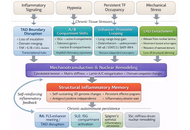
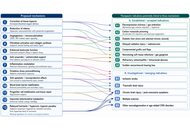
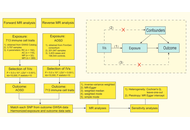


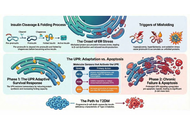

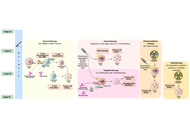

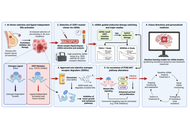




Autoimmune rheumatic diseases arise when the immune system transitions from a flexible, self-regulating network into a metabolically and epigenetically fixed inflammatory attractor state. This review synthesizes emerging evidence that immune tolerance is governed by a coupled epigenetic-metabolic axis integrating mitochondrial fitness, chromatin accessibility, redox balance, and nutrient flux across lymphoid, myeloid, and stromal compartments. We examine how chronic cytokine signaling, hypoxia, and oxidative stress destabilize regulatory programs, imprint glycolytic effector states, and remodel enhancer landscapes, thereby sustaining autoreactive circuits even after inflammatory pathways are pharmacologically suppressed. Multi-omic and spatial analyses reveal that pathogenic chromatin architectures, persistent mitochondrial dysfunction, and intercellular metabolite exchange cooperate to establish self-sustaining inflammatory ecosystems in rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), systemic sclerosis (SSc), and Sjögren’s syndrome. We further highlight therapeutic strategies aimed at tolerance reprogramming, including metabolic correction, chromatin-targeted agents, chimeric antigen receptor regulatory T cells (CAR-Tregs), tolerogenic dendritic cells, and integrative biomarkers that quantify metabolic-epigenetic coherence. By reframing autoimmunity as a disorder of energetic and chromatin desynchronization rather than isolated immune activation, this review outlines a mechanistic path toward durable, drug-free remission through deliberate restoration of the molecular architecture that maintains immune self-recognition.
Autoimmune rheumatic diseases arise when the immune system transitions from a flexible, self-regulating network into a metabolically and epigenetically fixed inflammatory attractor state. This review synthesizes emerging evidence that immune tolerance is governed by a coupled epigenetic-metabolic axis integrating mitochondrial fitness, chromatin accessibility, redox balance, and nutrient flux across lymphoid, myeloid, and stromal compartments. We examine how chronic cytokine signaling, hypoxia, and oxidative stress destabilize regulatory programs, imprint glycolytic effector states, and remodel enhancer landscapes, thereby sustaining autoreactive circuits even after inflammatory pathways are pharmacologically suppressed. Multi-omic and spatial analyses reveal that pathogenic chromatin architectures, persistent mitochondrial dysfunction, and intercellular metabolite exchange cooperate to establish self-sustaining inflammatory ecosystems in rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), systemic sclerosis (SSc), and Sjögren’s syndrome. We further highlight therapeutic strategies aimed at tolerance reprogramming, including metabolic correction, chromatin-targeted agents, chimeric antigen receptor regulatory T cells (CAR-Tregs), tolerogenic dendritic cells, and integrative biomarkers that quantify metabolic-epigenetic coherence. By reframing autoimmunity as a disorder of energetic and chromatin desynchronization rather than isolated immune activation, this review outlines a mechanistic path toward durable, drug-free remission through deliberate restoration of the molecular architecture that maintains immune self-recognition.
DOI: https://doi.org/10.37349/ei.2026.1003255

This Perspective explores why hyperbaric oxygen therapy (HBOT) deserves closer clinical and scientific attention as a possible treatment for ischemic stroke and a potential neuroprotective strategy in chronic central nervous system disorders, especially multiple sclerosis and other progressive or age-related neurological conditions. While HBOT is not yet widely accepted for these indications, three factors justify reconsideration: its strong safety profile, a biologically plausible mechanistic rationale, and recurrent signals of benefit from selected neurological studies. We review HBOT’s historical development, current accepted indications, and evidence that adverse effects are generally mild, uncommon, and reversible under modern protocols. In neurology, efficacy remains unproven, yet interest persists due to preliminary findings and repeated patient-reported improvements. A central argument is that the main barrier to progress is not safety, but evidence generation. Conventional randomized controlled trials face major challenges: difficult blinding, wide variation in dosing protocols, and uncertainty about meaningful outcomes in chronic neurological disease. Moreover, HBOT is non-patentable, which limits commercial investment and leaves a potentially valuable intervention underexplored. Mechanistically, we move beyond explanations centred solely on oxygen delivery or oxidative stress. As a working hypothesis, neurological benefits may partly arise from cumulative adaptive responses—including a rebound hormesis following repeated hyperoxic exposure. We conclude with a pragmatic research agenda: continuous, low-cost physiological monitoring in patients already receiving HBOT, coupled with a medium-term goal of adequately powered efficacy trials.
This Perspective explores why hyperbaric oxygen therapy (HBOT) deserves closer clinical and scientific attention as a possible treatment for ischemic stroke and a potential neuroprotective strategy in chronic central nervous system disorders, especially multiple sclerosis and other progressive or age-related neurological conditions. While HBOT is not yet widely accepted for these indications, three factors justify reconsideration: its strong safety profile, a biologically plausible mechanistic rationale, and recurrent signals of benefit from selected neurological studies. We review HBOT’s historical development, current accepted indications, and evidence that adverse effects are generally mild, uncommon, and reversible under modern protocols. In neurology, efficacy remains unproven, yet interest persists due to preliminary findings and repeated patient-reported improvements. A central argument is that the main barrier to progress is not safety, but evidence generation. Conventional randomized controlled trials face major challenges: difficult blinding, wide variation in dosing protocols, and uncertainty about meaningful outcomes in chronic neurological disease. Moreover, HBOT is non-patentable, which limits commercial investment and leaves a potentially valuable intervention underexplored. Mechanistically, we move beyond explanations centred solely on oxygen delivery or oxidative stress. As a working hypothesis, neurological benefits may partly arise from cumulative adaptive responses—including a rebound hormesis following repeated hyperoxic exposure. We conclude with a pragmatic research agenda: continuous, low-cost physiological monitoring in patients already receiving HBOT, coupled with a medium-term goal of adequately powered efficacy trials.
DOI: https://doi.org/10.37349/ent.2026.1004157

