





Endocrine resistance in estrogen receptor-positive (ER+) breast cancer has undergone a fundamental reconceptualization over the past decade. The discovery that activating mutations in the ESR1 gene encoding ERα emerge under aromatase inhibitor (AI) selection pressure and drive ligand-independent receptor activation established a shift from empirical treatment sequencing to molecularly guided intervention. This review provides a synopsis of the structural biology underlying constitutive ER activation, the evolutionary dynamics of ESR1-mutant clones detectable through circulating tumor DNA (ctDNA), and the clinical evidence demonstrating that early molecular detection can trigger therapeutic switches that alter disease trajectory. The regulatory approval of elacestrant for ESR1-mutant disease and randomized trial data showing progression-free survival (PFS) benefit from ctDNA-guided endocrine switching (PADA-1, SERENA-6) position ESR1 genotyping as a dynamic biomarker with direct therapeutic implications. We examine the integration of oral selective ER degraders (SERDs) into treatment algorithms, the role of co-occurring alterations in the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway, and emerging directions, including machine learning approaches to ctDNA kinetics and adaptive trial designs that treat clonal evolution as an actionable target. The convergence of structural mechanisms, liquid biopsy technology, and biomarker-driven drug development provides a framework for precision oncology in endocrine-resistant breast cancer. While these advances are substantial, important challenges remain, including the lack of mature overall survival (OS) data from interception trials, cost and accessibility barriers to serial ctDNA monitoring in diverse global healthcare settings, the unresolved question of optimal therapeutic sequencing in patients with concurrent ESR1 and PI3K pathway alterations, and the need to distinguish clinically actionable low-variant allele frequency (VAF) ESR1 calls from background noise in liquid biopsies.
Endocrine resistance in estrogen receptor-positive (ER+) breast cancer has undergone a fundamental reconceptualization over the past decade. The discovery that activating mutations in the ESR1 gene encoding ERα emerge under aromatase inhibitor (AI) selection pressure and drive ligand-independent receptor activation established a shift from empirical treatment sequencing to molecularly guided intervention. This review provides a synopsis of the structural biology underlying constitutive ER activation, the evolutionary dynamics of ESR1-mutant clones detectable through circulating tumor DNA (ctDNA), and the clinical evidence demonstrating that early molecular detection can trigger therapeutic switches that alter disease trajectory. The regulatory approval of elacestrant for ESR1-mutant disease and randomized trial data showing progression-free survival (PFS) benefit from ctDNA-guided endocrine switching (PADA-1, SERENA-6) position ESR1 genotyping as a dynamic biomarker with direct therapeutic implications. We examine the integration of oral selective ER degraders (SERDs) into treatment algorithms, the role of co-occurring alterations in the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway, and emerging directions, including machine learning approaches to ctDNA kinetics and adaptive trial designs that treat clonal evolution as an actionable target. The convergence of structural mechanisms, liquid biopsy technology, and biomarker-driven drug development provides a framework for precision oncology in endocrine-resistant breast cancer. While these advances are substantial, important challenges remain, including the lack of mature overall survival (OS) data from interception trials, cost and accessibility barriers to serial ctDNA monitoring in diverse global healthcare settings, the unresolved question of optimal therapeutic sequencing in patients with concurrent ESR1 and PI3K pathway alterations, and the need to distinguish clinically actionable low-variant allele frequency (VAF) ESR1 calls from background noise in liquid biopsies.
DOI: https://doi.org/10.37349/etat.2026.1002375
This article belongs to the special issue Breaking Boundaries in Breast Cancer Care: Emerging Controversies and Innovation in Surgical and Medical Approaches

Chemotherapy has profoundly shaped modern oncology, evolving from early cytotoxic approaches to biologically informed strategies. In glioblastoma (GBM), the highly aggressive primary brain tumor, precision medicine does not rely on the identification of a single crucial oncogenic driver but integrates biologically informed stratification strategies to predict treatment response, resistance, and therapy-induced adaptation. This review recapitulates the historical milestones of chemotherapeutic development in neuro-oncology, with particular emphasis on GBM. As the main chemotherapeutic agent currently used in GBM, temozolomide (TMZ) initially represented a major therapeutic breakthrough; however, its clinical use has progressively unveiled the biological and clinical limitations of conventional cytotoxic paradigms. While TMZ exerts antitumor activity through DNA damage–induced apoptosis, accumulating experimental evidence indicates that it may also elicit adaptive responses, ultimately supporting tumor progression and therapy resistance. By integrating historical milestones with recent molecular understanding, this review highlights how improved knowledge of therapy-induced adaptations may inform emerging precision medicine strategies in GBM, underscoring the need for tailored treatments to overcome tumor heterogeneity and adaptive responses.
Chemotherapy has profoundly shaped modern oncology, evolving from early cytotoxic approaches to biologically informed strategies. In glioblastoma (GBM), the highly aggressive primary brain tumor, precision medicine does not rely on the identification of a single crucial oncogenic driver but integrates biologically informed stratification strategies to predict treatment response, resistance, and therapy-induced adaptation. This review recapitulates the historical milestones of chemotherapeutic development in neuro-oncology, with particular emphasis on GBM. As the main chemotherapeutic agent currently used in GBM, temozolomide (TMZ) initially represented a major therapeutic breakthrough; however, its clinical use has progressively unveiled the biological and clinical limitations of conventional cytotoxic paradigms. While TMZ exerts antitumor activity through DNA damage–induced apoptosis, accumulating experimental evidence indicates that it may also elicit adaptive responses, ultimately supporting tumor progression and therapy resistance. By integrating historical milestones with recent molecular understanding, this review highlights how improved knowledge of therapy-induced adaptations may inform emerging precision medicine strategies in GBM, underscoring the need for tailored treatments to overcome tumor heterogeneity and adaptive responses.
DOI: https://doi.org/10.37349/etat.2026.1002374
This article belongs to the special issue Novel Precision Medicine Approaches to Brain Tumors (Primary and Metastatic)

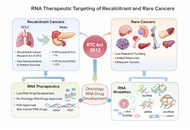
In 2013, more than a decade ago, the “Recalcitrant Cancer Research Act of 2012” was signed into law in the USA. Recalcitrant cancers are among the leading causes of global cancer morbidity and mortality. At the inception of the act, priority was placed on lungs and pancreatic cancers. Despite the tremendous advancement achieved in the research and treatment of said ‘recalcitrant cancers’ in the form of developing novel or modified small molecule and antibody drugs, modest improvement has been recorded for patients’ survival. Also, current mortality and morbidity for recalcitrant cancers keep increasing. Similarly, rare cancers only enjoy very meager research and drug development efforts globally. Consequently, very limited advancement has been made towards therapeutic development targeting rare cancers. Hence, the current situation calls for re-strategizing research efforts and exploring different treatment modalities towards combating recalcitrant and rare cancers. On this note, RNA therapeutics strategy holds a unique and vital prospect because of its propensity to target coding and non-coding RNA transcripts in the biological system. Moreover, RNA therapeutics such as lncRNAs and circRNAs have been established to even modulate protein expressions and biological phenotypic activity through RNA-protein interactions. Therefore, the current review aimed at summarizing existing literature, clinical trials, and elucidating the important prospect of RNA therapeutics in mitigating the recalcitrant and rare cancers menace.
In 2013, more than a decade ago, the “Recalcitrant Cancer Research Act of 2012” was signed into law in the USA. Recalcitrant cancers are among the leading causes of global cancer morbidity and mortality. At the inception of the act, priority was placed on lungs and pancreatic cancers. Despite the tremendous advancement achieved in the research and treatment of said ‘recalcitrant cancers’ in the form of developing novel or modified small molecule and antibody drugs, modest improvement has been recorded for patients’ survival. Also, current mortality and morbidity for recalcitrant cancers keep increasing. Similarly, rare cancers only enjoy very meager research and drug development efforts globally. Consequently, very limited advancement has been made towards therapeutic development targeting rare cancers. Hence, the current situation calls for re-strategizing research efforts and exploring different treatment modalities towards combating recalcitrant and rare cancers. On this note, RNA therapeutics strategy holds a unique and vital prospect because of its propensity to target coding and non-coding RNA transcripts in the biological system. Moreover, RNA therapeutics such as lncRNAs and circRNAs have been established to even modulate protein expressions and biological phenotypic activity through RNA-protein interactions. Therefore, the current review aimed at summarizing existing literature, clinical trials, and elucidating the important prospect of RNA therapeutics in mitigating the recalcitrant and rare cancers menace.
DOI: https://doi.org/10.37349/etat.2026.1002373

