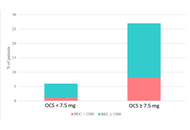

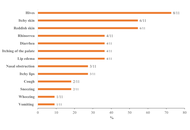



Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare systemic vasculitis with heterogeneous clinical manifestations. Identifying reliable biomarkers is crucial to predicting disease evolution and guiding therapy. We analyzed clinical, biological, and functional data from 33 patients with EGPA in the vasculitic phase. Blood eosinophil count (BEC), eosinophilic cationic protein (ECP), antineutrophil cytoplasmic antibodies (ANCAs), specific IgE to staphylococcal enterotoxins (SE-IgE), and serum free light chains (FLCs) were evaluated. Severe eosinophilic asthma (SEA) and chronic rhinosinusitis with nasal polyps (CRSwNPs) were present in 100% and 88.5% of patients, respectively. Median BEC was 1950 cells/mm3, with elevated ECP (60.0 µg/L). SE-IgE was detected in 54.2% of patients. A significant negative correlation emerged between λ FLCs and oral corticosteroid (OCS) dose (r = –0.58, p = 0.009). Forced expiratory volume in 1 second (FEV1) was significantly lower in C-ANCA+ patients (p = 0.006). ECP and SE-IgE may serve as markers of eosinophilic activity and epithelial barrier damage in EGPA. λ FLCs might be a useful indicator of OCS exposure and treatment response. These biomarkers could support the evaluation of disease evolution and treatment response.
Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare systemic vasculitis with heterogeneous clinical manifestations. Identifying reliable biomarkers is crucial to predicting disease evolution and guiding therapy. We analyzed clinical, biological, and functional data from 33 patients with EGPA in the vasculitic phase. Blood eosinophil count (BEC), eosinophilic cationic protein (ECP), antineutrophil cytoplasmic antibodies (ANCAs), specific IgE to staphylococcal enterotoxins (SE-IgE), and serum free light chains (FLCs) were evaluated. Severe eosinophilic asthma (SEA) and chronic rhinosinusitis with nasal polyps (CRSwNPs) were present in 100% and 88.5% of patients, respectively. Median BEC was 1950 cells/mm3, with elevated ECP (60.0 µg/L). SE-IgE was detected in 54.2% of patients. A significant negative correlation emerged between λ FLCs and oral corticosteroid (OCS) dose (r = –0.58, p = 0.009). Forced expiratory volume in 1 second (FEV1) was significantly lower in C-ANCA+ patients (p = 0.006). ECP and SE-IgE may serve as markers of eosinophilic activity and epithelial barrier damage in EGPA. λ FLCs might be a useful indicator of OCS exposure and treatment response. These biomarkers could support the evaluation of disease evolution and treatment response.
DOI: https://doi.org/10.37349/eaa.2026.1009127
This article belongs to the special issue Environment, Infectious Diseases, and Allergy

Aim:
This study aimed to assess the prevalence and patterns of aeroallergen sensitisation in steroid-naïve, newly diagnosed adult patients with bronchial asthma using skin prick test (SPT), and to examine its association with lung function parameters measured by oscillometry and spirometry.
Methods:
Consecutive adult patients with bronchial asthma who were naïve to systemic and inhaled corticosteroids were recruited. Following a detailed clinical history and blood investigations, lung function was assessed using oscillometry and spirometry. SPT was performed using a panel of 13 aeroallergens in accordance with established guidelines. Based on SPT results, patients were stratified into two groups: atopic asthma (sensitisation to ≥ 1 allergen) and non-atopic asthma (no sensitisation). Demographic characteristics, blood parameters, and lung function parameters were compared between the two groups.
Results:
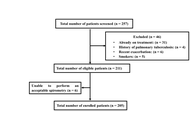
Of 257 patients screened, 205 were enrolled (59% men; mean age 36.9 years). Allergic rhinitis was present in 58% of patients, and 69.8% had atopic asthma (95% CI 62.9–75.6). Sensitisation was most common to house dust mites (47.8%), followed by cockroach (37.6%) and weed pollen (29.8%). Atopy was more prevalent in men than in women and was associated with higher serum total IgE levels. Peripheral blood eosinophil counts, oscillometric parameters, severity of airflow obstruction, and bronchodilator responses did not differ significantly between atopic and non-atopic patients.
Conclusions:
The majority of asthma patients exhibited sensitisation to one or more aeroallergens. Despite differences in immunopathogenesis, no significant differences in oscillometric parameters were observed between atopic and non-atopic asthma, suggesting that atopic status has a limited influence on the severity of small airway dysfunction.
Aim:
This study aimed to assess the prevalence and patterns of aeroallergen sensitisation in steroid-naïve, newly diagnosed adult patients with bronchial asthma using skin prick test (SPT), and to examine its association with lung function parameters measured by oscillometry and spirometry.
Methods:
Consecutive adult patients with bronchial asthma who were naïve to systemic and inhaled corticosteroids were recruited. Following a detailed clinical history and blood investigations, lung function was assessed using oscillometry and spirometry. SPT was performed using a panel of 13 aeroallergens in accordance with established guidelines. Based on SPT results, patients were stratified into two groups: atopic asthma (sensitisation to ≥ 1 allergen) and non-atopic asthma (no sensitisation). Demographic characteristics, blood parameters, and lung function parameters were compared between the two groups.
Results:
Of 257 patients screened, 205 were enrolled (59% men; mean age 36.9 years). Allergic rhinitis was present in 58% of patients, and 69.8% had atopic asthma (95% CI 62.9–75.6). Sensitisation was most common to house dust mites (47.8%), followed by cockroach (37.6%) and weed pollen (29.8%). Atopy was more prevalent in men than in women and was associated with higher serum total IgE levels. Peripheral blood eosinophil counts, oscillometric parameters, severity of airflow obstruction, and bronchodilator responses did not differ significantly between atopic and non-atopic patients.
Conclusions:
The majority of asthma patients exhibited sensitisation to one or more aeroallergens. Despite differences in immunopathogenesis, no significant differences in oscillometric parameters were observed between atopic and non-atopic asthma, suggesting that atopic status has a limited influence on the severity of small airway dysfunction.
DOI: https://doi.org/10.37349/eaa.2026.1009126