Aim:
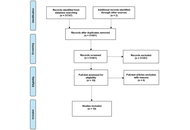
Adult-onset Still’s disease (AOSD) is a rare systemic inflammatory disorder marked by fever, rash, joint pain, and hyperferritinemia. While immune dysregulation is implicated in AOSD, the exact causal mechanisms remain unclear. This study aimed to investigate the genetic causal relationship between 731 immune cell phenotypes and AOSD, and to identify protective or risk-associated profiles.
Methods:
Using a two-sample Mendelian randomization (TSMR) approach, we applied inverse variance weighted (IVW) as the primary method, supplemented by MR-Egger, weighted median, simple mode, and weighted mode methods for robustness. Genetic instrumental variables for immune traits were sourced from recent genome-wide association studies (GWAS), and AOSD genetic predispositions were derived from the finn-b-STILL_ADULT cohort, comprising 201,947 individuals of European ancestry (3,403 AOSD cases and 198,544 controls).
Results:
We identified 49 immune cell-related traits showing nominally significant associations with AOSD (all adjusted P > 0.05 after FDR correction). Among these, 34 traits showed nominally protective trends, while 15 showed nominally risk-associated trends. Reciprocally, AOSD showed nominally suggestive effects on 40 immune cell traits, with 25 exhibiting a trend toward decreased levels and 15 toward increased levels. Additionally, we conducted multiple sensitivity analyses to explore potential heterogeneity and pleiotropy, though the primary findings did not survive FDR correction.
Conclusions:
These nominally significant associations between immune cell traits and AOSD, though not surviving FDR correction, may offer hypothesis-generating insights for future therapeutic research. The observed directional trends—with certain traits showing nominally protective or risk-associated patterns—suggest potential avenues for further exploration in the development of targeted treatment approaches for AOSD.
Aim:
Adult-onset Still’s disease (AOSD) is a rare systemic inflammatory disorder marked by fever, rash, joint pain, and hyperferritinemia. While immune dysregulation is implicated in AOSD, the exact causal mechanisms remain unclear. This study aimed to investigate the genetic causal relationship between 731 immune cell phenotypes and AOSD, and to identify protective or risk-associated profiles.
Methods:
Using a two-sample Mendelian randomization (TSMR) approach, we applied inverse variance weighted (IVW) as the primary method, supplemented by MR-Egger, weighted median, simple mode, and weighted mode methods for robustness. Genetic instrumental variables for immune traits were sourced from recent genome-wide association studies (GWAS), and AOSD genetic predispositions were derived from the finn-b-STILL_ADULT cohort, comprising 201,947 individuals of European ancestry (3,403 AOSD cases and 198,544 controls).
Results:
We identified 49 immune cell-related traits showing nominally significant associations with AOSD (all adjusted P > 0.05 after FDR correction). Among these, 34 traits showed nominally protective trends, while 15 showed nominally risk-associated trends. Reciprocally, AOSD showed nominally suggestive effects on 40 immune cell traits, with 25 exhibiting a trend toward decreased levels and 15 toward increased levels. Additionally, we conducted multiple sensitivity analyses to explore potential heterogeneity and pleiotropy, though the primary findings did not survive FDR correction.
Conclusions:
These nominally significant associations between immune cell traits and AOSD, though not surviving FDR correction, may offer hypothesis-generating insights for future therapeutic research. The observed directional trends—with certain traits showing nominally protective or risk-associated patterns—suggest potential avenues for further exploration in the development of targeted treatment approaches for AOSD.
DOI: https://doi.org/10.37349/emed.2026.1001406

Tricuspid valve endocarditis has potential associations with various conditions, but it is commonly related to intravenous drug abuse. Often, its eradication can become very challenging due to high post-operative mortality and high rate of recurrence due to persistence of drug abuse habits. The bidirectional Glenn shunt (BGS), typically employed in congenital heart surgery, combined with tricuspid valvectomy, has occasionally been used for recurrent endocarditis. Herein, we present a 31-year-old woman with drug addiction scheduled for her fourth reintervention due to the early degeneration and infection of a previous pulmonary homograft used for recurring tricuspid valve endocarditis. The final surgical strategy was valvectomy and BGS, aimed at eliminating all potential sources of infection and providing more time for the patient to overcome drug addiction, enabling further treatment if necessary.
Tricuspid valve endocarditis has potential associations with various conditions, but it is commonly related to intravenous drug abuse. Often, its eradication can become very challenging due to high post-operative mortality and high rate of recurrence due to persistence of drug abuse habits. The bidirectional Glenn shunt (BGS), typically employed in congenital heart surgery, combined with tricuspid valvectomy, has occasionally been used for recurrent endocarditis. Herein, we present a 31-year-old woman with drug addiction scheduled for her fourth reintervention due to the early degeneration and infection of a previous pulmonary homograft used for recurring tricuspid valve endocarditis. The final surgical strategy was valvectomy and BGS, aimed at eliminating all potential sources of infection and providing more time for the patient to overcome drug addiction, enabling further treatment if necessary.
DOI: https://doi.org/10.37349/emed.2026.1001407

Aim:
This study focused on evaluating the bioaccessibility and antioxidant potential of the protein and phenolic compounds present in a beverage (
Methods:
The
Results:
After SGID, protein digestibility of
Conclusions:
This product demonstrated potential as an antioxidant functional beverage, which should be further confirmed by additional in vitro and in vivo assays. It also remains pending sensory and microbiological safety evaluations to obtain a final product that is both organoleptically acceptable and safe for consumption.
Aim:
This study focused on evaluating the bioaccessibility and antioxidant potential of the protein and phenolic compounds present in a beverage (
Methods:
The
Results:
After SGID, protein digestibility of
Conclusions:
This product demonstrated potential as an antioxidant functional beverage, which should be further confirmed by additional in vitro and in vivo assays. It also remains pending sensory and microbiological safety evaluations to obtain a final product that is both organoleptically acceptable and safe for consumption.
DOI: https://doi.org/10.37349/eff.2026.1010155
This article belongs to the special issue Natural Bioactive Compounds in Functional Foods: From Foodomics Insights to Clinical Relevance

Aim:
This study aimed to develop a rice and sautéed mung bean meal using freeze-drying to preserve its safety, nutritional quality, and sensory attributes, providing a nutrient-dense meal suitable for disaster response and emergency feeding.
Methods:
White rice, mung beans, smoked herring, horseradish leaves, and seasonings were prepared, cooked, and freeze-dried. The freeze-dried product was vacuum-packed and evaluated for microbiological safety, physicochemical and proximate composition, micronutrient content (iron and vitamin A), and sensory acceptability by 50 Filipino panelists using a nine-point hedonic scale. Statistical comparisons with the traditional cooked meal were performed using paired t-tests.
Results:
The freeze-dried meal exhibited low microbial counts water activity, and moisture content, confirming its safety. Proximate analysis showed high protein (21.69 g/100 g), moderate carbohydrates (65.53 g/100 g), low fat (7.03 g/100 g), and total energy of 412.15 kcal/100 g. Micronutrient content per 117 g serving was 0.35 mg iron and 10.56 μg retinol equivalents vitamin A. Sensory evaluation revealed high acceptability for aroma, taste, and texture, while appearance and color showed minor reductions compared to the control, with statistically significant differences (p < 0.05) in some attributes.
Conclusions:
Freeze-drying effectively produced a safe, nutrient-rich, and sensorially acceptable instant rice and mung bean meal. The product demonstrates strong potential for long-shelf-life and is a convenient option for disaster response and emergency feeding, though further optimization may improve visual appeal.
Aim:
This study aimed to develop a rice and sautéed mung bean meal using freeze-drying to preserve its safety, nutritional quality, and sensory attributes, providing a nutrient-dense meal suitable for disaster response and emergency feeding.
Methods:
White rice, mung beans, smoked herring, horseradish leaves, and seasonings were prepared, cooked, and freeze-dried. The freeze-dried product was vacuum-packed and evaluated for microbiological safety, physicochemical and proximate composition, micronutrient content (iron and vitamin A), and sensory acceptability by 50 Filipino panelists using a nine-point hedonic scale. Statistical comparisons with the traditional cooked meal were performed using paired t-tests.
Results:
The freeze-dried meal exhibited low microbial counts water activity, and moisture content, confirming its safety. Proximate analysis showed high protein (21.69 g/100 g), moderate carbohydrates (65.53 g/100 g), low fat (7.03 g/100 g), and total energy of 412.15 kcal/100 g. Micronutrient content per 117 g serving was 0.35 mg iron and 10.56 μg retinol equivalents vitamin A. Sensory evaluation revealed high acceptability for aroma, taste, and texture, while appearance and color showed minor reductions compared to the control, with statistically significant differences (p < 0.05) in some attributes.
Conclusions:
Freeze-drying effectively produced a safe, nutrient-rich, and sensorially acceptable instant rice and mung bean meal. The product demonstrates strong potential for long-shelf-life and is a convenient option for disaster response and emergency feeding, though further optimization may improve visual appeal.
DOI: https://doi.org/10.37349/eff.2026.1010156