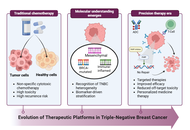
Triple-negative breast cancer (TNBC) is an aggressive breast cancer subtype defined by the absence of estrogen receptors, progesterone receptors, and human epidermal growth factor receptor 2 (HER2) expression. Consequently, standard hormone and HER2-targeted therapies are ineffective, necessitating reliance on chemotherapy, immunotherapy, antibody-drug conjugates (ADCs), and poly(ADP-ribose) polymerase (PARP) inhibitors for BRCA-mutated cases. TNBC exhibits rapid growth, a high risk of early recurrence, and disproportionately affects younger women, Black women, and BRCA1 mutation carriers. Standard management typically involves neoadjuvant chemotherapy followed by surgery and potential radiation. However, TNBC treatment remains challenging due to its severe biological heterogeneity, high metastatic potential, and the toxicity associated with systemic therapies. This review discusses the current understanding of TNBC biology, highlighting the urgent need for advanced diagnostics, integrated molecular subtyping, and personalized targeted therapies.
Triple-negative breast cancer (TNBC) is an aggressive breast cancer subtype defined by the absence of estrogen receptors, progesterone receptors, and human epidermal growth factor receptor 2 (HER2) expression. Consequently, standard hormone and HER2-targeted therapies are ineffective, necessitating reliance on chemotherapy, immunotherapy, antibody-drug conjugates (ADCs), and poly(ADP-ribose) polymerase (PARP) inhibitors for BRCA-mutated cases. TNBC exhibits rapid growth, a high risk of early recurrence, and disproportionately affects younger women, Black women, and BRCA1 mutation carriers. Standard management typically involves neoadjuvant chemotherapy followed by surgery and potential radiation. However, TNBC treatment remains challenging due to its severe biological heterogeneity, high metastatic potential, and the toxicity associated with systemic therapies. This review discusses the current understanding of TNBC biology, highlighting the urgent need for advanced diagnostics, integrated molecular subtyping, and personalized targeted therapies.
DOI: https://doi.org/10.37349/etat.2026.1002372

Aim:
The aim of this study is to investigate the molecular and functional features underlying the clinical heterogeneity between oligometastatic (OM) and polymetastatic (PM) colon cancer.
Methods:
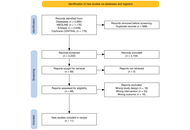
We performed a genotype-phenotype analysis in a homogeneous cohort of 127 patients with metastatic colon cancer (mCC) profiled using the same next-generation sequencing platform (TruSight Oncology® 500). OM disease was defined as the presence of one to three metastatic lesions per involved organ, involving no more than two organs overall, with all lesions measuring < 70 mm in maximum diameter and no single lesion > 25 mm. Molecular alterations, microsatellite instability (MSI), tumor mutational burden (TMB), and overall survival (OS) were analyzed. Gene Ontology (GO) enrichment and Phenolyzer network analyses were applied to explore functional differences between prognostically distinct molecular subgroups.
Results:
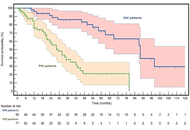
OM patients showed a striking survival advantage compared with PM patients [median OS not reached versus 29 months; hazard ratio (HR): 0.20, P < 0.0001], validating the clinical distinction between the two phenotypes. PM disease was significantly enriched for RAS mutations, whereas OM disease was associated with MSI-high status and elevated TMB. Canonical driver alterations were largely shared between groups, and Phenolyzer analysis revealed similar core oncogenic networks centered on adenomatous polyposis coli (APC), tumor protein p53 (TP53), and epidermal growth factor receptor (EGFR). In contrast, GO analysis demonstrated selective enrichment in PM tumors for molecular functions related to ATP binding, nucleotide binding, and protein kinase activity, consistent with enhanced bioenergetic demand and signaling intensity.
Conclusions:
These findings support refined biological stratification of mCC and the exploration of personalized, metastasis-directed strategies, potentially incorporating immunological modulation in OM disease.
Aim:
The aim of this study is to investigate the molecular and functional features underlying the clinical heterogeneity between oligometastatic (OM) and polymetastatic (PM) colon cancer.
Methods:
We performed a genotype-phenotype analysis in a homogeneous cohort of 127 patients with metastatic colon cancer (mCC) profiled using the same next-generation sequencing platform (TruSight Oncology® 500). OM disease was defined as the presence of one to three metastatic lesions per involved organ, involving no more than two organs overall, with all lesions measuring < 70 mm in maximum diameter and no single lesion > 25 mm. Molecular alterations, microsatellite instability (MSI), tumor mutational burden (TMB), and overall survival (OS) were analyzed. Gene Ontology (GO) enrichment and Phenolyzer network analyses were applied to explore functional differences between prognostically distinct molecular subgroups.
Results:
OM patients showed a striking survival advantage compared with PM patients [median OS not reached versus 29 months; hazard ratio (HR): 0.20, P < 0.0001], validating the clinical distinction between the two phenotypes. PM disease was significantly enriched for RAS mutations, whereas OM disease was associated with MSI-high status and elevated TMB. Canonical driver alterations were largely shared between groups, and Phenolyzer analysis revealed similar core oncogenic networks centered on adenomatous polyposis coli (APC), tumor protein p53 (TP53), and epidermal growth factor receptor (EGFR). In contrast, GO analysis demonstrated selective enrichment in PM tumors for molecular functions related to ATP binding, nucleotide binding, and protein kinase activity, consistent with enhanced bioenergetic demand and signaling intensity.
Conclusions:
These findings support refined biological stratification of mCC and the exploration of personalized, metastasis-directed strategies, potentially incorporating immunological modulation in OM disease.
DOI: https://doi.org/10.37349/etat.2026.1002371

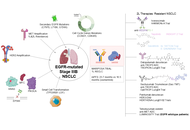
The treatment paradigm for advanced non-small cell lung cancer (NSCLC) harboring EGFR mutations is undergoing a significant transition. While third-generation tyrosine kinase inhibitors (TKIs) like osimertinib have long served as the frontline standard, the emergence of heterogeneous resistance mechanisms requires more robust therapeutic strategies. This review evaluates the clinical impact of the MARIPOSA trial, which demonstrated the superior efficacy of combining the bispecific antibody amivantamab with lazertinib. Beyond improving progression-free and overall survival, this dual-inhibition approach fundamentally alters the clonal evolution of the disease by suppressing common escape routes, such as MET amplifications and secondary EGFR mutations. Furthermore, we explore the diversifying landscape of second-line interventions, including the rise of antibody-drug conjugates (ADCs) like Sac-TMT and patritumab-deruxtecan, dual PD-1/VEGF inhibitors, and novel fourth-generation TKIs. By integrating preclinical insights on drug-tolerant persister cells with late-phase clinical data, this article outlines a future for EGFR-mutant NSCLC management defined by precision sequencing and the proactive mitigation of molecular resistance.
The treatment paradigm for advanced non-small cell lung cancer (NSCLC) harboring EGFR mutations is undergoing a significant transition. While third-generation tyrosine kinase inhibitors (TKIs) like osimertinib have long served as the frontline standard, the emergence of heterogeneous resistance mechanisms requires more robust therapeutic strategies. This review evaluates the clinical impact of the MARIPOSA trial, which demonstrated the superior efficacy of combining the bispecific antibody amivantamab with lazertinib. Beyond improving progression-free and overall survival, this dual-inhibition approach fundamentally alters the clonal evolution of the disease by suppressing common escape routes, such as MET amplifications and secondary EGFR mutations. Furthermore, we explore the diversifying landscape of second-line interventions, including the rise of antibody-drug conjugates (ADCs) like Sac-TMT and patritumab-deruxtecan, dual PD-1/VEGF inhibitors, and novel fourth-generation TKIs. By integrating preclinical insights on drug-tolerant persister cells with late-phase clinical data, this article outlines a future for EGFR-mutant NSCLC management defined by precision sequencing and the proactive mitigation of molecular resistance.
DOI: https://doi.org/10.37349/etat.2026.1002370

