosilibinin, silydianin, silychristin, isosilychristin, and taxifolin. Among these, silybin is the most prominent for its anti-cancer properties, demonstrating antioxidant, anti-inflammatory, anti-proliferative, pro-apoptotic, antimetastatic, and anti-angiogenic effects [115]. Silybin shows enhanced therapeutic efficacy when combined with other agents. For example, in combination with doxorubicin, it induces cell cycle arrest at the G2-M phase by regulating the cdc25C-cyclin B1-cdc2 pathway and promotes apoptosis [116]. When paired with sorafenib, silibinin effectively suppresses the self-renewal capacity of cancer stem cells by downregulating stemness-related markers such as Nanog and Klf4 [117]. Additionally, silybin inhibits tumor progression in HCC by downregulating MMPs and modulating key signaling pathways, including the ERK1/2 cascade, Slit-2/Robo-1, and by suppressing PD-L1 expression, thereby enhancing T-cell activation [118, 119].
Garlic (Allium sativum L.), a member of the Liliaceae family, is a herbaceous plant widely cultivated in India, Central Asia, and surrounding regions. Known for its pungent aroma and flavor, garlic is rich in sulfur-containing phytochemicals that contribute to its extensive nutritional, physiological, and medicinal benefits [120, 121]. Approximately 82% of garlic’s sulfur compounds include allicin, diallyl sulfide (DAS), diallyl trisulfide (DATS), E-/Z-ajoene, and S-allyl cysteine (SAC) sulfoxide. It also contains other organosulfur compounds such as SAC, N-acetylcysteine, and S-allyl mercapto cysteine (SAMC) [121, 122]. Among these, allicin (diallyl thiosulfinate) is the principal bioactive component with significant anticancer activity [123].
Although naturally derived from garlic, allicin is liposoluble and unstable post-synthesis, rapidly converting in vitro into secondary metabolites such as DADS (diallyl disulfide), DATS, SAC, SAMC, and γ-glutamyl-SAC (GSAC), which can also be synthesized artificially [122, 124]. Studies show that allicin induces apoptosis via the p38/MAPK signaling pathway in gastric carcinoma cells [125] and through p53-mediated autophagy in Hep3B cells [85]. Additionally, allicin suppresses telomerase activity, contributing to cancer cell apoptosis and senescence [126, 127]. It also inhibits the NF-κB pathway and modulates inflammatory cytokines by promoting pro-inflammatory signaling and inducing autophagy through TAMs in hepatoma cells [128–130].
Allicin exhibits antimetastatic properties across various cancers. In gastric cancer, it inhibits cell migration by upregulating miR-383-5p and blocking ERBB4/PI3K/Akt signaling, while in cholangiocarcinoma, it suppresses MMP-2 and MMP-9 activity [131, 132]. In breast cancer, it enhances doxorubicin sensitivity by inhibiting the Nrf2/HO-1 pathway and promoting apoptosis [133, 134]. DATS, another allicin metabolite, also shows anticancer activity in HCC. In HepG2 cells, DATS induces apoptosis by activating the AMPK/SIRT1 pathway [135] and disrupts the ubiquitination of APPL1 via inhibition of TRAF6-mediated K63-linked polyubiquitination, leading to the inactivation of STAT3, Akt, and ERK1/2 pathways and suppression of tumor progression [136]. However, these findings are based on in vitro models, and further in vivo studies are necessary to validate DATS’s therapeutic potential in HCC.
Azadirachta indica, commonly known as neem or “The Village Pharmacy”, belongs to the Meliaceae family. Valued in modern medicine as well as traditional systems such as Unani, Ayurveda, and Homeopathy, neem is recognized for its wide range of therapeutic properties. Its bioactive compounds fall into two major categories: isoprenoids and non-isoprenoids. Key constituents include azadirachtin, nimbolide, gedunin, nimbin, nimbidin, quercetin, and limonoids. These compounds contribute to disease prevention by reducing inflammation, inducing apoptosis, inhibiting angiogenesis, modulating immune responses, and providing antioxidant effects. Notably, neem exhibits significant anticancer activity by altering cancer cell behavior, making it a promising agent for cancer therapy [137].
Nimbolide, a potent limonoid derived from neem flowers and leaves, possesses diverse pharmacological properties including antimalarial, antibacterial, antiviral, antioxidant, anti-inflammatory, anti-invasive, neuroprotective, hepatoprotective, and pro-apoptotic effects. It enhances antioxidant defence by increasing the activity of enzymes such as glutathione (GSH) peroxidase, catalase, and superoxide dismutase (SOD) [138, 139]. Nimbolide also regulates lipid metabolism by modulating genes like liver X receptor-α (LXR-α), peroxisome proliferator-activated receptor-γ (PPARγ), and sterol regulatory element-binding protein-1c (SREBP1c) in hepatocytes [140]. In autoimmune hepatitis, nimbolide exerts anti-inflammatory effects by targeting histone deacetylase 3 (HDAC3), thereby reducing the expression of proinflammatory cytokines such as IL-1β, IL-6, and TNF-α [141]. In HCC, nimbolide enhances tight junction integrity by upregulating proteins such as ZO-1 and occludin, thereby preserving membrane polarity and reducing cell migration. Our previous study confirmed these effects, along with its anti-inflammatory properties [142]. However, further investigation is needed to fully elucidate nimbolide’s role in metastasis prevention in HCC.
