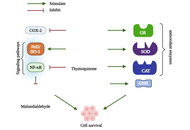






Thymoquinone (TQ), the main bioactive constituent of Nigella sativa, has gained great attention for its neuroprotective properties, especially for Alzheimer’s disease (AD), which is a progressive neurodegenerative disorder with limited therapeutic options. This review provides several experimental evidence on the effects of TQ in AD models. The evidences indicate that TQ reduces the amyloid-β accumulation, reduces the oxidative stress and neuroinflammation, and improves cognitive and behavioral outcomes. Additionally, TQ should be able to promote the neuronal survival and neurogenesis while reducing biological markers that indicate brain damage or neuron loss. Although these findings clearly highlight and show the promising therapeutic potential of the TQ molecule in the AD, it is important to note that further in-depth studies are still needed to fully understand its underlying molecular mechanisms and to determine its clinical relevance in patients.
Thymoquinone (TQ), the main bioactive constituent of Nigella sativa, has gained great attention for its neuroprotective properties, especially for Alzheimer’s disease (AD), which is a progressive neurodegenerative disorder with limited therapeutic options. This review provides several experimental evidence on the effects of TQ in AD models. The evidences indicate that TQ reduces the amyloid-β accumulation, reduces the oxidative stress and neuroinflammation, and improves cognitive and behavioral outcomes. Additionally, TQ should be able to promote the neuronal survival and neurogenesis while reducing biological markers that indicate brain damage or neuron loss. Although these findings clearly highlight and show the promising therapeutic potential of the TQ molecule in the AD, it is important to note that further in-depth studies are still needed to fully understand its underlying molecular mechanisms and to determine its clinical relevance in patients.
DOI: https://doi.org/10.37349/en.2026.1006137
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection (Vol II)

For this review paper, data on protein misfolding and aggregation in progressive myoclonus epilepsies and some developmental encephalopathies are gathered. There is evidence that in some cases of monogenic epilepsies, misfolding of the mutated protein takes place, often leading to protein aggregation. On one hand, protein aggregation reduces the amount of protein and its activity; on the other, it exerts generic toxicity to neurons. Understanding the molecular causes due to loss of normal function and gain of toxic function of the mutated aggregate-prone proteins is important to obtain new therapies. By observing the symptomatology of progressive and developmental epileptic syndromes, one can derive some conclusions about the relevance of protein misfolding and aggregation in the picture. A plausible view seems that the most severe symptoms of dementia, behavioral and psychiatric symptoms, are linked to protein aggregation and downstream effects on cellular degradation and energy systems. Finally, I discuss the potential of targeting the proteostasis network to develop novel anti-seizure and neuroprotective therapies.
For this review paper, data on protein misfolding and aggregation in progressive myoclonus epilepsies and some developmental encephalopathies are gathered. There is evidence that in some cases of monogenic epilepsies, misfolding of the mutated protein takes place, often leading to protein aggregation. On one hand, protein aggregation reduces the amount of protein and its activity; on the other, it exerts generic toxicity to neurons. Understanding the molecular causes due to loss of normal function and gain of toxic function of the mutated aggregate-prone proteins is important to obtain new therapies. By observing the symptomatology of progressive and developmental epileptic syndromes, one can derive some conclusions about the relevance of protein misfolding and aggregation in the picture. A plausible view seems that the most severe symptoms of dementia, behavioral and psychiatric symptoms, are linked to protein aggregation and downstream effects on cellular degradation and energy systems. Finally, I discuss the potential of targeting the proteostasis network to develop novel anti-seizure and neuroprotective therapies.
DOI: https://doi.org/10.37349/en.2026.1006136
This article belongs to the special issue Advances in Epilepsy Research

Aim:
Redox-oxidative dysregulation is implicated in the aetiology of several diseases, including schizophrenia, with a possible influence on clinical symptoms. This study investigated the influence of redox, lipid peroxidation, and micronutrient antioxidants on the expression of clinical phenotypes of schizophrenia.
Methods:
A total of 220 consenting drug-naïve volunteers, including 120 participants with schizophrenia and 100 apparently healthy controls, were recruited. Schizophrenia symptoms were evaluated using the Positive and Negative Syndrome Scale (PANSS). Lipid peroxidation (malondialdehyde; MDA) was quantified using the thiobarbituric acid reactive substances (TBARS) spectrophotometric method; glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT) were assessed using established enzymatic activity assays; total antioxidant capacity (TAC) was determined by the phosphomolybdenum colorimetric method; vitamins C and E were measured using spectrophotometric biochemical assays; and zinc (Zn) and selenium (Se) concentrations were quantified using atomic absorption spectrophotometry (AAS).
Results:
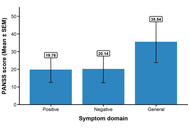
Enzymatic antioxidants, SOD (19.58 ± 0.80; 10.12 ± 0.45 U/L) and CAT (41.73 ± 1.81; 21.33 ± 0.98 U/L), increased in schizophrenia compared with controls (p < 0.05), but decreased non-enzymatic antioxidants; GSH (14.5 ± 0.28; 15.9 ± 1.59 µmol/L, p < 0.05). Furthermore, serum levels of zinc (1.8 ± 0.01; 2.7 ± 0.02 mg/L), selenium (0.08 ± 0.01; 0.10 ± 0.01 mg/L), and vitamin C (12.98 ± 0.49; 15.08 ± 0.37 mg/L) were lowered in schizophrenia compared with controls (p < 0.05). GSH had a negative correlation with positive symptoms (r = –0.285, p = 0.013) while SOD (r = 0.281, p = 0.001) and CAT (r = 0.179, p = 0.034) correlated positively with MDA (p < 0.05). In contrast, GSH (r = –0.247, p = 0.003) and TAC (r = –0.221, p = 0.009) correlated negatively with MDA (p < 0.05).
Conclusions:
Drug-naïve Nigerian individuals with schizophrenia appear to exhibit a pattern of redox imbalance, including increased lipid peroxidation, altered antioxidant enzyme activity, and reduced non-enzymatic antioxidants, with lower GSH levels modestly associated with greater positive symptom severity.
Aim:
Redox-oxidative dysregulation is implicated in the aetiology of several diseases, including schizophrenia, with a possible influence on clinical symptoms. This study investigated the influence of redox, lipid peroxidation, and micronutrient antioxidants on the expression of clinical phenotypes of schizophrenia.
Methods:
A total of 220 consenting drug-naïve volunteers, including 120 participants with schizophrenia and 100 apparently healthy controls, were recruited. Schizophrenia symptoms were evaluated using the Positive and Negative Syndrome Scale (PANSS). Lipid peroxidation (malondialdehyde; MDA) was quantified using the thiobarbituric acid reactive substances (TBARS) spectrophotometric method; glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT) were assessed using established enzymatic activity assays; total antioxidant capacity (TAC) was determined by the phosphomolybdenum colorimetric method; vitamins C and E were measured using spectrophotometric biochemical assays; and zinc (Zn) and selenium (Se) concentrations were quantified using atomic absorption spectrophotometry (AAS).
Results:
Enzymatic antioxidants, SOD (19.58 ± 0.80; 10.12 ± 0.45 U/L) and CAT (41.73 ± 1.81; 21.33 ± 0.98 U/L), increased in schizophrenia compared with controls (p < 0.05), but decreased non-enzymatic antioxidants; GSH (14.5 ± 0.28; 15.9 ± 1.59 µmol/L, p < 0.05). Furthermore, serum levels of zinc (1.8 ± 0.01; 2.7 ± 0.02 mg/L), selenium (0.08 ± 0.01; 0.10 ± 0.01 mg/L), and vitamin C (12.98 ± 0.49; 15.08 ± 0.37 mg/L) were lowered in schizophrenia compared with controls (p < 0.05). GSH had a negative correlation with positive symptoms (r = –0.285, p = 0.013) while SOD (r = 0.281, p = 0.001) and CAT (r = 0.179, p = 0.034) correlated positively with MDA (p < 0.05). In contrast, GSH (r = –0.247, p = 0.003) and TAC (r = –0.221, p = 0.009) correlated negatively with MDA (p < 0.05).
Conclusions:
Drug-naïve Nigerian individuals with schizophrenia appear to exhibit a pattern of redox imbalance, including increased lipid peroxidation, altered antioxidant enzyme activity, and reduced non-enzymatic antioxidants, with lower GSH levels modestly associated with greater positive symptom severity.
DOI: https://doi.org/10.37349/en.2026.1006134