Background:
Diabetes mellitus (DM) refers to a group of metabolic diseases characterized by reduced insulin synthesis and release, leading to hyperglycemia, oxidative stress, and inflammation. The prevalence of DM continues to increase worldwide, primarily driven by type 2 DM (T2DM). While numerous prescription drugs are widely available for the management of T2DM, these treatments must be coupled with significant changes in diet and lifestyle to have long-term effects. This comparative review aims to summarize and critically evaluate some of the dietary antioxidant and anti-inflammatory functional foods (FFs), including bioactive compounds on glycemic regulation, oxidative stress, inflammatory markers, and other outcomes in T2DM patients.
Methods:
A comprehensive literature search was performed from June 2014 to December 2025 in Cochrane Library, Embase, Google Scholar, PubMed/Medline, and Scopus databases.
Results:
Thirty-one meta-analyses describing the effects of specific FFs on glycemic outcomes, including fasting blood glucose, HbA1c, insulin levels, insulin resistance, oxidative or inflammatory stress, and/or the doses needed to achieve these endpoints, in T2DM patients were included. Comparative analyses of the data for green tea in T2DM showed significant antioxidant effects, but conflicting data for glycemic regulation, and no significant effect on inflammatory markers. Flaxseed products significantly reduced oxidative stress and HbA1c but had no anti-inflammatory effects. Both anthocyanins and resveratrol from fruits and vegetables significantly reduced fasting blood glucose, HbA1c, as well as oxidative and inflammatory markers. Turmeric and curcumin significantly reduced fasting blood glucose, but the data for ginger were conflicted. In T2DM patients, turmeric and ginger significantly reduced markers of oxidation and inflammation.
Discussion:
Overall, improvements in diet, including the incorporation of specific FFs and exercise, along with standard treatments, may be cost-effective methods to reduce the public health impact of T2DM. Limitations included the significant heterogeneity, high risk of bias, and low to moderate quality of the randomized controlled trial (RCT).
Background:
Diabetes mellitus (DM) refers to a group of metabolic diseases characterized by reduced insulin synthesis and release, leading to hyperglycemia, oxidative stress, and inflammation. The prevalence of DM continues to increase worldwide, primarily driven by type 2 DM (T2DM). While numerous prescription drugs are widely available for the management of T2DM, these treatments must be coupled with significant changes in diet and lifestyle to have long-term effects. This comparative review aims to summarize and critically evaluate some of the dietary antioxidant and anti-inflammatory functional foods (FFs), including bioactive compounds on glycemic regulation, oxidative stress, inflammatory markers, and other outcomes in T2DM patients.
Methods:
A comprehensive literature search was performed from June 2014 to December 2025 in Cochrane Library, Embase, Google Scholar, PubMed/Medline, and Scopus databases.
Results:
Thirty-one meta-analyses describing the effects of specific FFs on glycemic outcomes, including fasting blood glucose, HbA1c, insulin levels, insulin resistance, oxidative or inflammatory stress, and/or the doses needed to achieve these endpoints, in T2DM patients were included. Comparative analyses of the data for green tea in T2DM showed significant antioxidant effects, but conflicting data for glycemic regulation, and no significant effect on inflammatory markers. Flaxseed products significantly reduced oxidative stress and HbA1c but had no anti-inflammatory effects. Both anthocyanins and resveratrol from fruits and vegetables significantly reduced fasting blood glucose, HbA1c, as well as oxidative and inflammatory markers. Turmeric and curcumin significantly reduced fasting blood glucose, but the data for ginger were conflicted. In T2DM patients, turmeric and ginger significantly reduced markers of oxidation and inflammation.
Discussion:
Overall, improvements in diet, including the incorporation of specific FFs and exercise, along with standard treatments, may be cost-effective methods to reduce the public health impact of T2DM. Limitations included the significant heterogeneity, high risk of bias, and low to moderate quality of the randomized controlled trial (RCT).
DOI: https://doi.org/10.37349/eemd.2026.101474

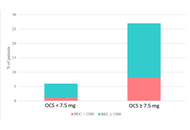
Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare systemic vasculitis with heterogeneous clinical manifestations. Identifying reliable biomarkers is crucial to predicting disease evolution and guiding therapy. We analyzed clinical, biological, and functional data from 33 patients with EGPA in the vasculitic phase. Blood eosinophil count (BEC), eosinophilic cationic protein (ECP), antineutrophil cytoplasmic antibodies (ANCAs), specific IgE to staphylococcal enterotoxins (SE-IgE), and serum free light chains (FLCs) were evaluated. Severe eosinophilic asthma (SEA) and chronic rhinosinusitis with nasal polyps (CRSwNPs) were present in 100% and 88.5% of patients, respectively. Median BEC was 1950 cells/mm3, with elevated ECP (60.0 µg/L). SE-IgE was detected in 54.2% of patients. A significant negative correlation emerged between λ FLCs and oral corticosteroid (OCS) dose (r = –0.58, p = 0.009). Forced expiratory volume in 1 second (FEV1) was significantly lower in C-ANCA+ patients (p = 0.006). ECP and SE-IgE may serve as markers of eosinophilic activity and epithelial barrier damage in EGPA. λ FLCs might be a useful indicator of OCS exposure and treatment response. These biomarkers could support the evaluation of disease evolution and treatment response.
Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare systemic vasculitis with heterogeneous clinical manifestations. Identifying reliable biomarkers is crucial to predicting disease evolution and guiding therapy. We analyzed clinical, biological, and functional data from 33 patients with EGPA in the vasculitic phase. Blood eosinophil count (BEC), eosinophilic cationic protein (ECP), antineutrophil cytoplasmic antibodies (ANCAs), specific IgE to staphylococcal enterotoxins (SE-IgE), and serum free light chains (FLCs) were evaluated. Severe eosinophilic asthma (SEA) and chronic rhinosinusitis with nasal polyps (CRSwNPs) were present in 100% and 88.5% of patients, respectively. Median BEC was 1950 cells/mm3, with elevated ECP (60.0 µg/L). SE-IgE was detected in 54.2% of patients. A significant negative correlation emerged between λ FLCs and oral corticosteroid (OCS) dose (r = –0.58, p = 0.009). Forced expiratory volume in 1 second (FEV1) was significantly lower in C-ANCA+ patients (p = 0.006). ECP and SE-IgE may serve as markers of eosinophilic activity and epithelial barrier damage in EGPA. λ FLCs might be a useful indicator of OCS exposure and treatment response. These biomarkers could support the evaluation of disease evolution and treatment response.
DOI: https://doi.org/10.37349/eaa.2026.1009127
This article belongs to the special issue Environment, Infectious Diseases, and Allergy

