Affiliation:
Department of Radiology, University of Washington, Seattle, WA 98195, USA
Email: aiphuong.s.tong@gmail.com
ORCID: https://orcid.org/0000-0002-2771-9504
Explor Neurosci. 2026;5:1006131 DOI: https://doi.org/10.37349/en.2026.1006131
Received: December 08, 2025 Accepted: March 02, 2026 Published: March 25, 2026
Academic Editor: Jinwei Zhang, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, China
The article belongs to the special issue Advances in Epilepsy Research
Human behavior depends on a collection of cognitive capacities that are expressed with complexity in humans. Although animal models have been essential for identifying fundamental neural mechanisms, many aspects of human cognition require direct investigation in the human brain. Studies of social decision-making, communication, and spatial navigation increasingly rely on intracranial electrophysiology to probe the neural basis. Related to these topics, reward processing warrants emphasis. It is not uniquely human, but it provides a central organizing signal linking motivation, learning, emotion, and choice across many human behaviors. Disruptions of reward circuits are a hallmark of numerous neurological and psychiatric conditions, giving this domain specific relevance for patient care. Fifteen studies published between 2009 and 2024 used human intracranial recordings to examine reward-related processes, nearly all in patients undergoing invasive monitoring for drug-resistant epilepsy. These studies investigated 17 neocortical and subcortical regions, most frequently the orbitofrontal cortex, using intracranial EEG, deep brain stimulation, and single-unit recordings. Recent work increasingly incorporates social interactions and computational models of learning. The purpose of this narrative review is to provide an overview of human reward processing, emphasizing how intracranial recordings have clarified the neural circuits that underlie a range of human cognitive capacities. Beyond advancing basic neuroscience, intracranial electrophysiology can inform circuit-guided interventions for neurological and psychiatric disorders.
Humans have internal states, motivations, and cognitive goals that are distinct from other animal species, and these internal states strongly influence how reward expectations bias behavior and facilitate learning [1, 2]. Rodent and non-human primate models have provided many insights into the neural architecture of reinforcement learning, decision-making, and value representation [3, 4]. However, human aspects of reward processing, including abstract goal formation, metacognition, social evaluation, and self-referential thought, remain critically understudied due to historical limitations in human electrophysiology [5, 6]. Only a small minority of individuals require invasive intracranial recordings for clinical purposes. Thus, opportunities to interrogate human reward circuits at cellular and network resolution have been limited [7]. With expanding indications for stereo-electroencephalography (sEEG) and deep brain stimulation (DBS), as well as improvements in electrode technology, there will be increasing opportunities to investigate what makes human reward learning unique and to identify potential therapeutic targets when these processes are disrupted [8, 9].
Although reward processing is not unique to humans, it occupies a privileged position in human cognition, shaping goal selection, learning, social behavior, and motivation. As a result, reward circuits represent one of the most clinically and mechanistically informative ways for understanding how internal states and abstract goals are instantiated in the human brain, and how disruption gives rise to psychiatric and neurological disease. Human reward processing is highly dynamic, involving rapid fluctuations in valuation, prediction error signaling, and cognitive control [10, 11]. Capturing these processes requires neural recordings with very high temporal resolution, something invasive electrophysiology is uniquely positioned to provide [12, 13]. Many human decisions are also temporally extended, requiring maintenance of goals, prospective planning, and sequential information sampling over long time horizons [14]. Long-duration, high-density intracranial recordings allow study of these processes with unprecedented specificity, revealing how distributed networks encode evolving reward expectations and integrate them into behavior [15]. As the field continues to grow, invasive human recordings will play a central role in refining understanding of the neural dynamics that underlie reward, decision-making, social cognition, language, and memory, ultimately deepening insight into the computations that make humans capable.
In the review, reward processing is examined in greater depth because it integrates communication, learning, and decision-making and is broadly implicated in neurological and psychiatric disorders. Studies were identified through targeted literature searches of PubMed and Google Scholar using combinations of keywords including “intracranial EEG”, “single-unit recording”, “deep brain stimulation”, “reward”, “value”, “prediction error”, and “human”. Eligible studies included original research reports involving direct intracranial recordings from neocortical or subcortical regions in human participants performing tasks related to reward valuation, learning, decision-making, or reinforcement processes. Studies providing mechanistic insight into neural dynamics, including high-frequency activity or single-neuron responses, are discussed in more depth. Studies limited to non-invasive recordings or lacking task-based reward components were excluded unless relevant to conceptual framing. This review is narrative rather than systematic and is not intended to be exhaustive. While efforts were made to capture influential and representative findings, some areas of the literature remain relatively sparse, particularly regarding subcortical human recordings and longitudinal clinical data. These gaps reflect both methodological constraints and the current state of the field.
Fifteen studies published between 2009 and 2024 were identified that specifically employed human intracranial recordings to study reward processing. These studies investigated 17 neocortical and subcortical regions (Table 1). The orbitofrontal cortex (OFC) was most frequently examined (7 studies, 46.7%). Sample sizes averaged 12.4 ± 8.6 participants (range 1–36). Recording techniques included intracranial EEG (iEEG; 8 studies, 53.3%), DBS (5 studies, 33.3%), and single-unit arrays (2 studies, 13.3%). Social interactions were assessed in a subset of recent studies (3, 20%; 2021–2024), and behavioral modeling of learning was reported in another 3 studies (20%). These studies emphasize the investigation of internal goals, intentions, and decision-making (Tables 2 and 3). From a review of trends in human electrophysiology studies, the number of publications has increased in recent years, particularly since 2023, and most studies included 10–15 participants (Figure 1).
Summary of human intracranial studies on reward processing published between 2009 and 2024.
| Study information | Number of studies | % of studies |
|---|---|---|
| Participants | ||
| Epilepsy | 10 | 66.7% |
| Depression | 2 | 13.3% |
| Movement disorders | 3 | 20.0% |
| Electrophysiological implants | ||
| DBS-local field potentials | 3 | 20.0% |
| DBS/FSCV-dopamine | 2 | 13.3% |
| Single units | 2 | 13.3% |
| Electrocorticography (strips/grid) | 5 | 33.3% |
| Stereo EEG | 3 | 20.0% |
| Research questions | ||
| Social | 3 | 20.0% |
| Non-social | 12 | 80.0% |
| Behavioral modeling | 3 | 20.0% |
| No behavioral model | 12 | 80.0% |
| Cross-region synchronization | 2 | 13.3% |
| No synchronization | 13 | 86.7% |
| Regions of interest—neocortical | ||
| OFC (lateral/medial) | 7 (3/1) | 46.7 (20.0/6.7)% |
| vmPFC/dlPFC/dmPFC | 4/1/3 | 26.7%/6.7%/20% |
| Frontal pole | 1 | 6.7% |
| Cingulate cortex | 1 | 6.7% |
| Amygdala | 2 | 13.3% |
| Parahippocampal cortex | 1 | 6.7% |
| Insula | 3 | 20.0% |
| Supramarginal gyrus | 1 | 6.7% |
| Angular gyrus | 1 | 6.7% |
| Pre-supplementary motor area | 1 | 6.7% |
| Regions of interest—subcortical | ||
| Nucleus accumbens | 1 | 6.7% |
| Ventral striatum | 1 | 6.7% |
| SNr | 1 | 6.7% |
| Caudate | 1 | 6.7% |
| Putamen | 1 | 6.7% |
| Thalamus | 1 | 6.7% |
| Hippocampus | 3 | 20.0% |
Studies are categorized by participant population, electrophysiological implant type, research question, use of behavioral modeling, cross-region synchronization analysis, and regions of interest. Percentages indicate the proportion of studies within each category. Neocortical and subcortical regions are listed separately. DBS: deep brain stimulation; dlPFC: dorsolateral prefrontal cortex; dmPFC: dorsomedial prefrontal cortex; FSCV: fast-scan cyclic voltammetry; OFC: orbitofrontal cortex; SNr: substantia nigra reticulata; vmPFC: ventromedial prefrontal cortex.