Aim:
Stroke represents a leading cause of mortality and disability globally, yet comprehensive epidemiological analyses for Saudi Arabia remain limited. This study aimed to analyze temporal trends in stroke incidence, mortality, and disability-adjusted life years (DALYs) in Saudi Arabia from 1990 to 2021, examine stroke subtype distributions and risk factor attributions, and compare findings with regional and international benchmarks.
Methods:
We conducted a comprehensive analysis of the Global Burden of Disease (GBD) 2021 study data for Saudi Arabia, supplemented by systematic review findings and hospital-based registry data. Age-standardized incidence rates (ASIRs), mortality rates (ASMRs), and DALY rates were extracted for ischemic stroke, intracerebral hemorrhage, and subarachnoid hemorrhage. Annual percent change (APC) was calculated using Joinpoint regression. Risk factor attribution was analyzed using comparative risk assessment methodology. Poisson regression models examined sex differences.
Results:
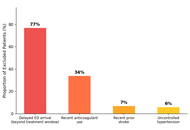
The pooled annual stroke incidence in Saudi Arabia was 29 per 100,000 population (95% CI: 15–47). Ischemic stroke predominated, comprising 79–87% of cases. Age-standardized DALYs showed significant yearly decreases of 9.28 per 100,000 (95% CI: 6.31–12.26, p < 0.001). The age-standardized death rate in the Middle East and North Africa (MENA) region was 87.7 per 100,000 [95% uncertainty interval (UI): 78.2–97.6] in 2019, representing a 27.8% regional decrease from 1990. Mean age at first stroke in Saudi Arabia was 63 years—six years younger than Western populations. Hypertension (57.7%), diabetes mellitus (49.4%), and obesity (42.0%) were the major modifiable risk factors. Intravenous thrombolysis utilization remained critically low at 1–3.6%.
Conclusions:
Despite declining mortality and DALYs, Saudi Arabia faces a substantial stroke burden characterized by a younger onset age and significant treatment gaps. Achieving Vision 2030 health targets requires accelerated primary prevention addressing metabolic risk factors and expansion of acute stroke treatment capacity nationwide.
Aim:
Stroke represents a leading cause of mortality and disability globally, yet comprehensive epidemiological analyses for Saudi Arabia remain limited. This study aimed to analyze temporal trends in stroke incidence, mortality, and disability-adjusted life years (DALYs) in Saudi Arabia from 1990 to 2021, examine stroke subtype distributions and risk factor attributions, and compare findings with regional and international benchmarks.
Methods:
We conducted a comprehensive analysis of the Global Burden of Disease (GBD) 2021 study data for Saudi Arabia, supplemented by systematic review findings and hospital-based registry data. Age-standardized incidence rates (ASIRs), mortality rates (ASMRs), and DALY rates were extracted for ischemic stroke, intracerebral hemorrhage, and subarachnoid hemorrhage. Annual percent change (APC) was calculated using Joinpoint regression. Risk factor attribution was analyzed using comparative risk assessment methodology. Poisson regression models examined sex differences.
Results:
The pooled annual stroke incidence in Saudi Arabia was 29 per 100,000 population (95% CI: 15–47). Ischemic stroke predominated, comprising 79–87% of cases. Age-standardized DALYs showed significant yearly decreases of 9.28 per 100,000 (95% CI: 6.31–12.26, p < 0.001). The age-standardized death rate in the Middle East and North Africa (MENA) region was 87.7 per 100,000 [95% uncertainty interval (UI): 78.2–97.6] in 2019, representing a 27.8% regional decrease from 1990. Mean age at first stroke in Saudi Arabia was 63 years—six years younger than Western populations. Hypertension (57.7%), diabetes mellitus (49.4%), and obesity (42.0%) were the major modifiable risk factors. Intravenous thrombolysis utilization remained critically low at 1–3.6%.
Conclusions:
Despite declining mortality and DALYs, Saudi Arabia faces a substantial stroke burden characterized by a younger onset age and significant treatment gaps. Achieving Vision 2030 health targets requires accelerated primary prevention addressing metabolic risk factors and expansion of acute stroke treatment capacity nationwide.
DOI: https://doi.org/10.37349/en.2026.1006135
This article belongs to the special issue The Science of Ischemic Stroke