Background:
Targeted radioligand therapy (TRT) is an emerging theranostic modality in oncology. While well established in neuroendocrine and prostate cancers, its role in small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC) remains investigational. This systematic review summarizes current evidence evaluating TRT in lung cancer.
Methods:
A Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA)-guided systematic review of PubMed, Embase, and Scopus (2000–November 2025) was conducted. Original studies evaluating TRT in SCLC or NSCLC were included. Primary outcomes were tumor response, disease-control rate, and treatment-related toxicity. Secondary outcomes included progression-free survival, overall survival, and dosimetry. Risk of bias was assessed using the Risk Of Bias In Non-randomized Studies—of Interventions (ROBINS-I) tool.
Results:
From 2,453 records, 15 studies were included, reporting 358 lung cancer patients, of whom 105 received TRT. Disease-control rates reached up to 78% in mixed NSCLC/SCLC cohorts. In SCLC, somatostatin receptor-targeted peptide receptor radionuclide therapy demonstrated heterogeneous disease control (0–50%), with [177Lu]Lu-labeled agents showing more favorable outcomes than [90Y]Y-based therapy. The most favorable outcomes were a median progression-free survival of 11.9 months and an overall survival of 16 months in responders. In NSCLC, fibroblast activation protein (FAP)-targeted agents such as [177Lu]Lu-FAP-2286 demonstrated partial metabolic responses, including a 44.4% response rate and 78% disease control in a mixed cohort. Severe toxicities were infrequent.
Discussion:
TRT is a promising but experimental option for advanced lung cancer. Early efficacy signals exist for strong somatostatin receptor (SSTR)-targeted therapy in SCLC and FAP-targeted therapy in NSCLC, but evidence remains limited. Prospective trials with standardized protocols and dosimetry are needed to define TRT’s role in lung cancer treatment.
Background:
Targeted radioligand therapy (TRT) is an emerging theranostic modality in oncology. While well established in neuroendocrine and prostate cancers, its role in small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC) remains investigational. This systematic review summarizes current evidence evaluating TRT in lung cancer.
Methods:
A Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA)-guided systematic review of PubMed, Embase, and Scopus (2000–November 2025) was conducted. Original studies evaluating TRT in SCLC or NSCLC were included. Primary outcomes were tumor response, disease-control rate, and treatment-related toxicity. Secondary outcomes included progression-free survival, overall survival, and dosimetry. Risk of bias was assessed using the Risk Of Bias In Non-randomized Studies—of Interventions (ROBINS-I) tool.
Results:
From 2,453 records, 15 studies were included, reporting 358 lung cancer patients, of whom 105 received TRT. Disease-control rates reached up to 78% in mixed NSCLC/SCLC cohorts. In SCLC, somatostatin receptor-targeted peptide receptor radionuclide therapy demonstrated heterogeneous disease control (0–50%), with [177Lu]Lu-labeled agents showing more favorable outcomes than [90Y]Y-based therapy. The most favorable outcomes were a median progression-free survival of 11.9 months and an overall survival of 16 months in responders. In NSCLC, fibroblast activation protein (FAP)-targeted agents such as [177Lu]Lu-FAP-2286 demonstrated partial metabolic responses, including a 44.4% response rate and 78% disease control in a mixed cohort. Severe toxicities were infrequent.
Discussion:
TRT is a promising but experimental option for advanced lung cancer. Early efficacy signals exist for strong somatostatin receptor (SSTR)-targeted therapy in SCLC and FAP-targeted therapy in NSCLC, but evidence remains limited. Prospective trials with standardized protocols and dosimetry are needed to define TRT’s role in lung cancer treatment.
DOI: https://doi.org/10.37349/etat.2026.1002368

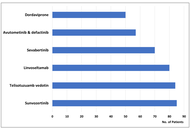
This commentary discusses the FDA’s drug approvals in 2025, with a particular focus on cancer therapies and the role of companion diagnostics (CDx). Cancer has emerged as the leading therapeutic area, accounting for 35% of all new drug approvals, largely driven by targeted therapies, with kinase inhibitors representing nearly half of these drugs. Many of the drugs have received orphan drug designations and/or have utilized the Accelerated Approval Program. A key finding was the widespread adoption of the drug-diagnostic co-development model, in which a CDx assay is developed along with the drug and used for patient selection in clinical trials. However, a significant challenge is the frequent lack of concurrent drug and CDx assay approvals. The absence of an analytically and clinically validated CDx assay may pose a challenge for healthcare providers in accurately identifying eligible patients, potentially delaying access to appropriate therapy. The FDA’s cancer drug approvals for 2025 highlight an ongoing commitment to precision medicine, with several new targeted treatments, such as antibody-drug conjugates and kinase inhibitors, where CDx assays play an important role in identifying the appropriate patient population.
This commentary discusses the FDA’s drug approvals in 2025, with a particular focus on cancer therapies and the role of companion diagnostics (CDx). Cancer has emerged as the leading therapeutic area, accounting for 35% of all new drug approvals, largely driven by targeted therapies, with kinase inhibitors representing nearly half of these drugs. Many of the drugs have received orphan drug designations and/or have utilized the Accelerated Approval Program. A key finding was the widespread adoption of the drug-diagnostic co-development model, in which a CDx assay is developed along with the drug and used for patient selection in clinical trials. However, a significant challenge is the frequent lack of concurrent drug and CDx assay approvals. The absence of an analytically and clinically validated CDx assay may pose a challenge for healthcare providers in accurately identifying eligible patients, potentially delaying access to appropriate therapy. The FDA’s cancer drug approvals for 2025 highlight an ongoing commitment to precision medicine, with several new targeted treatments, such as antibody-drug conjugates and kinase inhibitors, where CDx assays play an important role in identifying the appropriate patient population.
DOI: https://doi.org/10.37349/etat.2026.1002369
This article belongs to the special issue Precision Oncology: Molecular Classification, Efficacy Prediction, and Treatment Decision-Making

