








Aim:
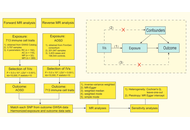
Adult-onset Still’s disease (AOSD) is a rare systemic inflammatory disorder marked by fever, rash, joint pain, and hyperferritinemia. While immune dysregulation is implicated in AOSD, the exact causal mechanisms remain unclear. This study aimed to investigate the genetic causal relationship between 731 immune cell phenotypes and AOSD, and to identify protective or risk-associated profiles.
Methods:
Using a two-sample Mendelian randomization (TSMR) approach, we applied inverse variance weighted (IVW) as the primary method, supplemented by MR-Egger, weighted median, simple mode, and weighted mode methods for robustness. Genetic instrumental variables for immune traits were sourced from recent genome-wide association studies (GWAS), and AOSD genetic predispositions were derived from the finn-b-STILL_ADULT cohort, comprising 201,947 individuals of European ancestry (3,403 AOSD cases and 198,544 controls).
Results:
We identified 49 immune cell-related traits showing nominally significant associations with AOSD (all adjusted P > 0.05 after FDR correction). Among these, 34 traits showed nominally protective trends, while 15 showed nominally risk-associated trends. Reciprocally, AOSD showed nominally suggestive effects on 40 immune cell traits, with 25 exhibiting a trend toward decreased levels and 15 toward increased levels. Additionally, we conducted multiple sensitivity analyses to explore potential heterogeneity and pleiotropy, though the primary findings did not survive FDR correction.
Conclusions:
These nominally significant associations between immune cell traits and AOSD, though not surviving FDR correction, may offer hypothesis-generating insights for future therapeutic research. The observed directional trends—with certain traits showing nominally protective or risk-associated patterns—suggest potential avenues for further exploration in the development of targeted treatment approaches for AOSD.
Aim:
Adult-onset Still’s disease (AOSD) is a rare systemic inflammatory disorder marked by fever, rash, joint pain, and hyperferritinemia. While immune dysregulation is implicated in AOSD, the exact causal mechanisms remain unclear. This study aimed to investigate the genetic causal relationship between 731 immune cell phenotypes and AOSD, and to identify protective or risk-associated profiles.
Methods:
Using a two-sample Mendelian randomization (TSMR) approach, we applied inverse variance weighted (IVW) as the primary method, supplemented by MR-Egger, weighted median, simple mode, and weighted mode methods for robustness. Genetic instrumental variables for immune traits were sourced from recent genome-wide association studies (GWAS), and AOSD genetic predispositions were derived from the finn-b-STILL_ADULT cohort, comprising 201,947 individuals of European ancestry (3,403 AOSD cases and 198,544 controls).
Results:
We identified 49 immune cell-related traits showing nominally significant associations with AOSD (all adjusted P > 0.05 after FDR correction). Among these, 34 traits showed nominally protective trends, while 15 showed nominally risk-associated trends. Reciprocally, AOSD showed nominally suggestive effects on 40 immune cell traits, with 25 exhibiting a trend toward decreased levels and 15 toward increased levels. Additionally, we conducted multiple sensitivity analyses to explore potential heterogeneity and pleiotropy, though the primary findings did not survive FDR correction.
Conclusions:
These nominally significant associations between immune cell traits and AOSD, though not surviving FDR correction, may offer hypothesis-generating insights for future therapeutic research. The observed directional trends—with certain traits showing nominally protective or risk-associated patterns—suggest potential avenues for further exploration in the development of targeted treatment approaches for AOSD.
DOI: https://doi.org/10.37349/emed.2026.1001406

Tricuspid valve endocarditis has potential associations with various conditions, but it is commonly related to intravenous drug abuse. Often, its eradication can become very challenging due to high post-operative mortality and high rate of recurrence due to persistence of drug abuse habits. The bidirectional Glenn shunt (BGS), typically employed in congenital heart surgery, combined with tricuspid valvectomy, has occasionally been used for recurrent endocarditis. Herein, we present a 31-year-old woman with drug addiction scheduled for her fourth reintervention due to the early degeneration and infection of a previous pulmonary homograft used for recurring tricuspid valve endocarditis. The final surgical strategy was valvectomy and BGS, aimed at eliminating all potential sources of infection and providing more time for the patient to overcome drug addiction, enabling further treatment if necessary.
Tricuspid valve endocarditis has potential associations with various conditions, but it is commonly related to intravenous drug abuse. Often, its eradication can become very challenging due to high post-operative mortality and high rate of recurrence due to persistence of drug abuse habits. The bidirectional Glenn shunt (BGS), typically employed in congenital heart surgery, combined with tricuspid valvectomy, has occasionally been used for recurrent endocarditis. Herein, we present a 31-year-old woman with drug addiction scheduled for her fourth reintervention due to the early degeneration and infection of a previous pulmonary homograft used for recurring tricuspid valve endocarditis. The final surgical strategy was valvectomy and BGS, aimed at eliminating all potential sources of infection and providing more time for the patient to overcome drug addiction, enabling further treatment if necessary.
DOI: https://doi.org/10.37349/emed.2026.1001407

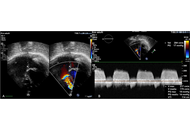
The recent improvements of the ultrasounds technology and the probes have led the dental community to start to apply this technology at macrovascular and microvascular level. This scoping review aims to investigate the diagnostic and research applications of ultrasonography in periodontics. A comprehensive literature search was conducted in PubMed, Scopus, and Web of Science, to identify relevant studies in periodontology: The search strategy included the following terms: “ultrasonography”, “echography”, “ultrasound”, “dentistry”. The review was conducted in accordance with the PRISMA-ScR guidelines. In addition, a manual search was conducted through the following journals from the last 10 years: Journal of Clinical Periodontology, the Journal of Dental Research and Oral Surgery, and Oral Medicine, Oral Pathology and Oral Radiology. Ten studies were included, covering different applications of ultrasonography in periodontology. Ultrasonography was employed in peri-implant and periodontal diagnosis, assessment of soft tissue thickness and vascularization, palatal wound and bone healing. This review highlights the effectiveness of ultrasonography in diagnosis and surgical evaluation. Further research, standardized protocols, and randomized clinical trials are needed, expanding the investigation to more fields relevant to the maxillofacial district.
The recent improvements of the ultrasounds technology and the probes have led the dental community to start to apply this technology at macrovascular and microvascular level. This scoping review aims to investigate the diagnostic and research applications of ultrasonography in periodontics. A comprehensive literature search was conducted in PubMed, Scopus, and Web of Science, to identify relevant studies in periodontology: The search strategy included the following terms: “ultrasonography”, “echography”, “ultrasound”, “dentistry”. The review was conducted in accordance with the PRISMA-ScR guidelines. In addition, a manual search was conducted through the following journals from the last 10 years: Journal of Clinical Periodontology, the Journal of Dental Research and Oral Surgery, and Oral Medicine, Oral Pathology and Oral Radiology. Ten studies were included, covering different applications of ultrasonography in periodontology. Ultrasonography was employed in peri-implant and periodontal diagnosis, assessment of soft tissue thickness and vascularization, palatal wound and bone healing. This review highlights the effectiveness of ultrasonography in diagnosis and surgical evaluation. Further research, standardized protocols, and randomized clinical trials are needed, expanding the investigation to more fields relevant to the maxillofacial district.
DOI: https://doi.org/10.37349/emed.2026.1001405

