143 results in Exploration of Neuroprotective Therapy
Latest
Sort by :
- Latest
- Most Viewed
- Most Downloaded
- Most Cited
Open Access
Mini Review
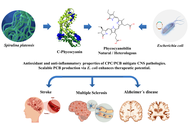
Neuroprotective potential of phycocyanobilin and C-phycocyanin: from molecular targets to translational applications
Sheyla Delgado-Lora ... Giselle Pentón-Rol
Published: April 03, 2026 Explor Neuroprot Ther. 2026;6:1004143
This article belongs to the special issue Natural Products in Neurotherapeutic Applications
Open Access
Review
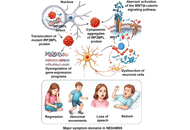
IRF2BPL mutations in rare childhood brain disorders: from genetics to pathophysiology
Mauricio Javier Sanchez-Castellanos ... Manoj Kumar Pandey
Published: April 02, 2026 Explor Neuroprot Ther. 2026;6:1004142
Open Access
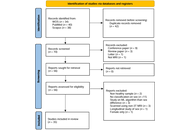
Systematic Review
Machine learning or morphometric scaling? A systematic review of methodological confounds and the generalizability of sex classification in neuroimaging
Abdul Halim Sapuan ... Sherzod Turaev
Published: March 23, 2026 Explor Neuroprot Ther. 2026;6:1004141
Open Access
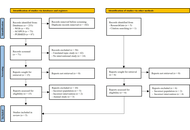
Systematic Review
Systematic review of probiotic supplementation in Alzheimer’s patients with results on cognitive function, health biomarkers, and microbiota
Diego Fernández-Lázaro ... Juan Mielgo-Ayuso
Published: March 16, 2026 Explor Neuroprot Ther. 2026;6:1004140
This article belongs to the special issue Role of Microbiota in Neurological Diseases
Open Access
Original Article
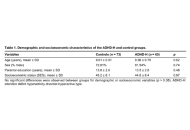
Visuospatial attention and intelligence in children with ADHD-hyperactive type
Daniela Smirni ... Michele Roccella
Published: March 11, 2026 Explor Neuroprot Ther. 2026;6:1004139
This article belongs to the special issue Advances in the Pathogenesis, Diagnosis and Treatment of Attention Deficit Hyperactivity Disorder
Open Access
Systematic Review
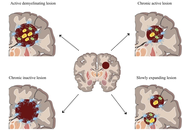
Therapeutic strategies in multiple sclerosis: a systematic review of current and emerging approaches
Carolina Machado, Ana Valado
Published: February 09, 2026 Explor Neuroprot Ther. 2026;6:1004138
Open Access
Review
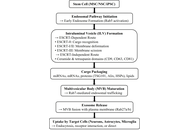
Stem cell-derived exosomes as neurotherapeutic agents: mechanisms of immunomodulation and neural regeneration in neurodegenerative disorders
Afra Wasama Islam ... Saumya Rajesh Kadam
Published: January 27, 2026 Explor Neuroprot Ther. 2026;6:1004137
This article belongs to the special issue Breakthroughs in Mechanisms and Treatments for Neurodegenerative Diseases
Open Access
Review
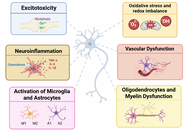
Neuroprotection beyond neurons: integrated biomarker-based and astroglia- or microglia-targeted approaches to combat neurodegenerative diseases
Cinzia Volonté ... Rafael Franco
Published: January 04, 2026 Explor Neuroprot Ther. 2026;6:1004136
Open Access
Original Article
Geothermal pteridophyte endophyte as a potential producer of anti-aggregation metabolites with relevance to neuroprotection
Agustina Lulustyaningati Nurul Aminin ... Muhammad Ajmal Shah
Published: December 30, 2025 Explor Neuroprot Ther. 2025;5:1004135
This article belongs to the special issue Natural Products in Neurotherapeutic Applications
Open Access
Original Article
Cognitive control in children with ADHD and subthreshold ADHD: an eye-tracking study
Rosa Angela Fabio ... Pina Filippello
Published: December 30, 2025 Explor Neuroprot Ther. 2025;5:1004134
This article belongs to the special issue Advances in the Pathogenesis, Diagnosis and Treatment of Attention Deficit Hyperactivity Disorder
Open Access
Perspective
Can we find early phase biomarkers for ALS: What are the prospects and challenges?
Christopher A. Shaw ... Leal Marakoff
Published: December 26, 2025 Explor Neuroprot Ther. 2025;5:1004133

