
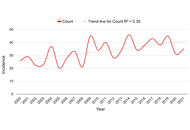






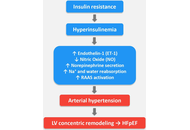


Cardiovascular disease (CVD) is the leading cause of mortality in women worldwide. While increasing parity has been associated with greater CVD risk in several populations, limited data exist on this association in South Asian women who experience some of the highest fertility rates globally. This narrative review synthesizes current literature examining the relationship between multiparity and CVD in South Asian women, including epidemiologic patterns, proposed biological mechanisms, and the influence of sociocultural factors. Evidence from South Asia suggests a possible association between high parity (particularly ≥ 4 or 5 births) and increased risk of hypertension, obesity, metabolic syndrome, and coronary heart disease. However, the available data are limited, largely cross-sectional, and occasionally contradictory. Some studies found no association or even protective effects at lower parity levels, suggesting a potential threshold or nonlinear effect. Biologically, proposed mechanisms include insulin resistance, endothelial dysfunction, and dysregulation of adipokines. Sociocultural factors such as male child preference, restricted contraceptive access, and limited autonomy in family planning decisions may also contribute to high parity and indirectly affect cardiovascular health. Although global research supports a positive association between multiparity and CVD, the evidence specific to South Asian populations remains inconsistent and underexplored. Further region-specific, longitudinal research is essential to clarify causality and inform culturally tailored screening and prevention strategies.
Cardiovascular disease (CVD) is the leading cause of mortality in women worldwide. While increasing parity has been associated with greater CVD risk in several populations, limited data exist on this association in South Asian women who experience some of the highest fertility rates globally. This narrative review synthesizes current literature examining the relationship between multiparity and CVD in South Asian women, including epidemiologic patterns, proposed biological mechanisms, and the influence of sociocultural factors. Evidence from South Asia suggests a possible association between high parity (particularly ≥ 4 or 5 births) and increased risk of hypertension, obesity, metabolic syndrome, and coronary heart disease. However, the available data are limited, largely cross-sectional, and occasionally contradictory. Some studies found no association or even protective effects at lower parity levels, suggesting a potential threshold or nonlinear effect. Biologically, proposed mechanisms include insulin resistance, endothelial dysfunction, and dysregulation of adipokines. Sociocultural factors such as male child preference, restricted contraceptive access, and limited autonomy in family planning decisions may also contribute to high parity and indirectly affect cardiovascular health. Although global research supports a positive association between multiparity and CVD, the evidence specific to South Asian populations remains inconsistent and underexplored. Further region-specific, longitudinal research is essential to clarify causality and inform culturally tailored screening and prevention strategies.
DOI: https://doi.org/10.37349/ec.2026.1012108
This article belongs to the special issue Cardiovascular Risk for Mothers and Offspring Resulting from Complicated Pregnancy

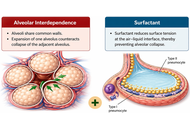
Cardiopulmonary interaction is a fundamental physiological process during spontaneous breathing, but it is profoundly altered in critically ill patients receiving mechanical ventilation (MV). Positive-pressure ventilation modifies intrathoracic, pleural, and transpulmonary pressures, with major effects on pulmonary vascular hemodynamics and right ventricular performance. Among these consequences, acute pulmonary hypertension (aPH) has emerged as a clinically relevant yet frequently underrecognized complication. This review summarizes the current evidence on cardiopulmonary interaction during spontaneous breathing and MV, with particular emphasis on the mechanisms driving aPH and right ventricular dysfunction in critically ill patients. A narrative review was performed using PubMed, Embase, Scopus, Web of Science, and the Cochrane Library. Free-text terms and controlled vocabulary related to positive-pressure ventilation, right ventricular dysfunction, pulmonary hypertension (PH), pulmonary vascular resistance (PVR), right heart catheterization, intensive care, and respiratory compliance were combined using Boolean operators. Priority was given to studies involving adult patients, including systematic reviews, observational studies, clinical trials, and relevant reference lists. During spontaneous breathing, cardiopulmonary interaction is governed by negative intrathoracic pressure, venous return (VR), transpulmonary pressure, and physiological ventilation-perfusion relationships. In contrast, MV reverses this physiological pressure profile and may reduce VR, increase right ventricular afterload, impair ventricular interdependence, and increase PVR. High tidal volumes, excessive positive end-expiratory pressure, increased plateau pressure, hypercapnia, hypoxemia, alveolar overdistension, and diffuse lung injury all contribute to aPH, potentially disrupting right ventricle-pulmonary artery coupling and promoting right ventricular dysfunction. MV profoundly reshapes cardiopulmonary physiology and may precipitate aPH and right ventricular dysfunction. Early recognition of these mechanisms and the application of protective ventilatory strategies are essential to reduce pulmonary and hemodynamic complications.
Cardiopulmonary interaction is a fundamental physiological process during spontaneous breathing, but it is profoundly altered in critically ill patients receiving mechanical ventilation (MV). Positive-pressure ventilation modifies intrathoracic, pleural, and transpulmonary pressures, with major effects on pulmonary vascular hemodynamics and right ventricular performance. Among these consequences, acute pulmonary hypertension (aPH) has emerged as a clinically relevant yet frequently underrecognized complication. This review summarizes the current evidence on cardiopulmonary interaction during spontaneous breathing and MV, with particular emphasis on the mechanisms driving aPH and right ventricular dysfunction in critically ill patients. A narrative review was performed using PubMed, Embase, Scopus, Web of Science, and the Cochrane Library. Free-text terms and controlled vocabulary related to positive-pressure ventilation, right ventricular dysfunction, pulmonary hypertension (PH), pulmonary vascular resistance (PVR), right heart catheterization, intensive care, and respiratory compliance were combined using Boolean operators. Priority was given to studies involving adult patients, including systematic reviews, observational studies, clinical trials, and relevant reference lists. During spontaneous breathing, cardiopulmonary interaction is governed by negative intrathoracic pressure, venous return (VR), transpulmonary pressure, and physiological ventilation-perfusion relationships. In contrast, MV reverses this physiological pressure profile and may reduce VR, increase right ventricular afterload, impair ventricular interdependence, and increase PVR. High tidal volumes, excessive positive end-expiratory pressure, increased plateau pressure, hypercapnia, hypoxemia, alveolar overdistension, and diffuse lung injury all contribute to aPH, potentially disrupting right ventricle-pulmonary artery coupling and promoting right ventricular dysfunction. MV profoundly reshapes cardiopulmonary physiology and may precipitate aPH and right ventricular dysfunction. Early recognition of these mechanisms and the application of protective ventilatory strategies are essential to reduce pulmonary and hemodynamic complications.
DOI: https://doi.org/10.37349/ec.2026.1012109

