Affiliation:
1Foxabell-Laboratorium Investigativum, Laboratorium Scientiae et Studiorum Investigativorum, Nashik 422101, Maharashtra, India
Email: saurabh_bhandare@yahoo.com
ORCID: https://orcid.org/0000-0003-3138-2809
Affiliation:
2Pharmaceutical technology, KKWCOP Hirabai Haridas Vidyanagari, Amrutdham Sanitorium, Panchavati, Nashik 422003, Maharashtra, India
ORCID: https://orcid.org/0000-0003-4268-8475
Explor Drug Sci. 2026;4:1008151 DOI: https://doi.org/10.37349/eds.2026.1008151
Received: August 23, 2025 Accepted: November 12, 2025 Published: February 25, 2026
Academic Editor: Michio Kurosu, University of Tennessee Health Science Center, USA
The significant medicinal constituents and pharmacological potential of several botanicals suggest promising therapeutic applications. Scorzonera undulata displayed a diverse phytochemical profile, with 25 volatile and 21 phenolic compounds identified, including quinic and chlorogenic acids, along with flavonoids such as kaempferol, apigenin, luteolin derivatives, quercitrin, and naringin—mostly concentrated in the aerial parts. These extracts exhibited notable antioxidant, antimicrobial, anti-inflammatory, and cytotoxic activities, especially methanolic extracts against MCF-7 breast cancer cells, indicating therapeutic relevance. Andrographis paniculata extracts, rich in andrographolide, showed clinical potential in alleviating mild COVID-19 symptoms. However, the compound’s nonlinear pharmacokinetics highlight the need for optimized delivery strategies. Morinda citrifolia fruit extracts demonstrated considerable in vitro antimicrobial effects and moderate cytotoxicity, supported by UPLC–Orbitrap MS identification of unique bioactives. These findings reinforce the need for further pharmacological and clinical validation. The antiviral efficacy of Houttuynia cordata against dengue virus type 2 was evident, with aqueous extracts showing strong virucidal action and inhibition of viral replication. Hyperoside was identified as the dominant active constituent, supported by a rich phytochemical profile including flavonoids, aristolactams, and triterpenoids. Genotoxicity assessments indicated a favorable safety profile, suggesting potential for phytotherapeutic development. Achillea millefolium (yarrow) contained essential oils enriched in camphor, 1,8-cineole, artemisia ketone, and azulene derivatives, alongside phenolic acids and flavonoids like chlorogenic acid, apigenin, luteolin, and quercetin. These contributed to its antioxidant, anti-inflammatory, antimicrobial, and hemostatic effects, validating traditional medicinal applications and warranting clinical standardization. Flavonoids such as luteolin and apigenin offered anticancer and cardiovascular benefits by inhibiting PD-L1 via STAT3 suppression and promoting autophagy to counter vascular calcification. Bryophyllum pinnatum demonstrated broad pharmacological activity attributed to bufadienolides, flavonoids, and phenolic acids, supporting its ethnomedicinal use while emphasizing the need for clinical safety validation.
Since ancient times, humans have relied on nature to meet essential needs, particularly for medicinal purposes to treat a wide variety of illnesses. Historical records from Mesopotamia, dating back to around 2600 BCE, document nearly 1,000 plant-based remedies, highlighting the central role of plants in traditional healing practices [1]. Among these remedies were oils derived from Cedrus (cedar) and Cupressus sempervirens (cypress), preparations from Glycyrrhiza glabra (licorice), various species of Commiphora (myrrh), and Papaver somniferum (the opium poppy). These substances were esteemed in antiquity for their therapeutic virtues, and many continue to be used today in the management of respiratory disorders, parasitic infections, and inflammation [1].
Natural products have long served as a crucial foundation for identifying novel lead compounds in pharmaceutical research. A significant proportion of modern therapeutic drugs can be traced back to natural sources, with plants playing a particularly vital role [2]. However, in recent decades, interest in natural product-based drug discovery declined, mainly due to the complexity of isolating and characterizing bioactive compounds. Recent advances in analytical and extraction technologies have helped overcome these challenges, resulting in renewed scientific enthusiasm for exploring nature-derived compounds in drug development [2].
Throughout history, natural substances have been central to traditional and folk medicine, applied to a wide range of health conditions. The application of classical techniques in natural product chemistry led to the discovery of numerous biologically active secondary metabolites from both terrestrial and marine organisms, many of which progressed to become promising drug candidates in modern pharmaceutical development [3]. Since a large number of contemporary pharmaceuticals originate from natural sources, traditional medicine and ethnopharmacological knowledge provide valuable opportunities for uncovering potential leads in drug discovery [4, 5].
In contemporary medicine, natural products still hold a crucial position in therapeutic development. Despite the rise of combinatorial chemistry and high-throughput screening techniques, a considerable number of approved medications are either directly derived from natural compounds or structurally inspired by them [4]. Plant-derived compounds contribute significantly to this field: through photosynthesis, plants generate essential primary metabolites for growth and survival, while also producing diverse secondary metabolites that serve biological roles such as defense, signaling, and adaptation to environmental stress [6]. These secondary metabolites often possess pharmacological activities, making them indispensable resources for drug discovery and modern pharmaceutical formulations.
Medicinal plant extracts contain bioactive compounds with therapeutic potential, making them valuable in both the prevention and treatment of disease. They are utilized in various dosage forms, ranging from oral preparations such as tablets, capsules, and droplet pills to parenteral formulations including injectables, drawing attention to their versatility in both traditional and clinical healthcare practices [7, 8]. Indeed, plant-derived compounds represent a major reservoir of active pharmaceutical ingredients, contributing to approximately 40% of approved therapeutic drugs and nearly 25% of FDA-authorized products [9].
Phytotherapy, defined as the use of medicinal plants for the treatment and prevention of illnesses, has its roots in ancient cultures [10]. Today, complementary and alternative medicine increasingly emphasizes the development of safe, effective, and accessible plant-based therapies. Herbal medicines are especially valued in underdeveloped regions for their affordability and availability [10]. Although some plant-derived compounds still lack complete clinical validation and detailed mechanistic understanding, natural products from botanicals remain central to global medicine—providing treatment options for nearly 80% of the world’s population [2]. Remarkably, over half of modern pharmaceuticals are of plant origin, and about 65% of the global population relies on phytotherapy for healthcare needs [10]. Numerous scientific studies continue to support the efficacy and health benefits of herbal medicines [10].
In this context, recent investigations into plants such as Scorzonera undulata have clarifiedtheir rich phytochemical profiles. Gas chromatography-mass spectrometry (GC-MS) analysis of various extracts from S. undulata revealed 25 volatile compounds across different chemical classes, including phenols (e.g., 2,4-Di-tert-butylphenol), fatty alcohols (e.g., 1-hexadecanol), organic acids (lactic, malic, and carbonic acids), steroids, alkanes, and their derivatives. Notably, 2,4-Di-tert-butylphenol and palmitic acid were consistently present across plant parts and extraction techniques. Among the tested preparations, ethanolic extracts from the tuberous roots yielded the highest number of volatile constituents, with 2,4-Di-tert-butylphenol uniformly detected in all samples. These findings highlight the chemical diversity and therapeutic potential of S. undulata, supporting its relevance as a candidate for pharmacological exploration [10–12].
This manuscript was prepared as a narrative review. Relevant scientific literature was identified through searches in PubMed, Scopus, Web of Science, and Google Scholar up to September 2025. Keywords included the scientific names of the selected medicinal plants [e.g., S. undulata, Andrographis paniculata, Houttuynia cordata (HC), and Achillea millefolium] combined with terms such as “phytochemistry,” “bioactive compounds,” “pharmacological activity,” “toxicology,” and “clinical trial”. Additional references were retrieved by screening the bibliographies of relevant articles.
No formal restrictions on publication year were applied, but preference was given to studies published in the past two decades and to peer-reviewed sources. Both in vitro, in vivo, and clinical studies were considered. Evidence strength was qualitatively judged according to study type: in vitro (preliminary mechanistic evidence), animal models (preclinical evidence), and human studies (clinical evidence). Safety information was also extracted where available (Figure 1).
Andrographis paniculata (Burm. f.) Nees, an herbaceous plant of the Acanthaceae family, traditionally known in Thailand as Fah-Talai-Jone, has gained significant attention for its antiviral potential against SARS-CoV-2. Recent clinical trials indicate its effectiveness in alleviating mild to moderate COVID-19 symptoms [13]. Phytochemical analysis of Andrographis paniculata powder identified thirteen bioactive constituents, comprising seven diterpenoids, five flavonoids, and one phenolic compound [13]. The co-administration of andrographolide with solubilizing and bioenhancing agents markedly improved its pharmacokinetic profile: β-cyclodextrin (50% w/w), sodium dodecyl sulphate (1% w/w), and piperine (10% w/w), either individually or in combination, significantly increased systemic absorption. This enhancement resulted in oral bioavailability ranging from 131.01% to 196.05% after both single and multiple dosing regimens [13]. The detection of glucuronide metabolites in urine and feces indicates glucuronidation as a primary metabolic pathway for andrographolide. These findings highlight the potential of formulation strategies to improve the absorption and therapeutic efficacy of Andrographis paniculata, encouraging further clinical research into its applications [13].
The aerial parts of S. suberosa, S. latifolia, and S. laciniata have also been investigated for their phytochemical content, mineral composition, and antioxidant potential [11]. High-performance liquid chromatography (HPLC) revealed the presence of diverse bioactive molecules, including vitamins (D, K, α-tocopherol, and retinol), flavonoids (rutin, myricetin, morin, quercetin, and kaempferol), resveratrol, and sugars (arabinose, fructose, glucose, sucrose, and maltose) [11]. Mineral analysis by atomic absorption spectrometry demonstrated essential elements such as: calcium, sodium, potassium, iron, manganese, zinc, and magnesium, whereas copper, cobalt, and nickel were not detected [11]. Antioxidant assays using DPPH radical scavenging revealed a dose-dependent activity across all species, confirming their rich phytochemical composition and strong antioxidant potential [11].
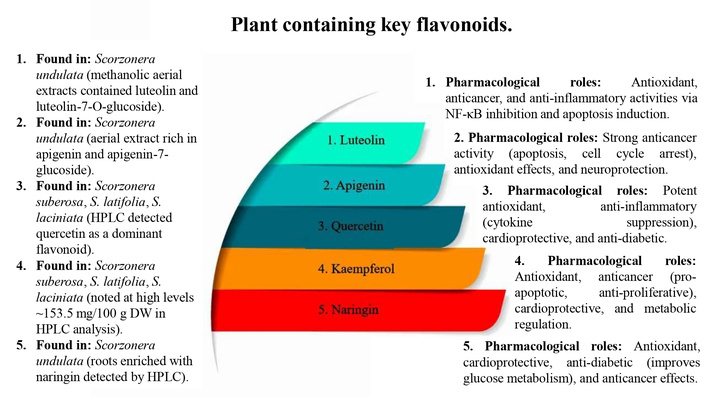
The methanolic extracts of S. undulata were evaluated for anticancer activity against Michigan Cancer Foundation-7 (MCF7), breast cancer cells [10]. HPLC analysis of the root extract of Scorzonera undulata (RSU) and the aerial part extract of Scorzonera undulata (ASU) demonstrated distinct phytochemical profiles, including flavonoids, polyphenols, and tannins. ASU exhibited stronger cytotoxicity (IC50 = 4.22 ± 0.06 mg/mL) compared to RSU (IC50 = 5.89 ± 0.08 mg/mL) after 24 hours, reducing cancer cell viability through apoptosis and cellular disintegration [10]. The ASU was particularly rich in apigenin (1.489 mg/mL), gallic acid (0.562 mg/mL), quercetin (0.062 mg/mL), luteolin-7-glucoside (0.108 mg/mL), and ferulic acid (0.086 mg/mL). In contrast, the root extract contained higher amounts of luteolin (0.427 mg/mL), chlorogenic acid (0.350 mg/mL), and naringin (0.098 mg/mL). Apigenin-7-glucoside and gallic acid were exclusive to the aerial parts, whereas caffeic acid, vanillic acid, and verbascoside were detected in both roots and aerial parts, albeit at varying concentrations [10]. Overall, ASU demonstrated a broader and more potent flavonoid profile, correlating with its superior antioxidant and anticancer activities [10].