Major depressive disorder (MDD) is increasingly understood as a multifactorial psychiatric disorder involving interacting neural, immune, metabolic, and microbial processes. Within this framework, the microbiota–gut–brain axis and mitochondrial bioenergetics have emerged as potentially intersecting contributors to depressive symptomatology. Preclinical studies suggest that microbial metabolites—especially short-chain fatty acids (SCFAs)—can influence oxidative phosphorylation, redox balance, neuroinflammation, and synaptic plasticity, whereas inflammatory signals such as lipopolysaccharide may disrupt mitochondrial dynamics. However, the strength of evidence is uneven: mechanistic support is strongest in cell and animal models, whereas human data remain heterogeneous and largely associative. This narrative review critically synthesizes current evidence on microbiota–mitochondria crosstalk in MDD, distinguishing established findings from emerging hypotheses. It also examines recent psychobiotic trials, metabolomic and biomarker studies, and microglia–mitochondria mechanisms, and discusses the translational limitations that currently constrain clinical application. Overall, this axis represents a plausible and clinically relevant framework for hypothesis generation and adjunctive intervention development, but it should not yet be regarded as a fully validated causal pathway or stand-alone therapeutic target in MDD.
Major depressive disorder (MDD) is increasingly understood as a multifactorial psychiatric disorder involving interacting neural, immune, metabolic, and microbial processes. Within this framework, the microbiota–gut–brain axis and mitochondrial bioenergetics have emerged as potentially intersecting contributors to depressive symptomatology. Preclinical studies suggest that microbial metabolites—especially short-chain fatty acids (SCFAs)—can influence oxidative phosphorylation, redox balance, neuroinflammation, and synaptic plasticity, whereas inflammatory signals such as lipopolysaccharide may disrupt mitochondrial dynamics. However, the strength of evidence is uneven: mechanistic support is strongest in cell and animal models, whereas human data remain heterogeneous and largely associative. This narrative review critically synthesizes current evidence on microbiota–mitochondria crosstalk in MDD, distinguishing established findings from emerging hypotheses. It also examines recent psychobiotic trials, metabolomic and biomarker studies, and microglia–mitochondria mechanisms, and discusses the translational limitations that currently constrain clinical application. Overall, this axis represents a plausible and clinically relevant framework for hypothesis generation and adjunctive intervention development, but it should not yet be regarded as a fully validated causal pathway or stand-alone therapeutic target in MDD.
DOI: https://doi.org/10.37349/en.2026.1006133
This article belongs to the special issue Depression: From Pathophysiology to Treatment Innovation

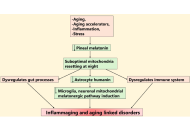
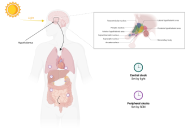
There is a growing appreciation of the role of mitochondria in determining the interactions of CNS astrocytes, microglia, and neurons. The influence of circadian and systemic processes in regulating these interactions is relatively underexplored. Recent work has indicated the importance of night-time dampening and resetting in the pathoetiology of a diverse array of aging-associated medical conditions, including neurodegenerative disorders. The 10-fold decrease in pineal melatonin at night between childhood and the 9th decade of life is a major determinant of how aging associates with neurodegenerative disorders, cardiovascular disorders, and a wide range of tumors. It is proposed that the beneficial effects of pineal melatonin are mediated via its upregulation of the mitochondria-derived peptides (MDPs), including humanin. Although potentially induced in all mitochondria-containing cells, humanin is primarily produced in the CNS by astrocytes. The capacity of pineal melatonin to increase astrocyte humanin leads to the induction of the local melatonergic pathway in microglia to shift microglia from a pro-inflammatory M1-like to prophagocytic M2-like phenotype. In neurons, astrocyte-derived humanin optimizes mitochondrial function and decreases oxidant production to increase function and survival, possibly also involving mitochondrial melatonergic pathway upregulation. Concurrent effects of pineal melatonin in decreasing gut dysbiosis/permeability and stimulating oxytocin to activate the vagal nerve contribute to more optimized dampening and resetting that influences CNS interactions of glia and neurons. Overall, the conceptualizations of how astrocyte, microglial, and neuronal mitochondria interact require integration with wider circadian and systemic processes. A plethora of novel research implications are highlighted.
There is a growing appreciation of the role of mitochondria in determining the interactions of CNS astrocytes, microglia, and neurons. The influence of circadian and systemic processes in regulating these interactions is relatively underexplored. Recent work has indicated the importance of night-time dampening and resetting in the pathoetiology of a diverse array of aging-associated medical conditions, including neurodegenerative disorders. The 10-fold decrease in pineal melatonin at night between childhood and the 9th decade of life is a major determinant of how aging associates with neurodegenerative disorders, cardiovascular disorders, and a wide range of tumors. It is proposed that the beneficial effects of pineal melatonin are mediated via its upregulation of the mitochondria-derived peptides (MDPs), including humanin. Although potentially induced in all mitochondria-containing cells, humanin is primarily produced in the CNS by astrocytes. The capacity of pineal melatonin to increase astrocyte humanin leads to the induction of the local melatonergic pathway in microglia to shift microglia from a pro-inflammatory M1-like to prophagocytic M2-like phenotype. In neurons, astrocyte-derived humanin optimizes mitochondrial function and decreases oxidant production to increase function and survival, possibly also involving mitochondrial melatonergic pathway upregulation. Concurrent effects of pineal melatonin in decreasing gut dysbiosis/permeability and stimulating oxytocin to activate the vagal nerve contribute to more optimized dampening and resetting that influences CNS interactions of glia and neurons. Overall, the conceptualizations of how astrocyte, microglial, and neuronal mitochondria interact require integration with wider circadian and systemic processes. A plethora of novel research implications are highlighted.
DOI: https://doi.org/10.37349/en.2026.1006132