Aim:
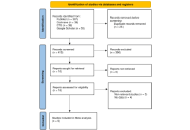
This study aims to evaluate how demographic and treatment variables, including age, tumor type, resectability, and metastasis, affect survival outcomes across prognostic subgroups of primary malignant cardiac tumors (PMCTs).
Methods:
PMCT cases diagnosed between 2000 and 2021 were identified from Surveillance, Epidemiology, and End Results (SEER) 18 registries. 730 patients were analyzed and categorized into prognosis groups based on 5-year survival (< 50%, 50–95%, > 95%). Demographic, histologic, and treatment varieties were examined using descriptive statistics, Kaplan-Meier estimates, and Cox proportional hazards models.
Results:
Among 730 patients, most were middle-aged (56.6%), male (52.5%), and White (60.1%). Soft tissue sarcomas predominated in the poorest-prognosis group (80.3%), while hematologic malignancies were most common in intermediate prognosis (52.0%). Younger age significantly reduced mortality risk [hazard ratio (HR) 0.49–0.52; p < 0.01]. Brain and lung metastases increased mortality (HR 2.04 and 1.89; p < 0.05). Surgical resection improved survival in sarcoma-dominant tumors, while systemic metastasis predicted poorer outcomes in hematologic malignancies. Chemotherapy improved survival in sarcomas (ρ = 0.457; p < 0.0001) but was associated with poorer outcomes in hematologic cancers (ρ = −0.337; p < 0.0001).
Conclusions:
PMCT prognosis is primarily influenced by histologic subtype and resectability. Sarcoma-dominant tumors benefit from aggressive surgical and chemotherapeutic management, conversely systemic control is key for hematologic malignancies. Younger age and absence of metastasis consistently predict better outcomes. Histology-specific and early intervention strategies are critical to improving survival in this rare malignancy.
Aim:
This study aims to evaluate how demographic and treatment variables, including age, tumor type, resectability, and metastasis, affect survival outcomes across prognostic subgroups of primary malignant cardiac tumors (PMCTs).
Methods:
PMCT cases diagnosed between 2000 and 2021 were identified from Surveillance, Epidemiology, and End Results (SEER) 18 registries. 730 patients were analyzed and categorized into prognosis groups based on 5-year survival (< 50%, 50–95%, > 95%). Demographic, histologic, and treatment varieties were examined using descriptive statistics, Kaplan-Meier estimates, and Cox proportional hazards models.
Results:
Among 730 patients, most were middle-aged (56.6%), male (52.5%), and White (60.1%). Soft tissue sarcomas predominated in the poorest-prognosis group (80.3%), while hematologic malignancies were most common in intermediate prognosis (52.0%). Younger age significantly reduced mortality risk [hazard ratio (HR) 0.49–0.52; p < 0.01]. Brain and lung metastases increased mortality (HR 2.04 and 1.89; p < 0.05). Surgical resection improved survival in sarcoma-dominant tumors, while systemic metastasis predicted poorer outcomes in hematologic malignancies. Chemotherapy improved survival in sarcomas (ρ = 0.457; p < 0.0001) but was associated with poorer outcomes in hematologic cancers (ρ = −0.337; p < 0.0001).
Conclusions:
PMCT prognosis is primarily influenced by histologic subtype and resectability. Sarcoma-dominant tumors benefit from aggressive surgical and chemotherapeutic management, conversely systemic control is key for hematologic malignancies. Younger age and absence of metastasis consistently predict better outcomes. Histology-specific and early intervention strategies are critical to improving survival in this rare malignancy.
DOI: https://doi.org/10.37349/ec.2026.1012107

Diabetes mellitus significantly increases the burden of coronary artery disease and peripheral arterial disease, frequently necessitating vascular bypass surgery. Imaging plays a central role across the perioperative continuum, from preoperative risk stratification and surgical planning to intraoperative guidance and postoperative surveillance. This narrative review synthesizes current evidence on the application of multimodality imaging in diabetic patients undergoing coronary artery bypass grafting and peripheral arterial bypass procedures. A structured literature search of major databases was undertaken to identify contemporary studies evaluating echocardiography, computed tomography angiography, duplex ultrasound, magnetic resonance angiography, digital subtraction angiography, and functional nuclear imaging. Diabetic-specific factors such as diffuse multivessel disease, medial arterial calcification, renal impairment, and microvascular dysfunction substantially influence imaging performance and modality selection. While noninvasive imaging increasingly guides surgical decision-making and follow-up, the available evidence remains heterogeneous and largely observational. Advanced and hybrid imaging techniques show promise for individualized care but are limited by cost, availability, and a lack of robust outcome-driven data. Standardized imaging pathways and higher-quality comparative studies are required to optimize perioperative imaging strategies in this high-risk population.
Diabetes mellitus significantly increases the burden of coronary artery disease and peripheral arterial disease, frequently necessitating vascular bypass surgery. Imaging plays a central role across the perioperative continuum, from preoperative risk stratification and surgical planning to intraoperative guidance and postoperative surveillance. This narrative review synthesizes current evidence on the application of multimodality imaging in diabetic patients undergoing coronary artery bypass grafting and peripheral arterial bypass procedures. A structured literature search of major databases was undertaken to identify contemporary studies evaluating echocardiography, computed tomography angiography, duplex ultrasound, magnetic resonance angiography, digital subtraction angiography, and functional nuclear imaging. Diabetic-specific factors such as diffuse multivessel disease, medial arterial calcification, renal impairment, and microvascular dysfunction substantially influence imaging performance and modality selection. While noninvasive imaging increasingly guides surgical decision-making and follow-up, the available evidence remains heterogeneous and largely observational. Advanced and hybrid imaging techniques show promise for individualized care but are limited by cost, availability, and a lack of robust outcome-driven data. Standardized imaging pathways and higher-quality comparative studies are required to optimize perioperative imaging strategies in this high-risk population.
DOI: https://doi.org/10.37349/ec.2026.1012106