Further profiling of S. undulata through GC-MS identified 25 volatile compounds across multiple chemical classes [12]. These included phenolic compounds (2,4-di-tert-butylphenol; phenol, 2,2’-methylenebis[6-(1,1-dimethylethyl)-4-methyl-]), fatty alcohols (1-hexadecanol and its methylated derivatives), fatty acid esters (9-hexadecenoic acid, 9-octadecenyl ester), saturated fatty acids (hexanoic, decanoic, and palmitic acids), organic acids (carbonic, lactic, butanedioic, and malic acids), steroids (e.g., 2(4H)-benzofuranone derivatives), and alkanes such as heneicosane, octadecane, and heptadecane [12]. Compounds including 2,4-di-tert-butylphenol, palmitic acid, and lactic acid were consistently observed across extraction methods, solvents, and plant parts. Among them, ethanolic extracts of the tuberous roots exhibited the greatest diversity and abundance of volatile constituents, with 2,4-di-tert-butylphenol present in all tested specimens [12].
Taken together, these findings demonstrate that S. undulata possesses a wide spectrum of bioactive compounds capable of suppressing cancer cell growth through apoptosis and lysis, alongside strong antioxidant activity [10, 12].
Liquid chromatography-mass spectrometry (LC-MS) profiling of S. undulata revealed a total of 21 phenolic compounds comprising 11 phenolic acids and 10 flavonoids, with ultrasound-assisted extraction (UAE) outperforming maceration by enhancing the release of bioactive constituents, especially in leaves and flowers. Among the identified phenolic acids, caffeic acid exhibited antioxidant and anti-inflammatory properties, chlorogenic acid (196.34 mg/100 g DW in flowers) showed potent antioxidant, anti-diabetic, and cardioprotective activity, ferulic acid contributed to anti-diabetic and cardioprotective effects, gallic acid was associated with antioxidant and anti-inflammatory actions, quinic acid (165.55 mg/100 g DW in leaves) demonstrated antioxidant, anti-inflammatory, anti-diabetic, and anticancer potential, and vanillic acid contributed to antioxidant activity. The flavonoid fraction was particularly enriched in aerial parts, with apigenin exerting anticancer and cytoprotective effects, kaempferol (153.50 mg/100 g DW) displaying anticancer and cytoprotective activities, luteolin-7-O-glucoside (245.11 mg/100 g DW) offering strong anticancer and antioxidant effects, naringin contributing to anti-diabetic, cardioprotective, and anticancer roles, and quercetin derivatives enhancing antioxidant and cytoprotective capacity. Tissue distribution analysis confirmed that leaves and flowers contained the highest levels of both phenolic acids and flavonoids (up to 322.39 mg/100 g DW by UAE), whereas tubers exhibited minimal amounts (phenolic acids ~35.87 mg/100 g DW; flavonoids < 1 mg/100 g DW). Collectively, these findings establish S. undulata as a rich source of pharmacologically relevant phytochemicals with strong antioxidant, anti-inflammatory, anti-diabetic, cardioprotective, and anticancer potential, while also highlighting UAE as a superior extraction technique for maximizing bioactive compound recovery [12] Table 1.
Pharmacological and medicinal activities of identified phytochemicals in S. undulata (phenolic acids and flavonoids in Scorzonera species) [12].
| Compound name | Pharmacological/medicinal activities |
|---|---|
| Quinic acid | Antioxidant, anti-inflammatory, anti-diabetic, anticancer, cardioprotective. |
| 1,3-Dicaffeoylquinic acid | Antioxidant, hepatoprotective, anti-inflammatory, antiviral. |
| Gallic acid | Antioxidant, anti-inflammatory, anticancer, antimicrobial. |
| Protocatechuic acid | Antioxidant, anti-inflammatory, anti-diabetic, neuroprotective, anticancer. |
| Chlorogenic acid | Potent antioxidant, anti-diabetic, cardioprotective, anti-obesity. |
| Caffeic acid | Antioxidant, anti-inflammatory, anticancer, anti-diabetic, hepatoprotective. |
| Syringic acid | Antioxidant, anti-inflammatory, anti-hyperglycemic, cardioprotective. |
| p-Coumaric acid | Antioxidant, anti-inflammatory, antimicrobial, anti-diabetic. |
| Ferulic acid | Antioxidant, anti-inflammatory, cardioprotective, anti-diabetic, anticancer. |
| Rosmarinic acid | Strong antioxidant, anti-inflammatory, antiviral, anticancer, neuroprotective. |
| Cinnamic acid | Antioxidant, antimicrobial, anti-inflammatory, anti-diabetic. |
| Rutin | Antioxidant, anti-inflammatory, vasoprotective, anticancer, cardioprotective. |
| Quercetin | Antioxidant, anti-inflammatory, anticancer, cardioprotective, antiviral. |
| Hyperoside | Antioxidant, anti-inflammatory, anticancer, neuroprotective. |
| Luteolin-7-O-glucoside | Strong antioxidant, anticancer, anti-inflammatory, cytoprotective. |
| Naringin | Antioxidant, anti-diabetic, cardioprotective, anticancer, anti-inflammatory. |
| Quercitrin | Antioxidant, anti-inflammatory, hepatoprotective, anticancer. |
| Apigenin | Anticancer, anti-inflammatory, antioxidant, cytoprotective, neuroprotective. |
| Kaempferol | Antioxidant, anticancer, anti-inflammatory, cardioprotective, neuroprotective. |
| Naringenin | Antioxidant, anti-inflammatory, anti-diabetic, cardioprotective, anticancer. |
| Apigenin-7-O-glucoside | Antioxidant, anticancer, anti-inflammatory, hepatoprotective. |
Thus, common flavonoid scaffolds provide a shared therapeutic foundation across diverse medicinal plants. This suggests that the pharmacological relevance of these plants may lie not only in unique compounds but also in the synergistic reinforcement of widely distributed flavonoids with overlapping molecular targets.
Hence, the comparative synthesis showed that recurrent flavonoids (luteolin, apigenin, quercetin, kaempferol, and naringin) exhibit overlapping antioxidant, anti-inflammatory, anticancer, cardioprotective, and neuroprotective effects across multiple plants. This highlights a common set of mechanisms—ROS scavenging, cytokine modulation, apoptosis induction, and vascular protection—that collectively reinforce the therapeutic relevance of these plants. Integrating these shared pathways provides a clearer mechanistic and deterministic architecture.
Hence, the phytochemical profiling across different Scorzonera species and related medicinal plants confirms that recurrent flavonoids—luteolin, apigenin, quercetin, kaempferol, and naringin—are present in multiple extracts. These flavonoids consistently demonstrate overlapping antioxidant, anti-inflammatory, anticancer, cardioprotective, and neuroprotective activities. For instance, S. undulata provides luteolin, apigenin, and naringin, while S. suberosa, S. latifolia, and S. laciniata contribute: rutin, myricetin, morin, quercetin, and kaempferol. Together, these findings justify that the therapeutic potential of these plants is largely driven by a shared flavonoid framework.
Distribution and bioactivities of major flavonoids—luteolin, apigenin, quercetin, kaempferol, and naringin—identified in various Scorzonera species. Methanolic and HPLC analyses revealed their presence in aerial parts and roots, with notable concentrations. These compounds exhibit a wide range of pharmacological effects, including antioxidant, anticancer, anti-inflammatory, cardioprotective, anti-diabetic, and neuroprotective activities through mechanisms such as NF-κB inhibition, apoptosis induction, cytokine suppression, and metabolic regulation (Figure 2).
Hydroalcoholic extracts of dried Morinda citrifolia L. (noni) fruit pulp were prepared using ethanol (NE) and methanol (NM) solvents with an intelligent-flash extractor (KBE-I5), followed by freeze-vacuum drying [14]. The process yielded 16.8% for NE and 25.8% for NM. Antimicrobial activity was assessed by disc diffusion assay against bacterial and fungal strains, while cytotoxic effects on human colon adenocarcinoma (HT-29) and bladder cancer (UMUC-3) cell lines were evaluated using CCK-8 viability assays, acridine orange staining, and western blot analysis, and the outcomes are discussed below in section [14].
i. Antimicrobial efficacy: NE extract displayed minimum inhibitory concentrations (MICs) of 177 mg/mL for Escherichia coli, 52 mg/mL for Saccharomyces cerevisiae, 388 mg/mL for Staphylococcus aureus, and 283 mg/mL for Streptococcus thermophilus. Comparatively, NM extract exhibited stronger effects with MICs of 105 mg/mL for E. coli, 47 mg/mL for S. cerevisiae, 312 mg/mL for S. aureus, and 135 mg/mL for S. thermophilus [14].
ii. Anticancer efficacy: NE extract inhibited HT-29 and UMUC-3 cells with IC50 values of 0.758 mg/mL and 0.899 mg/mL, respectively, whereas NM extract showed IC50 values of 1.231 mg/mL and 1.173 mg/mL for the same lines. Morphological alterations in cellular organelles, elevated autophagy marker expression, and increased acridine orange fluorescence confirmed apoptosis and autophagic cell death as the principal mechanisms of cytotoxicity [14].
iii. Phytochemical profiling: Advanced UPLC-Q-Exactive Orbitrap-MS/MS analysis in dual ion mode identified 17 unique bioactive compounds, including 14 reported for the first time in M. citrifolia fruit [14].
Phenolic acids: 2-Hydroxycinnamic acid and 4-Hydroxycinnamic acid (antioxidant, anticancer, and anti-inflammatory activities).
Vitamins: Riboflavin (oncological relevance, metabolic regulation, antiviral activity).
Oligosaccharides and glycosides: Gentiotriose, umbelliferose, sakebiose (prebiotic, metabolic, and immunomodulatory roles).
Specialized metabolites: Wyomin and armillaripin (antimicrobial and cytotoxic potential).
Thus, both ethanol and methanol extracts of M. citrifolia demonstrated significant antimicrobial and anticancer activities, with NM showing greater antimicrobial potency and NE exhibiting stronger anticancer efficacy. The broad spectrum of phytochemicals—ranging from phenolic acids and vitamins to oligosaccharides and rare metabolites—provides a mechanistic basis for these bioactivities. Collectively, these findings validate M. citrifolia as a promising candidate for therapeutic and nutraceutical applications with antimicrobial, antioxidant, anticancer, and immunomodulatory potential [14].
The antiviral activity of aqueous decoctions prepared from HC was investigated against dengue virus serotype 2 (strain 16681). The extract displayed efficacy across multiple stages of infection in HepG2 liver cells. When administered either prior to or following viral exposure, it markedly reduced intracellular dengue viral RNA levels, which corresponded with decreased viral protein production, demonstrating inhibition of viral replication [15].
In addition to these preventive and therapeutic effects, the extract exhibited strong virucidal properties in a direct-contact assay. Pre-incubation of the virus with the extract effectively neutralized viral particles, leading to inhibition of RNA replication at a remarkably low effective concentration (EC50 = 0.8 µg/mL). On top of that, at concentrations ranging from 10–40 µg/mL, it significantly reduced the release of new virions from infected LLC-MK2 monkey kidney cells, thereby restricting viral spread [15].
Phytochemical analysis using HPLC identified hyperoside as the predominant bioactive constituent in the extract. This flavonoid is likely responsible for the observed antiviral activity, particularly its ability to disrupt viral replication and enhance direct virucidal effects. Importantly, genotoxicity assays confirmed that the extract was non-damaging to human blood cells, supporting its safety profile at therapeutically effective doses [15].
From a practical standpoint, HC—a medicinal plant long incorporated into traditional remedies and diets across East and Southeast Asia—emerges as a promising natural candidate for dengue therapy. Its aqueous extract exerts a threefold mode of action: preventing viral entry, treating established infections, and directly neutralizing viral particles. In the absence of licensed antivirals or universally effective vaccines against dengue, these findings highlight the pharmaceutical potential of hyperoside-enriched extracts for further development as anti-dengue agents [15].
