Affiliation:
Department of Neurobiology and Anatomy, Drexel University College of Medicine, Philadelphia, PA 19129, USA
Email: tjc29@drexel.edu
Explor Drug Sci. 2026;4:1008150 DOI: https://doi.org/10.37349/eds.2026.1008150
Received: December 22, 2025 Accepted: February 08, 2026 Published: February 24, 2026
Academic Editor: Alessandra Tolomelli, University of Bologna, Italy
The article belongs to the special issue Peptide Science Without Borders: Novel Insights for Drug Discovery
Aim: A seven amino acid cyclic peptide has been applied to human blood plasma treated with glucose metabolite methylglyoxal (MG) in “proof of concept” experiments to determine the peptide’s ability to counteract pathologies associated with hyperglycemia. Similar pathologies are evident during aging and in age-related disorders. In fact, elevated MG levels in the blood lead directly to diabetic complications and accelerated aging, including cognitive decline. These changes are attributed to oxidant stress and amyloidogenesis, the latter involving toxic accumulations of blood and tissue proteins.
Methods: cSKE7 was redesigned from cell survival-promoting and anti-inflammatory fragments near the N-terminus of human/primate “orphan” protein DSEP/Dermcidin and incubated at low micromolar concentrations with the MG-stressed human plasma for 24 hours. The modified design of the new compound offers several practical advantages over predecessors including cyclic stability and a marked increase in aqueous solubility.
Results: The peptide dispersed thioflavin-T-stained amyloid aggregates and reduced oxidant stress as measured by plasma levels of free thiols and of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) activity. Since these N-terminal fragments of DSEP/Dermcidin have been shown to bind and influence the activity of heat shock protein 70 (HSP70), HSP70 inhibitor pifithrin-μ was added to the plasma prior to peptide treatment. The inhibitor disrupted amyloid dispersion and both peptide-induced and, in some cases, normally occurring antioxidant effects, suggesting these reparative activities are HSP70 dependent.
Conclusions: The results are discussed in terms of their potential use in new therapies for the complications of metabolic disease and disorders of aging that result from a deterioration of the quality control mechanisms of proteostasis.
Attention to protein aggregate diseases has increased dramatically over the last several decades due to their involvement in age-related neurodegenerative disorders. Toxic protein accumulations were essentially rediscovered for the brain, since for most of the last century they were understood as features of relatively rare blood diseases, classified generally as amyloidosis [1–3]. While the causes of amyloidosis, or other instances of aggregate pathology, are diverse, one generalized approach to these disorders has remained therapeutically elusive. That approach consists of enhancing endogenous mechanisms of protein quality control, an important part of the balanced process of proteostasis. For this, the prominent role of heat shock proteins (HSPs), especially HSP70, has been emphasized, although attempts at regulation of this protein so far have not been successfully translated to a workable therapy for human aggregate diseases. HSP70 also exists in the circulation, where its role is less well understood but may have functions both similar and distinct from its intracellular versions [4–6].
Targeting toxic protein aggregation and associated oxidative stress has also been considered for metabolic diseases like diabetes and its complications [7–9]. This is especially the case as these pertain to vascular disorders, but also pancreatic and kidney disease, as well as peripheral neuropathies [10–12]. It is now well-known that chronic elevations in blood glucose and its metabolites lead to toxic protein aggregation [7, 9, 13, 14]. The aggregates form from blood and tissue proteins and accumulate in blood vessel walls and in the parenchyma of organs like the pancreas, kidney, brain, and peripheral nerves, establishing a direct link between hyperglycemia and diabetic complications, including age-related cognitive decline [15–20]. The proteins aggregate in a hyperglycemic environment, at least in part, because elevations in glucose’s principal metabolite, methylglyoxal (MG), stimulate the nonenzymatic formation of advanced glycation end products (AGEs); the activities of AGEs are diverse, but a primary effect of their excess is to block redox balance elevating oxidative stress through a complex chemistry that also promotes disruption of protein structure via direct glycation of protein amines as well as by thiol oxidation and crosslinking [21–24]. These changes create exposed hydrophobic patches in the proteins that stick together, often in an amyloid configuration [25], disrupting interactions with binding partners and protein function [26–28]. Thus, the destructive cycle of persistent hyperglycemia is perpetuated, further highlighting the role that a failure of protein quality control could play in the pathogenesis of diabetes and its complications.
It is not surprising, therefore, that HSP70 has been proposed as a therapeutic target for diabetes and its many complications [10, 11, 29, 30], as well as other conditions generally associated with inflammatory and oxidant stressors, especially as these relate to aging [31, 32]. Our laboratory identified peptide fragments near the N-terminus of an endogenous human protein best known as Dermcidin, but identified by us simultaneously as DSEP [33–35]. The fragments included Y-P30, and two shorter peptides, CHEASAAQC (CHEC-9), and CHEASQC (CHEC-7), all with cell survival-promoting and anti-inflammatory properties, in vitro and in vivo [33, 35–39]. The shorter peptides were cyclized by adding a terminal cysteine and verifying cross-linking prior to use by the loss of both 5,5'-dithio-bis(2-nitrobenzoic acid) (DTNB) absorbance and of 2 protons by mass spectroscopy [39]. Subsequently, and most importantly for the present study, a DSEP/Dermcidin fragment that included the CHEC-9 sequence was shown independently to bind HSP70 in detailed peptide mapping studies [40]. Both CHEC-9 and CHEC-7 were also shown to bind and influence HSP70 activity, including the dissolution of amyloid-forming aggregates, and for CHEC-7, this was demonstrated for oxidant-stressed human plasma ex vivo [41]. The present study is a proof-of-concept report designed to test a newly designed peptide fragment in a hyperglycemic model using MG-stressed human plasma, a compound that also has been implicated in cognitive decline [42].