Although emotions play a fundamental role in modulating pain perception, their objective assessment in clinical contexts remains challenging. Recent advances in artificial intelligence (AI) have opened new opportunities to measure emotional states through facial expression analysis, physiological signal modeling, natural language processing (NLP), and multimodal data integration. In affective computing, the field that focuses on technologies designed to recognize, interpret, process, and simulate human emotions, facial expression-based emotion recognition has progressed from traditional machine learning methods to advanced deep learning approaches, including convolutional neural networks (CNNs), attention-based hybrid models, and transformer architectures. Similarly, recurrent neural networks and self-supervised learning methods have been implemented for developing models from physiological signals such as electrocardiography, photoplethysmography, galvanic skin response, and related biosignals. Additionally, NLP systems can extract affective information from naturalistic text, using both lexicon-based and transformer-based models. Finally, multimodal fusion and alignment techniques allow the integration of heterogeneous data streams, providing richer and more ecologically valid emotion representations. Collectively, these strategies offer powerful tools for advancing automatic pain assessment (APA) in cancer care, with the potential to support personalized, emotion-aware therapeutic approaches. However, from an AI perspective, several open challenges remain, including multimodal representation learning under weak supervision, robustness to missing or degraded modalities, limited explainability of affective inference models, lack of standardized benchmarking protocols, and the presence of bias and domain shift in emotion datasets. Given the inherently subjective, context-dependent, and culturally mediated features of the emotional experience, further research is needed to address these technical limitations, integrating technological advances with the intrinsic complexity of emotion interpretation.
Although emotions play a fundamental role in modulating pain perception, their objective assessment in clinical contexts remains challenging. Recent advances in artificial intelligence (AI) have opened new opportunities to measure emotional states through facial expression analysis, physiological signal modeling, natural language processing (NLP), and multimodal data integration. In affective computing, the field that focuses on technologies designed to recognize, interpret, process, and simulate human emotions, facial expression-based emotion recognition has progressed from traditional machine learning methods to advanced deep learning approaches, including convolutional neural networks (CNNs), attention-based hybrid models, and transformer architectures. Similarly, recurrent neural networks and self-supervised learning methods have been implemented for developing models from physiological signals such as electrocardiography, photoplethysmography, galvanic skin response, and related biosignals. Additionally, NLP systems can extract affective information from naturalistic text, using both lexicon-based and transformer-based models. Finally, multimodal fusion and alignment techniques allow the integration of heterogeneous data streams, providing richer and more ecologically valid emotion representations. Collectively, these strategies offer powerful tools for advancing automatic pain assessment (APA) in cancer care, with the potential to support personalized, emotion-aware therapeutic approaches. However, from an AI perspective, several open challenges remain, including multimodal representation learning under weak supervision, robustness to missing or degraded modalities, limited explainability of affective inference models, lack of standardized benchmarking protocols, and the presence of bias and domain shift in emotion datasets. Given the inherently subjective, context-dependent, and culturally mediated features of the emotional experience, further research is needed to address these technical limitations, integrating technological advances with the intrinsic complexity of emotion interpretation.
DOI: https://doi.org/10.37349/emed.2026.1001404
This article belongs to the special issue Innovative Approaches to Chronic Pain Management: from Multidisciplinary Strategies to Artificial Intelligence Perspectives

Aim:
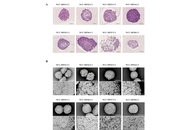
Myxofibrosarcoma (MFS) is characterized by high local recurrence and complex microenvironmental interactions. Although three-dimensional (3D) culture systems better mimic in vivo tumor architecture than conventional two-dimensional (2D) monolayer cultures, the global proteomic consequences of dimensionality in MFS remain incompletely defined.
Methods:
We performed quantitative mass spectrometry-based proteomic profiling of eight independently established patient-derived MFS cell lines cultured under 2D monolayer and 3D spheroid conditions. Differential protein expression and pathway enrichment analyses were conducted to delineate dimensionality-driven molecular programs.
Results:
Culture dimensionality emerged as the principal determinant of proteomic variation across all cell lines. Compared with monolayers, 3D spheroids exhibited significant enrichment of hypoxia response, autophagy-related processes, extracellular matrix organization, and PI3K-Akt signaling pathways. In contrast, 2D cultures preferentially upregulated DNA replication, RNA processing, and cell-cycle-associated pathways. These findings indicate that 3D architecture alone is sufficient to induce coordinated stress-adaptive and survival-oriented proteomic reprogramming in MFS cells.
Conclusions:
This study provides a comprehensive proteomic atlas defining dimensionality-dependent molecular states in MFS. While functional validation was beyond the scope of this work, the pathway rewiring identified here establishes a mechanistic framework for future hypothesis-driven investigations targeting autophagy- and PI3K-Akt-associated vulnerabilities in 3D MFS models.
Aim:
Myxofibrosarcoma (MFS) is characterized by high local recurrence and complex microenvironmental interactions. Although three-dimensional (3D) culture systems better mimic in vivo tumor architecture than conventional two-dimensional (2D) monolayer cultures, the global proteomic consequences of dimensionality in MFS remain incompletely defined.
Methods:
We performed quantitative mass spectrometry-based proteomic profiling of eight independently established patient-derived MFS cell lines cultured under 2D monolayer and 3D spheroid conditions. Differential protein expression and pathway enrichment analyses were conducted to delineate dimensionality-driven molecular programs.
Results:
Culture dimensionality emerged as the principal determinant of proteomic variation across all cell lines. Compared with monolayers, 3D spheroids exhibited significant enrichment of hypoxia response, autophagy-related processes, extracellular matrix organization, and PI3K-Akt signaling pathways. In contrast, 2D cultures preferentially upregulated DNA replication, RNA processing, and cell-cycle-associated pathways. These findings indicate that 3D architecture alone is sufficient to induce coordinated stress-adaptive and survival-oriented proteomic reprogramming in MFS cells.
Conclusions:
This study provides a comprehensive proteomic atlas defining dimensionality-dependent molecular states in MFS. While functional validation was beyond the scope of this work, the pathway rewiring identified here establishes a mechanistic framework for future hypothesis-driven investigations targeting autophagy- and PI3K-Akt-associated vulnerabilities in 3D MFS models.
DOI: https://doi.org/10.37349/emed.2026.1001403