A comprehensive phytochemical investigation of the whole herb of HC revealed a wide spectrum of natural compounds, including both newly identified and previously reported constituents. Two novel molecules, houttuynoside A and houttuynamide A, were isolated alongside 38 known compounds [16].
From a pharmacological perspective, several constituents displayed bioactivity in antiviral, antioxidant, and enzyme inhibitory assays. Among the antiviral compounds, the 4,5-dioxoaporphine norcepharadione B exhibited significant suppression of herpes simplex virus type 1 (HSV-1) replication [16].
In terms of antioxidant potential, the flavonoids quercitrin and quercetin-3-O-β-D-galactopyranoside demonstrated pronounced free radical-scavenging activities, with IC50 values of 31 µM and 63 µM, respectively [16].
Regarding enzyme inhibition, the oxoaporphine alkaloid cepharadione B showed potent anti-tyrosinase activity, yielding an IC50 of 170 µM, highlighting its role in melanogenesis regulation [16].
Beyond these pharmacologically validated molecules, several other structural classes of phytochemicals were also identified. These include aristolactam derivatives (aristolactam A II, aristolactam B II, piperolactam A, 3,4-dimethoxy-N-methyl aristolactam), oxoaporphines (splendidine, lysicamine), 4,5-dioxoaporphines (cepharadione B, norcepharadione B, noraristolodione), amide derivatives [N-(1-hydroxymethyl-2-phenylethyl)benzamide, N-(4-hydroxyphenylethyl)benzamide, benzamide, 4-hydroxybenzamide, 4-hydroxy-3-methoxybenzamide, 6,7-dimethyl-1-ribitol-1-yl-1,4-dihydroquinoxaline-2,3-dione, and a (1H)-quinolinone], as well as the indolic compound indole-3-carboxylic acid. The study further reported ionone derivatives (vomifoliol, dehydrovomifoliol, reseoside, 7-(3,5,6-trihydroxy-2,6,6-trimethylcyclohexyl)-but-3-en-2-one, 6-(9-hydroxy-but-7-ethyl)-1,1,5-trimethylcyclohexane-3,5,6-triol), additional flavonoids (afzelin), and numerous benzoenoid compounds (cis- and trans-isomers of methyl ferulate, benzyl-β-D-glucopyranoside, vanillic acid, methyl vanillate, vanillin, protocatechuic acid, 4-hydroxybenzoic acid, methylparaben, and p-hydroxybenzaldehyde). Structural lipophilic components included steroids (β-sitosterol, β-sitosteryl glucoside) and a triterpenoid, cycloart-25-ene-3β,24-diol [16]. Taken together, the phytochemical richness of HC reflects a diverse reservoir of bioactive metabolites, among which norcepharadione B, quercitrin, quercetin-3-O-β-D-galactopyranoside, and cepharadione B stand out as key contributors to antiviral, antioxidant, and enzyme inhibitory activities, respectively [16].
HC, a traditional medicinal herb, has shown strong antiviral potential against Coxsackievirus A4 (CVA4), a major cause of hand, foot, and mouth disease (HFMD) in children under five, particularly in China. Laboratory studies demonstrated that HC significantly reduces viral replication and cytopathic effects in infected cells, with a favorable safety profile at concentrations exceeding 1 mg/mL and an IC50 of 88.96 μg/mL. In animal models, HC treatment improved survival rates, delayed disease onset, reduced tissue damage, and effectively suppressed the excessive inflammatory responses triggered by CVA4 infection. Mechanistic analysis revealed that HC primarily acts after viral entry by inhibiting key host signaling pathways—specifically, the p38 mitogen-activated protein kinase (MAPK) and JNK pathways—which are known to be involved in both viral replication and inflammation. Chemical profiling identified bioactive compounds such as quercetin, hyperoside, and chlorogenic acid, which likely contribute to these effects. Moreover, HC outperformed the antiviral drug ribavirin in vivo, suggesting it may be a more effective option. These findings highlight HC’s promise as a low-toxicity, plant-based therapeutic candidate for the treatment of CVA4-induced HFMD (Table 2) [17].
| Section | Morinda citrifolia (Noni) | Houttuynia cordata |
|---|---|---|
| Extracts | Ethanol (NE), methanol (NM). | Aqueous decoction. |
| Main bioactivities |
| 1. Antiviral.2. Anti-inflammatory.3. Antioxidant. |
| Mechanisms | Apoptosis & autophagy induction, cell cycle arrest, bacterial/fungal inhibition. | Viral entry inhibition, RNA replication suppression, and MAPK/JNK pathway modulation. |
| Target pathogens/cells | E. coli, S. aureus, S. cerevisiae, and cancer cell lines HT-29, UMUC-3. | Dengue virus, HSV-1, CVA4. |
| Phytochemicals identified | 17 bioactives, including new compounds: 1. Phenolic acids.2. Riboflavin.3. Wyomin, armillaripin.4. Glycosides & oligosaccharides. | 38+ compounds:1. Hyperoside, quercitrin, and quercetin.2. Norcepharadione B (HSV-1 inhibitor).3. Cepharadione B (anti-tyrosinase).4. Houttuynoside A.5. Houttuynamide A.6. Chlorogenic acid.7. Cycloart-25-ene-3β,24-diol. |
| Highlights | NE: Stronger anticancer activity.NM: Better antimicrobial activity. | HC outperformed ribavirin in the HFMD model (CVA4).Effective at EC50 = 0.8 µg/mL for dengue. |
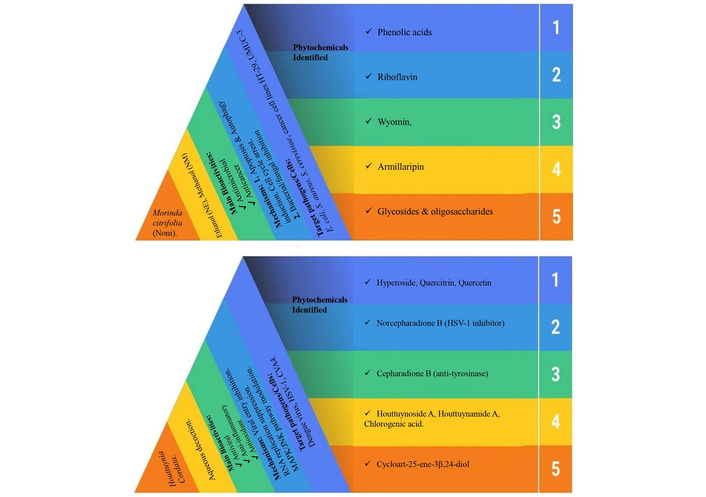
A comparative overview of the phytochemical composition, pharmacological activities, and mechanisms of action of M. citrifolia (noni) and HC extracts.
Noni extracts (ethanolic and methanolic) demonstrated significant antimicrobial and anticancer effects through induction of apoptosis and autophagy, supported by 17 identified bioactives, including phenolic acids, riboflavin, and novel metabolites such as wyomin and armillaripin.
HC, traditionally used in East Asia, exhibited potent antiviral and anti-inflammatory activity against dengue virus, HSV-1, and CVA4, acting via viral entry inhibition and modulation of host signaling pathways (MAPK, JNK). Identified compounds included hyperoside, quercitrin, norcepharadione B, and cepharadione B, contributing to its antiviral and enzyme inhibitory properties.
These findings accentuate the therapeutic potential of both plants as natural sources of multi-target bioactives Figure 3.
The HC extract was chemically profiled using HPLC, identifying key bioactive compounds such as quercetin, hyperoside, rutin, and chlorogenic acid. These components are known for their antiviral and anti-inflammatory properties, potentially contributing to the observed effects [17].
Thus, research offered the first comprehensive evidence that HC can effectively inhibit CVA4 infection both in cell culture and in a live animal model. Its ability to suppress viral replication and modulate host inflammatory responses—primarily through inhibition of the p38 MAPK and JNK pathways—highlights its promise as a novel therapeutic agent. Given its low toxicity and traditional usage, HC represents a strong candidate for further development as a treatment for HFMD caused by CVA4 [17].
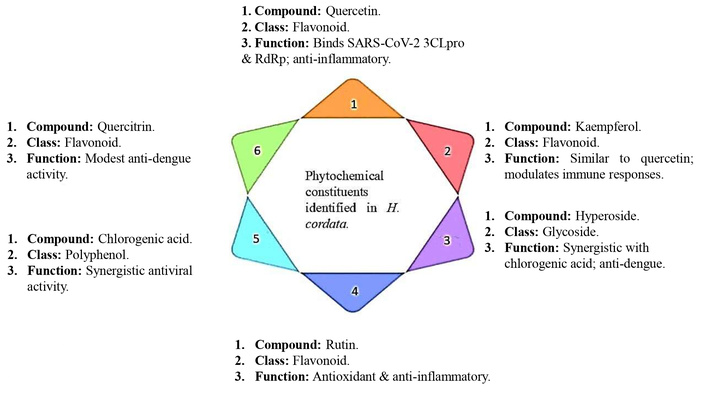
A network pharmacology and molecular simulation-based investigation highlighted the molecular mechanisms through which Houttuynia cordata Thunb. (HCT) may act against COVID-19 and its associated cytokine storm. Using the Traditional Chinese Medicine Systems Pharmacology Database (TCMSP), six bioactive compounds were identified, interacting with 45 COVID-19-related targets. A “component-target-pathway” interaction network was constructed in Cytoscape, and protein-protein interactions were mapped using the STRING database. Enrichment analysis revealed 18 major signaling pathways, with modulation of protein kinase C activity, suppression of NF-κB signaling, and inhibition of T cell responses being particularly relevant. The key target proteins were RELA (NF-κB p65), interleukin-6 (IL-6), and MAPK1. Molecular docking using AutoDockTools showed that quercetin and kaempferol, two prominent HCT constituents, bound with high affinity to SARS-CoV-2’s 3CL protease (3CLpro) and RNA-dependent RNA polymerase (RdRp), comparable to established antiviral drugs. Molecular dynamics simulations further validated these interactions, confirming that quercetin and kaempferol could regulate immune-inflammatory responses and suppress viral replication, thereby mitigating COVID-19 progression and cytokine storm [18].
Parallel investigations assessed the antiviral efficacy of HC against dengue virus serotype 2 (DENV-2, New Guinea C strain). Ethyl acetate (EA) extracts from two plant collections, along with five EA fractions and four polyphenolic compounds, were tested for inhibitory effects. The complete EA extracts showed potent activity during the early phase of infection, significantly reducing viral infectivity when pre-incubated with DENV-2 before adsorption. However, their activity was weaker when applied six hours post-infection, with no clear dose-dependent effect. The IC50 values of the EA extracts were remarkably low (0.24 ± 3.1 μg/mL and 0.04 ± 4.6 μg/mL), indicating strong antiviral efficacy. Of the five EA fractions, only one demonstrated measurable activity, but with a weaker IC50 of 333 μg/mL. Among the tested polyphenols, chlorogenic acid, hyperoside, quercetin, and quercitrin exhibited modest antiviral effects, though synergistic activity was observed when chlorogenic acid and hyperoside were combined. Nonetheless, their efficacy did not surpass that of the complete EA extract, suggesting that additional unidentified compounds may contribute synergistically to the extract’s overall potency. Importantly, the EA extracts were non-cytotoxic, bringing attention to their safety for potential therapeutic use [19].
From a pharmacological perspective, the phytochemicals of HC can be grouped according to their primary activities. Chlorogenic acid and hyperoside contribute synergistically to antiviral action against dengue [19]. Quercetin and kaempferol not only enhance antiviral efficacy but also target COVID-19-related proteins such as 3CLpro and RdRp while regulating inflammatory mediators, including IL-6 and NF-κB [18, 19]. Quercitrin, another flavonoid, supports additional antiviral potential against dengue [19]. Collectively, these compounds provide a mechanistic basis for the observed therapeutic effects, reinforcing the role of HC as a promising phytotherapeutic agent against both COVID-19 and dengue infections.