Human behavior depends on a collection of cognitive capacities that are expressed with complexity in humans. Although animal models have been essential for identifying fundamental neural mechanisms, many aspects of human cognition require direct investigation in the human brain. Studies of social decision-making, communication, and spatial navigation increasingly rely on intracranial electrophysiology to probe the neural basis. Related to these topics, reward processing warrants emphasis. It is not uniquely human, but it provides a central organizing signal linking motivation, learning, emotion, and choice across many human behaviors. Disruptions of reward circuits are a hallmark of numerous neurological and psychiatric conditions, giving this domain specific relevance for patient care. Fifteen studies published between 2009 and 2024 used human intracranial recordings to examine reward-related processes, nearly all in patients undergoing invasive monitoring for drug-resistant epilepsy. These studies investigated 17 neocortical and subcortical regions, most frequently the orbitofrontal cortex, using intracranial EEG, deep brain stimulation, and single-unit recordings. Recent work increasingly incorporates social interactions and computational models of learning. The purpose of this narrative review is to provide an overview of human reward processing, emphasizing how intracranial recordings have clarified the neural circuits that underlie a range of human cognitive capacities. Beyond advancing basic neuroscience, intracranial electrophysiology can inform circuit-guided interventions for neurological and psychiatric disorders.
Human behavior depends on a collection of cognitive capacities that are expressed with complexity in humans. Although animal models have been essential for identifying fundamental neural mechanisms, many aspects of human cognition require direct investigation in the human brain. Studies of social decision-making, communication, and spatial navigation increasingly rely on intracranial electrophysiology to probe the neural basis. Related to these topics, reward processing warrants emphasis. It is not uniquely human, but it provides a central organizing signal linking motivation, learning, emotion, and choice across many human behaviors. Disruptions of reward circuits are a hallmark of numerous neurological and psychiatric conditions, giving this domain specific relevance for patient care. Fifteen studies published between 2009 and 2024 used human intracranial recordings to examine reward-related processes, nearly all in patients undergoing invasive monitoring for drug-resistant epilepsy. These studies investigated 17 neocortical and subcortical regions, most frequently the orbitofrontal cortex, using intracranial EEG, deep brain stimulation, and single-unit recordings. Recent work increasingly incorporates social interactions and computational models of learning. The purpose of this narrative review is to provide an overview of human reward processing, emphasizing how intracranial recordings have clarified the neural circuits that underlie a range of human cognitive capacities. Beyond advancing basic neuroscience, intracranial electrophysiology can inform circuit-guided interventions for neurological and psychiatric disorders.
DOI: https://doi.org/10.37349/en.2026.1006131
This article belongs to the special issue Advances in Epilepsy Research

Malignant tumors of the nervous system, such as gliomas, medulloblastomas, and neuroblastomas, pose a greater clinical challenge due to their aggressive and invasive nature and their resistance to current treatment options. The blood-brain barrier (BBB) impairs the delivery of therapeutic agents, which is associated with poor prognosis. The natural flavonoid fisetin has demonstrated potential for cancer treatment by regulating major cancer-related signaling pathways, including PI3K/Akt/mTOR, NF-κB, and MAPK. Preclinical studies suggest that fisetin induces apoptosis, suppresses tumor invasion, and reduces malignancy in glioma, medulloblastoma, and neuroblastoma models. However, evidence for fisetin’s effectiveness remains preclinical and in vitro, with no clinical trials in humans to date. One solution to this challenge is to use nanotechnology-based delivery systems to increase fisetin’s stability and solubility and facilitate its crossing of the BBB, thereby enhancing its therapeutic efficacy. Such advancements have made fisetin a promising option for neuro-oncology treatment. Further clinical trials are needed to assess the safety, efficacy, and effectiveness of fisetin in combination therapies. When conjugated with nanotechnology-based delivery, fisetin may enable a digital transformation in treatment outcomes for patients with malignant nervous system tumors.
Malignant tumors of the nervous system, such as gliomas, medulloblastomas, and neuroblastomas, pose a greater clinical challenge due to their aggressive and invasive nature and their resistance to current treatment options. The blood-brain barrier (BBB) impairs the delivery of therapeutic agents, which is associated with poor prognosis. The natural flavonoid fisetin has demonstrated potential for cancer treatment by regulating major cancer-related signaling pathways, including PI3K/Akt/mTOR, NF-κB, and MAPK. Preclinical studies suggest that fisetin induces apoptosis, suppresses tumor invasion, and reduces malignancy in glioma, medulloblastoma, and neuroblastoma models. However, evidence for fisetin’s effectiveness remains preclinical and in vitro, with no clinical trials in humans to date. One solution to this challenge is to use nanotechnology-based delivery systems to increase fisetin’s stability and solubility and facilitate its crossing of the BBB, thereby enhancing its therapeutic efficacy. Such advancements have made fisetin a promising option for neuro-oncology treatment. Further clinical trials are needed to assess the safety, efficacy, and effectiveness of fisetin in combination therapies. When conjugated with nanotechnology-based delivery, fisetin may enable a digital transformation in treatment outcomes for patients with malignant nervous system tumors.
DOI: https://doi.org/10.37349/en.2026.1006130
This article belongs to the special issue Current Approaches to Malignant Tumors of the Nervous System

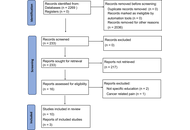
Chronic pain, defined as pain persisting beyond 12 weeks, is known to be challenging to manage. Despite increasing opioid prescribing, pain is still an issue for many patients. Patient education has emerged as a key element in supporting self-management and improving pain-related outcomes. This narrative review explores the impact of patient education alongside pain management interventions, with a focus on pain intensity, pain-related beliefs, and pain-related disability. A structured search using PubMed, Medline, EBSCOhost, and UCL Explore identified ten studies (six RCTs, two systematic reviews, two cohort studies) and three national guidelines meeting the inclusion criteria. Inclusion criteria focused on adult patients with chronic non-malignant pain who received some form of education alongside or prior to pain interventions. Pain education was associated with 29% reduction in opioid use, with 7% patients discontinuing opioids entirely in the usual group. Improvements in emotional functioning (41%), pain understanding (75%), and overall functioning (38%) were observed across various trials. Existing research shows promising results; however, the clarity of the type of education needed needs to be established. Short, focused education sessions, especially those incorporating pain neuroscience education (PNE) or cognitive behavioural therapy (CBT) elements, were as effective as longer programs. Further research is required to identify how education improves the outcome of pain management interventions. Integrating targeted patient education into the chronic pain care pathway can significantly reduce disability, improve quality of life, and decrease opioid reliance. These findings support implementing structured education sessions as part of routine pain management services to enhance long-term patient outcomes.
Chronic pain, defined as pain persisting beyond 12 weeks, is known to be challenging to manage. Despite increasing opioid prescribing, pain is still an issue for many patients. Patient education has emerged as a key element in supporting self-management and improving pain-related outcomes. This narrative review explores the impact of patient education alongside pain management interventions, with a focus on pain intensity, pain-related beliefs, and pain-related disability. A structured search using PubMed, Medline, EBSCOhost, and UCL Explore identified ten studies (six RCTs, two systematic reviews, two cohort studies) and three national guidelines meeting the inclusion criteria. Inclusion criteria focused on adult patients with chronic non-malignant pain who received some form of education alongside or prior to pain interventions. Pain education was associated with 29% reduction in opioid use, with 7% patients discontinuing opioids entirely in the usual group. Improvements in emotional functioning (41%), pain understanding (75%), and overall functioning (38%) were observed across various trials. Existing research shows promising results; however, the clarity of the type of education needed needs to be established. Short, focused education sessions, especially those incorporating pain neuroscience education (PNE) or cognitive behavioural therapy (CBT) elements, were as effective as longer programs. Further research is required to identify how education improves the outcome of pain management interventions. Integrating targeted patient education into the chronic pain care pathway can significantly reduce disability, improve quality of life, and decrease opioid reliance. These findings support implementing structured education sessions as part of routine pain management services to enhance long-term patient outcomes.
DOI: https://doi.org/10.37349/en.2026.1006129