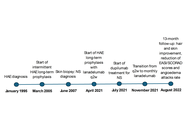
Netherton syndrome (NS) is a rare autosomal recessive disorder caused by SPINK5 mutations, leading to impaired skin barrier function and severe atopic manifestations. Hereditary angioedema due to C1 inhibitor deficiency (HAE-C1-INH) is a rare autosomal dominant disorder characterised by recurrent bradykinin-mediated angioedema. Their coexistence has not previously been reported, and evidence on combined biologic therapy is lacking. We report a 30-year-old woman with confirmed NS and long-standing HAE-C1-INH presenting with severe pruritus, xerosis, widespread eczema, elevated IgE, eosinophilia, and trichorrhexis invaginata. Dupilumab was initiated to target T helper (Th)2-mediated inflammation. Due to persistent angioedema attacks despite prior prophylaxis, lanadelumab was introduced. Dupilumab improved eczema severity, hyperkeratosis, and hair abnormalities over 13 months. Lanadelumab reduced angioedema attacks by 88.50%, allowing dose spacing while maintaining disease control. No adverse effects or drug interactions were observed. This is the first reported case of NS and HAE-C1-INH successfully treated with dual biologic therapy. Targeting distinct immunological pathways simultaneously may represent an effective and safe strategy for complex rare disease phenotypes.
Netherton syndrome (NS) is a rare autosomal recessive disorder caused by SPINK5 mutations, leading to impaired skin barrier function and severe atopic manifestations. Hereditary angioedema due to C1 inhibitor deficiency (HAE-C1-INH) is a rare autosomal dominant disorder characterised by recurrent bradykinin-mediated angioedema. Their coexistence has not previously been reported, and evidence on combined biologic therapy is lacking. We report a 30-year-old woman with confirmed NS and long-standing HAE-C1-INH presenting with severe pruritus, xerosis, widespread eczema, elevated IgE, eosinophilia, and trichorrhexis invaginata. Dupilumab was initiated to target T helper (Th)2-mediated inflammation. Due to persistent angioedema attacks despite prior prophylaxis, lanadelumab was introduced. Dupilumab improved eczema severity, hyperkeratosis, and hair abnormalities over 13 months. Lanadelumab reduced angioedema attacks by 88.50%, allowing dose spacing while maintaining disease control. No adverse effects or drug interactions were observed. This is the first reported case of NS and HAE-C1-INH successfully treated with dual biologic therapy. Targeting distinct immunological pathways simultaneously may represent an effective and safe strategy for complex rare disease phenotypes.
DOI: https://doi.org/10.37349/eaa.2026.1009125

Obesity is a determinant of the risk of developing various diseases, including asthma. It can also contribute to asthma severity. Obvious determinants of the risk of developing obesity and conditions associated with obesity are the diet an individual consumes and their energy expenditure, as determined by activity. Therefore, diet and exercise are important non-pharmacological components in the management and prevention of many diseases. Several individual elements in diet, including certain fatty acids and vitamins, as well as types of diets, notably the Mediterranean diet, have been studied in asthmatic patients, but the literature is not consistent. This review explores the relationship between asthma and obesity, exercise, and multiple dietary components and regimens, including the Mediterranean diet; polyunsaturated fats; vitamins A, C, D, and E; flavonoids; probiotics; and sodium intake in the published randomized clinical trials. Overall, the data have many shortcomings, but there is no single component of diet that is consistently associated with improved asthma outcomes, nor any component found to be clearly harmful. However, a diet that helps an individual lose weight may indirectly improve their lung function and asthma control, even if the diet itself does not impact asthma outcomes. Exercise, now known to be safe and widely recommended in asthma, has various forms. This review looked at meta-analyses, as well as recently published data addressing this question, categorizing exercise as aerobic activity, pulmonary rehabilitation, and yoga. The most evidence for benefit is for aerobic exercise, but yoga also has potential for modest improvement in asthma symptoms. There is conflicting data as to whether supervised exercise programs are superior to unsupervised physical activity. Overall, exercise is helpful in asthma, but it is still unclear how much exercise should be done, and this should be tailored to each individual.
Obesity is a determinant of the risk of developing various diseases, including asthma. It can also contribute to asthma severity. Obvious determinants of the risk of developing obesity and conditions associated with obesity are the diet an individual consumes and their energy expenditure, as determined by activity. Therefore, diet and exercise are important non-pharmacological components in the management and prevention of many diseases. Several individual elements in diet, including certain fatty acids and vitamins, as well as types of diets, notably the Mediterranean diet, have been studied in asthmatic patients, but the literature is not consistent. This review explores the relationship between asthma and obesity, exercise, and multiple dietary components and regimens, including the Mediterranean diet; polyunsaturated fats; vitamins A, C, D, and E; flavonoids; probiotics; and sodium intake in the published randomized clinical trials. Overall, the data have many shortcomings, but there is no single component of diet that is consistently associated with improved asthma outcomes, nor any component found to be clearly harmful. However, a diet that helps an individual lose weight may indirectly improve their lung function and asthma control, even if the diet itself does not impact asthma outcomes. Exercise, now known to be safe and widely recommended in asthma, has various forms. This review looked at meta-analyses, as well as recently published data addressing this question, categorizing exercise as aerobic activity, pulmonary rehabilitation, and yoga. The most evidence for benefit is for aerobic exercise, but yoga also has potential for modest improvement in asthma symptoms. There is conflicting data as to whether supervised exercise programs are superior to unsupervised physical activity. Overall, exercise is helpful in asthma, but it is still unclear how much exercise should be done, and this should be tailored to each individual.
DOI: https://doi.org/10.37349/eaa.2026.1009124
This article belongs to the special issue The Complex Interactions Between Lifestyles and Asthma

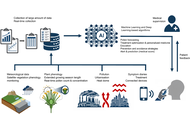
Pollen-related allergic diseases, including allergic rhinoconjunctivitis and asthma, affect a growing proportion of the population and have substantial consequences for quality of life and healthcare systems. Conventional pollen forecasts, which rely on fixed pollen traps and meteorological data, are limited in spatial granularity, real-time responsiveness, and individual relevance. Recent advances in artificial intelligence (AI) and machine learning (ML) offer a paradigm shift in the modelling of pollen release, forecast exposure, and alert allergic patients. This article provides a comprehensive overview of ML-based pollen forecasting systems, clarifying their underlying principles in accessible terms for clinicians and presenting practical and published tools that allergologists can integrate into routine practice. By combining aerobiological data, meteorological models, and patient-reported outcomes, ML enables more personalized, precise, and timely allergy management. We review the fundamental mechanisms of pollen release and dispersion and illustrate how ML models can improve predictive accuracy. Key platforms are compared in terms of clinical usability. We present real-world use cases showing how ML-driven alerts can help optimize treatment plans and support patient education. Practical insights are provided on the evaluation, implementation, and limitations of these tools. ML is not a distant technology—it is already transforming pollen forecasting and allergy alerts. This article aims to equip allergologists with the knowledge needed to evaluate and adopt these tools, enabling a more proactive and personalized approach to managing pollen allergies.
Pollen-related allergic diseases, including allergic rhinoconjunctivitis and asthma, affect a growing proportion of the population and have substantial consequences for quality of life and healthcare systems. Conventional pollen forecasts, which rely on fixed pollen traps and meteorological data, are limited in spatial granularity, real-time responsiveness, and individual relevance. Recent advances in artificial intelligence (AI) and machine learning (ML) offer a paradigm shift in the modelling of pollen release, forecast exposure, and alert allergic patients. This article provides a comprehensive overview of ML-based pollen forecasting systems, clarifying their underlying principles in accessible terms for clinicians and presenting practical and published tools that allergologists can integrate into routine practice. By combining aerobiological data, meteorological models, and patient-reported outcomes, ML enables more personalized, precise, and timely allergy management. We review the fundamental mechanisms of pollen release and dispersion and illustrate how ML models can improve predictive accuracy. Key platforms are compared in terms of clinical usability. We present real-world use cases showing how ML-driven alerts can help optimize treatment plans and support patient education. Practical insights are provided on the evaluation, implementation, and limitations of these tools. ML is not a distant technology—it is already transforming pollen forecasting and allergy alerts. This article aims to equip allergologists with the knowledge needed to evaluate and adopt these tools, enabling a more proactive and personalized approach to managing pollen allergies.
DOI: https://doi.org/10.37349/eaa.2026.1009123
This article belongs to the special issue Climate Change, Allergy, and Immunotherapy