Summary of human intracranial studies examining reward-related neural activity, including the number of participants, brain regions recorded, electrophysiological features, behavioral tasks, and analytical approaches.
| First author, Year | Number of participants | Regions of interest | Electrophysio-logic features | Task | Analysis |
|---|---|---|---|---|---|
| Cohen et al., 2009 [16] | N = 5, DBS for treatment-resistant depression | Nucleus accumbens | Alpha (8–12 Hz), gamma (40–80 Hz), gamma-alpha synchronization | Rewarding shape learning task, select shapes on left versus right, given text feedback of money won/lost with sound (tada!/buzzer); not instructed; win (75%) or lose (25%) €0.12, 152 trials | Stimulus-locked changes in frequency over time; examined whether higher frequencies were coupled with activity in lower-frequency bands for each condition; examined possible differences between gamma-alpha couplings across different conditions. |
| Lega et al., 2011 [17] | N = 1, DBS for treatment-resistant depression | Bilateral ventral striatum | Firing rate: theta (4–8 Hz), alpha (10–14 Hz), beta (16–24 Hz), and low/high gamma | Video game reward task, 104 trials total | Firing rate and local field potential power for each of the three conditions: positive feedback, negative feedback, and reward-neutral feedback. |
| Kishida et al., 2016 [18] | N = 17, DBS for PD | Caudate (N = 14), putamen (N = 3). | Estimate dopamine concentration, measured by fast-scan cyclic voltammetry using linear regression models trained on in vitro data and the EN algorithm | Sequential investment game; 120 investment decisions (20 choices × 6 markets); adjust and submit investment, 0 to 100% of portfolio in 10% increments, experience gain or loss | Reward prediction error (RPE) is computed at each trial, measured as the difference between outcome and expected value (average return up to trial), and counterfactual prediction error (CPE). |
| Li et al., 2016 [19] | N = 8, sEEG for treatment-resistant epilepsy | Medial and lateral OFC | Local field potential amplitude; 86 contacts (35% code reward prob) | Probabilistic reward learning task; five slot machines with five reward probabilities (0/0.25/0.5/0.75/1) | “Expected value”—reward probability estimate since the first trial.“Risk”—emerges during reward anticipation or can also be found at the time of reward outcome.“Experienced value”—response to reward or no reward.Comparing amplitude (uV) and reaction times for different reward probabilities. |
| Saez et al., 2018 [13] | N = 10, ECoG (grids/strips for treatment-resistant epilepsy) | OFC (n = 192) | High frequency activity (70–200 Hz) | A neuroeconomic task that captures the tradeoff between risk and reward | Defined two sets of reward-related signals associated with both choice and outcome evaluation processes |
| Lopez-Persem et al., 2020 [20] | N = 36, iEEG for treatment-resistant epilepsy | Electrodes in vmPFC (n = 146); HC (n = 280); lOFC (n = 304); PHC (n = 122); total n =4,273 electrodes | High gamma (50–150 Hz) | Judgment tasks.Phase 1: ‘age rating and confidence task’ (120 trials, two blocks, faces and paintings; age 21-step scale, 100-step confidence).Phase 2: ‘likeability rating and confidence task’ (180 trials, three blocks, same faces and paintings, added food items, likeability 21-step scale, −10 to 10).Phase 3: forced binary choice task among prior stimuli (food, face, or painting). | Value representation for different categories of items (food, faces, paintings), value-based and non-value-based, first-order judgments (food/nonfood likeability and age ratings), and second-order judgments (confidence ratings).“Subjective value”: regression estimates of OFC (pooling vmPFC and lOFC) and (P)HC (pooling hippocampus and PHC) activity against food likeability rating |
| Gueguen et al., 2021 [21] | N = 20, iEEG for treatment-resistant epilepsy | Electrodes in aINS (n = 83)dlPFC (n = 74)vmPFC (n = 54)lOFC (n = 70) | Broadband gamma activity (BGA, in the 50−150 Hz range) and beta band (13–33 Hz), theta/alpha (4–8 and 8–13 Hz) | Instrumental learning task.Choose between two cues to either maximize monetary gains (for reward cues) or minimize monetary losses (for punishment cues). The pairs of cues associated with reward and punishment learning were intermingled within three to six sessions of 96 trials. In each pair, the two cues were associated with the two possible outcomes (0/1€ in the reward condition and 0/–1€ in the punishment condition) with reciprocal probabilities (0.75/0.25 and 0.25/0.75). | Generate trial-wise expected values and prediction errors, we fitted a Q-learning model (QL) to behavioral data |
| Jamali et al., 2021 [22] | N = 11, single-neuron recordings DBS for essential tremor (N = 7), PD (N = 3), and dystonia (N = 1) | Single cells in the human dmPFC (n = 212) | Firing rate | Verbal version of the false-belief task: other-belief versus physical trials; true- versus false-belief trials; true-physical versus false-physical trials | A Fisher discriminant was used to evaluate whether and to what degree the activity of each neuron during questioning could be used to predict specific trial conditions on a trial-by-trial basis.Model-switch decoding—a drop in decoding accuracy on model-switching would suggest that neuronal responses were selective for another’s beliefs. |
| Manssuer et al., 2022 [23] | N = 16, sEEG for treatment-resistant epilepsy | LFPs from amygdala (n = 16); OFC (n = 9); hippocampus (n = 9) | High frequency gamma 60–250 Hz; theta 4–8 Hz synchrony between amygdala and OFC | Monetary incentive delay task | Monetary reward processing |
| Aquino et al., 2023 [24] | N = 20, Behnke-Fried depth electrodes (macro- and micro-recordings) for treatment-resistant epilepsy | Pre-SMA (n = 137), vmPFC (n = 191) and dACC (n = 108) neurons (436 total) | Firing rate | Two-armed bandit task; contained 20 blocks of 15 trials, a total of 300 trials; split into two recording sessions with ten blocks each.Trial began with a baseline period (sampled randomly from a uniform distribution of 0.75–1.25 s), followed by a choice screen showing the two available slot machines presented on the left or on the right of the screen; after button press, chosen slot machine was shown for a period of 1–2 s (sampled randomly from a uniform distribution), followed by outcome screen shown for 2 s. The outcome screen showed either a golden coin to represent winning a reward or a crossed-out coin to represent not winning. | Logistic regression model to describe how the past history of rewards, sampling history, stimulus exposure history and their interactions with trial number correlated with decisions; defined Q values (denoted as Qs) as the mean of a β distribution that estimates the probability of receiving a reward from a bandit, as determined by the history of wins and losses after sampling a stimulus s, as well as δQ = Qleft – Qright, the difference between left and right Q values; defined an uncertainty value U as the variance of the same β distribution, as well as its corresponding differential δU = Uleft – Uright; defined novelty (N) as the variance of a β distribution in which β = 1 and the α parameter is the number of times patients were exposed to a stimulus s in the entire session, as well as its corresponding differential δN = Nleft – Nright. |
