




Background:
Diabetes mellitus (DM) refers to a group of metabolic diseases characterized by reduced insulin synthesis and release, leading to hyperglycemia, oxidative stress, and inflammation. The prevalence of DM continues to increase worldwide, primarily driven by type 2 DM (T2DM). While numerous prescription drugs are widely available for the management of T2DM, these treatments must be coupled with significant changes in diet and lifestyle to have long-term effects. This comparative review aims to summarize and critically evaluate some of the dietary antioxidant and anti-inflammatory functional foods (FFs), including bioactive compounds on glycemic regulation, oxidative stress, inflammatory markers, and other outcomes in T2DM patients.
Methods:
A comprehensive literature search was performed from June 2014 to December 2025 in Cochrane Library, Embase, Google Scholar, PubMed/Medline, and Scopus databases.
Results:
Thirty-one meta-analyses describing the effects of specific FFs on glycemic outcomes, including fasting blood glucose, HbA1c, insulin levels, insulin resistance, oxidative or inflammatory stress, and/or the doses needed to achieve these endpoints, in T2DM patients were included. Comparative analyses of the data for green tea in T2DM showed significant antioxidant effects, but conflicting data for glycemic regulation, and no significant effect on inflammatory markers. Flaxseed products significantly reduced oxidative stress and HbA1c but had no anti-inflammatory effects. Both anthocyanins and resveratrol from fruits and vegetables significantly reduced fasting blood glucose, HbA1c, as well as oxidative and inflammatory markers. Turmeric and curcumin significantly reduced fasting blood glucose, but the data for ginger were conflicted. In T2DM patients, turmeric and ginger significantly reduced markers of oxidation and inflammation.
Discussion:
Overall, improvements in diet, including the incorporation of specific FFs and exercise, along with standard treatments, may be cost-effective methods to reduce the public health impact of T2DM. Limitations included the significant heterogeneity, high risk of bias, and low to moderate quality of the randomized controlled trial (RCT).
Background:
Diabetes mellitus (DM) refers to a group of metabolic diseases characterized by reduced insulin synthesis and release, leading to hyperglycemia, oxidative stress, and inflammation. The prevalence of DM continues to increase worldwide, primarily driven by type 2 DM (T2DM). While numerous prescription drugs are widely available for the management of T2DM, these treatments must be coupled with significant changes in diet and lifestyle to have long-term effects. This comparative review aims to summarize and critically evaluate some of the dietary antioxidant and anti-inflammatory functional foods (FFs), including bioactive compounds on glycemic regulation, oxidative stress, inflammatory markers, and other outcomes in T2DM patients.
Methods:
A comprehensive literature search was performed from June 2014 to December 2025 in Cochrane Library, Embase, Google Scholar, PubMed/Medline, and Scopus databases.
Results:
Thirty-one meta-analyses describing the effects of specific FFs on glycemic outcomes, including fasting blood glucose, HbA1c, insulin levels, insulin resistance, oxidative or inflammatory stress, and/or the doses needed to achieve these endpoints, in T2DM patients were included. Comparative analyses of the data for green tea in T2DM showed significant antioxidant effects, but conflicting data for glycemic regulation, and no significant effect on inflammatory markers. Flaxseed products significantly reduced oxidative stress and HbA1c but had no anti-inflammatory effects. Both anthocyanins and resveratrol from fruits and vegetables significantly reduced fasting blood glucose, HbA1c, as well as oxidative and inflammatory markers. Turmeric and curcumin significantly reduced fasting blood glucose, but the data for ginger were conflicted. In T2DM patients, turmeric and ginger significantly reduced markers of oxidation and inflammation.
Discussion:
Overall, improvements in diet, including the incorporation of specific FFs and exercise, along with standard treatments, may be cost-effective methods to reduce the public health impact of T2DM. Limitations included the significant heterogeneity, high risk of bias, and low to moderate quality of the randomized controlled trial (RCT).
DOI: https://doi.org/10.37349/eemd.2026.101474

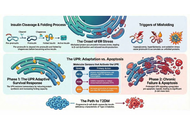
Establishing remission as a clinical goal for carefully selected patients with Type 2 Diabetes Mellitus (T2DM) represents a fundamental shift from the traditional paradigm of lifelong disease management. Short disease duration, preserved C-peptide levels, and overweight or obesity are the central criteria for T2DM remission. T2DM remission may help reduce the psychological burden on patients, improve quality of life, delay disease progression, and reduce the risk of long-term complications. This consensus aims to help physicians establish standardized clinical diagnosis and treatment protocols for T2DM remission in overweight or obese patients with T2DM, and support related research to develop safer and more effective interventions. Notably, T2DM remission is conditional, reversible and population-selective, which is not universally applicable to all T2DM patients across different disease stages and healthcare settings.
Establishing remission as a clinical goal for carefully selected patients with Type 2 Diabetes Mellitus (T2DM) represents a fundamental shift from the traditional paradigm of lifelong disease management. Short disease duration, preserved C-peptide levels, and overweight or obesity are the central criteria for T2DM remission. T2DM remission may help reduce the psychological burden on patients, improve quality of life, delay disease progression, and reduce the risk of long-term complications. This consensus aims to help physicians establish standardized clinical diagnosis and treatment protocols for T2DM remission in overweight or obese patients with T2DM, and support related research to develop safer and more effective interventions. Notably, T2DM remission is conditional, reversible and population-selective, which is not universally applicable to all T2DM patients across different disease stages and healthcare settings.
DOI: https://doi.org/10.37349/eemd.2026.101473

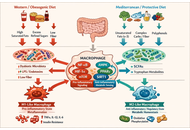
The emerging field of immunometabolism has established that the metabolic pathways governing immune cell function—glycolysis, oxidative phosphorylation, fatty acid oxidation, and amino acid metabolism—are fundamental determinants of immune responses in health and disease. This review synthesizes current evidence on how dietary inputs serve as primary environmental modulators of this immunometabolism programming. We detail the mechanisms by which macronutrients (carbohydrates, lipids, proteins), micronutrients, and specific dietary patterns (e.g., Western, Mediterranean, ketogenic) engage key nutrient-sensing pathways (mechanistic target of rapamycin [mTOR], AMP-activated protein kinase [AMPK], hypoxia-inducible factor-1α [HIF-1α], peroxisome proliferator-activated receptor [PPARs]) to rewire immune cell metabolism and influence functional phenotypes. A central role is afforded to the gut microbiota as a critical intermediary, translating diet into immunoregulatory signals, such as short-chain fatty acids. We further explore how obesogenic diets disrupt this network to fuel chronic low-grade inflammation (metaflammation), driving the pathophysiology of common metabolic diseases, including obesity, type 2 diabetes, non-alcoholic fatty liver disease (NAFLD), and atherosclerosis. Finally, we evaluate therapeutic nutritional strategies—from nutraceuticals and probiotics to the promise of precision nutrition—designed to recalibrate immunometabolism. This review underscores that diet is a powerful, modifiable lever of immunity, positioning targeted nutritional intervention as a pivotal strategy for preventing and managing metabolic disease.
The emerging field of immunometabolism has established that the metabolic pathways governing immune cell function—glycolysis, oxidative phosphorylation, fatty acid oxidation, and amino acid metabolism—are fundamental determinants of immune responses in health and disease. This review synthesizes current evidence on how dietary inputs serve as primary environmental modulators of this immunometabolism programming. We detail the mechanisms by which macronutrients (carbohydrates, lipids, proteins), micronutrients, and specific dietary patterns (e.g., Western, Mediterranean, ketogenic) engage key nutrient-sensing pathways (mechanistic target of rapamycin [mTOR], AMP-activated protein kinase [AMPK], hypoxia-inducible factor-1α [HIF-1α], peroxisome proliferator-activated receptor [PPARs]) to rewire immune cell metabolism and influence functional phenotypes. A central role is afforded to the gut microbiota as a critical intermediary, translating diet into immunoregulatory signals, such as short-chain fatty acids. We further explore how obesogenic diets disrupt this network to fuel chronic low-grade inflammation (metaflammation), driving the pathophysiology of common metabolic diseases, including obesity, type 2 diabetes, non-alcoholic fatty liver disease (NAFLD), and atherosclerosis. Finally, we evaluate therapeutic nutritional strategies—from nutraceuticals and probiotics to the promise of precision nutrition—designed to recalibrate immunometabolism. This review underscores that diet is a powerful, modifiable lever of immunity, positioning targeted nutritional intervention as a pivotal strategy for preventing and managing metabolic disease.
DOI: https://doi.org/10.37349/eemd.2026.101472