Aim:
Dysfunctional breathing (DB) is a common comorbidity in asthma, yet its objective characterisation remains challenging due to reliance on subjective questionnaires. This study aimed to determine whether quantifiable breathing pattern parameters can be used to characterise asthma patients with perceived DB from those without DB.
Methods:
This observational cross-sectional study involved 122 adults with physician-diagnosed asthma (GINA Steps 2–5). Participants completed the Nijmegen Questionnaire (NQ) to determine DB (NQ > 23) or non-DB (NDB). Resting breathing was recorded for 5 minutes using structured light plethysmography (SLP), a contactless optical motion-analysis system that quantifies thoracoabdominal displacement. Extracted parameters included respiratory rate (RR), inspiration time (Ti), expiration time (Te), Ti/total breath cycle duration (Ttot), and the ratio of ribcage to abdominal displacement during the inspiration phase (RCampinsp/ABampinsp). Both absolute values and within-subject variability [coefficient of variation expressed in percentage (CoV%)] were estimated. Between-group comparisons used Mann-Whitney U tests. Two binary logistic regression models evaluated the predictive value of mean parameters and their variability for characterising DB.
Results:
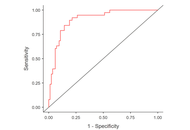
Of the 122 participants, 38 asthmatic patients were determined with DB. The DB group showed significantly higher RR and lower Ti and Te, but no differences in Ti/Ttot or RCampinsp/ABampinsp. In contrast, within-subject variability across all parameters was significantly greater in the DB group. The regression model using absolute values showed limited predictive power (R2 = 0.251). The model incorporating variability demonstrated substantially improved predictive power (R2 = 0.540), with CoV% RR, Te, Ti/Ttot, and RCampinsp/ABampinsp emerging as significant predictors.
Conclusions:
Asthma patients with DB exhibit breathing-pattern alterations, most notably increased within-subject variability of timing and TA movement parameters. Breathing pattern variability may be a promising surrogate marker to characterise DB, highlighting the value of dynamic objective physiological assessment of breathing beyond conventional symptom-based assessments of DB.
Aim:
Dysfunctional breathing (DB) is a common comorbidity in asthma, yet its objective characterisation remains challenging due to reliance on subjective questionnaires. This study aimed to determine whether quantifiable breathing pattern parameters can be used to characterise asthma patients with perceived DB from those without DB.
Methods:
This observational cross-sectional study involved 122 adults with physician-diagnosed asthma (GINA Steps 2–5). Participants completed the Nijmegen Questionnaire (NQ) to determine DB (NQ > 23) or non-DB (NDB). Resting breathing was recorded for 5 minutes using structured light plethysmography (SLP), a contactless optical motion-analysis system that quantifies thoracoabdominal displacement. Extracted parameters included respiratory rate (RR), inspiration time (Ti), expiration time (Te), Ti/total breath cycle duration (Ttot), and the ratio of ribcage to abdominal displacement during the inspiration phase (RCampinsp/ABampinsp). Both absolute values and within-subject variability [coefficient of variation expressed in percentage (CoV%)] were estimated. Between-group comparisons used Mann-Whitney U tests. Two binary logistic regression models evaluated the predictive value of mean parameters and their variability for characterising DB.
Results:
Of the 122 participants, 38 asthmatic patients were determined with DB. The DB group showed significantly higher RR and lower Ti and Te, but no differences in Ti/Ttot or RCampinsp/ABampinsp. In contrast, within-subject variability across all parameters was significantly greater in the DB group. The regression model using absolute values showed limited predictive power (R2 = 0.251). The model incorporating variability demonstrated substantially improved predictive power (R2 = 0.540), with CoV% RR, Te, Ti/Ttot, and RCampinsp/ABampinsp emerging as significant predictors.
Conclusions:
Asthma patients with DB exhibit breathing-pattern alterations, most notably increased within-subject variability of timing and TA movement parameters. Breathing pattern variability may be a promising surrogate marker to characterise DB, highlighting the value of dynamic objective physiological assessment of breathing beyond conventional symptom-based assessments of DB.
DOI: https://doi.org/10.37349/eaa.2026.1009122

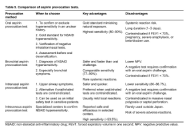
Non-steroidal anti-inflammatory drug (NSAID)-exacerbated respiratory disease (N-ERD) is a respiratory illness characterized by chronic eosinophilic airway inflammation. Although a typical clinical history of asthma, chronic rhinosinusitis with nasal polyps (CRSwNP), and respiratory reactions to NSAIDs may strongly suggest the diagnosis, history alone is often unreliable due to NSAID avoidance, atypical reactions, or incomplete symptom recognition. In this context, aspirin provocation tests remain the diagnostic gold standard for confirming N-ERD. Beyond diagnostic confirmation, aspirin provocation testing provides critical insights into disease heterogeneity and phenotyping. Different challenge routes—nasal, bronchial, and oral—allow assessment of organ-specific sensitivity and inflammatory dominance. Nasal aspirin provocation primarily reflects upper airway involvement and is particularly informative in patients with severe CRSwNP, whereas inhalational lysine-aspirin challenges highlight lower-airway bronchial hyperresponsiveness. Oral aspirin provocation, through systemic exposure, captures the full spectrum of respiratory and extra-respiratory responses and therefore best reflects global disease severity. The pattern, timing, and intensity of reactions observed during provocation testing contribute to the identification of clinically relevant N-ERD subphenotypes, such as upper-airway-dominant disease, bronchial-predominant disease, or blended reactions involving both compartments. These phenotypic distinctions have direct therapeutic implications, influencing the selection of targeted treatments, including aspirin desensitization, biologic therapies, or surgical interventions. Moreover, provocation testing remains essential prior to aspirin desensitization to ensure both diagnostic accuracy and patient safety. Aspirin provocation tests are not merely confirmatory tools but represent a cornerstone of precision-based evaluation of N-ERD, enabling refined phenotyping, risk stratification, and individualized treatment planning in this complex and heterogeneous disease.
Non-steroidal anti-inflammatory drug (NSAID)-exacerbated respiratory disease (N-ERD) is a respiratory illness characterized by chronic eosinophilic airway inflammation. Although a typical clinical history of asthma, chronic rhinosinusitis with nasal polyps (CRSwNP), and respiratory reactions to NSAIDs may strongly suggest the diagnosis, history alone is often unreliable due to NSAID avoidance, atypical reactions, or incomplete symptom recognition. In this context, aspirin provocation tests remain the diagnostic gold standard for confirming N-ERD. Beyond diagnostic confirmation, aspirin provocation testing provides critical insights into disease heterogeneity and phenotyping. Different challenge routes—nasal, bronchial, and oral—allow assessment of organ-specific sensitivity and inflammatory dominance. Nasal aspirin provocation primarily reflects upper airway involvement and is particularly informative in patients with severe CRSwNP, whereas inhalational lysine-aspirin challenges highlight lower-airway bronchial hyperresponsiveness. Oral aspirin provocation, through systemic exposure, captures the full spectrum of respiratory and extra-respiratory responses and therefore best reflects global disease severity. The pattern, timing, and intensity of reactions observed during provocation testing contribute to the identification of clinically relevant N-ERD subphenotypes, such as upper-airway-dominant disease, bronchial-predominant disease, or blended reactions involving both compartments. These phenotypic distinctions have direct therapeutic implications, influencing the selection of targeted treatments, including aspirin desensitization, biologic therapies, or surgical interventions. Moreover, provocation testing remains essential prior to aspirin desensitization to ensure both diagnostic accuracy and patient safety. Aspirin provocation tests are not merely confirmatory tools but represent a cornerstone of precision-based evaluation of N-ERD, enabling refined phenotyping, risk stratification, and individualized treatment planning in this complex and heterogeneous disease.
DOI: https://doi.org/10.37349/eaa.2026.1009121
This article belongs to the special issue Update on Chronic Rhinosinusitis