Adult human hippocampal neurogenesis has been debated for decades, with methodological differences producing conflicting reports. Radiocarbon birth-dating provided population-level evidence of sustained dentate gyrus neuron turnover, while immunohistochemical studies produced variable results depending on fixation protocols. Optimized post-mortem handling has reported higher detectability of immature-neuron markers across adulthood, whereas longer post-mortem delays and prolonged fixation can reduce signal and contribute to apparent null findings; however, marker-based interpretations remain debated and require cautious, multi-marker validation. Recent single-nucleus and spatial transcriptomics further support persistent neurogenesis, identifying immature granule-cell signatures and niche programs into late life. This article critically appraises evidence from radiocarbon dating, immunohistochemistry, and transcriptomics, highlighting sources of discrepancy and convergence. Practical standards for human tissue handling, antigen retrieval, and multimarker panels are proposed to minimize methodological artefacts. Collectively, convergent evidence favors low-level, lifelong neurogenesis with potential contributions to memory precision and affective regulation, albeit at lower rates than rodents. It is concluded that integrating radiocarbon baselining, optimized immunohistochemistry, and transcriptomic validation provides a robust framework for resolving the controversy and advancing translational relevance in cognition, aging, and psychiatry.
Adult human hippocampal neurogenesis has been debated for decades, with methodological differences producing conflicting reports. Radiocarbon birth-dating provided population-level evidence of sustained dentate gyrus neuron turnover, while immunohistochemical studies produced variable results depending on fixation protocols. Optimized post-mortem handling has reported higher detectability of immature-neuron markers across adulthood, whereas longer post-mortem delays and prolonged fixation can reduce signal and contribute to apparent null findings; however, marker-based interpretations remain debated and require cautious, multi-marker validation. Recent single-nucleus and spatial transcriptomics further support persistent neurogenesis, identifying immature granule-cell signatures and niche programs into late life. This article critically appraises evidence from radiocarbon dating, immunohistochemistry, and transcriptomics, highlighting sources of discrepancy and convergence. Practical standards for human tissue handling, antigen retrieval, and multimarker panels are proposed to minimize methodological artefacts. Collectively, convergent evidence favors low-level, lifelong neurogenesis with potential contributions to memory precision and affective regulation, albeit at lower rates than rodents. It is concluded that integrating radiocarbon baselining, optimized immunohistochemistry, and transcriptomic validation provides a robust framework for resolving the controversy and advancing translational relevance in cognition, aging, and psychiatry.
DOI: https://doi.org/10.37349/en.2026.1006128

The circadian clock orchestrates cellular physiology by synchronizing transcriptional, metabolic, and signaling networks with the environmental light-dark cycle. Basic helix-loop-helix ARNT-like protein 1 (BMAL1), a core transcriptional regulator of circadian timing, contributes to rhythmic gene expression and is implicated in cellular responses to stress and energy demand. Emerging evidence suggests an interplay between BMAL1 and the phosphatidylinositol 3-kinase (PI3K)/AKT pathway, a central hub controlling cell survival, metabolism, and angiogenesis. In ischemic conditions, BMAL1 is associated with increased PI3K/AKT activity and downstream mTOR signaling, which may help preserve mitochondrial integrity, limit oxidative stress, and support neuronal and vascular recovery. Conversely, BMAL1 deficiency is linked to impaired AKT phosphorylation and redox imbalance, exacerbating ischemic injury. Proteomic and functional studies further suggest that BMAL1 may contribute to metabolic reprogramming through PI3K/AKT-dependent regulation of oxidative phosphorylation and antioxidant defenses. This review is based on a focused narrative evaluation of experimental and translational studies retrieved from PubMed, emphasizing circadian regulation of PI3K/AKT signaling in ischemic and vascular contexts. Collectively, these findings support the concept that BMAL1 functions as a temporal modulator of PI3K/AKT signaling, integrating circadian and metabolic cues to promote cellular resilience. Understanding this regulatory axis may offer novel therapeutic perspectives for ischemic and neurovascular disorders associated with circadian misalignment.
The circadian clock orchestrates cellular physiology by synchronizing transcriptional, metabolic, and signaling networks with the environmental light-dark cycle. Basic helix-loop-helix ARNT-like protein 1 (BMAL1), a core transcriptional regulator of circadian timing, contributes to rhythmic gene expression and is implicated in cellular responses to stress and energy demand. Emerging evidence suggests an interplay between BMAL1 and the phosphatidylinositol 3-kinase (PI3K)/AKT pathway, a central hub controlling cell survival, metabolism, and angiogenesis. In ischemic conditions, BMAL1 is associated with increased PI3K/AKT activity and downstream mTOR signaling, which may help preserve mitochondrial integrity, limit oxidative stress, and support neuronal and vascular recovery. Conversely, BMAL1 deficiency is linked to impaired AKT phosphorylation and redox imbalance, exacerbating ischemic injury. Proteomic and functional studies further suggest that BMAL1 may contribute to metabolic reprogramming through PI3K/AKT-dependent regulation of oxidative phosphorylation and antioxidant defenses. This review is based on a focused narrative evaluation of experimental and translational studies retrieved from PubMed, emphasizing circadian regulation of PI3K/AKT signaling in ischemic and vascular contexts. Collectively, these findings support the concept that BMAL1 functions as a temporal modulator of PI3K/AKT signaling, integrating circadian and metabolic cues to promote cellular resilience. Understanding this regulatory axis may offer novel therapeutic perspectives for ischemic and neurovascular disorders associated with circadian misalignment.
DOI: https://doi.org/10.37349/en.2026.1006127