Aim:
Two empirical methods were used to predict the absolute bioavailability (F) of medicines in adults and children following oral administration in the absence of intravenous (IV) dosing. This study systematically evaluates the predictive performance of Equation 1 to predict F in adults and children.
Methods:
Equation 3 [F = Q/(Q + CLoral)] was used for the prediction of F in adults and children. In Equation 3, clearance is the observed oral clearance following oral administration of a medicine and Q is either liver blood or plasma flow rate. The predictive performance of Equation 3 was evaluated in adults and children for three categories of medicines; medicines which are mainly metabolized in the liver, medicines which are metabolized both in the liver and the gut, and medicines which are mainly renally excreted. From the literature, oral clearance and F values for adults and children were obtained. The predictive performance of these two methods (blood or plasma flow rate) was assessed by comparing the predicted F of the medicines used in this study with the observed F (obtained from clinical studies).
Results:
More than 90% predicted F values were within 0.5–2-fold prediction error in adults and children by both methods for all three categories of medicines. Plasma flow rate provided slightly better results than the blood flow rate.
Conclusions:
The proposed methods indicate that the estimation of F of medicines in adults and children is possible with reasonable accuracy (within 0.5–2-fold prediction error). The method is useful to estimate F, especially in children, because it is not ethical to administer medicines by both IV and oral routes to children just for the sake of estimating F.
Aim:
Two empirical methods were used to predict the absolute bioavailability (F) of medicines in adults and children following oral administration in the absence of intravenous (IV) dosing. This study systematically evaluates the predictive performance of Equation 1 to predict F in adults and children.
Methods:
Equation 3 [F = Q/(Q + CLoral)] was used for the prediction of F in adults and children. In Equation 3, clearance is the observed oral clearance following oral administration of a medicine and Q is either liver blood or plasma flow rate. The predictive performance of Equation 3 was evaluated in adults and children for three categories of medicines; medicines which are mainly metabolized in the liver, medicines which are metabolized both in the liver and the gut, and medicines which are mainly renally excreted. From the literature, oral clearance and F values for adults and children were obtained. The predictive performance of these two methods (blood or plasma flow rate) was assessed by comparing the predicted F of the medicines used in this study with the observed F (obtained from clinical studies).
Results:
More than 90% predicted F values were within 0.5–2-fold prediction error in adults and children by both methods for all three categories of medicines. Plasma flow rate provided slightly better results than the blood flow rate.
Conclusions:
The proposed methods indicate that the estimation of F of medicines in adults and children is possible with reasonable accuracy (within 0.5–2-fold prediction error). The method is useful to estimate F, especially in children, because it is not ethical to administer medicines by both IV and oral routes to children just for the sake of estimating F.
DOI: https://doi.org/10.37349/eds.2026.1008160

Immunotherapy is a promising treatment strategy for treating colorectal cancer (CRC). Despite significant advances in this field, resistance and low efficacy of immunotherapy remain a principal problem. One of the most important factors affecting the response to immunotherapy is the tumor microenvironment (TME). Among the components of the TME, tumor-associated macrophages (TAMs) are key immune cells involved in cancer progression by stimulating tumor cell proliferation, angiogenesis, epithelial-mesenchymal transition, metastasis, and tumor immune evasion. This review presents currently investigated combination therapy based on the immune checkpoint inhibitors and inhibitors of diverse components of the TME, including TAMs, that can potentially increase the effectiveness of CRC treatment. Therapeutic efficacy, together with the functional activity of TAMs, is estimated in multiple preclinical data obtained with diverse in vitro and in vivo models. Ongoing clinical trials demonstrated the association of treatment effectiveness with TAM phenotypes and functions.
Immunotherapy is a promising treatment strategy for treating colorectal cancer (CRC). Despite significant advances in this field, resistance and low efficacy of immunotherapy remain a principal problem. One of the most important factors affecting the response to immunotherapy is the tumor microenvironment (TME). Among the components of the TME, tumor-associated macrophages (TAMs) are key immune cells involved in cancer progression by stimulating tumor cell proliferation, angiogenesis, epithelial-mesenchymal transition, metastasis, and tumor immune evasion. This review presents currently investigated combination therapy based on the immune checkpoint inhibitors and inhibitors of diverse components of the TME, including TAMs, that can potentially increase the effectiveness of CRC treatment. Therapeutic efficacy, together with the functional activity of TAMs, is estimated in multiple preclinical data obtained with diverse in vitro and in vivo models. Ongoing clinical trials demonstrated the association of treatment effectiveness with TAM phenotypes and functions.
DOI: https://doi.org/10.37349/edd.2026.1005124
This article belongs to the special issue Immunotherapy for Cancer of Digestive System

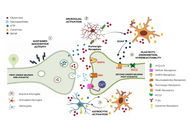
The intricate involvement of glial cells in chronic pain mechanisms represents a paradigm shift in our understanding of pain processing. From microglial-mediated neuroinflammation to astrocytic modulation of synaptic function, glial cells emerge as critical players in the complex neurobiology of chronic pain. As research continues to unravel the multifaceted roles of these cells, novel therapeutic targets and strategies are likely to emerge, potentially revolutionizing the management of chronic pain conditions. Chronic pain is complicated by frequent comorbidities such as fatigue, sleep disturbances, cognitive impairment, and mood disorders. Here, we hypothesize that neuroinflammatory processes are at the root of pain chronification and serve as the common thread linking characteristic hypersensitivity with the cognitive and emotional comorbidities typically associated with chronic pain.
The intricate involvement of glial cells in chronic pain mechanisms represents a paradigm shift in our understanding of pain processing. From microglial-mediated neuroinflammation to astrocytic modulation of synaptic function, glial cells emerge as critical players in the complex neurobiology of chronic pain. As research continues to unravel the multifaceted roles of these cells, novel therapeutic targets and strategies are likely to emerge, potentially revolutionizing the management of chronic pain conditions. Chronic pain is complicated by frequent comorbidities such as fatigue, sleep disturbances, cognitive impairment, and mood disorders. Here, we hypothesize that neuroinflammatory processes are at the root of pain chronification and serve as the common thread linking characteristic hypersensitivity with the cognitive and emotional comorbidities typically associated with chronic pain.
DOI: https://doi.org/10.37349/ei.2026.1003254
This article belongs to the special issue Immunology and Pain