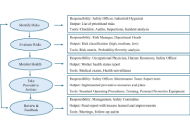
Occupational allergens are an important cause of diseases emerging in the workplace, both indoor and outdoor, causing conditions such as allergic asthma, dermatitis, and rhinitis. Numerous national and international institutions focus on sensitizing agents able to induce allergies in workplaces, which are also considered by the International Classification of Diseases (ICD)-11, underlining their increasing worldwide importance. There is thus the need to develop and implement new, multidisciplinary approaches to study and monitor these agents, taking into account different sources of exposure, environmental concentrations, co-factors of exposure, and individual susceptibility. This includes the integration between traditional and innovative methodologies applied to environmental and biological matrices, as well as the use of “omic” techniques. In this picture, information, training, and communication emerge as fundamental for workers. This kind of approach will permit us to attain a better management of exposure to allergens in the workplace, improving the well-being of workers worldwide.
Occupational allergens are an important cause of diseases emerging in the workplace, both indoor and outdoor, causing conditions such as allergic asthma, dermatitis, and rhinitis. Numerous national and international institutions focus on sensitizing agents able to induce allergies in workplaces, which are also considered by the International Classification of Diseases (ICD)-11, underlining their increasing worldwide importance. There is thus the need to develop and implement new, multidisciplinary approaches to study and monitor these agents, taking into account different sources of exposure, environmental concentrations, co-factors of exposure, and individual susceptibility. This includes the integration between traditional and innovative methodologies applied to environmental and biological matrices, as well as the use of “omic” techniques. In this picture, information, training, and communication emerge as fundamental for workers. This kind of approach will permit us to attain a better management of exposure to allergens in the workplace, improving the well-being of workers worldwide.
DOI: https://doi.org/10.37349/eaa.2026.1009120
This article belongs to the special issue Environment, Infectious Diseases, and Allergy

Climate change is reshaping the aeroallergen landscape, with rising temperatures, elevated CO2, shifting precipitation, and land-use change extending pollen seasons, increasing pollen loads and allergenicity, and expanding the geographic range of allergenic plants. These changes are accompanied by escalating air pollution from fossil fuel combustion and wildfires that act as an adjuvant with co-exposure with allergen exacerbate allergic airway disease. Vulnerable populations—particularly those in socioeconomically disadvantaged and marginalized communities in the US—experience disproportionate exposure to pollutants and allergens due to structural inequities that result in some populations being exposed to more environmental hazards than other groups. Climate-amplified aeroallergen exposure and air pollution are associated with higher sensitization, symptom burden, exacerbations, and healthcare use. Structural inequities magnify exposures to allergens and air pollution, while also influencing the social environment through concentration of poverty and diminished access to resources. This review synthesizes evidence linking climate change-related effects on aeroallergens and air pollution with allergic disease risk and the modification of this relationship by social vulnerability, with a focus on Europe and North America. We also highlight established and emerging strategies to mitigate the effects of climate change on allergic disease prevalence and morbidity, including anticipatory guidance, digital forecasting, community adaptation measures, and local, regional, and national policies that promote responsible land use, healthy housing, and equity-focused public health initiatives.
Climate change is reshaping the aeroallergen landscape, with rising temperatures, elevated CO2, shifting precipitation, and land-use change extending pollen seasons, increasing pollen loads and allergenicity, and expanding the geographic range of allergenic plants. These changes are accompanied by escalating air pollution from fossil fuel combustion and wildfires that act as an adjuvant with co-exposure with allergen exacerbate allergic airway disease. Vulnerable populations—particularly those in socioeconomically disadvantaged and marginalized communities in the US—experience disproportionate exposure to pollutants and allergens due to structural inequities that result in some populations being exposed to more environmental hazards than other groups. Climate-amplified aeroallergen exposure and air pollution are associated with higher sensitization, symptom burden, exacerbations, and healthcare use. Structural inequities magnify exposures to allergens and air pollution, while also influencing the social environment through concentration of poverty and diminished access to resources. This review synthesizes evidence linking climate change-related effects on aeroallergens and air pollution with allergic disease risk and the modification of this relationship by social vulnerability, with a focus on Europe and North America. We also highlight established and emerging strategies to mitigate the effects of climate change on allergic disease prevalence and morbidity, including anticipatory guidance, digital forecasting, community adaptation measures, and local, regional, and national policies that promote responsible land use, healthy housing, and equity-focused public health initiatives.
DOI: https://doi.org/10.37349/eaa.2026.1009119
This article belongs to the special issue Climate Change, Allergy, and Immunotherapy

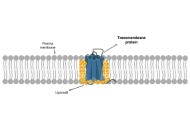
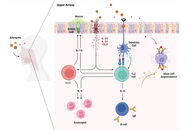
The composition and biophysical characteristics of the plasma membrane are pivotal in regulating mast cell immune functions by influencing receptor distribution, activation, and intracellular signaling pathways. This article highlights the impact of plasma membrane components, such as cholesterol and lipid rafts, on the function of the Mas-related G protein-coupled receptor X2 (MRGPRX2), a key mediator of IgE-independent mast cell activation and pseudoallergic reactions. We discuss how variations in membrane fluidity, lipid composition, and microdomain organization influence MRGPRX2 conformational dynamics, ligand accessibility, and downstream signaling efficiency. These membrane-driven effects may help explain the heterogeneity of mast cell responsiveness across tissues and disease states. Integrating insights from structural biology, biophysics, and clinical immunology emphasizes that plasma membrane composition and dynamics regulate MRGPRX2-mediated signaling, positioning the membrane environment as a promising therapeutic target for modulating mast-cell hyperreactivity. By outlining this conceptual framework, we introduce a unifying hypothesis that membrane-driven regulation is a critical, yet underrecognized, determinant of MRGPRX2 responsiveness in different tissues and disease states.
The composition and biophysical characteristics of the plasma membrane are pivotal in regulating mast cell immune functions by influencing receptor distribution, activation, and intracellular signaling pathways. This article highlights the impact of plasma membrane components, such as cholesterol and lipid rafts, on the function of the Mas-related G protein-coupled receptor X2 (MRGPRX2), a key mediator of IgE-independent mast cell activation and pseudoallergic reactions. We discuss how variations in membrane fluidity, lipid composition, and microdomain organization influence MRGPRX2 conformational dynamics, ligand accessibility, and downstream signaling efficiency. These membrane-driven effects may help explain the heterogeneity of mast cell responsiveness across tissues and disease states. Integrating insights from structural biology, biophysics, and clinical immunology emphasizes that plasma membrane composition and dynamics regulate MRGPRX2-mediated signaling, positioning the membrane environment as a promising therapeutic target for modulating mast-cell hyperreactivity. By outlining this conceptual framework, we introduce a unifying hypothesis that membrane-driven regulation is a critical, yet underrecognized, determinant of MRGPRX2 responsiveness in different tissues and disease states.
DOI: https://doi.org/10.37349/eaa.2026.1009118
This article belongs to the special issue Bridging Experimental and Translational Allergology