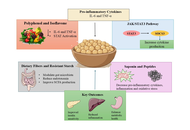
Metabolic diseases, including obesity, insulin resistance, and type 2 diabetes mellitus, are increasingly recognized as conditions characterized by chronic low-grade inflammation driven by dysregulated cytokine signaling. Persistent elevation of pro-inflammatory mediators disrupts insulin signaling and contributes to metabolic dysfunction. Nutritional interventions capable of restoring cytokine balance are emerging as promising adjunct strategies for metabolic disease management. Chickpea (Cicer arietinum L.), a widely consumed legume, is rich in polyphenols, saponins, dietary fiber, and bioactive peptides with potential immunometabolic benefits. This review critically summarizes the role of chickpea bioactives in modulating cytokine signaling networks, with particular emphasis on the IL-6-JAK-STAT3-SOCS3 axis and NF-κB pathway. Chickpea components exert antioxidant and anti-inflammatory effects, suppress pro-inflammatory cytokine production, and may improve insulin sensitivity. Additionally, their potential chemoprotective effects in obesity-associated metabolic disorders are discussed. Furthermore, this review highlights key gaps in mechanistic and translational research and proposes future directions for pathway-oriented investigations. Although current evidence supports the therapeutic potential of chickpea, further mechanistic and clinical validation is required.
Metabolic diseases, including obesity, insulin resistance, and type 2 diabetes mellitus, are increasingly recognized as conditions characterized by chronic low-grade inflammation driven by dysregulated cytokine signaling. Persistent elevation of pro-inflammatory mediators disrupts insulin signaling and contributes to metabolic dysfunction. Nutritional interventions capable of restoring cytokine balance are emerging as promising adjunct strategies for metabolic disease management. Chickpea (Cicer arietinum L.), a widely consumed legume, is rich in polyphenols, saponins, dietary fiber, and bioactive peptides with potential immunometabolic benefits. This review critically summarizes the role of chickpea bioactives in modulating cytokine signaling networks, with particular emphasis on the IL-6-JAK-STAT3-SOCS3 axis and NF-κB pathway. Chickpea components exert antioxidant and anti-inflammatory effects, suppress pro-inflammatory cytokine production, and may improve insulin sensitivity. Additionally, their potential chemoprotective effects in obesity-associated metabolic disorders are discussed. Furthermore, this review highlights key gaps in mechanistic and translational research and proposes future directions for pathway-oriented investigations. Although current evidence supports the therapeutic potential of chickpea, further mechanistic and clinical validation is required.
DOI: https://doi.org/10.37349/eemd.2026.101471
This article belongs to the special issue Role of Dysregulated Cytokine Signaling Pathways in Metabolic Disease

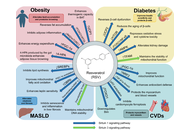
Obesity, diabetes mellitus (DM), metabolic dysfunction-associated steatotic liver disease (MASLD), and cardiovascular diseases (CVDs) share pathogenic mechanisms like oxidative stress and inflammation. Resveratrol (RSV) offers therapeutic potential by activating the sirtuin (SIRT) signaling network. This review synthesizes RSV’s pharmacological impacts on adipose, pancreatic, hepatic, and cardiovascular tissues, focusing on the AMPK/SIRT1/PGC-1α axis, PI3K/AKT pathways, and epigenetic modulations. Despite robust preclinical data, a significant translational gap exists. Clinical evidence is heterogeneous, often contradicting animal studies due to varying dosages, durations, and population characteristics. RSV acts as a pan-SIRT activator, though its precise SIRT1 activation mechanism, direct or via NAD+ modulation, remains debated. In obesity, RSV promotes adipose beiging and thermogenesis, yet clinical weight loss is modest. For DM, it preserves β-cell function and improves insulin sensitivity, primarily benefiting diabetic populations with variable glycemic outcomes. In MASLD, RSV ameliorates steatosis and fibrosis in models, but large-scale human trials confirming histological benefits are lacking. Regarding CVDs, RSV protects against endothelial dysfunction and inflammation, showing minor improvements in biomarkers like blood pressure, though hard endpoints need validation. Major limitations hinder clinical efficacy, such as poor oral bioavailability, rapid metabolism, and significant interindividual pharmacokinetic variability. The lack of standardized formulations further complicates systemic exposure. Nevertheless, the RSV-SIRT axis remains a unified metabolic and epigenetic modulator, stabilizing cellular microenvironments across organ systems. It represents a promising target for complex metabolic syndromes. Future research should prioritize overcoming bioavailability challenges through novel delivery systems and investigating synergistic combinatorial therapies to bridge the gap between preclinical promise and clinical reality.
Obesity, diabetes mellitus (DM), metabolic dysfunction-associated steatotic liver disease (MASLD), and cardiovascular diseases (CVDs) share pathogenic mechanisms like oxidative stress and inflammation. Resveratrol (RSV) offers therapeutic potential by activating the sirtuin (SIRT) signaling network. This review synthesizes RSV’s pharmacological impacts on adipose, pancreatic, hepatic, and cardiovascular tissues, focusing on the AMPK/SIRT1/PGC-1α axis, PI3K/AKT pathways, and epigenetic modulations. Despite robust preclinical data, a significant translational gap exists. Clinical evidence is heterogeneous, often contradicting animal studies due to varying dosages, durations, and population characteristics. RSV acts as a pan-SIRT activator, though its precise SIRT1 activation mechanism, direct or via NAD+ modulation, remains debated. In obesity, RSV promotes adipose beiging and thermogenesis, yet clinical weight loss is modest. For DM, it preserves β-cell function and improves insulin sensitivity, primarily benefiting diabetic populations with variable glycemic outcomes. In MASLD, RSV ameliorates steatosis and fibrosis in models, but large-scale human trials confirming histological benefits are lacking. Regarding CVDs, RSV protects against endothelial dysfunction and inflammation, showing minor improvements in biomarkers like blood pressure, though hard endpoints need validation. Major limitations hinder clinical efficacy, such as poor oral bioavailability, rapid metabolism, and significant interindividual pharmacokinetic variability. The lack of standardized formulations further complicates systemic exposure. Nevertheless, the RSV-SIRT axis remains a unified metabolic and epigenetic modulator, stabilizing cellular microenvironments across organ systems. It represents a promising target for complex metabolic syndromes. Future research should prioritize overcoming bioavailability challenges through novel delivery systems and investigating synergistic combinatorial therapies to bridge the gap between preclinical promise and clinical reality.
DOI: https://doi.org/10.37349/eemd.2026.101470