For this study, we redesigned one of the DSEP/Dermcidin peptides to increase stability and solubility. These modifications were considered essential as human therapeutic applications were contemplated (see Materials and methods). The new peptide is cycloSQKCHEA (cSKE7). The data show that cSKE7 inhibits and reverses aspects of MG toxicity in human blood plasma, including amyloidogenesis and elements of oxidative stress as measured by glyceraldehyde-3-phosphate dehydrogenase (GAPDH) activity and thiol redox status, all in an HSP70-dependent manner. We used well-tested assays of the entirety of blood plasma, rather than focusing on specific plasma proteins, to document the potential reparative properties of this peptide. We also used human-derived plasma, noting that the peptide is a re-designed fragment of a particularly human/primate protein (“orphan”) that is not present in other mammals [34]. The goal was to encourage further work with this peptide that might lead to a new treatment strategy for the complications of persistent hyperglycemia and other systemic metabolic disorders. We also considered the possibility of a novel approach to other aggregate and age-related disorders where proteostasis mechanisms become overwhelmed and simply wear out [43, 44].
cSKE7 was synthesized, cyclized, and purified by Creative Peptides. Purity was > 98% by LC/MS. Endotoxin levels were ≤ 0.01 EU/μg. The peptide was constructed by substituting the native lysine at the C-terminal end of the CHEC-7 peptide and then cyclizing the new peptide end-to-end giving cSKE7 (Figure 1). This strategy eliminated the nonnative disulfide bond, susceptible to reduction under various uncontrolled conditions. End-to-end cyclization was expected to provide a more robust and stable therapeutic [45]. In addition, we found limitations of the solubility of CHEC-7 in scaling up formulation efforts for potential human applications. While this problem may eventually be overcome as more sophisticated peptide delivery methods are developed, Savjani et al. (and many others more recently) [46–48] have noted that “Low aqueous solubility is the major problem encountered with formulation development of new chemical entities as well as generic development”. Substitution of the lysine for the second cysteine dramatically increases aqueous solubility, both theoretically and practically, mainly by increasing the polarity of the peptide (see Results).
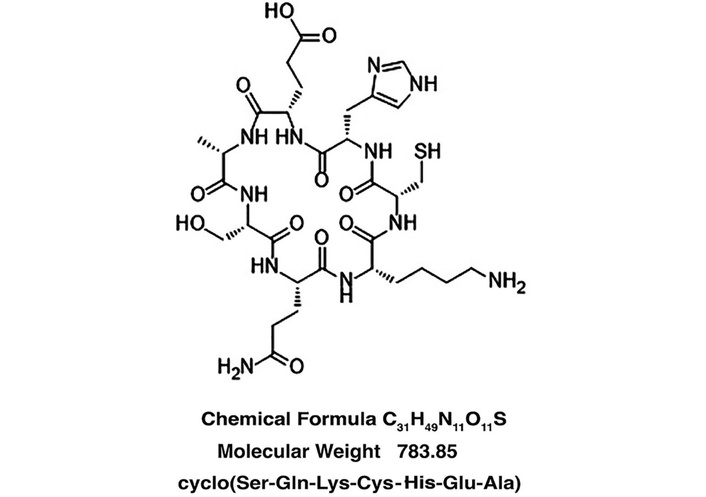
Composition of cSKE7. (Structure provided by Creative Peptides using ChemDraw software, version 25). cSKE7: cycloSQKCHEA.
Plasma from individuals of known age and both genders was prepared in acid-citrate-dextrose (ACD) anticoagulant by BioIVT and frozen within one hour of collection. BioIVT provides viral-negative normal human blood products collected from consented donors under IRB-approved protocols at facilities located in the United States and Europe. The plasma was from both genders and pooled according to age groups, with 3–4 individuals per group, as shown in Table 1. Pooled samples were treated with 0.05% NaN3 (Sigma Aldrich, S2002) prior to use to retard microbial growth.
Genders, ages of donors, and experimental use of plasma pools.
| Pool1 | Gender | Ages | Experiments2 |
|---|---|---|---|
| 70 yo (a) | 3 M | 71, 72, 75 | Thioflavin-T3 |
| Free thiols | |||
| GAPDH activity | |||
| 30 yo | 2 F, 1 M | 30, 36, 30 | Thioflavin-T3 |
| GAPDH activity | |||
| Mixed | 4 M | 25, 34, 25, 67 | Thioflavin-T |
| Free thiols | |||
| 70 yo (b) | 2 M, 1 F | 72, 72, 73 | Thioflavin-T |
| Fructosamine | |||
| HSP70 binding |
1: 70 yo = 70-year-old; 30 yo = 30-year-old; 2: Samples used in individual experiments and from different groups are noted in the figure legends and associated statistical tables; 3: Plasma smears from all pools were examined with fluorescent microscopy but only a subset of samples from 70 yo (a) and 30 yo pools was used for the quantitative thioflavin-T assay. M: male; F: female; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; HSP70: heat shock protein 70.
The purpose of these pools was not to systematically compare the response of plasma samples from different-aged or gendered individuals to MG treatment, as there was uneven availability of samples from the different pools. We aimed simply to pool samples collected from diverse groups of individuals to test the general applicability of the chosen hyperglycemic model for producing pathological changes in plasma and then test the effect of the cSKE7 peptide on this modified plasma chemistry.