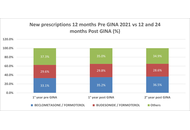
APPaRENT2 was a multi-country, cross-sectional, online survey of patients with asthma and physicians conducted in five countries (Argentina, Brazil, France, Mexico, and Italy), aimed at assessing physicians’ and patients’ preferred treatment strategies and goals. Education level, age, and place of residence differed between European and Latin American patients; furthermore, most Italian doctors worked within the Public Health Service. For the purpose of identifying the critical issues in the management of asthma in Italy, the data were analysed in greater detail, comparing those collected in Italy with those from Latin American countries and those provided by Italian patients and doctors. The data are reported as frequencies or means. The differences found in comparing data were not analysed for statistical significance, given the absence of any a priori hypothesis in the original paper. The Italian results were consistently within the range of the Latin American countries’ results. In Italy, despite the physicians’ prioritization of symptom control, many patients had poor asthma control but gave a surprisingly optimistic evaluation of their disease, given the high frequency of symptoms and limitations in everyday life. Physicians and patients had quite different evaluations of symptoms and outcomes of asthma. Finally, the combination of ICS/LABA with SABA as needed was the preferred treatment compared with the maintenance and reliever therapy (MART) strategy, suggested as preferential in the Global Initiative for Asthma (GINA) document. In conclusion, the absence of shared assessments and expectations between doctors and patients appears to be the primary issue to address in order to improve asthma treatment in Italy.
APPaRENT2 was a multi-country, cross-sectional, online survey of patients with asthma and physicians conducted in five countries (Argentina, Brazil, France, Mexico, and Italy), aimed at assessing physicians’ and patients’ preferred treatment strategies and goals. Education level, age, and place of residence differed between European and Latin American patients; furthermore, most Italian doctors worked within the Public Health Service. For the purpose of identifying the critical issues in the management of asthma in Italy, the data were analysed in greater detail, comparing those collected in Italy with those from Latin American countries and those provided by Italian patients and doctors. The data are reported as frequencies or means. The differences found in comparing data were not analysed for statistical significance, given the absence of any a priori hypothesis in the original paper. The Italian results were consistently within the range of the Latin American countries’ results. In Italy, despite the physicians’ prioritization of symptom control, many patients had poor asthma control but gave a surprisingly optimistic evaluation of their disease, given the high frequency of symptoms and limitations in everyday life. Physicians and patients had quite different evaluations of symptoms and outcomes of asthma. Finally, the combination of ICS/LABA with SABA as needed was the preferred treatment compared with the maintenance and reliever therapy (MART) strategy, suggested as preferential in the Global Initiative for Asthma (GINA) document. In conclusion, the absence of shared assessments and expectations between doctors and patients appears to be the primary issue to address in order to improve asthma treatment in Italy.
DOI: https://doi.org/10.37349/eaa.2026.1009117

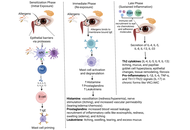
Allergic conjunctivitis (AC) is an inflammatory disorder of the ocular surface caused by allergic reactions to environmental substances. It presents with symptoms such as itching, redness of the eye, excessive tearing, and swelling/irritation in the eyes and eyelids. While many AC episodes occur on their own and go away, some forms of this disease are present in a chronic fashion or have the potential to cause serious loss of vision. In recent years, AC has been viewed as primarily an episodic irritative condition to a mucosal inflammatory condition in which the ocular surface provides an environment for the initiation and perpetuation of local immune responses. The molecular basis of AC represents a phase-linked inflammatory cascade: an immediate (minutes) mediator-driven response followed by a late (6–12 hours) cytokine/chemokine-driven cellular recruitment phase that can sustain symptoms and, in severe phenotypes, contribute to tissue remodeling. The initial response is due to the activation of mast cells via IgE-dependent pathways, producing the early phase response. The sustained response seen in the late phase of the disease is mediated by the action of lipid mediators and cytokines/chemokines involved in the recruitment of eosinophils and Th2-associated leukocytes. This narrative review synthesizes evidence on epithelial “alarmins” (TSLP, IL-33, IL-25) as upstream signals that may amplify type-2 inflammation in a phenotype-dependent manner, particularly in more severe or chronic disease, alongside established IgE/mast-cell biology. Further, we discuss neuroimmune mechanisms implicated in histamine-independent itch and symptom persistence, while noting that their clinical contribution likely varies across AC phenotypes. Finally, we will discuss how the mechanistic pathways relate to current limitations and to developing new therapeutic approaches.
Allergic conjunctivitis (AC) is an inflammatory disorder of the ocular surface caused by allergic reactions to environmental substances. It presents with symptoms such as itching, redness of the eye, excessive tearing, and swelling/irritation in the eyes and eyelids. While many AC episodes occur on their own and go away, some forms of this disease are present in a chronic fashion or have the potential to cause serious loss of vision. In recent years, AC has been viewed as primarily an episodic irritative condition to a mucosal inflammatory condition in which the ocular surface provides an environment for the initiation and perpetuation of local immune responses. The molecular basis of AC represents a phase-linked inflammatory cascade: an immediate (minutes) mediator-driven response followed by a late (6–12 hours) cytokine/chemokine-driven cellular recruitment phase that can sustain symptoms and, in severe phenotypes, contribute to tissue remodeling. The initial response is due to the activation of mast cells via IgE-dependent pathways, producing the early phase response. The sustained response seen in the late phase of the disease is mediated by the action of lipid mediators and cytokines/chemokines involved in the recruitment of eosinophils and Th2-associated leukocytes. This narrative review synthesizes evidence on epithelial “alarmins” (TSLP, IL-33, IL-25) as upstream signals that may amplify type-2 inflammation in a phenotype-dependent manner, particularly in more severe or chronic disease, alongside established IgE/mast-cell biology. Further, we discuss neuroimmune mechanisms implicated in histamine-independent itch and symptom persistence, while noting that their clinical contribution likely varies across AC phenotypes. Finally, we will discuss how the mechanistic pathways relate to current limitations and to developing new therapeutic approaches.
DOI: https://doi.org/10.37349/eaa.2026.1009116
This article belongs to the special issue Innate Immune Mechanisms in Allergic Diseases