Aim:
The aim of this study was to compare the effects of high-intensity interval training (HIIT) and concurrent training (CONC) on body composition and metabolic profile in postmenopausal women.
Methods:
Forty-five postmenopausal women with overweight or obesity (50–65 years) were randomly assigned to one of three groups: HIIT group, CONC group, or control group (CG). All groups underwent a 12-week intervention, consisting of three training sessions per week, scheduled on alternate days. All dependent variables (body composition, lipid, or glycemic markers) were assessed before and after a 12-week intervention period.
Results:
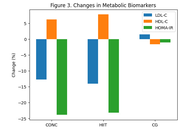
The CONC and HIIT groups showed anthropometric improvements compared to the CG. In this regard, CONC and HIIT significantly (P < 0.05) reduced their body weight and body mass index (BMI) compared with the CG. This reduction was due to a decrease in fat mass, mainly in the abdominal area, as reflected by the reduction in the waist-to-hip ratio. Participants in the CONC group showed greater reductions in body weight, BMI, and body fat percentage compared with the HIIT group, while both exercise interventions produced similar improvements in metabolic biomarkers. Regarding circulating parameters, CONC and HIIT significantly (P < 0.05) reduced circulating triglycerides, total cholesterol, and LDL-cholesterol, which resulted in a decrease in the atherogenic index of plasma (AIP, calculated as log[TG/HDL-C]) compared with CG. Blood glucose and insulin levels also decreased significantly (P < 0.05) in CONC and HIIT compared with CG, with a consequent reduction in the homeostatic model assessment-insulin resistance (HOMA-IR).
Conclusions:
A 12-week program of HIIT or CONC training represents an effective strategy for improving body fat reduction and metabolic profile in sedentary postmenopausal women with overweight or obesity [ClinicalTrials.gov, identifier (ID NCT07302191)].
Aim:
The aim of this study was to compare the effects of high-intensity interval training (HIIT) and concurrent training (CONC) on body composition and metabolic profile in postmenopausal women.
Methods:
Forty-five postmenopausal women with overweight or obesity (50–65 years) were randomly assigned to one of three groups: HIIT group, CONC group, or control group (CG). All groups underwent a 12-week intervention, consisting of three training sessions per week, scheduled on alternate days. All dependent variables (body composition, lipid, or glycemic markers) were assessed before and after a 12-week intervention period.
Results:
The CONC and HIIT groups showed anthropometric improvements compared to the CG. In this regard, CONC and HIIT significantly (P < 0.05) reduced their body weight and body mass index (BMI) compared with the CG. This reduction was due to a decrease in fat mass, mainly in the abdominal area, as reflected by the reduction in the waist-to-hip ratio. Participants in the CONC group showed greater reductions in body weight, BMI, and body fat percentage compared with the HIIT group, while both exercise interventions produced similar improvements in metabolic biomarkers. Regarding circulating parameters, CONC and HIIT significantly (P < 0.05) reduced circulating triglycerides, total cholesterol, and LDL-cholesterol, which resulted in a decrease in the atherogenic index of plasma (AIP, calculated as log[TG/HDL-C]) compared with CG. Blood glucose and insulin levels also decreased significantly (P < 0.05) in CONC and HIIT compared with CG, with a consequent reduction in the homeostatic model assessment-insulin resistance (HOMA-IR).
Conclusions:
A 12-week program of HIIT or CONC training represents an effective strategy for improving body fat reduction and metabolic profile in sedentary postmenopausal women with overweight or obesity [ClinicalTrials.gov, identifier (ID NCT07302191)].
DOI: https://doi.org/10.37349/eemd.2026.101469
This article belongs to the special issue Metabolic Syndrome in Menopause

The global rise in consumption of added sugars and confectionery parallels the increasing prevalence of obesity, metabolic dysfunction, and male hypogonadism. Testosterone is essential for male reproductive, metabolic, and cardiovascular health, and growing evidence indicates that excessive sugar intake may disrupt hormonal regulation. This narrative review synthesizes data from human observational and interventional studies and experimental animal models to evaluate the effects of sugar consumption on testosterone homeostasis and male reproductive outcomes. Acute glucose ingestion is associated with hyperinsulinemia and inflammatory cytokine release, which may contribute to transient reductions in circulating testosterone of approximately 20–30%, although findings vary across populations and study designs. Chronic high-sugar intake, particularly from sugar-sweetened beverages, promotes oxidative stress, adiposity, insulin resistance, and leptin dysregulation, which may collectively impair Leydig cell steroidogenesis and contribute to testosterone suppression. Animal studies consistently demonstrate testicular structural damage, downregulation of steroidogenic enzymes, and impaired spermatogenesis under high-sugar dietary conditions. Epidemiological studies associate frequent intake of sugar-sweetened beverages and confectionery with lower testosterone levels and poorer semen parameters. Emerging evidence suggests partial reversibility with dietary interventions emphasizing reduced sugar intake and increased antioxidant consumption. Overall, current data support a biologically plausible link between excessive sugar intake and impaired testosterone regulation, underscoring the need for longitudinal studies and public health strategies targeting added sugar reduction.
The global rise in consumption of added sugars and confectionery parallels the increasing prevalence of obesity, metabolic dysfunction, and male hypogonadism. Testosterone is essential for male reproductive, metabolic, and cardiovascular health, and growing evidence indicates that excessive sugar intake may disrupt hormonal regulation. This narrative review synthesizes data from human observational and interventional studies and experimental animal models to evaluate the effects of sugar consumption on testosterone homeostasis and male reproductive outcomes. Acute glucose ingestion is associated with hyperinsulinemia and inflammatory cytokine release, which may contribute to transient reductions in circulating testosterone of approximately 20–30%, although findings vary across populations and study designs. Chronic high-sugar intake, particularly from sugar-sweetened beverages, promotes oxidative stress, adiposity, insulin resistance, and leptin dysregulation, which may collectively impair Leydig cell steroidogenesis and contribute to testosterone suppression. Animal studies consistently demonstrate testicular structural damage, downregulation of steroidogenic enzymes, and impaired spermatogenesis under high-sugar dietary conditions. Epidemiological studies associate frequent intake of sugar-sweetened beverages and confectionery with lower testosterone levels and poorer semen parameters. Emerging evidence suggests partial reversibility with dietary interventions emphasizing reduced sugar intake and increased antioxidant consumption. Overall, current data support a biologically plausible link between excessive sugar intake and impaired testosterone regulation, underscoring the need for longitudinal studies and public health strategies targeting added sugar reduction.
DOI: https://doi.org/10.37349/eemd.2026.101468

