129 results in Exploration of Neuroscience
Most Viewed
Sort by :
- Latest
- Most Viewed
- Most Downloaded
- Most Cited
Open Access
Review
Current therapeutics for Alzheimer’s disease and clinical trials
Danqing Xiao, Chen Zhang
Published: June 27, 2024 Explor Neurosci. 2024;3:255–271
This article belongs to the special issue Alzheimer’s Disease
Open Access
Review
Effects mediated by melatonin and cortisol of artificial light and noise, alone and in combination, on sleep and health
Nahum M. Gabinet
Published: September 13, 2024 Explor Neurosci. 2024;3:382–417
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Review
Impact of circadian clock dysfunction on human health
Saptadip Samanta, Sk Asif Ali
Published: September 29, 2022 Explor Neurosci. 2022;1:4–30
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Review
Negative environmental influences on the developing brain mediated by epigenetic modifications
Maya Komar-Fletcher ... Joanna Michalina Jurek
Published: September 28, 2023 Explor Neurosci. 2023;2:193–211
Open Access
Review
Neuropharmacologic modulation of the melatonergic system
Utku Aykan ... Canan Uluoglu
Published: December 22, 2023 Explor Neurosci. 2023;2:287–306
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Review
An intricate relationship between circadian rhythm dysfunction and psychiatric diseases
Saptadip Samanta, Debasis Bagchi
Published: August 23, 2024 Explor Neurosci. 2024;3:321–351
This article belongs to the special issue Circadian Rhythm and Melatonin
Open Access
Review
Novel treatments of depression: bridging the gap in current therapeutic approaches
Amit Jagtiani
Published: July 09, 2024 Explor Neurosci. 2024;3:272–286
This article belongs to the special issue Novel Therapeutic Approaches for the Treatment of Depression
Open Access
Review
Update for astrocytomas: medical and surgical management considerations
Matthew Willman ... Brandon Lucke-Wold
Published: February 23, 2023 Explor Neurosci. 2023;2:1–26
Open Access
Review
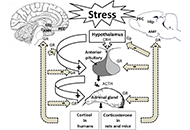
Cellular and molecular mechanisms of stress-induced memory impairment
Ameneh Rezayof ... Shiva Hashemizadeh
Published: December 30, 2022 Explor Neurosci. 2022;1:100–119
Open Access
Review
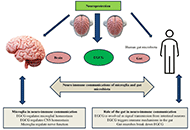
Neuroprotective insights into epigallocatechin gallate (EGCG) for neurodegenerative disorders
Neha Kamboj ... Rahul Kumar
Published: February 24, 2025 Explor Neurosci. 2025;4:100673
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection
Open Access
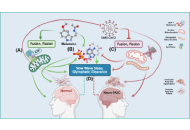
Review
Melatonin regulation of phase separation in Neuro-PASC: out-maneuvering Janus-faced amyloids
Doris Loh, Russel J. Reiter
Published: March 24, 2025 Explor Neurosci. 2025;4:100678
This article belongs to the special issue Identification of Therapeutic Targets in the Pathogenesis of Neurological Diseases
Open Access
Review
Stigma and psychosocial problems in patients with epilepsy
Kubra Yeni
Published: December 06, 2023 Explor Neurosci. 2023;2:251–263
This article belongs to the special issue Epilepsy
Open Access
Mini Review
Neuroprotective compounds from three common medicinal plants of West Bengal, India: a mini review
Suvendu Ghosh ... Debosree Ghosh
Published: December 26, 2023 Explor Neurosci. 2023;2:307–317
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection

Open Access
Original Article
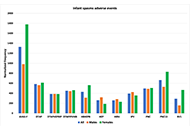
Epilepsy adverse events post vaccination
Darrell O. Ricke
Published: October 16, 2024 Explor Neurosci. 2024;3:508–519
This article belongs to the special issue Epilepsy
Open Access
Review
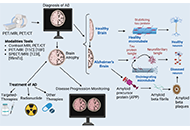
The role of neuroimaging in Alzheimer’s disease: implications for the diagnosis, monitoring disease progression, and treatment
Julius Mulumba ... Yong Yang
Published: February 25, 2025 Explor Neurosci. 2025;4:100675
This article belongs to the special issue Alzheimer's Disease
Open Access
Review
Psychology of bipolar depression: revisiting past and present researches, prospects ahead, and moving toward future directions
Behrooz Afshari
Published: December 29, 2023 Explor Neurosci. 2023;2:331–349
This article belongs to the special issue Novel Therapeutic Approaches for the Treatment of Depression
Open Access
Review
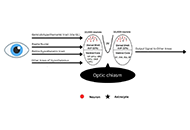
Functional interactions between neurotransmitters and neuropeptides in regulating suprachiasmatic nucleus function and circadian rhythms
Vallath Reghunandanan
Published: September 24, 2024 Explor Neurosci. 2024;3:434–477
Open Access
Perspective
What might melatonin supplementation provide for humans beyond improved onset to sleep?
Leticia A. Shea
Published: November 26, 2024 Explor Neurosci. 2024;3:551–558
This article belongs to the special issue Circadian Rhythm and Melatonin
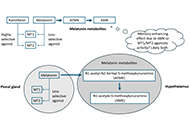
Open Access
Review
Interventional procedures for refractory neuropathic pain
Hannah G. Matejowsky ... Alan D. Kaye
Published: December 22, 2023 Explor Neurosci. 2023;2:276–286
This article belongs to the special issue Neuropathic Pain
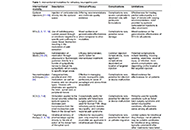
Open Access
Review
Current advances in epilepsy among patients with arteriovenous malformations
Joham Choque-Velasquez ... Alder Fernando Valenzuela-Rangel
Published: May 13, 2024 Explor Neurosci. 2024;3:175–197
This article belongs to the special issue Epilepsy
Journal Information
 Previous
Previous