In pilot experiments, plasma from each pool was treated with different concentrations of MG (MP Biomedicals, 02155558-CF) and compared daily with parallel samples without MG treatment using plasma smears and thioflavin-T (ThT) fluorescent staining to detect amyloidogenesis. All age groups showed significant amyloid accumulations without peptide treatment at 8–9 days when incubated with 1 mM MG. Plasma samples were handled as described previously [41] in order to account for variations in baseline chemistry that might result from ex vivo procedures [49–51]. Briefly, aliquots from individual pools were treated with MG, ATP levels were restored at 100 µM, and only then was the aliquot divided into different peptide or vehicle control groups for side-by-side assay analysis. In some cases, the plasma was also pretreated with HSP70 inhibitor pifithrin-µ [200 µM, Cayman Chemical, 10748, or dimethyl sulfoxide (DMSO) vehicle, Sigma Aldrich, D8418] for 1 hour at 37°C prior to group assignment. The specific targeting of this inhibitor to plasma HSP70 was confirmed in a previous study demonstrating identical effects on plasma HSP70 activity using an HSP70 antibody [41].
We applied the Chemistry of Health software, University of Cambridge (https://www-cohsoftware.ch.cam.ac.uk/index.php/camsolintrinsic), which assigns a solubility value for each residue taking into account its charge, polarity, and capability to interact with water molecules, as well as the properties of the surrounding amino acids (Pietro Somanni, University of Cambridge, personal communication). To test software predictions directly, we used different water/2-propanol mixtures (Fisher Scientific, 040983-K2) in a common aqueous two-phase system that allows determination of aqueous solubility following adjustments of solvent polarity [52].
Plasma samples were incubated at 37°C with or without 1 mM MG. The plasma was diluted to 10% in 4 mM phosphate buffer and reacted overnight at 4°C with nitrocellulose dots (Primorigen Biosciences, 2060-96) to which 4 µL of different concentrations of cSKE7 had been added and dried. The dots were blocked with blocking buffer and incubated overnight (4°C) with HSP70 Rabbit pAb (AbClonal, A12948). Secondary antibodies and all processing reagents were from Licor Biosciences, and the blots were viewed and captured on the Licor Odyssey M Imaging System with Image Studio 6.1 software.
Fructosamine, an AGE intermediate, was measured after incubation of plasma with or without MG for 8 days at 37°C. We applied the method of Johnson et al. [53] with minor modifications. This method clearly distinguishes diabetic patients from healthy controls and correlates well with fasting glucose. Briefly, 20 µL plasma samples were reacted with nitroblue tetrazolium (Gold Bio, NBT-1) in carbonate buffer (pH 10.1) at 37°C with 5 minutes of agitation. The samples were read after 20 additional minutes at 550 nm with the Tecan Infinite 200 PRO reader (Tecan Life Sciences) using i-control software (version 1.10). MG-exposed and non-MG-exposed samples were run side by side and compared (see Data analysis). Additional sets of cSKE7-treated samples were measured and compared to these initial baseline values to determine if peptide treatment interfered with fructosamine formation.
After 8 days of MG exposure, the samples of MG-treated plasma were centrifuged at 20,000 × g to sediment amyloid fibrils and other aggregates [54]. Supernatants were removed and stored at –80°C for later assays of free thiols and GAPDH enzyme activity. Fifteen microliters of 4 mM phosphate buffer (pH 7.5) were added to the tube and the precipitate (not always visible) was disrupted with a tissue homogenizer, subsequent vigorous scraping and vortexing, followed by the addition of 7.5 µL of 1 mM ThT (Cayman Chemical, 32553). After further vortexing, a 4 µL smear of the plasma sample was prepared on poly-L-lysine-coated gelatinized slides, examined, and photographed with a Leitz Orthoplan fluorescent microscope (Leica Microsystems). For the ThT solution assay, a subset of samples, as well as those pretreated with the HSP70 inhibitor, was further diluted 1:25 because of the different concentration requirements of the solution assay [55]. The samples were read with the Tecan reader at 450/510 nm (ex/em).
The measurements were conducted in 100 µL of 50 mM Tris (pH 8.2) to which was added 10 µL plasma, and finally 20 µL 10 mM DTNB (Gold Bio, D-250-50) in 0.4 M Tris (pH 8.2). The progress of the absorbance at 415 nm was measured using the kinetic software in the Tecan reader to determine the time to maximum absorbance and the values recorded.
We used a standard method as described for serum and plasma samples, measuring the conversion of NAD+ to NADH at 340 nm absorbance [56]. Ten microliters of plasma were mixed with 40 µL 3× sample buffer, and the reaction was initiated by the addition of 100 µL of a 1.5× mix of NAD+ (Cayman Chemical, 16077) and glyceraldehyde-3-phosphate (G3P, Cayman Chemical, 17865). Final concentrations of buffer reagents were 50 mM triethanolamine, EDTA 1 mM in 50 mM Tris (pH 8.9), and of reactants G3P 3 mM and NAD+ 1 mM. The maximum velocities reached during the initial 5 minutes of the reaction were recorded for each peptide and control pair and taken as a proportional estimate of the concentration of active enzyme in each sample. The procedure was repeated 3 times for each sample pair with systematic variation of the position of the samples on the plate. The average of the measurements was normalized and analyzed as described below.
Data sets from different plasma pools and different assay procedures were obtained after side-by-side processing in all experiments to avoid variations introduced by ex vivo handling of blood plasma (see above). The data points from each data set and for a particular assay session were normalized to the average vehicle/control value for that session and expressed as a percentage change from this average value according to a standard normalization formula.
This procedure reflects the variability of measurements around an average baseline control value. The resulting data were compared statistically by ANOVA with Dunnett’s test for multiple comparisons, except for GAPDH, for which measurements from individual side-by-side comparisons were analyzed via t-test because of session and age variability. Statistical analysis was via Prism software (10.6.1, GraphPad).