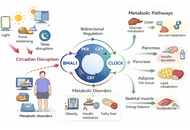
Circadian rhythms are intrinsic 24-hour cycles that coordinate key metabolic processes, including glucose homeostasis, lipid metabolism, and energy expenditure. Disruption of these rhythms, due to sleep disturbances, shift work, or irregular feeding schedules, contributes to the development of obesity, insulin resistance, and dyslipidemia. Core clock genes, including circadian locomotor output cycles kaput (CLOCK), brain and muscle ARNT-like 1 (BMAL1), period gene (PER), and cryptochrome (CRY), play a central role in orchestrating these metabolic pathways. Chronotherapy, aligning lifestyle, behavioral, and pharmacologic interventions with circadian timing, represents a promising, yet underexplored, strategy for metabolic disease management. Evidence suggests that interventions such as time-restricted feeding, light therapy, pharmacologic modulators of clock gene expression, and wearable technology can improve glucose control, lipid profiles, and body weight. This review synthesizes current knowledge on the molecular regulation of metabolism by circadian clocks, elucidates mechanistic links between rhythm disruption and metabolic dysfunction, and explores translational strategies to restore circadian homeostasis. By targeting circadian rhythms, personalized and cost-effective interventions can be developed to mitigate the global burden of metabolic disorders. Future research should focus on large-scale clinical trials, precision chronotherapy, and integration of wearable devices to optimize the timing of interventions, ultimately enhancing treatment efficacy and long-term metabolic health.
Circadian rhythms are intrinsic 24-hour cycles that coordinate key metabolic processes, including glucose homeostasis, lipid metabolism, and energy expenditure. Disruption of these rhythms, due to sleep disturbances, shift work, or irregular feeding schedules, contributes to the development of obesity, insulin resistance, and dyslipidemia. Core clock genes, including circadian locomotor output cycles kaput (CLOCK), brain and muscle ARNT-like 1 (BMAL1), period gene (PER), and cryptochrome (CRY), play a central role in orchestrating these metabolic pathways. Chronotherapy, aligning lifestyle, behavioral, and pharmacologic interventions with circadian timing, represents a promising, yet underexplored, strategy for metabolic disease management. Evidence suggests that interventions such as time-restricted feeding, light therapy, pharmacologic modulators of clock gene expression, and wearable technology can improve glucose control, lipid profiles, and body weight. This review synthesizes current knowledge on the molecular regulation of metabolism by circadian clocks, elucidates mechanistic links between rhythm disruption and metabolic dysfunction, and explores translational strategies to restore circadian homeostasis. By targeting circadian rhythms, personalized and cost-effective interventions can be developed to mitigate the global burden of metabolic disorders. Future research should focus on large-scale clinical trials, precision chronotherapy, and integration of wearable devices to optimize the timing of interventions, ultimately enhancing treatment efficacy and long-term metabolic health.
DOI: https://doi.org/10.37349/eemd.2026.101467

Osteoarthritis (OA) is the most prevalent form of arthritis and a leading cause of disability worldwide. Epidemiological studies indicate that its global prevalence is rising, particularly among women, who often experience more severe disease than men. This sex disparity is most pronounced around menopause and is influenced by female-specific biological and hormonal factors, joint morphology, reproductive history, and sex-specific comorbidities that contribute to disease onset and progression. A mechanistic understanding of these factors is essential to identify individuals at risk, implement preventive strategies, enable early diagnosis, and mitigate complications, ultimately preserving function and quality of life. This review focuses on the determinants underlying the higher incidence and severity of OA in women, highlighting the interplay between hormonal changes, anatomy, and systemic factors.
Osteoarthritis (OA) is the most prevalent form of arthritis and a leading cause of disability worldwide. Epidemiological studies indicate that its global prevalence is rising, particularly among women, who often experience more severe disease than men. This sex disparity is most pronounced around menopause and is influenced by female-specific biological and hormonal factors, joint morphology, reproductive history, and sex-specific comorbidities that contribute to disease onset and progression. A mechanistic understanding of these factors is essential to identify individuals at risk, implement preventive strategies, enable early diagnosis, and mitigate complications, ultimately preserving function and quality of life. This review focuses on the determinants underlying the higher incidence and severity of OA in women, highlighting the interplay between hormonal changes, anatomy, and systemic factors.
DOI: https://doi.org/10.37349/eemd.2026.101466

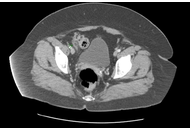
Epiploic appendagitis is a rare, self-limiting inflammatory condition of the colonic epiploic appendages. It commonly presents with acute focal lower quadrant abdominal pain and may mimic appendicitis, colitis, or diverticulitis. Rapid weight loss has been proposed as a potential predisposing factor, although clinical evidence remains limited. This report describes a case of epiploic appendagitis occurring in temporal association with substantial and relatively rapid weight reduction during treatment with tirzepatide, a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. A 45-year-old individual with a history of obesity, prediabetes, hypertension, hypothyroidism, and inflammatory bowel syndrome presented to primary care with right lower quadrant abdominal pain. Initial evaluation attributed symptoms to constipation; however, pain progressed despite management. Computed tomography (CT) of the abdomen and pelvis with contrast was obtained due to concern for appendicitis and demonstrated imaging findings consistent with epiploic appendagitis localized to the cecum. The patient had experienced clinically significant and relatively rapid weight loss during tirzepatide dose escalation prior to presentation. Management was conservative with nonsteroidal anti-inflammatory drugs and supportive care, without surgical intervention. Symptoms resolved completely within two weeks, and tirzepatide therapy was continued without recurrence. Although epiploic appendagitis has not been identified as a safety signal in randomized trials, systematic reviews, or pharmacovigilance analyses of GLP-1/GIP receptor agonists, this case highlights epiploic appendagitis as an important diagnostic consideration in patients presenting with focal abdominal pain during periods of substantial weight loss. Awareness of this uncommon condition may help prevent misdiagnosis and unnecessary interventions as pharmacologic weight loss therapies become more widely used.
Epiploic appendagitis is a rare, self-limiting inflammatory condition of the colonic epiploic appendages. It commonly presents with acute focal lower quadrant abdominal pain and may mimic appendicitis, colitis, or diverticulitis. Rapid weight loss has been proposed as a potential predisposing factor, although clinical evidence remains limited. This report describes a case of epiploic appendagitis occurring in temporal association with substantial and relatively rapid weight reduction during treatment with tirzepatide, a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. A 45-year-old individual with a history of obesity, prediabetes, hypertension, hypothyroidism, and inflammatory bowel syndrome presented to primary care with right lower quadrant abdominal pain. Initial evaluation attributed symptoms to constipation; however, pain progressed despite management. Computed tomography (CT) of the abdomen and pelvis with contrast was obtained due to concern for appendicitis and demonstrated imaging findings consistent with epiploic appendagitis localized to the cecum. The patient had experienced clinically significant and relatively rapid weight loss during tirzepatide dose escalation prior to presentation. Management was conservative with nonsteroidal anti-inflammatory drugs and supportive care, without surgical intervention. Symptoms resolved completely within two weeks, and tirzepatide therapy was continued without recurrence. Although epiploic appendagitis has not been identified as a safety signal in randomized trials, systematic reviews, or pharmacovigilance analyses of GLP-1/GIP receptor agonists, this case highlights epiploic appendagitis as an important diagnostic consideration in patients presenting with focal abdominal pain during periods of substantial weight loss. Awareness of this uncommon condition may help prevent misdiagnosis and unnecessary interventions as pharmacologic weight loss therapies become more widely used.
DOI: https://doi.org/10.37349/eemd.2026.101465