The major bioactive compounds were identified in HC through chemical profiling, particularly via HPLC. The six highlighted constituents—quercetin, kaempferol, hyperoside, rutin, chlorogenic acid, and quercitrin—belong to flavonoid, glycoside, and polyphenol classes. These phytochemicals exhibit diverse antiviral mechanisms, including inhibition of SARS-CoV-2 viral enzymes (3CLpro and RdRp), modulation of host immune responses, and synergistic action against dengue virus. Their known anti-inflammatory and antioxidant properties further contribute to HC’s therapeutic potential Figure 4.
The antiviral potential of A. japonica extracts has been extensively investigated for their efficacy against influenza and coronaviruses. Recent research highlighted their effectiveness against the avian influenza virus subtype H9N2, addressing a pressing issue of rising viral resistance to conventional antiviral agents and the demand for safer, natural alternatives [20].
In this study, extracts were prepared from the leaves, bark (stem), heartwood, and flowers of A. japonica through methanol extraction and dissolved in DMSO at a concentration of 20 mg/mL. Specific-pathogen-free embryonated chicken eggs infected with the low-pathogenic avian influenza strain KBNP-0028 (H9N2) were used for in vitro assays. Antiviral efficacy was evaluated via hemagglutination assays, while cytotoxicity was assessed using MTT assays on chorioallantoic membrane cells [20]. Results demonstrated a concentration-dependent inhibition of viral replication, with complete viral suppression observed at 400 µg/mL for leaf extracts, 300 µg/mL for bark extracts, and 200 µg/mL for flower extracts. While flower extracts exhibited reduced cell viability at higher concentrations, no cytotoxic effects were observed in stem or leaf-derived samples. These findings emphasized the strong antiviral activity of A. japonica, particularly from leaf and bark extracts, without compromising host cell viability [20].
Beyond influenza, diarylheptanoids isolated from A. japonica have been evaluated for inhibitory effects against the papain-like protease (PLpro) of Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV), a crucial enzyme in viral replication and a key antiviral drug target [21]. Through activity-guided fractionation of ethanol extracts, nine diarylheptanoids were purified and structurally characterized: hirsutenone, hirsutanonol, oregonin, platyphyllenone, platyphyllone, platyphyllonol-5-xylopyranoside, rubranol, rubranoside A, and rubranoside B [21]. Fluorometric assays confirmed that six of these compounds demonstrated clear dose-dependent suppression of PLpro activity, with reversible inhibition observed upon increasing enzyme concentrations. Among them, hirsutenone exhibited the strongest effect (IC50 = 4.1 µM), with structural analysis revealing that its catechol group and α, β-unsaturated carbonyl moiety were essential for potent inhibitory action [21].
Collectively, the phytochemicals of A. japonica contribute diverse antiviral properties. Diarylheptanoids (hirsutenone, hirsutanonol, oregonin, platyphyllenone, platyphyllone, platyphyllonol-5-xylopyranoside, rubranol, rubranoside A, and rubranoside B) exhibit inhibitory activity against SARS-CoV PLpro, with hirsutenone being the most potent inhibitor [21]. Methanolic extracts of leaves, bark, and flowers act as strong suppressors of avian influenza virus replication, showing complete inhibition at specific concentrations without notable cytotoxicity [20]. Forkhead Box M1 (FOXM1), a transcription factor involved in cell growth, survival, and cancer development, is highly expressed in glioblastoma (GBM) compared to normal tissue. The therapeutic potential of a plant-derived lignan, (−)-(2R,3R)-1,4-O-diferuloylsecoisolariciresinol (DFS), isolated from A. japonica, in targeting GBM tumorspheres (TSs), which are known to contribute to tumor growth and resistance [22].
DFS was found to induce degradation of FOXM1 protein, which in turn disrupted its interaction with β-catenin, a key regulator of oncogenic signaling. This inhibition prevented β-catenin from translocating into the nucleus and thereby blocked the activation of downstream genes involved in proliferation, stemness, and invasion [22].
In vitro, DFS significantly reduced cell viability, lowered ATP production, induced apoptosis, and suppressed both the formation and invasive behavior of GBM TSs. It also downregulated several transcription factors associated with tumor aggressiveness and stem cell-like characteristics [22].
In vivo, DFS treatment markedly slowed tumor growth and improved survival in mice bearing orthotopic GBM xenografts, providing strong evidence for its anti-tumor efficacy [22].
The findings demonstrate that DFS exerts potent anti-glioblastoma effects by targeting and destabilizing FOXM1, leading to inhibition of β-catenin nuclear signaling and suppression of tumor-promoting gene expression. These results highlight DFS as a promising natural compound with therapeutic potential for treating aggressive glioblastoma, particularly by targeting tumorsphere-driven growth and resistance mechanisms [22].
These findings not only validate the traditional medicinal use of A. japonica but also position its phytochemicals as promising leads for the development of natural antiviral therapeutics against both influenza and coronaviruses.
The key phytochemicals and their mechanisms of action contribute to the therapeutic effects of A. japonica. The antiviral activity is linked to methanolic extracts (from leaves, bark, and flowers) that inhibit H9N2 avian influenza virus replication and diarylheptanoids (e.g., hirsutenone) that suppress SARS-CoV PLpro enzyme activity. The anticancer activity, particularly against glioblastoma, is attributed to the lignan DFS [(−)-(2R,3R)-1,4-O-diferuloylsecoisolariciresinol], which degrades FOXM1 protein, blocks β-catenin nuclear translocation, and downregulates genes related to proliferation, stemness, and invasion. The diagram categorizes bioactivity by compound class and biological target, providing a consolidated overview of A. japonica’s pharmacological potential (Figure 5).
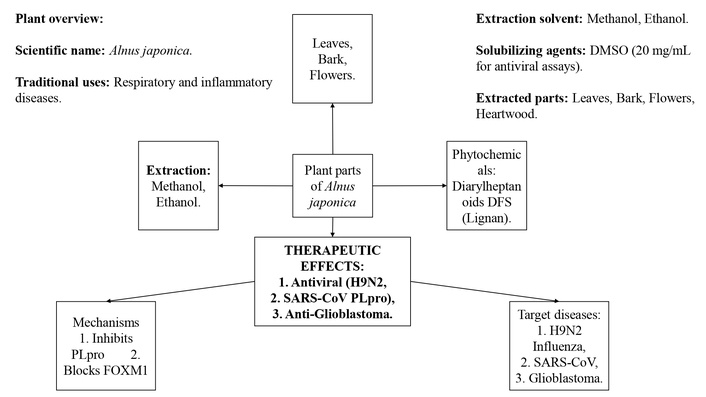
Mechanistic basis of the therapeutic potential of Alnus japonica: An analysis of its antiviral and anticancer activities [20–22]. DMSO: dimethyl sulfoxide; DFS: disease-free survival; PLpro: papain-like protease; FOXM1: (Forkhead Box M1). FOXM1 is a transcription factor involved in cell cycle regulation, cell proliferation, and cancer progression; H9N2: influenza A virus subtype H9N2; SARS-CoV: severe acute respiratory syndrome coronavirus.
Overview of the antiviral activity of A. japonica against avian influenza virus (H9N2) and SARS-CoV. The methanolic extracts from flowers, bark, and leaves showed dose-dependent inhibition of H9N2 replication in embryonated chicken eggs, with complete suppression observed at 200–400 µg/mL, though flower extracts exhibited mild cytotoxicity at higher doses. In parallel, nine diarylheptanoids isolated from A. japonica demonstrated inhibitory effects against the papain-like protease (PLpro) of SARS-CoV in fluorometric assays. Hirsutenone was the most potent compound (IC50 = 4.1 µM), with reversible enzyme inhibition dependent on its catechol and α, β-unsaturated carbonyl moieties. These findings underline the broad-spectrum antiviral potential of A. japonica via multiple phytochemical pathways (Figure 6).
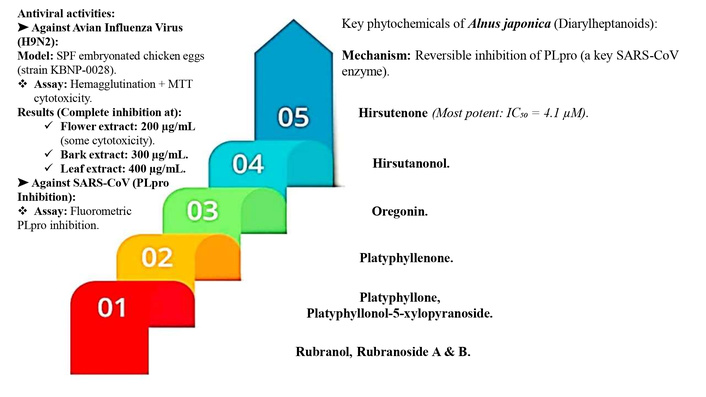
Antiviral effects of Alnus japonica extracts and diarylheptanoids against H9N2 influenza and SARS-CoV [20–22]. H9N9: influenza A virus subtype H9N9; SARS-CoV: severe acute respiratory syndrome coronavirus; SPF: specific pathogen-free embryonated chicken eggs; Strain KBNP-0028: KBNP-0028 viral, strain (strain designation; has no expanded full form); MTT cytotoxicity: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide cytotoxicity assay; PLpro: papain-like protease.
Achillea millefolium (common yarrow) is widely distributed in Poland, where it grows in meadows, roadsides, hillsides, field edges, and fallow lands, and is also cultivated for culinary and ornamental purposes. The plant is easily identified by its aromatic shoots, trichome-covered stems, and finely divided tripinnate or tetrapinnate leaves. Flowering generally occurs from June to October, occasionally persisting until the first frost [23]. Both folk and modern medicine in Europe and Asia have long valued yarrow for its therapeutic and cosmetic applications. Medicinally, its herb and flowers—collected during early bloom—are rich in essential oils (EOs), phenolic acids, flavonoids, sterols, and saponins, making them suitable for treating skin conditions, menstrual and renal disorders, memory issues, and digestive problems [23].
Historically, yarrow has been used since ancient Greece and the Trojan War, and it was revered among Native American tribes for its healing properties. In Polish culture, the flowers are included in Assumption Day bouquets as a symbol of health and prosperity [24]. Traditional uses include wound healing, fever reduction through sweat induction, and treatment of coughs, toothaches, headaches, nausea, digestive upset, burns, wounds, boils, and skin irritations. The plant has also been used as a mild analgesic, anti-inflammatory agent, and gentle laxative, and to ease anxiety. Notably, achilleine, one of its primary alkaloids, promotes rapid blood clotting, accounting for its effectiveness in stopping bleeding [24].
The EOs of Achillea millefolium contain a diverse chemical profile. Monoterpenes such as: α-pinene, β-pinene, borneol, bornyl acetate, camphor, 1,8-cineole (eucalyptol), limonene, sabinene, terpin-4-ol, terpineol, and α-thujone dominate, with oxygenated monoterpenes enriched in aerial parts compared to floral oils [23]. Sesquiterpenes and sesquiterpene lactones are also abundant, including: achillicin, achillin, caryophyllene, milefin, millefolide, chamazulene, azulene, and isoartemisia ketone, alongside sterols and saponins [25, 26]. Phenolic acids and flavonoids include apigenin derivatives (apigenin 4’-O-glucoside, apigenin 7-O-glucoside), chlorogenic acid, chrysoeriol, luteolin derivatives (luteolin 4’-O-glucoside, luteolin 7-O-glucoside), rutin, and several dicaffeoylquinic acid isomers (1,3-, 1,4-, 3,4-, and 3,5-dicaffeoylquinic acids) [27–29]. Other compounds include: megastigmanes (3α-hydroxy-5β,6β-epoxy-β-ionone; 3α-hydroxy-5α,6β-epoxy-β-ionone), (+)-dehydrovomifoliol, loliolide, (+)-isololiolide, (−)-10-hydroxydihydroactinidiolide, 8-hydroxycarvotanacetone, and phenylpropanoid 3-(4-hydroxyphenyl)-1-propanol [30]. Novel sesquiterpenoids such as millefoliumons A–F [31] and millefoliumines A–Q [32] have also been reported, the latter significantly reducing nitric oxide and cytokine (TNF-α, IL-6) production in macrophages.