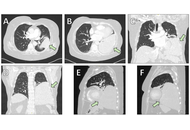
Solitary fibrous tumors (SFTs) are rare mesenchymal neoplasms that typically arise from the pleura but may occur in various extrathoracic sites. Primary intraparenchymal pulmonary SFTs without pleural attachment are exceptionally uncommon and often pose diagnostic and therapeutic challenges. We report the case of a middle-aged female patient presenting with progressive dyspnea and a large mass in the left lower lobe on imaging. Computed tomography revealed a well-circumscribed, hypervascular mass occupying the left lower lobe. Bronchoscopic and percutaneous biopsies were nondiagnostic, and surgical resection was pursued. Intraoperatively, the tumor was found to arise from the lung parenchyma without pleural involvement. Histopathological examination demonstrated a spindle-cell neoplasm with the typical “patternless pattern,” and immunohistochemistry confirmed nuclear STAT6 positivity, establishing the diagnosis of SFT. The postoperative course was uneventful apart from a transient pulmonary embolism, which was successfully treated. The patient was discharged in good condition and is under regular radiologic surveillance. SFTs of the lung are rare and often mimic more common pulmonary tumors radiologically. Histologic confirmation with STAT6 immunohistochemistry is crucial for accurate diagnosis. Complete surgical excision remains the mainstay of treatment. Given the risk of late recurrence—especially in large tumors—long-term imaging follow-up is mandatory. This case highlights the importance of considering SFT in the differential diagnosis of large pulmonary masses, the critical role of STAT6-based histopathologic confirmation, and the necessity for prolonged surveillance even after complete resection.
Solitary fibrous tumors (SFTs) are rare mesenchymal neoplasms that typically arise from the pleura but may occur in various extrathoracic sites. Primary intraparenchymal pulmonary SFTs without pleural attachment are exceptionally uncommon and often pose diagnostic and therapeutic challenges. We report the case of a middle-aged female patient presenting with progressive dyspnea and a large mass in the left lower lobe on imaging. Computed tomography revealed a well-circumscribed, hypervascular mass occupying the left lower lobe. Bronchoscopic and percutaneous biopsies were nondiagnostic, and surgical resection was pursued. Intraoperatively, the tumor was found to arise from the lung parenchyma without pleural involvement. Histopathological examination demonstrated a spindle-cell neoplasm with the typical “patternless pattern,” and immunohistochemistry confirmed nuclear STAT6 positivity, establishing the diagnosis of SFT. The postoperative course was uneventful apart from a transient pulmonary embolism, which was successfully treated. The patient was discharged in good condition and is under regular radiologic surveillance. SFTs of the lung are rare and often mimic more common pulmonary tumors radiologically. Histologic confirmation with STAT6 immunohistochemistry is crucial for accurate diagnosis. Complete surgical excision remains the mainstay of treatment. Given the risk of late recurrence—especially in large tumors—long-term imaging follow-up is mandatory. This case highlights the importance of considering SFT in the differential diagnosis of large pulmonary masses, the critical role of STAT6-based histopathologic confirmation, and the necessity for prolonged surveillance even after complete resection.
DOI: https://doi.org/10.37349/etat.2026.1002367

Aim:
This study aimed to evaluate the real-world efficacy and safety of lorlatinib in patients with anaplastic lymphoma kinase (ALK)-rearranged metastatic non-small cell lung cancer (NSCLC) after the failure of at least one prior ALK tyrosine kinase inhibitor (TKI).
Methods:
The dataset included 82 subjects with metastatic NSCLC, who received lorlatinib upon compassionate use program or routine treatment between January 2017 and May 2025. All patients involved in this study responded to a prior ALK inhibitor for at least 4 months and switched to the above drug due to disease progression.
Results:
The overall objective response rate (ORR) was 64.6%, with the disease control rate (DCR) of 96.3%. Among 65 patients with brain metastases, the intracranial ORR and DCR were 66.2% and 96.9%, respectively. After a median follow-up of 82.7 months, the median progression-free survival (PFS) was 66.7 months (95% CI, 40.5–75.0 months), while the median overall survival (OS) was not reached (NR) (95% CI, NR–NR). Patients who had benefited from prior ALK TKI for more than 12 months achieved significantly longer PFS (NR vs. 34.0 months; p = 0.013) and OS (NR vs. 39.4 months; p = 0.002). Multivariate analysis showed that prior response to ALK TKI of less than 12 months was an independent negative predictor of survival (PFS: p = 0.039, OS: p = 0.027). Treatment-related adverse events (AEs) were reported in 75.6% of patients, with 8.1% experiencing grade 3 or higher toxicity; no treatment-related AEs led to permanent discontinuation of lorlatinib.
Conclusions:
This real-world dataset demonstrates an unusually pronounced benefit from lorlatinib in selected patients who progressed on early-generation TKIs, especially in long-term responders to prior therapy. However, the observed outcomes should be interpreted within the context of patient selection. The enrichment for prior responders limits the generalizability to unselected post-TKI populations, including those with primary resistance.
Aim:
This study aimed to evaluate the real-world efficacy and safety of lorlatinib in patients with anaplastic lymphoma kinase (ALK)-rearranged metastatic non-small cell lung cancer (NSCLC) after the failure of at least one prior ALK tyrosine kinase inhibitor (TKI).
Methods:
The dataset included 82 subjects with metastatic NSCLC, who received lorlatinib upon compassionate use program or routine treatment between January 2017 and May 2025. All patients involved in this study responded to a prior ALK inhibitor for at least 4 months and switched to the above drug due to disease progression.
Results:
The overall objective response rate (ORR) was 64.6%, with the disease control rate (DCR) of 96.3%. Among 65 patients with brain metastases, the intracranial ORR and DCR were 66.2% and 96.9%, respectively. After a median follow-up of 82.7 months, the median progression-free survival (PFS) was 66.7 months (95% CI, 40.5–75.0 months), while the median overall survival (OS) was not reached (NR) (95% CI, NR–NR). Patients who had benefited from prior ALK TKI for more than 12 months achieved significantly longer PFS (NR vs. 34.0 months; p = 0.013) and OS (NR vs. 39.4 months; p = 0.002). Multivariate analysis showed that prior response to ALK TKI of less than 12 months was an independent negative predictor of survival (PFS: p = 0.039, OS: p = 0.027). Treatment-related adverse events (AEs) were reported in 75.6% of patients, with 8.1% experiencing grade 3 or higher toxicity; no treatment-related AEs led to permanent discontinuation of lorlatinib.
Conclusions:
This real-world dataset demonstrates an unusually pronounced benefit from lorlatinib in selected patients who progressed on early-generation TKIs, especially in long-term responders to prior therapy. However, the observed outcomes should be interpreted within the context of patient selection. The enrichment for prior responders limits the generalizability to unselected post-TKI populations, including those with primary resistance.
DOI: https://doi.org/10.37349/etat.2026.1002366

