Affiliation:
1University Centre for Research & Development (UCRD), Chandigarh University, Mohali 140413, Punjab, India
Email: kumar.ajay1250@gmail.com
ORCID: https://orcid.org/0000-0002-5312-9823
Affiliation:
2Department of Physiotherapy, Guru Jambheshwar University of Science & Technology, Hisar 125001, Haryana, India
Email: amitapuniaphysio@gmail.com
ORCID: https://orcid.org/0000-0003-2397-8282
Explor Neurosci. 2026;5:1006130 DOI: https://doi.org/10.37349/en.2026.1006130
Received: November 18, 2025 Accepted: February 13, 2026 Published: March 23, 2026
Academic Editor: Dirk M. Hermann, University of Duisburg-Essen, Germany
The article belongs to the special issue Current Approaches to Malignant Tumors of the Nervous System
Malignant tumors of the nervous system, such as gliomas, medulloblastomas, and neuroblastomas, pose a greater clinical challenge due to their aggressive and invasive nature and their resistance to current treatment options. The blood-brain barrier (BBB) impairs the delivery of therapeutic agents, which is associated with poor prognosis. The natural flavonoid fisetin has demonstrated potential for cancer treatment by regulating major cancer-related signaling pathways, including PI3K/Akt/mTOR, NF-κB, and MAPK. Preclinical studies suggest that fisetin induces apoptosis, suppresses tumor invasion, and reduces malignancy in glioma, medulloblastoma, and neuroblastoma models. However, evidence for fisetin’s effectiveness remains preclinical and in vitro, with no clinical trials in humans to date. One solution to this challenge is to use nanotechnology-based delivery systems to increase fisetin’s stability and solubility and facilitate its crossing of the BBB, thereby enhancing its therapeutic efficacy. Such advancements have made fisetin a promising option for neuro-oncology treatment. Further clinical trials are needed to assess the safety, efficacy, and effectiveness of fisetin in combination therapies. When conjugated with nanotechnology-based delivery, fisetin may enable a digital transformation in treatment outcomes for patients with malignant nervous system tumors.
Malignant neoplasms of the nervous system are a diverse group of tumors arising from various cellular lineages in the central and peripheral nervous systems [1]. Among these, gliomas, medulloblastomas, and neuroblastomas are the major subgroups, each characterized by distinct clinical, histopathological, and molecular features that contribute significantly to morbidity and mortality worldwide [2]. Glioblastoma is among the most malignant forms of brain tumors, with a dismal prognosis [3]. The most common subtype is isocitrate dehydrogenase (IDH)-wildtype glioblastoma, characterized by rapid progression and treatment resistance. Pediatric high-grade gliomas, although rare, exhibit characteristics of glioblastoma [4, 5]. Glioblastoma is the most common and fatal primary brain tumor in adults, characterized by aggressive growth, extensive invasiveness, and genetic/epigenetic heterogeneity [6]. Glioblastomas are characterized by necrosis, microvascular proliferation, and resistance to apoptosis; yet patients have a median survival of approximately 15 months, even with aggressive multimodal treatment, including maximal resection, radiotherapy, and temozolomide chemotherapy [7]. Medulloblastoma is the most frequent malignant brain tumor in children, and survivors often suffer from long-term neurocognitive and endocrine handicaps despite aggressive treatment strategies such as surgery, irradiation, and high-dose chemotherapy [8]. Medulloblastoma is categorized into four primary subgroups: WNT, SHH, Group 3, and Group 4 [9]. Distinct molecular profiles and clinical features characterize these subgroups. The WNT subgroup is associated with a favourable prognosis, whereas the SHH subgroup can be subdivided into molecular categories, each with distinct prognostic significance [10]. Recent advances in molecular subtyping have led to more accurate prognostic and therapeutic strategies, but resistance and relapse remain critical issues that require novel targeted therapies [11]. Tumors derived from sympathetic nervous system progenitors, such as neuroblastoma, exhibit heterogeneous clinical behavior, with high-risk patients often displaying chemotherapy resistance associated with genomic instability and tumor heterogeneity [12]. These tumor types exhibit the blood-brain barrier (BBB), which limits drug delivery and treatment; these challenges can be overcome through strategies that involve drug modifications, BBB disruption, and nanotechnology to enhance therapeutic efficacy [13–15]. In the last few decades, natural products have been recognized in oncology for their multitarget activity, low toxicity, and modulation of molecular pathways involved in carcinogenesis [16–19]. Because of their antioxidative, anti-inflammatory, immunomodulatory, and anticancer effects on tumor development and chemotherapeutic resistance, flavonoids could be promising therapeutic agents in anticancer therapy [20–23].
Fisetin (3,3',4',7-tetrahydroxyflavone), a typical flavonoid with a broad and highly active pharmacological profile, has been shown to be effective against several types of cancer, including human malignant nervous system tumors [24–27]. Fisetin, a bioactive ingredient abundant in strawberries, apples, grapes, cucumbers, and onions, modulates oncogenic signaling pathways [24, 28]. Fisetin also helps reduce oxidative stress in brain tumors, lowers neuronal apoptosis, and protects cognitive function and neurological behavior [29, 30].
Due to fisetin’s low water solubility, its metabolic conjugation and, consequently, low bioavailability, along with poor BBB penetration, limit its therapeutic application [28, 31–34]. Liposomes and polymeric or solid lipid nanoparticles (SLNs) have been used to overcome these limitations, thereby improving stability, enabling controlled drug delivery, and facilitating effective brain targeting in preclinical studies [35]. The lack of clinical studies limits its current application in clinical settings, making it an interesting but not yet ready-for-translation pharmaceutical for malignant nervous system malignancies. Given the complex aetiology of nervous system tumors and the limitations of current therapies, fisetin’s ability to modulate multiple molecular targets and to employ nanodelivery strategies suggests it is a promising therapeutic option in neuro-oncology. In particular, this narrative review explores the status of nervous system malignant tumors, fisetin’s biochemical and pharmacological characterization, including its structural-activity relationships, and nanotechnology to address drug challenges for clinical application (Figure 1).
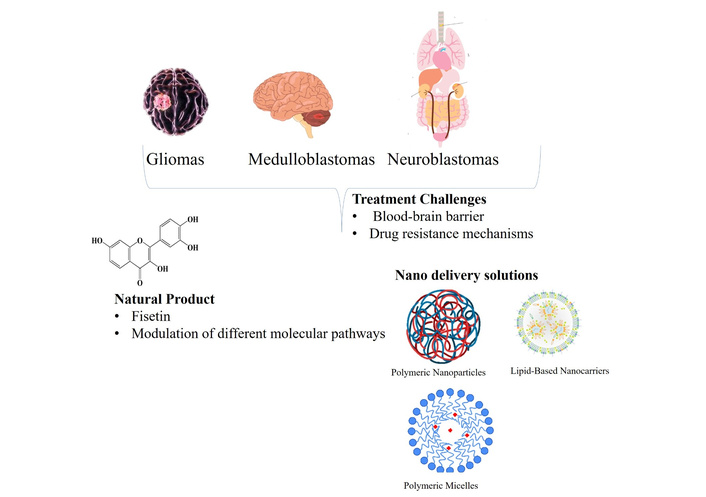
Overview of malignant nervous system tumors, their molecular characteristics, treatment challenges, and the therapeutic potential of fisetin enhanced by nanodelivery.
This narrative review identified relevant peer-reviewed articles on the chemistry, pharmacological properties, and metabolism of fisetin, as well as nanodelivery systems, from literature databases including PubMed, Scopus, Google Scholar, and Web of Science. Keywords such as “fisetin,” “pharmacokinetics,” “antioxidant activity,” “anticancer mechanisms,” “nanoparticles,” and nervous system tumors were used. Relevant review articles, original research papers, and clinical trials published up to 2025 were included. Articles were selected based on rigorous research design, their relevance to fisetin’s potential therapeutic use in nervous system cancers, and the clarity of the available data on molecular mechanisms and delivery challenges. Inclusion criteria were studies that employed both in vitro and in vivo models, with priority given to those with sample sizes greater than 10 in in vivo studies. Articles were selected to ensure representation of diverse species, including murine models and human cell lines. Data on chemical structure, in vivo metabolism, pharmacodynamics/pharmacokinetics, and developments in nanotechnology-based formulations were collected. The information was integrated to highlight biochemical associations, pharmacological applications, manufacturing strategies, and potential hurdles for fisetin in clinical settings. The focus was on a multidisciplinary dataset to inform future research on neuro-oncology therapies involving fisetin.
Fisetin (3,3',4',7-tetrahydroxyflavone) is a plant flavonol from the flavonoid group of polyphenols found in significantly high levels in various fruits and vegetables and is one of the most well-known compounds of this group (Figure 2). The molecular formula for fisetin is C15H10O6, and the molecular weight is 286.24 g/mol, with different properties shown in Table 1 [36]. The chemical structure of fisetin consists of two aromatic rings (A and B) connected by a heterocyclic pyran ring (C), typical of flavonoids in general. The antioxidant potential was seen due to the hydroxylation at positions 3, 7, 3', and 4' that allowed for free radical scavenging ability, together with chelation of metals, as well as regulation of redox-sensitive signaling pathways [28, 37, 38].