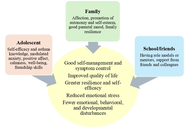
Background:
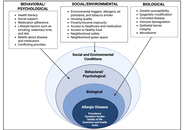
Adolescence is a vulnerable and constantly changing stage of life. Experiencing chronic illnesses such as bronchial asthma during this period can lead to heightened physical, psychological, and social problems in addition to the wide scope of challenges that coincide with the stage. The objective of this research was to identify risk and protective factors for mental health disorders, as well as the preventive and treatment strategies recommended to preserve mental health in adolescents with asthma (AA).
Methods:
A search was conducted in Medline, Web of Science, EBSCO Host, PsycINFO, ScienceDirect, and Scopus for articles published in English between 2020 and 2025 using the following search terms: i) asthma in adolescents and ii) psychosocial or emotional problems. The items were checked using the PRISMA checklist.
Results:
Thirty-eight articles were found: eight on mental health problems, fourteen on biopsychosocial risk factors, ten on biopsychosocial protection factors, and six on biopsychosocial interventions in AA.
Discussion:
Internalizing problems such as anxiety and depression, or externalizing problems such as attention deficit hyperactivity disorder (ADHD) or conduct disorders, are prevalent in AA. Several biopsychosocial risk factors, both individual and familial, have been identified as being related to mental health problems in AA. Protective biopsychosocial factors have also been found in AA, such as certain personal characteristics, family types or structures, friends, or schoolmates. Prevention or treatment strategies for mental health problems in AA should consider a personalized approach, taking into account the family system, friendships, and the school environment.
Background:
Adolescence is a vulnerable and constantly changing stage of life. Experiencing chronic illnesses such as bronchial asthma during this period can lead to heightened physical, psychological, and social problems in addition to the wide scope of challenges that coincide with the stage. The objective of this research was to identify risk and protective factors for mental health disorders, as well as the preventive and treatment strategies recommended to preserve mental health in adolescents with asthma (AA).
Methods:
A search was conducted in Medline, Web of Science, EBSCO Host, PsycINFO, ScienceDirect, and Scopus for articles published in English between 2020 and 2025 using the following search terms: i) asthma in adolescents and ii) psychosocial or emotional problems. The items were checked using the PRISMA checklist.
Results:
Thirty-eight articles were found: eight on mental health problems, fourteen on biopsychosocial risk factors, ten on biopsychosocial protection factors, and six on biopsychosocial interventions in AA.
Discussion:
Internalizing problems such as anxiety and depression, or externalizing problems such as attention deficit hyperactivity disorder (ADHD) or conduct disorders, are prevalent in AA. Several biopsychosocial risk factors, both individual and familial, have been identified as being related to mental health problems in AA. Protective biopsychosocial factors have also been found in AA, such as certain personal characteristics, family types or structures, friends, or schoolmates. Prevention or treatment strategies for mental health problems in AA should consider a personalized approach, taking into account the family system, friendships, and the school environment.
DOI: https://doi.org/10.37349/eaa.2026.1009115
This article belongs to the special issue Asthma and its Relationship with Psychological and Psychopathological Factors

Aim:
To describe the first major epidemic thunderstorm asthma (ETSA) event detected in France in June 2023.
Methods:
Data on local meteorology, visits to the emergency room (ER) for asthma and hospitalization after a visit, aerobiological composition of the atmosphere (pollens and spores), phenological information on the flowering of grasses, and regional air pollution were collected, aggregated, and analyzed.
Results:
The ETSA was centered on the Paris region. An excess of 1,900 emergency visits for asthma was recorded over the period 10, 11, and 12 June. The people most affected were men aged 14 to 44. The hospitalization rate following a visit to the ER for asthma increased to 13%. ER visits for asthma began at around 6 pm on 10 June, just after an intense gust (15 m/s) triggering a PM10 resuspension episode, and peaked at around 10 pm on 11 June. Concentrations of mold spores (Cladosporium and Ascosporium) rose sharply during the night of 10–11 June, at the same time as the intake peak. The ETSA occurred during the grass and Urticaceae pollen season, with pollen concentrations lower (< 100 pollen grains/m3) compared to the days preceding the event (> 200 pollen grains/m3). A fraction of the pollen was observed without cytoplasm, but there was no apparent link with the ETSA. Phenological observations in the Paris pollinarium showed that the ETSA coincided with the start of the Lolium perenne (ryegrass) pollen season.
Conclusions:
Although the data collected did not allow the identification of a single cause for the occurrence of the ETSA, they pointed to multifactorial causes such as the occurrence of an ozone pollution episode, strong winds before the storm, an episode of resuspension of PM10 particles, the presence of broken pollen, and the significant increase in mold spores just after the stormy episode.
Aim:
To describe the first major epidemic thunderstorm asthma (ETSA) event detected in France in June 2023.
Methods:
Data on local meteorology, visits to the emergency room (ER) for asthma and hospitalization after a visit, aerobiological composition of the atmosphere (pollens and spores), phenological information on the flowering of grasses, and regional air pollution were collected, aggregated, and analyzed.
Results:
The ETSA was centered on the Paris region. An excess of 1,900 emergency visits for asthma was recorded over the period 10, 11, and 12 June. The people most affected were men aged 14 to 44. The hospitalization rate following a visit to the ER for asthma increased to 13%. ER visits for asthma began at around 6 pm on 10 June, just after an intense gust (15 m/s) triggering a PM10 resuspension episode, and peaked at around 10 pm on 11 June. Concentrations of mold spores (Cladosporium and Ascosporium) rose sharply during the night of 10–11 June, at the same time as the intake peak. The ETSA occurred during the grass and Urticaceae pollen season, with pollen concentrations lower (< 100 pollen grains/m3) compared to the days preceding the event (> 200 pollen grains/m3). A fraction of the pollen was observed without cytoplasm, but there was no apparent link with the ETSA. Phenological observations in the Paris pollinarium showed that the ETSA coincided with the start of the Lolium perenne (ryegrass) pollen season.
Conclusions:
Although the data collected did not allow the identification of a single cause for the occurrence of the ETSA, they pointed to multifactorial causes such as the occurrence of an ozone pollution episode, strong winds before the storm, an episode of resuspension of PM10 particles, the presence of broken pollen, and the significant increase in mold spores just after the stormy episode.
DOI: https://doi.org/10.37349/eaa.2026.1009114
This article belongs to the special issue Climate Change, Allergy, and Immunotherapy

This review describes the eosinophil journey through the various physiological and pathophysiological phases, from production, maturation, and activation by chemokines and cytokines [especially eotaxin, interleukin (IL)-5, IL-3, and granulocyte-macrophage colony-stimulating factor (GM-CSF)], to interaction with the innate and adaptive immune system and tissue homing. Excessive production and activation of eosinophils lead to the release of granule proteins, such as major basic protein, eosinophil cationic protein, eosinophil peroxidase, and others, resulting in inflammation, cell cytotoxicity, and oxidative stress. The pathogenesis, clinical features, diagnostic processes, and the latest therapeutic approaches to the resulting diseases—which affect the upper and lower airways, gastrointestinal tract, skin, myocardium, and may occur systemically—are discussed.
This review describes the eosinophil journey through the various physiological and pathophysiological phases, from production, maturation, and activation by chemokines and cytokines [especially eotaxin, interleukin (IL)-5, IL-3, and granulocyte-macrophage colony-stimulating factor (GM-CSF)], to interaction with the innate and adaptive immune system and tissue homing. Excessive production and activation of eosinophils lead to the release of granule proteins, such as major basic protein, eosinophil cationic protein, eosinophil peroxidase, and others, resulting in inflammation, cell cytotoxicity, and oxidative stress. The pathogenesis, clinical features, diagnostic processes, and the latest therapeutic approaches to the resulting diseases—which affect the upper and lower airways, gastrointestinal tract, skin, myocardium, and may occur systemically—are discussed.
DOI: https://doi.org/10.37349/eaa.2026.1009111

