Affiliation:
1Biomedical Research, Pharmaceutical Department, Center for Genetic Engineering and Biotechnology (CIGB), Havana 10600, Cuba
†These authors share the first authorship.
ORCID: https://orcid.org/0009-0006-0182-8143
Affiliation:
1Biomedical Research, Pharmaceutical Department, Center for Genetic Engineering and Biotechnology (CIGB), Havana 10600, Cuba
†These authors share the first authorship.
ORCID: https://orcid.org/0000-0001-9428-5606
Affiliation:
1Biomedical Research, Pharmaceutical Department, Center for Genetic Engineering and Biotechnology (CIGB), Havana 10600, Cuba
2Physiological Sciences, Immunology Department, Latin American School of Medicine, Havana 19108, Cuba
Email: giselle.penton@cigb.edu.cu
ORCID: https://orcid.org/0000-0002-3719-7287
Explor Neuroprot Ther. 2026;6:1004143 DOI: https://doi.org/10.37349/ent.2026.1004143
Received: September 30, 2025 Accepted: March 10, 2026 Published: April 03, 2026
Academic Editor: Marcello Iriti, Università degli Studi di Milano, Italy
The article belongs to the special issue Natural Products in Neurotherapeutic Applications
Phycocyanobilin (PCB), the covalently bound chromophore of the cyanobacterial protein C-phycocyanin (CPC), is recognized as a bioactive molecule with neuroprotective and anti-inflammatory properties. PCB and CPC, frequently coexisting in Spirulina extracts or experimental formulations, have demonstrated beneficial effects in preclinical models of multiple sclerosis, ischemic stroke, and Alzheimer’s disease. Reported mechanisms include attenuation of oxidative stress, reduction of neuroinflammation, and preservation of mitochondrial function, thereby contributing to a reparative microenvironment within the central nervous system. PCB can be obtained through two complementary approaches: Extraction from cyanobacterial biomass, where it remains covalently bound to CPC, and heterologous biosynthesis in Escherichia coli (E. coli), which enables production of free PCB as a high-purity, scalable linear tetrapyrrole suitable for translational applications. This mini-review summarizes current evidence on the neuroprotective actions of PCB and CPC, highlights their molecular targets, and discusses biotechnological advances that support their potential role in remyelination. By bridging natural pigment pharmacology with recombinant production strategies, PCB is positioned as a multitarget candidate of growing interest for the development of future neuroprotective and neurorepair therapies.
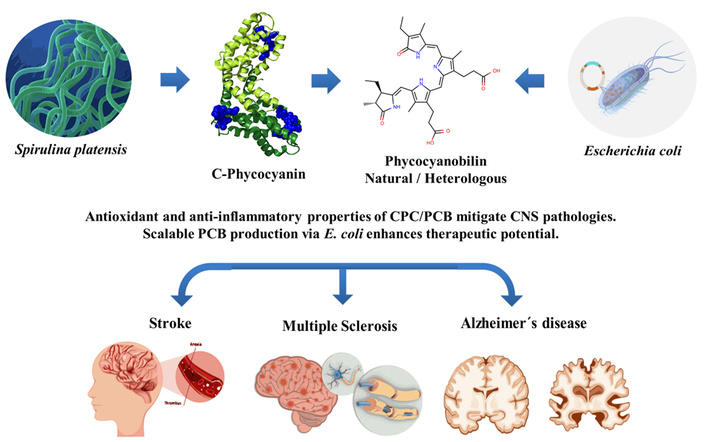
Neurological and neurodegenerative diseases such as multiple sclerosis (MS), ischemic stroke, and Alzheimer’s disease (AD) continue to represent major public health challenges, with limited therapeutic options capable of preventing or reversing neuronal and glial damage [1, 2]. Increasing evidence indicates that these disorders share convergent pathological mechanisms (including oxidative stress, chronic neuroinflammation, mitochondrial dysfunction, and impaired remyelination) that collectively drive disease progression [3, 4]. Targeting these interconnected processes is therefore a key strategy for developing multitarget therapies applicable across different central nervous system (CNS) conditions.
In this context, phycocyanobilin (PCB), the covalently bound prosthetic group of the cyanobacterial protein C-phycocyanin (CPC), emerges as a promising bioactive compound with antioxidant, anti-inflammatory, and neuroprotective properties. Experimental studies in both animal and cellular models have shown that PCB mitigates behavioral, histological, and molecular alterations associated with autoimmune and neurodegenerative damage [5]. Moreover, transcriptomic and histological evidence support its ability to promote oligodendrocyte survival and remyelination, thereby adding a regenerative dimension to its protective profile [6].
Recent advances in heterologous PCB biosynthesis, particularly in E. coli systems, enable the production of reproducible, high-purity, and scalable material suitable for mechanistic and translational research [7]. Such progress bridges natural pigment pharmacology with modern biotechnology, paving the way for clinical development.
This mini-review integrates current evidence on PCB’s neuroprotective actions, elucidates its molecular mechanisms, and highlights recent progress in its biotechnological production and translational prospects.
PCB serves as the covalently bound prosthetic group of phycobiliproteins (most prominently CPC) in cyanobacteria such as Spirulina platensis. Within CPC, PCB is attached via thioether linkages to conserved cysteine residues within the α/β subunits [8], as illustrated in Figure 1A.