Hydrocephalus is a complex neurological disease characterized by abnormal cerebrospinal fluid (CSF) accumulation, ventricular enlargement, and progressive neurologic dysfunction. Existing therapies are predominantly surgical, with high complication rates, with high complication rates, prompting ongoing efforts to develop alternative modalities. This review integrates developments across mechanistic platforms and integrated disease models—including genetic and induced animals, patient-derived organoids, and organ-on-a-chip systems—to evaluate their ability to recapitulate CSF kinetics and subventricular zone (SVZ) biology. New therapies, including surgical enhancements, drugs, stem cell-based repair, and gene-targeted therapies, are discussed for translation potential. Ethics and regulatory frameworks, 3Rs, and validation and scalability issues are discussed critically. Finally, computational modeling and AI are introduced as ways to integrate multi-scale data and enable precision medicine. Each of these perspectives outlines a roadmap in which bioengineering, precision medicine, and ethical rigor converge to accelerate discovery and improve outcomes for patients with hydrocephalus.
Hydrocephalus is a complex neurological disease characterized by abnormal cerebrospinal fluid (CSF) accumulation, ventricular enlargement, and progressive neurologic dysfunction. Existing therapies are predominantly surgical, with high complication rates, with high complication rates, prompting ongoing efforts to develop alternative modalities. This review integrates developments across mechanistic platforms and integrated disease models—including genetic and induced animals, patient-derived organoids, and organ-on-a-chip systems—to evaluate their ability to recapitulate CSF kinetics and subventricular zone (SVZ) biology. New therapies, including surgical enhancements, drugs, stem cell-based repair, and gene-targeted therapies, are discussed for translation potential. Ethics and regulatory frameworks, 3Rs, and validation and scalability issues are discussed critically. Finally, computational modeling and AI are introduced as ways to integrate multi-scale data and enable precision medicine. Each of these perspectives outlines a roadmap in which bioengineering, precision medicine, and ethical rigor converge to accelerate discovery and improve outcomes for patients with hydrocephalus.
DOI: https://doi.org/10.37349/en.2026.1006126

DOI: https://doi.org/10.37349/en.2026.1006125

Aim:
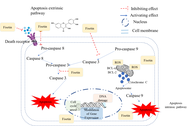
Tissue transglutaminase [transglutaminase 2 (TG2)] is implicated in central neuronal apoptosis and is expressed in the peripheral nervous system; however, its role in sensory neuron survival and neuropathic pain after nerve injury remains poorly defined. This study examined whether TG2 knockout (KO) affects dorsal root ganglion (DRG) neuron survival and pain-related behaviors following sciatic nerve injury.
Methods:
TG2 KO mice and wild-type (WT) controls underwent complete sciatic nerve transection (axotomy). Pain-related behavior was evaluated using detailed autotomy scoring over 14 days. DRG neuron survival was assessed using unbiased stereological counts.
Results:
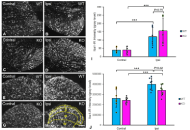
TG2 KO resulted in a distinct, previously unreported “atypical autotomy” pattern, with lesions localized mainly to the midplantar paw region. In contrast, WT mice exhibited typical autotomy directed primarily at the toes. Despite this clear difference in pain phenotype, stereological analysis revealed that TG2 KO did not alter neuronal counts in intact or axotomized DRGs, with both groups showing comparable, significant neuronal loss after injury.
Conclusions:
These findings indicate that TG2 functions as an important modulator of neuropathic pain but is not required for neuronal survival in the adult DRG following nerve injury.
Aim:
Tissue transglutaminase [transglutaminase 2 (TG2)] is implicated in central neuronal apoptosis and is expressed in the peripheral nervous system; however, its role in sensory neuron survival and neuropathic pain after nerve injury remains poorly defined. This study examined whether TG2 knockout (KO) affects dorsal root ganglion (DRG) neuron survival and pain-related behaviors following sciatic nerve injury.
Methods:
TG2 KO mice and wild-type (WT) controls underwent complete sciatic nerve transection (axotomy). Pain-related behavior was evaluated using detailed autotomy scoring over 14 days. DRG neuron survival was assessed using unbiased stereological counts.
Results:
TG2 KO resulted in a distinct, previously unreported “atypical autotomy” pattern, with lesions localized mainly to the midplantar paw region. In contrast, WT mice exhibited typical autotomy directed primarily at the toes. Despite this clear difference in pain phenotype, stereological analysis revealed that TG2 KO did not alter neuronal counts in intact or axotomized DRGs, with both groups showing comparable, significant neuronal loss after injury.
Conclusions:
These findings indicate that TG2 functions as an important modulator of neuropathic pain but is not required for neuronal survival in the adult DRG following nerve injury.
DOI: https://doi.org/10.37349/en.2026.1006124

Aim:
Male infertility resulting from neurological disorders, oxidative stress, and hormonal imbalance is a growing health concern. This study, therefore, investigated the effects of Aframomum melegueta and Aframomum danielli-supplemented diets on sperm quality and testicular oxidative damage in a scopolamine-induced rat model.
Methods:
Adult male rats were randomly allocated into seven groups: normal group; scopolamine-induced group; donepezil-treated scopolamine group and four treatment groups receiving 4% or 8% dietary supplementation of Aframomum melegueta or Aframomum danielli, respectively. Sperm motility, count, and morphology were evaluated. In addition, serum testosterone and follicle stimulating hormone levels, testicular oxidative stress markers, inflammatory cytokines, and antioxidant activities were assessed to determine reproductive and biochemical responses. High performance liquid chromatography (HPLC) profiling was also conducted to identify the major phenolic compounds in both seeds.
Results:
Scopolamine administration impaired sperm quality, decreased hormonal levels, promoted oxidative stress, and altered inflammatory responses. These alterations were, however, reversed by diets supplemented with Aframomum melegueta and Aframomum danielli in a dose-dependent manner. The 8% supplementation produced better outcomes than 4% supplementation and donepezil treatment in most parameters, indicating protective effects on sperm quality and other reproduction-related indices. HPLC profiling revealed bioactive compounds that may collectively account for the observed restorative effects of the seeds.
Conclusions:
These findings demonstrate that Aframomum melegueta and Aframomum danielli seeds effectively reversed the adverse reproductive alterations caused by scopolamine-induced neurotoxicity. Both species significantly improved sperm quality and testicular function, which may suggest their possible development as plant-based nutraceuticals for protecting male reproductive health in future studies. Their phytochemical abundance further supports their potential as plant-based nutraceuticals.
Aim:
Male infertility resulting from neurological disorders, oxidative stress, and hormonal imbalance is a growing health concern. This study, therefore, investigated the effects of Aframomum melegueta and Aframomum danielli-supplemented diets on sperm quality and testicular oxidative damage in a scopolamine-induced rat model.
Methods:
Adult male rats were randomly allocated into seven groups: normal group; scopolamine-induced group; donepezil-treated scopolamine group and four treatment groups receiving 4% or 8% dietary supplementation of Aframomum melegueta or Aframomum danielli, respectively. Sperm motility, count, and morphology were evaluated. In addition, serum testosterone and follicle stimulating hormone levels, testicular oxidative stress markers, inflammatory cytokines, and antioxidant activities were assessed to determine reproductive and biochemical responses. High performance liquid chromatography (HPLC) profiling was also conducted to identify the major phenolic compounds in both seeds.
Results:
Scopolamine administration impaired sperm quality, decreased hormonal levels, promoted oxidative stress, and altered inflammatory responses. These alterations were, however, reversed by diets supplemented with Aframomum melegueta and Aframomum danielli in a dose-dependent manner. The 8% supplementation produced better outcomes than 4% supplementation and donepezil treatment in most parameters, indicating protective effects on sperm quality and other reproduction-related indices. HPLC profiling revealed bioactive compounds that may collectively account for the observed restorative effects of the seeds.
Conclusions:
These findings demonstrate that Aframomum melegueta and Aframomum danielli seeds effectively reversed the adverse reproductive alterations caused by scopolamine-induced neurotoxicity. Both species significantly improved sperm quality and testicular function, which may suggest their possible development as plant-based nutraceuticals for protecting male reproductive health in future studies. Their phytochemical abundance further supports their potential as plant-based nutraceuticals.
DOI: https://doi.org/10.37349/en.2026.1006123
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection (Vol II)