Aim:
Adherence to antihypertensive medication is essential for positive outcomes among patients diagnosed with hypertension. Yet, up to 72% of individuals prescribed antihypertensives do not take medication as prescribed. Understanding mechanisms of behavior change (MoBCs) for antihypertensive adherence provides essential insight for how to change adherence behavior. This study examined the association between 4 potential MoBCs and self-reported antihypertensive adherence.
Methods:
This exploratory, cross-sectional study recruited 101 patients prescribed antihypertensive medications to complete self-report questionnaires on adherence and potential mechanisms of nonadherence. Antihypertensive adherence was assessed using the Hill-Bone Compliance to High Blood Pressure Therapy Scale (HB-HBP). The 4 putative MoBCs for nonadherence included self-efficacy, self-regulation, behavioral automaticity, and hypertension knowledge. Associations between self-reported antihypertensive adherence and MoBCs were examined using independent samples t-tests and multivariate median regression with covariate adjustment for participant characteristics.
Results:
The sample had a mean age of 61.7 years (SD = 13.1 years), was 59.4% female (n = 60), 66.3% White (n = 67), and 7.9% Hispanic (n = 8). Low adherence was reported by 16.8% of the study sample. Participants who reported low adherence to antihypertensive medications had lower scores on the self-efficacy questionnaire (p < 0.001) and hypertension knowledge (p = 0.045). Self-efficacy and hypertension knowledge remained significantly associated with self-reported adherence in multivariate regression with covariate adjustment.
Conclusions:
The current study supports the hypothesis that self-efficacy for adherence and additionally hypertension knowledge are the MoBCs most strongly associated with self-reported adherence behavior. Behavioral interventions to improve medication adherence should consider focusing on self-efficacy and hypertension knowledge as potentially important target MoBCs. The authors recommend that future research should consider tailored intervention approaches that target specific mechanisms of adherence and specific self-reported reasons for nonadherence.
Aim:
Adherence to antihypertensive medication is essential for positive outcomes among patients diagnosed with hypertension. Yet, up to 72% of individuals prescribed antihypertensives do not take medication as prescribed. Understanding mechanisms of behavior change (MoBCs) for antihypertensive adherence provides essential insight for how to change adherence behavior. This study examined the association between 4 potential MoBCs and self-reported antihypertensive adherence.
Methods:
This exploratory, cross-sectional study recruited 101 patients prescribed antihypertensive medications to complete self-report questionnaires on adherence and potential mechanisms of nonadherence. Antihypertensive adherence was assessed using the Hill-Bone Compliance to High Blood Pressure Therapy Scale (HB-HBP). The 4 putative MoBCs for nonadherence included self-efficacy, self-regulation, behavioral automaticity, and hypertension knowledge. Associations between self-reported antihypertensive adherence and MoBCs were examined using independent samples t-tests and multivariate median regression with covariate adjustment for participant characteristics.
Results:
The sample had a mean age of 61.7 years (SD = 13.1 years), was 59.4% female (n = 60), 66.3% White (n = 67), and 7.9% Hispanic (n = 8). Low adherence was reported by 16.8% of the study sample. Participants who reported low adherence to antihypertensive medications had lower scores on the self-efficacy questionnaire (p < 0.001) and hypertension knowledge (p = 0.045). Self-efficacy and hypertension knowledge remained significantly associated with self-reported adherence in multivariate regression with covariate adjustment.
Conclusions:
The current study supports the hypothesis that self-efficacy for adherence and additionally hypertension knowledge are the MoBCs most strongly associated with self-reported adherence behavior. Behavioral interventions to improve medication adherence should consider focusing on self-efficacy and hypertension knowledge as potentially important target MoBCs. The authors recommend that future research should consider tailored intervention approaches that target specific mechanisms of adherence and specific self-reported reasons for nonadherence.
DOI: https://doi.org/10.37349/emed.2026.1001402
This article belongs to the special issue Drug Adherence in Hypertension

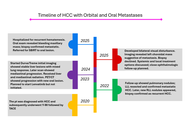
Hepatocellular carcinoma (HCC) is an aggressive primary liver malignancy with a high propensity for extrahepatic spread, most commonly to the lungs, lymph nodes, and bones. Metastases to atypical sites like the orbital and oral cavity are uncommon and often underrecognized. We report the case of a 74-year-old male with hepatitis C-related cirrhosis who was diagnosed with HCC and initially treated with locoregional therapies. Disease progression was marked by the development of biopsy-confirmed pulmonary metastases, followed by treatment with durvalumab plus tremelimumab immunotherapy and external beam radiation. Despite therapy, further progression occurred with mediastinal lymphadenopathy. The patient subsequently developed visual disturbances, and ophthalmologic evaluation identified a solitary choroidal mass consistent with metastatic HCC. Around the same time, he presented with recurrent oral bleeding, and a biopsy of a maxillary gingival lesion confirmed metastatic HCC. This case highlights the aggressive nature of advanced HCC and its potential for unusual metastatic spread. To our knowledge, this is among the few reported cases of HCC with orbital and oral metastases. Awareness of atypical metastatic presentations and a multidisciplinary approach are essential for timely diagnosis and optimal management.
Hepatocellular carcinoma (HCC) is an aggressive primary liver malignancy with a high propensity for extrahepatic spread, most commonly to the lungs, lymph nodes, and bones. Metastases to atypical sites like the orbital and oral cavity are uncommon and often underrecognized. We report the case of a 74-year-old male with hepatitis C-related cirrhosis who was diagnosed with HCC and initially treated with locoregional therapies. Disease progression was marked by the development of biopsy-confirmed pulmonary metastases, followed by treatment with durvalumab plus tremelimumab immunotherapy and external beam radiation. Despite therapy, further progression occurred with mediastinal lymphadenopathy. The patient subsequently developed visual disturbances, and ophthalmologic evaluation identified a solitary choroidal mass consistent with metastatic HCC. Around the same time, he presented with recurrent oral bleeding, and a biopsy of a maxillary gingival lesion confirmed metastatic HCC. This case highlights the aggressive nature of advanced HCC and its potential for unusual metastatic spread. To our knowledge, this is among the few reported cases of HCC with orbital and oral metastases. Awareness of atypical metastatic presentations and a multidisciplinary approach are essential for timely diagnosis and optimal management.
DOI: https://doi.org/10.37349/emed.2026.1001401