| Collomb-Clerc et al., 2023 [25] | N = 8, DBS for treatment-resistant epilepsy | Anterior thalamus (ATN) and dorsomedial thalamus (DMTN) | Low frequency oscillations (4–12 Hz) | Probabilistic instrumental reinforcement learning task.Cues were abstract visual stimuli taken from the agathodaimon alphabet. The four cue pairs were divided into two conditions (2 pairs of reward and 2 pairs of punishment cues), associated with different pairs of outcomes (winning 1€ versus nothing or losing 1€ versus nothing). | A standard QL algorithm was used to model choice behavior. |
| Hoy et al., 2023 [26] | N = 10, sEEG and ECoG for treatment-resistant epilepsy | Insula (n = 9) and dMPFC (n = 10) (primarily mid-cingulate cortex) | High frequency activity (7–150 Hz) | Interval timing task, consisting of four blocks (two easy, two hard), 75 trials each, with initial instruction indicating difficulty level. | HFA power as a marker of local population dynamics, compared the performance of three different linear mixed models in explaining single-trial dMPFC and INS responses to positive, negative, and neutral feedback during the task. |
| Marciano et al., 2023 [27] | N = 10, sEEG for treatment-resistant epilepsy | OFC n = 144 electrodes | High frequency activity (70–150 Hz) | Repeated trial dictator game; a single player (the ‘‘dictator’’) decides how to split different pots of money between themselves and a social counterpart to study social decision making.In each trial, patients chose between two allocations of money for themselves and another anonymous social counterpart. | OFC ECoG/single-unit activity analysis with time-resolved regression models, using predictors (reward magnitude, advantageous inequity, and disadvantageous inequity) to characterize modulation of neural firing rates; trial-wise encoding analyses to identify neurons and time windows where activity significantly tracked inequity-dependent value signals. |
| Batten et al., 2024 [28] | N = 4, DBS for PD, two intra-op recordings, spaced 14–28 days | SNr | Human electrochemistry, carbon-fibre electrodes | Ultimatum game, a two-person ‘take-it-or-leave-it’ game probing social fairness norms | Electrochemical estimates of dopamine and serotonin in the SNr during the ultimatum game |
| Man et al., 2024 [29] | N = 10, iEEG for treatment-resistant epilepsy (16 recording sessions) | Frontal pole, vmPFC, OFC, supramarginal gyrus, angular gyrus, putamen, hippocampus, amygdala, anterior and posterior insula428 contacts | Average contact activity (1–250 Hz) | Presented with two cards drawn sequentially and without replacement on each trial; cards were shuffled from a deck of 10 cards (ace to ten), excluding face cards. Instructed to treat an ace card as denoting “1”; cards were reshuffled after every trial; prior to drawing a card, participants predicted whether the second card have higher or lower numerical value than the first. | Understand how the brain temporally organizes reward and risk representations |
N: number of patients; n: number of electrodes; aINS: anterior insula; DBS: deep brain stimulation; dlPFC: dorsolateral prefrontal cortex; dmPFC: dorsomedial prefrontal cortex; lOFC: lateral orbitofrontal cortex; OFC: orbitofrontal cortex; PD: Parkinson’s disease; pre-SMA: pre-supplementary motor area; sEEG: stereo-electroencephalography; SNr: substantia nigra reticulata; vmPFC: ventromedial prefrontal cortex.
Summary of key findings from human intracranial recording studies of reward, value, and decision-making.
| Study | Title | Key takeaway |
|---|---|---|
| Cohen et al., 2009 [16] | Good Vibrations: Cross-frequency Coupling in the Human Nucleus Accumbens during Reward Processing | Gamma-alpha coupling in the nucleus accumbens acts as a gating mechanism encoding reward information via phase coding. |
| Lega et al., 2011 [17] | Neuronal and oscillatory activity during reward processing in the human ventral striatum | Reward processing in the ventral striatum involves reward-sensitive neurons and distinct oscillatory patterns differentiating positive versus negative feedback. |
| Kishida et al., 2016 [18] | Subsecond dopamine fluctuations in human striatum encode superposed error signals about actual and counterfactual reward | Subsecond dopamine fluctuations encode integrated RPEs, including actual and counterfactual outcomes. |
| Li et al., 2016 [19] | The neural dynamics of reward value and risk coding in the human orbitofrontal cortex | Different OFC subregions encode reward probability, risk, and experienced value at distinct time points during reward anticipation and delivery. |
| Saez et al., 2018 [13] | Encoding of Multiple Reward-Related Computations in Transient and Sustained High-Frequency Activity in Human OFC | Human OFC shows fast transient and sustained high-frequency activity reflecting multiple valuation signals, including risk and outcome regret. |
| Lopez-Persem et al., 2020 [20] | Four core properties of the human brain valuation system demonstrated in intracranial signals | Subjective value is encoded in both OFC and parahippocampal cortex, modulated by pre-stimulus activity, and follows linear and quadratic functions. |
| Gueguen et al., 2021 [21] | Anatomical dissociation of intracerebral signals for reward and punishment prediction errors in humans | Broadband gamma activity encodes reward and punishment outcomes and expectations in anatomically distinct human brain regions. |
| Jamali et al., 2021 [22] | Single-neuronal predictions of others’ beliefs in humans | Neurons in human dmPFC encode others’ beliefs, supporting theory of mind processing at the single-neuron level. |
| Manssuer et al., 2022 [23] | Integrated Amygdala, Orbitofrontal and Hippocampal Contributions to Reward and Loss Coding Revealed with Human Intracranial EEG | Reward anticipation synchronizes hippocampus, amygdala, and OFC activity, implicating these regions in memory and motivation integration. |
| Aquino et al., 2023 [24] | Neurons in human pre-supplementary motor area encode key computations for value-based choice | PreSMA neurons integrate separate choice variables into unified value signals guiding decision making. |
| Collomb-Clerc et al., 2023 [25] | Human thalamic low-frequency oscillations correlate with expected value and outcomes during reinforcement learning | Low-frequency oscillations in thalamic subregions correlate with expected value signals during reward and punishment learning. |
| Hoy et al., 2023 [26] | Asymmetric coding of reward prediction errors in human insula and dorsomedial prefrontal cortex | Insula leads dmPFC in processing and communicating asymmetric RPE signals based on valence and salience. |
| Marciano et al., 2023 [27] | Electrophysiological signatures of inequity-dependent reward encoding in the human OFC | OFC high-frequency activity encodes self and others’ rewards, including social inequity types, reflecting social valuation processes. |
| Batten et al., 2024 [28] | Dopamine and serotonin in human substantia nigra track social context and value signals during economic exchange | Dopamine in the substantia nigra tracks RPEs, while serotonin tracks offer value modulated by social context during economic exchanges. |
| Man et al., 2024 [29] | Temporally organized representations of reward and risk in the human brain | Reward and risk representations in the human brain are temporally organized, revealing how these computations unfold over time. |
This table provides the key takeaway for each study, highlighting electrophysiological signatures, neural computations, and brain regions involved in processing reward, risk, prediction errors, and social valuation. dmPFC: dorsomedial prefrontal cortex; HC: hippocampus; HFA: high-frequency activity; INS: insula; LFO: low-frequency oscillations; OFC: orbitofrontal cortex; PHC: parahippocampal cortex; preSMA: pre-supplementary motor area; RPE: reward prediction error.