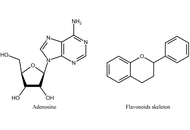
Flavonoids are a large class of natural polyphenolic substances ubiquitously synthesized in the plant kingdom. When entering the human body, these compounds can exert a wide range of biological activities, including immunomodulatory, antiinflammatory, and anticancer effects. Over the recent years, the mechanisms underlying these actions have become increasingly clear, also indicating the important involvement of G protein-coupled receptors (GPCRs), such as adenosine receptors, in signal transduction networks. In this perspective article, the potential role of flavonoids as adenosine receptor antagonists on the development, progression, and spread of glioblastoma is discussed, blocking the tumor-promoting and immunosuppressive actions of elevated levels of endogenous adenosine. Therefore, flavonoids can be considered as structural leads for developing novel antiglioblastoma agents, applied either alone or as boosters of chemo- or immunotherapy to improve the quality of life and outcome of patients. The importance of these studies is, in turn, emphasized by the current lack of effective treatment strategies for this highly aggressive and fast-growing brain tumor, associated with poor prognosis.
Flavonoids are a large class of natural polyphenolic substances ubiquitously synthesized in the plant kingdom. When entering the human body, these compounds can exert a wide range of biological activities, including immunomodulatory, antiinflammatory, and anticancer effects. Over the recent years, the mechanisms underlying these actions have become increasingly clear, also indicating the important involvement of G protein-coupled receptors (GPCRs), such as adenosine receptors, in signal transduction networks. In this perspective article, the potential role of flavonoids as adenosine receptor antagonists on the development, progression, and spread of glioblastoma is discussed, blocking the tumor-promoting and immunosuppressive actions of elevated levels of endogenous adenosine. Therefore, flavonoids can be considered as structural leads for developing novel antiglioblastoma agents, applied either alone or as boosters of chemo- or immunotherapy to improve the quality of life and outcome of patients. The importance of these studies is, in turn, emphasized by the current lack of effective treatment strategies for this highly aggressive and fast-growing brain tumor, associated with poor prognosis.
DOI: https://doi.org/10.37349/en.2026.1006122
This article belongs to the special issue Current Approaches to Malignant Tumors of the Nervous System

Cerebral amyloid angiopathy (CAA), characterized by amyloid β deposition in cerebral vasculature, is increasingly recognized as a major contributor to both cognitive decline and lobar intracerebral hemorrhage (ICH) in older adults and often coexists with Alzheimer’s disease (AD). Understanding CAA is a crucial step for improving health outcomes and the development of effective therapies. However, significant gaps remain in our understanding of CAA’s pathophysiology, diagnostic approaches, biomarker development, and clinical management. A comprehensive review is therefore essential to synthesize existing knowledge and highlight key directions for future research. This review goes beyond prior summaries by critically synthesizing recent evidence on diagnostic innovations—including the Boston criteria v2.0 and emerging plasma biomarkers—and addressing pressing clinical dilemmas such as anticoagulation management in patients with coexisting atrial fibrillation and CAA. It also highlights ongoing research into multimodal diagnostic frameworks and precision treatment strategies aimed at bridging current diagnostic and therapeutic gaps. Together, these updates underscore how advancing biomarker validation, individualized risk stratification, and amyloid-targeted approaches may shape future CAA management and prevention.
Cerebral amyloid angiopathy (CAA), characterized by amyloid β deposition in cerebral vasculature, is increasingly recognized as a major contributor to both cognitive decline and lobar intracerebral hemorrhage (ICH) in older adults and often coexists with Alzheimer’s disease (AD). Understanding CAA is a crucial step for improving health outcomes and the development of effective therapies. However, significant gaps remain in our understanding of CAA’s pathophysiology, diagnostic approaches, biomarker development, and clinical management. A comprehensive review is therefore essential to synthesize existing knowledge and highlight key directions for future research. This review goes beyond prior summaries by critically synthesizing recent evidence on diagnostic innovations—including the Boston criteria v2.0 and emerging plasma biomarkers—and addressing pressing clinical dilemmas such as anticoagulation management in patients with coexisting atrial fibrillation and CAA. It also highlights ongoing research into multimodal diagnostic frameworks and precision treatment strategies aimed at bridging current diagnostic and therapeutic gaps. Together, these updates underscore how advancing biomarker validation, individualized risk stratification, and amyloid-targeted approaches may shape future CAA management and prevention.
DOI: https://doi.org/10.37349/en.2026.1006121