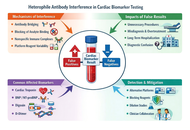
High-sensitivity cardiac biomarkers have transformed modern cardiology by enabling earlier diagnosis and refined risk stratification in acute coronary syndromes and heart failure. However, increasing analytical sensitivity has also amplified the clinical impact of immunoassay interferences, particularly those caused by heterophile antibodies. These endogenous antibodies can interact with assay antibodies and generate false-positive or false-negative results, most notably in sandwich immunoassays used for cardiac troponins and natriuretic peptides. Persistent, clinically implausible biomarker elevations related to heterophile antibody interference have led to unnecessary invasive procedures, inappropriate pharmacologic treatment, repeated hospital admissions, and prolonged diagnostic uncertainty. Conversely, false-negative results may delay diagnosis, result in inappropriate discharge, and contribute to adverse clinical outcomes in high-risk patients. This review summarizes the immunological basis and analytical mechanisms of heterophile antibody interference, highlights cardiac biomarkers most commonly affected, and outlines key clinical red flags that should prompt suspicion. Practical laboratory strategies include polyethylene glycol precipitation as an initial approach to detect antibody-mediated interference, followed by dilution studies, heterophile blocking reagents, and cautiously interpreted alternative platform testing within assay-specific reference frameworks, together with close clinician-laboratory collaboration. Greater awareness of analytical interference is critical as cardiology becomes increasingly biomarker-driven. Integrating clinical judgment with structured laboratory verification can substantially reduce diagnostic error, improve patient safety, and prevent avoidable healthcare utilization in contemporary cardiovascular practice.
High-sensitivity cardiac biomarkers have transformed modern cardiology by enabling earlier diagnosis and refined risk stratification in acute coronary syndromes and heart failure. However, increasing analytical sensitivity has also amplified the clinical impact of immunoassay interferences, particularly those caused by heterophile antibodies. These endogenous antibodies can interact with assay antibodies and generate false-positive or false-negative results, most notably in sandwich immunoassays used for cardiac troponins and natriuretic peptides. Persistent, clinically implausible biomarker elevations related to heterophile antibody interference have led to unnecessary invasive procedures, inappropriate pharmacologic treatment, repeated hospital admissions, and prolonged diagnostic uncertainty. Conversely, false-negative results may delay diagnosis, result in inappropriate discharge, and contribute to adverse clinical outcomes in high-risk patients. This review summarizes the immunological basis and analytical mechanisms of heterophile antibody interference, highlights cardiac biomarkers most commonly affected, and outlines key clinical red flags that should prompt suspicion. Practical laboratory strategies include polyethylene glycol precipitation as an initial approach to detect antibody-mediated interference, followed by dilution studies, heterophile blocking reagents, and cautiously interpreted alternative platform testing within assay-specific reference frameworks, together with close clinician-laboratory collaboration. Greater awareness of analytical interference is critical as cardiology becomes increasingly biomarker-driven. Integrating clinical judgment with structured laboratory verification can substantially reduce diagnostic error, improve patient safety, and prevent avoidable healthcare utilization in contemporary cardiovascular practice.
DOI: https://doi.org/10.37349/ec.2026.1012105

Implantable cardioverter defibrillators (ICDs) play a central role in the prevention of sudden cardiac death. Although implantation is generally safe, rare but clinically significant complications may occur. One such complication is inadvertent placement of a transvenous ICD lead into the left ventricle via a patent foramen ovale (PFO), which carries a risk of systemic thromboembolism and may remain clinically silent for prolonged periods. We report a case of an ICD lead malpositioned in the left ventricle through a PFO, detected 17 months after implantation during transthoracic echocardiography performed for worsening dyspnoea. Imaging demonstrated the lead traversing the interatrial septum and mitral valve into the left ventricle. The patient underwent successful transvenous extraction and reimplantation of a new right ventricular lead without complications. This case highlights the limitations of electrocardiographic clues alone and underscores the importance of multimodality imaging and meticulous implantation technique. A focused narrative review integrates current literature regarding mechanisms, diagnostic strategies, management options, and preventive considerations.
Implantable cardioverter defibrillators (ICDs) play a central role in the prevention of sudden cardiac death. Although implantation is generally safe, rare but clinically significant complications may occur. One such complication is inadvertent placement of a transvenous ICD lead into the left ventricle via a patent foramen ovale (PFO), which carries a risk of systemic thromboembolism and may remain clinically silent for prolonged periods. We report a case of an ICD lead malpositioned in the left ventricle through a PFO, detected 17 months after implantation during transthoracic echocardiography performed for worsening dyspnoea. Imaging demonstrated the lead traversing the interatrial septum and mitral valve into the left ventricle. The patient underwent successful transvenous extraction and reimplantation of a new right ventricular lead without complications. This case highlights the limitations of electrocardiographic clues alone and underscores the importance of multimodality imaging and meticulous implantation technique. A focused narrative review integrates current literature regarding mechanisms, diagnostic strategies, management options, and preventive considerations.
DOI: https://doi.org/10.37349/ec.2026.1012104