Background:
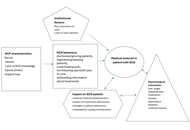
Sickle cell disease (SCD) is a group of heritable conditions with significant morbidity, burden of disease management and healthcare delivery issues. Medical mistrust (MM) is a psychological outcome of healthcare delivery issues. The purpose of this review was to assess the concept of MM in the literature on SCD, summarize the findings and gaps, and was guided by three questions: 1) How has MM been measured and/or described in patients with SCD? 2) What factors have been described in the literature that predispose patients with SCD to develop MM? 3) What consequences or outcomes have been described because of MM in patients with SCD?
Methods:
The methodological framework of Arksey and O’Malley was used to review articles from PubMed, Scopus, Web of Science, CINAHL, PsycInfo, and EMBASE. Inclusion criteria were quantitative, qualitative, and mixed methods peer-reviewed studies published in English between 1994 and 2025; articles focused on patients with SCD and those that described concepts found in the existing MM instruments.
Results:
Forty-two studies were included; 26 were strictly qualitative, 11 were mixed methods, and 5 were strictly quantitative. No study used an existing MM measure; yet concepts from MM measures were described: group disparities and suspicion. Negative healthcare staff communication, poor pain control, transition of care, and lack of provider transparency predisposed patients toward MM. Outcomes of MM included avoidance of care, nonadherence, psychological distress, and maladaptive coping.
Discussion:
This review highlights the predictors and outcomes of MM in patients with SCD and identifies the notable gaps in the state of the SCD MM literature. More studies are needed to assess the development and consequences of MM in patients with SCD. The findings highlight the experiences of patients with SCD and offer researchers insights into possible interventions to decrease MM and improve outcomes.
Background:
Sickle cell disease (SCD) is a group of heritable conditions with significant morbidity, burden of disease management and healthcare delivery issues. Medical mistrust (MM) is a psychological outcome of healthcare delivery issues. The purpose of this review was to assess the concept of MM in the literature on SCD, summarize the findings and gaps, and was guided by three questions: 1) How has MM been measured and/or described in patients with SCD? 2) What factors have been described in the literature that predispose patients with SCD to develop MM? 3) What consequences or outcomes have been described because of MM in patients with SCD?
Methods:
The methodological framework of Arksey and O’Malley was used to review articles from PubMed, Scopus, Web of Science, CINAHL, PsycInfo, and EMBASE. Inclusion criteria were quantitative, qualitative, and mixed methods peer-reviewed studies published in English between 1994 and 2025; articles focused on patients with SCD and those that described concepts found in the existing MM instruments.
Results:
Forty-two studies were included; 26 were strictly qualitative, 11 were mixed methods, and 5 were strictly quantitative. No study used an existing MM measure; yet concepts from MM measures were described: group disparities and suspicion. Negative healthcare staff communication, poor pain control, transition of care, and lack of provider transparency predisposed patients toward MM. Outcomes of MM included avoidance of care, nonadherence, psychological distress, and maladaptive coping.
Discussion:
This review highlights the predictors and outcomes of MM in patients with SCD and identifies the notable gaps in the state of the SCD MM literature. More studies are needed to assess the development and consequences of MM in patients with SCD. The findings highlight the experiences of patients with SCD and offer researchers insights into possible interventions to decrease MM and improve outcomes.
DOI: https://doi.org/10.37349/emed.2026.1001400

The involvement of the Internet of Things (IoT) technology and artificial intelligence (AI) in the matter of maternal healthcare has allowed monitoring pregnant women in real-time and predicting poor pregnancy outcomes, including stillbirth and premature birth. Nevertheless, to diminish the risks of devices, the introduction of such technologies has to be accompanied by harsh safety monitoring programs. Materiovigilance, as the systematic sensing and monitoring of adverse events associated with medical instruments, is also crucial to patient safety in high-risk obstetric environments. Wearable sensors, e.g., fetal Dopplers, smart fabrics, and adhesive patches, have enhanced the prediction of stillbirth by offering continuous acquisition of physiological data but presents a hazard of ill effects if not controlled adequately. The AI introduction into the sphere of Materiovigilance enhances regulatory conformity, real-time decision-making, and raises the possibility of risk identification. Despite the massive potential, issues such as inaccurate data, poor infrastructure, and underreporting persist, particularly in low- and middle-income countries. In such circumstances, the Materiovigilance Programme of India is another staged effort to enhance the device safety supervision and reporting systems. Engagement of different stakeholders such as clinicians, engineers, regulatory agencies, and technology developers provides an opportunity to secure the safety and efficacy of AI-equipped medical devices. Enhancement of Materiovigilance systems is required to preserve proper maternal-fetal health and sustain clinics in digital obstetrics, as any error in maternal-fetal monitoring may lead to preventable death.
The involvement of the Internet of Things (IoT) technology and artificial intelligence (AI) in the matter of maternal healthcare has allowed monitoring pregnant women in real-time and predicting poor pregnancy outcomes, including stillbirth and premature birth. Nevertheless, to diminish the risks of devices, the introduction of such technologies has to be accompanied by harsh safety monitoring programs. Materiovigilance, as the systematic sensing and monitoring of adverse events associated with medical instruments, is also crucial to patient safety in high-risk obstetric environments. Wearable sensors, e.g., fetal Dopplers, smart fabrics, and adhesive patches, have enhanced the prediction of stillbirth by offering continuous acquisition of physiological data but presents a hazard of ill effects if not controlled adequately. The AI introduction into the sphere of Materiovigilance enhances regulatory conformity, real-time decision-making, and raises the possibility of risk identification. Despite the massive potential, issues such as inaccurate data, poor infrastructure, and underreporting persist, particularly in low- and middle-income countries. In such circumstances, the Materiovigilance Programme of India is another staged effort to enhance the device safety supervision and reporting systems. Engagement of different stakeholders such as clinicians, engineers, regulatory agencies, and technology developers provides an opportunity to secure the safety and efficacy of AI-equipped medical devices. Enhancement of Materiovigilance systems is required to preserve proper maternal-fetal health and sustain clinics in digital obstetrics, as any error in maternal-fetal monitoring may lead to preventable death.
DOI: https://doi.org/10.37349/emed.2026.1001399

Idiopathic pulmonary fibrosis (IPF) is a progressive, fatal interstitial lung disease of undefined etiology. In recent years, its global incidence has shown an upward trend, with a median survival of approximately 3–5 years after diagnosis. Currently, clinical treatment outcomes for this disease remain limited. Approved therapeutic agents are nintedanib, pirfenidone, and nerandomilast, all of which are predominantly administered orally. The oral route renders drugs susceptible to degradation in the gastrointestinal tract, leading to reduced drug bioavailability and limited therapeutic efficacy. In this work, we believe that nanodelivery systems (NDSs) represent a promising approach to address the limitations of traditional therapies. Most importantly, we emphasized that inhalation NDSs should be prioritized to enhance the accumulation efficiency of targeted drugs at pulmonary lesion sites. Collectively, this study aims to provide insights into the developmental prospects of NDSs for IPF, paving the way for more efficient and personalized therapeutic approaches to enhance treatment efficacy while minimizing side effects.
Idiopathic pulmonary fibrosis (IPF) is a progressive, fatal interstitial lung disease of undefined etiology. In recent years, its global incidence has shown an upward trend, with a median survival of approximately 3–5 years after diagnosis. Currently, clinical treatment outcomes for this disease remain limited. Approved therapeutic agents are nintedanib, pirfenidone, and nerandomilast, all of which are predominantly administered orally. The oral route renders drugs susceptible to degradation in the gastrointestinal tract, leading to reduced drug bioavailability and limited therapeutic efficacy. In this work, we believe that nanodelivery systems (NDSs) represent a promising approach to address the limitations of traditional therapies. Most importantly, we emphasized that inhalation NDSs should be prioritized to enhance the accumulation efficiency of targeted drugs at pulmonary lesion sites. Collectively, this study aims to provide insights into the developmental prospects of NDSs for IPF, paving the way for more efficient and personalized therapeutic approaches to enhance treatment efficacy while minimizing side effects.
DOI: https://doi.org/10.37349/emed.2026.1001397