These experiments were intended to test the aqueous solubility of the cSKE7 peptide compared to the predecessor peptide CHEC-7. The Camsol software suggested a marked solubility difference between the redesigned peptide and CHEC-7. This was tested directly and confirmed by adjustments of solvent polarity with various aqueous mixtures of 2-propanol [52] (Figure 2).
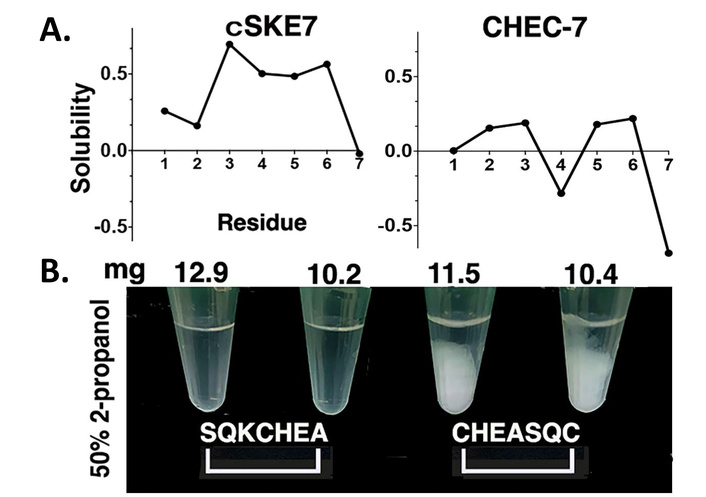
Peptide solubility. (A) Solubility predictions of cSKE7 and CHEC-7 peptides based on amino acid composition using the online software from Cambridge University (https://www-cohsoftware.ch.cam.ac.uk/index.php/camsolintrinsic). (B) The differences were tested directly with different aqueous mixtures of 2-propanol (shown are 4 examples of the solubility of mg quantities of peptide in 200 µL of 50% 2-propanol/water mixture). cSKE7: cycloSQKCHEA; CHEC-7: CHEASQC.
Plasma stressed for 8 days with MG had increased levels of HSP70 and/or increased HSP70 affinity for cSKE7 (or both; Figure 3). The results are consistent with previous reports of HSP70 affinity for peptide fragments near the N-terminus of the DSEP/Dermcidin protein [40].
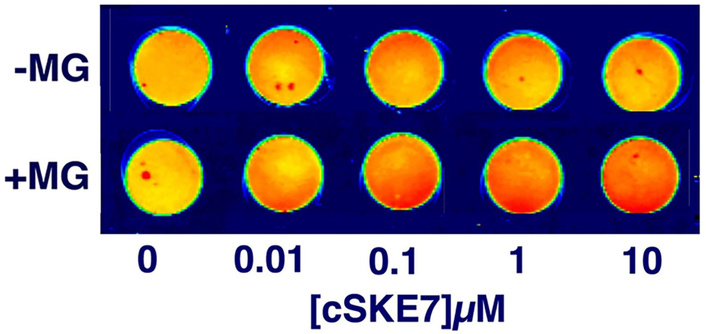
Plasma HSP70 binds with cSKE7. HSP70 immunostaining (red) with different concentrations of cSKE7 immobilized on nitrocellulose discs. The plasma was incubated with and without MG for 8 days at 37°C. Shown is a representative of two experiments with samples from each 70 yo pool. MG: methylglyoxal; cSKE7: cycloSQKCHEA; HSP70: heat shock protein 70; 70 yo: 70-year-old.
Fructosamine represents early-stage glycation by glucose and MG and is an intermediate in the formation of AGEs [57, 58]. We compared samples incubated with or without MG for 8 days and found that fructosamine formation was increased by over 50% (Figure 4; Table 2). Pretreatment of the samples with bioactive concentrations of cSKE7 was ineffective at reducing fructosamine levels, either following MG treatment (Figure 4) or that which occurs spontaneously without MG treatment (latter data not shown).
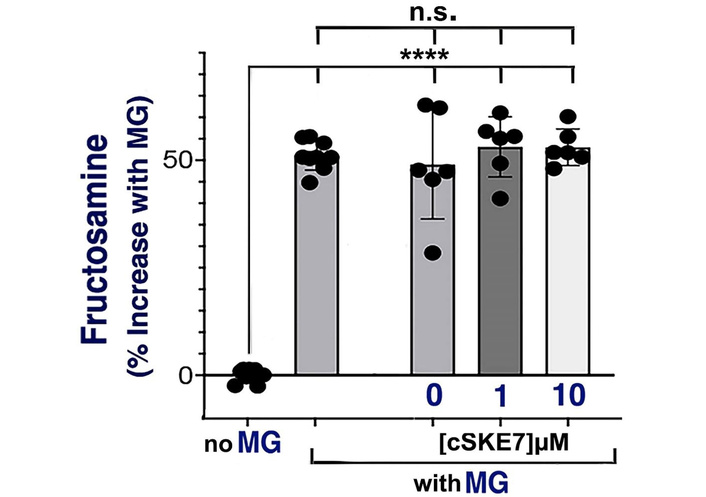
Fructosamine measurements. Increased fructosamine levels in plasma treated with methylglyoxal (MG). The plasma was treated with MG for 8 days at 37°C. Values were normalized to the average of control samples (no MG). MG increased fructosamine both with and without pretreatment with bioactive concentrations of cSKE7. Statistical calculations with corrections for multiple comparisons via Dunnett’s test are shown in Table 2. MG-treated samples, whether peptide-treated or not, were not statistically different. ****: p < 0.0001; n.s.: not significant; cSKE7: cycloSQKCHEA.