Open Access
Original Article
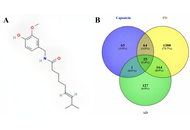
Molecular mechanism analysis and network pharmacology of capsaicin in Alzheimer’s and Parkinson’s diseases: an in silico study
Luis Antonio Ramirez-Contreras ... Andrés Frausto de Alba
Published: December 25, 2025 Explor Neuroprot Ther. 2025;5:1004132
This article belongs to the special issue Natural Products in Neurotherapeutic Applications
Open Access
Review
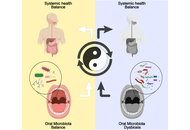
The role of oral microbiome in neurological diseases: mechanisms and clinical significance
Zhengrui Li ... Jing Li
Published: December 22, 2025 Explor Neuroprot Ther. 2025;5:1004131
This article belongs to the special issue Role of Microbiota in Neurological Diseases
Open Access
Perspective
Neurodivergence as environmental adaptation
Lur Carreras
Published: December 18, 2025 Explor Neuroprot Ther. 2025;5:1004130

Open Access
Review
Therapeutic role of probiotics in motor and non-motor symptoms of Parkinson’s disease
Salomón Páez-García ... Miguel Germán Borda
Published: December 15, 2025 Explor Neuroprot Ther. 2025;5:1004129
This article belongs to the special issue Role of Microbiota in Neurological Diseases

Open Access
Original Article
Exploring the therapeutic utility of apocynin in multifactorial neurodegenerative Alzheimer’s disease in a preclinical animal model
Sneha Bagle ... Sadhana Sathaye
Published: December 11, 2025 Explor Neuroprot Ther. 2025;5:1004128
This article belongs to the special issue Natural Products in Neurotherapeutic Applications
Open Access
Systematic Review
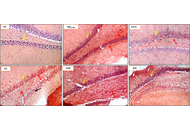
The long-term neuroprotective effects of calcium (Ca2+) modulation in ischemic stroke: a systematic review from animal models
Poppy Kristina Sasmita ... Bernadus Bernardino Bramantyo
Published: December 11, 2025 Explor Neuroprot Ther. 2025;5:1004127
This article belongs to the special issue Therapeutic Targets for Neuroprotection in Ischemic Stroke
Open Access
Perspective
Missed cervicocephalic dissections and the role of digital subtraction angiography and MRA with vessel wall imaging
Debabrata Chakraborty
Published: December 07, 2025 Explor Neuroprot Ther. 2025;5:1004126

Open Access
Systematic Review
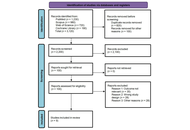
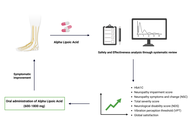
Alpha lipoic acid: advancing insights in diabetic neuropathy through updated systematic review and meta-analysis
Susmita Das ... Shylaja Hanumanthappa
Published: December 05, 2025 Explor Neuroprot Ther. 2025;5:1004125
This article belongs to the special issue Natural Products in Neurotherapeutic Applications
Open Access
Review
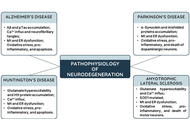
Resveratrol’s potential in the prevention and treatment of neurodegenerative diseases: molecular mechanisms
Mac Dionys Rodrigues da Costa ... Tiago Lima Sampaio
Published: November 20, 2025 Explor Neuroprot Ther. 2025;5:1004124
This article belongs to the special issue Neuro-Inflammation as a Target in the Design of Multifunctional Drug Candidates for Neurodegenerative Diseases
Journal Information
 Previous
Previous