Aim:
Polypharmacy is increasingly prevalent among older adults and has been suggested as a potential risk factor for adverse health outcomes, including cognitive impairment and functional decline. Therefore, this study aimed to investigate the associations of polypharmacy with cognitive impairment and functional status among community-dwelling older adults using nationally representative data from the 2023 Korean Elderly Survey.
Methods:
A cross-sectional analysis was conducted using data from 9,898 community-dwelling older adults without a diagnosis of dementia. Polypharmacy was defined as the concurrent use of five or more physician-prescribed medications. Cognitive function was assessed using the Korean version of the Mini-Mental State Examination (K-MMSE), with cognitive impairment defined as a score ≤ 23. Functional status was evaluated using the Korean Activities of Daily Living (K-ADL) and Korean Instrumental Activities of Daily Living (K-IADL). Logistic regression was used to estimate odds ratios and 95% confidence intervals (CIs) for cognitive impairment, while multiple linear regression analyses examined associations with functional status. Models were sequentially adjusted for sociodemographic characteristics, health behaviors, and the number of chronic diseases.
Results:
Polypharmacy was associated with increased odds of cognitive impairment in the crude model (OR = 1.70, 95% CI: 1.40–2.05); however, this association was attenuated and became non-significant after adjustment for sociodemographic and health-related factors. In contrast, polypharmacy remained independently associated with poorer functional status in fully adjusted models, showing higher K-ADL scores (B = 0.14, p = 0.007) and K-IADL scores (B = 0.43, p < 0.001).
Conclusions:
Polypharmacy was independently associated with functional impairment but not with cognitive impairment after comprehensive adjustment, suggesting that functional decline may represent a more sensitive and immediate consequence of complex medication use in older adults. These findings underscore the need for comprehensive geriatric assessment approaches that integrate medication review with functional evaluation.
Aim:
Polypharmacy is increasingly prevalent among older adults and has been suggested as a potential risk factor for adverse health outcomes, including cognitive impairment and functional decline. Therefore, this study aimed to investigate the associations of polypharmacy with cognitive impairment and functional status among community-dwelling older adults using nationally representative data from the 2023 Korean Elderly Survey.
Methods:
A cross-sectional analysis was conducted using data from 9,898 community-dwelling older adults without a diagnosis of dementia. Polypharmacy was defined as the concurrent use of five or more physician-prescribed medications. Cognitive function was assessed using the Korean version of the Mini-Mental State Examination (K-MMSE), with cognitive impairment defined as a score ≤ 23. Functional status was evaluated using the Korean Activities of Daily Living (K-ADL) and Korean Instrumental Activities of Daily Living (K-IADL). Logistic regression was used to estimate odds ratios and 95% confidence intervals (CIs) for cognitive impairment, while multiple linear regression analyses examined associations with functional status. Models were sequentially adjusted for sociodemographic characteristics, health behaviors, and the number of chronic diseases.
Results:
Polypharmacy was associated with increased odds of cognitive impairment in the crude model (OR = 1.70, 95% CI: 1.40–2.05); however, this association was attenuated and became non-significant after adjustment for sociodemographic and health-related factors. In contrast, polypharmacy remained independently associated with poorer functional status in fully adjusted models, showing higher K-ADL scores (B = 0.14, p = 0.007) and K-IADL scores (B = 0.43, p < 0.001).
Conclusions:
Polypharmacy was independently associated with functional impairment but not with cognitive impairment after comprehensive adjustment, suggesting that functional decline may represent a more sensitive and immediate consequence of complex medication use in older adults. These findings underscore the need for comprehensive geriatric assessment approaches that integrate medication review with functional evaluation.
DOI: https://doi.org/10.37349/emed.2026.1001398

Background:
Obstructive sleep apnea (OSA) is a syndrome characterized by episodes of complete cessation of breathing (apnea) or inadequate breathing (hypopnea) during sleep.
Methods:
A systematic search of PubMed, Cochrane Library, Embase, Scopus, Web of Science, and Lilacs databases was conducted following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We identified 1,532 records; after screening, 9 randomized controlled trials (RCTs), published between 2009 and 2020, met the inclusion criteria. These studies included 698 participants aged 5–75 years.
Results:
Nine randomized trials (n = 698; 2009–2020) showed that myofunctional therapy (MFT), alone or as an adjunct, for example, continuous positive airway pressure (CPAP) and nasal washing, reduced apnea-hypopnea index (AHI) versus control in adults and children. Snoring intensity improved in trials that measured it; several studies reported gains in oxygen saturation and mouth-breathing reduction. Protocols targeted the soft palate, tongue, and facial muscles with daily home exercises.
Discussion:
MFT appears to be a promising non-invasive treatment for reducing AHI, especially in pediatric patients. Its benefits extend beyond AHI reduction, supporting orofacial function and nasal breathing. However, its clinical integration remains limited due to a lack of standardized protocols and inconsistent reporting of patient adherence. Most studies also have short follow-up periods, which makes it difficult to assess long-term efficacy. To advance evidence-based use of MFT, future research should adopt standardized outcomes, monitor adherence systematically, and include long-term follow-up.
Background:
Obstructive sleep apnea (OSA) is a syndrome characterized by episodes of complete cessation of breathing (apnea) or inadequate breathing (hypopnea) during sleep.
Methods:
A systematic search of PubMed, Cochrane Library, Embase, Scopus, Web of Science, and Lilacs databases was conducted following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We identified 1,532 records; after screening, 9 randomized controlled trials (RCTs), published between 2009 and 2020, met the inclusion criteria. These studies included 698 participants aged 5–75 years.
Results:
Nine randomized trials (n = 698; 2009–2020) showed that myofunctional therapy (MFT), alone or as an adjunct, for example, continuous positive airway pressure (CPAP) and nasal washing, reduced apnea-hypopnea index (AHI) versus control in adults and children. Snoring intensity improved in trials that measured it; several studies reported gains in oxygen saturation and mouth-breathing reduction. Protocols targeted the soft palate, tongue, and facial muscles with daily home exercises.
Discussion:
MFT appears to be a promising non-invasive treatment for reducing AHI, especially in pediatric patients. Its benefits extend beyond AHI reduction, supporting orofacial function and nasal breathing. However, its clinical integration remains limited due to a lack of standardized protocols and inconsistent reporting of patient adherence. Most studies also have short follow-up periods, which makes it difficult to assess long-term efficacy. To advance evidence-based use of MFT, future research should adopt standardized outcomes, monitor adherence systematically, and include long-term follow-up.
DOI: https://doi.org/10.37349/emed.2026.1001396