EO displayed moderate activity against Streptococcus pneumoniae, Clostridium perfringens, and Candida albicans, but weaker inhibition of Mycobacterium smegmatis, Acinetobacter lwoffii, and Candida krusei (MICs 4.5–72.0 mg/mL). Ethanolic flower extracts inhibited Shigella dysenteriae, though they were ineffective against E. coli and Salmonella enteritidis, as their potential in antimicrobial activity [26]. Infusions of dried flower heads (1 mg/mL) protected human blood cells from oxidative stress, elevating catalase, superoxide dismutase, glutathione peroxidase, and intracellular glutathione while lowering lipid peroxidation, as its antioxidant activity [26]. A 5% hot water extract reduced recalcification time in human plasma by 43% versus saline (p < 0.001). Interestingly, pressed plant juice delayed clotting (p < 0.05–0.001); it shows a hemostatic effect [26]. In mice, aqueous flower extracts reduced yeast-induced paw oedema by 35%, comparable to indomethacin (44%) and phenylbutazone (26%). The active agents were water-soluble protein–carbohydrate complexes with low toxicity [26]. Additional in vitro studies confirmed that flavonoids, sesquiterpene lactones, and proteins downregulated inflammatory markers such as ICAM-1 in endothelial cells and nitric oxide in LPS-stimulated macrophages, demonstrating their anti-inflammatory properties [30–34].
Compounds such as: achillicin, achillin, azulene, chamazulene, isoartemisia ketone, millefoliumines A–Q, and millefoliumons A–F have shown anticancer and anti-inflammatory activities, whereas α-pinene, β-pinene, borneol, camphor, 1,8-cineole, sabinene, caryophyllene, terpinen-4-ol, and α-thujone have demonstrated antimicrobial activity. Along with this, apigenin (glucosides), chlorogenic acid, chrysoeriol, luteolin (glucosides), rutin, and dicaffeoylquinic acid isomers have exhibited antioxidant effects, while loliolide, (+)-isololiolide, (+)-dehydrovomifoliol, 3α-hydroxy-epoxy-ionones, (−)-10-hydroxydihydroactinidiolide, 8-hydroxycarvotanacetone, and 3-(4-hydroxyphenyl)-1-propanol have shown cytoprotective and metabolic regulatory activities. On top of that, achilleine possesses hemostatic (rapid blood-clotting) activity. Luteolin, a naturally occurring tetrahydroxy flavonoid widely distributed in medicinal herbs and foods, exhibits a broad spectrum of pharmacological activities encompassing anti-aging, anticancer, antioxidant, anti-inflammatory, anti-migratory, anti-osteoporotic, and hepatoprotective effects [35–40].
Anti-aging/anti-degenerative activity: Luteolin targets cellular senescence by modulating oxidative-stress pathways. Network pharmacology revealed 145 shared senescence-related targets, enriched in p53 and PI3K-Akt signaling. Molecular dynamics simulations confirmed strong binding to p53, CDK2, and CDK4, while functional assays in SW1353 cells showed reduced β-galactosidase staining and downregulation of MMP13, p53, and p21, with concomitant increases in CDK2, CDK4, and COL2A1 expression [38].
Anticancer effects: In lung adenocarcinoma A549 cells, luteolin induced concentration- and time-dependent apoptosis by activating caspase-3 and caspase-9, downregulating Bcl-2, and upregulating Bax through the MEK-ERK and Akt signaling cascades. Pharmacological inhibition of MEK-ERK attenuated these pro-apoptotic effects, confirming pathway dependence [37]. Molecular docking identified the androgen receptor (AR) as a high-affinity target; Western blot analysis demonstrated reduced AR expression, and mutation of the AR phosphorylation site at position 877 diminished luteolin’s growth-inhibitory action. In vivo, luteolin significantly curtailed A549 xenograft expansion by days 25 and 28 with a corresponding decline in AR expression [35].
Immunomodulatory/immune-checkpoint inhibition: Luteolin, together with the flavonoid apigenin, suppressed IFN-γ-induced PD-L1 expression in KRAS-mutant non-small cell lung cancer (NSCLC) by blocking STAT3 phosphorylation. Both flavonoids enhanced T-cell infiltration and apoptosis in H358 xenografts and Lewis lung carcinoma models, and apigenin additionally reduced tumor burden in KRASLA2 mice. Combination therapy with PD-1 antibodies (e.g., Keytruda) further amplified antitumor effects, indicating a synergistic role for luteolin or apigenin with immune-checkpoint blockade [36].
Hepatoprotective activity: Luteolin exerts protective effects in metabolic-associated fatty liver disease, hepatic fibrosis, and hepatocellular carcinoma by limiting oxidative stress, dampening inflammatory mediators, modulating lipid metabolism, inhibiting extracellular matrix deposition, and promoting both apoptosis and autophagy in malignant hepatocytes. Despite promising efficacy, its clinical translation requires strategies to overcome low oral bioavailability caused by rapid metabolism [39].
Cardiovascular protection/vascular calcification (VC): In high-fat diet and vitamin D3-induced rat models of VC, luteolin reduced calcification, oxidative stress, and apoptosis. It decreased malondialdehyde (MDA), lactate dehydrogenase (LDH), and reactive oxygen species (ROS), and downregulated cleaved caspase-3, cleaved caspase-9, β-catenin, and BMP-2. Concurrently, luteolin enhanced the expression of autophagy markers and activated Sirtuin 1 (SIRT1), as supported by molecular docking that revealed direct binding. These actions were mediated through the SIRT1/CXCR4 signaling axis, pointing out luteolin’s therapeutic potential against VC and related cardiovascular disorders [40].
Apigenin-flavonoid; immunomodulatory, anti-inflammatory, and anticancer (synergistic PD-1/PD-L1 inhibition in KRAS-mutant NSCLC) [36].
Luteolin-flavonoid; anti-aging/anti-degenerative [38], anticancer and anti-migratory [35, 37], antioxidant and anti-inflammatory [35–40], hepatoprotective [39], and vasculoprotective via SIRT1 activation [40].
A fragrant herb from the Lamiaceae family is primarily cultivated for its EO, which is highly valued across cosmetics, fragrances, food, beverages, and pharmaceutical industries due to its distinctive aroma and bioactive compounds [41]. The main active component, patchouli alcohol (PA), serves as a key quality marker, while norpatchoulene contributes to the unique warm and spicy fragrance. Java is the leading producer, contributing over 66% of the global supply, followed by China and Malaysia. In traditional Chinese medicine, patchouli has been employed to treat headaches, nausea, colds, and diarrhea, and is also applied in hair care and aromatherapy. It is a major ingredient in well-known Chinese patent medicines such as Huoxiang Zhengqi Koufuye (oral liquid) and Baoji Pian (tablets). Patchouli has a long history of use in traditional Chinese and Indian herbal medicine and Ayurvedic therapies for treating various ailments, and extensive pharmacological and pharmacognostic studies support its wide therapeutic potential [41].
Skin aging is driven by intrinsic factors, including genetic and metabolic processes, and extrinsic factors, such as environmental exposure, which cumulatively alter skin structure and function. Cellular senescence, oxidative damage, and telomere shortening are major hallmarks of intrinsic aging, while telomerase activity, which could counteract telomere loss, is largely inactive in skin cells [42]. In silico studies suggest that compounds derived from P. cablin may activate telomerase via the PI3K-Akt signaling pathway. Molecular docking and dynamics simulations showed strong binding of patchouli constituents to key proteins AKT1, HSP90AA1, and HSP90AB1, implicating their role in promoting telomerase activity and cellular longevity [42].
P. cablin EO and its nanoemulsion exhibit potent insecticidal activity against the maize weevil, Sitophilus zeamais. Chemical profiling identified fifteen constituents, dominated by patchoulol (~55%). Both bulk oil and nanoemulsion caused rapid mortality (median lethal time ~53 h) across nine geographically distinct populations under topical, contact, and fumigation exposure. Lethal concentrations ranged from 0.30–6.37 µL mL–1 (contact) and 0.029–0.037 µL mL–1 (fumigation). Repellency was significant, and reproductive indices dropped below replacement levels at ≥ 50 µL kg–1, indicating strong suppression of population growth and grain consumption. Some geographic variation in susceptibility was observed, with Viçosa insects most sensitive and Sete Lagoas least, but the overall pattern demonstrates consistent insecticidal and anti-reproductive effects, supporting PEO as an eco-friendly alternative to synthetic pesticides [43].
PCEE (P. cablin ethanolic extract) demonstrates anti-aging potential in doxorubicin-induced 3T3-L1 fibroblasts by reducing ROS, suppressing the senescence-associated β-galactosidase gene GLB-1, and maintaining telomere length. These findings highlight its role as a natural antioxidant that may delay cellular aging and prevent degenerative changes. While current evidence includes in vitro and limited in vivo studies, further animal studies and human clinical trials are required to confirm its therapeutic relevance for promoting healthy aging [44].
Regional variations in PEO composition were observed across five cultivation sites in Aceh Province, Indonesia. Steam-distilled oils analyzed via GC-MS showed that Sabang produced the highest oil yield (~4%), while Gayo Lues exhibited the highest total sesquiterpene content (~85%), including β-patchoulene, caryophyllene, α-guaiene, and aciphyllene. The inverse relationship between oil yield and sesquiterpene content corroborates the influence of local climate and geography on secondary metabolite profiles. These findings suggest Gayo Lues as the preferred site for large-scale cultivation when high-quality, sesquiterpene-rich oil is desired, while highlighting the need to consider environmental factors to maintain consistent commercial quality [45].
Oral administration of patchouli essential oil (PEO) alleviates anxiety- and depression-like behaviors in a chronic unpredictable mild stress (CUMS) rat model. Open-field and forced-swim tests indicated reversal of stress-induced locomotor and depressive deficits. Gut microbiome analysis revealed increased abundance of beneficial genera (Bacteroides, Blautia) and reduced Ruminococcus_1 and Ruminococcus_2. Short-chain fatty acids were modulated, with elevated propionic acid and decreased caproic acid, and hippocampal serotonin (5-HT) levels were restored. These results suggest that PCO exerts anxiolytic and antidepressant effects via the gut–brain axis, presenting a potential natural therapy for stress-related mood disorders with minimal side effects compared to conventional drugs [46].
Phytochemical composition and pharmacological activities of P. cablin include flavonoids, terpenoids, phytosterols, organic acids, lignins, glycosides, alcohols, pyrones, and aldehydes. Key compounds such as: PA, patchoulene epoxide, α- and β-patchoulene, α-bulnesene, seychellene, norpatchoulenol, pogostone (PO), eugenol, pogostol, and pachypodol contribute to antioxidant, antimicrobial, antitumor, antidiabetic, anti-inflammatory, anti-ulcer, antihypertensive, antiplatelet, antidepressant, and aphrodisiac activities. The high therapeutic value and global demand make P. cablin an economically important crop, and modern in vitro culture techniques support the large-scale production of uniform, pathogen-free plants with consistent bioactive profiles [47].
In silico analysis of PEO identified 44 constituents, of which 26 passed ADME and toxicity screenings, indicating favorable pharmacokinetic properties and safety. Reverse docking against skin-aging enzymes MMP13, MMP9, and hyaluronidase highlighted strong binding of 3,7,11-trimethyl-dodeca-2,4,6,10-tetraenal to MMP9 (binding energy –8.2 kcal/mol), with molecular dynamics simulations confirming interaction stability. These findings suggest that PEO bioactive compounds may inhibit collagenase and hyaluronidase activity, supporting anti-aging and skincare applications [48].
Defense mechanisms of P. cablin against bacterial wilt (SY1 pathogen) involve coordinated lipid and hormone signaling. Integrated transcriptomic and metabolomic analyses revealed activation of lipid metabolic pathways and increased ceramide accumulation. Hormone assays showed elevated abscisic acid (ABA) levels with upregulation of ABA biosynthesis and downstream genes. Treatment with the ceramide synthase inhibitor FB1 reduced ceramide and ABA levels, demonstrating that ceramide accumulation and ABA signaling function synergistically in pathogen defense, providing targets for enhancing resistance and promoting sustainable cultivation [49].