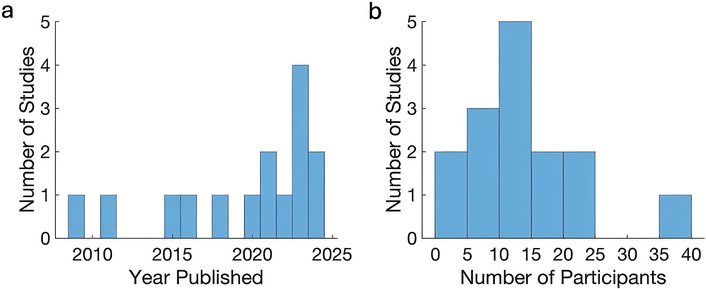
Characteristics of studies surveyed to assess trends in human electrophysiology research. (a) Distribution of study publication years. (b) Distribution of the number of participants per study.
Much of what we currently know about human reward processing and large-scale neural dynamics comes from non-invasive techniques such as magnetoencephalography (MEG) and scalp electroencephalography (scEEG) [30, 31]. These methods provide millisecond temporal resolution and have been used to study a wide range of cognitive and affective processes while participants remain relatively sedentary [32, 33]. MEG and scEEG have offered insight into decision-making, value computation, sensory processing, and motor planning, but are limited in resolving activity from deep structures such as the amygdala, hippocampus, ventral striatum, and thalamus [34].
The skull substantially attenuates high-frequency activity, particularly broadband high gamma (70–200 Hz), a signal closely correlated with local neuronal spiking [35, 36]. As a result, high-frequency signatures of reward computation, memory operations, and rapid cognitive transitions often remain inaccessible to MEG or scEEG. Spatial smearing and depth biases further limit their ability to distinguish fine-grained circuits or heterogeneous subpopulations within the cortex [37]. Because human cognition relies on rapid, high-frequency, and spatially distributed computations across both neocortical and subcortical networks, invasive electrophysiology offers a window into neural dynamics that non-invasive methods cannot reliably capture [5–7, 38].
Invasive intracranial recordings-including electrocorticography (ECoG), sEEG, microelectrode arrays, and depth electrodes-provide high spatial and temporal resolution with direct access to deep structures such as the hippocampus, nucleus accumbens, amygdala, basal ganglia, and thalamus [12, 39, 40]. These structures support reward learning, emotional processing, episodic memory, and goal-directed behavior, yet remain sparsely accessed by non-invasive recordings. Through high-density intracranial recordings, hierarchical organization within the human prefrontal cortex has been demonstrated. Anterior prefrontal regions encode long-horizon predictions, whereas posterior regions encode immediate action-outcome contingencies [41, 42]. Similarly, hippocampal-prefrontal circuits exhibit grid-like and map-like representations during predictive and memory-guided behavior [43, 44]. Specialization is also evident in motor and language cortices. Dorsal-ventral gradients in the precentral gyrus support error monitoring, articulatory sequencing, and phonological encoding [45, 46].
Within reward-related regions, including dorsomedial prefrontal cortex (dmPFC), anterior cingulate cortex (ACC), OFC, and insula, invasive recordings reveal small, intermingled populations selective for reward prediction errors (RPEs), value signals, and belief-state representations, patterns difficult to detect non-invasively [47, 48]. Such fine-grained circuit resolution is essential for understanding reinforcement learning, credit assignment, and human decision-making.
Invasive electrophysiology includes both non-penetrating and penetrating modalities:
ECoG uses subdural grids to record local field potentials with excellent signal-to-noise and broad cortical coverage [38, 49].
sEEG and depth microelectrodes target deep structures and support multi-site sampling across distributed networks [50, 51].
Although penetrating electrodes can cause mechanical injury, edema, or hemorrhage, placements are clinically driven, such as for epilepsy localization, often yielding rich datasets spanning multiple functional networks simultaneously [52, 53]. These recordings provide unparalleled opportunities to link neural dynamics with complex human cognition, behavior, and psychiatric dysfunction [7, 54]. It is important to acknowledge that invasive intracranial recordings are typically obtained from clinical populations such as patients with epilepsy or Parkinson’s disease. While these studies provide critical mechanistic insights, factors including underlying pathology, medication effects, and electrode placement can influence observed neural signals. This context highlights the value and inherent limitations of intracranial research in clinical populations.
Humans possess cognitive abilities like symbolic language and causal reasoning that set them apart from other animals [1, 2]. Key areas are highlighted in which human intracranial recordings have yielded mechanistic insights into human cognition (Table 2).
Adaptive, intelligent human behavior depends on learning from outcomes, evaluating context-dependent value, and updating beliefs about uncertain environments [10, 32, 55]. Reward learning in humans is shaped by internal state, motivational context, episodic memory, social goals, and future planning, making it more complex than in animals [56, 57]. Computational models such as Q-learning, actor-critic, and Bayesian belief-state models have been widely applied to characterize human reward learning [25, 58].