Aim:
To assess the effect of a synbiotic supplement composed of Enterococcus faecium and agave inulin on cognitive function in older adults with mild cognitive impairment (MCI).
Methods:
In a triple-blind randomized crossover trial, nineteen adults aged 64–85 years with MCI received either the synbiotic or an isocaloric vehicle for eight weeks, followed by a three-week washout and treatment crossover. Cognitive outcomes were assessed at baseline, after the first intervention, and after crossover using the Modified Mini-Mental State Examination (MMSE), Rey-Osterrieth Complex Figure Test (RCFT; copy and memory), and Abbreviated Instrument for Expectations of Self-Efficacy for Daily Activities in Older Adults (AERAC) self-efficacy scale. Paired and unpaired Student’s t-tests were used for statistical comparisons (p < 0.05).
Results:
The synbiotic group showed significant improvement relative to baseline across all domains: MMSE (p = 0.05), AERAC (p = 0.005), RCFT-copy (p = 0.03), and RCFT-memory (p = 0.03). Post-treatment comparisons between groups also favored the synbiotic, with significant differences in MMSE (p = 0.001), AERAC (p = 0.001), RCFT-copy (p = 0.0095), and RCFT-memory (p = 0.001). After crossover, cognitive gains were sustained and reproduced. MMSE scores reached 17.89 ± 1.45 in the synbiotic-first group versus 18.20 ± 0.63 in the control-first group (p < 0.001). RCFT-copy remained high (29.83 ± 4.18 vs. 29.52 ± 5.60, p = 0.0157), while RCFT-memory scores differed (17.56 ± 6.73 vs. 17.20 ± 3.29, p = 0.0005). AERAC scores continued to improve during crossover (82.60 ± 10.49 vs. 85.46 ± 8.28, p < 0.001). No adverse effects occurred.
Conclusions:
Synbiotic supplementation significantly improved global cognition, visuoconstructive ability, memory, and functional self-efficacy in older adults with MCI. Benefits persisted beyond the initial intervention and were replicated when the control group received the synbiotic, supporting its potential as a safe and effective strategy to mitigate age-related cognitive decline.
Aim:
To assess the effect of a synbiotic supplement composed of Enterococcus faecium and agave inulin on cognitive function in older adults with mild cognitive impairment (MCI).
Methods:
In a triple-blind randomized crossover trial, nineteen adults aged 64–85 years with MCI received either the synbiotic or an isocaloric vehicle for eight weeks, followed by a three-week washout and treatment crossover. Cognitive outcomes were assessed at baseline, after the first intervention, and after crossover using the Modified Mini-Mental State Examination (MMSE), Rey-Osterrieth Complex Figure Test (RCFT; copy and memory), and Abbreviated Instrument for Expectations of Self-Efficacy for Daily Activities in Older Adults (AERAC) self-efficacy scale. Paired and unpaired Student’s t-tests were used for statistical comparisons (p < 0.05).
Results:
The synbiotic group showed significant improvement relative to baseline across all domains: MMSE (p = 0.05), AERAC (p = 0.005), RCFT-copy (p = 0.03), and RCFT-memory (p = 0.03). Post-treatment comparisons between groups also favored the synbiotic, with significant differences in MMSE (p = 0.001), AERAC (p = 0.001), RCFT-copy (p = 0.0095), and RCFT-memory (p = 0.001). After crossover, cognitive gains were sustained and reproduced. MMSE scores reached 17.89 ± 1.45 in the synbiotic-first group versus 18.20 ± 0.63 in the control-first group (p < 0.001). RCFT-copy remained high (29.83 ± 4.18 vs. 29.52 ± 5.60, p = 0.0157), while RCFT-memory scores differed (17.56 ± 6.73 vs. 17.20 ± 3.29, p = 0.0005). AERAC scores continued to improve during crossover (82.60 ± 10.49 vs. 85.46 ± 8.28, p < 0.001). No adverse effects occurred.
Conclusions:
Synbiotic supplementation significantly improved global cognition, visuoconstructive ability, memory, and functional self-efficacy in older adults with MCI. Benefits persisted beyond the initial intervention and were replicated when the control group received the synbiotic, supporting its potential as a safe and effective strategy to mitigate age-related cognitive decline.
DOI: https://doi.org/10.37349/ent.2026.1004156
This article belongs to the special issue Role of Microbiota in Neurological Diseases

Endocrine resistance in estrogen receptor-positive (ER+) breast cancer has undergone a fundamental reconceptualization over the past decade. The discovery that activating mutations in the ESR1 gene encoding ERα emerge under aromatase inhibitor (AI) selection pressure and drive ligand-independent receptor activation established a shift from empirical treatment sequencing to molecularly guided intervention. This review provides a synopsis of the structural biology underlying constitutive ER activation, the evolutionary dynamics of ESR1-mutant clones detectable through circulating tumor DNA (ctDNA), and the clinical evidence demonstrating that early molecular detection can trigger therapeutic switches that alter disease trajectory. The regulatory approval of elacestrant for ESR1-mutant disease and randomized trial data showing progression-free survival (PFS) benefit from ctDNA-guided endocrine switching (PADA-1, SERENA-6) position ESR1 genotyping as a dynamic biomarker with direct therapeutic implications. We examine the integration of oral selective ER degraders (SERDs) into treatment algorithms, the role of co-occurring alterations in the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway, and emerging directions, including machine learning approaches to ctDNA kinetics and adaptive trial designs that treat clonal evolution as an actionable target. The convergence of structural mechanisms, liquid biopsy technology, and biomarker-driven drug development provides a framework for precision oncology in endocrine-resistant breast cancer. While these advances are substantial, important challenges remain, including the lack of mature overall survival (OS) data from interception trials, cost and accessibility barriers to serial ctDNA monitoring in diverse global healthcare settings, the unresolved question of optimal therapeutic sequencing in patients with concurrent ESR1 and PI3K pathway alterations, and the need to distinguish clinically actionable low-variant allele frequency (VAF) ESR1 calls from background noise in liquid biopsies.
Endocrine resistance in estrogen receptor-positive (ER+) breast cancer has undergone a fundamental reconceptualization over the past decade. The discovery that activating mutations in the ESR1 gene encoding ERα emerge under aromatase inhibitor (AI) selection pressure and drive ligand-independent receptor activation established a shift from empirical treatment sequencing to molecularly guided intervention. This review provides a synopsis of the structural biology underlying constitutive ER activation, the evolutionary dynamics of ESR1-mutant clones detectable through circulating tumor DNA (ctDNA), and the clinical evidence demonstrating that early molecular detection can trigger therapeutic switches that alter disease trajectory. The regulatory approval of elacestrant for ESR1-mutant disease and randomized trial data showing progression-free survival (PFS) benefit from ctDNA-guided endocrine switching (PADA-1, SERENA-6) position ESR1 genotyping as a dynamic biomarker with direct therapeutic implications. We examine the integration of oral selective ER degraders (SERDs) into treatment algorithms, the role of co-occurring alterations in the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway, and emerging directions, including machine learning approaches to ctDNA kinetics and adaptive trial designs that treat clonal evolution as an actionable target. The convergence of structural mechanisms, liquid biopsy technology, and biomarker-driven drug development provides a framework for precision oncology in endocrine-resistant breast cancer. While these advances are substantial, important challenges remain, including the lack of mature overall survival (OS) data from interception trials, cost and accessibility barriers to serial ctDNA monitoring in diverse global healthcare settings, the unresolved question of optimal therapeutic sequencing in patients with concurrent ESR1 and PI3K pathway alterations, and the need to distinguish clinically actionable low-variant allele frequency (VAF) ESR1 calls from background noise in liquid biopsies.
DOI: https://doi.org/10.37349/etat.2026.1002375
This article belongs to the special issue Breaking Boundaries in Breast Cancer Care: Emerging Controversies and Innovation in Surgical and Medical Approaches