Antifungal activity of patchouli leaf extract was demonstrated on heat-cured acrylic resin denture bases. Immersion in extract concentrations of 25%, 50%, and 75% significantly inhibited Candida albicans growth, suggesting its utility as a natural denture-cleaning agent to prevent denture stomatitis. Further studies are required to assess the impact of prolonged exposure on the physical and mechanical properties of acrylic resins [50].
The antibacterial activity of Aceh patchouli leaf ethanol extract was evaluated against Enterococcus faecalis, a common pathogen in secondary root canal infections. GC-MS analysis revealed a high content of sesquiterpenes and fatty acids, with PA as the major constituent. The extract demonstrated significant activity, with a MIC of 12.5%, highlighting its potential as a natural alternative or adjunctive therapy to conventional antibiotics, helping to mitigate antibiotic resistance risks [51].
PO, a major constituent of P. cablin, demonstrates potent activity against nonalcoholic fatty liver disease (NAFLD) [52]. In vitro, PO reduced intracellular lipid accumulation, promoted fatty acid oxidation (FAO), and downregulated pro-inflammatory mediators, including NLRP3, while upregulating FAO-related genes. In high-fat-diet animal models, PO administration decreased body weight, liver steatosis, fibrosis, and serum pro-inflammatory cytokines, with diminished efficacy in NLRP3-knockout mice, confirming NLRP3 as a primary molecular target [52]. Molecular docking further supported strong PO–NLRP3 binding [52]. PO also attenuated adipose-tissue inflammation linked to obesity by activating SIRT1, thereby suppressing MAPK and NF-κB signaling, reducing macrophage migration, and elevating anti-inflammatory IL-10 in LPS-challenged mice; SIRT1 inhibition reversed these benefits, thus demonstrating hepatoprotective and metabolic regulation [53].
Patchouli oil contains a diverse range of sesquiterpenes and monoterpenes that collectively modulate inflammatory pathways, particularly through platelet-activating factor (PAF) signaling [54]. Among these constituents, α-bulnesene contributes antioxidant potential, while α-guaiene provides fragrance along with additional bioactivity. Likewise, α-patchoulene influences anti-inflammatory mechanisms, and α-selinene offers antioxidant support. Likewise, β-caryophyllene interacts with cannabinoid receptors, delivering anti-inflammatory and analgesic effects, whereas β-cubebene imparts aromatic qualities and exhibits mild antimicrobial activity. Besides, β-elemene demonstrates notable anticancer and anti-inflammatory properties, and β-patchoulene enhances the oil’s overall therapeutic synergy [54].
Patchouli oil comprises a range of bioactive sesquiterpenes and related compounds that contribute to its diverse pharmacological profile. α-Bulnesene exhibits antioxidant and anti-inflammatory properties, whereas α-guaiene functions as an antioxidant and aromatic contributor [54]. The principal constituent, PA (patchoulol), is the dominant fragrance compound and exhibits antioxidant, anti-inflammatory, antiviral (notably against influenza H1N1 and H2N2), antidepressant, photoprotective, and skin-protective effects through the enhancement of endogenous antioxidant enzymes (SOD, CAT, and GPx) [54–56]. Plus, PO provides hepatoprotective effects in non-alcoholic fatty liver disease (NAFLD) and exerts anti-inflammatory activity via NLRP3 inflammasome inhibition, acting as a metabolic regulator through the SIRT1/MAPK/NF-κB pathways [52]. Finally, pogostol exhibits antiemetic activity, further contributing to the therapeutic potential of patchouli oil [57].
Gastroprotection through maintenance of intestinal barrier integrity and modulation of TNF-α and nitric oxide; antifungal activity of patchoulol and PO against Aspergillus spp. and Candida albicans; anti-mutagenic suppression of UMU gene expression; antiemetic action of PA and pogostol; anti-allergic and anti-anaphylactic effects; antinociceptive analgesia (delayed writhing, prolonged hot-plate latency); antidepressant effects enhanced by microencapsulation; strong antioxidant protection of neuronal and skin cells; and antibacterial activity against Staphylococcus aureus and Helicobacter pylori via urease inhibition and membrane disruption [57–75].
The therapeutic breadth of P. cablin is largely attributable to PA and PO [52], complemented by a suite of sesquiterpenes (α-bulnesene, α-guaiene, α-patchoulene, α-selinene, β-caryophyllene, β-cubebene, β-elemene, β-patchoulene, β-pinene, and γ-patchoulene) [52, 54, 56, 59, 60, 62–65, 67–70, 73, 74, 76]. These compounds collectively confer anti-inflammatory, antioxidant, antimicrobial, antiviral, gastroprotective, neuroprotective, antinociceptive, and dermatoprotective activities, substantiating the plant’s long-standing traditional and modern medicinal applications.
Patchouli oil contained a variety of bioactive sesquiterpenes and monoterpenes that collectively contributed to its pharmacological and aromatic properties. α-Bulnesene was identified as a sesquiterpene with anti-inflammatory and antioxidant potential, and it also contributed to the characteristic aroma of the oil [54]. Likewise, α-guaiene supported the earthy fragrance profile and exhibited possible anti-inflammatory effects [54], whereas α-patchoulene, a minor sesquiterpene, influenced anti-inflammatory mechanisms [54]. Along with this, α-selinene functioned as an antioxidant compound that helped reduce oxidative stress [54]. Along with that, β-caryophyllene interacted with cannabinoid type-2 (CB2) receptors, thereby exerting anti-inflammatory and analgesic effects [54], while β-cubebene displayed mild antimicrobial and anti-inflammatory activity [54]. The principal constituent, PA (patchoulol), demonstrated potent anti-inflammatory and antimicrobial activity, as it inhibited PAF signaling and reduced hypersensitivity and anaphylactic responses [54, 56–59, 62, 63, 65, 68–71, 73, 74, 76]. Over and above that, PO suppressed NLRP3 inflammasome activation, reduced hepatic lipid accumulation, and alleviated adipose tissue inflammation through the SIRT1/MAPK/NF-κB pathways [52, 53].
Patchouli oil and its constituents exhibited a range of biological activities. α-Selinene, β-elemene, PA, and PO demonstrated antioxidant and cytoprotective effects, as they reduced ROS, protected skin and neuronal cells, and enhanced endogenous antioxidant enzymes such as SOD, CAT, and GPx [53, 56–59, 62–65, 68–71, 73, 74]. Likewise, PA showed dose-dependent antiviral activity by inhibiting influenza viruses H1N1 and H2N2 and reducing plaque formation [58, 62, 65, 74], whereas PO exerted direct antiviral and anti-inflammatory effects, which were particularly relevant to NAFLD and metabolic inflammation [58, 62, 65, 74]. As well as that, PA displayed antibacterial activity against Staphylococcus aureus and Helicobacter pylori through urease inhibition and membrane disruption, while PO exhibited strong antifungal activity against Aspergillus spp. and Candida albicans, and its structural analogs were noted to enhance efficacy [55, 56, 59, 62, 64–67, 71, 72, 74, 76].
Moreover, PA demonstrated gastroprotective effects by preserving intestinal barrier integrity and modulating membrane fluidity, TNF-α, and nitric oxide levels [59–62, 65, 74], and it also exerted neurological, antidepressant, and antinociceptive activities, as evidenced by behavioral models showing antidepressant effects, delayed writhing, and increased hot-plate latency [59–62, 65, 74]. Together with this, PA provided dermatological and anti-photoaging benefits through up-regulation of antioxidant enzymes SOD, CAT, and GPx [56, 59, 62, 63, 65–67, 69–72, 74, 75], and it exhibited anti-mutagenic activity by suppressing UMU gene expression in Salmonella typhimurium, indicating DNA-protective action [56, 59, 62, 63, 65–67, 69–72, 74, 75].
Bryophyllum pinnatum (family Crassulaceae) is a perennial succulent commonly known as the air plant, cathedral bells, life plant, or miracle plant. Although native to Madagascar, it is now widely naturalized across tropical and subtropical regions [77, 78]. For generations, it has been valued in traditional medicine for its broad therapeutic spectrum, offering a natural alternative to synthetic drugs because of its ready availability and comparatively low risk of adverse effects [77].
The plant is used to manage a variety of conditions: respiratory complaints such as asthma and chest colds; skin problems including: eczema, burns, blisters, and insect bites; digestive disturbances like abdominal pain and constipation; inflammatory disorders such as rheumatoid arthritis; neurological issues including epilepsy and certain psychiatric illnesses; infectious diseases like cholera and chickenpox; ocular ailments such as conjunctivitis; and circulatory problems including edema and piles. It also holds significance as a tocolytic agent that helps prevent premature labor [77, 78].
Phytochemical investigations reveal a rich profile of flavonoids, phenolic acids, and steroids that underpin these effects [77]. The major constituents and their principal pharmacological activities are listed below, within each activity:
Caffeic acid, coumaric acid, gallic acid, isorhamnetin, kaempferol, quercetin, and quercitrin exhibit strong free-radical scavenging, membrane-stabilizing, and cytokine-modulating properties that contribute to the management of inflammatory disorders and oxidative-stress–related conditions [77, 78].
Bryophyllin A and Bryophyllin C display pronounced antibacterial and antiviral actions, supporting traditional uses against infections such as cholera and chickenpox [77, 78].
Bersaldegenin, Bryophollone, Bryophyllol, and Bryophynol have been reported to exert cytotoxic effects on tumor cells, suggesting potential anticancer relevance [77, 78].
Campesterol and stigmasterol assist in regulating cholesterol metabolism and exhibit mild anti-inflammatory and immunomodulatory effects [77, 78].
Thus, collectively, these diverse bioactive molecules explain the wide therapeutic range of Bryophyllum pinnatum, validating its continued use across cultures as a multipurpose medicinal plant [77, 78].
Bufadienolides in Bryophyllum pinnatum demonstrate strong tocolytic activity at low concentrations (as little as 1 µg/mL), making them highly potent. However, this potency comes with pharmacological risk, as bufadienolides can interfere with cardiac Na⁺/K⁺ ATPase activity, potentially causing arrhythmias and cardiac toxicity if overdosed. While in vitro studies show no reduction in myometrial cell viability at effective doses, previous research has documented toxic effects in animal models at relatively modest dose increases. These findings suggest that although bufadienolides are effective uterine relaxants, their narrow therapeutic window warrants careful consideration in clinical applications [79].
While bufadienolides in Bryophyllum pinnatum exhibit potent tocolytic effects at low concentrations without compromising myometrial cell viability in vitro, similar compounds isolated from Kalanchoe daigremontiana—a related Crassulaceae species—have demonstrated significantly higher toxicity. In K. daigremontiana, five structurally related bufadienolides, particularly the orthoacetates daigremontianin and bersaldegenin 1,3,5-orthoacetate, have been identified as the main contributors to the plant’s pronounced toxic, sedative, and positive inotropic effects. Unlike B. pinnatum, where bufadienolides show therapeutic potential with a relatively safe in vitro profile, the bufadienolides from K. daigremontiana are associated with systemic toxicity and a clear risk of poisoning. This comparison highlights the variability in bufadienolide toxicity across Crassulaceae species, emphasizing the importance of precise compound characterization and dose regulation in medicinal applications [80].
The systemic and cardiotoxic effects associated with bufadienolides from Bryophyllum pinnatum and particularly the more hazardous Kalanchoe daigremontiana, the primary toxicity concern with Achillea millefolium (yarrow) lies in its potential to induce allergic contact dermatitis. Although not systemically toxic at therapeutic or topical doses, yarrow contains a group of previously unidentified guaianolide-type sesquiterpene lactones—particularly alpha-peroxyachifolid—which act as strong sensitizers. Guinea pig studies and clinical follow-ups of Compositae-sensitive patients revealed that more than 50% exhibited allergic reactions upon exposure to yarrow extracts. Unlike bufadienolides, which exert potent pharmacological and toxic cardiovascular effects, the sesquiterpene lactones in yarrow present a localized dermatological hazard, highlighting the importance of screening for contact allergens in herbal preparations, even in the absence of systemic toxicity [81].