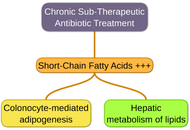
The hypothesis that early-life antibiotic exposure predisposes to obesity has, over the past decade, gained substantial traction across both biomedical literature and public discourse. Its appeal lies in a seemingly coherent mechanistic framework: disruption of the developing intestinal microbiota is presumed to induce long-lasting alterations in metabolic homeostasis, thereby promoting increased adiposity. However, reported effect sizes are consistently modest, with odds ratios typically ranging from 1.1 to 1.3, values that approach the limits of residual confounding and statistical imprecision. Studies incorporating rigorous adjustment for familial environment, socioeconomic status, and dietary patterns observe a complete loss of the association, underscoring the dominant influence of these confounders. This review revisits the conceptual emergence and sustained prominence of the antibiotic-obesity paradigm, positioning it as a case study in the amplification of weak epidemiological signals through mechanistic plausibility. Notwithstanding its methodological limitations, the hypothesis has exerted a constructive influence by fostering more judicious antibiotic use and stimulating renewed investigation into host-microbiota interactions within the context of metabolic disease.
The hypothesis that early-life antibiotic exposure predisposes to obesity has, over the past decade, gained substantial traction across both biomedical literature and public discourse. Its appeal lies in a seemingly coherent mechanistic framework: disruption of the developing intestinal microbiota is presumed to induce long-lasting alterations in metabolic homeostasis, thereby promoting increased adiposity. However, reported effect sizes are consistently modest, with odds ratios typically ranging from 1.1 to 1.3, values that approach the limits of residual confounding and statistical imprecision. Studies incorporating rigorous adjustment for familial environment, socioeconomic status, and dietary patterns observe a complete loss of the association, underscoring the dominant influence of these confounders. This review revisits the conceptual emergence and sustained prominence of the antibiotic-obesity paradigm, positioning it as a case study in the amplification of weak epidemiological signals through mechanistic plausibility. Notwithstanding its methodological limitations, the hypothesis has exerted a constructive influence by fostering more judicious antibiotic use and stimulating renewed investigation into host-microbiota interactions within the context of metabolic disease.
DOI: https://doi.org/10.37349/eemd.2026.101464

Aim:
Necrotizing fasciitis (NF) is a severe and early challenging-to-identify complication of diabetic foot (DF). This study aimed to develop and validate a novel risk assessment model for NF with DF patients utilizing conventional clinical indicators.
Methods:
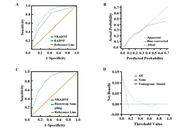
A retrospective analysis was conducted on 815 DF patients admitted to the First Affiliated Hospital of Chongqing Medical University between October 2018 and April 2022. Based on the presence of NF, patients were stratified into a DF group (n = 703) and a DF complicated with NF (DNF) group (n = 112). Clinically and statistically significant variables were converted into categorical form. A new risk assessment for DNF (NRADNF) nomogram was developed via multivariable stepwise logistic regression. Model performance was evaluated using the area under the receiver operating characteristic curve (AUC) for discriminative ability, the Hosmer-Lemeshow goodness-of-fit test for calibration, decision curve analysis (DCA) for clinical utility, and bootstrap resampling for stability.
Results:
The final NRADNF model incorporated six indicators: age < 60 years, body temperature ≥ 38°C, foot skin necrosis, neutrophil-to-lymphocyte ratio (NLR) ≥ 8.5, hypersensitive C-reactive protein > 20 mg/L, and hemoglobin ≤ 100 g/L. The model demonstrated favorable predictive performance with an AUC of 0.815 (95% CI: 0.773, 0.857), and it was significantly superior to the RADNF model by our team (P = 0.027). Calibration curves and the Hosmer-Lemeshow test indicated good accuracy. DCA confirmed the model’s clinical net benefit, and internal validation via bootstrap resampling supported its stability.
Conclusions:
Based on its favorable predictive performance and accessible indicators, the NRADNF model is suitable for preliminary screening of DNF in clinical practice.
Aim:
Necrotizing fasciitis (NF) is a severe and early challenging-to-identify complication of diabetic foot (DF). This study aimed to develop and validate a novel risk assessment model for NF with DF patients utilizing conventional clinical indicators.
Methods:
A retrospective analysis was conducted on 815 DF patients admitted to the First Affiliated Hospital of Chongqing Medical University between October 2018 and April 2022. Based on the presence of NF, patients were stratified into a DF group (n = 703) and a DF complicated with NF (DNF) group (n = 112). Clinically and statistically significant variables were converted into categorical form. A new risk assessment for DNF (NRADNF) nomogram was developed via multivariable stepwise logistic regression. Model performance was evaluated using the area under the receiver operating characteristic curve (AUC) for discriminative ability, the Hosmer-Lemeshow goodness-of-fit test for calibration, decision curve analysis (DCA) for clinical utility, and bootstrap resampling for stability.
Results:
The final NRADNF model incorporated six indicators: age < 60 years, body temperature ≥ 38°C, foot skin necrosis, neutrophil-to-lymphocyte ratio (NLR) ≥ 8.5, hypersensitive C-reactive protein > 20 mg/L, and hemoglobin ≤ 100 g/L. The model demonstrated favorable predictive performance with an AUC of 0.815 (95% CI: 0.773, 0.857), and it was significantly superior to the RADNF model by our team (P = 0.027). Calibration curves and the Hosmer-Lemeshow test indicated good accuracy. DCA confirmed the model’s clinical net benefit, and internal validation via bootstrap resampling supported its stability.
Conclusions:
Based on its favorable predictive performance and accessible indicators, the NRADNF model is suitable for preliminary screening of DNF in clinical practice.
DOI: https://doi.org/10.37349/eemd.2026.101463

Aim:
In Mexico, 18.3% of patients over 20 years of age suffer from type 2 diabetes mellitus (T2DM). Visceral fat has been identified as a risk factor for developing T2DM, considering that its effect depends mainly on the amount, it has been proposed that its location also participates in the genesis of T2DM. This paper aims to analyze whether the location of fatty depots participates in the appearance of T2DM in patients with metabolic syndrome.
Methods:
A total of 101 patients with metabolic syndrome according to the criteria of the International Diabetes Federation were included. In all of them, epicardial fat (EF) was measured by echocardiography, according to the Iacobelis criteria. Preperitoneal fat (PreF), mesenteric fat (MF), and perirenal fat (PF) were also measured by ultrasound. The statistical methods used were the odds ratio (OR) and Fisher’s exact test.
Results:
We found a significant association between EF thickness (OR 8.28, 95% CI 2.16–31.70, p = 0.001) and MF (OR 4.27, 95% CI 1.63–11.13, p = 0.0037) with T2DM, 60% of patients with increased PF presented T2DM. Interestingly, no patient with a PF less than 10 mm presented with T2DM. We found no association between PreF and T2DM (OR 0.88, 95% CI 0.32–2.40).
Conclusions:
The results suggest that the location of adipose tissue plays an important role in the development of T2DM. This may be due to the type of adipocytokines secreted by the various fat deposits and their metabolic effects.
Aim:
In Mexico, 18.3% of patients over 20 years of age suffer from type 2 diabetes mellitus (T2DM). Visceral fat has been identified as a risk factor for developing T2DM, considering that its effect depends mainly on the amount, it has been proposed that its location also participates in the genesis of T2DM. This paper aims to analyze whether the location of fatty depots participates in the appearance of T2DM in patients with metabolic syndrome.
Methods:
A total of 101 patients with metabolic syndrome according to the criteria of the International Diabetes Federation were included. In all of them, epicardial fat (EF) was measured by echocardiography, according to the Iacobelis criteria. Preperitoneal fat (PreF), mesenteric fat (MF), and perirenal fat (PF) were also measured by ultrasound. The statistical methods used were the odds ratio (OR) and Fisher’s exact test.
Results:
We found a significant association between EF thickness (OR 8.28, 95% CI 2.16–31.70, p = 0.001) and MF (OR 4.27, 95% CI 1.63–11.13, p = 0.0037) with T2DM, 60% of patients with increased PF presented T2DM. Interestingly, no patient with a PF less than 10 mm presented with T2DM. We found no association between PreF and T2DM (OR 0.88, 95% CI 0.32–2.40).
Conclusions:
The results suggest that the location of adipose tissue plays an important role in the development of T2DM. This may be due to the type of adipocytokines secreted by the various fat deposits and their metabolic effects.
DOI: https://doi.org/10.37349/eemd.2026.101462
This article belongs to the special issue Current Views on Pathogenesis, Diagnosis and Management of Type 2 Diabetes Mellitus and Its Complications and Related Conditions