Aim:
The study investigated the nutritional, functional, antioxidant, and enzyme inhibitory properties of tigernut-wheat composite flours.
Methods:
Composite samples were prepared by substituting wheat flour with tigernut flour at 5–20%, while 100% wheat flour served as the control. Proximate, functional, antioxidant, and enzyme inhibitory analyses were carried out on the composite flours to determine the effects of tigernut substitution.
Results:
Proximate analysis revealed that tigernut addition significantly increased fiber (1.08–3.22%), ash (0.83–3.20%), and fat (2.61–7.32%) contents. While the crude protein content decreased slightly at higher substitution (13.00–6.91%), the carbohydrate content did not follow any specific pattern. Functional properties such as water absorption (73.13–85.26%) improved with tigernut incorporation, while bulk density and foaming capacity also showed positive trends. Antioxidant indices demonstrated substantial enhancement: total phenolic content (2.20–8.87 mg GAE/g) and improved radical scavenging activities. In addition, α-glucosidase inhibition rose from 27.25% in the control to 64.86% in the highest blend, while α-amylase inhibition declined, indicating potential benefits for moderating postprandial glycemia.
Conclusions:
Tigernut substitution enriched the mineral and phytochemical content of wheat flour and enhanced its functional and antioxidant properties. These findings suggest that tigernut-wheat composite flour could serve as a functional ingredient for bakery and snack formulations, offering improved nutritional quality and preliminary benefits related to glycemic modulation.
Aim:
The study investigated the nutritional, functional, antioxidant, and enzyme inhibitory properties of tigernut-wheat composite flours.
Methods:
Composite samples were prepared by substituting wheat flour with tigernut flour at 5–20%, while 100% wheat flour served as the control. Proximate, functional, antioxidant, and enzyme inhibitory analyses were carried out on the composite flours to determine the effects of tigernut substitution.
Results:
Proximate analysis revealed that tigernut addition significantly increased fiber (1.08–3.22%), ash (0.83–3.20%), and fat (2.61–7.32%) contents. While the crude protein content decreased slightly at higher substitution (13.00–6.91%), the carbohydrate content did not follow any specific pattern. Functional properties such as water absorption (73.13–85.26%) improved with tigernut incorporation, while bulk density and foaming capacity also showed positive trends. Antioxidant indices demonstrated substantial enhancement: total phenolic content (2.20–8.87 mg GAE/g) and improved radical scavenging activities. In addition, α-glucosidase inhibition rose from 27.25% in the control to 64.86% in the highest blend, while α-amylase inhibition declined, indicating potential benefits for moderating postprandial glycemia.
Conclusions:
Tigernut substitution enriched the mineral and phytochemical content of wheat flour and enhanced its functional and antioxidant properties. These findings suggest that tigernut-wheat composite flour could serve as a functional ingredient for bakery and snack formulations, offering improved nutritional quality and preliminary benefits related to glycemic modulation.
DOI: https://doi.org/10.37349/eff.2026.1010153

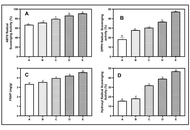
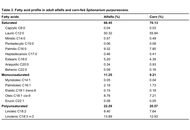
Aim:
To explore the potential contribution of ω-3 fatty acids and fiber from traditionally cooked adult chapulines (Sphenarium purpurascens) to the diet of consumers in the Sierra Sur region of Oaxaca.
Methods:
Exploratory study in two phases. In the first stage, preliminary interviews were conducted with retailers of the edible insect S. purpurascens at the traveling market in the city of Miahuatlán de Porfirio Díaz, Oaxaca. Proximate analyses were also performed in triplicate and fatty acid profiles were determined by gas chromatography on samples of cooked adult S. purpurascens fed on alfalfa and corn (Mesoamerican polyculture), collected at the same market. The information was analyzed using descriptive statistics, confidence interval comparisons, and Welch’s t-test. The second phase consisted of a survey (n = 144) to estimate the average intake (g/day) and frequency of consumption of S. purpurascens by its buyers in the aforementioned market. The information was analyzed using descriptive statistics and the Kruskal-Wallis test.
Results:
Alfalfa-fed S. purpurascens had 6.4 times more fiber (57.80% vs. 8.96%) than their corn-fed counterpart (p < 0.001), with the ω-6:ω-3 ratios being 0.60 and 0.59 in the corn- and alfalfa-fed samples, respectively. The most frequent consumption of S. purpurascens reported by buyers was 1–2 days per week (51%), and the average intake of all respondents was 27.5 g/day. This amount would provide 12.4% of the average daily requirement of omega-3 for adults and 39.7% of fiber if the alfalfa-fed insect is considered.
Conclusions:
Alfalfa-fed adult S. purpurascens provide a high fiber content to the diet of their consumers, and those fed alfalfa or corn provide a moderate amount of ω-3 fatty acids, which can help balance the ω-6:ω-3 ratio.
Aim:
To explore the potential contribution of ω-3 fatty acids and fiber from traditionally cooked adult chapulines (Sphenarium purpurascens) to the diet of consumers in the Sierra Sur region of Oaxaca.
Methods:
Exploratory study in two phases. In the first stage, preliminary interviews were conducted with retailers of the edible insect S. purpurascens at the traveling market in the city of Miahuatlán de Porfirio Díaz, Oaxaca. Proximate analyses were also performed in triplicate and fatty acid profiles were determined by gas chromatography on samples of cooked adult S. purpurascens fed on alfalfa and corn (Mesoamerican polyculture), collected at the same market. The information was analyzed using descriptive statistics, confidence interval comparisons, and Welch’s t-test. The second phase consisted of a survey (n = 144) to estimate the average intake (g/day) and frequency of consumption of S. purpurascens by its buyers in the aforementioned market. The information was analyzed using descriptive statistics and the Kruskal-Wallis test.
Results:
Alfalfa-fed S. purpurascens had 6.4 times more fiber (57.80% vs. 8.96%) than their corn-fed counterpart (p < 0.001), with the ω-6:ω-3 ratios being 0.60 and 0.59 in the corn- and alfalfa-fed samples, respectively. The most frequent consumption of S. purpurascens reported by buyers was 1–2 days per week (51%), and the average intake of all respondents was 27.5 g/day. This amount would provide 12.4% of the average daily requirement of omega-3 for adults and 39.7% of fiber if the alfalfa-fed insect is considered.
Conclusions:
Alfalfa-fed adult S. purpurascens provide a high fiber content to the diet of their consumers, and those fed alfalfa or corn provide a moderate amount of ω-3 fatty acids, which can help balance the ω-6:ω-3 ratio.
DOI: https://doi.org/10.37349/eff.2026.1010154

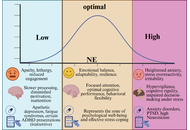
Norepinephrine (NE), a central catecholamine neurotransmitter synthesized primarily in the locus coeruleus (LC), plays a critical role in regulating arousal, attention, emotional processing, and stress responsiveness. While contemporary personality neuroscience has established the role of NE in acute psychological states, its contribution to stable personality traits remains underexplored. This review synthesizes neurobiological, psychological, genetic, and psychopharmacological evidence to propose a NE-personality continuum that links tonic and phasic dynamics of the LC-NE system to enduring individual differences in alertness, anxiety, and adaptability. Alertness is associated with optimal noradrenergic tone and efficient phasic signaling, which enhances the signal-to-noise ratio and attentional focus. Anxiety arises from chronic hyperactivation or dysregulated NE release, particularly involving excessive α1- and β-adrenergic receptor activity and impaired modulation from the prefrontal cortex. Adaptability denotes a harmonious interaction between the limbic system and prefrontal cortex, which facilitates cognitive flexibility and emotional regulation in response to changing environmental demands. The connection between NE activity and personality traits follows an inverted U-shaped pattern. Low tone leads to apathy and less engagement, moderate tone helps with resilience and optimal functioning, and high tone leads to hypervigilance and rigidity. This model combines findings from fundamental neuroscience and clinical research to provide a physiologically based framework for understanding how long-term variations in noradrenergic regulation affect personality traits, as described in established trait theories. The findings underline the feasibility of adding noradrenergic biomarkers and pharmaceutical therapies into clinical practice, as well as the importance of longitudinal and multimodal research to determine trait-level causality. This is especially important for understanding how to use these elements to improve treatment plans for personality disorders.
Norepinephrine (NE), a central catecholamine neurotransmitter synthesized primarily in the locus coeruleus (LC), plays a critical role in regulating arousal, attention, emotional processing, and stress responsiveness. While contemporary personality neuroscience has established the role of NE in acute psychological states, its contribution to stable personality traits remains underexplored. This review synthesizes neurobiological, psychological, genetic, and psychopharmacological evidence to propose a NE-personality continuum that links tonic and phasic dynamics of the LC-NE system to enduring individual differences in alertness, anxiety, and adaptability. Alertness is associated with optimal noradrenergic tone and efficient phasic signaling, which enhances the signal-to-noise ratio and attentional focus. Anxiety arises from chronic hyperactivation or dysregulated NE release, particularly involving excessive α1- and β-adrenergic receptor activity and impaired modulation from the prefrontal cortex. Adaptability denotes a harmonious interaction between the limbic system and prefrontal cortex, which facilitates cognitive flexibility and emotional regulation in response to changing environmental demands. The connection between NE activity and personality traits follows an inverted U-shaped pattern. Low tone leads to apathy and less engagement, moderate tone helps with resilience and optimal functioning, and high tone leads to hypervigilance and rigidity. This model combines findings from fundamental neuroscience and clinical research to provide a physiologically based framework for understanding how long-term variations in noradrenergic regulation affect personality traits, as described in established trait theories. The findings underline the feasibility of adding noradrenergic biomarkers and pharmaceutical therapies into clinical practice, as well as the importance of longitudinal and multimodal research to determine trait-level causality. This is especially important for understanding how to use these elements to improve treatment plans for personality disorders.
DOI: https://doi.org/10.37349/ent.2026.1004155