Summary of the important chemical and physical characteristics of fisetin in pharmaceutical applications.
| Property | Value/Description |
|---|---|
| Chemical identity | Name: fisetinIUPAC name: 2-(3,4-dihydroxyphenyl)-3,7-dihydroxychromen-4-one |
| Physical state | Yellow crystalline powder |
| Molecular characteristics | Formula: C15H10O6Molecular mass: 286.24 g/mol |
| Thermal properties | Melting point: 330°CBoiling point: 599.4°C (at 760 mm Hg) |
| Density and solubility | Density: 1.688 g/mLSolubility: Poorly soluble in water (10.45 μg/mL); soluble in ethanol (5 mg/mL), highly soluble in DMSO (30 mg/mL) at 25°C |
| Chemical behavior | LogP (partition coefficient): 3.2pKa (acidity constant): 7.42 |
| Flammability | Flash point: 233°C |
DMSO: dimethyl sulfoxide.
The tetrahydroxy functional groups are effective hydrogen-bond donors that interact with proteins, enzymes, and nucleic acids, a characteristic of fisetin’s multitarget bioactivity. Its structural composition and purity have been characterized using spectroscopic methods, such as nuclear magnetic resonance (NMR) and mass spectrometry, to confirm its identity and enable direct synthetic modification [39]. It has been found that fisetin exhibits favorable biological properties but low aqueous solubility (10 µg/mL) and moderate lipophilicity (logP ≈ 3.2), resulting in reduced bioavailability and challenging scale-up formulation. It had a crystalline form that remained stable above 330°C, with a melting point confirming its physical-chemical stability, a crucial quality in drug development [40, 41].
Fisetin’s high dietary content, including strawberries, apples, persimmons, grapes, onions, and cucumbers, has been well documented [24]. The standard isolation procedure consists of extracting the plant matrix with a solvent (methanol, ethanol, or acetone) under controlled conditions to ensure compound integrity [39]. Further purification is carried out using chromatographic methods such as high-performance liquid chromatography (HPLC), preparative thin-layer chromatography (PTLC), and column chromatography to isolate pure fisetin to pharmaceutical-grade quality, as shown in Figure 3 [39].
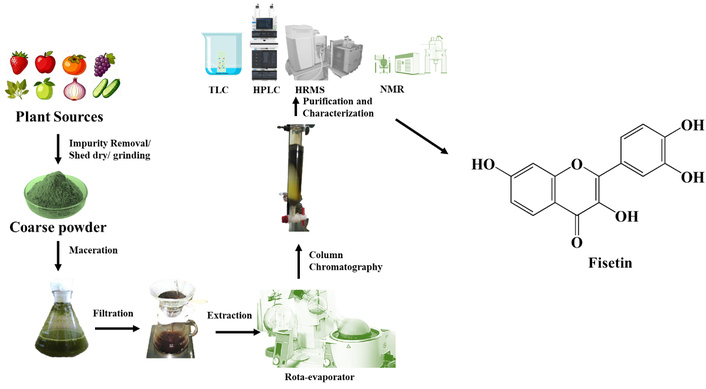
Gradual demonstration of the extraction of fisetin in the diet, purification by chromatographic methods, quality control, and biotechnological and synthetic manufacturing methods. TLC: thin-layer chromatography; HPLC: high-performance liquid chromatography; HRMS: high-resolution mass spectrometry; NMR: nuclear magnetic resonance.
Eliminating chlorophyll, wax, and other polyphenols that may confound the lectin assay is achieved through purification, followed by monitoring of samples using UV-Vis spectroscopy and mass spectrometry to ensure high quality [42]. In biotechnological production, including microbial fermentation and plant cell cultures, there is significant potential to produce fisetin sustainably to address issues of intermittent crop harvests and inefficient extraction [43]. Fisetin analogues have also been synthesised by chemical methods, with improved solubility and biological activity, providing scalable methods for drug discovery [44].
Fisetin has broad pharmacological potential, with potent antioxidant, anti-inflammatory, neuroprotective, and anticancer activity (Figure 4) [24, 26, 32, 38]. The major antioxidant agent directly scavenges reactive oxygen species (ROS) and activates the human endogenous antioxidant defense system by activating nuclear factor erythroid 2-related factor 2 (Nrf2), which promotes the expression of phase II detoxifying enzymes, such as heme oxygenase-1 (HO-1) [28]. This reduces oxidative stress, an important contributor to the development of nervous system tumors. Fisetin has anti-inflammatory properties that have been ascribed to its ability to block the nuclear factor-kappa B (NF-κB) pathway, resulting in the inhibition of the expression of pro-inflammatory cytokines, including tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-6, as well as the cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), which suppress tumor-promotive inflammatory diseases [29, 30, 33]. The neuroprotective properties include reducing glutamate-induced excitotoxicity and preserving mitochondrial function, which are especially important in cancerous diseases of neural tissue, where neural tissue preservation is essential [32]. The compound fisetin has been shown to significantly enhance anticancer activity against nervous system tumors by regulating multiple cancer-promoting signaling pathways. This affects the phosphatidylinositol 3-kinase/protein kinase B/mechanistic (mammalian) target of rapamycin (PI3K/Akt/mTOR) pathway, leading to reduced cell growth and increased cell apoptosis by decreasing phospho-Akt and mTOR levels, and increasing pro-apoptotic protein Bax and decreasing anti-apoptotic protein Bcl-2 [29, 30]. Fisetin modulates extracellular signal-regulated kinase (ERK) 1/2 signaling, thereby inhibiting tumor cell proliferation and promoting apoptosis [25, 27]. Fisetin prevents tumor invasion and metastasis through the inhibition of epithelial-mesenchymal transition (EMT), the expression of E-cadherin, and the expression of the matrix metalloproteinases (MMP-2 and MMP-9), which are required to degrade the extracellular matrix (ECM) [28]. This anti-invasive effect of gliomas and neuroblastomas is important given their aggressive invasiveness and metastatic potential. Table 2 summarizes key preclinical studies examining fisetin’s biological effects in central nervous system (CNS) tumor models. Such multitarget effects make fisetin an attractive candidate for the treatment of malignant tumors, including those of the nervous system.
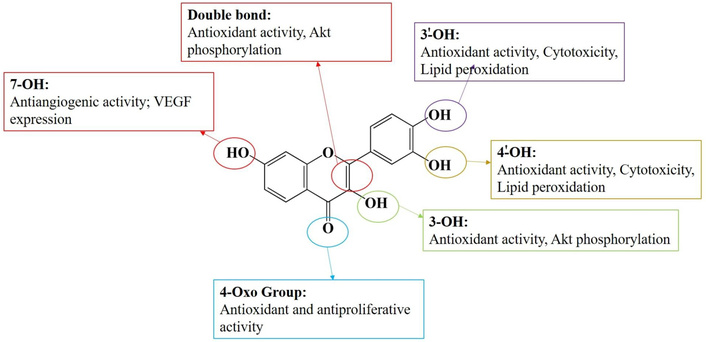
Pharmacological activity of fisetin based on its biochemical structure. Akt: protein kinase B; VEGF: vascular endothelial growth factor.
Summarizes key pharmacological studies examining fisetin’s biological effects in CNS tumor models.
| Model | Dose/Formulation | Main findings | Reference |
|---|---|---|---|
| U-138 MG glioblastoma cells | Fisetin (FIS) & FIS-PLGA NPs; 25 µM and 50 µM |
| [45] |
| LN229 glioblastoma cells | 40–80 µM |
| [46] |
| T98G glioblastoma cells | 1–500 µM |
| [47] |
| GBM8401 glioma cells | Fisetin (10–40 µM) |
| [48] |
| U-87 MG glioblastoma cells; EA.hy926 endothelial cells | Fisetin + cisplatin co-loaded liposomes (DOPC/chol/PEG, ~ 1.7% fisetin, ~ 0.8% cisplatin loading); fisetin-loaded liposomes (1.2 mg/mL fisetin) |
| [49] |
| BV2 cells; Spinal Cord Injury (SCI) Sprague-Dawley Rat Model | Fisetin (25–50 µM)Fisetin-treated; fisetin (40 mg/kg) + autophagy inhibitor [chloroquine (20 mg/kg)] (oral dose) |
| [50] |
CNS: central nervous system; PLGA: poly(lactic-co-glycolic acid); NPs: nanoparticles; IC50: half-maximal inhibitory concentration; ADAM9: A disintegrin and metalloproteinase 9; ERK: extracellular signal-regulated kinase; DOPC: 1,2-dioleoyl-sn-glycero-3-phosphocholine; PEG: polyethylene glycol; AMPK: AMP-activated protein kinase; mTOR: mechanistic (mammalian) target of rapamycin; IL-6: interleukin-6; TNF-α: tumor necrosis factor-alpha.
Fisetin has potent pharmacodynamic properties, whereas poor pharmacokinetic profiles, such as low water solubility, rapid metabolism, and low bioavailability, limit its use [27]. Fisetin, when given orally, is extensively metabolised by phase II enzymes in the liver and intestine, with extensive first-pass metabolism [24]. The formation of hydrophilic metabolites following glucuronidation and sulfation also allows their excretion via the kidneys and bile. This metabolic conversion results in a pronounced reduction in free plasma fisetin levels; however, it does not abolish all biological activity, as some conjugated metabolites remain therapeutically active [41].