Coronary vasospasm, affecting both epicardial arteries and the coronary microcirculation, is a significant yet frequently underdiagnosed and undertreated cause of coronary syndromes. When promptly identified, it carries a relatively benign prognosis. Recognition can be straightforward in non-cardiology settings when triggered by known spasmogenic agents such as misoprostol (obstetrics) or 5-fluorouracil (oncology). Vasospasm may also be incidentally revealed during noninvasive functional testing, typically presenting as ST-segment elevation during early recovery phases, or after administration of agents like aminophylline following dipyridamole or β-blockers following dobutamine. In patients with high clinical suspicion but negative Holter or stress test findings, targeted provocation with ergonovine or hyperventilation protocols can safely induce vasospasm and unmask regional wall motion abnormalities, indicating epicardial involvement. Hyperventilation-Doppler echocardiography enables the detection of microvascular dysfunction through reductions in coronary flow velocity in the mid-distal left anterior descending coronary artery. A multi-stress, multi-marker functional testing approach offers a noninvasive, safe, and effective diagnostic strategy. Inducible wall motion abnormalities are specific for epicardial spasm, while Doppler-detected flow reduction is more sensitive for microvascular dysfunction. Early diagnosis is essential, as coronary vasospasm, though potentially life-threatening, is highly manageable with appropriate therapy. Management of patients with proven epicardial coronary artery or microvascular vasospasm involves starting therapy with calcium channel blockers and nitrates, and avoiding β-blockers, as they can worsen vasospasm by blocking β2-mediated vasodilation and leaving α1-mediated vasoconstriction unopposed.
Coronary vasospasm, affecting both epicardial arteries and the coronary microcirculation, is a significant yet frequently underdiagnosed and undertreated cause of coronary syndromes. When promptly identified, it carries a relatively benign prognosis. Recognition can be straightforward in non-cardiology settings when triggered by known spasmogenic agents such as misoprostol (obstetrics) or 5-fluorouracil (oncology). Vasospasm may also be incidentally revealed during noninvasive functional testing, typically presenting as ST-segment elevation during early recovery phases, or after administration of agents like aminophylline following dipyridamole or β-blockers following dobutamine. In patients with high clinical suspicion but negative Holter or stress test findings, targeted provocation with ergonovine or hyperventilation protocols can safely induce vasospasm and unmask regional wall motion abnormalities, indicating epicardial involvement. Hyperventilation-Doppler echocardiography enables the detection of microvascular dysfunction through reductions in coronary flow velocity in the mid-distal left anterior descending coronary artery. A multi-stress, multi-marker functional testing approach offers a noninvasive, safe, and effective diagnostic strategy. Inducible wall motion abnormalities are specific for epicardial spasm, while Doppler-detected flow reduction is more sensitive for microvascular dysfunction. Early diagnosis is essential, as coronary vasospasm, though potentially life-threatening, is highly manageable with appropriate therapy. Management of patients with proven epicardial coronary artery or microvascular vasospasm involves starting therapy with calcium channel blockers and nitrates, and avoiding β-blockers, as they can worsen vasospasm by blocking β2-mediated vasodilation and leaving α1-mediated vasoconstriction unopposed.
DOI: https://doi.org/10.37349/ec.2026.1012103

Aim:
Cardiac memory is classically expressed as persistent T wave changes following restoration of normal ventricular activation. Its manifestations in idiopathic intermittent left bundle branch block (ILBBB) remain incompletely characterized. The aim of this report is to describe a distinct electrocardiographic phenotype potentially related to cardiac memory.
Methods:
We retrospectively analyzed a homogeneous series of 14 patients with idiopathic, rate-dependent ILBBB, absence of demonstrable structural heart disease, homophasic T waves in leads V5–V6 during left bundle branch block (LBBB), and pseudo-ischemic T wave inversion in the right precordial leads during phases of normal conduction.
Results:
In all patients, memory-related negative T waves were observed during normal conduction, while concordant (homophasic) T waves were consistently present during LBBB. A close relationship emerged between the width of the intraventricular conduction zone and the severity of repolarization abnormalities: narrower conduction zones were associated with deeper T wave inversion during normal conduction. During follow-up, most patients progressed to permanent LBBB, without documented major arrhythmic events.
Conclusions:
These findings describe a distinctive and reproducible electrocardiographic phenotype within idiopathic ILBBB, likely related to cardiac memory. This pattern, provisionally designated homophasic idiopathic intermittent left bundle branch block and cardiac memory (OIL-CAME), warrants further confirmation in larger prospective studies.
Aim:
Cardiac memory is classically expressed as persistent T wave changes following restoration of normal ventricular activation. Its manifestations in idiopathic intermittent left bundle branch block (ILBBB) remain incompletely characterized. The aim of this report is to describe a distinct electrocardiographic phenotype potentially related to cardiac memory.
Methods:
We retrospectively analyzed a homogeneous series of 14 patients with idiopathic, rate-dependent ILBBB, absence of demonstrable structural heart disease, homophasic T waves in leads V5–V6 during left bundle branch block (LBBB), and pseudo-ischemic T wave inversion in the right precordial leads during phases of normal conduction.
Results:
In all patients, memory-related negative T waves were observed during normal conduction, while concordant (homophasic) T waves were consistently present during LBBB. A close relationship emerged between the width of the intraventricular conduction zone and the severity of repolarization abnormalities: narrower conduction zones were associated with deeper T wave inversion during normal conduction. During follow-up, most patients progressed to permanent LBBB, without documented major arrhythmic events.
Conclusions:
These findings describe a distinctive and reproducible electrocardiographic phenotype within idiopathic ILBBB, likely related to cardiac memory. This pattern, provisionally designated homophasic idiopathic intermittent left bundle branch block and cardiac memory (OIL-CAME), warrants further confirmation in larger prospective studies.
DOI: https://doi.org/10.37349/ec.2026.1012102

Aim:
To evaluate the health literacy (HL) of patients with acute coronary syndrome (ACS) admitted to a public hospital of high complexity in the interior of Minas Gerais, Brazil, through the application of the instrument Short Assessment of Health Literacy for Portuguese Speaking Adults (SAHLPA)-18.
Methods:
Retrospective cross-sectional study, from the Good Clinical Practices (GCP) project developed in a tertiary hospital. The 175 patients with ACS were analyzed, in which the profile and the SAHLPA-18 were evaluated.
Results:
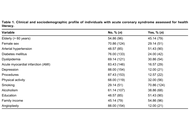
It was found that 55.43% (97 among 175; 95% CI: 48.07–62.79) were considered with inadequate HL, and 40.00% of patients who have completed elementary school or higher education had inadequate HL (36 among 90). Female sex and complete primary education or higher increased the HL, and diabetes decreased the HL.
Conclusions:
We observed low literacy even in the presence of formal education, which, combined with the presence of diabetes, may represent a risk to patients with ACS, highlighting the need for continuous health education in this group regardless of the profile.
Aim:
To evaluate the health literacy (HL) of patients with acute coronary syndrome (ACS) admitted to a public hospital of high complexity in the interior of Minas Gerais, Brazil, through the application of the instrument Short Assessment of Health Literacy for Portuguese Speaking Adults (SAHLPA)-18.
Methods:
Retrospective cross-sectional study, from the Good Clinical Practices (GCP) project developed in a tertiary hospital. The 175 patients with ACS were analyzed, in which the profile and the SAHLPA-18 were evaluated.
Results:
It was found that 55.43% (97 among 175; 95% CI: 48.07–62.79) were considered with inadequate HL, and 40.00% of patients who have completed elementary school or higher education had inadequate HL (36 among 90). Female sex and complete primary education or higher increased the HL, and diabetes decreased the HL.
Conclusions:
We observed low literacy even in the presence of formal education, which, combined with the presence of diabetes, may represent a risk to patients with ACS, highlighting the need for continuous health education in this group regardless of the profile.
DOI: https://doi.org/10.37349/ec.2026.1012101