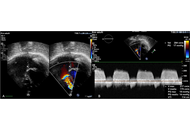
The recent improvements of the ultrasounds technology and the probes have led the dental community to start to apply this technology at macrovascular and microvascular level. This scoping review aims to investigate the diagnostic and research applications of ultrasonography in periodontics. A comprehensive literature search was conducted in PubMed, Scopus, and Web of Science, to identify relevant studies in periodontology: The search strategy included the following terms: “ultrasonography”, “echography”, “ultrasound”, “dentistry”. The review was conducted in accordance with the PRISMA-ScR guidelines. In addition, a manual search was conducted through the following journals from the last 10 years: Journal of Clinical Periodontology, the Journal of Dental Research and Oral Surgery, and Oral Medicine, Oral Pathology and Oral Radiology. Ten studies were included, covering different applications of ultrasonography in periodontology. Ultrasonography was employed in peri-implant and periodontal diagnosis, assessment of soft tissue thickness and vascularization, palatal wound and bone healing. This review highlights the effectiveness of ultrasonography in diagnosis and surgical evaluation. Further research, standardized protocols, and randomized clinical trials are needed, expanding the investigation to more fields relevant to the maxillofacial district.
The recent improvements of the ultrasounds technology and the probes have led the dental community to start to apply this technology at macrovascular and microvascular level. This scoping review aims to investigate the diagnostic and research applications of ultrasonography in periodontics. A comprehensive literature search was conducted in PubMed, Scopus, and Web of Science, to identify relevant studies in periodontology: The search strategy included the following terms: “ultrasonography”, “echography”, “ultrasound”, “dentistry”. The review was conducted in accordance with the PRISMA-ScR guidelines. In addition, a manual search was conducted through the following journals from the last 10 years: Journal of Clinical Periodontology, the Journal of Dental Research and Oral Surgery, and Oral Medicine, Oral Pathology and Oral Radiology. Ten studies were included, covering different applications of ultrasonography in periodontology. Ultrasonography was employed in peri-implant and periodontal diagnosis, assessment of soft tissue thickness and vascularization, palatal wound and bone healing. This review highlights the effectiveness of ultrasonography in diagnosis and surgical evaluation. Further research, standardized protocols, and randomized clinical trials are needed, expanding the investigation to more fields relevant to the maxillofacial district.
DOI: https://doi.org/10.37349/emed.2026.1001405

Aim:
To benchmark three deep learning-based retinal image registration methods RetinaRegNet, EyeLiner, and GeoFormer on the Fundus Image Registration (FIRE) dataset to compare registration accuracy and computational efficiency using mean landmark error (MLE) as the primary outcome measure.
Methods:
The three image registration approaches were evaluated using the FIRE dataset under consistent conditions across varying image overlap conditions (Classes S, A, and P). These included: (a) RetinaRegNet, which incorporates diffusion features, dual keypoint sampling through Scale-Invariant Feature Transform (SIFT) and random, two-stage outlier removal, and a multilevel registration hierarchy progressing from homography to polynomial transforms; (b) EyeLiner, which integrates anatomical segmentation with SuperPoint feature extraction, LightGlue matching, and thin-plate spline warping; (c) GeoFormer, which builds on Local Feature Transformers (LoFTR) through cross-attention mechanisms and Random Sampling Consensus (RANSAC)-based refinement. Registration performance was quantified using MLE.
Results:
Across all 134 FIRE image pairs, RetinaRegNet achieved the lowest overall MLE (3.12 pixels), outperforming EyeLiner (3.81 pixels) and GeoFormer (6.06 pixels). Class-specific analysis showed that RetinaRegNet delivered the highest accuracy in Class S images (1.70 pixels), competitive performance in Class A (5.24 pixels), and the strongest results in the most challenging Class P cases (4.57 pixels). GeoFormer demonstrated the shortest processing time at 0.32 seconds per image pair, compared with 4.92 seconds for EyeLiner and 31.23 seconds for RetinaRegNet. In Class P, RetinaRegNet achieved a 59.2% improvement in accuracy relative to GeoFormer (4.57 vs 11.20 pixels). The code is available at: https://github.com/ThenukaDharmaseelan/image_Registration.
Conclusions:
Overall, the evaluation reveals a clear trade-off between registration precision and computational speed. RetinaRegNet achieves the lowest MLE for complex clinical cases despite higher computational cost. EyeLiner balances precision and speed for routine use, while GeoFormer prioritizes rapid throughput where processing speed is critical.
Aim:
To benchmark three deep learning-based retinal image registration methods RetinaRegNet, EyeLiner, and GeoFormer on the Fundus Image Registration (FIRE) dataset to compare registration accuracy and computational efficiency using mean landmark error (MLE) as the primary outcome measure.
Methods:
The three image registration approaches were evaluated using the FIRE dataset under consistent conditions across varying image overlap conditions (Classes S, A, and P). These included: (a) RetinaRegNet, which incorporates diffusion features, dual keypoint sampling through Scale-Invariant Feature Transform (SIFT) and random, two-stage outlier removal, and a multilevel registration hierarchy progressing from homography to polynomial transforms; (b) EyeLiner, which integrates anatomical segmentation with SuperPoint feature extraction, LightGlue matching, and thin-plate spline warping; (c) GeoFormer, which builds on Local Feature Transformers (LoFTR) through cross-attention mechanisms and Random Sampling Consensus (RANSAC)-based refinement. Registration performance was quantified using MLE.
Results:
Across all 134 FIRE image pairs, RetinaRegNet achieved the lowest overall MLE (3.12 pixels), outperforming EyeLiner (3.81 pixels) and GeoFormer (6.06 pixels). Class-specific analysis showed that RetinaRegNet delivered the highest accuracy in Class S images (1.70 pixels), competitive performance in Class A (5.24 pixels), and the strongest results in the most challenging Class P cases (4.57 pixels). GeoFormer demonstrated the shortest processing time at 0.32 seconds per image pair, compared with 4.92 seconds for EyeLiner and 31.23 seconds for RetinaRegNet. In Class P, RetinaRegNet achieved a 59.2% improvement in accuracy relative to GeoFormer (4.57 vs 11.20 pixels). The code is available at: https://github.com/ThenukaDharmaseelan/image_Registration.
Conclusions:
Overall, the evaluation reveals a clear trade-off between registration precision and computational speed. RetinaRegNet achieves the lowest MLE for complex clinical cases despite higher computational cost. EyeLiner balances precision and speed for routine use, while GeoFormer prioritizes rapid throughput where processing speed is critical.
DOI: https://doi.org/10.37349/edht.2026.101194
This article belongs to the special issue Deep Learning Methods and Applications for Biomedical Imaging