Studies in animals have shown that fisetin has poor oral bioavailability due to its low aqueous solubility, low permeability, and rapid clearance. Its half-life is short, and the dose is high or repeated for adequate blood levels [51]. Fisetin’s lipophilicity enables it to cross the BBB to some extent, but not sufficiently to achieve effective concentrations, indicating a need for better delivery systems [31].
Nanotechnology-based delivery formulations, including polymeric nanoparticles, liposomes, and SLNs, have substantially enhanced the solubility, stability, and brain permeability of fisetin. These nanoformulations exhibit sustained release kinetics, passive targeting, and low systemic toxicity, resulting in improved pharmacokinetic/pharmacodynamic profiles in preclinical models of CNS tumors [35].
Fisetin has been shown to have anticancer effects on nervous system tumors, including gliomas, medulloblastomas, and neuroblastomas [32]. Antitumor activity is mainly due to the modulation of key oncogenic signaling pathways, including the PI3K/Akt/mTOR pathway, leading to inhibition of cellular proliferation and induction of apoptosis [30]. Fisetin also suppresses NF-κB activation, reduces anti-apoptotic proteins, and drives programmed cell death [52]. Moreover, it modulates mitogen-activated protein kinase (MAPK) signaling and inhibits EMT, thereby reducing tumor cell motility and invasiveness [53, 54]. It possesses anti-angiogenic activity by inhibiting vascular endothelial growth factor (VEGF) expression and by interrupting tumor energetics through metabolic reprogramming [55]. Fisetin regulates several oncogenic signaling pathways, including PI3K/Akt/mTOR, DNA damage response, and apoptosis. A summary of these effects is provided in Table 3, which describes the specific molecular mechanisms and biological responses observed across various tumor types. It is crucial to emphasize that, although there are encouraging results, fisetin has not yet undergone clinical trials in humans; thus, it remains a preclinical candidate for cancer treatment.
FIS mechanism of action and biological effects.
| Mechanism | FIS’s action | Biological effect | Tumor types affected | Reference |
|---|---|---|---|---|
| Cell cycle modulation | Induces G1/S arrest | Modifies cell cycle distribution | Glioblastoma (U-138 MG) | [45] |
| Apoptosis induction | Activates caspases-3, -8, and -9 | Increases apoptosis in glioblastoma cells | ||
| Nanoparticle encapsulation | Encapsulated in PLGA nanoparticles (FIS-PLGA-NP4) | Enhances stability and drug loading, cytotoxicity improvement | ||
| Radiostability | Exposure to ionizing radiation (25 kGy) | Maintains structural integrity, suitable for sterilization | ||
| Combination therapy | Synergistic effect with chemotherapeutics | Enhances efficacy and reduces resistance | ||
| DNA damage | Induces DNA strand breaks, measured by alkaline comet and γH2AX assay | Initiates genotoxicity and the DNA damage response via p53 signaling | Glioblastoma | [46] |
| Apoptosis induction | Induces apoptosis through flow cytometry analysis | Increases cell death through apoptosis, thereby lowering overall cell survival | ||
| Senescence reduction | Reduces senescence induced by TMZ | Decreases the number of senescent cells, thereby increasing apoptosis | ||
| DNA repair inhibition | Inhibits DNA repair pathways such as BER | Increases DNA damage, thereby increasing cell death | ||
| Enhancement of chemotherapy | Enhances the genotoxic effect of TMZ | Increases the efficiency of temozolomide by enhancing cell death and inhibiting senescence | ||
| Cell proliferation inhibition | Inhibits cell proliferation in T98G cells | Inhibits cell viability and growth in MTT assays as indicated by a lower IC50 in T98G cells | Glioblastoma (T98G cells) | [47] |
| Apoptosis activation | Induces apoptosis in T98G cells via caspases 3, 8, 9, and Bax | Enhances apoptotic indices by assessing caspase activation, mitochondrial destabilisation, and DNA fragmentation | ||
| Cytotoxicity comparison | More cytotoxic in T98G cells than carmustine (positive control) | Causes cell death and cytotoxicity at lower concentrations in T98G cells than carmustine | ||
| Gene expression modulation | Increases CASPASE-3, CASPASE-9, BAX, and decreases BCL-2 | Regulates the expression of genes involved in apoptosis, thereby promoting apoptosis | ||
| Cell migration inhibition | Suppresses glioma cell migration at non-cytotoxic concentrations | Reduces the migration and motility ability of malignant cells | Glioma (GBM8401 cells) | [48] |
| Cell invasion suppression | Inhibits invasion through the extracellular matrix | Inhibits the limits of the invasion potential without causing cytotoxicity | ||
| ADAM9 downregulation | Decreases ADAM9 protein and mRNA expression | Insulates glioma development and invasion-related proteins | ||
| ERK1/2 signaling modulation | Induces sustained phosphorylation of ERK1/2 | ERK1/2 is activated, leading to the inhibition of the ADAM9 expression | ||
| ERK1/2-dependent anti-invasive effect | ERK1/2 inhibition (U0126 or siERK) reverses FIS’s effects | Elevates the ERK1/2 as a requisite mediator of the anti-invasive effect of FIS | ||
| Tumor microenvironment modulation | Alters the tumor microenvironment, affects autophagy and apoptosis pathways | Enhances a better treatment response and reduces treatment resistance | Glioblastoma, glioma | [49] |
| Active targeting | Modification with cRGD peptide | Penetrates the blood-brain barrier to reach glioma cells | Glioblastoma, glioma GL261 or U87-MG cells | [56] |
| Redox-responsive release | Disulfide bond-containing nanoparticles | Breaks down under the condition of increased glutathione, thus, liberating FIS and doxorubicin | ||
| Enhancement of drug uptake | FIS inhibits P-glycoprotein (P-gp) | Modifies the cellular localization of doxorubicin and other chemotherapeutic agents | ||
| Cell cycle arrest | Induction of G2/M phase arrest | Prevents the multiplication and movement of glioma cells | ||
| Apoptosis promotion | Activation of the caspase cascade | Causes oligonucleotide apoptosis via exogenous and endogenous mechanisms | ||
| Synergistic effects with DOX | Combination with DOX-loaded nanoparticles | A synergistic increase in tumor suppression and a decrease in drug resistance | ||
| Cell proliferation | Decreases cell proliferation in both TMZ-sensitive and TMZ-resistant cells | Reduced cell viability and growth inhibition | Glioblastoma (TMZ-resistant T98G23 and TMZ-sensitive A172 cells) | [57] |
| Cell migration | Inhibits migration in TMZ-treated GB cells, enhancing the effects of TMZ | Suppression of the migratory behavior of glioblastoma cells | ||
| Combination therapy | TMZ + FIS combination treatment | Enhanced chemotherapeutic efficacy, overcoming resistance in glioblastoma models | ||
| PI3K/Akt/CREB pathway | Activates PI3K/Akt/CREB signaling | Promotes neuronal survival and neuroprotection | Neurodegenerative disorders | [58] |
FIS: fisetin; PLGA: poly(lactic-co-glycolic acid); NP: nanoparticle; γH2AX: gamma H2A histone family member X; TMZ: temozolomide; IC50: half-maximal inhibitory concentration; ADAM9: A disintegrin and metalloproteinase 9; ERK: extracellular signal-regulated kinase; BER: base excision repair; MTT: methylthiazolyldiphenyl-tetrazolium bromide; cRGD: cyclic arginine-glycine-aspartic acid (peptide); DOX: doxorubicin; PI3K: phosphatidylinositol 3-kinase; Akt: protein kinase B; mTOR: mechanistic (mammalian) target of rapamycin. CREB: cAMP response element-binding protein.
Fisetin induces apoptosis and cell cycle arrest through intricate molecular pathways that determine its antitumor activity against CNS tumors such as gliomas, medulloblastomas, and neuroblastomas (Figure 5) [59, 60]. Fisetin-induced apoptosis is mediated by activation of an intrinsic mitochondrial pathway, leading to the initiation of the caspase cascade, a key determinant of programmed cell death that removes cancerous cells [60]. The chemosensitization effect of fisetin, a plant flavonoid, against cisplatin was evaluated via in silico and in vitro approaches. Preclinical data revealed that fisetin disrupts the mitochondrial membrane potential (MMP) and induces cytochrome c release to the cytosol, an essential apoptogenic factor. This event also initiates the activation of initiator caspase-9, followed later by executioner caspases-3 and -7, resulting in DNA fragmentation and cell death [28]. Fisetin regulates apoptosis by suppressing antiapoptotic factors, such as Bcl-2 proteins and hTERT, and activating proapoptotic factors, such as Bak/Bax, in tumor cells, thereby promoting apoptosis [59]. It downregulates the NF-κB pathway, which is involved in cell survival and chemoresistance, thereby promoting an apoptotic response [52]. The cell cycle regulatory effects of fisetin are linked to its antioxidant properties, as oxidative stress plays an essential role in cell cycle dysregulation and tumor development [61]. Fisetin scavenges ROS and reduces oxidative stress, allowing defective cells to reestablish multiple cell cycle checkpoints and preventing damaged cells from entering mitosis [62]. Furthermore, the ability of fisetin to induce cell cycle arrest and apoptosis is not confined to a single tumor type but has been observed across other models, including glioblastoma, an extremely aggressive brain tumor [27, 63]. In addition to inducing apoptosis, fisetin arrests the cell cycle at specific checkpoints, primarily G2/M or S phases, preventing uncontrolled proliferation of cancer cells. It induces the cyclin-dependent kinase inhibitors p21 and p27 while inhibiting cyclins and CDKs required for cell cycle progression. This cell cycle control mechanism restricts tumor growth and is one component of fisetin’s anti-tumor action [64, 65]. As shown in Table 3, fisetin promotes apoptosis in various tumor cells by activating caspases and arresting the cell cycle.