Intracranial recordings in humans reveal rich spatiotemporal signatures of value, RPEs, uncertainty, and expectation across OFC, medial prefrontal cortex, ACC, hippocampus, insula, striatum, and midbrain structures [13, 15, 59]. Widespread cortical representations of reward expectation signals have been observed beyond canonical dopaminergic pathways, and direct recordings of dopaminergic neurons encoding reward expectation and prediction errors have been reported [57]. For example, Gueguen et al. (2021) [21] used iEEG in epilepsy patients and showed broadband gamma activity scaled with outcome valence and inversely with reward expectation, with anatomically dissociable coding of reward and punishment across prefrontal subregions. Collomb-Clerc et al. (2023) [25] found that low-frequency thalamic oscillations tracked expected value during reward and punishment learning, demonstrating thalamic involvement in human value computation. Aquino et al. (2023) [24] recorded single units in pre-supplementary motor area (preSMA) and showed neurons encoded value estimates for each option, linking medial frontal motor areas with computational decision variables. Foundational studies include Li et al. (2016) [19] showing distinct coding of expected value, risk, and experienced value across medial versuslateral OFC, Rutishauser et al. (2015) [60] demonstrating medial temporal lobe (MTL) neurons encoding surprise and conflict, and Saez et al. (2018) [13] observing RPEs in thalamus and OFC.
Theory of mind (ToM) is essential for understanding how humans learn from and about others’ rewards, intentions, and actions [61–63]. Social reward learning, such as predicting others’ behavior, evaluating fairness, or updating beliefs about partners, requires inferring hidden mental states and mapping them onto reward outcomes. Intracranial recordings demonstrate that ToM-related regions, such as temporoparietal junction (TPJ) [61], encode belief states, social outcomes, and inequity, often using signals associated with reward valuation. Jamali et al. (2021) [22] performed single-unit recordings in dmPFC and identified neurons encoding others’ beliefs, including false beliefs, with selective model-switch decoding. Marciano et al. (2023) [27] recorded OFC high-frequency activity during a repeated dictator game, showing encoding of self-reward, partner reward, and inequity type. Together, these findings indicate that social inference engages neural mechanisms closely aligned with reward learning, extending value-based computations into social domains.
Beyond language, intracranial recordings reveal the fine-grained neural dynamics that orchestrate movement. Action selection, motor vigor, and movement adaptation are shaped by RPEs and dopaminergic signals, particularly within cortico-basal ganglia circuits [64–66]. Intracranial recordings during movement in humans, primates, and rodents show how reward-related neuromodulatory dynamics interact with motor control signals, revealing how learning signals influence not just what decisions are made, but how they are executed [67, 68]. These studies demonstrate how value signals guide behavior in real time.
Faces are among the most important social stimuli humans encounter and serve as key sources of reward-related information, signaling trust, threat, affiliation, and social value [69–71]. Efficient reward learning in social environments depends on rapidly extracting and evaluating facial cues. Invasive recordings reveal highly selective neural responses to facial identity, emotion, and social salience in ventral temporal cortex, amygdala, and OFC, regions also central to reward valuation [72]. These findings show how perceptual systems interface with reward circuitry to support learning from socially relevant cues, with implications for disorders involving disrupted social reward processing.
Humans integrate reward with abstract rules, goals, autobiographical memory, and social reasoning, supporting flexible behaviors across diverse contexts [3, 4, 73]. Invasive human intracranial recordings, including ECoG, sEEG, depth microelectrodes, and single-unit recordings, provide access to fast and spatially precise neural signals that are critical for studying these human capacities, particularly in deep and association cortical networks that are poorly sampled by non-invasive approaches [5, 6].
While reward processing plays a crucial modulatory role in shaping cognitive functions, it is not necessarily the dominant organizing principle of human cognition. Alternative theoretical frameworks emphasize that multiple interacting systems, including attention, memory, and executive control, contribute to cognitive flexibility. Reward processing is a key mechanism influencing the integration of cognitive processes. This perspective acknowledges the richness of cognitive architecture beyond reward-centric models.
Reward-related signals play a role in shaping learning and decision making under uncertainty, delay, and social context [3, 74]. Single-neuron and population-level recordings demonstrate that reward-related learning signals are expressed across prefrontal, cingulate, striatal, and thalamic circuits. For example, Gueguen et al. (2021) [21] showed that broadband gamma activity in the prefrontal cortex scaled with outcome valence and inversely with reward expectation, with anatomically dissociable coding of reward and punishment. Collomb-Clerc et al. (2023) [25] reported that low-frequency thalamic oscillations tracked expected value during reward and punishment learning. Aquino et al. (2023) [24] identified neurons in preSMA encoding option-specific value estimates, linking medial frontal motor areas with computational decision variables. These studies illustrate how reward-guided learning is implemented across distributed neural circuits in humans.
Naturalistic behaviors such as language, movement, and navigation provide rich contexts in which reward signals interact with cognition. Interestingly, language can facilitate abstract reward learning by allowing humans to represent and communicate value information symbolically rather than solely through direct reinforcement [69, 75–77]. Intracranial recordings reveal how cortical populations encode phonological, syntactic, and semantic structure with high temporal precision, providing the substrate through which information can be parsed and integrated. High-gamma activity in the superior temporal gyrus encodes phonetic features [75]. The motor cortex reflects articulatory and phonological encoding [45], and temporal lobe single units represent semantic categories and word meanings. Distributed population codes further support context-dependent interpretations of words [78]. These mechanisms support higher-order processing, linking symbolic representations to motivational and decision-making systems.
Reward-related signals also shape motor control and action execution. Intracranial recordings during movement reveal how cortico-basal ganglia circuits integrate value and learning signals to guide action selection, vigor, and adaptation. McCrimmon et al. (2018) [79] demonstrated modulation of broadband gamma activity in human primary motor cortex by stride phase and walking speed. Hell et al. (2018) [80] showed phase-dependent modulation of subthalamic nucleus beta activity during gait, and Louie et al. (2022) [81] reported that chronic ambulatory deep brain recordings encode locomotor state and freezing severity in Parkinson’s disease. These studies highlight how reward-related neuromodulatory dynamics influence not only decisions but also their execution.
In spatial navigation, invasive recordings have revealed neural representations that support goal-directed behavior, memory, and reward-guided learning. Studies have demonstrated place- and view-selective firing in human MTL neurons during virtual navigation [82] and grid-like representations in the entorhinal cortex [43], highlighting conserved spatial coding principles across species. These spatial maps provide a scaffold for integrating reward information, allowing the brain to encode the location and value of goals. More recent work extends these findings to naturalistic settings: medial temporal theta oscillations track both memory and spatial location during immersive virtual navigation [83], linking spatial representations with goal-directed and reward-related behavior.
A key strength of human intracranial recordings is the ability to directly relate neural dynamics to behavior at millisecond timescales. By combining invasive electrophysiology with naturalistic tasks and computational modeling, recent studies demonstrate how reward-related signals interact with internal goals, beliefs, and contextual information to guide behavior in real time. Rather than acting in isolation, reward emerges as a coordinating signal that links perception, learning, decision making, and action across distributed neural circuits.