Aim:
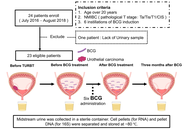
Intravesical Bacillus Calmette–Guérin (BCG) is the standard therapy for non-muscle invasive bladder cancer (NMIBC); however, many patients experience recurrence or progression. We examined how urinary immune signals and the urinary microbiome change across BCG and are related to outcomes.
Methods:
In this single-center prospective cohort study, adults with NMIBC underwent transurethral resection of bladder tumor (TURBT), followed by BCG induction. Urine was collected before TURBT, before BCG, after BCG induction, and three months later. Urine sediment mRNA (PD-L1, PD-L2, CD33, and CD204) was quantified using TaqMan ΔCt. The urinary microbiome was profiled using 16S rRNA gene sequencing, and diversity, composition, and taxon balance were evaluated using nonparametric tests, PERMANOVA, repeated-measures correlations, and mixed-effects models. We analyzed the relationship between the urinary microbiome and prognosis.
Results:
Twenty-three patients were analyzed; ten recurrences, eight progressions, and three deaths were observed. Relative to baseline, CD33 increased after BCG and after three months; PD-L2 increased immediately after BCG and returned to baseline by three months; PD-L1 and CD204 increased after BCG. Shannon alpha-diversity was unchanged, but total read count was higher at three months, with stable beta-diversity and dispersion. Higher PD-L1 expression was associated with lower Actinobacteria abundance in the bladder cancer microenvironment. A higher post-BCG Firmicutes/Bacteroidetes ratio was associated with worse prognosis, with the clearest signal for progression-free survival (PFS) observed in the univariate Cox models. Higher post-BCG Corynebacterium and Enterobacteriaceae abundance was associated with better PFS.
Conclusions:
BCG was associated with higher urinary PD-L1/PD-L2 and myeloid marker transcripts, while overall community structure remained stable. These exploratory data support that pre-BCG microbial features may be related to early response, and post-BCG profiles may reflect durability and survival. Urine immune-microbiome profiling could be a feasible, noninvasive adjunct for monitoring and risk stratification in NMIBC.
Aim:
Intravesical Bacillus Calmette–Guérin (BCG) is the standard therapy for non-muscle invasive bladder cancer (NMIBC); however, many patients experience recurrence or progression. We examined how urinary immune signals and the urinary microbiome change across BCG and are related to outcomes.
Methods:
In this single-center prospective cohort study, adults with NMIBC underwent transurethral resection of bladder tumor (TURBT), followed by BCG induction. Urine was collected before TURBT, before BCG, after BCG induction, and three months later. Urine sediment mRNA (PD-L1, PD-L2, CD33, and CD204) was quantified using TaqMan ΔCt. The urinary microbiome was profiled using 16S rRNA gene sequencing, and diversity, composition, and taxon balance were evaluated using nonparametric tests, PERMANOVA, repeated-measures correlations, and mixed-effects models. We analyzed the relationship between the urinary microbiome and prognosis.
Results:
Twenty-three patients were analyzed; ten recurrences, eight progressions, and three deaths were observed. Relative to baseline, CD33 increased after BCG and after three months; PD-L2 increased immediately after BCG and returned to baseline by three months; PD-L1 and CD204 increased after BCG. Shannon alpha-diversity was unchanged, but total read count was higher at three months, with stable beta-diversity and dispersion. Higher PD-L1 expression was associated with lower Actinobacteria abundance in the bladder cancer microenvironment. A higher post-BCG Firmicutes/Bacteroidetes ratio was associated with worse prognosis, with the clearest signal for progression-free survival (PFS) observed in the univariate Cox models. Higher post-BCG Corynebacterium and Enterobacteriaceae abundance was associated with better PFS.
Conclusions:
BCG was associated with higher urinary PD-L1/PD-L2 and myeloid marker transcripts, while overall community structure remained stable. These exploratory data support that pre-BCG microbial features may be related to early response, and post-BCG profiles may reflect durability and survival. Urine immune-microbiome profiling could be a feasible, noninvasive adjunct for monitoring and risk stratification in NMIBC.
DOI: https://doi.org/10.37349/etat.2026.1002365

Aim:
Glioblastoma (GBM), a rare, highly aggressive and chemoresistant brain cancer, exhibits profound metabolic plasticity that relies, in part, on aberrant transforming growth factor-β (TGF-β) signaling. Such plasticity was recently associated with TGF-β-regulated apoptosis and autophagy. Here, we questioned whether TGF-β-regulated apoptotic/autophagic phenotypes are recapitulated in a preclinical in vitro 3D spheroid culture model of human U87 GBM-derived cells, and how metabolic alterations affect such phenotypes.
Methods:
3D U87 spheroids were cultured using the hanging drop method. Western blotting was used to assess protein expression, while RT-qPCR was used to assess gene expression levels.
Results:
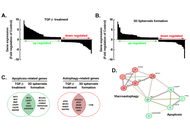
3D spheroids exhibited decreased AKT phosphorylation, and increased TGF-β, fibronectin, and Smad2 phosphorylation, indicative of both cell death signaling and epithelial-mesenchymal transition molecular signatures. 2-Deoxy-D-glucose (2DG), a glycolytic inhibitor, depleted ATP dose-dependently (30–300 μM) and prevented those increases both at the protein and transcriptional levels. This was also observed in 3D spheroids upon TGF-β transient siRNA-mediated silencing or when TGF-βR1 kinase activity was inhibited by galunisertib. Transcriptomic profiling revealed shared upregulation of apoptosis-related (BCL2, CASP7, FAS, FASLG, GADD45A) and autophagy-related (ATG7, ATG16L1, IRGM, PIK3C3, ULK1) genes in response to TGF-β or upon 3D spheroid formation. 2DG, transient silencing of TGF-β, or galunisertib treatment prevented these increases.
Conclusions:
3D spheroids require ATP and a TGF-β/TGF-βR1 autocrine signaling axis to recapitulate the apoptosis/autophagy phenotypes. Combining glycolysis inhibition with TGF-β signaling inhibition could offer a promising therapeutic strategy for this rare and lethal brain cancer.
Aim:
Glioblastoma (GBM), a rare, highly aggressive and chemoresistant brain cancer, exhibits profound metabolic plasticity that relies, in part, on aberrant transforming growth factor-β (TGF-β) signaling. Such plasticity was recently associated with TGF-β-regulated apoptosis and autophagy. Here, we questioned whether TGF-β-regulated apoptotic/autophagic phenotypes are recapitulated in a preclinical in vitro 3D spheroid culture model of human U87 GBM-derived cells, and how metabolic alterations affect such phenotypes.
Methods:
3D U87 spheroids were cultured using the hanging drop method. Western blotting was used to assess protein expression, while RT-qPCR was used to assess gene expression levels.
Results:
3D spheroids exhibited decreased AKT phosphorylation, and increased TGF-β, fibronectin, and Smad2 phosphorylation, indicative of both cell death signaling and epithelial-mesenchymal transition molecular signatures. 2-Deoxy-D-glucose (2DG), a glycolytic inhibitor, depleted ATP dose-dependently (30–300 μM) and prevented those increases both at the protein and transcriptional levels. This was also observed in 3D spheroids upon TGF-β transient siRNA-mediated silencing or when TGF-βR1 kinase activity was inhibited by galunisertib. Transcriptomic profiling revealed shared upregulation of apoptosis-related (BCL2, CASP7, FAS, FASLG, GADD45A) and autophagy-related (ATG7, ATG16L1, IRGM, PIK3C3, ULK1) genes in response to TGF-β or upon 3D spheroid formation. 2DG, transient silencing of TGF-β, or galunisertib treatment prevented these increases.
Conclusions:
3D spheroids require ATP and a TGF-β/TGF-βR1 autocrine signaling axis to recapitulate the apoptosis/autophagy phenotypes. Combining glycolysis inhibition with TGF-β signaling inhibition could offer a promising therapeutic strategy for this rare and lethal brain cancer.
DOI: https://doi.org/10.37349/etat.2026.1002364
This article belongs to the special issue Novel Precision Medicine Approaches to Brain Tumors (Primary and Metastatic)