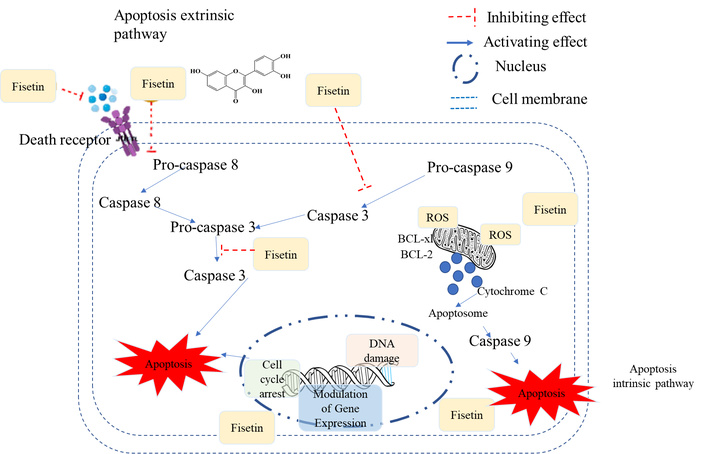
Apoptosis induction and cell cycle arrest modulated by fisetin. ROS: reactive oxygen species.
Fisetin, a dietary flavonoid, targets critical oncogenic signaling proteins, such as PI3K/Akt/mTOR and NF-κB, that are integral to the proliferation, survival, and metastasis of nervous system-type cancers [29, 30]. It acts as a potent dual inhibitor of the PI3K/Akt and mTOR pathways, which are frequently overactive in gliomas, medulloblastomas, and neuroblastomas, thereby preventing tumor growth and inducing apoptosis. Fisetin was shown to inhibit the PI3K/Akt pathway and ERK1/2 signaling and to enhance the expression of JNK, c-Jun, and p38 MAPK. The study also reported increased ROS production and decreased mitochondrial membrane potential, suggesting that fisetin may be a promising therapeutic agent against osteosarcoma by modulating key signaling pathways [63].
Lim et al. [64] reported that fisetin effectively modulates the PI3K/Akt signaling pathway and inhibits PI3K activity in a dose-dependent manner. Fisetin at 30 µM induced apoptosis by silencing the anti-apoptotic cIAP-2 protein and by preventing Akt phosphorylation, thereby suppressing cell growth. It was also shown that fisetin’s inhibitory effect on PI3K was accompanied by a decrease in mTOR activity, indicating that fisetin acts as a dual inhibitor of PI3K and mTOR, thereby amplifying its pro-apoptotic effects. The findings of this study point to fisetin as a potentially effective chemotherapeutic agent for cancers with PI3K/Akt pathologic signaling, and to its considerable potential when used in combination with standard treatment to overcome treatment resistance and improve patient outcomes. Zhang et al. [58] also found that fisetin prevents neurotoxicity induced by high glucose (HG) in HT22 hippocampal neuronal cells by inhibiting the PI3K/Akt/CREB signaling cascade. The researchers established that fisetin enhanced the viability of cells, diminished oxidative stress (regulating the activity of SODs and MDA levels), and greatly reduced the level of apoptosis of HT22 cells exposed to HG. The therapeutic effect associated with these functions was attributed to fisetin’s capacity to restore phosphorylation of PI3K, Akt, and CREB, which were suppressed by HG treatment. Notably, partial inhibition of this neuroprotective effect by the PI3K-specific inhibitor LY294002 indicates that the PI3K/Akt/CREB pathway will likely play a significant role in fisetin’s protective effect. The study found that fisetin was a potential therapeutic agent for preventing neuronal damage caused by hyperglycemia, and its neuroprotective properties were associated with activation of the PI3K/Akt/CREB pathway, as well as its potential use in conditions such as diabetic encephalopathy. Yang et al. [66] researched the neuroprotective effect of fisetin in traumatic brain injury (TBI) models. They have shown that fisetin reduces neurological injury and cognitive impairment by inhibiting ferroptosis and oxidative stress. Fisetin decreased brain edema, neuron survival, and cognitive performance in mouse models. In vitro analysis revealed that fisetin enhanced cell survival under stress in HT22 cells, reduced ferroptosis by regulating related proteins, and reduced ROS production. Fisetin engages the PI3K/Akt/NRF2 signaling pathway, which is essential for brain protection. These results indicate that fisetin is a candidate treatment for TBI targeting ferroptosis and oxidative stress. Alamoudi et al. [67] tested the anticancer efficacy of fisetin and significantly modulated the PI3K/Akt/mTOR pathway and induced apoptosis. Results revealed that fisetin suppressed the growth of cells in a time and dose-dependent manner (60 µM and 90 µM). The analysis found that fisetin suppressed PI3K, mTOR, and NF-κB, and stimulated Bax and suppressed Bcl-2, thereby inducing apoptosis. The PI3K/Akt/mTOR pathway was identified as a major mechanism underlying fisetin’s antiproliferative effect. In conclusion, the study hypothesized that microtubules are anticancer targets regulated by fisetin, which explains its proposed use as a colorectal cancer treatment.
Fisetin lowers the generation of both regulatory (p85) and catalytic (p110) subunits of PI3K, thereby concomitantly inhibiting the phosphorylation of Akt at Thr308 and Ser473 in a dose-dependent manner, which is necessary for complete Akt activation [28, 29]. This competes with downstream signaling pathways that promote cell survival and growth. In addition, fisetin inhibits phosphorylation of mTOR at Ser2448 and thus the formation of mTORC1 and mTORC2 complexes. Following fisetin treatment, major constituents of these complexes, such as Raptor, Rictor, GβL, and PRAS40, are down-regulated, thereby influencing protein synthesis and cellular metabolism, which are important for tumor progression [27, 29, 61]. Fisetin also exerts neuroprotective effects by modulating PI3K/Akt signaling, thereby attenuating oxidative stress and apoptosis in cancer cells [68]. These combined actions induce cell cycle arrest and apoptosis, thereby reducing cancer cell viability. In addition, fisetin suppresses the expression of MMP-2 and MMP-9, which are involved in tumor invasion and metastasis, by abrogating NF-κB- and AP-1-dependent transcriptional activity [69]. The multitargeting action of fisetin, through pathways that regulate proliferation, survival, and metastasis, provides a promising therapeutic strategy for CNS malignancies (Figure 6). Fisetin-induced inhibition of PI3K/Akt/mTOR and NF-κB signaling may represent an interesting molecular mechanism underlying its anticancer potential in nervous system cancers and warrants further study as a candidate for cancer therapeutic applications in neuro-oncology.
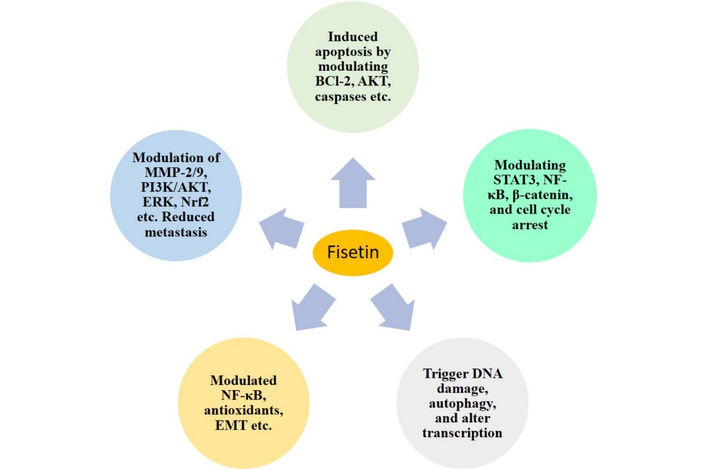
Fisetin’s potential anticancer effects involve modulation of multiple signaling pathways. AKT: protein kinase B; MMP: matrix metalloproteinase; PI3K: phosphatidylinositol 3-kinase; ERK: extracellular signal-regulated kinase; Nrf2: nuclear factor erythroid 2-related factor 2; STAT3: signal transducer and activator of transcription 3; NF-κB: nuclear factor-kappa B; EMT: epithelial-mesenchymal transition.
In sum, the effects of fisetin on several pivotal oncogenic signaling pathways, including PI3K/Akt, NF-κB, mTOR, and MAPK, underscore the multifaceted role of this plant polyphenol in cancer therapy. Fisetin also blocks these pathways, inhibiting tumor cell proliferation and viability, inducing apoptosis, and suppressing migration and angiogenesis. Because of its broad-spectrum action, fisetin may also be a promising therapeutic option, particularly for nervous system cancers, which are often dysregulated. Further investigations addressing the molecular targets of fisetin, its combination with other drug(s) or agents, may also yield valuable information towards how it could be potentially employed for adjuvant cancer therapy.