Butea monosperma (Palash)—a revered Ayurvedic plant—has long been valued for both medicinal and cosmetic applications, and it enriches culinary products with natural color and aroma [78]. The plant demonstrates wide-ranging pharmacological effects, including: antidiabetic, antifertility, anti-inflammatory, antimicrobial, anthelmintic, anticataract, anticonvulsant, antidiarrheal, antigiardial, antihypertensive, antioxidant, chemopreventive, free-radical scavenging, hepatoprotective, radioprotective, tumor-suppressive, and wound-healing properties [78, 82]. Traditionally, Palash treats chickenpox, smallpox, fevers, colds, viral infections, and numerous digestive disorders such as gastritis, esophagitis, irritable bowel syndrome, and enteritis, supporting overall gastrointestinal health [82].
Palash flowers are also prepared as herbal tea infused with saffron, blending potent antioxidant and anti-inflammatory actions with mood-enhancing and immune-modulating effects. The tea aids digestion, promotes blood purification and respiratory relief, and provides a soothing, aromatic experience [78]. Although consumers often associate the tea with antidiabetic activity and its distinctive aroma, its antioxidant and immune-boosting potential adds further therapeutic value [78].
Antioxidant/free-radical scavenging: Flavonoids (butrin and isobutrin), tannins, and triterpenoids protect against oxidative stress, supporting chemopreventive and anticataract activity [77, 78, 82].
Anti-inflammatory and antimicrobial: Butrin and butein reduce inflammation and combat microbial infections, contributing to wound healing and relief of respiratory infections [77, 78, 82].
Antidiabetic and hepatoprotective: Chalcones (butein) and flavonoids improve glucose regulation and protect hepatic tissue [77, 78, 82].
Antifertility and anticonvulsant: Isoflavonoids exhibit estrogenic modulation and neuroprotective effects, reducing seizure risk [77, 78, 82].
Antihypertensive and antitumor: Phytosterols and triterpenoids assist in vascular relaxation and inhibit tumor cell growth [77, 78, 82].
Peumus boldo (Boldo), native to Chile and integral to South American traditional medicine, was first documented in the 1700s by Jesuit scholar Juan Ignacio Molina [83]. Boldine, the plant’s principal alkaloid, underpins its medicinal efficacy. Boldine exhibits strong choleretic and cholagogue effects, enhancing bile flow and supporting digestive function [83].
Advanced molecular modelling shows boldine forms stable, non-covalent interactions with the farnesoid X receptor (FXR), paralleling natural ligand chenodeoxycholic acid (CDA) and synthetic agonist colifexor (CFX) [83]. This interaction explains boldine’s therapeutic promise in primary sclerosing cholangitis and non-alcoholic steatohepatitis (NASH) [83].
Boldine: Potent antioxidant and hepatoprotective agent; choleretic and cholagogue, aiding bile production and lipid metabolism.
Chromolaena odorata is traditionally applied for epistaxis (nosebleeds) and wound care [84]. GC-MS identified eight bioactive compounds with antithrombotic, antioxidant, and anti-inflammatory activities [84]. Using a rabbit model, a leaf-extract gel—applied twice daily for 21 days—significantly shortened clotting time and promoted angiogenesis (p = 0.018) without altering CD34/CD68 expression or fibroblast proliferation. These effects correlated with modulation of G-protein–coupled receptors, ion channels, and enzymes, and inhibition of kinases, nuclear receptors, and proteases, confirming the extract’s hemostatic and tissue-repair potential [84].
Antioxidant and anti-inflammatory compounds (eight GC-MS–identified constituents): Stimulate angiogenesis and immune modulation, enhancing blood clotting and tissue regeneration.
Antithrombotic agents: Facilitate haemostasis and protect vascular integrity.
Collectively, Butea monosperma, Peumus boldo, and Chromolaena odorata illustrate how diverse phytochemicals—boldine, butein, butrin, chalcones, flavonoids, isoflavonoids, phytosterols, tannins, triterpenoids, and multiple GC-MS–detected metabolites—exert antioxidant, anti-inflammatory, antimicrobial, hepatoprotective, antidiabetic, choleretic, angiogenic, antithrombotic, and chemopreventive actions [78, 82–84].
These plants exemplify the integration of ethnobotany and modern pharmacology, validating traditional knowledge through contemporary biochemical and computational evidence.
Mango (Mangifera indica L.) leaves are nutrient-dense, providing essential vitamins and minerals that support cellular function, immunity, and overall physiological health. Quantified vitamins include vitamin A (121 µg), vitamin B (189 µg), vitamin C (30 µg), and vitamin E (10 µg). Key minerals comprise calcium (368 mg), iron (343 mg), magnesium (98 mg), manganese (3 mg), nitrogen (2 mg), phosphorus (480 mg), potassium (589 mg), sodium (28 mg), and zinc (14 mg) [85].
Known in Chinese medicine as Folium mangiferae, mango leaves have a long history of therapeutic application. Early documentation appears in classical texts such as South of the Five Ridges Medicinal Records, Luchuan Book on Chinese Herbal Medicine, and Nanning City’s Materia Medica (1959, Vol. I). Official standards were later included in the Chinese Materia Medica Dictionary (1977) and in the Guangxi, Guangdong (2004), and Yunnan pharmacopoeias. Cultivation spans Taiwan, Yunnan, Guangxi, Guangdong, Hainan, and Fujian, making the leaves an abundant herbal resource [86].
A patented preparation process subjects mango leaves to reflux extraction with an aliphatic alcohol (1–4 carbon atoms), followed by purification with a low-polarity organic solvent to yield a highly soluble mango leaf extract. This extract, rich in mangiferin and homomangiferin, lowers serum uric acid and exerts a hypoglycemic effect in hyperuricemic mice, outperforming mangiferin alone and supporting its use in the prevention or management of metabolic syndrome (MS) [86].
MS—often termed the “deadly quartet”—is characterized by abdominal obesity, insulin resistance or impaired glucose tolerance, hypertension, hypertriglyceridemia, reduced HDL-cholesterol, hyperuricemia, and gout, all of which synergistically elevate the risk of type 2 diabetes mellitus (T2DM) and cardiovascular or cerebrovascular diseases [86].
Catechol, Ellagic acid, Gallic acid, Kaempferol acid, Mangiferin, Quercetin, Shikimic acid: Suppress acute, subacute, and chronic inflammation; ethanolic kernel extracts reduce pain and swelling; leaf extracts inhibit Bacillus subtilis, Staphylococcus albus, and Vibrio cholera [85].
Strong antioxidant and antibacterial activities [85].
Astragalin, Fisetin, Gallic acid, Isoquercitrin, Methylgallate, Quercetin: Neutralize free radicals, protect DNA, and inhibit colon and breast tumor growth [85].
Mangiferin: Demonstrates notable in-vitro activity against Herpes Simplex Virus type 2 [85].
Dietary fibre, Pectin, Potassium, and vitamin C: Reduce serum cholesterol, regulate heart rate, and help maintain normal blood pressure [85].
Homomangiferin, Mangiferin: Provides hypoglycemic and uric-acid-lowering effects, improving insulin sensitivity and mitigating risk factors of MS [86].
Mangiferin, Tannins: Soothe and repair inflamed gastrointestinal mucosa, alleviating gout-related intestinal irritation [85].
Vitamin E: Supports hormonal balance and sexual vitality, contributing to mango’s traditional reputation as a “love fruit” [85].
Different plant parts—bark, root, leaves, fruit, and flowers—have been used globally for conditions such as: syphilis, uterine disorders, wounds, ulcers, vomiting, bleeding disorders, and other gastrointestinal ailments [85].
Phytochemicals found in Mangifera indica (mango leaves and other parts) with their respective pharmacological or medicinal activities (Table 3, Table 4).
| Phytochemical | Pharmacological/Medicinal activities |
|---|---|
| Mangiferin | Antioxidant, anti-inflammatory, hypoglycemic, hypouricemic, antiviral (HSV-2), antiasthmatic, anti-allergic. |
| Homomangiferin | Enhanced solubility contributes to hypoglycemic and uric acid-lowering effects. |
| Quercetin | Antioxidant, anticancer (colon, breast), cytoprotective. |
| Gallic acid | Antioxidant, anti-inflammatory, anticancer. |
| Catechol | Antioxidant (general), possible antimicrobial. |
| Shikimic acid | Antiviral precursor (used in the synthesis of antiviral drugs like oseltamivir). |
| Kaempferol | Antioxidant, anticancer, cytoprotective. |
| Kaempferol acid | Antioxidant, anti-inflammatory, anticancer. |
| Ellagic acid | Antioxidant, anticancer. |
| Thujene (volatile) | Antimicrobial, anti-inflammatory (based on general knowledge of monoterpenes). |
| Flavonoids (general) | Antioxidant, anti-inflammatory, antimicrobial, cytoprotective. |
| Tannins | Antioxidant, antimicrobial, anti-inflammatory, detoxifying. |
| Alkaloids | Analgesic, antimicrobial, anti-inflammatory (varies by specific type). |
| Saponins | Immune-boosting, antimicrobial, anti-inflammatory. |
| Cardiac glycosides | Cardiotonic, detoxifying (caution: can be toxic in high doses). |
| Phenolic compounds | Antioxidant, anti-inflammatory, anticancer. |
| Isoquercitrin | Antioxidant, anticancer. |
| Astragalin | Antioxidant, anticancer, anti-inflammatory. |
| Fisetin | Antioxidant, anticancer, neuroprotective. |
| Methylgallate | Antioxidant, anticancer. |
| Vimang (mangiferin-rich extract) | Anthelmintic, anti-allergic. |
| Compound/Nutrient | Medicinal or physiological benefit |
|---|---|
| Vitamin A | Vision, immunity, and cell growth. |
| Vitamin B (various) | Metabolism, energy production, and nervous system health. |
| Vitamin C | Antioxidant, immune support, and collagen synthesis. |
| Vitamin E | Antioxidant, supports hormonal balance, and has aphrodisiac properties. |
| Sodium, Potassium | Electrolyte balance, blood pressure regulation. |
| Calcium, Magnesium | Bone health, enzyme activation, muscle and nerve function. |
| Iron, Zinc | Blood formation, immunity. |
| Phosphorus, Nitrogen | Cellular function, protein, and DNA synthesis. |
| Manganese | Antioxidant enzyme cofactor, bone formation. |
| Pectin (fiber) | Cholesterol-lowering, gut health. |
| Dietary fiber | Digestive health, cholesterol reduction. |
The scientific investigations into the phytochemistry and bioactivity of several medicinal plants revealed a diverse array of therapeutic potentials, primarily attributed to their rich secondary metabolite profiles. S. undulata was studied through GC-MS and LC-ESI-MS analyses, which uncovered 25 volatile and 21 phenolic compounds across different plant parts. The aerial portions, especially leaves and flowers, exhibited a greater diversity and abundance of volatiles and phenolics, including key compounds such as: chlorogenic acid, quinic acid, kaempferol, apigenin, and luteolin-7-O-glucoside. These constituents appeared to contribute significantly to the plant’s bioactivities.
In terms of antioxidant potential, the aerial extracts of S. undulata showed superior activity in DPPH assays compared to the tubers, with ultrasound-assisted ethanolic extracts achieving up to 25.06% inhibition at 50 µg/mL. Cytotoxic assessments further highlighted the bioefficacy of the plant, with methanolic extracts from aerial parts (ASU) and roots (RSU) demonstrating significant inhibition of MCF-7 breast cancer cells. The aerial extracts showed stronger activity (IC50 ≈ 4.22 mg/mL), possibly due to higher flavonoid content. These extracts induced apoptosis and mitigated oxidative stress, suggesting a multifaceted mechanism of action. Likewise, antimicrobial and anti-inflammatory activities were noted, especially in volatile-rich aerial parts containing terpenoids and sesquiterpenes. Polysaccharides from the roots also exhibited anti-inflammatory and antioxidant effects, alongside cytotoxicity against HeLa cells. However, the extraction method and solvent type significantly influenced the yield and bioactivity, indicating that standardization across plant parts remains a challenge.