Understanding how errors in reward or belief processing are coded in the brain has important implications for the development of new diagnostic and rehabilitative treatments for disorders such as depression and obsessive-compulsive disorder (OCD), in which reward processing is disrupted.
Intracranial recordings reveal that psychiatric symptoms emerge from dynamic dysregulation within cortico-striatal-thalamo-cortical loops rather than static dysfunction of isolated regions. In depression, medial prefrontal and orbitofrontal neurons show blunted reward prediction signals and altered theta-gamma coupling, reflecting impaired integration of outcome expectations with ongoing behavior. In OCD, disrupted corticostriatal processing biases behavior toward habitual responding, leading to repetitive actions that persist despite reduced sensitivity to reward outcomes [84]. Continuous recordings allow these patterns to be tracked over time, showing how network interactions fluctuate with mood states or compulsive urges [85]. This level of precision links specific temporal and spectral neural signatures to symptom expression, guiding individualized interventions such as DBS targeting nodes where reward and action signals converge.
Chronic pain involves dynamic fluctuations in both sensory and affective neural circuits, which can be leveraged to improve neuromodulation strategies. DBS has been shown to provide short-term relief for refractory pain disorders. However, pain circuits can adapt to stimulation or fluctuate naturally in chronic pain states. Conventional continuous stimulation that does not adapt to these fluctuations has limited long-term efficacy. Advances in adaptive DBS are critical to appropriately modulate maladaptive responses to painful stimuli. By capturing natural, spontaneous fluctuations in chronic pain using intracranial recordings, Shirvalkar et al. (2023) [85] distinguished transient, acute, and sustained pain states represented in frontal cortical regions, including the ACC and OFC. Recent machine-learning approaches have further demonstrated that chronic pain states can be decoded from sEEG signals, offering a framework for closed-loop, personalized interventions.
Across studies over the past 15 years, human intracranial recordings reveal that components of reward, error, and belief processing operate on timescales of milliseconds to seconds, while learning unfolds over hours to weeks. Similarly, speech comprehension and production operate on millisecond timescales, whereas language acquisition occurs across multiple exposures and social interactions. Motor planning, integration, and execution operate over seconds, while motor learning and rehabilitation span hours to weeks. In disorders such as treatment-resistant depression, chronic pain, or movement disorders such as Parkinson’s disease, neural activity can fluctuate transiently, persist, or progress over longer timescales.
ECoG recordings are typically collected between six and 14 days with continuous video and audio monitoring, which allows for the study of cognitive dynamics across extended timescales. Recording durations may be shorter or longer depending on clinical need. Signals are commonly sampled at ∼1,000 Hz, allowing analysis of frequencies up to ∼500 Hz [86]. In practice, lower sampling rates are often sufficient for many cognitive questions, whereas higher rates are sometimes used to better capture fast transients and high-frequency activity. Signal amplitudes are typically on the order of tens of microvolts (∼50–100 μV), but vary substantially with electrode size, cortical depth, referencing scheme, and local tissue properties. High-frequency activity derived from these signals is frequently used as an index of local neuronal population firing, though the relationship to single-unit spiking is indirect and task dependent.
Typical ECoG arrays include ∼70–100 electrodes arranged in grids or strips, providing broad regional coverage. The coverage is largely based on clinical indication, for seizure localization, so it can be biased and limit generalizability. However, both smaller, high-density arrays and larger clinical montages are increasingly used. The effective spatial resolution of ECoG is on the order of a few millimeters [87], reflecting the scale of local cortical populations, but it can degrade with increased inter-electrode spacing or distance from the cortical surface. Depth sEEG electrodes generally have ∼3 mm inter-contact spacing, though spacing and contact size vary across manufacturers and clinical protocols. iEEG offers relatively high spectral resolution (Figure 2), allowing reliable measurement of high-frequency and neuronal signals critical for studying human cognition.
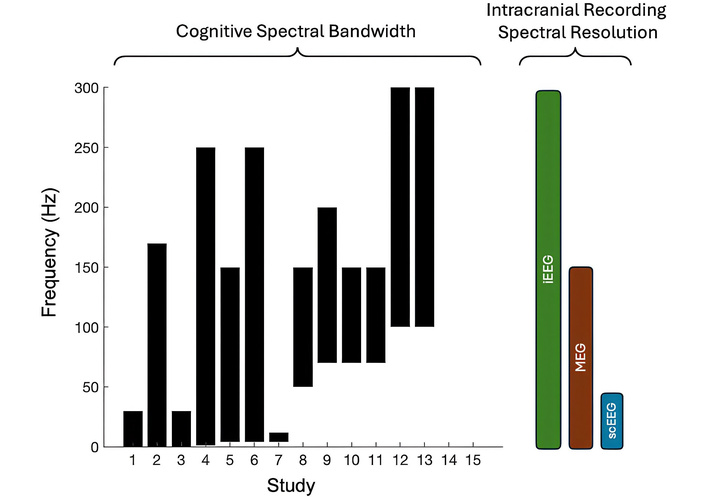
Spectral frequencies assessed in human electrophysiology studies were surveyed. Comparison of spectral bandwidth and resolution of cognition and intracranial recordings. Studies that used local field potentials most often assessed ranges above 50 Hz. Two studies (12 and 13) assessed neuronal firing rates, reflected here as frequencies above 100 Hz. iEEG provides the greatest spectral resolution compared to MEG and scEEG. MEG: magnetoencephalography; scEEG: scalp electroencephalography.
By contrast, non-invasive scEEG offers spatial resolution typically on the centimeter scale (∼5–9 cm), more sensitivity to low-frequency activity (< 40 Hz), and smaller signal amplitudes (∼10–20 μV) [88]. Although scEEG systems can be sampled at high rates and record activity at higher frequencies, these signals are strongly attenuated and susceptible to muscle, eye-movement, and environmental artifacts due to volume conduction and skull filtering, limiting reliable interpretation of fast, localized neural dynamics. These capabilities position intracranial recordings as a unique tool for understanding rapid neural dynamics underlying cognition, while providing a translational bridge to novel interventions in neuropsychiatric and pain disorders [85].
Recent advances in intracranial electrophysiology are rapidly expanding the capacity to understand human cognition through the lens of reward processing with increasingly high spatial and temporal resolution, in more naturalistic contexts, and with devices that are safer, more scalable, and more adaptable.