Fisetin, a known flavonoid with diverse pharmacological activities, exhibits potent inhibitory activity against cancer metastasis by blocking multiple critical processes, including cellular invasion, cell migration, and the epithelial-to-mesenchymal transition [54]. These processes are crucial for tumor proliferation and invasion, but when deregulated in cancer cells, they enable escape from the organ of origin and dissemination to other organs. More specifically, fisetin exerts its anti-metastatic potential by inhibiting molecular events that govern EMT, a key process by which cancer cells acquire migration and invasion properties during metastasis [70]. Fisetin is known to be a multitargeted signaling inhibitor, and to inhibit the migration of some cancer cells; migration and invasiveness are key steps in forming metastasis; thereby, the ability of tumor cells to move inside tissues dictates their potential for producing a metastatic colony in a site such as neuro-oncology, for which treatment for metastases is one of the most important clinical challenges [71].
EMT is a cellular phenomenon that confers on epithelial cells the capacity to assume a mesenchymal-like fate, thus permitting these cancer-forming cells, on one hand, to detach from the primary tumor mass and travel through the stromal compartment, and, on the other hand, to invade surrounding organs [72]. This transition is accompanied by downregulation of EMT markers, such as E-cadherin, and upregulation of mesenchymal markers, like N-cadherin, vimentin, and fibronectin [73]. Activation of EMT-related signaling pathways favours cell motility and invasion, two critical processes during metastasis [74]. Fisetin’s capacity to suppress cell migration and invasion is strongly associated with its ability to modulate the activity of the MMPs, a group of proteolytic enzymes that degrade the ECM. MMPs, especially MMP-2 and -9, are largely involved in tumor cell invasion by degrading the ECM, enabling tumor cells to move through tissues. Fisetin has been shown to reduce the synthesis and activity of MMP-2 and -9, which mediate tumor cell invasion into adjacent tissues [27]. By inhibiting these MMPs, fisetin prevents cancer cells from degrading the ECM and spreading to other parts of the body, a final stage in metastasis. This is particularly important in nervous system tumors, where implanted properties of glioblastoma and other malignant brain tumors that confer invasiveness are associated with poor prognosis [75].
In addition to EMT and MMPs, fisetin regulates other key signaling pathways involved in migration and invasion. The PI3K/Akt and MAPK cascades are key pathways that induce cell motility and invasiveness in cancer cells [76]. Fisetin can suppress both pathways that inhibit downstream activities and those that activate Rac1, Cdc42, and RhoA, which are responsible for actin cytoskeletal organization and cell motility [77]. Fisetin also impairs malignant cells’ ability to execute pliable motility/invasion-associated structural remodelling by preventing activation of these signaling pathways. Furthermore, fisetin inhibits focal adhesion kinase (FAK) phosphorylation, a mediator of cell adhesion and migration. Fisetin inhibits focal adhesion formation by disrupting FAK signaling, which is indispensable for cells to adhere to the ECM and generate contractile forces for cell motility [78]. Fisetin modulates invasion and migration in tumor tissues, particularly by promoting anti-angiogenesis in the tumor microenvironment (TME). So, to be able to feed that tumor like an infant, it needs angiogenesis, which is the growth of new blood vessels, very important for feeding the hypoxic regions in developing tumors and necessary if you’re going to have metastatic spread. Fisetin suppresses angiogenesis by decreasing the levels of VEGF and inhibiting activation of the hypoxia-inducible factor-1 alpha (HIF-1α) pathway, an important regulator of angiogenesis [79]. Through its effects on angiogenesis, fisetin not only obstructs tumor growth but also deprives tumor cells of their ability to infiltrate distant sites in the body via the bloodstream (and thus prevents metastasis). In summary, fisetin has striking anti-metastatic effects to suppress key aspects of the metastasis process, including invasion, migration, and EMT (Figure 6). Regarding metastatic and invasive potential, fisetin reciprocally chemosensitizes distant cells through molecular crosstalk cascades involving MMP-kinase regulation (Akt-EMT, MAPKs/FAK) and suppresses EMT/angiogenic footprints of cancer. These effects lead to the suggestion of fisetin as a promising candidate in the therapeutic control of metastasis development in several cancer types, including brain cancers such as glioblastoma. Further clinical trials are warranted to fully evaluate the effects of fisetin, combined with other treatments, on metastatic disease and on improving patient survival.
TME plays a pivotal role in cancer development, influencing tumor growth, metastasis, and therapeutic response [80]. It comprises diverse cellular and noncellular components, including neoplastic cells, stromal cells, immune cells, vascular elements, and ECM [81]. Tumor invasion and metastasis depend on angiogenesis, the formation of new blood vessels from existing vasculature, a central process in the TME that provides nutrients and oxygen to an enlarging neoplasm [82]. Fisetin significantly suppresses the TME and angiogenesis; therefore, it is a potential therapeutic agent for cancer treatment, including nervous system tumors such as glioblastoma, in which vasculature is essential for tumor expansion and invasion [76]. The effect of fisetin on the TME is multifaceted, as it can modulate immune responses, stromal cells, and ECM function. Its effects have also been shown to inhibit inflammatory signaling in the developing tumor environment, thereby reducing the accumulation and activation of immune cells (such as macrophages and neutrophils) that are often pro-tumor. Chronic inflammation in the TME plays a pivotal role in cancer growth and metastasis by supporting tumor cell proliferation, survival, and invasion [78]. Fisetin blocks the activity of major pro-inflammatory transcription factors, such as NF-κB, and, by inhibiting the overexpression of inflammatory cytokines (e.g., IL-6, TNF-α), disrupts a set of cancer metastasis-promoting inflammatory networks [83]. By inhibiting the immune contexture that generally permits cancer to escape immune effector mechanisms, as well as by exerting chemopreventive effects through its anti-inflammatory properties and effects on tumor cell growth, fisetin attenuates an immunosuppressive microenvironment that supports cancer and allows it to remain unchecked by immune effector mechanisms [71]. Kim et al. [55] also found that fisetin significantly modulated signaling pathways, including GSK-3β, NF-κB, c-Myc, EpCAM, and VEGF, thereby inhibiting angiogenesis and cellular proliferation. In vitro, a dose-dependent inhibitory effect on cell viability was observed at 25–100 µM, with an estimated half-maximal inhibitory concentration (IC50) of 43–50 µM. Systemic administration of 100–200 mg/kg in vivo delayed tumor development with no apparent toxicity. Collectively, this body of data indicates that fisetin could be used as a chemotherapeutic agent in colorectal carcinoma, owing to its anti-angiogenic effects. Fisetin significantly affects the TME by modulating the ECM and angiogenesis, two key factors in cancer progression. ECM, comprising proteins such as collagen and fibronectin, provides a framework for tumor cell structure and facilitates tumor cell invasion into surrounding tissues. Fisetin has also been shown to suppress MMP-2 and -9, reducing tumor cell migration and invasiveness by mitigating ECM degradation. This function is especially important in nervous system tumors such as gliomas, in which tumor infiltration is strongly influenced by ECM organization and remodeling [84]. Moreover, angiogenesis provides the oxygen and nutrients critical for tumor growth. Fisetin interferes with it by repressing the expression level of VEGF at the transcriptional level via suppression of HIF-1α, a key stimulator of VEGF. Fisetin effectively blocks angiogenesis by downregulating VEGF and other angiogenic factors, such as fibroblast growth factor (FGF) and platelet-derived growth factor (PDGF), and thus may be a promising therapeutic candidate, particularly for hypervascularized tumors such as glioblastoma [85]. The anti-angiogenic properties of fisetin enhance the efficacy of conventional cancer therapies, such as chemotherapy and radiotherapy, because tumors with reduced angiogenesis are less likely to develop treatment resistance [76]. Furthermore, fisetin can enhance therapeutic delivery to tumor lesions by normalizing tumor vasculature. In conclusion, the potential of fisetin to modulate the inflammatory signaling pathway, ECM remodeling, and angiogenesis makes it an attractive adjunct anticancer therapy agent, particularly for tumors such as glioblastoma, which exhibit highly TME-mediated dynamics that significantly influence disease progression and therapeutic efficacy (Figure 6). As shown in Table 3, fisetin exerts multiple effects on the TME, including inhibiting invasion and migration.
Conventional cytotoxic chemotherapy remains the main treatment for malignant nervous system tumors, but its effectiveness is hampered by various factors such as chemoresistance and neurotoxicity, leading to only modest survival rates for patients with glioblastoma and other high-grade CNS neoplasms [86]. This has spurred interest in combination strategies that incorporate non-toxic phytochemicals to enhance treatments, reduce dosages, and minimize side effects [87].