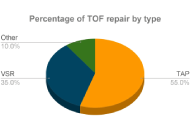
The aim is to evaluate the transannular patch (TAP) repair and valve-sparing repair (VSR) techniques following tetralogy of Fallot (TOF) correction, focusing on post-operative complications and cardiac function. A comprehensive search was performed in PubMed, EMBASE, and Scopus using relevant terms like “Tetralogy of Fallot, right ventricular outflow tract (RVOT), VSR, pulmonary valve replacement, transannular-patch repair”. Results indicated that VSR is favored due to its shorter cardiopulmonary bypass duration, preservation of the pulmonary valve, less demanding surgical requirements, shorter post-operative hospital stays, lower mortality rates, survival of at least 30 years, reduced pulmonary regurgitation, decreased right ventricular dysfunction, and improved physical activity tolerance and neurodevelopment. While TAP alleviates RVOT obstruction (RVOTO), it is associated with long-term pulmonary regurgitation. Both TAP and VSR are effective in managing TOF, but VSR provides better valve function preservation and long-term outcomes.
The aim is to evaluate the transannular patch (TAP) repair and valve-sparing repair (VSR) techniques following tetralogy of Fallot (TOF) correction, focusing on post-operative complications and cardiac function. A comprehensive search was performed in PubMed, EMBASE, and Scopus using relevant terms like “Tetralogy of Fallot, right ventricular outflow tract (RVOT), VSR, pulmonary valve replacement, transannular-patch repair”. Results indicated that VSR is favored due to its shorter cardiopulmonary bypass duration, preservation of the pulmonary valve, less demanding surgical requirements, shorter post-operative hospital stays, lower mortality rates, survival of at least 30 years, reduced pulmonary regurgitation, decreased right ventricular dysfunction, and improved physical activity tolerance and neurodevelopment. While TAP alleviates RVOT obstruction (RVOTO), it is associated with long-term pulmonary regurgitation. Both TAP and VSR are effective in managing TOF, but VSR provides better valve function preservation and long-term outcomes.
DOI: https://doi.org/10.37349/ec.2026.1012100

In the editorial game, authors are the players, reviewers are the linesmen on the sidelines, and the editor is the umpire. However, it is not uncommon for the umpire, to have a limited view of the pitch, only to be called back for revision through the video-assisted review. In the editorial context, the video-assisted review assistants are the readers, who critically evaluate the journal’s content and integrate, expand, and correct it when necessary. The journal remains open to this process, and we are well aware that only through dialectics—the comparison of differing views—can a sharper truth emerge. I described the role of the Editor-in-Chief as it is, based on my 40+ years of experience. Daly and Teixeira da Silva describe it as it should be, and this perspective is stimulating, refreshing, and timely.
In the editorial game, authors are the players, reviewers are the linesmen on the sidelines, and the editor is the umpire. However, it is not uncommon for the umpire, to have a limited view of the pitch, only to be called back for revision through the video-assisted review. In the editorial context, the video-assisted review assistants are the readers, who critically evaluate the journal’s content and integrate, expand, and correct it when necessary. The journal remains open to this process, and we are well aware that only through dialectics—the comparison of differing views—can a sharper truth emerge. I described the role of the Editor-in-Chief as it is, based on my 40+ years of experience. Daly and Teixeira da Silva describe it as it should be, and this perspective is stimulating, refreshing, and timely.
DOI: https://doi.org/10.37349/ec.2026.101299

DOI: https://doi.org/10.37349/ec.2026.101298

Infective endocarditis (IE) is a rare but potentially life-threatening condition with a wide spectrum of clinical presentations, often leading to diagnostic delay. The COVID-19 pandemic has added further complexity by overlapping clinical features and imposing constraints on diagnostic pathways. We report the case of a 48-year-old male who presented with prolonged non-specific symptoms following a recent COVID-19 infection. Multiple emergency department visits resulted in an initial diagnosis of viral illness. Subsequent clinical deterioration prompted further evaluation, which revealed severe aortic regurgitation due to a large aortic valve vegetation on transthoracic echocardiography. Blood cultures grew α-haemolytic Streptococcus mitis fulfilling the modified Duke criteria for IE. The patient developed complications, including heart failure and peripheral arterial embolisation, necessitating urgent surgical aortic valve replacement. This case highlights the diagnostic challenges of IE in the context of recent COVID-19 infection, where overlapping symptoms and altered healthcare pathways may contribute to delayed recognition. Clinicians should maintain a high index of suspicion for IE in patients presenting with persistent or atypical symptoms following COVID-19 infection. Early recognition and prompt intervention are essential to prevent serious complications.
Infective endocarditis (IE) is a rare but potentially life-threatening condition with a wide spectrum of clinical presentations, often leading to diagnostic delay. The COVID-19 pandemic has added further complexity by overlapping clinical features and imposing constraints on diagnostic pathways. We report the case of a 48-year-old male who presented with prolonged non-specific symptoms following a recent COVID-19 infection. Multiple emergency department visits resulted in an initial diagnosis of viral illness. Subsequent clinical deterioration prompted further evaluation, which revealed severe aortic regurgitation due to a large aortic valve vegetation on transthoracic echocardiography. Blood cultures grew α-haemolytic Streptococcus mitis fulfilling the modified Duke criteria for IE. The patient developed complications, including heart failure and peripheral arterial embolisation, necessitating urgent surgical aortic valve replacement. This case highlights the diagnostic challenges of IE in the context of recent COVID-19 infection, where overlapping symptoms and altered healthcare pathways may contribute to delayed recognition. Clinicians should maintain a high index of suspicion for IE in patients presenting with persistent or atypical symptoms following COVID-19 infection. Early recognition and prompt intervention are essential to prevent serious complications.
DOI: https://doi.org/10.37349/ec.2026.101297