Three major areas of progress that are particularly relevant for reward-centered models of cognition include: (1) high-density and single-unit recordings that reveal cellular-level value representations, (2) naturalistic and ambulatory recordings that capture reward processing during real behavior, and (3) emerging implantable devices that allow for long-term, closed-loop interrogation of reward circuits. Stereotactically implanted intracranial electrodes, sEEG depth electrodes, and subdural grids and strips remain a gold standard for clinical monitoring and for research on epilepsy, movement disorders, and psychiatric disease [5]. However, traditional clinical electrodes primarily capture local field potentials and lack the spatial resolution needed to resolve how reward, belief, and decision variables are encoded at the level of individual neurons and microcircuits. More recent technologies, such as Neuropixels probes and Neuropixels 2.0, have revolutionized large-scale cellular-resolution recordings in animal models by offering dramatically increased channel density, laminar sampling along the probe shank, and the ability to record from hundreds to thousands of neurons simultaneously [89]. By enabling simultaneous recordings from hundreds to thousands of neurons across cortical layers, these probes offer the potential to directly observe how reward signals are distributed across populations.
Reward learning and decision-making rarely occur in isolation. A major limitation of traditional neuroimaging, such as functional MRI (fMRI), MEG, and traditional EEG, has been the inability to study reward processing during freely moving, real-world activity. Naturalistic intracranial recording paradigms now overcome this barrier. Ambulatory and chronic intracranial systems facilitate high-resolution recordings during gait and locomotion [90], spatial navigation, and memory [83, 90]; sleep and mood fluctuations [91], and social interaction, spontaneous speech, and unconstrained decision-making. Innovation is accelerating the translation of intracranial recording technologies from short-term experimental tools into scalable platforms for long-term study of human cognition. Fully implantable, bidirectional systems now make it possible to simultaneously record and stimulate, allowing causal interrogation of reward-related circuits and adaptive modulation based on neural state [92, 93].
Neural prostheses are evolving technologies designed to restore lost or impaired function by directly interfacing with the brain’s electrical activity. These systems decode neural signals that correspond to intended movements, speech, or cognitive goals, enabling control of external devices such as robotic limbs or speech synthesizers. Early foundational studies demonstrated that neural ensembles in motor areas could be trained to control a computer cursor or robotic arm through imagined or attempted movement signals, providing proof of concept for brain-machine interfaces [94, 95]. More recently, Moses et al. (2021) [96] achieved a groundbreaking milestone by decoding attempted speech from cortical recordings, allowing real-time synthesis of speech in paralyzed individuals, a development that offers profound implications for restoring communication. In addition to decoding motor commands, advancing prosthetic function requires access to internal goal states, motivation, and learning signals that shape behavior over time. Reward-related neural processes play a critical role in this adaptation: reinforcement signals guide error correction, promote skill acquisition, and stabilize control policies [97, 98].
Over the past decade, the National Institutes of Health (NIH) has prioritized funding in areas critical to advancing human intracranial electrophysiology research (Figure 3; Table 4). A substantial portion of resources has been dedicated to understanding language comprehension and production, including speech, reflecting its foundational role in human cognition and communication [96, 99]. Parallel investments have supported research into motor function and rehabilitation, targeting not only voluntary movement but also gait and other complex motor behaviors affected by neurological disease [97]. Notably, there has been growing emphasis on mood and psychiatric disorders, particularly treatment-resistant depression and OCD. These conditions are increasingly understood as disorders of reward processing, valuation, and motivational control, making them well-suited to investigation using intracranial recordings and neuromodulation [84]. These funding trends highlight a strategic shift toward integrating cognitive neuroscience, reward-based models of behavior, and translational neurotechnology. Note, the table is not comprehensive and is intended to illustrate funding trends rather than provide an exhaustive record.
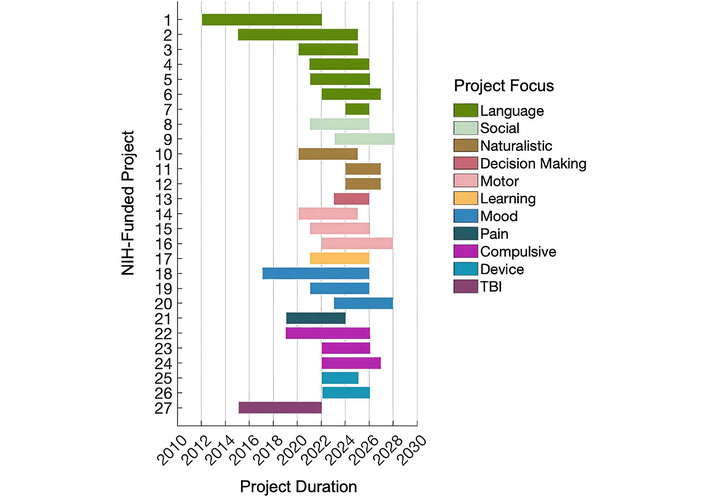
Timeline of past and ongoing NIH-funded projects using human intracranial recordings. Projects are grouped into 11 research focus areas, each represented by a distinct color. Horizontal bars indicate the duration of each project, from start to end year.
Selected NIH-funded projects over the past decade employing human electrophysiology.
| Focus | Title | Project start year | Project end year | Grant | Study section | Institute/center |
|---|---|---|---|---|---|---|
| Language | Functional organization of the superior temporal gyrus for speech perception | 2012 | 2022 | R01 | Auditory System | NIDCD |
| Language | Engaging new cognitive and motor signals to improve communication prostheses | 2015 | 2025 | R01 | BNVT | NIDCD |
| Language | The neural coding of speech across human languages | 2020 | 2025 | U01 | ZNS1-SRB-K | NINDS |
| Language | A Pilot Clinical Trial for Speech Neuroprosthesis | 2021 | 2026 | U01 | Special Emphasis Panel | NIDCD |
| Language | Single-neuron population dynamics in human speech motor cortex for a speech prosthesis | 2021 | 2026 | U01 | ZDC1-SRB-E(30) | NIDCD |
| Language | Studying semantic processing during language comprehension in humans at the single-cellular level | 2022 | 2027 | R01 | Language and Communication | NIDCD |
| Language | A fully implantable speech neuroprosthesis | 2024 | 2026 | UG3 | Special Emphasis Panel | NINDS |
| Social | An Integrated Single-Neuronal, Population-, Local Network- and Stimulation-Based Prefrontal Investigation of Human Social Cognition | 2021 | 2026 | U01 | ZNS1-SRB-P(01) | NINDS |
| Social | Neural Mechanisms of Social Attribution from Faces | 2023 | 2028 | R01 | Human Complex Mental Function | NIMH |
| Naturalistic | Neurostimulation and Recording of Real World Spatial Navigation in Humans | 2020 | 2025 | U01 | ZNS1-SRB-K(20) | NINDS |
| Naturalistic | Synchronized neuronal and peripheral biomarker recordings in freely moving humans | 2024 | 2027 | R61 | Special Emphasis Panel [ZMH1 ERB-E(01)] | NIMH |
| Naturalistic | Capturing Autobiographical memory formation in People moving Through real-world spaces Using synchronized wearables and intracranial Recordings of EEG | 2024 | 2027 | R61 | Special Emphasis Panel [ZMH1 ERB-E(01)] | NIMH |
| Decision Making | Identifying the neural mechanisms of goal-directed decision-making in Parkinson’s disease using closed-loop deep brain stimulation | 2023 | 2026 | F32 | ZMH1-ERB-E(03) | NIMH |