The potential synergistic effects among fisetin, other bioactive compounds, and conventional chemotherapeutic agents are encouraging for improving therapeutic efficacy and reducing the toxicities of chemotherapeutic drugs (Rahmani et al. [61]). Despite its high cytotoxicity, chemotherapy has a limited therapeutic effect, mainly resulting from chemoresistance and lack of selectivity, causing severe side effects and poor prognosis in patients [86]. Recent studies have begun to unmask the importance of these properties, particularly in nervous system cancers. Chen et al. [48] demonstrated that fisetin suppresses ADAM9 expression and markedly reduces the invasiveness and migration of human glioma cells through prolonged ERK1/2 activation and subsequent repression of pro-invasive pathways, providing a critical factor in CNS tumor progression rather than non-specific cytostasis in vitro. Beltzig and co-workers reported that in LN229 glioblastoma cells, fisetin (40–80 µM) induces p53-dependent apoptosis [46]. Emerging data indicate that therapy-induced senescence and the senescence-associated secretory phenotype (SASP) are significant contributors to glioma recurrence. Consequently, the capacity of a well-tolerated flavonoid to selectively eliminate senescent tumor cells holds substantial translational relevance [88]. Nanotechnology-based approaches have enhanced the justification for fisetin-containing combination therapies in CNS malignancies. Renault-Mahieux and colleagues [49] developed liposomes that co-encapsulate fisetin and cisplatin for the treatment of glioma. This formulation preserved fisetin’s anti-angiogenic activity, maintained cisplatin’s cytotoxicity, and demonstrated at least additive anti-glioblastoma effects in vitro, while also addressing fisetin’s poor aqueous solubility. Wang et al. [56] recently reported a microenvironment-responsive, cRGD-modified nanoparticle engineered to cross the BBB and co-deliver fisetin and doxorubicin. In animal models of glioma, fisetin release enhanced doxorubicin uptake, promoted apoptosis, induced G2/M arrest, and inhibited proliferation and migration, resulting in significant anti-glioma activity in vivo, demonstrating the potential of rational delivery systems to utilise the chemosensitizing properties of fisetin. Fisetin has been demonstrated to modulate the PI3K/Akt/mTOR, NF-κB, and MAPK pathways at the signalling level across various tumor systems, including astrocytic models. It also reverses EMT-like programs and suppresses matrix metalloproteinase-driven invasion and angiogenesis [76]. This multi-targeted modulation is essential in the heterogeneous molecular landscape of nervous system tumors, where redundant oncogenic circuits often compromise the efficacy of single-pathway inhibitors. The pleiotropic effects of fisetin, combined with its ability to enhance the cytotoxicity of alkylating agents and platinum compounds while reducing off-target toxicities in non-neural tissues, suggest its potential as a valuable adjunct to conventional chemotherapy in CNS oncology [76].
The convergence of mechanistic, nanotechnological, and preclinical efficacy data strongly supports further research on fisetin-containing regimens for malignant nervous system tumors, with a focus on glioblastoma and high-risk gliomas, despite clinical translation remaining in its early stages. Fisetin significantly enhances the chemotherapeutic effects of chemotherapy, enabling a reduction in the chemotherapeutic dose while maintaining the same cytotoxic effect. As a result, the patient’s quality of life improves, and survival rates increase due to the innovative approach of combining chemotherapy with bioactive compounds [78]. Fisetin had synergistic properties with most chemotherapeutic drugs and other natural compounds [61]. Traditional chemotherapy, a backbone in cancer treatment, has significant disadvantages in chemoresistance and dose-limiting side effects, which significantly decrease its overall advantage and deteriorate patient outcomes [89]. The integration of phytochemicals and conventional chemotherapy provides an avenue to increase efficacy and reduce the use of conventional drugs, thereby reducing the incidence and intensity of adverse effects and improving patient quality of life and survival [90]. Fisetin, a phytochemical, has been a highly promising chemosensitiser and anticancer agent [76]. Fisetin, when combined with cabazitaxel, has been reported to enhance tumor proliferation and metastasis and to augment apoptosis, yielding a more robust anti-tumor effect than either drug alone. Combining carnosic acid with fisetin further enhances fisetin’s ability to regulate apoptosis-related proteins in lung cancer cells, downregulating the anti-apoptotic proteins Bcl-2/Bcl-xL and Bax, and the DR and p53 upregulator caspase-3/8/9 [91, 92]. Tripathi et al. [92] showed that the combination of fisetin and cisplatin was 4 times more effective than either drug alone, suggesting a strong interaction. Fisetin also disrupts the cell cycle machinery, which is tightly regulated in cancer cells, typically in combination with other agents such as etoposide. This co-administration increases the percentage of cells in S/G2 and decreases the percentage in G2-M, thereby impeding passage through major checkpoints required for DNA replication and division and effectively halting cell proliferation in the tumor cell lineage [93]. The proliferation of in vivo studies, along with the development of several in vivo models, also supports the efficacy of fisetin-based combination therapy [28, 34, 35, 83]. Overall, the mounting evidence indicates that fisetin is a powerful adjuvant that can enhance the efficacy of current chemotherapeutic protocols, reduce both drug burden and toxicity, and improve treatment outcomes. The interaction of fisetin with chemotherapy agents, as presented in Table 3, has the potential to overcome drug resistance and increase cell death in glioblastoma.
Fisetin, despite its great anticancer effects, faces many challenges for clinical use due to its low water solubility, limited systemic bioavailability, rapid metabolism, and poor BBB permeability [27]. Pharmacokinetic limitations can greatly hinder achieving effective therapeutic concentrations in tumor tissues, particularly for malignant nervous system neoplasms [94]. Nanotechnology-based carriers have provided new approaches to overcome these limitations and improve the therapeutic efficiency of fisetin. Nanoformulations incorporating fisetin, such as liposomes, polymeric nanoparticles, SLNs, and micelles, are used to increase its solubility and stability (Figure 7). These nanoformulations improve fisetin delivery by enhancing its solubility, pharmacokinetics, and bioavailability [63]. The poly(lactic-co-glycolic acid) (PLGA)-based biocompatible polymer has been used to prepare polymeric nanoparticles that exhibit sustained release and enable long-term administration of fisetin to the tumor site, with minimal systemic side effects [95, 96]. In summary, nanodelivery offers a novel approach to maximizing the therapeutic effects of fisetin for CNS cancers and overcoming its significant pharmacokinetic limitations, thereby creating clinical opportunities. Nanotechnology is changing the paradigm of drug administration regimens, such as fisetin, which has low solubility, poor oral bioavailability, and limited CNS penetration. Such systems, through nanoparticles at the nanoscale, enhance the solubility, stability, and biological accessibility of fisetin, including its permeability across the BBB. Therefore, nanoformulations can improve bioavailability and deliver fisetin more efficiently and sustainably into nervous system tumors.
Fisetin exhibits enhanced anti-tumor activity when administered via polymeric nanoparticulate systems, especially against malignant nervous tissue tumors [56]. Feng et al. [96] reported that fisetin was encapsulated into poly(lactic acid) (PLA) using a spontaneous emulsification solvent diffusion technique, with an entrapment efficiency of 90.35% and a particle size of around 227 nm. In vivo experiments using the 4T1 breast cancer model showed that fisetin-PLA nanoparticles exhibited greater therapeutic efficacy than free fisetin, resulting in a significant reduction in tumor volume. This provides a promising rationale for the use of PLA nanocarriers to enhance the antitumor effects of fisetin as a sustained-exposure treatment for brain tumors. Another study reported that monomethyl poly(ethylene glycol)-poly(ε-caprolactone) (MPEG-PCL) copolymers to fisetin have been proven to be an efficient system for inhibiting tumor growth and weight in a mouse model, when it was administered via intravenous injection in vivo; the cell viability is up to 76%, reflecting low systemic toxicity. Nanocarrier systems provide a promising approach to improve fisetin delivery by enhancing its solubility, release behavior, and antitumor efficacy [96, 97]. In addition, fisetin encapsulated in polyvinylpyrrolidone (PVP) nanoparticles exhibited a more pronounced antiproliferative effect in MDA-MB-231 cells than free fisetin, reducing cell viability by up to 35%. The more rapid release of fisetin from the polymeric carriers may lead to enhanced efficacy, which is generally necessary to retain sufficient drug in fast-clearing, poorly perfused tumor spaces [98]. Sechi et al. [99] reported that fisetin-loaded nanoparticles based on PLGA-block-PEG-carboxylic acid (PLGA-PEG-COOH) and PCL exhibited pH-sensitive release behavior. Under gastric-simulated conditions, these formulations could protect fisetin from acid degradation, with sustained-release profiles, and are therefore suitable for oral administration in neuro-oncology.
The intestinal permeability of fisetin was also measured using everted gut sac models, demonstrating a remarkable 4.9-fold increase with PLGA/PVA nanoparticle delivery compared with fisetin suspension. These nanoparticles were kept stable for 60 days. The significantly increased intestinal uptake observed here, combined with pH-independent release, suggests that they can overcome fisetin’s low oral availability, a major obstacle to successful drug delivery in nervous system tumors [100].
Finally, pharmacokinetic experiments in rats demonstrated that fisetin-PLA nanoparticles significantly prolonged the elimination half-life (3.42 h) and increased the area under the concentration-time curve (AUC) (19.28 μg/mL). Such a pharmacokinetic advantage is attributed to prolonged release and restricted detachment in systemic circulation, mainly due to the nanoparticles’ negative charge [96, 100]. Regarding the therapy of malignant nervous system tumors, these polymeric fisetin nanoparticles with both increased systemic availability and controlled release capacity would present an advantageous approach to address biopharmaceutical challenges as well as improve delivery efficacy across the barrier into the CNS.
Polymeric micelles offer a promising approach to improve fisetin delivery, as its poor solubility limits its therapeutic application [101]. Ultra-small fisetin-loaded MPEG-PCL micelles [22 nm in diameter, encapsulation efficiency (EE) 98.5%] showed remarkable cellular uptake and cytotoxicity compared with free fisetin, as well as prolonged survival in tumor-bearing mice [101]. In other studies, the MPEG-PCL fisetin micelles exhibited 1.3-fold greater tumor growth inhibition and increased apoptosis in SKOV3 cells [102], while tocopherol-PEG-based micelles decreased the IC50 to approximately 2.9-fold in MCF-7 cells and significantly enhanced apoptosis at 24–48 h [103]. These gains in potency and intracellular accumulation are particularly pertinent for neurosystem cancers, where high metabolic stress, resistant pathways, and dense stroma barriers require more efficacious intracellular penetration systems.