The biopsychosocial model is the prevailing framework for chronic orofacial pain (COP). While COP is a heterogeneous clinical entity involving nociceptive and neuropathic components, it is increasingly defined by its nociplastic features—a systemic, non-nociceptive state in which psychological factors significantly influence symptoms. Current research frequently suffers from the conflation of constructs. Psychosocial predictors (e.g., self-efficacy) and outcome measures (e.g., pain interference) are often conceptually inseparable. To advance beyond this, we advocate for the integration of the brain-heart axis (BHA). The BHA provides objective, quantifiable markers of autonomic nervous system (ANS) dysregulation, the physical manifestation of chronic stress rooted in large-scale brain network imbalance. The present study proposes a theoretical framework in which psychological distress is reflected in corrected QT interval (QTc) changes, while low self-efficacy is mirrored by reduced heart rate variability (HRV). This integration is supported by the neurochemical roles of N-methyl-D-aspartate (NMDA) receptors in central sensitization and dopamine D2 receptor dysfunction in the basal ganglia. The present paper delineates a framework for research and clinical implementation within advanced dental training.
The biopsychosocial model is the prevailing framework for chronic orofacial pain (COP). While COP is a heterogeneous clinical entity involving nociceptive and neuropathic components, it is increasingly defined by its nociplastic features—a systemic, non-nociceptive state in which psychological factors significantly influence symptoms. Current research frequently suffers from the conflation of constructs. Psychosocial predictors (e.g., self-efficacy) and outcome measures (e.g., pain interference) are often conceptually inseparable. To advance beyond this, we advocate for the integration of the brain-heart axis (BHA). The BHA provides objective, quantifiable markers of autonomic nervous system (ANS) dysregulation, the physical manifestation of chronic stress rooted in large-scale brain network imbalance. The present study proposes a theoretical framework in which psychological distress is reflected in corrected QT interval (QTc) changes, while low self-efficacy is mirrored by reduced heart rate variability (HRV). This integration is supported by the neurochemical roles of N-methyl-D-aspartate (NMDA) receptors in central sensitization and dopamine D2 receptor dysfunction in the basal ganglia. The present paper delineates a framework for research and clinical implementation within advanced dental training.
DOI: https://doi.org/10.37349/emed.2026.1001395

Aim:
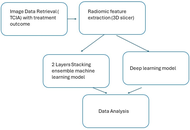
This study aimed to develop and evaluate a stacking ensemble machine learning (SEML) model that integrates deep learning (DL) algorithms to improve the accuracy of prognostic predictions for patients with head and neck squamous cell carcinoma (HNSCC).
Methods:
A cohort of 215 HNSCC patients’ CT images, featuring gross tumor volume (GTV) and planning target volume (PTV) contours, was analyzed. Radiomics features were extracted and converted into quantitative data. These features were then used to train and compare a novel SEML model against standard DL algorithms to predict patient prognosis.
Results:
The proposed SEML model demonstrated superior predictive performance compared to the DL model, achieving 93% accuracy, 100% sensitivity, and 83% specificity. Statistical analysis using the chi-square test indicated no substantial difference in prediction performance between features derived from GTV and PTV contours (p > 0.05).
Conclusions:
The SEML model effectively enhances the prognostic prediction accuracy for HNSCC based on radiomic features. This approach shows significant potential to inform clinical decision-making and support the development of customized treatment strategies for improved patient care.
Aim:
This study aimed to develop and evaluate a stacking ensemble machine learning (SEML) model that integrates deep learning (DL) algorithms to improve the accuracy of prognostic predictions for patients with head and neck squamous cell carcinoma (HNSCC).
Methods:
A cohort of 215 HNSCC patients’ CT images, featuring gross tumor volume (GTV) and planning target volume (PTV) contours, was analyzed. Radiomics features were extracted and converted into quantitative data. These features were then used to train and compare a novel SEML model against standard DL algorithms to predict patient prognosis.
Results:
The proposed SEML model demonstrated superior predictive performance compared to the DL model, achieving 93% accuracy, 100% sensitivity, and 83% specificity. Statistical analysis using the chi-square test indicated no substantial difference in prediction performance between features derived from GTV and PTV contours (p > 0.05).
Conclusions:
The SEML model effectively enhances the prognostic prediction accuracy for HNSCC based on radiomic features. This approach shows significant potential to inform clinical decision-making and support the development of customized treatment strategies for improved patient care.
DOI: https://doi.org/10.37349/emed.2026.1001394
This article belongs to the special issue Artificial Intelligence in Precision Imaging: Innovations Shaping the Future of Clinical Diagnostics

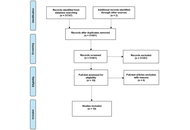
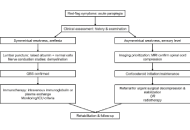
Acute paraplegia is a frequent and high-stake presentation in emergency departments. Spinal cord compression (SCC), particularly malignant epidural SCC, is a common oncologic emergency requiring urgent intervention, whereas Guillain-Barré syndrome (GBS) is a rarer but potentially life-threatening autoimmune polyradiculoneuropathy. Early differentiation between these conditions is essential, as delays in diagnosis and treatment are associated with irreversible neurological deficits and increased morbidity. Our objective is to synthesize recent evidence on the pathophysiology, clinical presentation, diagnosis, and management of SCC and GBS, with emphasis on early differentiation and multidisciplinary care strategies in emergency and rehabilitation settings. A scoping review was conducted following the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) guidelines. Electronic databases (PubMed, MEDLINE, Scopus, ScienceDirect, and CINAHL) were searched for English language studies published between January 2020 and June 2025. The review focused on clinical studies and reports addressing early differentiation and multidisciplinary management of SCC and GBS in emergency settings. Nineteen studies met the inclusion criteria. The review found that GBS diagnosis relies heavily on recognizing progressive symmetric weakness and preceding infectious triggers, while SCC requires immediate imaging and prompt corticosteroid administration. The limited number of studies highlights a gap in integrated emergency protocols for distinguishing these conditions. Surgical decompression remains the cornerstone of SCC management, with emerging evidence suggesting potential benefits even beyond the traditional 24-h window. This scoping review reinforces the critical need for early differentiation between SCC and GBS. Although the available literature is limited, it underscores the importance of coordinated multidisciplinary care. Clinicians must remain attentive to evolving diagnostic algorithms, particularly in light of new evidence supporting extended surgical windows for SCC.
Acute paraplegia is a frequent and high-stake presentation in emergency departments. Spinal cord compression (SCC), particularly malignant epidural SCC, is a common oncologic emergency requiring urgent intervention, whereas Guillain-Barré syndrome (GBS) is a rarer but potentially life-threatening autoimmune polyradiculoneuropathy. Early differentiation between these conditions is essential, as delays in diagnosis and treatment are associated with irreversible neurological deficits and increased morbidity. Our objective is to synthesize recent evidence on the pathophysiology, clinical presentation, diagnosis, and management of SCC and GBS, with emphasis on early differentiation and multidisciplinary care strategies in emergency and rehabilitation settings. A scoping review was conducted following the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) guidelines. Electronic databases (PubMed, MEDLINE, Scopus, ScienceDirect, and CINAHL) were searched for English language studies published between January 2020 and June 2025. The review focused on clinical studies and reports addressing early differentiation and multidisciplinary management of SCC and GBS in emergency settings. Nineteen studies met the inclusion criteria. The review found that GBS diagnosis relies heavily on recognizing progressive symmetric weakness and preceding infectious triggers, while SCC requires immediate imaging and prompt corticosteroid administration. The limited number of studies highlights a gap in integrated emergency protocols for distinguishing these conditions. Surgical decompression remains the cornerstone of SCC management, with emerging evidence suggesting potential benefits even beyond the traditional 24-h window. This scoping review reinforces the critical need for early differentiation between SCC and GBS. Although the available literature is limited, it underscores the importance of coordinated multidisciplinary care. Clinicians must remain attentive to evolving diagnostic algorithms, particularly in light of new evidence supporting extended surgical windows for SCC.
DOI: https://doi.org/10.37349/emed.2026.1001393
This article belongs to the special issue Practical Tips for Cancer Care: Guidance for Patients, Caregivers, and Healthcare Professionals