In comparative studies, Andrographis paniculata was investigated for its antiviral efficacy. Clinical trials revealed that ethanolic extracts containing andrographolide hastened recovery from COVID-related symptoms like headaches and anosmia without causing hepatic or renal toxicity. Though overall disease progression did not significantly differ from placebo, the compound showed immunomodulatory benefits. Pharmacokinetic studies further showed nonlinear oral bioavailability, suggesting a saturation effect in absorption or metabolism at higher doses.
Noni (M. citrifolia) also demonstrated promising pharmacological properties. Ethanol and methanol extracts from its fruit yielded high extraction rates and exhibited antimicrobial and cytotoxic effects. Ethanol extracts were particularly potent against UMUC-3 cancer cells and showed greater efficacy than methanolic counterparts. The anticancer activity of noni was supported by multiple mechanisms, including apoptosis induction and anti-angiogenic effects.
For HC, strong antiviral efficacy against the dengue virus was established through direct-contact assays, where the extract significantly suppressed viral RNA (EC50 ≈ 0.8 µg/mL) and reduced virion release in infected cells. Hyperoside was identified as a major constituent and likely contributor to this activity. Importantly, the extract was non-genotoxic at effective doses. The plant’s chemical complexity—encompassing aristolactams, flavonoids, oxoaporphines, and sterols—was thought to underlie its broad-spectrum antiviral effects.
Achillea millefolium (common yarrow) was historically used for various ailments, and scientific analyses confirmed its phytochemical richness and therapeutic properties. The EO contained constituents like camphor, 1,8-cineole, and artemisia ketone, with variations in chemotype across regions significantly affecting both its chemical makeup and biological activity. Phenolic-rich methanolic extracts contained flavonoids like luteolin, quercetin, and hyperoside. The oil and extracts exhibited anti-inflammatory, antioxidant, antimicrobial, and antifungal activities. Particularly, the oil reduced oxidative stress and inflammatory markers in macrophage assays. Despite its general safety, overuse could result in side effects like headaches or dermatitis.
Further mechanistic insights were obtained with flavonoids such as luteolin and apigenin in the context of KRAS-mutant NSCLC. These molecules reduced PD-L1 levels through the inhibition of STAT3 signaling pathways, thereby enhancing the cytotoxic function of T-cells. When used alongside PD-1 monoclonal antibodies, they enhanced therapeutic outcomes in xenograft and mouse models. Luteolin demonstrated vascular protective effects by modulating autophagy and SIRT1 expression in models of VC.
Bryophyllum pinnatum also displayed therapeutic versatility due to its diverse secondary metabolites. The plant contained bufadienolides (e.g., bryophyllin A and C), flavonoids like afzelin and quercitrin, and phenolic acids. Traditionally used for respiratory and gastrointestinal ailments, the plant was validated for several pharmacological effects—anti-inflammatory, analgesic, antidiabetic, antimicrobial, and anticancer. Specifically, bufadienolides demonstrated the ability to kill cervical cancer cells. Extracts also exhibited neuroprotective, hepatoprotective, and nephroprotective effects. However, bufadienolides were flagged for potential cardiac toxicity in animals, necessitating further safety studies in humans.
Overall, the scientific discussions demonstrated the pharmacological promise of these botanicals, with many aligning closely with traditional uses. Each plant exhibited unique bioactive profiles and mechanisms, reinforcing the importance of compound standardization, optimized extraction methods, and detailed safety assessments for future therapeutic development (Table 5).
Comparative analysis of medicinal plants, phytochemicals, and pharmacological roles.
| Plant (species). | Major phytochemicals identified. | Pharmacological/medicinal activities. | Overlapping mechanisms/shared effects. | References |
|---|---|---|---|---|
| Pogostemon cablin Benth. (Patchouli). | Patchouli alcohol, pogostone, patchoulene epoxide, α-/β-patchoulene, α-bulnesene, seychellene, eugenol, pogostol, pachypodol, flavonoids, terpenoids, phytosterols, and glycosides. | Antioxidant, antimicrobial, anti-inflammatory, anti-diabetic, antihypertensive, antidepressant, aphrodisiac, anti-peptic ulcer, antifungal (C. albicans inhibition), antiaging (ROS suppression, telomere maintenance), and antibacterial (E. faecalis inhibition). | Flavonoid-based antioxidant & anti-inflammatory effects; terpenoid-driven antimicrobial action. | [42, 52, 53, 55] |
| Scorzonera undulata. | Flavonoids (apigenin, luteolin, luteolin-7-O-glucoside, quercetin, kaempferol, naringin), phenolic acids (caffeic, chlorogenic, gallic, ferulic, quinic, vanillic, syringic, protocatechuic), tannins, steroids, fatty acids (palmitic, lactic, malic), and volatile compounds (2,4-di-tert-butylphenol). | Strong antioxidant, anticancer (MCF7 inhibition via apoptosis), anti-inflammatory, anti-diabetic, cardioprotective, and cytoprotective. | The flavonoids luteolin, apigenin, quercetin, and kaempferol replicate antioxidant/anticancer mechanisms. | [10–12] |
| Scorzonera suberosa, S. latifolia, and S. laciniata. | Vitamins (D, K, α-tocopherol, retinol), flavonoids (rutin, myricetin, morin, quercetin, kaempferol), resveratrol, sugars (fructose, glucose, sucrose), and minerals (Ca, K, Fe, Zn, Mg). | Antioxidant, nutritional, anticancer, cardioprotective. | Shared quercetin and kaempferol confirm common antioxidant and cardioprotective pathways. | [11] |
| Andrographis paniculata (Burm. f.) Nees. | Andrographolide (diterpenoid lactone), flavonoids, and phenolics. | Antiviral (esp. SARS-CoV-2), anti-inflammatory, hepatoprotective, and immunomodulatory. | Flavonoid antioxidants parallel Scorzonera species; diterpenoid adds a unique antiviral action. | [13] |
| Houttuynia cordata (mentioned in the methods screening). | Flavonoids (quercitrin, hyperoside, and rutin), volatile oils, and alkaloids. | Antiviral, antimicrobial, antioxidant, and immune modulation. | Flavonoid-driven antioxidant activity overlaps with Scorzonera and Patchouli. | [15, 16, 18] |
| Achillea millefolium (Yarrow). | Flavonoids (apigenin, luteolin, and quercetin), sesquiterpene lactones, tannins, and phenolic acids. | Anti-inflammatory, wound healing, antioxidant, digestive aid. | Flavonoid antioxidants (apigenin, luteolin, and quercetin) overlap with S. undulata & patchouli. | [23, 25, 26, 27, 30–34] |
| Scorzonera species (general). | Multiple flavonoids (luteolin, apigenin, quercetin, kaempferol, and naringin), phenolic acids, fatty acids, steroids, and sugars. | Antioxidant, anticancer, cardioprotective, and neuroprotective. | Common flavonoid mechanisms across plants. | [11] |
| Pogostemon cablin (Aceh variety, dental use). | Patchouli alcohol, sesquiterpenes, fatty acids. | Antibacterial (E. faecalis), antifungal (C. albicans in dentures).Modulate inflammatory pathways, particularly platelet-activating factor (PAF) signaling. | Shares antimicrobial terpenoids + phenolics with other plants. | [53, 55–63, 66, 68–75, 77] |
Hence, based on the accumulated evidence and research, it can be consistently concluded that flavonoids such as: apigenin, luteolin, quercetin, kaempferol, and naringin—commonly found in multiple medicinal plants including S. undulata, S. suberosa, Achillea millefolium, HC, and P. cablin—exhibit recurrent antioxidant, anti-inflammatory, anticancer, cardioprotective, and neuroprotective properties. Phenolic acids, including gallic, caffeic, chlorogenic, ferulic, quinic, syringic, and protocatechuic acids, are particularly abundant in Scorzonera species and contribute significantly to their antioxidant, anti-diabetic, and anti-inflammatory effects. Terpenoids such as andrographolide (in Andrographis paniculata) and PA (in P. cablin) offer distinct antiviral and antimicrobial activities while also complementing flavonoid-mediated pathways. Likewise, minerals and vitamins present in Scorzonera suberosa, S. latifolia, and S. laciniata enhance their nutritional value and supportive antioxidant activity, further amplifying their therapeutic potential. Despite the differences in phytochemical composition, the shared mechanisms of action among these plants primarily converge on ROS scavenging, modulation of key inflammatory signaling pathways (e.g., NF-κB and MAPK), induction of apoptosis in cancer cells, and regulation of metabolic functions related to glucose, lipids, and cardiovascular health.
This body of research collectively validates the pharmacological relevance of traditional medicinal plants and illustrates how modern analytical chemistry can accelerate drug discovery by pinpointing key bioactive markers. S. undulata was shown to contain a wide array of volatile and phenolic compounds throughout its aerial and subterranean parts, with leaves and flowers displaying the greatest antioxidant, antimicrobial, and anti-inflammatory activity, and methanolic extracts exhibiting notable cytotoxicity against breast cancer cells—yet its therapeutic promise depends on standardized, optimized extraction methods and further mechanistic and toxicological studies. Andrographis paniculata provided symptomatic relief in mild COVID-19 but revealed challenges with the non-linear pharmacokinetics of andrographolide, accentuating the need for improved delivery systems. Both M. citrifolia and S. undulata demonstrated potent antimicrobial, antioxidant, and anticancer effects in preclinical models, though their efficacy still requires confirmation through rigorous in vivo and clinical trials, with their rich flavonoid and phenolic profiles serving as the primary drivers of activity. The aqueous extract of HC displayed strong virucidal action against dengue by inhibiting viral replication and preventing virion release, with hyperoside emerging as the principal antiviral agent and a favorable safety record supporting further in vivo testing, dose optimization, and formulation work. Traditional uses of yarrow (Achillea millefolium) were reinforced by evidence of anti-inflammatory, antioxidant, antimicrobial, and hemostatic properties, though regional variation in EO composition highlights the importance of chemotypic standardization and dosage control. Flavonoids such as luteolin and apigenin offered dual benefits—anticancer effects via immune checkpoint suppression in lung cancer and cardiovascular protection through SIRT1-mediated autophagy—yet poor oral bioavailability demands advanced pharmacokinetic and dosing studies. Bryophyllum pinnatum exhibited wide-ranging pharmacological actions, including analgesic, hepatoprotective, nephroprotective, CNS-modulating, antimicrobial, and anticancer effects that align with its ethnopharmacological use, but bufadienolide content raises toxicity concerns requiring careful safety evaluation. Across these botanicals, chemotypic variation, seasonal shifts, and extraction differences necessitate validated chemical markers and Good Manufacturing Practices for consistent potency, while chronic-use toxicity, herb–drug interactions, and dose–response relationships remain underexplored. Although current evidence is largely preclinical, the demonstrated antioxidant, antiviral, antimicrobial, anti-inflammatory, and anticancer activities provide a compelling proof-of-concept for therapeutic development; translating this potential into clinically useful products now hinges on standardized extraction, advanced delivery strategies, systems-biology approaches to clarify molecular targets and synergy, and well-designed human trials to bridge traditional knowledge with evidence-based modern medicine.
5-HT: 5-Hydroxytryptamine (Serotonin)
ABA: abscisic acid
AR: androgen receptor
CUMS: chronic unpredictable mild stress
EA: ethyl acetate
EOs: essential oils
GC-MS: gas chromatography-mass spectrometry
HC: Houttuynia cordata
HCT: Houttuynia cordata Thunb.
HPLC: high-performance liquid chromatography
IL-6: interleukin-6
MAPK: mitogen-activated protein kinase
MICs: minimum inhibitory concentrations
NAFLD: non-alcoholic fatty liver disease
P. cablin: Pogostemon cablin
PA: patchouli alcohol
PCEE: Pogostemon cablin ethanolic extract
PEO: patchouli essential oil
ROS: reactive oxygen species
SCFAs: short-chain fatty acids
SIRT1: Sirtuin 1
TSs: tumorspheres
UAE: ultrasound-assisted extraction
VC: vascular calcification
SDB and SSM: Conceptualization, Methodology, Investigation, Data curation, Formal analysis, Validation, Visualization, Writing—original draft, Writing—review & editing, Funding acquisition, Project administration, Supervision, Resources. Both authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 2295
Download: 45
Times Cited: 0