Folic acid-targeted pluronic F127 micelles (≈ 103 nm, EE ≈ 82%) also exhibited pH-responsive, continuous release and significant pharmacokinetic advantages, including 1.8 times higher Cmax, 2 times longer t1/2, and a 6.3-fold increase in AUC [104]. These improvements suggest long-term systemic exposure and better accumulation in the acidic microenvironment typical of high-grade gliomas and CNS metastases.
Taken together, the results demonstrated that fisetin incorporated into polymeric micelles could achieve small size, sustained release, improved bioavailability, and tumor-selective accumulation, enabling efficient delivery to malignant nervous system tumors and their immunosuppressive microenvironment.
Nanostructured lipid carriers (NLCs) are a promising nanotechnology approach to address the major hurdles that limit fisetin’s application in malignant nervous system tumors, including low solubility, rapid clearance, and limited brain distribution [105–107]. SLNs form a solid matrix of lipids stabilized by surfactants, enabling controlled release and protection of labile molecules within physiological lipids to decrease toxicity [108]. Fisetin-loaded SLNs co-encapsulated with the photosensitizer IR-780 (134 nm size, EE ≈ 41%) led to 50% stable viability reduction in cancer cells after net therapy of combined electroporation and near-infrared (NIR) photodynamic therapy, accompanied by cytoskeletal alterations and p53 and manganese superoxide dismutase (MnSOD) over-expression [109]. These similarly multifunctional SLNs can be translated directly to neuro-oncology, where image-guided PDT and oxidative stress-mediated killing are being investigated for applications in glioblastoma and other CNS tumors.
Conventional and novel liposomes have also been employed to encapsulate fisetin, thereby enhancing its solubility, circulation half-life, and bioavailability. DOPC/DODA-PEG2000 fisetin liposomes (173 nm) slowed tumor growth in vivo and, relative to free fisetin after intraperitoneal dosing, produced a 47-fold increase in bioavailability; co-encapsulation with cisplatin improved antiangiogenic and cytotoxic activity, including additive effects on U-87 MG glioblastoma cells [49]. These results support the conclusion that fisetin liposomes can enhance the efficacy of conventional chemotherapies and remodel the tumor vasculature in nervous system malignancies.
Even more sophisticated lipid structures, such as spherulites and nanocochleates, optimise loading and pharmacokinetics. The entrapment of fisetin in spherulites was approximately 5-fold greater than for simple liposomes, whereas IC50 values were lower, indicating a retarded release. Fisetin nanocochleates (275 nm; EE ≈ 84%) maintained a 141-fold increase in bioavailability and improved in vitro antitumor effects, with slower elimination [110]. This combination of high stability, slow release, and prolonged systemic exposure is especially appealing for maintaining therapeutic levels of fisetin in diffuse treatment-refractory nervous system tumors.
Preclinical data indicate a relatively wide acute safety margin for fisetin; however, its toxicity profile is route-dependent and not well established in neuro-oncology. No obvious clinical toxicity or histological changes in the main viscera (heart, lung, liver, kidney) were observed in the acute toxic test with a single dose of up to 2 g/kg [111]. Reported median lethal dose (LD50) values vary markedly with the route of administration: about 1,700 mg/kg when given orally, 400 mg/kg by subcutaneous injection, 250 mg/kg intraperitoneally, and only 200 mg/kg by the intravenous route in mice, showing a narrower margin for parenteral dosing. In tumors of the nervous system, such as malignant gliomas, both systemic and local delivery routes may be considered, and route-specific differences in distribution, toxicity, and drug exposure can be important in designing dosing regimens [112].
In vitro, fisetin reduces the survival of numerous cancer cell types in a dose- and time-dependent manner, with IC50 values often in the low micromolar range. IC50 values of SCC lines from the tongue and gingiva (CAL-27, Ca9-22, HSC3, SCC-4) vary between 10 and 50 µM, indicating diversity in sensitivity between cell types. Laryngeal cancer cell lines (TU212, Hep-2, M2e) exhibit the highest sensitivity, with IC50 values of 10 µM. These data identify a window of pharmacological activity whereby fisetin is shown to be selectively cytotoxic toward rapidly replicating tumor cells but provide little insight into possible off-target neurotoxicity within nonmalignant neurons, oligodendrocytes, or astrocytes, an important consideration for malignant nervous system tumors, where preservation of adjacent functional tissue often remains paramount [113–115].
There have been isolated reports of signals that suggest organ toxicity. Fisetin administration increased alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels, reflecting possible hepatocellular stress in the orthotopic mammary cancer model, while protecting kidney function, with no systemic toxicity observed. In contrast, most preclinical studies provide no evidence of significant toxicity at usual anti-tumor doses, thereby supporting an overall favourable safety profile [116, 117]. The major outstanding question in neuro-oncology is the potential impact of long-term or high-dose fisetin, particularly when combined with temozolomide, radiation therapy, or targeted agents, on hepatic stress responses and drug metabolism, as well as on antiseizure medications commonly prescribed to patients with brain tumors.
Taken together, current data suggest that fisetin is generally well tolerated at low to moderate doses and has an adequate preclinical safety window for further development. However, further studies are needed to establish CNS-specific and long-term organ toxicity, as well as interactions with standard chemoradiotherapy and antiepileptic treatment, if procaspase-activating compound (PAC) is to have a role in the treatment of malignant nervous system tumors, and hence should be pursued in well-designed phase I/II neuro-oncology trials.
Fisetin is an anticancer agent and, as shown in preclinical studies, exhibits significant anticancer potential; however, major limitations to its use as a clinical agent for treating malignant nervous system tumors remain. These include low aqueous solubility, which affects bioavailability; rapid metabolism, leading to reduced efficacy; and poor BBB penetration. Even though improvements in nanotechnology-based delivery systems are likely to enhance fisetin’s stability and improve brain targeting, this does not imply that any phase I/II clinical trials have been conducted in humans. Further studies, such as trials assessing dosage, safety, and efficacy, are needed to translate fisetin into clinical practice for CNS malignancies.
Fisetin also shows potential for the management of CNS tumors, particularly glioblastoma, though it presents challenges [46]. These issues include poor solubility, formulation-related delivery challenges, and the need to further optimize nanoparticle-based fisetin delivery for glioblastoma treatment [45]. Comparison with other flavonoids should demonstrate exceptional benefits or successful treatment, and proceeding to a phase I clinical trial will require extensive research on pharmacokinetics, toxicology, and biomarkers.
BBB presents a major problem in the management of glioblastoma, and full functionality of the BBB, particularly at invasive limits, impedes active drug administration [118, 119]. This heterogeneity is associated with tumour recurrence and resistance to treatment because tumor cells that infiltrate the mass are not exposed to therapeutic agents, making the management of even those parts of the tumor that have not been radiologically affected very difficult [120, 121]. Fisetin is a natural flavonoid that has shown anticancer potential in experimental models [76]. However, its application in brain tumors is challenged by delivery-related limitations, including BBB penetration. Fisetin can also enter tumor tissues via ultrasound-mediated or osmotic disruption of the BBB. These approaches can enhance the clinical translation of fisetin for the treatment of glioblastoma by improving the brain penetration of therapeutic agents. To be used in a human clinical trial, these techniques should first be optimised in preclinical models [45].
Radiotherapy turns out to be the main treatment of glioblastoma, but radioresistance becomes a major challenge. Cancer cells can be radiosensitized by radiosensitizers, making them more susceptible to radiation-induced damage and thereby reducing this barrier. Fisetin is a naturally occurring flavonoid that may be a radiosensitizer in most malignancies, but its role in glioblastoma remains unknown. Fisetin enhances apoptosis, a primary mechanism by which radiation kills cancer cells. In p53-mutant HT-29 colorectal cancer cells, fisetin pretreatment prolonged radiation-induced G2/M arrest and enhanced caspase-dependent apoptosis, suggesting radiosensitizing potential in non-glioblastoma cancer models [122]. Fisetin can enhance the effectiveness of radiation by increasing apoptotic signalling and inhibiting DNA repair [123]. Fisetin shows potential as an adjunct to radiotherapy in some cancer models, although direct evidence in glioblastoma remains limited [45, 124]. In preclinical colorectal cancer models, fisetin enhanced radiation-induced cytotoxicity and tumor-suppressive activity. Future studies will investigate its capacity to modulate DNA repair and oxidative stress pathways to enhance radiosensitivity in glioblastoma cells. Additionally, its effectiveness in minimizing normal tissue toxicity while enhancing tumor-specific effects warrants further investigation in preclinical glioblastoma models.