Aim:
Chromodomain-helicase-DNA-binding protein 4 (CHD4) is a core NURD remodeling complex ATPase that plays a crucial role as a gene repressor. Its overexpression has been reported in several cancers. In papillary thyroid carcinomas (PTCs), CHD4 is overexpressed and associated with aggressive features of the tumor, such as proliferation, migration, and epithelial-mesenchymal transition (EMT). We previously showed in PTCs that NADPH oxidase NOX4 expression is positively regulated by BRAFV600E mutation, which is the most aggressive alteration in PTCs. In this retrospective study, we wondered whether there is a link between CHD4 and NOX4 protein expression in malignant thyroid tissues.
Methods:
We explored CHD4 protein expression by immunostaining analysis in 86 human thyroid tissues: 44 thyroid tumor tissues [28 classical forms of PTCs (C-PTCs), 13 follicular variants of PTCs (F-PTCs), and three anaplastic thyroid carcinomas (ATCs)] and 42 of their normal adjacent tissues (NATs). The detection of BRAFV600E mutation was performed using Sanger sequencing and digital droplet PCR. Statistical analyses were conducted using GraphPad Prism 8 software. Various tests were used to assess the statistical relevance of different correlations, such as the chi-square test, Fisher’s exact test, and the Pearson correlation coefficient. A p-value of less than 0.05 indicates statistical significance.
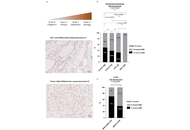
Results:
The CHD4 protein expression analysis with already published data from our group (BRAFV600E status and NOX4 expression) reveals a highly significant level of CHD4 protein expression in C-PTCs compared to F-PTCs and ATC. Importantly, 70% of C-PTCs-BRAFV600E overexpress CHD4 at the protein level, confirming the positive correlation between the CHD4 expression and BRAFV600E mutation. Furthermore, a high level of CHD4 is associated with the presence of capsular breach and vascular emboli, affirming the involvement of CHD4 in thyroid tumor aggressiveness. Interestingly, we showed for the first time, to our knowledge, a positive correlation between CHD4 and NOX4 protein expression in malignant thyroid tissues.
Conclusions:
The results of this study suggest that CHD4 could be used as a complementary molecular marker to improve the diagnosis and the management of PTCs-BRAFV600E.
Aim:
Chromodomain-helicase-DNA-binding protein 4 (CHD4) is a core NURD remodeling complex ATPase that plays a crucial role as a gene repressor. Its overexpression has been reported in several cancers. In papillary thyroid carcinomas (PTCs), CHD4 is overexpressed and associated with aggressive features of the tumor, such as proliferation, migration, and epithelial-mesenchymal transition (EMT). We previously showed in PTCs that NADPH oxidase NOX4 expression is positively regulated by BRAFV600E mutation, which is the most aggressive alteration in PTCs. In this retrospective study, we wondered whether there is a link between CHD4 and NOX4 protein expression in malignant thyroid tissues.
Methods:
We explored CHD4 protein expression by immunostaining analysis in 86 human thyroid tissues: 44 thyroid tumor tissues [28 classical forms of PTCs (C-PTCs), 13 follicular variants of PTCs (F-PTCs), and three anaplastic thyroid carcinomas (ATCs)] and 42 of their normal adjacent tissues (NATs). The detection of BRAFV600E mutation was performed using Sanger sequencing and digital droplet PCR. Statistical analyses were conducted using GraphPad Prism 8 software. Various tests were used to assess the statistical relevance of different correlations, such as the chi-square test, Fisher’s exact test, and the Pearson correlation coefficient. A p-value of less than 0.05 indicates statistical significance.
Results:
The CHD4 protein expression analysis with already published data from our group (BRAFV600E status and NOX4 expression) reveals a highly significant level of CHD4 protein expression in C-PTCs compared to F-PTCs and ATC. Importantly, 70% of C-PTCs-BRAFV600E overexpress CHD4 at the protein level, confirming the positive correlation between the CHD4 expression and BRAFV600E mutation. Furthermore, a high level of CHD4 is associated with the presence of capsular breach and vascular emboli, affirming the involvement of CHD4 in thyroid tumor aggressiveness. Interestingly, we showed for the first time, to our knowledge, a positive correlation between CHD4 and NOX4 protein expression in malignant thyroid tissues.
Conclusions:
The results of this study suggest that CHD4 could be used as a complementary molecular marker to improve the diagnosis and the management of PTCs-BRAFV600E.
DOI: https://doi.org/10.37349/etat.2026.1002363

Aim:
Triple-negative breast cancer (TNBC) is an aggressive subtype with limited therapeutic options and poor survival outcomes. Prognostic models developed in Western cohorts rarely assess algorithmic fairness. This study aimed to develop and internally validate a clinically interpretable Cox survival model for TNBC using baseline diagnostic variables and to evaluate its fairness according to ISO/IEC TR 24027:2021 guidelines in a Middle East and North Africa (MENA) cohort.
Methods:
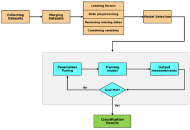
A total of 138 TNBC patients were included after merging two institutional datasets and removing variables with > 25% missingness. Baseline features comprised age, tumor size, lymph node involvement, tumor grade, Ki-67, type of surgery, metastasis at diagnosis, chemotherapy, and radiotherapy. A Cox proportional hazards (CoxPH) model with six clinically established predictors was fitted to reduce overfitting. Model performance was assessed through five-fold stratified cross-validation using Harrell’s concordance index (C-index), receiver operating characteristic area under the curve (AUROC), and calibration curves. Fairness was evaluated using demographic parity, equality of opportunity, predictive equality, and equalized odds metrics following ISO/IEC TR 24027:2021.
Results:
During follow-up, 34 patients (24.6%) died. Metastasis at diagnosis, high tumor grade, and radical mastectomy were significantly associated with mortality. The CoxPH model achieved a C-index of 0.80 [SE = 0.04; 95% confidence interval (CI): 0.72–0.87] and an AUROC of 0.81 (95% CI: 0.72–0.90). Calibration plots showed strong agreement between predicted and observed survival probabilities, with a modest overall bias of –8.8%. Fairness assessment revealed small but notable disparities in false-positive rates across age groups and surgical categories, while lymph node status and other variables showed no significant bias.
Conclusions:
This study presents a robust and fairness-aware survival prediction model for TNBC using routinely available clinical features. The model demonstrates strong discrimination, good calibration, and quantifiable fairness across patient subgroups, offering a clinically interpretable and ethically aligned tool to support TNBC risk stratification and decision-making in the MENA region.
Aim:
Triple-negative breast cancer (TNBC) is an aggressive subtype with limited therapeutic options and poor survival outcomes. Prognostic models developed in Western cohorts rarely assess algorithmic fairness. This study aimed to develop and internally validate a clinically interpretable Cox survival model for TNBC using baseline diagnostic variables and to evaluate its fairness according to ISO/IEC TR 24027:2021 guidelines in a Middle East and North Africa (MENA) cohort.
Methods:
A total of 138 TNBC patients were included after merging two institutional datasets and removing variables with > 25% missingness. Baseline features comprised age, tumor size, lymph node involvement, tumor grade, Ki-67, type of surgery, metastasis at diagnosis, chemotherapy, and radiotherapy. A Cox proportional hazards (CoxPH) model with six clinically established predictors was fitted to reduce overfitting. Model performance was assessed through five-fold stratified cross-validation using Harrell’s concordance index (C-index), receiver operating characteristic area under the curve (AUROC), and calibration curves. Fairness was evaluated using demographic parity, equality of opportunity, predictive equality, and equalized odds metrics following ISO/IEC TR 24027:2021.
Results:
During follow-up, 34 patients (24.6%) died. Metastasis at diagnosis, high tumor grade, and radical mastectomy were significantly associated with mortality. The CoxPH model achieved a C-index of 0.80 [SE = 0.04; 95% confidence interval (CI): 0.72–0.87] and an AUROC of 0.81 (95% CI: 0.72–0.90). Calibration plots showed strong agreement between predicted and observed survival probabilities, with a modest overall bias of –8.8%. Fairness assessment revealed small but notable disparities in false-positive rates across age groups and surgical categories, while lymph node status and other variables showed no significant bias.
Conclusions:
This study presents a robust and fairness-aware survival prediction model for TNBC using routinely available clinical features. The model demonstrates strong discrimination, good calibration, and quantifiable fairness across patient subgroups, offering a clinically interpretable and ethically aligned tool to support TNBC risk stratification and decision-making in the MENA region.
DOI: https://doi.org/10.37349/etat.2026.1002362