Polycystic ovary syndrome (PCOS) is a common endocrine–metabolic condition that carries a higher cardiovascular risk than currently reflected by traditional screening tools. Emerging evidence suggests that resting tachycardia and autonomic dysfunction may serve as early, non-invasive indicators of cardiovascular dysregulation in this population. This review synthesizes current data on resting heart rate (RHR), heart rate variability (HRV), and direct autonomic markers in women with PCOS, drawing from human studies published between 2000 and 2025. Across 32 eligible studies, most reported increased sympathetic activity, reduced parasympathetic tone, elevated RHR, and impaired HRV patterns observed even in normal-weight or metabolically mild PCOS phenotypes. These alterations correlate with endothelial dysfunction, arterial stiffness, and subclinical atherosclerosis, underscoring their cardiovascular relevance. Mechanistic insights highlight the contributions of insulin resistance, hyperandrogenism, inflammation, adipokine imbalance, chemoreflex sensitization, and altered cortisol metabolism to autonomic disruption. Despite consistent findings, methodological variability in HRV protocols and inadequate adjustment for major confounders limit definitive interpretation. RHR, due to its simplicity and accessibility, including through wearable devices, holds promise as a supportive early risk signal; however, it should not be used in isolation. Future studies must adopt standardized autonomic measurements, including diverse cohorts, and evaluate whether modifying autonomic markers translates into improved cardiometabolic outcomes. Integrating RHR and HRV with metabolic and endocrine markers may enhance early cardiovascular risk stratification in women with PCOS.
Polycystic ovary syndrome (PCOS) is a common endocrine–metabolic condition that carries a higher cardiovascular risk than currently reflected by traditional screening tools. Emerging evidence suggests that resting tachycardia and autonomic dysfunction may serve as early, non-invasive indicators of cardiovascular dysregulation in this population. This review synthesizes current data on resting heart rate (RHR), heart rate variability (HRV), and direct autonomic markers in women with PCOS, drawing from human studies published between 2000 and 2025. Across 32 eligible studies, most reported increased sympathetic activity, reduced parasympathetic tone, elevated RHR, and impaired HRV patterns observed even in normal-weight or metabolically mild PCOS phenotypes. These alterations correlate with endothelial dysfunction, arterial stiffness, and subclinical atherosclerosis, underscoring their cardiovascular relevance. Mechanistic insights highlight the contributions of insulin resistance, hyperandrogenism, inflammation, adipokine imbalance, chemoreflex sensitization, and altered cortisol metabolism to autonomic disruption. Despite consistent findings, methodological variability in HRV protocols and inadequate adjustment for major confounders limit definitive interpretation. RHR, due to its simplicity and accessibility, including through wearable devices, holds promise as a supportive early risk signal; however, it should not be used in isolation. Future studies must adopt standardized autonomic measurements, including diverse cohorts, and evaluate whether modifying autonomic markers translates into improved cardiometabolic outcomes. Integrating RHR and HRV with metabolic and endocrine markers may enhance early cardiovascular risk stratification in women with PCOS.
DOI: https://doi.org/10.37349/ec.2026.101295

The Gerbode defect is characterized by a high ventricular septal defect associated with a defect in the septal leaflet of the tricuspid valve, allowing blood to enter the right atrium from the left ventricle. It accounts for approximately 0.08% of intracardiac shunts and may be congenital or acquired. We describe a rare case of Gerbode defect secondary to tricuspid valve endocarditis. A 58-year-old male patient presented with acute infective endocarditis due to Staphylococcus aureus, related to central venous access. Echocardiography showed a tricuspid valve with thickened leaflets and a small mobile image on the atrial side of the septal leaflet, as well as moderate to severe regurgitation. After completion of the antibiotic regimen with resolution of the infectious condition, the patient was discharged asymptomatic, and a new echocardiogram showed no vegetation on the tricuspid valve. During outpatient follow-up, he presented dyspnea on mild exertion, and consecutive echocardiograms showed moderate tricuspid insufficiency and significant pulmonary hypertension with a pulmonary artery systolic pressure of 83 mmHg (reference: 30 mmHg). He underwent right and left cardiac catheterization, which showed a Gerbode defect, and a transesophageal echocardiogram showed a shunt in the subaortic region measuring 6 to 8 mm, with a maximum gradient of 56 mmHg. He underwent elective surgery to correct the Gerbode defect and tricuspid valve repair, with a good clinical result. The Gerbode defect is rare, and the diagnosis can be challenging because it simulates other conditions. Treatment consists of closing the defect when it generates refractory symptoms or complications. The reported case was surgically corrected, with a good result and favorable evolution.
The Gerbode defect is characterized by a high ventricular septal defect associated with a defect in the septal leaflet of the tricuspid valve, allowing blood to enter the right atrium from the left ventricle. It accounts for approximately 0.08% of intracardiac shunts and may be congenital or acquired. We describe a rare case of Gerbode defect secondary to tricuspid valve endocarditis. A 58-year-old male patient presented with acute infective endocarditis due to Staphylococcus aureus, related to central venous access. Echocardiography showed a tricuspid valve with thickened leaflets and a small mobile image on the atrial side of the septal leaflet, as well as moderate to severe regurgitation. After completion of the antibiotic regimen with resolution of the infectious condition, the patient was discharged asymptomatic, and a new echocardiogram showed no vegetation on the tricuspid valve. During outpatient follow-up, he presented dyspnea on mild exertion, and consecutive echocardiograms showed moderate tricuspid insufficiency and significant pulmonary hypertension with a pulmonary artery systolic pressure of 83 mmHg (reference: 30 mmHg). He underwent right and left cardiac catheterization, which showed a Gerbode defect, and a transesophageal echocardiogram showed a shunt in the subaortic region measuring 6 to 8 mm, with a maximum gradient of 56 mmHg. He underwent elective surgery to correct the Gerbode defect and tricuspid valve repair, with a good clinical result. The Gerbode defect is rare, and the diagnosis can be challenging because it simulates other conditions. Treatment consists of closing the defect when it generates refractory symptoms or complications. The reported case was surgically corrected, with a good result and favorable evolution.
DOI: https://doi.org/10.37349/ec.2026.101296