| Motor | Cortical basal ganglia network dynamics during human gait control | 2020 | 2025 | U01 | ZNS1-SRB-K(20) | NINDS |
| Motor | Cortical basis of complex motor sequences in humans for neural interfaces | 2021 | 2026 | U01 | ZNS1-SRB-P(02) | NINDS |
| Motor | Motor Recovery through Plasticity-Inducing Cortical Stimulation | 2022 | 2028 | UH3 | Neurological Sciences and Disorders C | NINDS |
| Learning | Mapping Algorithmic State Space in the Human Brain | 2021 | 2026 | U01 | ZNS1-SRB-P(01) | NINDS |
| Mood | Deep Brain Stimulation for Depression Using Directional Current Steering and Individualized Network Targeting | 2017 | 2026 | UH3 | ZNS1-SRB-E(10)1 | NINDS |
| Mood | Invasive decoding and stimulation of altered reward computations in depression | 2021 | 2026 | R01 | ZMH1-ERB-Q(03) | NIMH |
| Mood | Intracranial Investigation of Neural Circuity Underlying Human Mood | 2023 | 2028 | R01 | Human Complex Mental Function | NIMH |
| Pain | Technology development for closed-loop deep brain stimulation to treat refractory neuropathic pain | 2019 | 2024 | UH3 | ZNS1-SRB-T(32) | NINDS |
| Compulsive | Responsive Neurostimulation for Loss of Control Eating | 2019 | 2026 | UH3 | ZNS1-SRB-E(10) | NINDS |
| Compulsive | The role of distributional reinforcement learning in human neurons during impulsive choices | 2022 | 2026 | R01 | Human Complex Mental Function | NIMH |
| Compulsive | Investigating the role of nucleus accumbens delta signaling in loss of control | 2022 | 2027 | R01 | ZMH1-ERB-Q(03) | NIMH |
| Device | Advancing epilepsy diagnosis with flexible, high-resolution thin-film electrodes | 2022 | 2025 | UG3 | Neurological Sciences and Disorders C | NINDS |
| Device | An Ecosystem of Technology and Protocols for Adaptive Neuromodulation Research in Humans | 2022 | 2026 | U01 | ZNS1-SRB-O(18) | NINDS |
| TBI | Central thalamic stimulation for traumatic brain injury (TBI) | 2015 | 2022 | UH3 | ZNS1-SRB-E(06) | NINDS |
Projects are categorized by research focus, with project title, start and end year, grant mechanism, study section, and institute/center listed. This table is not comprehensive. BNVT: Bioengineering of Neuroscience, Vision, and Low Vision Technologies; NIH: National Institutes of Health.
Despite advances, significant technical and clinical challenges remain. Signal stability over time is variable, with electrode encapsulation, tissue response, and hardware degradation affecting long-term performance. Decoding algorithms often require extensive calibration and may not generalize well across tasks, contexts, or individuals. Access to internal cognitive states remains methodologically complex. The precise contribution of reward-related processes to adaptive prosthetic control requires further empirical validation. Intracranial recordings are typically obtained from patients with neurological or psychiatric disorders, which may limit generalizability. Safety considerations, surgical risk, regulatory requirements, and scalability remain substantial barriers to widespread implementation. While funding trends reflect growing interest in neuromodulation and cognitive neurotechnology, these investments do not eliminate the need for careful validation, longitudinal outcome studies, and rigorous ethical oversight. The development of neurotechnology remains iterative and contingent on overcoming substantial methodological, biological, and clinical limitations.
As intracranial recording and neuroprosthetic technologies advance, ethical considerations become increasingly important. The privacy of neural data is paramount, given that brain signals can reveal deeply personal thoughts and intentions. Robust protocols must ensure informed consent, data security, and transparency regarding how neural information is used. Informed consent requires explicit disclosure of risks, including privacy breaches, cybersecurity vulnerabilities, and potential effects on identity and agency, and safeguards against therapeutic misconception, particularly in neurologically vulnerable populations [100, 101]. Neural data governance presents distinct challenges, as intracranial recordings can reveal intentions, preferences, and cognitive states. Dual-use risks are critical considerations, as neurotechnologies developed for therapy may enable cognitive manipulation or surveillance when combined with AI-driven closed-loop systems [102]. Finally, posttrial responsibilities, including continued device access, maintenance, and long-term care, represent a critical ethical obligation that should be explicitly planned and reported.
Human intracranial electrophysiology is entering an exciting phase, where advances in technology and clinical applications are beginning to reveal how reward processing emerges from distributed, temporally coordinated neural circuits. Single-cell and population recordings provide precise insights into activity patterns across distinct frequency bands. These mechanistic insights lay the groundwork for interventions such as adaptive neurostimulation and targeted therapies, while future integration with molecular- and micro-level recording techniques promises to expand our understanding of the neural computations underlying cognition and behavior.
ACC: anterior cingulate cortex
DBS: deep brain stimulation
dmPFC: dorsomedial prefrontal cortex
ECoG: electrocorticography
fMRI: functional magnetic resonance imaging
iEEG: intracranial electroencephalography
MEG: magnetoencephalography
MTL: medial temporal lobe
OCD: obsessive-compulsive disorder
OFC: orbitofrontal cortex
preSMA: pre-supplementary motor area
RPEs: reward prediction errors
scEEG: scalp electroencephalography
sEEG: stereo-electroencephalography
STS: superior temporal sulcus
ToM: theory of mind
TPJ: temporoparietal junction
The author sincerely thanks Lawrence Hunt and Ignacio Saez for their thoughtful and constructive comments on the Oxford thesis work that formed the basis of this review. Deep gratitude is also extended to Mark Woolrich, Huiling Tan, Chet Gohil, Mats van Es, Julio Chapeton, Zane Xie, and Ashwin Ramayya for their insightful perspectives, which improved the conceptual development of this work. While this work emphasizes the unique insights afforded by human intracranial recordings, increasingly even more precise approaches targeting circuits at the level of specific cell types and receptors are emerging as powerful ways to unravel and modulate brain processes.
APST: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing—original draft, Writing—review & editing. The author read and approved the submitted version.
The author declares no conflicts of interest related to this work.
Not applicable.
Not applicable.
Not applicable.
No new data were generated or analyzed in support of this review. All cited data are publicly available in the referenced publications.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 2662
Download: 20
Times Cited: 0
Hoong Wei Gan ... William P. Whitehouse
Jerónimo Auzmendi, Alberto Lazarowski
Toshimitsu Suzuki ... Kazuhiro Yamakawa
Rodolfo Cesar Callejas-Rojas ... Ildefonso Rodriguez-Leyva
Anwaar M. Bennour ... Heba A. El-Zawawi