Breast cancer is a leading cause of cancer death in women worldwide. One of the major causes of death from breast cancer is metastatic disease, which results from the malignant cells invading and migrating through blood vessels to distant sites. Several studies have shown that metastasis is facilitated by haemostatic proteins. Breast cancer is characterized by a haemostatic imbalance, which is tilted more to a procoagulant state with resultant thrombotic complications. These elements that are involved in thrombosis also play key roles in different aspects of breast cancer growth, including cancer proliferation and progression, cancer survival, angiogenesis, and metastasis. Some of these elements include platelets, endothelial cells, coagulation factors, and fibrinolytic proteins. There is a close relationship between cancer and many of the haemostatic elements. They are usually increased in metastatic breast cancer and have found use as predictive and prognostic markers. Some have been validated in breast cancer. Due to their seemingly active roles in breast cancer progression, some of the haemostatic proteins are being developed as diagnostic tools in the management of breast cancer. They are equally seen as potential targets for the development of novel therapies in breast cancer or repurposing drugs in current use for the same gain. This review highlights the role haemostatic proteins play in breast cancer progression, and their diagnostic and therapeutic relevance.
Breast cancer is a leading cause of cancer death in women worldwide. One of the major causes of death from breast cancer is metastatic disease, which results from the malignant cells invading and migrating through blood vessels to distant sites. Several studies have shown that metastasis is facilitated by haemostatic proteins. Breast cancer is characterized by a haemostatic imbalance, which is tilted more to a procoagulant state with resultant thrombotic complications. These elements that are involved in thrombosis also play key roles in different aspects of breast cancer growth, including cancer proliferation and progression, cancer survival, angiogenesis, and metastasis. Some of these elements include platelets, endothelial cells, coagulation factors, and fibrinolytic proteins. There is a close relationship between cancer and many of the haemostatic elements. They are usually increased in metastatic breast cancer and have found use as predictive and prognostic markers. Some have been validated in breast cancer. Due to their seemingly active roles in breast cancer progression, some of the haemostatic proteins are being developed as diagnostic tools in the management of breast cancer. They are equally seen as potential targets for the development of novel therapies in breast cancer or repurposing drugs in current use for the same gain. This review highlights the role haemostatic proteins play in breast cancer progression, and their diagnostic and therapeutic relevance.
DOI: https://doi.org/10.37349/etat.2026.1002361
This article belongs to the special issue Breaking Boundaries in Breast Cancer Care: Emerging Controversies and Innovation in Surgical and Medical Approaches

Background:
Docetaxel is a cornerstone chemotherapy for metastatic hormone-sensitive and castration-resistant prostate cancer. Although the standard triweekly regimen is widely used, weekly and biweekly schedules are often employed to improve tolerability, particularly in elderly or frail patients. The comparative efficacy and safety of these dosing strategies remain unclear. This study aimed to systematically compare weekly, biweekly, and triweekly docetaxel regimens using a network meta-analysis.
Methods:
MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials were searched from inception to February 2025. Randomized controlled trials and observational retrospective studies comparing weekly, biweekly, and triweekly docetaxel regimens were included. Outcomes assessed were prostate-specific antigen (PSA) response rate, time to treatment failure or progression, and adverse events. A frequentist random-effects network meta-analysis was conducted using R software.
Results:
Eleven studies involving 1,238 patients were included. PSA response rates did not differ significantly among regimens; triweekly docetaxel showed a numerically lower response compared with weekly dosing (RR = 0.79, 95% CI 0.52–1.22; I2 = 41.1%). Time to treatment failure was significantly longer with triweekly dosing compared with weekly dosing (mean difference = 10.91 months, 95% CI 6.94–14.87; I2 = 96.8%). Biweekly and triweekly regimens were associated with significantly higher hepatotoxicity compared with weekly dosing (RR = 3.71 and RR = 3.21, respectively; I2 = 0%). Vomiting was more frequent with triweekly docetaxel (RR = 2.47, 95% CI 1.31–4.63). No significant differences were observed for overall adverse events, hematologic toxicity, neuropathy, fatigue, febrile neutropenia, nausea, anorexia, or diarrhea.
Discussion:
Docetaxel dosing schedules show comparable PSA response rates. Triweekly dosing prolongs time to treatment failure but is associated with greater toxicity, whereas weekly dosing offers better tolerability. Treatment decisions should balance efficacy and safety based on individual patient characteristics.
Background:
Docetaxel is a cornerstone chemotherapy for metastatic hormone-sensitive and castration-resistant prostate cancer. Although the standard triweekly regimen is widely used, weekly and biweekly schedules are often employed to improve tolerability, particularly in elderly or frail patients. The comparative efficacy and safety of these dosing strategies remain unclear. This study aimed to systematically compare weekly, biweekly, and triweekly docetaxel regimens using a network meta-analysis.
Methods:
MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials were searched from inception to February 2025. Randomized controlled trials and observational retrospective studies comparing weekly, biweekly, and triweekly docetaxel regimens were included. Outcomes assessed were prostate-specific antigen (PSA) response rate, time to treatment failure or progression, and adverse events. A frequentist random-effects network meta-analysis was conducted using R software.
Results:
Eleven studies involving 1,238 patients were included. PSA response rates did not differ significantly among regimens; triweekly docetaxel showed a numerically lower response compared with weekly dosing (RR = 0.79, 95% CI 0.52–1.22; I2 = 41.1%). Time to treatment failure was significantly longer with triweekly dosing compared with weekly dosing (mean difference = 10.91 months, 95% CI 6.94–14.87; I2 = 96.8%). Biweekly and triweekly regimens were associated with significantly higher hepatotoxicity compared with weekly dosing (RR = 3.71 and RR = 3.21, respectively; I2 = 0%). Vomiting was more frequent with triweekly docetaxel (RR = 2.47, 95% CI 1.31–4.63). No significant differences were observed for overall adverse events, hematologic toxicity, neuropathy, fatigue, febrile neutropenia, nausea, anorexia, or diarrhea.
Discussion:
Docetaxel dosing schedules show comparable PSA response rates. Triweekly dosing prolongs time to treatment failure but is associated with greater toxicity, whereas weekly dosing offers better tolerability. Treatment decisions should balance efficacy and safety based on individual patient characteristics.
DOI: https://doi.org/10.37349/etat.2026.1002360

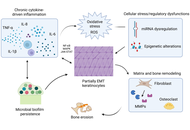
Acquired middle-ear cholesteatoma is a histologically benign keratinizing squamous epithelial lesion that paradoxically exhibits locally destructive, recurrent, and invasive behavior, often resulting in ossicular erosion, hearing loss, labyrinthine fistula, and, rarely, intracranial complications. Surgical excision remains the primary management strategy; however, recurrence is common due to persistent microenvironmental drivers. Recent mechanistic studies—including single-cell transcriptomics, spatial proteomics, and epigenetic profiling—reveal a multifactorial pathogenesis orchestrated by chronic inflammation, proteolytic extracellular-matrix remodeling, osteoclast activation via RANKL and activin A, epithelial plasticity with partial epithelial-to-mesenchymal transition (EMT), and a dysbiotic, biofilm-forming microbiome. Emerging evidence further implicates oxidative stress, RNA and epigenetic modifications, miRNA dysregulation, and immune cell infiltration as central modulators of lesion chronicity and bone resorption. Collectively, these processes establish a self-sustaining pro-osteolytic microenvironment that drives bone erosion and postoperative recurrence. Cholesteatoma recapitulates several features of malignant lesions—hyperproliferation, local invasion, and stromal/immune cell recruitment—yet remains fundamentally benign, lacking metastatic potential and genomic instability. Its aggression is ecological rather than genetic, highlighting the potential for microenvironment-directed, precision-based strategies. Adjunctive approaches may include local delivery of modulatory agents, targeted interference with inflammatory, proteolytic, osteoclastogenic, and microbial axes, and biomarker-guided patient stratification. Preclinical and early-phase experimental studies assessing target engagement, radiologic stabilization, and molecular surrogates of efficacy could inform safer, mechanism-driven interventions that complement surgery, reduce recurrence, and preserve hearing. Integrating molecular pathobiology with clinical strategy positions cholesteatoma as a model for benign yet locally aggressive, microenvironment-driven disease, providing a roadmap for translational therapies with direct relevance to surgical practice.
Acquired middle-ear cholesteatoma is a histologically benign keratinizing squamous epithelial lesion that paradoxically exhibits locally destructive, recurrent, and invasive behavior, often resulting in ossicular erosion, hearing loss, labyrinthine fistula, and, rarely, intracranial complications. Surgical excision remains the primary management strategy; however, recurrence is common due to persistent microenvironmental drivers. Recent mechanistic studies—including single-cell transcriptomics, spatial proteomics, and epigenetic profiling—reveal a multifactorial pathogenesis orchestrated by chronic inflammation, proteolytic extracellular-matrix remodeling, osteoclast activation via RANKL and activin A, epithelial plasticity with partial epithelial-to-mesenchymal transition (EMT), and a dysbiotic, biofilm-forming microbiome. Emerging evidence further implicates oxidative stress, RNA and epigenetic modifications, miRNA dysregulation, and immune cell infiltration as central modulators of lesion chronicity and bone resorption. Collectively, these processes establish a self-sustaining pro-osteolytic microenvironment that drives bone erosion and postoperative recurrence. Cholesteatoma recapitulates several features of malignant lesions—hyperproliferation, local invasion, and stromal/immune cell recruitment—yet remains fundamentally benign, lacking metastatic potential and genomic instability. Its aggression is ecological rather than genetic, highlighting the potential for microenvironment-directed, precision-based strategies. Adjunctive approaches may include local delivery of modulatory agents, targeted interference with inflammatory, proteolytic, osteoclastogenic, and microbial axes, and biomarker-guided patient stratification. Preclinical and early-phase experimental studies assessing target engagement, radiologic stabilization, and molecular surrogates of efficacy could inform safer, mechanism-driven interventions that complement surgery, reduce recurrence, and preserve hearing. Integrating molecular pathobiology with clinical strategy positions cholesteatoma as a model for benign yet locally aggressive, microenvironment-driven disease, providing a roadmap for translational therapies with direct relevance to surgical practice.
DOI: https://doi.org/10.37349/etat.2026.1002359