Heart failure (HF) is still one of the most common causes of death today. The vast majority of heart diseases end up leading to HF, which therefore has a high prevalence in the adult population (on average 1–2%), and which increases enormously (over 10%) after the age of 65, becoming the most frequent cause of hospitalization for these subjects. It is therefore necessary to increase efforts to deepen our understanding of the pathophysiological mechanisms that lead to HF and its worsening, particularly with regard to hormonal-metabolic derangements as contributors to HF development and progression. This, in the hope of being able, in the near future, to intervene on them, reducing the prevalence of this pathology and its economic impact on countries’ healthcare spending. To this aim, we performed a narrative review of the scientific literature on the interactions between both insulin and the growth hormone/insulin-like growth factor-1 axis and the cardiovascular system, and in particular, to verify the role that these hormones may play in the development and negative progression of HF.
Heart failure (HF) is still one of the most common causes of death today. The vast majority of heart diseases end up leading to HF, which therefore has a high prevalence in the adult population (on average 1–2%), and which increases enormously (over 10%) after the age of 65, becoming the most frequent cause of hospitalization for these subjects. It is therefore necessary to increase efforts to deepen our understanding of the pathophysiological mechanisms that lead to HF and its worsening, particularly with regard to hormonal-metabolic derangements as contributors to HF development and progression. This, in the hope of being able, in the near future, to intervene on them, reducing the prevalence of this pathology and its economic impact on countries’ healthcare spending. To this aim, we performed a narrative review of the scientific literature on the interactions between both insulin and the growth hormone/insulin-like growth factor-1 axis and the cardiovascular system, and in particular, to verify the role that these hormones may play in the development and negative progression of HF.
DOI: https://doi.org/10.37349/ec.2026.101294

Background:
Heart failure (HF) remains a growing global health problem, with nearly half of all cases attributed to HF with preserved ejection fraction (HFpEF) and its precursor, left ventricular diastolic dysfunction (LVDD). Although echocardiography is the diagnostic gold standard, its high cost and limited availability restrict its use for large-scale screening. In contrast, the electrocardiogram (ECG) is inexpensive and widely accessible. Recent advances in artificial intelligence (AI) have created opportunities to leverage ECG data for the early detection of cardiac dysfunction. The objective of this study was to systematically review and meta-analyze the diagnostic performance of AI-based ECG models for detecting cardiac dysfunction.
Methods:
The QUADAS-2 tool was used to assess the risk of bias. Pooled sensitivity and specificity were estimated using a bivariate random-effects model, with heterogeneity quantified using the I2 statistic. Pre-specified subgroup analyses were conducted according to clinical endpoint and AI model type.
Results:
Following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, nine eligible studies evaluating AI algorithms applied to ECG data for the detection of HFpEF were identified. Considerable methodological and population heterogeneity was observed across studies. Risk of bias was generally low for reference standards, although concerns were noted in patient selection. The pooled specificity of AI-ECG models was high at 0.83 [95% confidence interval (CI): 0.74–0.89], while pooled sensitivity was 0.82 (95% CI: 0.70–0.90). Both estimates demonstrated extremely high heterogeneity (I2 > 96%). Subgroup analyses by endpoint and model type did not explain this variability.
Discussion:
AI-enhanced ECG models show good diagnostic accuracy, specifically in ruling out cardiac dysfunction due to their high specificity. However, the high and unexplained heterogeneity across these studies limits the immediate generalizability of the results. Large, prospective validation studies across diverse populations are essential before these models can be confidently adopted into routine clinical practice.
Background:
Heart failure (HF) remains a growing global health problem, with nearly half of all cases attributed to HF with preserved ejection fraction (HFpEF) and its precursor, left ventricular diastolic dysfunction (LVDD). Although echocardiography is the diagnostic gold standard, its high cost and limited availability restrict its use for large-scale screening. In contrast, the electrocardiogram (ECG) is inexpensive and widely accessible. Recent advances in artificial intelligence (AI) have created opportunities to leverage ECG data for the early detection of cardiac dysfunction. The objective of this study was to systematically review and meta-analyze the diagnostic performance of AI-based ECG models for detecting cardiac dysfunction.
Methods:
The QUADAS-2 tool was used to assess the risk of bias. Pooled sensitivity and specificity were estimated using a bivariate random-effects model, with heterogeneity quantified using the I2 statistic. Pre-specified subgroup analyses were conducted according to clinical endpoint and AI model type.
Results:
Following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, nine eligible studies evaluating AI algorithms applied to ECG data for the detection of HFpEF were identified. Considerable methodological and population heterogeneity was observed across studies. Risk of bias was generally low for reference standards, although concerns were noted in patient selection. The pooled specificity of AI-ECG models was high at 0.83 [95% confidence interval (CI): 0.74–0.89], while pooled sensitivity was 0.82 (95% CI: 0.70–0.90). Both estimates demonstrated extremely high heterogeneity (I2 > 96%). Subgroup analyses by endpoint and model type did not explain this variability.
Discussion:
AI-enhanced ECG models show good diagnostic accuracy, specifically in ruling out cardiac dysfunction due to their high specificity. However, the high and unexplained heterogeneity across these studies limits the immediate generalizability of the results. Large, prospective validation studies across diverse populations are essential before these models can be confidently adopted into routine clinical practice.
DOI: https://doi.org/10.37349/ec.2026.101293