Statistics for fructosamine measurements.
| Comparisons | Samples | Mean diff. | 95.00% CI of diff. | Adjusted p-value |
|---|---|---|---|---|
| no MG vs. MG | 9 vs. 9 | –51.16 | –58.93 to –43.39 | < 0.0001 |
| no MG vs. 0 µM-MG | 9 vs. 6 | –49.03 | –57.71 to –40.34 | < 0.0001 |
| no MG vs. 1 µM-MG | 9 vs. 6 | –53.13 | –61.81 to –44.44 | < 0.0001 |
| no MG vs. 10 µM-MG | 9 vs. 6 | –53.02 | –61.70 to –44.33 | < 0.0001 |
MG: methylglyoxal; diff.: difference.
MG treatment induced a range of amyloid fibrillation and globulation, and consistent inhibition by cSKE7 addition at both 1 and 10 µM. Shown are ThT-stained smears of the resuspended precipitates of treated plasma and the typical appearance of fields from different plasma pools. The difference in amyloid staining between experimental groups and at both peptide concentrations was found upon inspection in all plasma groups. Large areas of fibers and globules were absent after peptide treatment. However, these accumulations were apparent once again following pretreatment with 200 µM pifithrin-µ [shown for the 70-year-old (70 yo) (a) pool] compared to vehicle, where the dissolution of amyloid accumulations was still observed (Figure 5A, far-right panels). Samples from the 30-year-old (30 yo) pool and the 70 yo (a) pools including after vehicle and 1 and 10 µM peptide treatment were selected for the ThT solution assay (Figure 5B; Table 3). Samples pretreated with the HSP70 inhibitor were not statistically different from vehicle controls without the inhibitor. The microplate fluorescent assay supported the staining observations and revealed a significant effect at 0.1 µM cSKE7, a result that was not consistently obvious upon examination of the plasma smears. The solution assay may be sensitive to prefibrillar amyloid forms, even though these have a less robust interaction with ThT [59], and disruption of these was realized at even the lowest concentration of the peptide tested in these experiments.
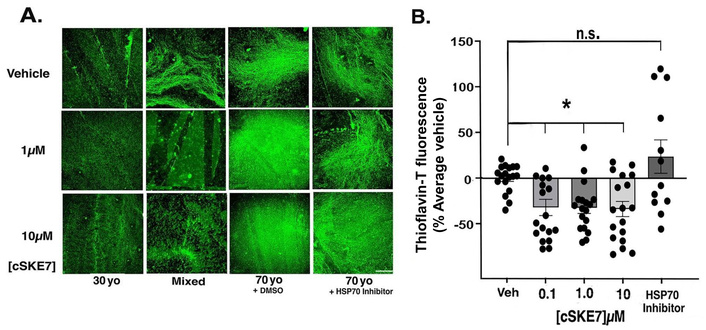
Thioflavin-T (ThT) fluorescence of plasma samples. (A) Images of smears from samples of the different plasma pools showing the range of amyloid accumulations after 8-day exposure to methylglyoxal (1 mM) and after subsequent treatment with vehicle, 1 or 10 µM cSKE7 for 24 hours. The micrographs on the right from the 70 yo pool were also treated with HSP70 inhibitor pifithrin-µ (200 µM) or DMSO vehicle for 1 hour prior to peptide treatment. Large areas of the smears were sampled for the micrographs (bar = 100 µm). (B) Samples from the 30 yo and 70 yo pools were processed for the ThT solution assay and normalized as described in the methods. The HSP inhibitor pifithrin-µ abrogated the loss of ThT fluorescence at all concentrations and in both pools. Values from companion samples pretreated with DMSO were not statistically different from initial samples. *: p < 0.05; n.s.: not significant (ANOVA with Dunnett’s correction for multiple comparisons, Table 3); cSKE7: cycloSQKCHEA; 30 yo: 30-year-old; 70 yo: 70-year-old; DMSO: dimethyl sulfoxide; HSP70: heat shock protein 70.
Statistics for thioflavin-T measurements.
| Comparisons | Samples | Mean diff. | 95.00% CI of diff. | Adjusted p-value |
|---|---|---|---|---|
| Veh vs. 0.1 µM | 9 vs. 9 | 33.08 | 1.64 to 64.51 | 0.0360 |
| Veh vs. 1 µM | 9 vs. 6 | 33.48 | 2.05 to 64.92 | 0.0331 |
| Veh vs. 10 µM | 9 vs. 6 | 34.88 | 3.44 to 66.31 | 0.0247 |
| Veh vs. inhib | 9 vs. 6 | –24.51 | –59.66 to 10.63 | 0.2530 |
Veh: vehicle; inhib: inhibitor; diff.: difference.
We investigated free thiol levels in MG-treated plasma because of the close relationship of thiol oxidation to the redox status in the circulation and the reported influence of HSP70 [60–63]. The measurements revealed a significant increase in free thiols (non-oxidized) with peptide treatment at both 1 and 10 µM cSKE7 (Figure 6; Table 4). The increase in free thiols was eliminated in a subset of samples from the 70 yo (a) pool pretreated with pifithrin-µ, suggesting the effect was HSP70 dependent.