Therapy-induced senescent cells that drive tumor resistance and recurrence complicate the treatment of recurrent glioma. Despite their irreversible cell cycle arrest induced by genotoxic stress (e.g., chemotherapy or radiation), senescent cells can release pro-inflammatory and pro-tumorigenic substances, collectively known as the SASP. Senescent cells can cause persistent inflammation, immunosuppression, and tumor development, particularly in recurrent glioma. Therefore, senolytic medicines that selectively target and remove senescent cells may improve traditional therapy [46]. Yousefzadeh et al. [117] found that fisetin was the most efficient senolytic agent among ten flavonoids. In the study, fisetin successfully regulated senescence in wild-type progeroid and aged mice (500 mg/kg) and appeared to reduce p16Ink4a and p21Cip1 levels. This treatment improved tissue health and extended lifespan, indicating that fisetin has potential as a senotherapeutic. The ideal in vitro concentration to reduce senescent cells was 5 µM, and higher in vivo doses (500 mg/kg) were effective without side effects. Preclinical experiments have shown that fisetin selectively induces apoptosis in senescent cells, particularly in TMZ-induced gliomas. Fisetin dramatically reduced the number of senescent LN229 and A172 glioma cells, thereby boosting senescent tumor cell death. These data suggest that fisetin may reduce senescent cells that drive glioma recurrence after standard treatment [45]. Mahoney et al. [125] found that intermittent fisetin supplementation (100 mg/kg/day) alleviated vascular cell senescence and SASP factors in old mice, thereby promoting endothelial function and easing aortic stasis. This is due to increased nitric oxide bioavailability and reduced oxidative stress. The combination of one week of treatment followed by two weeks off targeted senescent cells selectively and did not disrupt their physiological functions. These results validated fisetin’s senolytic properties in a genetic model. Fisetin targets senescent tumor-associated fibroblasts and immune cells in breast and colorectal cancer to induce senolysis, thereby reducing inflammatory cytokine release and tumour growth. It may support glioma treatment by targeting senescent cells and modulating inflammation, thereby preventing tumor recurrence and resistance [126]. Zhang et al. [127] investigated the effects of fisetin on bleomycin-induced pulmonary fibrosis in mice, revealing that a dosage of 100 mg/kg significantly diminished fibrosis markers, including collagen deposition and α-SMA expression. The reduction in inflammatory cytokines (IL-1β, IL-6, TNF-α) in bronchoalveolar lavage fluid was noted alongside fisetin’s ability to activate AMPK and inhibit NF-κB signaling. Additionally, fisetin treatment mitigated TGF-β/Smad3 signaling, underscoring its potential as a senolytic agent. While these findings underline fisetin’s promising senolytic effects, clinical validation in glioma models is needed. Future studies should assess fisetin’s senolytic activity in vivo, evaluate biomarker responses, and explore its combination with standard glioma treatments.
Plant flavonoids are polyphenolic compounds with anticancer effects that vary in their associated molecular targets and biological effects [128]. Fisetin, quercetin, luteolin, and curcumin are well-known flavonoids that share mechanistic overlaps, including modulation of apoptosis, oxidative stress, inflammation, and oncogenic signaling pathways (e.g., PI3K/Akt/mTOR and NF-κB). These compounds exhibited distinct properties that affect their therapeutic efficacy in oncology [129–131]. Overall, flavonoids exhibit significant anticancer effects that can be exploited to regulate apoptosis, prevent metastasis and angiogenesis, and enhance responses to conventional therapies across diverse malignancies [128].
Fisetin exerts diverse anticancer effects, including inducing apoptosis, inhibiting tumor proliferation, and regulating pro-survival signaling [132]. Its wide range of targeting possibilities has led to its potential use in neurodegenerative diseases, albeit with issues of bioavailability and BBB penetration [32]. It is the case of quercetin, which has antitumor activity similar to that of fisetin, acting by altering oxidative stress, drug transporters, and cell cycle regulators [133]. It modulates PI3K/Akt/mTOR signaling and may overcome multidrug resistance in cancer models, suggesting interactions with chemotherapeutic agents [134].
Luteolin has also demonstrated strong anticancer effects, particularly in inhibiting cell proliferation, migration, and EMT, including in glioma and other cancer types [135]. It regulates p-IGF-1R/PI3K/Akt/mTOR signalling and suppresses matrix metalloproteinase activity, suggesting its potential to reduce glioma invasiveness and the TME [136]. Also, luteolin has been reported to inhibit genetic damage and chromosomal aberrations in non-CNS experimental models [137]. There is a comparison of curcumin and flavonols based on their anticancer activity via pathways such as STAT3 and NF-κB, but this research has not been combined with clinical trials due to their low bioavailability and metabolic instability [138].
Finally, despite the similar mechanisms of the flavonoids mentioned, fisetin is unique for its senolytic effect and its potential to affect multiple targets. Fisetin would have specific advantages when combined with CNS-optimized delivery systems for the treatment of glioma. Direct comparative neuro-oncology studies are necessary to identify which specific flavonoid or combination of flavonoids should be considered to achieve appropriate clinical outcomes or to exclude them.
To translate fisetin into a Phase I CNS tumor clinical trial, a systematic translation process, encompassing evidence focus, regulatory guideline compliance, and a strategic approach to translation, is needed [77]. The future of drug development encompasses selecting a drug lead and conducting preclinical validation, including demonstrating biological activity and an excellent therapeutic index [139]. Although preclinical research has indicated the anticancer activity of fisetin, its lack of efficacy in clinical use is attributable to poor solubility, rapid metabolism, and limited CNS penetration [32]. Preclinical toxicology and pharmacology testing must be conducted before initiating first-in-human (FIH) studies [140]. This involves pharmacokinetic profiling of absorption, distribution, metabolism, and excretion (ADME) and safety research, which guides the selection of toxicity and dose [141]. The resulting information is then submitted in the form of an Investigational New Drug (IND) application, which must be approved by regulatory agencies such as the U.S. Food and Drug Administration (FDA) before human subjects can be tested [142, 143]. For CNS agents such as fisetin, BBB penetration and CNS distribution can be achieved through advanced formulation technologies, including nanoparticles and receptor-targeting [144]. In oncology, phase I CNS trials are used to determine dose escalation, with a major emphasis on safety and tolerability, and to identify the maximum tolerated dose (MTD) [145]. Program ethics approval and clinical plans, informed by safety monitoring and nonclinical toxicology, are needed to advance fisetin into the initial clinical phases.
The natural dietary flavonoid fisetin exerts broad anticancer effects against nervous system malignancies, including gliomas, medulloblastomas, and neuroblastomas. Fisetin suppresses tumor cell growth, stimulates apoptosis and autophagy, inhibits invasion and metastasis, and positively alters the TME by targeting key signalling pathways, including PI3K/Akt/mTOR, NF-κB, and MAPK. In vitro and in vivo preclinical studies demonstrate reduced tumor progression and increased survival, indicating the promise of fisetin in augmenting the effects of traditional treatments, including temozolomide and radiotherapy. Nevertheless, it should be emphasized that the existing data remain preclinical, and no phase I/II clinical trials in CNS tumors have been conducted. The available evidence is based on in vitro and animal research, and the clinical application of fisetin remains speculative. There are no human clinical trials available yet, so any claim of its therapeutic value should be viewed as hypothesis-forming rather than conclusive. The clinical translation of fisetin is currently limited by poor aqueous solubility, rapid metabolism, and nonoptimal BBB penetration. These barriers have also been addressed through nanotechnology-based delivery systems that enhance stability, allow selective delivery to the brain, and provide sustained release. Despite toxicology data indicating that fisetin is well tolerated and has a large therapeutic index, high-quality clinical trials are still needed to characterize its dose, long-term safety, and clinical efficacy. In general, fisetin remains a preclinical candidate with a multitarget therapeutic candidate in neuro-oncology. Incorporating enhanced drug-delivery technologies into a rational combination approach can ultimately realise the full clinical potential of fisetin-based treatments in patients with tumors of the malignant nervous system.
Akt: protein kinase B
AUC: area under the concentration-time curve
BBB: blood-brain barrier
CNS: central nervous system
ECM: extracellular matrix
EE: encapsulation efficiency
EMT: epithelial-mesenchymal transition
ERK: extracellular signal-regulated kinase
FAK: focal adhesion kinase
HG: high glucose
HIF-1α: hypoxia-inducible factor-1 alpha
IC50: half-maximal inhibitory concentration
IL: interleukin
JNK: c-Jun N-terminal kinase
MAPK: mitogen-activated protein kinase
MMP: matrix metalloproteinase
MPEG-PCL: monomethoxy poly(ethylene glycol)-poly(ε-caprolactone)
mTOR: mechanistic (mammalian) target of rapamycin
NF-κB: nuclear factor-kappa B
PEG: polyethylene glycol
PI3K: phosphatidylinositol 3-kinase
PLA: poly(lactic acid)
PLGA: poly(lactic-co-glycolic acid)
ROS: reactive oxygen species
SASP: senescence-associated secretory phenotype
SLNs: solid lipid nanoparticles
TBI: traumatic brain injury
TME: tumor microenvironment
TNF-α: tumor necrosis factor-alpha
VEGF: vascular endothelial growth factor
AK and A: Writing—review & editing, Conceptualization, Validation, Methodology, Formal analysis, Writing—original draft. Both authors read and approved the submitted version.
The authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1151
Download: 18
Times Cited: 0
Zohreh Khosravi Dehaghi
Maria Ciscar-Fabuel ... Andreu Gabarros-Canals
Maria Ciscar-Fabuel ... Andreu Gabarros-Canals
Julius Mulumba ... Yong Yang
Adam H. Lapidus, Malaka Ameratunga