Glioblastoma (GBM) is a complex condition with a poorly understood pathophysiology and no effective treatment to date. The present article highlights the role of canonical and non-canonical signal transducer and activator of transcription 3 (STAT3) interactions with nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) in the modulation of the mitochondrial melatonergic pathway in GBM microenvironment pathophysiology. The capacity of STAT3 and NF-κB to interact to upregulate the mitochondrial melatonergic pathway is suppressed systemically over the course of aging, thereby attenuating the capacity to achieve inflammation resolution. The suppressed capacity to induce the mitochondrial melatonergic pathway systemically is partly driven by the dramatic 10-fold decrease in pineal melatonin over aging. The attenuation of pineal melatonin in the first half of sleep over aging and aging-accelerating conditions disinhibits the effects of cortisol in the second half of sleep. This decrease in the melatonin/cortisol ratio alters the nature of night-time dampening and resetting in preparation for the coming day by altering cellular and intercellular homeostatic interactions. Aging and aging-accelerating conditions, by impacting the night-time melatonin/cortisol ratio, also suppress the capacity of the vagal nerve to resolve inflammation. This further contributes to systemic changes that influence GBM pathoetiology and ongoing pathophysiology. Aging-associated changes in night-time dampening and resetting provide a novel framework on which many previously disparate bodies of data on GBM pathophysiology can be collated. This has numerous future research, prevention, and treatment implications.
Glioblastoma (GBM) is a complex condition with a poorly understood pathophysiology and no effective treatment to date. The present article highlights the role of canonical and non-canonical signal transducer and activator of transcription 3 (STAT3) interactions with nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) in the modulation of the mitochondrial melatonergic pathway in GBM microenvironment pathophysiology. The capacity of STAT3 and NF-κB to interact to upregulate the mitochondrial melatonergic pathway is suppressed systemically over the course of aging, thereby attenuating the capacity to achieve inflammation resolution. The suppressed capacity to induce the mitochondrial melatonergic pathway systemically is partly driven by the dramatic 10-fold decrease in pineal melatonin over aging. The attenuation of pineal melatonin in the first half of sleep over aging and aging-accelerating conditions disinhibits the effects of cortisol in the second half of sleep. This decrease in the melatonin/cortisol ratio alters the nature of night-time dampening and resetting in preparation for the coming day by altering cellular and intercellular homeostatic interactions. Aging and aging-accelerating conditions, by impacting the night-time melatonin/cortisol ratio, also suppress the capacity of the vagal nerve to resolve inflammation. This further contributes to systemic changes that influence GBM pathoetiology and ongoing pathophysiology. Aging-associated changes in night-time dampening and resetting provide a novel framework on which many previously disparate bodies of data on GBM pathophysiology can be collated. This has numerous future research, prevention, and treatment implications.
DOI: https://doi.org/10.37349/etat.2026.1002358

Machine learning (ML) and deep learning (DL) models applied to electronic health records (EHRs) have substantial potential to improve oncology care across diagnosis, prognosis, treatment selection, and trial recruitment. However, opacity of many high-performing models limits clinician trust, regulatory acceptance, and safe deployment. Explainable artificial intelligence (XAI) methods aim to make model behavior understandable and actionable in clinical contexts. The present perspective summarizes current XAI approaches applied to EHR-based oncology tasks, identifies key challenges in evaluation, reproducibility, clinical utility, and equity, and proposes pragmatic recommendations and research directions to accelerate safe adoption in oncology. Common XAI categories used with EHR data include feature importance/interaction methods, intrinsically interpretable models, attention mechanisms, dimensionality reduction, and knowledge distillation or rule extraction. Tree-based models with SHapley Additive exPlanations (SHAP) explanations dominate recent EHR studies. Other interpretable strategies, such as generalized additive models and rule sets, appear in settings where transparency is prioritized. Gaps include inconsistent reporting, scarce formal evaluation of explanations for clinical utility, limited reproducibility for data and code availability, inadequate external validation, and insufficient consideration of fairness and equity that these issues are particularly important in oncology, where heterogeneity and stakes are high. Overall, integrating XAI with EHR-driven oncology models is promising but underdeveloped, which requires further progress by multi-stakeholder evaluation frameworks, reproducible pipelines, prospective and multicenter validations, and equity-aware design. The field should prioritize clinically meaningful explanations beyond ranking features and study how explanations affect clinician decision-making and patient outcomes.
Machine learning (ML) and deep learning (DL) models applied to electronic health records (EHRs) have substantial potential to improve oncology care across diagnosis, prognosis, treatment selection, and trial recruitment. However, opacity of many high-performing models limits clinician trust, regulatory acceptance, and safe deployment. Explainable artificial intelligence (XAI) methods aim to make model behavior understandable and actionable in clinical contexts. The present perspective summarizes current XAI approaches applied to EHR-based oncology tasks, identifies key challenges in evaluation, reproducibility, clinical utility, and equity, and proposes pragmatic recommendations and research directions to accelerate safe adoption in oncology. Common XAI categories used with EHR data include feature importance/interaction methods, intrinsically interpretable models, attention mechanisms, dimensionality reduction, and knowledge distillation or rule extraction. Tree-based models with SHapley Additive exPlanations (SHAP) explanations dominate recent EHR studies. Other interpretable strategies, such as generalized additive models and rule sets, appear in settings where transparency is prioritized. Gaps include inconsistent reporting, scarce formal evaluation of explanations for clinical utility, limited reproducibility for data and code availability, inadequate external validation, and insufficient consideration of fairness and equity that these issues are particularly important in oncology, where heterogeneity and stakes are high. Overall, integrating XAI with EHR-driven oncology models is promising but underdeveloped, which requires further progress by multi-stakeholder evaluation frameworks, reproducible pipelines, prospective and multicenter validations, and equity-aware design. The field should prioritize clinically meaningful explanations beyond ranking features and study how explanations affect clinician decision-making and patient outcomes.
DOI: https://doi.org/10.37349/etat.2026.1002357

DOI: https://doi.org/10.37349/etat.2026.1002356
 Previous
Previous