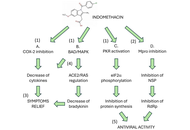
At the beginning of COVID-19 pandemic, due to the lack of guidance from evidence-based medicine on how to treat the infected patients, the medical class faced significant difficulties, not only with the unknown infection but also with the great number of cases. This led to a great number of hospitalizations and deaths. It would have been necessary to try drugs already available on the market, which might be effective against SARS-CoV-2. This is a short review of studies in the scientific literature dealing with the use of indomethacin as an antiviral drug against SARS-CoVs. We revised studies taken from the scientific literature in PubMed, Science Direct, Scopus, ResearchGate, Google Scholar etc., describing the effects of indomethacin as an antiviral drug against SARS-CoVs. To search for studies, we used the keywords: SARS-CoV, off-label drug, pandemic emergence, repurposed drug, Absence of EBM, Antiviral, Indomethacin, and Mechanisms. Among the studies reviewed, there is an interesting experimental study, published in 2006 by an Italian group, which considered the problem at the time of the previous SARS-CoV epidemic, that clearly demonstrated an antiviral effect of indomethacin, both in vitro and in vivo, against SARS-CoV. Two other studies, both clinical, one retrospective observational, and the other a prospective randomized trial comparing indomethacin with paracetamol, also showed good effects of indomethacin in the treatment of patients with COVID-19. On the basis of these notices, we wonder why, in such an emergency situation, indomethacin was not taken into consideration and was not tried in the treatment of COVID-19.
At the beginning of COVID-19 pandemic, due to the lack of guidance from evidence-based medicine on how to treat the infected patients, the medical class faced significant difficulties, not only with the unknown infection but also with the great number of cases. This led to a great number of hospitalizations and deaths. It would have been necessary to try drugs already available on the market, which might be effective against SARS-CoV-2. This is a short review of studies in the scientific literature dealing with the use of indomethacin as an antiviral drug against SARS-CoVs. We revised studies taken from the scientific literature in PubMed, Science Direct, Scopus, ResearchGate, Google Scholar etc., describing the effects of indomethacin as an antiviral drug against SARS-CoVs. To search for studies, we used the keywords: SARS-CoV, off-label drug, pandemic emergence, repurposed drug, Absence of EBM, Antiviral, Indomethacin, and Mechanisms. Among the studies reviewed, there is an interesting experimental study, published in 2006 by an Italian group, which considered the problem at the time of the previous SARS-CoV epidemic, that clearly demonstrated an antiviral effect of indomethacin, both in vitro and in vivo, against SARS-CoV. Two other studies, both clinical, one retrospective observational, and the other a prospective randomized trial comparing indomethacin with paracetamol, also showed good effects of indomethacin in the treatment of patients with COVID-19. On the basis of these notices, we wonder why, in such an emergency situation, indomethacin was not taken into consideration and was not tried in the treatment of COVID-19.
DOI: https://doi.org/10.37349/emed.2026.1001392

DOI: https://doi.org/10.37349/emed.2026.1001391

New guidelines for high blood pressure (BP) by the European Society of Cardiology (ESC) now consider BP of 120–140/70–90 mmHg to be elevated (“high”) BP and recommend pharmacotherapy for BP > 130/80 mmHg if the estimated 10-year risk of cardiovascular events exceeds 10%, regardless of age. These recommendations are given with reference to two meta-analyses, but the studies included in the referred meta-analyses lack direct relevance, as patients in the first meta-analysis had established hypertension and several cardiovascular risk conditions. Further, the estimation of risk levels in the second meta-analysis was not done a priori with the recommended SCORE2 risk tool, but through a post hoc analysis of cardiovascular death in the placebo group. We argue that no randomized study has provided BP treatment based on such risk algorithms, nor from healthy participants with “elevated BP” (120–139/70–89 mmHg). A further problem is the use of a fixed risk threshold of 10% regardless of age, which will shift treatment to older patients without any major risk factors, while younger individuals with early onset of high BP and other metabolic syndrome characteristics usually will have a 10-year estimated cardiovascular risk far below this threshold, thus treatment may be delayed. This concept of the 2024 ESC hypertension guidelines is not evidence-based and should, in our opinion, not be implemented in Norway or in any other country. Norwegian doctors should follow the National Directorate of Health’s guidelines with age-adjusted intervention thresholds and consult European Society of Hypertension guidelines if they need to consider more extensive patient recommendations.
New guidelines for high blood pressure (BP) by the European Society of Cardiology (ESC) now consider BP of 120–140/70–90 mmHg to be elevated (“high”) BP and recommend pharmacotherapy for BP > 130/80 mmHg if the estimated 10-year risk of cardiovascular events exceeds 10%, regardless of age. These recommendations are given with reference to two meta-analyses, but the studies included in the referred meta-analyses lack direct relevance, as patients in the first meta-analysis had established hypertension and several cardiovascular risk conditions. Further, the estimation of risk levels in the second meta-analysis was not done a priori with the recommended SCORE2 risk tool, but through a post hoc analysis of cardiovascular death in the placebo group. We argue that no randomized study has provided BP treatment based on such risk algorithms, nor from healthy participants with “elevated BP” (120–139/70–89 mmHg). A further problem is the use of a fixed risk threshold of 10% regardless of age, which will shift treatment to older patients without any major risk factors, while younger individuals with early onset of high BP and other metabolic syndrome characteristics usually will have a 10-year estimated cardiovascular risk far below this threshold, thus treatment may be delayed. This concept of the 2024 ESC hypertension guidelines is not evidence-based and should, in our opinion, not be implemented in Norway or in any other country. Norwegian doctors should follow the National Directorate of Health’s guidelines with age-adjusted intervention thresholds and consult European Society of Hypertension guidelines if they need to consider more extensive patient recommendations.
DOI: https://doi.org/10.37349/emed.2026.1001390
This article belongs to the special issue Drug Adherence in Hypertension