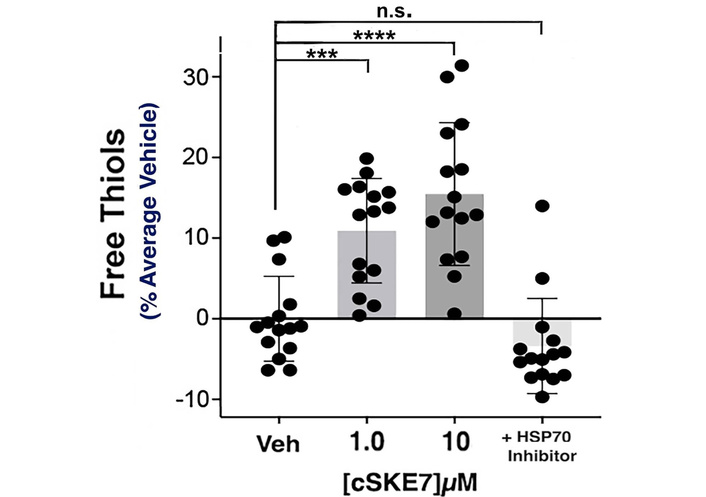
Free thiol measurement. Thiols were detected with 5,5'-dithio-bis(2-nitrobenzoic acid) (DTNB) and normalized to the average of vehicle-treated samples. Significant elevations (fewer oxidized thiols) were detected at 1 and 10 µM peptide concentrations, representing combined samples from the 70 yo (a) and mixed plasma pools. Additional samples from the 70 yo (a) pool were pretreated with pifithrin-µ or DMSO. The HSP70 inhibitor eliminated the increase. Values from companion samples pretreated with DMSO were not statistically different from initial samples. ***: p = 0.0001; ****: p < 0.0001; n.s.: not significant (ANOVA with Dunnett’s correction for multiple comparisons, Table 4); cSKE7: cycloSQKCHEA; HSP70: heat shock protein 70; 70 yo: 70-year-old; DMSO: dimethyl sulfoxide.
Statistics for free thiol measurements.
| Comparisons | Samples | Mean diff. | 95.00% CI of diff. | Adjusted p-value |
|---|---|---|---|---|
| Veh vs. 1 µM | 15 vs. 15 | –10.91 | –16.87 to –4.95 | 0.0001 |
| Veh vs. 10 µM | 15 vs. 15 | –15.46 | –21.42 to –9.50 | < 0.0001 |
| Veh vs. inhib | 15 vs. 15 | 3.38 | –2.58 to 9.34 | 0.3862 |
Veh: vehicle; inhib: inhibitor; diff.: difference.
GAPDH enzyme activity was examined because of its sensitivity to both MG levels and redox status [64, 65], and therefore, the possible relationship of enzyme activity to plasma HSP70 activity (see Discussion). These measurements were the most variable and required not only that we run individual cSKE7-treated samples side by side with individual vehicle-treated samples, but also that these samples be paired for statistical analysis. In this case also, we analyzed pairs of samples from the 70 yo (a) pool separately from those of the 30 yo pool because of near significant differences in baseline (vehicle-treated) responses to MG treatment between the two pools after 8 days (30 yo pool, 1,140.03 ± 46.7 vs. 70 yo pool, 880.44 ± 116.7 mAU/min/mL at 340 nm, p = 0.073). No firm conclusion was possible with this very limited sample, but the result may be consistent with the decline in GAPDH activity reported in aging mammalian systems [66–68]. Despite the variability in these measurements, both pools showed significant elevations in GAPDH activity with cSKE7 treatment after pairing and combining data from 1 and 10 µM peptide treatments (Figure 7A and B). Inhibition of HSP70 activity had a marked effect on GAPDH activity in MG-treated samples, whether peptide-treated or not. This is shown for samples from the 30 yo pool, where peptide and vehicle samples were also pretreated with pifithrin-µ or DMSO (Figure 7C; Table 5). This result would be expected if plasma GAPDH activity is supported generally (i.e., without peptide treatment) by plasma HSP70 under conditions of hyperglycemic stress.
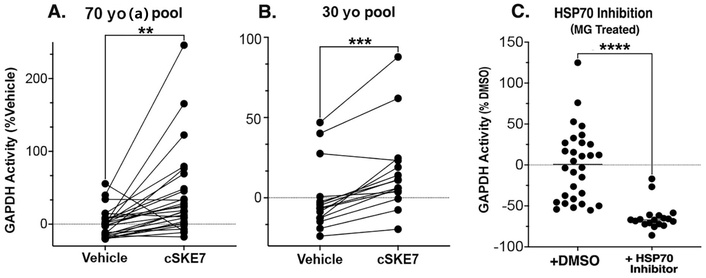
GAPDH measurement. (A–B) Paired samples from the 70 yo (a) and 30 yo pools after cSKE7 treatments of 1 and 10 µM, normalized to the average of vehicle-treated samples. (C) Additional MG-treated samples from the 30 yo pool were pretreated with pifithrin-µ or DMSO. The inhibitor eliminates both the peptide-mediated rescue effect of activity as well as endogenous surviving GAPDH activity. A “floor effect”, encountered with the present assay (too many values at or near the detection limit), precluded a similar analysis of inhibitor effects in the 70 yo (a) pool. **: p < 0.01; ***: p = 0.0001; ****: p < 0.0001. Significance levels via paired t-test for A&B and unpaired test for C with Welch’s correction for different variances (Table 5). 70 yo: 70-year-old; cSKE7: cycloSQKCHEA; 30 yo: 30-year-old; HSP70: heat shock protein 70; MG: methylglyoxal; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; DMSO: dimethyl sulfoxide.
Statistics for GAPDH measurement.
| Comparisons | Samples | Mean diff. | 95.00% CI of diff. | p-Value | p-Value (pairing)1 |
|---|---|---|---|---|---|
| Peptide vs. vehicle—30 yo | 15 vs. 15 | 16.37 | 9.69 to 23.05 | 0.0001 | < 0.0001 |
| Peptide vs. vehicle—70 yo (a) | 27 vs. 27 | 38.59 | 16.97 to 60.21 | 0.0011 | 0.0262 |
| DMSO vs. inhib2 | 18 vs. 18 | –57.10 | –77.49 to –36.72 | < 0.0001 | NA3 |
1: Significance levels for effective pairing; 2: significant difference of variances (p = 0.0099); 3: NA = not applicable; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; 30 yo: 30-year-old; 70 yo: 70-year-old; DMSO: dimethyl sulfoxide; inhib: inhibitor; diff.: difference.