Aim:
Recent studies suggest an association between sleep patterns and metabolic dysfunction-associated steatotic liver disease (MASLD) among American adults. Despite established sex-specific disparities in MASLD prevalence, the potential influence of sex on the sleep-MASLD relationship is not well defined. Our research aims to elucidate the sex-specific associations of sleep with MASLD by utilizing a nationally representative cohort from the United States.
Methods:
Data from United States adults aged 20 and older in the 2017–2020 NHANES were analyzed in this cross-sectional study. Sleep parameters were assessed via interviewer-administered questionnaires, while MASLD was defined using vibration-controlled transient elastography (VCTE). Sex-specific associations were investigated with sex-stratified multivariable logistic regression models, and their robustness was tested through subgroup and sensitivity analyses.
Results:
This study included 5,243 participants (51.2% female). A significant association was observed between sleep disorders and a greater likelihood of MASLD among male participants (OR = 1.50; 95% CI: 1.08–2.10). After full adjustment for covariates, the association remained significant in females (OR = 1.51; 95% CI: 1.06–2.16). Conversely, high sleep debt remained significantly associated with MASLD in both sexes (males: OR = 1.64; 95% CI: 1.14–2.37; females: OR = 1.51; 95% CI: 1.06–2.15). Subgroup analyses confirmed that sex did not modify these relationships.
Conclusions:
Sleep disorders and sleep debt were significantly associated with MASLD in both sexes. These findings suggest that sleep health may represent an important modifiable target in MASLD prevention strategies.
Aim:
Recent studies suggest an association between sleep patterns and metabolic dysfunction-associated steatotic liver disease (MASLD) among American adults. Despite established sex-specific disparities in MASLD prevalence, the potential influence of sex on the sleep-MASLD relationship is not well defined. Our research aims to elucidate the sex-specific associations of sleep with MASLD by utilizing a nationally representative cohort from the United States.
Methods:
Data from United States adults aged 20 and older in the 2017–2020 NHANES were analyzed in this cross-sectional study. Sleep parameters were assessed via interviewer-administered questionnaires, while MASLD was defined using vibration-controlled transient elastography (VCTE). Sex-specific associations were investigated with sex-stratified multivariable logistic regression models, and their robustness was tested through subgroup and sensitivity analyses.
Results:
This study included 5,243 participants (51.2% female). A significant association was observed between sleep disorders and a greater likelihood of MASLD among male participants (OR = 1.50; 95% CI: 1.08–2.10). After full adjustment for covariates, the association remained significant in females (OR = 1.51; 95% CI: 1.06–2.16). Conversely, high sleep debt remained significantly associated with MASLD in both sexes (males: OR = 1.64; 95% CI: 1.14–2.37; females: OR = 1.51; 95% CI: 1.06–2.15). Subgroup analyses confirmed that sex did not modify these relationships.
Conclusions:
Sleep disorders and sleep debt were significantly associated with MASLD in both sexes. These findings suggest that sleep health may represent an important modifiable target in MASLD prevention strategies.
DOI: https://doi.org/10.37349/edd.2026.1005123

Aim:
Sweet potato (Ipomoea batatas Lam.) is a nutritious root crop that remains underexploited. This study aimed to valorize sweet potato as a functional ingredient for biscuit formulation by reducing dependency on refined wheat flour with a focus on evaluating its nutritional value, bioactive phytochemicals, and mineral profile. By using purple-fleshed sweet potato flour (PFSPF) in biscuit recipes, food security and health concerns can be addressed by enhancing dietary intake.
Methods:
Biscuits developed with PFSPF in different percentages (10%, 20%, 30%, 40%, and 50%), whereas the control included only wheat flour, were evaluated for physicochemical, nutritional, mineral profile, and sensorial qualities. Sensorial analysis was conducted with 54 participants to evaluate the acceptability, appearance, color, flavor, taste, and texture of the developed biscuits.
Results:
Physicochemical analysis of the PFSPF revealed high crude fiber, protein, ash, and carbohydrate, along with bioactive phytochemicals, namely total phenolic content (TPC) (25.07 mg GAE/100 g), total carotenoid content (TCC) (27.90 mg/100 g), and antioxidant activity (DPPH: 43.51%). The PFSPF incorporated biscuits showed that dietary fiber and anthocyanin content increased significantly (p < 0.05), with values ranging from 0.46% to 6.38% and 0.28 to 2.64 mg/100 g, respectively. The TPC and TCC of the developed biscuits ranged from 1.44 to 6.91 mg GAE/100 g and 4.42 to 6.32 mg/100 g, respectively. The mineral profile also proportionally increased in the formulated biscuits. Biscuits fortifying 50% PFSPF (T6) showed more hardness (34.25 N) and the highest energy value (9.08 kcal/100 g). Sensory results indicated that the ideal acceptability level of the formulated biscuits achieved the maximum score for 50% PFSPF (T6) (7.60).
Conclusions:
The utilization of PFSPF in biscuit formulation appears to be a promising approach for baking applications, offering more nutritional benefits, including mineral and bioactive phytochemicals, along with a distinctive color, appealing texture, and improved consumer acceptability.
Aim:
Sweet potato (Ipomoea batatas Lam.) is a nutritious root crop that remains underexploited. This study aimed to valorize sweet potato as a functional ingredient for biscuit formulation by reducing dependency on refined wheat flour with a focus on evaluating its nutritional value, bioactive phytochemicals, and mineral profile. By using purple-fleshed sweet potato flour (PFSPF) in biscuit recipes, food security and health concerns can be addressed by enhancing dietary intake.
Methods:
Biscuits developed with PFSPF in different percentages (10%, 20%, 30%, 40%, and 50%), whereas the control included only wheat flour, were evaluated for physicochemical, nutritional, mineral profile, and sensorial qualities. Sensorial analysis was conducted with 54 participants to evaluate the acceptability, appearance, color, flavor, taste, and texture of the developed biscuits.
Results:
Physicochemical analysis of the PFSPF revealed high crude fiber, protein, ash, and carbohydrate, along with bioactive phytochemicals, namely total phenolic content (TPC) (25.07 mg GAE/100 g), total carotenoid content (TCC) (27.90 mg/100 g), and antioxidant activity (DPPH: 43.51%). The PFSPF incorporated biscuits showed that dietary fiber and anthocyanin content increased significantly (p < 0.05), with values ranging from 0.46% to 6.38% and 0.28 to 2.64 mg/100 g, respectively. The TPC and TCC of the developed biscuits ranged from 1.44 to 6.91 mg GAE/100 g and 4.42 to 6.32 mg/100 g, respectively. The mineral profile also proportionally increased in the formulated biscuits. Biscuits fortifying 50% PFSPF (T6) showed more hardness (34.25 N) and the highest energy value (9.08 kcal/100 g). Sensory results indicated that the ideal acceptability level of the formulated biscuits achieved the maximum score for 50% PFSPF (T6) (7.60).
Conclusions:
The utilization of PFSPF in biscuit formulation appears to be a promising approach for baking applications, offering more nutritional benefits, including mineral and bioactive phytochemicals, along with a distinctive color, appealing texture, and improved consumer acceptability.
DOI: https://doi.org/10.37349/eff.2026.1010152
 Previous
Previous