Background:
Transcatheter mitral valve intervention, including repair (e.g., edge-to-edge repair) and replacement, is now a cornerstone of treatment for severe mitral regurgitation (MR) in high-risk patients, providing a less invasive alternative to surgical treatment. The purpose of this systematic review is to assess the efficacy and safety of different antithrombotic strategies to prevent thromboembolic events and bleeding complications after transcatheter mitral valve repair (TMVR).
Methods:
According to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a systematic literature search on PubMed, Embase, Web of Science, Scopus, and Cochrane Library was conducted from January 2015 to January 2025. Eligible studies included adult patients who underwent transcatheter mitral valve intervention (including repair and replacement procedures) and compared different antithrombotic regimens, including direct oral anticoagulants (DOACs), vitamin K antagonists (VKAs), and antiplatelet therapies. Two reviewers independently extracted data and assessed quality. The studies were too heterogeneous, so a narrative synthesis was performed.
Results:
Fifteen studies involving 20,956 patients were included. DOACs were associated with a lower risk of major bleeding compared to VKAs [hazard ratio (HR): 0.21, p = 0.02, in one large study], with similar rates of stroke. Mortality was lower with DOACs in several analyses (e.g., HR: 0.67). Triple therapy and dual antiplatelet therapy (DAPT) were associated with increased bleeding risk without providing additional thromboembolic protection.
Discussion:
DOACs have a safer profile in post-TMVR patients, with reduced bleeding risk and lower mortality compared with VKAs. Triple therapy and DAPT should only be used in high-risk patients with specific indications due to their greater risk of bleeding. Optimizing outcomes requires a tailored approach to antithrombotic therapy, considering patient factors and procedural considerations. Definitive standards may still demand further investigation, such as multicenter randomized controlled trials evaluating antithrombotic treatments after TMVR.
Background:
Transcatheter mitral valve intervention, including repair (e.g., edge-to-edge repair) and replacement, is now a cornerstone of treatment for severe mitral regurgitation (MR) in high-risk patients, providing a less invasive alternative to surgical treatment. The purpose of this systematic review is to assess the efficacy and safety of different antithrombotic strategies to prevent thromboembolic events and bleeding complications after transcatheter mitral valve repair (TMVR).
Methods:
According to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a systematic literature search on PubMed, Embase, Web of Science, Scopus, and Cochrane Library was conducted from January 2015 to January 2025. Eligible studies included adult patients who underwent transcatheter mitral valve intervention (including repair and replacement procedures) and compared different antithrombotic regimens, including direct oral anticoagulants (DOACs), vitamin K antagonists (VKAs), and antiplatelet therapies. Two reviewers independently extracted data and assessed quality. The studies were too heterogeneous, so a narrative synthesis was performed.
Results:
Fifteen studies involving 20,956 patients were included. DOACs were associated with a lower risk of major bleeding compared to VKAs [hazard ratio (HR): 0.21, p = 0.02, in one large study], with similar rates of stroke. Mortality was lower with DOACs in several analyses (e.g., HR: 0.67). Triple therapy and dual antiplatelet therapy (DAPT) were associated with increased bleeding risk without providing additional thromboembolic protection.
Discussion:
DOACs have a safer profile in post-TMVR patients, with reduced bleeding risk and lower mortality compared with VKAs. Triple therapy and DAPT should only be used in high-risk patients with specific indications due to their greater risk of bleeding. Optimizing outcomes requires a tailored approach to antithrombotic therapy, considering patient factors and procedural considerations. Definitive standards may still demand further investigation, such as multicenter randomized controlled trials evaluating antithrombotic treatments after TMVR.
DOI: https://doi.org/10.37349/emed.2026.1001389

Opioid use disorder (OUD) is an emerging clinical and public health concern in cancer care. Although opioids remain essential for cancer pain management, a substantial subset of patients develops OUD, including many iatrogenic cases. This scoping review (ScR) synthesized evidence on the epidemiology, risk factors, screening approaches, and multidisciplinary management of OUD in adults with cancer. The review followed Joanna Briggs Institute (JBI) and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)-ScR guidelines. Four databases (PubMed, Medline, Scopus, ScienceDirect) were searched for English language randomized trials, quasi-experimental studies, and observational studies published from 2019–2025. Eligible studies included adults aged ≥ 18 years with cancer. Two independent reviewers conducted the study selection (OA and YI). The search identified 1,044 records; 403 abstracts were screened; 164 full-texts were assessed; and 46 studies met the inclusion criteria. OUD prevalence averaged 8% (range 6–50%), with 23.5% of patients classified as at risk. New persistent opioid use occurred in 19% within 8 weeks. Postoperative persistent opioid use was 33.3% overall, higher among patients with prior opioid exposure (48.3%) than opioid-naive individuals (18.5%). Reported risk factors included prior opioid use, younger age, male sex, substance use history, anxiety, and financial distress. Screening tools identified approximately 20% of patients as high risk. Multidisciplinary interventions—including buprenorphine/naloxone treatment and structured monitoring—were effective in managing concurrent cancer pain and OUD. OUD in cancer populations is preventable and manageable through systematic risk assessment and integrated multidisciplinary care. Early screening with validated tools, structured monitoring, and compassionate communication support safe opioid use while maintaining adequate analgesia. Evidence supports expanding access to medication-assisted therapies, psychosocial support, and harm-reduction strategies. These findings provide a foundation for improving clinical outcomes and guiding future research.
Opioid use disorder (OUD) is an emerging clinical and public health concern in cancer care. Although opioids remain essential for cancer pain management, a substantial subset of patients develops OUD, including many iatrogenic cases. This scoping review (ScR) synthesized evidence on the epidemiology, risk factors, screening approaches, and multidisciplinary management of OUD in adults with cancer. The review followed Joanna Briggs Institute (JBI) and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)-ScR guidelines. Four databases (PubMed, Medline, Scopus, ScienceDirect) were searched for English language randomized trials, quasi-experimental studies, and observational studies published from 2019–2025. Eligible studies included adults aged ≥ 18 years with cancer. Two independent reviewers conducted the study selection (OA and YI). The search identified 1,044 records; 403 abstracts were screened; 164 full-texts were assessed; and 46 studies met the inclusion criteria. OUD prevalence averaged 8% (range 6–50%), with 23.5% of patients classified as at risk. New persistent opioid use occurred in 19% within 8 weeks. Postoperative persistent opioid use was 33.3% overall, higher among patients with prior opioid exposure (48.3%) than opioid-naive individuals (18.5%). Reported risk factors included prior opioid use, younger age, male sex, substance use history, anxiety, and financial distress. Screening tools identified approximately 20% of patients as high risk. Multidisciplinary interventions—including buprenorphine/naloxone treatment and structured monitoring—were effective in managing concurrent cancer pain and OUD. OUD in cancer populations is preventable and manageable through systematic risk assessment and integrated multidisciplinary care. Early screening with validated tools, structured monitoring, and compassionate communication support safe opioid use while maintaining adequate analgesia. Evidence supports expanding access to medication-assisted therapies, psychosocial support, and harm-reduction strategies. These findings provide a foundation for improving clinical outcomes and guiding future research.
DOI: https://doi.org/10.37349/emed.2026.1001388
This article belongs to the special issue Practical Tips for Cancer Care: Guidance for Patients, Caregivers, and Healthcare Professionals
 Previous
Previous