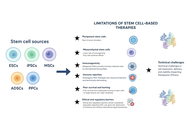
Diabetes and metabolic dysfunction represent growing global health challenges, and current therapies mitigate hyperglycemia without sufficiently altering the underlying disease processes. Rapid advances in regenerative medicine, gene editing, and nano-enabled drug delivery are reshaping therapeutic possibilities, offering the potential to restore β-cell function, enhance insulin sensitivity, and personalize care at an unprecedented scale. This review synthesizes emerging therapeutic strategies with the greatest translational promise, including stem cell-derived islet replacement, immune-evasive encapsulation devices, CRISPR-based gene correction, and targeted or glucose-responsive nanocarriers engineered for noninvasive insulin delivery. Complementary advances in multi-omics profiling, proteogenomics, microbiome science, and artificial intelligence are enabling stratification of patients based on molecular signatures, optimizing therapeutic selection, and predicting clinical outcomes. Despite remarkable breakthroughs—such as the early-phase clinical success of stem cell-derived β-cell replacement and organ-targeted lipid nanoparticles (LNPs)—significant barriers remain, including immunogenicity, off-target editing, scalability, ethical concerns, and long-term safety. By integrating mechanistic insights with translational developments, this review outlines a forward-looking perspective on next-generation treatments poised to shift diabetes care from glycemic management toward disease modification and personalized metabolic restoration.
Diabetes and metabolic dysfunction represent growing global health challenges, and current therapies mitigate hyperglycemia without sufficiently altering the underlying disease processes. Rapid advances in regenerative medicine, gene editing, and nano-enabled drug delivery are reshaping therapeutic possibilities, offering the potential to restore β-cell function, enhance insulin sensitivity, and personalize care at an unprecedented scale. This review synthesizes emerging therapeutic strategies with the greatest translational promise, including stem cell-derived islet replacement, immune-evasive encapsulation devices, CRISPR-based gene correction, and targeted or glucose-responsive nanocarriers engineered for noninvasive insulin delivery. Complementary advances in multi-omics profiling, proteogenomics, microbiome science, and artificial intelligence are enabling stratification of patients based on molecular signatures, optimizing therapeutic selection, and predicting clinical outcomes. Despite remarkable breakthroughs—such as the early-phase clinical success of stem cell-derived β-cell replacement and organ-targeted lipid nanoparticles (LNPs)—significant barriers remain, including immunogenicity, off-target editing, scalability, ethical concerns, and long-term safety. By integrating mechanistic insights with translational developments, this review outlines a forward-looking perspective on next-generation treatments poised to shift diabetes care from glycemic management toward disease modification and personalized metabolic restoration.
DOI: https://doi.org/10.37349/eemd.2026.101461
This article belongs to the special issue Innovative Strategies for Diabetes and Metabolic Disorders: Current and Future Directions

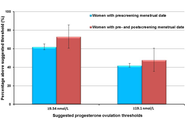
The dominant paradigm for healthy ageing in women+ (all genders) focuses on estrogen and sees the menopause, per se, as a major health problem (with low estrogen and progesterone levels). In reality, the risks for diseases that increase at older ages originate during the menstruating years. Rarely discussed evidence supports the central role of progesterone and normally ovulatory menstrual cycles in preventing early cardiovascular disease, fragility fractures, dementia, and cancers. Menstrual cycles with normal and predictable lengths but disturbed ovulation, including short luteal phases with lower progesterone production as well as anovulation without progesterone, likely occur in over 25% of all such cycles. These Subclinical Ovulatory Disturbances are usually an adaptive and protective response to physiological, sociocultural, or emotional stressors. Ovulatory disturbances and risks for health issues during ageing are intrinsically related to the social determinants of health—wholesome food, plentiful physical activity, strong communities, and access to timely and appropriate medical care. This review discusses the empirical evidence that normal ovulation and progesterone production during the premenopausal years lead to the prevention of early heart attacks and fragility fractures. Few studies document the effects of prevalent Subclinical Ovulatory Disturbances on brain issues (sleep, night sweats, ischemic strokes, pain, and addictions) and cancer risks. Serious gaps in women+’s fundamental reproductive physiology must be addressed with unbiased (population-based), rigorously collected longitudinal physiological, hormonal, and sociocultural data. Progesterone therapy during perimenopause and menopause also indirectly leads to healthy ageing through effective treatment of night sweats, hot flushes, and disturbed sleep, which are associated with cardiovascular problems and osteoporosis. Not only is progesterone effective for vasomotor symptoms in menopause, but also effective in perimenopause, a time of high and chaotic estrogen levels. In sum, strong summarized evidence suggests that progesterone and ovulation need further exploration for their important roles in promoting healthy ageing for women+.
The dominant paradigm for healthy ageing in women+ (all genders) focuses on estrogen and sees the menopause, per se, as a major health problem (with low estrogen and progesterone levels). In reality, the risks for diseases that increase at older ages originate during the menstruating years. Rarely discussed evidence supports the central role of progesterone and normally ovulatory menstrual cycles in preventing early cardiovascular disease, fragility fractures, dementia, and cancers. Menstrual cycles with normal and predictable lengths but disturbed ovulation, including short luteal phases with lower progesterone production as well as anovulation without progesterone, likely occur in over 25% of all such cycles. These Subclinical Ovulatory Disturbances are usually an adaptive and protective response to physiological, sociocultural, or emotional stressors. Ovulatory disturbances and risks for health issues during ageing are intrinsically related to the social determinants of health—wholesome food, plentiful physical activity, strong communities, and access to timely and appropriate medical care. This review discusses the empirical evidence that normal ovulation and progesterone production during the premenopausal years lead to the prevention of early heart attacks and fragility fractures. Few studies document the effects of prevalent Subclinical Ovulatory Disturbances on brain issues (sleep, night sweats, ischemic strokes, pain, and addictions) and cancer risks. Serious gaps in women+’s fundamental reproductive physiology must be addressed with unbiased (population-based), rigorously collected longitudinal physiological, hormonal, and sociocultural data. Progesterone therapy during perimenopause and menopause also indirectly leads to healthy ageing through effective treatment of night sweats, hot flushes, and disturbed sleep, which are associated with cardiovascular problems and osteoporosis. Not only is progesterone effective for vasomotor symptoms in menopause, but also effective in perimenopause, a time of high and chaotic estrogen levels. In sum, strong summarized evidence suggests that progesterone and ovulation need further exploration for their important roles in promoting healthy ageing for women+.
DOI: https://doi.org/10.37349/eemd.2026.101460
This article belongs to the special issue The Fountain of Youth: Decoding the Hormonal Regulation of Aging