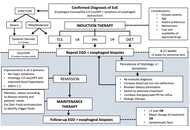
Eosinophilic esophagitis (EoE) is a chronic, immune-mediated inflammatory disease of the esophagus that has emerged as a major cause of esophageal dysfunction in all ages. Over the past two decades, its frequency has increased worldwide, reflecting both heightened recognition and a rise in occurrence. EoE predominantly affects males and frequently coexists with atopic conditions, underscoring its relationship with allergy. The pathogenesis involves genetic susceptibility, epithelial barrier dysfunction, and dysregulated type 2 immune responses. Variants in genes related to epithelial integrity and immune signaling, such as TSLP and CAPN14, predispose susceptible individuals to aberrant immune responses to food antigens, leading to eosinophil recruitment, mast cell activation, and chronic inflammation, which in turn promotes tissue remodeling and progression toward fibrostenotic disease. Clinical presentation varies with age. Infants and younger children often exhibit feeding difficulties, vomiting, and abdominal pain, whereas older children and adolescents usually present with dysphagia and food impaction. Diagnosis requires integration of clinical symptoms with histologic confirmation of esophageal eosinophilia (≥ 15 eosinophils per high-power field) and exclusion of alternative causes. Management of pediatric EoE aims to achieve and maintain clinical and histologic remission while preventing long-term complications and preserving quality of life. First-line therapeutic options include proton pump inhibitors, swallowed topical corticosteroids, and dietary elimination strategies. Biologic therapy has expanded treatment options for severe or refractory disease. Because symptom improvement alone does not reliably reflect disease control, objective reassessment with endoscopy and biopsies is recommended after treatment and during follow-up. Long-term outcomes of EoE are strongly influenced by diagnostic timing and adequacy of treatment. Early diagnosis, sustained anti-inflammatory therapy, and transition from pediatric to adult care are critical components of an appropriate management. Future directions include the development of precision medicine, identification of biomarkers to guide therapy selection, non-invasive tools for disease monitoring, and strategies aimed at disease modification.
Eosinophilic esophagitis (EoE) is a chronic, immune-mediated inflammatory disease of the esophagus that has emerged as a major cause of esophageal dysfunction in all ages. Over the past two decades, its frequency has increased worldwide, reflecting both heightened recognition and a rise in occurrence. EoE predominantly affects males and frequently coexists with atopic conditions, underscoring its relationship with allergy. The pathogenesis involves genetic susceptibility, epithelial barrier dysfunction, and dysregulated type 2 immune responses. Variants in genes related to epithelial integrity and immune signaling, such as TSLP and CAPN14, predispose susceptible individuals to aberrant immune responses to food antigens, leading to eosinophil recruitment, mast cell activation, and chronic inflammation, which in turn promotes tissue remodeling and progression toward fibrostenotic disease. Clinical presentation varies with age. Infants and younger children often exhibit feeding difficulties, vomiting, and abdominal pain, whereas older children and adolescents usually present with dysphagia and food impaction. Diagnosis requires integration of clinical symptoms with histologic confirmation of esophageal eosinophilia (≥ 15 eosinophils per high-power field) and exclusion of alternative causes. Management of pediatric EoE aims to achieve and maintain clinical and histologic remission while preventing long-term complications and preserving quality of life. First-line therapeutic options include proton pump inhibitors, swallowed topical corticosteroids, and dietary elimination strategies. Biologic therapy has expanded treatment options for severe or refractory disease. Because symptom improvement alone does not reliably reflect disease control, objective reassessment with endoscopy and biopsies is recommended after treatment and during follow-up. Long-term outcomes of EoE are strongly influenced by diagnostic timing and adequacy of treatment. Early diagnosis, sustained anti-inflammatory therapy, and transition from pediatric to adult care are critical components of an appropriate management. Future directions include the development of precision medicine, identification of biomarkers to guide therapy selection, non-invasive tools for disease monitoring, and strategies aimed at disease modification.
DOI: https://doi.org/10.37349/eaa.2026.1009112
This article belongs to the special issue Beyond Eosinophilic Gastrointestinal Diseases: Pathogenetic Mechanisms and Therapeutic Strategies

Scombroid syndrome is a frequent cause of fish poisoning and typically presents with mild, self-limiting symptoms. Severe anaphylaxis-like reactions are uncommon, and a biphasic clinical course has not previously been reported. We describe the case of a 19-year-old woman who developed flushing, headache, generalized urticaria, facial edema, dyspnea, and hypotension approximately 30 minutes after ingesting raw amberjack tartare. Emergency treatment with adrenaline, antihistamines, corticosteroids, and intravenous fluids led to initial clinical improvement; however, three hours later, she experienced a recurrence of cutaneous, respiratory, and gastrointestinal symptoms, consistent with a biphasic reaction. The patient subsequently developed chest pain associated with transient electrocardiographic changes and mild troponin elevation, prompting further evaluation for suspected Kounis syndrome. Allergy assessment, including specific IgE testing and skin-prick testing with fresh fish, was negative, supporting a toxic rather than IgE-mediated mechanism. This case expands the clinical spectrum of scombroid syndrome by documenting a biphasic anaphylaxis-like presentation and underscores the importance of considering histamine fish poisoning in the differential diagnosis of severe food-related reactions. Careful evaluation is essential to prevent misdiagnosis and unnecessary long-term dietary restrictions.
Scombroid syndrome is a frequent cause of fish poisoning and typically presents with mild, self-limiting symptoms. Severe anaphylaxis-like reactions are uncommon, and a biphasic clinical course has not previously been reported. We describe the case of a 19-year-old woman who developed flushing, headache, generalized urticaria, facial edema, dyspnea, and hypotension approximately 30 minutes after ingesting raw amberjack tartare. Emergency treatment with adrenaline, antihistamines, corticosteroids, and intravenous fluids led to initial clinical improvement; however, three hours later, she experienced a recurrence of cutaneous, respiratory, and gastrointestinal symptoms, consistent with a biphasic reaction. The patient subsequently developed chest pain associated with transient electrocardiographic changes and mild troponin elevation, prompting further evaluation for suspected Kounis syndrome. Allergy assessment, including specific IgE testing and skin-prick testing with fresh fish, was negative, supporting a toxic rather than IgE-mediated mechanism. This case expands the clinical spectrum of scombroid syndrome by documenting a biphasic anaphylaxis-like presentation and underscores the importance of considering histamine fish poisoning in the differential diagnosis of severe food-related reactions. Careful evaluation is essential to prevent misdiagnosis and unnecessary long-term dietary restrictions.
DOI: https://doi.org/10.37349/eaa.2026.1009113

Peanut allergy (PA) is a significant public health problem in Western countries; however, while some previous work has been conducted in Mexico among specific subgroups, the national prevalence of PA in the Mexican population remains unknown. This ENRADAL-MEX study aimed to estimate the prevalence of PA among Mexican schoolchildren. A total of 4,269 children aged 6–12 years were included (mean age: 8.7 years; 51.7% male). The national prevalence of adverse food reactions was 9.5%; among these, 16 cases (0.37%) were associated with peanut consumption, but only 11 presented symptoms within the first hour after ingestion, yielding a PA prevalence of 0.26% (95% CI: 0.14–0.47%). Five cases corresponded to convincing non-severe reactions, and the other five to convincing severe reactions (prevalence of 0.12%; 95% CI: 0.06–0.28%, each). Oral symptoms occurred in 54.5% of cases, and 63.6% also had tree nut allergy, with no reactions to other legumes. Since this national study is the first of its kind and indicates that PA is not currently a public health problem among Mexican schoolchildren, further research is encouraged for more comprehensive results.
Peanut allergy (PA) is a significant public health problem in Western countries; however, while some previous work has been conducted in Mexico among specific subgroups, the national prevalence of PA in the Mexican population remains unknown. This ENRADAL-MEX study aimed to estimate the prevalence of PA among Mexican schoolchildren. A total of 4,269 children aged 6–12 years were included (mean age: 8.7 years; 51.7% male). The national prevalence of adverse food reactions was 9.5%; among these, 16 cases (0.37%) were associated with peanut consumption, but only 11 presented symptoms within the first hour after ingestion, yielding a PA prevalence of 0.26% (95% CI: 0.14–0.47%). Five cases corresponded to convincing non-severe reactions, and the other five to convincing severe reactions (prevalence of 0.12%; 95% CI: 0.06–0.28%, each). Oral symptoms occurred in 54.5% of cases, and 63.6% also had tree nut allergy, with no reactions to other legumes. Since this national study is the first of its kind and indicates that PA is not currently a public health problem among Mexican schoolchildren, further research is encouraged for more comprehensive results.
DOI: https://doi.org/10.37349/eaa.2026.1009110
This article belongs to the special issue Practical Issues in Pediatric Allergy