Furthermore, in prostate cancer, nimbolide downregulates metastasis-associated genes like MMP-9 and ICAM1, reducing cell migration and invasion [143]. In oral cancer, it induces autophagy-dependent apoptosis by inhibiting the PI3K/Akt signaling pathway [144].
Nimbolide also reverses EMT markers in triple-negative breast cancer (TNBC) cells and inhibits the integrin-FAK (ITG-FAK) signaling pathway, affecting actin cytoskeleton remodeling and promoting apoptosis [145]. In pancreatic cancer, combining nimbolide with docetaxel (DTX) enhances DTX sensitivity, suppressing tumor growth and metastasis through inhibition of NF-κB signaling and promotion of apoptosis [146, 147].
Numerous medicinal plants exhibit promising antimetastatic effects in HCC through well-defined molecular mechanisms. Curcumin longa inhibits PI3K/Akt/mTOR/NF-κB pathway and downregulates MMPs-2/9 and VEGF, thereby impeding angiogenesis and invasion. Andrographis paniculata suppresses tumor progression by modulating HMGB1 and MMP-9 expression via upregulation of miR-22-3p. Camptotheca acuminata affects EMT markers by downregulating Nrf2 and N-cadherin while upregulating E-cadherin. Silybum marianum activates the AMPK-DR5 pathway, inhibiting glycolysis and inducing apoptosis. Collectively, these phytochemicals target critical oncogenic processes.
Herbal medicine is one of the oldest forms of therapeutic intervention and has significantly influenced modern pharmaceutical development. Many medicinal plants, including those used in traditional Indian healthcare systems like Ayurveda, have been historically employed to treat various diseases. However, several challenges limit the effective use of herbal remedies in treating complex diseases such as cancer, including HCC.
A major limitation is the lack of sufficient clinical studies to validate the efficacy of plant-based anticancer agents, particularly in preventing cancer metastasis. Additionally, poor systemic absorption—such as low gastrointestinal uptake—significantly reduces the therapeutic potency of compounds like curcumin in clinical trials. Other contributing factors to low bioavailability include rapid metabolism and fast systemic elimination [99]. While herbal medicines generally have fewer side effects compared to synthetic drugs, their therapeutic application is hindered by the difficulty in identifying and standardizing active compounds due to the complex mixture of constituents in plant extracts. More research is necessary to elucidate the molecular mechanisms of these active ingredients and improve their pharmacokinetic properties [148].
This comprehensive review highlights the potential of plant-based bioactive compounds in targeting HCC through diverse cellular mechanisms. Natural agents such as nimbolide, curcumin, crocetin, allicin, picroside II, and ginsenosides have shown promising anti-cancer effects in both in vitro and in vivo studies. Notably, many of these compounds are commonly found in dietary sources, including spices and fruits. Preclinical and clinical findings suggest that these phytochemicals can enhance the efficacy of conventional anti-cancer therapies while reducing their side effects, offering promising alternative strategies for controlling HCC progression and metastasis. Further exploration of their molecular pathways may pave the way for the development of more effective and less toxic cancer treatments.
BCLC: Barcelona Clinic Liver Cancer
DATS: diallyl trisulfide
DTX: docetaxel
ECM: extracellular matrix
EMT: epithelial-mesenchymal transition
GA: glycyrrhetinic acid
HBV: hepatitis B virus
HCC: hepatocellular carcinoma
HIF-1α: hypoxia inducible factor-1α
HSCs: hepatic stellate cells
LT: liver transplantation
MAFLD: metabolic dysfunction-associated fatty liver disease
MMPs: matrix metalloproteinases
MWA: microwave ablation
PD-L1: programmed death-ligand 1
PDGF: platelet-derived growth factor
PPD: protopanaxadiol
RFA: radiofrequency ablation
SAC: S-allyl cysteine
SAMC: S-allyl mercapto cysteine
SIRT: selective internal radiation therapy
TACE: transarterial chemoembolization
TAMs: tumor-associated macrophages
TGF-β1: transforming growth factor-β1
TME: tumor microenvironment
VEGFR: vascular endothelial growth factor receptor
We also extend our utmost gratitude to Dr. Dinesh Kumar Meena, Research Scientist-II, St John’s Research Institute, Bangalore, for his invaluable insights and feedback on this manuscript.
MKN: Writing—original draft. DS and BV: Writing—review & editing.
The authors declare that there are no conflicts of interest.
This article is a review of previously published studies and does not involve human participants, animal subjects, or the collection of primary data; therefore, ethical approval was not required.
Not applicable.
Not applicable.
This article is a review of previously published studies, and no new data were generated or analyzed in the course of this study. All data supporting the findings of this article are available in the cited references.
This work was supported by the JIPMER intramural research grant [JIP/Res/Intramural/Phase-4/2023-2024]. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2025.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2025. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 4862
Download: 60
Times Cited: 0