Aim:
Hypertriglyceridemia is linked to increased risk of diabetes diagnosis, incidence, and mortality. However, whether individuals with normal triglyceride levels (i.e., < 1.7 mmol/L) uniformly exhibit low diabetes risk remains underexplored. Specifically, it is unclear whether triglyceride levels within the normal range are associated with plasma glucose levels and the prevalence of type 2 diabetes (T2DM). This study aimed to address these gaps by examining the associations between triglyceride levels and fasting plasma glucose, as well as between triglyceride levels and T2DM, in individuals with triglycerides in the normal range.
Methods:
This cross-sectional study included 16,706 Chinese adults with triglyceride levels below 1.7 mmol/L. Among them, 1,067 had T2DM. Associations between triglyceride levels and fasting plasma glucose were assessed using linear regression, while associations with T2DM were evaluated using binary logistic regression. The optimal triglyceride cut-off for T2DM diagnosis was determined via receiver operating characteristic (ROC) curve analysis.
Results:
Triglyceride levels were positively associated with fasting plasma glucose after multivariate adjustment (β = 0.034, P < 0.001). A one-unit increase in the natural log of triglyceride levels was associated with a 61% higher adjusted odds of T2DM [odds ratio (OR), 1.61; 95% confidence interval (CI), 1.19–2.17; P = 0.002]. The optimal triglyceride cut-off for T2DM diagnosis was 1.09 mmol/L. Participants with triglyceride levels ≥ 1.09 mmol/L had a 28% higher odds of T2DM (OR, 1.28; 95% CI, 1.07–1.53; P = 0.006) compared to those with levels below the cut-off.
Conclusions:
Among individuals with normal triglyceride levels, higher triglyceride concentrations were associated with higher odds of T2DM diagnosis, with an optimal diagnostic cut-off of 1.09 mmol/L. These findings suggest that adults with triglyceride levels more than 1.09 mmol/L may benefit from closer monitoring for T2DM development.
Aim:
Hypertriglyceridemia is linked to increased risk of diabetes diagnosis, incidence, and mortality. However, whether individuals with normal triglyceride levels (i.e., < 1.7 mmol/L) uniformly exhibit low diabetes risk remains underexplored. Specifically, it is unclear whether triglyceride levels within the normal range are associated with plasma glucose levels and the prevalence of type 2 diabetes (T2DM). This study aimed to address these gaps by examining the associations between triglyceride levels and fasting plasma glucose, as well as between triglyceride levels and T2DM, in individuals with triglycerides in the normal range.
Methods:
This cross-sectional study included 16,706 Chinese adults with triglyceride levels below 1.7 mmol/L. Among them, 1,067 had T2DM. Associations between triglyceride levels and fasting plasma glucose were assessed using linear regression, while associations with T2DM were evaluated using binary logistic regression. The optimal triglyceride cut-off for T2DM diagnosis was determined via receiver operating characteristic (ROC) curve analysis.
Results:
Triglyceride levels were positively associated with fasting plasma glucose after multivariate adjustment (β = 0.034, P < 0.001). A one-unit increase in the natural log of triglyceride levels was associated with a 61% higher adjusted odds of T2DM [odds ratio (OR), 1.61; 95% confidence interval (CI), 1.19–2.17; P = 0.002]. The optimal triglyceride cut-off for T2DM diagnosis was 1.09 mmol/L. Participants with triglyceride levels ≥ 1.09 mmol/L had a 28% higher odds of T2DM (OR, 1.28; 95% CI, 1.07–1.53; P = 0.006) compared to those with levels below the cut-off.
Conclusions:
Among individuals with normal triglyceride levels, higher triglyceride concentrations were associated with higher odds of T2DM diagnosis, with an optimal diagnostic cut-off of 1.09 mmol/L. These findings suggest that adults with triglyceride levels more than 1.09 mmol/L may benefit from closer monitoring for T2DM development.
DOI: https://doi.org/10.37349/eemd.2026.101459
This article belongs to the special issue Current Views on Pathogenesis, Diagnosis and Management of Type 2 Diabetes Mellitus and Its Complications and Related Conditions

This commentary discusses a recent article (J Diabetes 2025;17(3):e70063), focusing on interpreting the study’s sex-stratified results in a broader clinical and mechanistic context. The authors of this systematic review and meta-analysis of 14 randomized trials demonstrate that women achieve greater weight loss induced by glucagon-like peptide-1 (GLP-1) receptor agonists compared to men (mean difference of 1.04 kg or 1.69%). Analyses specific to different drugs consistently show that women benefit more from dulaglutide, liraglutide, semaglutide, and retatrutide, with trials focused on obesity further emphasizing this gap. Sensitivity analyses confirm the reliability of these findings and indicate the absence of publication bias. We discuss the clinical implications of these results, suggesting that healthcare providers should consider sex differences when counseling, monitoring, and dosing patients. We also advocate for future trials that are adequately powered and stratified by sex to evaluate factors such as adherence, adverse events, and body composition. Mechanistic hypotheses, such as sex-related pharmacokinetics, estrogen-GLP-1 synergy, and varying inflammatory responses, should be investigated further to inform precision dosing. Lastly, we recommend that regulatory agencies revisit current labeling, which claims no sex differences, as more sex-stratified evidence becomes available. It is important to acknowledge the existing heterogeneity and remaining uncertainties in this area of research.
This commentary discusses a recent article (J Diabetes 2025;17(3):e70063), focusing on interpreting the study’s sex-stratified results in a broader clinical and mechanistic context. The authors of this systematic review and meta-analysis of 14 randomized trials demonstrate that women achieve greater weight loss induced by glucagon-like peptide-1 (GLP-1) receptor agonists compared to men (mean difference of 1.04 kg or 1.69%). Analyses specific to different drugs consistently show that women benefit more from dulaglutide, liraglutide, semaglutide, and retatrutide, with trials focused on obesity further emphasizing this gap. Sensitivity analyses confirm the reliability of these findings and indicate the absence of publication bias. We discuss the clinical implications of these results, suggesting that healthcare providers should consider sex differences when counseling, monitoring, and dosing patients. We also advocate for future trials that are adequately powered and stratified by sex to evaluate factors such as adherence, adverse events, and body composition. Mechanistic hypotheses, such as sex-related pharmacokinetics, estrogen-GLP-1 synergy, and varying inflammatory responses, should be investigated further to inform precision dosing. Lastly, we recommend that regulatory agencies revisit current labeling, which claims no sex differences, as more sex-stratified evidence becomes available. It is important to acknowledge the existing heterogeneity and remaining uncertainties in this area of research.
DOI: https://doi.org/10.37349/eemd.2026.101458