Aim:
This study aimed to assess the effectiveness of mepolizumab in enhancing asthma control, achieving clinical remission, and alleviating upper airway symptoms in patients with severe eosinophilic asthma (SEA) with comorbid nasal polyps and/or chronic rhinosinusitis (CRS). Additionally, it aimed to identify clinical and laboratory predictors of remission. The findings are based on real-world data from a single center.
Methods:
This retrospective, single-center, real-world study included 99 patients diagnosed with SEA. Patients were categorized into three groups based on the presence or absence of nasal polyps and CRS. Treatment response was evaluated using the asthma control test (ACT), spirometry, laboratory biomarkers, computed tomography (CT) scores, and nasal polyp scores. Remission was defined as the absence of asthma exacerbations and systemic corticosteroid use, along with improvements in both forced expiratory volume in 1 second (FEV1) and ACT scores.
Results:
After 12 months of mepolizumab therapy, there were significant improvements in FEV1 values, asthma exacerbation frequency, systemic corticosteroid requirements, and nasal symptom scores. The overall remission rate was 30.6%. Patients with higher baseline FEV1 and no prior exposure to omalizumab were more likely to achieve remission.
Conclusions:
This real-world evidence suggests that mepolizumab provides meaningful clinical, functional, and radiological improvements in patients with SEA, regardless of comorbid nasal polyps or CRS. Furthermore, the study highlights independent predictors associated with treatment-induced remission in this population.
Aim:
This study aimed to assess the effectiveness of mepolizumab in enhancing asthma control, achieving clinical remission, and alleviating upper airway symptoms in patients with severe eosinophilic asthma (SEA) with comorbid nasal polyps and/or chronic rhinosinusitis (CRS). Additionally, it aimed to identify clinical and laboratory predictors of remission. The findings are based on real-world data from a single center.
Methods:
This retrospective, single-center, real-world study included 99 patients diagnosed with SEA. Patients were categorized into three groups based on the presence or absence of nasal polyps and CRS. Treatment response was evaluated using the asthma control test (ACT), spirometry, laboratory biomarkers, computed tomography (CT) scores, and nasal polyp scores. Remission was defined as the absence of asthma exacerbations and systemic corticosteroid use, along with improvements in both forced expiratory volume in 1 second (FEV1) and ACT scores.
Results:
After 12 months of mepolizumab therapy, there were significant improvements in FEV1 values, asthma exacerbation frequency, systemic corticosteroid requirements, and nasal symptom scores. The overall remission rate was 30.6%. Patients with higher baseline FEV1 and no prior exposure to omalizumab were more likely to achieve remission.
Conclusions:
This real-world evidence suggests that mepolizumab provides meaningful clinical, functional, and radiological improvements in patients with SEA, regardless of comorbid nasal polyps or CRS. Furthermore, the study highlights independent predictors associated with treatment-induced remission in this population.
DOI: https://doi.org/10.37349/eaa.2026.1009109
This article belongs to the special issue Update on Chronic Rhinosinusitis

Aim:
Woodworking exposes carpenters to a higher risk of developing asthma or worsening pre-existing asthma. The objective of this study was to determine the prevalence of and factors associated with work-related asthma (WRA) among carpenters in Parakou in 2024.
Methods:
This was a cross-sectional, descriptive, and analytical study with prospective data collection conducted from June to September 2024. Following a voluntary survey, the included carpenters were interviewed using the Occupational Asthma Screening Questionnaire-11 (OASQ-11) to assess the relationship between asthma symptoms and the occupational environment. Peak expiratory flow (PEF) variability and spirometry were also measured. Data were analyzed using R software version 4.4.1. Factors associated with WRA were identified using simple and multiple logistic regression analyses.
Results:
Out of 153 carpenters/apprentices in 117 workshops, 144 (94.1%) were included, all of whom were male. The mean age was 35.9 ± 12.1 years. Among them, 15 (10.4%) had a WRA profile, including 10 (6.9%; 95% CI: 3.8–12.3) with occupational asthma and 5 (3.5%; 95% CI: 1.5–7.9) with work-aggravated asthma. Asthma was confirmed in 7 of the 15 carpenters suspected of having WRA through PEF variability measurement and spirometry. Simple and multiple logistic regression analyses identified a history of allergic rhinitis (aOR = 3.90; p = 0.033) and urticaria (aOR = 8.21; p = 0.002) as factors significantly associated with WRA.
Conclusions:
The prevalence of WRA among carpenters in Parakou is not negligible, particularly occupational asthma. Awareness campaigns, education for carpenters, regular monitoring of working conditions, and systematic medical follow-up of exposed workers could help preserve their respiratory health.
Aim:
Woodworking exposes carpenters to a higher risk of developing asthma or worsening pre-existing asthma. The objective of this study was to determine the prevalence of and factors associated with work-related asthma (WRA) among carpenters in Parakou in 2024.
Methods:
This was a cross-sectional, descriptive, and analytical study with prospective data collection conducted from June to September 2024. Following a voluntary survey, the included carpenters were interviewed using the Occupational Asthma Screening Questionnaire-11 (OASQ-11) to assess the relationship between asthma symptoms and the occupational environment. Peak expiratory flow (PEF) variability and spirometry were also measured. Data were analyzed using R software version 4.4.1. Factors associated with WRA were identified using simple and multiple logistic regression analyses.
Results:
Out of 153 carpenters/apprentices in 117 workshops, 144 (94.1%) were included, all of whom were male. The mean age was 35.9 ± 12.1 years. Among them, 15 (10.4%) had a WRA profile, including 10 (6.9%; 95% CI: 3.8–12.3) with occupational asthma and 5 (3.5%; 95% CI: 1.5–7.9) with work-aggravated asthma. Asthma was confirmed in 7 of the 15 carpenters suspected of having WRA through PEF variability measurement and spirometry. Simple and multiple logistic regression analyses identified a history of allergic rhinitis (aOR = 3.90; p = 0.033) and urticaria (aOR = 8.21; p = 0.002) as factors significantly associated with WRA.
Conclusions:
The prevalence of WRA among carpenters in Parakou is not negligible, particularly occupational asthma. Awareness campaigns, education for carpenters, regular monitoring of working conditions, and systematic medical follow-up of exposed workers could help preserve their respiratory health.
DOI: https://doi.org/10.37349/eaa.2026.1009108
 Previous
Previous