The goal of the present study was to model the well-documented cumulative effect of elevated MG levels on plasma protein amyloidogenesis and oxidative stress (and related metabolic disease states) and to test the redesigned peptide, cSKE7, for its reparative properties under these conditions. Elevated levels of fructosamine after MG treatment are consistent with an accelerated process of non-enzymatic protein glycation and the resulting toxic changes documented in this study. MG is a highly reactive dicarbonyl compound, making it a much more potent glycating agent than glucose and therefore more efficient in the eventual production of toxic AGEs [23, 24]. “Fructation” [58] is an early stage of this process targeting not only proteins but also free amino acids, lipids, and nucleic acids. The lack of a measurable influence of peptide treatment on this process suggested that the peptide’s effects were realized after these modifications and directed to activities associated specifically with aspects of HSP70 activity like disaggregation and lowering of stress levels. With respect to the former, it is important to emphasize that we provided evidence only for amyloid dissolution and not protein repair.
Our selection of a 1 mM MG concentration and of an 8-day incubation was based on the consistent and marked appearance of increased amyloid globular and fibrous aggregates without peptide treatment. This is also the level measured in the plasma of mice fed MG to model metabolic and vascular dysfunction [69]. Studies of MG effects on amyloidogenesis of identified plasma proteins generally apply higher concentrations [70–73], while sporadic measures of MG are lower in diabetic patients but significantly higher than in healthy subjects [74, 75]. MG, fructosamine, and fasting glucose levels are all measures of glycemic control. Importantly, persistent elevations of any of these are associated with heightened risk for diabetic complications [76–79]. Therefore, we suggest that the present model is sufficiently robust to demonstrate cSKE7 influence on hyperglycemic-instigated amyloidogenesis as well as the involvement of HSP70 in disrupting this process.
The use of human plasma for these experiments may also be important since, as noted above, the parent DSEP/Dermcidin is restricted to humans and primates, i.e., an “orphan” protein, not present in other mammals [35]. It was also assumed that the human versions of accessory molecules or co-chaperones required for circulating HSP70 activity are also present in the plasma samples [80, 81]. These considerations make the choice of the appropriate animal model uncertain at this stage of therapeutic development. However, the reliance on ex vivo plasma in the absence of blood cells and whole-body influences also introduced obvious limitations that prevent conclusions about eventual clinical efficacy. This study has therefore been aptly considered “proof of concept”. Nonetheless, as noted throughout this paper, the ex vivo modifications of plasma proteins are well-documented under hyperglycemic and other toxic stressors, as are the in vivo and clinical counterparts and correlated disease states.
The loss of free thiols in blood plasma, although not a specific biomarker per se, nevertheless indicates pro-oxidant changes that accompany or contribute to several systemic disorders [60–62]. Intracellular HSP70 is an antioxidant and promotes thiol reduction [63]. The simple explanation for the increase in free thiols after peptide treatment, therefore, was the reduction of oxidant conditions due to the action of HSP70 as a modulator of the thiol-disulfide system. This has been demonstrated in cells where there is a direct effect of HSP70 on the glutathione (GSH) redox balance and suggested by several studies in patient serum, where HSP70 levels are negatively correlated with stress enzymes in metabolic disorders [29]. Altered levels of thiols in plasma have been linked with specific pathological conditions, including increased risk for cardiovascular and cerebrovascular diseases. In addition, the pro-oxidant effects of MG in the hyperglycemic condition are also strongly linked to the progression of age-related and chronic metabolic diseases [21, 57, 82, 83].
GAPDH is an ancient and multifunctional protein with properties that go well beyond intracellular glycolysis [84]. Plasma and serum GAPDH are less well understood, but the circulating enzyme is described as an enzymatically active multimer consisting of several high-molecular-weight subunits besides the ~37 kDa monomer [56]. In the present study, GAPDH activity was examined because of its role in redox balance and the relationship between MG levels and inhibition of the enzyme [64, 65, 71, 85]. It is unclear how the increased GAPDH activity occurred in plasma after cSKE7 treatment, as well as the demonstrated relationship to HSP70 activity, but several possibilities are evident. First, HSP70 might simply reduce or dissociate GAPDH aggregates as one of its target proteins, such that there is an increase in the relative concentration of one of the active multimeric forms. Our previous data showing a GAPDH gel shift after treatment of CNS cytosol with a cSKE7 predecessor peptide, CHEC-9, which also influences HSP70 activity, is consistent with this suggestion [36]. In fact, the pro-oxidant conditions present in the current model can induce GAPDH to form inactive amyloid-like structures, an effect attributed to oxidation of the enzyme’s active site cystine under such conditions [86, 87]. For example, oxidative stress produced by hyperglycemic MG levels occurs via a decrease in the ratio of reduced GSH to oxidized GSH (GSSG), which can further induce the reversible S-glutathionylation of GAPDH, thus inactivating the enzyme [88]. Finally, intracellular GAPDH’s interaction with multiple protein partners may allow cells to utilize the enzyme as a cleaning agent that sequesters and flushes out misfolded molecules arising because of cellular stress, and, in a hypoxic cellular environment, HSP70 targets and disaggregates GAPDH to assist in this activity [89, 90]. As noted, however, most of the reports reviewed above relate to the activity of intracellular GAPDH, so it remains to be determined if similar processes occur in the circulation. Nonetheless, the human pathologies associated with the oxidant-induced dysregulation of GAPDH highlight the importance of the redox regulation of the enzyme, especially in metabolic and neurodegenerative disorders [65], and there are several possible mechanisms by which cSKE7 and HSP70 can intervene to elevate enzyme activity.