A pro-inflammatory state with elevated cytokines influenced by both environmental and genetic factors is a key characteristic of both type 1 diabetes (T1DM) and type 2 diabetes (T2DM). Cytokines promote immune cell infiltration and degradation of the pancreatic islets, which play a direct role in the development of insulin resistance in T1DM. Cytokines also interfere with insulin signaling pathways and lead to metabolic dysfunction, contributing to the development of insulin resistance in patients with T2DM. In this narrative review, we have discussed the mechanisms of action and specific effects on insulin resistance of different cytokines, the influence of single-nucleotide polymorphisms and genetic factors that alter cytokine levels, and the development of insulin resistance. Further, we have discussed the complication of diabetes with a focus on diabetic foot ulcers, wounds, impaired wound healing, and reduced angiogenesis in association with the role of cytokines. Finally, the discussion addresses interventions for managing cytokines, such as Treg-based therapies, along with the various challenges presented by therapies targeting cytokine dysregulation and their effects on insulin resistance.
A pro-inflammatory state with elevated cytokines influenced by both environmental and genetic factors is a key characteristic of both type 1 diabetes (T1DM) and type 2 diabetes (T2DM). Cytokines promote immune cell infiltration and degradation of the pancreatic islets, which play a direct role in the development of insulin resistance in T1DM. Cytokines also interfere with insulin signaling pathways and lead to metabolic dysfunction, contributing to the development of insulin resistance in patients with T2DM. In this narrative review, we have discussed the mechanisms of action and specific effects on insulin resistance of different cytokines, the influence of single-nucleotide polymorphisms and genetic factors that alter cytokine levels, and the development of insulin resistance. Further, we have discussed the complication of diabetes with a focus on diabetic foot ulcers, wounds, impaired wound healing, and reduced angiogenesis in association with the role of cytokines. Finally, the discussion addresses interventions for managing cytokines, such as Treg-based therapies, along with the various challenges presented by therapies targeting cytokine dysregulation and their effects on insulin resistance.
DOI: https://doi.org/10.37349/eemd.2026.101457
This article belongs to the special issue Role of Dysregulated Cytokine Signaling Pathways in Metabolic Disease

Osteoporosis is a disabling disease with a significant impact on the global population, particularly among older men and postmenopausal women. Several factors contribute to the increasing prevalence of osteoporosis, including greater life expectancy and the absence of symptoms in its early stages. The morbidity, mortality, and substantial economic burden associated with osteoporosis, especially due to hip fractures and related complications, constitute a major public health concern. Diagnosis should involve a comprehensive biochemical profile, along with additional tests to rule out secondary causes, which are often underdiagnosed and can influence the progression of the disease. Preventive measures and early diagnosis are essential to maintaining bone health and preventing fractures and disability. This review will focus on the definition, diagnostic approach, and key considerations prior to initiating treatment in patients with osteoporosis. Fracture risk prediction tools, including Fracture Risk Assessment Tool (FRAX), and treatment strategies are not addressed, as this review focuses on the appropriate diagnostic evaluation of osteoporosis and the systematic exclusion of secondary causes.
Osteoporosis is a disabling disease with a significant impact on the global population, particularly among older men and postmenopausal women. Several factors contribute to the increasing prevalence of osteoporosis, including greater life expectancy and the absence of symptoms in its early stages. The morbidity, mortality, and substantial economic burden associated with osteoporosis, especially due to hip fractures and related complications, constitute a major public health concern. Diagnosis should involve a comprehensive biochemical profile, along with additional tests to rule out secondary causes, which are often underdiagnosed and can influence the progression of the disease. Preventive measures and early diagnosis are essential to maintaining bone health and preventing fractures and disability. This review will focus on the definition, diagnostic approach, and key considerations prior to initiating treatment in patients with osteoporosis. Fracture risk prediction tools, including Fracture Risk Assessment Tool (FRAX), and treatment strategies are not addressed, as this review focuses on the appropriate diagnostic evaluation of osteoporosis and the systematic exclusion of secondary causes.
DOI: https://doi.org/10.37349/eemd.2026.101456

Background:
Metabolic syndrome and dyslipidaemia increase the risk of death by two or three times. In this context, the role of apolipoprotein A-I (Apo A-I), the main structural protein of high-density lipoprotein (HDL), stands out, since its anti-inflammatory potential reduces cardiovascular risk. Further, genetic modifications, such as the rs670 single-nucleotide polymorphism (SNP), in the promoter region of the APOA1 gene are associated with the development of cardiovascular events, dyslipidemia, and diabetes, as well as metabolic syndrome. Thus, this study aims to investigate the relation between the occurrence of dyslipidemia and the rs670 SNP genotypes.
Methods:
An integrative and systematic review was performed with the LitVar2 database according to the PRISMA protocol standards. Studies were researched up to August 2025. Then, a meta-analysis was performed using the fixed-effects model, since the study was considered homogeneous based on the I2 value (< 50%).
Results:
Of the 99 found articles in the database, 5 referred to metabolic disorders (n = 7,705—4 Chinese studies and 1 Iranian study) and were published between 2015 and 2018. Three (n = 2,784 patients or 36.13%) of the articles indicated an association between the polymorphic allele and a higher risk of developing dyslipidemia with a relative risk of 1.16 (IC 95% 1.09–1.23, p < 0.01, I2 = 0%). Relative risk (IC 95%) was presented, and p < 0.05 was defined as the significance criterion.
Discussion:
This study reinforces a possible association between the influence of SNP rs670 and dyslipidemia. This emphasizes the importance of conducting further research incorporating a larger and more diverse study group, as well as investigating the genetic and environmental influence on the phenotypic expression of the rs670 SNP.
Background:
Metabolic syndrome and dyslipidaemia increase the risk of death by two or three times. In this context, the role of apolipoprotein A-I (Apo A-I), the main structural protein of high-density lipoprotein (HDL), stands out, since its anti-inflammatory potential reduces cardiovascular risk. Further, genetic modifications, such as the rs670 single-nucleotide polymorphism (SNP), in the promoter region of the APOA1 gene are associated with the development of cardiovascular events, dyslipidemia, and diabetes, as well as metabolic syndrome. Thus, this study aims to investigate the relation between the occurrence of dyslipidemia and the rs670 SNP genotypes.
Methods:
An integrative and systematic review was performed with the LitVar2 database according to the PRISMA protocol standards. Studies were researched up to August 2025. Then, a meta-analysis was performed using the fixed-effects model, since the study was considered homogeneous based on the I2 value (< 50%).
Results:
Of the 99 found articles in the database, 5 referred to metabolic disorders (n = 7,705—4 Chinese studies and 1 Iranian study) and were published between 2015 and 2018. Three (n = 2,784 patients or 36.13%) of the articles indicated an association between the polymorphic allele and a higher risk of developing dyslipidemia with a relative risk of 1.16 (IC 95% 1.09–1.23, p < 0.01, I2 = 0%). Relative risk (IC 95%) was presented, and p < 0.05 was defined as the significance criterion.
Discussion:
This study reinforces a possible association between the influence of SNP rs670 and dyslipidemia. This emphasizes the importance of conducting further research incorporating a larger and more diverse study group, as well as investigating the genetic and environmental influence on the phenotypic expression of the rs670 SNP.
DOI: https://doi.org/10.37349/eemd.2026.101455
 Previous
Previous