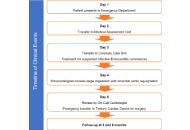
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is a rare but potentially life-threatening inherited arrhythmia disorder, often presenting in childhood or adolescence. Early and accurate diagnosis is critical, as untreated CPVT carries a high risk of sudden cardiac death, particularly in young individuals. This case underscores the importance of maintaining a high index of clinical suspicion and employing a systematic diagnostic approach. We highlight the value of integrating clinical history, family background, and targeted investigations in evaluating young adults presenting with sudden cardiac arrest. Prompt recognition and diagnosis of CPVT may be lifesaving and have significant implications for both patients and their families.
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is a rare but potentially life-threatening inherited arrhythmia disorder, often presenting in childhood or adolescence. Early and accurate diagnosis is critical, as untreated CPVT carries a high risk of sudden cardiac death, particularly in young individuals. This case underscores the importance of maintaining a high index of clinical suspicion and employing a systematic diagnostic approach. We highlight the value of integrating clinical history, family background, and targeted investigations in evaluating young adults presenting with sudden cardiac arrest. Prompt recognition and diagnosis of CPVT may be lifesaving and have significant implications for both patients and their families.
DOI: https://doi.org/10.37349/ec.2026.101292

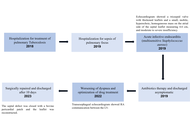
Left ventricular pseudoaneurysm is a rare acquired cardiac abnormality that frequently occurs after myocardial infarction or a previous cardiac procedure. Blunt chest trauma accounts for this uncommon entity in sporadic cases. However, this disease does not have any specific clinical findings, so it is necessary to monitor the suspected patient closely. The standard noninvasive techniques for diagnosing left ventricular pseudoaneurysms are transthoracic echocardiography and thoracic computed tomography. Untreated ventricular pseudoaneurysms carry a considerable risk of rupture, ranging from 30% to 45%. So, an urgent surgical treatment is often required. Herein, we aimed to present a 34-year-old male who underwent emergency surgery as a result of cardiac perforation three hours after a traffic accident and developed a giant left ventricular pseudoaneurysm 19 months after discharge. The giant pseudoaneurysm was successfully repaired. This case highlights the need for long‑term surveillance after blunt cardiac trauma to detect late pseudoaneurysm formation.
Left ventricular pseudoaneurysm is a rare acquired cardiac abnormality that frequently occurs after myocardial infarction or a previous cardiac procedure. Blunt chest trauma accounts for this uncommon entity in sporadic cases. However, this disease does not have any specific clinical findings, so it is necessary to monitor the suspected patient closely. The standard noninvasive techniques for diagnosing left ventricular pseudoaneurysms are transthoracic echocardiography and thoracic computed tomography. Untreated ventricular pseudoaneurysms carry a considerable risk of rupture, ranging from 30% to 45%. So, an urgent surgical treatment is often required. Herein, we aimed to present a 34-year-old male who underwent emergency surgery as a result of cardiac perforation three hours after a traffic accident and developed a giant left ventricular pseudoaneurysm 19 months after discharge. The giant pseudoaneurysm was successfully repaired. This case highlights the need for long‑term surveillance after blunt cardiac trauma to detect late pseudoaneurysm formation.
DOI: https://doi.org/10.37349/ec.2026.101291

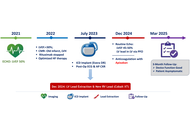
Redesigning cardiovascular services at the local level is a pressing task for decentralized health systems facing the rising burden of chronic cardiovascular disease. In northern Modena (Emilia-Romagna, Italy), a post-restructuring reorganization exposed the limits of hospital-centric models and the need for integrated, patient-centered care. In 2021, Santa Maria Bianca Hospital, Mirandola—a first-level, non-interventional facility serving a largely rural population—launched a program to build a digitally integrated, prevention-oriented cardiology network. This review distills that field experience into a scalable framework for organizing peripheral cardiovascular services. The Mirandola Cardiology Network evolved along six operational domains: (1) reactivation of the cardiology unit with community outreach; (2) expansion of outpatient services and telecardiology; (3) a day hospital platform for chronic heart failure management; (4) digital transformation of the echocardiography service; (5) development of an advanced imaging center integrating coronary computed tomography (CT) angiography and planned cardiac magnetic resonance imaging (MRI); and (6) consolidation of professional education, research, and network-wide governance. By combining digital tools, non-invasive imaging, and multidisciplinary collaboration, the model established continuity of care across inpatient, outpatient, and community settings while improving access to diagnostics and appropriateness of care. Although prospective or comparative outcomes are not presented, process indicators and implementation milestones suggest scalability and sustainability, with potential to reduce avoidable admissions and streamline clinical pathways. The Mirandola experience shows that innovation in cardiology is feasible in peripheral settings when investment in technology, governance, and training is aligned with a coherent, value-based vision. It offers actionable guidance for decentralized systems seeking to implement digitally enabled, community-focused cardiology consistent with contemporary recommendations on territorial care and chronic disease management.
Redesigning cardiovascular services at the local level is a pressing task for decentralized health systems facing the rising burden of chronic cardiovascular disease. In northern Modena (Emilia-Romagna, Italy), a post-restructuring reorganization exposed the limits of hospital-centric models and the need for integrated, patient-centered care. In 2021, Santa Maria Bianca Hospital, Mirandola—a first-level, non-interventional facility serving a largely rural population—launched a program to build a digitally integrated, prevention-oriented cardiology network. This review distills that field experience into a scalable framework for organizing peripheral cardiovascular services. The Mirandola Cardiology Network evolved along six operational domains: (1) reactivation of the cardiology unit with community outreach; (2) expansion of outpatient services and telecardiology; (3) a day hospital platform for chronic heart failure management; (4) digital transformation of the echocardiography service; (5) development of an advanced imaging center integrating coronary computed tomography (CT) angiography and planned cardiac magnetic resonance imaging (MRI); and (6) consolidation of professional education, research, and network-wide governance. By combining digital tools, non-invasive imaging, and multidisciplinary collaboration, the model established continuity of care across inpatient, outpatient, and community settings while improving access to diagnostics and appropriateness of care. Although prospective or comparative outcomes are not presented, process indicators and implementation milestones suggest scalability and sustainability, with potential to reduce avoidable admissions and streamline clinical pathways. The Mirandola experience shows that innovation in cardiology is feasible in peripheral settings when investment in technology, governance, and training is aligned with a coherent, value-based vision. It offers actionable guidance for decentralized systems seeking to implement digitally enabled, community-focused cardiology consistent with contemporary recommendations on territorial care and chronic disease management.
DOI: https://doi.org/10.37349/ec.2026.101290
 Previous
Previous