After their discovery by Ferruccio Ritossa in the early 1960s, appreciation of the importance of HSPs to the many aspects of biological repair was slow [91]. Even today, therapeutic strategies for targeting HSP70 activity appear to be at cross-purposes: Inhibition for neoplastic disease if the focus is on its cell survival and anti-tumor suppression, but enhancement for inflammatory and protein aggregate disorders where the emphasis is on targeting systemic pathologies and unwanted cell death [81, 92–94]. However, even in the case of putative cancer therapies, the same duality or “double-edged sword” exists, especially for the diverse immunological functions of HSP70 [95, 96]. For example, the release of HSP70-containing vesicles from cancer cells can trigger a “full-scale” anti-cancer immune response [97]. In fact, for p53 mutations, cancer has also been considered an aggregate disease [98]. Furthermore, Ambrose and Chapman [92] suggest the importance of different HSP70 isoforms in therapeutic targeting, a sentiment echoed by Singh et al. [99] in their review of HSP70 functions.
Therefore, until we better know the isoform identities and properties of circulating HSP70s, it is just as reasonable to suggest that they are distinct, or more likely repurposed intracellular HSP70s, perhaps housed in extracellular microvesicles and now in a systemic environment [100–102]. Here, they can be released and recruited to combat systemic oxidant stressors and amyloidogenesis of blood proteins. In support of this suggestion are the reports of specific effects of systemically administered recombinant human HSP70 on protection against muscle denervation, including in the SOD mouse model of amyotrophic lateral sclerosis [103, 104]. Similar results have been reported for HSP70 administration in models of CNS vascular injury [105, 106] as well as for clinical manifestations of sepsis in an LPS-treated rat model. The latter studies may be related to previous work from our laboratory where the cSKE7 predecessor CHEC-7, applied orally to LPS-injected rats or to an autoimmune model (experimental autoimmune encephalomyelitis), reduced inflammation and promoted recovery of motor function [38, 107]. However, one difficulty with direct application of HSP70 in clinical models, a problem that is likely to reappear in human applications, may be the limited bioavailability demonstrated in pharmacokinetic studies of the protein [108]. Several investigators, therefore, have opted for indirect stimulation of HSP70 via stressors such as hyperthermia, or alternatively, have intensified the search for stimulants of HSP70 activity [80, 94, 109]. The CHEC peptides, including the current modification cSKE7, are among the latter category, and so far, the only compounds with demonstrated HSP70-mediated activities in human blood plasma, including for cSKE7 in the current model of hyperglycemic stress. Present results from this model may be significant because promising clinical outcomes in diabetic monkeys and humans have been obtained with indirect stimulation of HSP70 via hyperthermia [8, 110]. The possibility of cSKE7 peptide therapy may also be relevant to disorders involving serum amyloidosis, especially when anti-inflammatory treatments are not effective [111, 112].
The link between glycative stress and accelerated aging has been suggested for decades, as has the role of HSP70 and an intact system of proteostasis as an important factor in establishing this relationship [44, 113, 114]. For example, the contribution of HSP70 activity to extended lifespan was advanced by studies in Drosophila and C. elegans, providing the conceptual framework for similar proposals in mammals, including humans [115–118]. As already noted, the protein handling functions in cells and in the blood get overburdened, regardless of glycative status. Hyperglycemia and accompanying inflammation and oxidative stress contribute significantly to this burden [43, 44]. Future studies may help determine if cSKE7 will provide some relief for the accumulated aggregate toxicity in aging paradigms, whether under metabolically compromised or other disease conditions, or simply as the expected age-related decline in the detoxification functions of proteostasis. The work of Njemini et al. [4] suggests clearly that circulating HSP70 declines with advancing patient age, only to increase with the heightened systemic stress of frail health. While the eventual relationship of the current results to aging pathology is speculative, it still may be important to determine if there is a critical period in the aging process when the capacity to combat aggregate disease and concomitant stressors will respond to therapeutic intervention.
30 yo: 30-year-old
70 yo: 70-year-old
AGEs: advanced glycation end products
CHEC-7: CHEASQC
CHEC-9: CHEASAAQC
cSKE7: cycloSQKCHEA
DMSO: dimethyl sulfoxide
DTNB: 5,5'-dithio-bis(2-nitrobenzoic acid)
G3P: glyceraldehyde-3-phosphate
GAPDH: glyceraldehyde-3-phosphate dehydrogenase
GSH: glutathione
HSP70: heat shock protein 70
MG: methylglyoxal
ThT: thioflavin-T
The author gratefully acknowledges the support of the Marion Murray Spinal Cord Research Center at the Department of Neurobiology and Anatomy, Drexel University College of Medicine, and Center manager Theresa Connors for expert technical assistance.
TJC: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Validation, Visualization, Writing—original draft, Writing—review & editing. The author read and approved the submitted version.
Drexel University and the author have applied for patent protection of the peptide compound that is the subject of this paper. Partial financial support was provided by CHEC-PR LLC, a company established to develop peptide therapies. The company had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
BioIVT provides viral-negative normal human blood products collected from consented donors under Seratrials LLC IRB protocol #2010-017 approved by WCG (WIRB-Copernicus Group), expiring 9/07/2026.
Informed consent to participate in the study was obtained from all participants.
Not applicable.
All data sets and limited samples of cSKE7 peptide for independent testing are available at no cost to qualified research laboratories. Please contact Timothy Cunningham at tjc29@drexel.edu.
Partial financial support was provided by the Department of Neurobiology & Anatomy, Drexel University College of Medicine, and by CHEC-PR LLC, a company established to develop peptide therapies. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1278
Download: 14
Times Cited: 0