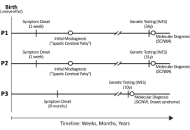
Neurogenetic disorders remain genetically uncharacterized in many populations, including Libya. We report three Libyan patients from two consanguineous families with pathogenic variants in sodium channel genes. Two adult sisters (Patients 1 & 2) presented with global developmental delay and progressive spastic paraparesis without epilepsy. Whole exome sequencing identified the same heterozygous SCN8A variant (c.142G>A; p.Asp48Asn) in both sisters, classified as a variant of uncertain significance (VUS). Its occurrence in two affected siblings with a consistent phenotype and the absence of other explanatory variants provide supporting evidence for its potential pathogenicity. These cases represent the first documented instances of a suspected SCN8A-related disorder in Libya. A third, unrelated 10-year-old boy (Patient 3) with a phenotype consistent with Dravet syndrome, including refractory seizures and neurodevelopmental regression, was found to harbor a likely pathogenic heterozygous SCN1A variant (c.2113del; p.Glu705Lysfs*10). This report expands the genetic and phenotypic spectrum of neurological disorders in Libya and underscores the critical role of genetic testing, while also highlighting the need for segregation studies to achieve a definitive molecular diagnosis.
Neurogenetic disorders remain genetically uncharacterized in many populations, including Libya. We report three Libyan patients from two consanguineous families with pathogenic variants in sodium channel genes. Two adult sisters (Patients 1 & 2) presented with global developmental delay and progressive spastic paraparesis without epilepsy. Whole exome sequencing identified the same heterozygous SCN8A variant (c.142G>A; p.Asp48Asn) in both sisters, classified as a variant of uncertain significance (VUS). Its occurrence in two affected siblings with a consistent phenotype and the absence of other explanatory variants provide supporting evidence for its potential pathogenicity. These cases represent the first documented instances of a suspected SCN8A-related disorder in Libya. A third, unrelated 10-year-old boy (Patient 3) with a phenotype consistent with Dravet syndrome, including refractory seizures and neurodevelopmental regression, was found to harbor a likely pathogenic heterozygous SCN1A variant (c.2113del; p.Glu705Lysfs*10). This report expands the genetic and phenotypic spectrum of neurological disorders in Libya and underscores the critical role of genetic testing, while also highlighting the need for segregation studies to achieve a definitive molecular diagnosis.
DOI: https://doi.org/10.37349/en.2025.1006120
This article belongs to the special issue Advances in Epilepsy Research

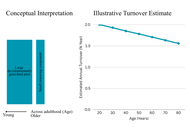
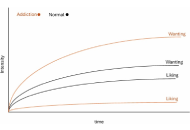
Although addiction is a complex and contextually embedded disorder that extends beyond individual pathology and neurobiological dysfunction, prevailing computational and clinical models often reduce addiction to a chronic brain disease. While such frameworks have shaped dominant approaches to treatment and theory, they remain poorly aligned with the lived experience and behavioral phenomena of addiction, ignoring its psychological, social, and systemic dimensions. This paper examines the limitations of various disease and compulsion models both critically and in-depth, highlighting their empirical and conceptual shortcomings. In doing so, it argues for the development of context-sensitive and psychologically grounded computational models, ones capable of capturing the nuanced realities of addiction and informing more effective, personalized interventions.
Although addiction is a complex and contextually embedded disorder that extends beyond individual pathology and neurobiological dysfunction, prevailing computational and clinical models often reduce addiction to a chronic brain disease. While such frameworks have shaped dominant approaches to treatment and theory, they remain poorly aligned with the lived experience and behavioral phenomena of addiction, ignoring its psychological, social, and systemic dimensions. This paper examines the limitations of various disease and compulsion models both critically and in-depth, highlighting their empirical and conceptual shortcomings. In doing so, it argues for the development of context-sensitive and psychologically grounded computational models, ones capable of capturing the nuanced realities of addiction and informing more effective, personalized interventions.
DOI: https://doi.org/10.37349/en.2025.1006119

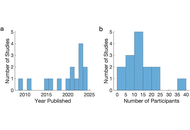
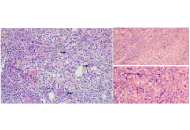
Primary central nervous system lymphoma is a rare form of extranodal non-Hodgkin lymphoma that is confined to the brain, spinal cord, leptomeninges, or eyes, representing less than one percent of all non-Hodgkin lymphomas and approximately four percent of primary brain tumors. When the disease is truly isolated to the central nervous system, with no evidence of systemic spread, it poses unique diagnostic and therapeutic challenges, particularly in immunocompetent patients. We reviewed nine recently published cases from 2021 to 2024 that described isolated primary central nervous system lymphoma without extracranial involvement. Patients ranged in age from forty-four to eighty-five years, with both immunocompetent and immunosuppressed individuals represented. Presenting symptoms include focal neurological deficits, seizures, progressive confusion, cranial neuropathies, and neurolymphomatosis. Magnetic resonance imaging findings were diverse, including intra-axial masses, leptomeningeal and cranial nerve enhancement, and mass effect. Cerebrospinal fluid analysis was variably positive for lymphoma cells. Histopathological analysis confirmed diffuse large B-cell lymphoma in all cases, although initial biopsies were sometimes inconclusive, underscoring the importance of repeat tissue sampling and expert pathology review. Treatment strategies most often included high-dose methotrexate-based chemotherapy, monoclonal antibody therapy, and radiotherapy, with some patients undergoing surgical decompression or diagnostic craniotomy. Follow-up data revealed variable survival outcomes, with a subset of patients achieving disease-free survival beyond one year. These cases highlight the wide clinical spectrum and diagnostic complexity of isolated primary central nervous system lymphoma and reinforce the need for a high index of suspicion, timely advanced imaging, multidisciplinary discussion, and appropriate tissue diagnosis to guide individualized management.
Primary central nervous system lymphoma is a rare form of extranodal non-Hodgkin lymphoma that is confined to the brain, spinal cord, leptomeninges, or eyes, representing less than one percent of all non-Hodgkin lymphomas and approximately four percent of primary brain tumors. When the disease is truly isolated to the central nervous system, with no evidence of systemic spread, it poses unique diagnostic and therapeutic challenges, particularly in immunocompetent patients. We reviewed nine recently published cases from 2021 to 2024 that described isolated primary central nervous system lymphoma without extracranial involvement. Patients ranged in age from forty-four to eighty-five years, with both immunocompetent and immunosuppressed individuals represented. Presenting symptoms include focal neurological deficits, seizures, progressive confusion, cranial neuropathies, and neurolymphomatosis. Magnetic resonance imaging findings were diverse, including intra-axial masses, leptomeningeal and cranial nerve enhancement, and mass effect. Cerebrospinal fluid analysis was variably positive for lymphoma cells. Histopathological analysis confirmed diffuse large B-cell lymphoma in all cases, although initial biopsies were sometimes inconclusive, underscoring the importance of repeat tissue sampling and expert pathology review. Treatment strategies most often included high-dose methotrexate-based chemotherapy, monoclonal antibody therapy, and radiotherapy, with some patients undergoing surgical decompression or diagnostic craniotomy. Follow-up data revealed variable survival outcomes, with a subset of patients achieving disease-free survival beyond one year. These cases highlight the wide clinical spectrum and diagnostic complexity of isolated primary central nervous system lymphoma and reinforce the need for a high index of suspicion, timely advanced imaging, multidisciplinary discussion, and appropriate tissue diagnosis to guide individualized management.
DOI: https://doi.org/10.37349/en.2025.1006117
 Previous
Previous