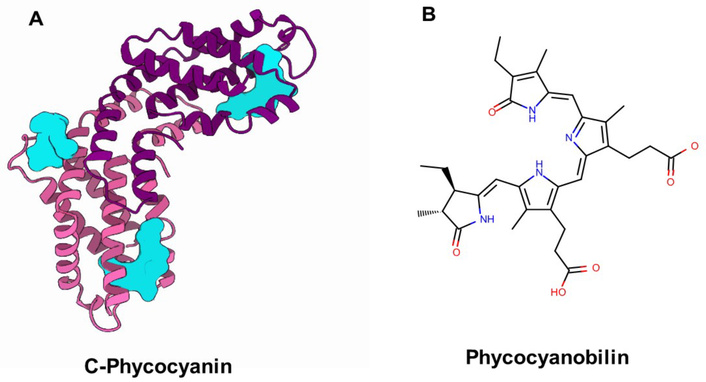
Structural representation of C-phycocyanin and its prosthetic group, phycocyanobilin. (A) Three-dimensional structure of the phycobiliprotein CPC with PCB covalently bound as a prosthetic group through thioether linkages to conserved cysteine residues in the α/β subunits. Structure was obtained from the Protein Data Bank (PDB ID: 2BV8) and visualized using MolViewer* (https://www.rcsb.org/structure/2BV8); (B) chemical structure of the free PCB molecule, a linear tetrapyrrole chromophore structurally related to biliverdin and bilirubin. These complementary panels illustrate PCB’s natural protein context and its molecular features that are relevant to antioxidant and neuroprotective functions. CPC: C-phycocyanin; PCB: phycocyanobilin.
Structurally related to biliverdin and bilirubin, PCB exhibits potent antioxidant and anti-inflammatory activities through the modulation of redox-sensitive signaling pathways and the preservation of mitochondrial homeostasis [9, 10]. The free PCB molecule, represented in Figure 1B, retains the conjugated tetrapyrrolic structure that confers these redox and radical-scavenging properties.
PCB can be obtained through two complementary and translationally relevant production routes: Natural extraction from cyanobacterial biomass (most commonly Spirulina) which yields PCB covalently bound as the prosthetic group of CPC [11, 12]; and heterologous biosynthesis, primarily in E. coli, which enables the production of free PCB as a recombinant linear tetrapyrrole molecule with enhanced purity, reproducibility, and scalability [13, 14].
These advances in biotechnological production are pivotal for translational development, providing consistent material for mechanistic and preclinical studies while overcoming the intrinsic limitations of natural biomass sources.
Taken together, the structural versatility and scalable production routes of PCB provide the basis for advancing its neuroprotective and remyelinating potential in experimental models.
MS is a chronic, immune-mediated disorder of the CNS characterized by inflammation, demyelination, and progressive neurodegeneration. Disease-modifying therapies (DMTs) mainly target immune mechanisms but have limited efficacy in halting disability progression and promoting remyelination.
In preclinical MS models, PCB has demonstrated significant neuroprotective potential. Oral administration of PCB-enriched fractions (0.2, 1, or 5 mg/kg) in mice with experimental autoimmune encephalomyelitis (EAE) markedly reduced clinical scores, attenuated inflammatory cell infiltration, and decreased pro-inflammatory cytokines, including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interferon-γ (IFN-γ), in the brain and spinal cord [15]. In vitro studies using human neuroblastoma cells subjected to excitotoxic damage with 60 mM glutamate have shown that treatment with 1 µM PCB downregulated cytochrome b-245 beta chain (CYBB), also known as nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 2 (NOX2), and enhanced antioxidant defenses. These included HMOX1, the gene encoding heme oxygenase-1 (HO-1), superoxide dismutase 2 (SOD2), and catalase (CAT), thereby contributing to the preservation of neuronal and oligodendrocyte integrity [16]. Taken together, the findings support the role of PCB as a multitarget molecule capable of suppressing neuroinflammation while promoting a permissive environment for remyelination, as indicated by an increased expression of myelin-related markers and reduced glial activation [12].
The parent biliprotein CPC, which contains PCB covalently bound as its prosthetic group, also exerts protective effects in EAE. Intraperitoneal administration of 25 mg/kg/day CPC for 12 days, in both prophylactic and early therapeutic regimens, reduced clinical scores, preserved myelin integrity, and increased regulatory T cell frequency, suggesting an immunoregulatory action beyond oxidative stress modulation [17]. In addition, CPC promoted oligodendrocyte survival and contributed to a remyelination-permissive environment [18].
Ischemic stroke involves excitotoxicity, oxidative stress, neuroinflammation, and blood-brain barrier (BBB) disruption. In rodent models of transient cerebral hypoperfusion, PCB, administered intraperitoneally at cumulative doses of 47 or 213 µg/kg for 30 min, 1, 3, and 6 h after surgery, significantly reduced oxidative damage, preserved neuronal morphology, and modulated immune-related genes, including IL-6 and HMOX1 [19]. PCB also improved mitochondrial function and cell survival. In focal ischemia models (endothelin-1-induced middle cerebral artery occlusion), PCB was administered intraperitoneally at 200 μg/kg at 30 min, 1, 3, and 6 h after surgery, which decreased infarct volume and neuronal loss while improving neurological scores. These effects were associated with preserved mitochondrial integrity and suppression of oxidative stress pathways [20].
In vitro, treatment with cumulative doses ranging from 1 to 15 µM PCB protected PC12 cells from H2O2-induced oxidative stress and glutamate-induced toxicity, maintaining mitochondrial membrane potential and reducing apoptosis [19].
CPC has demonstrated parallel cerebroprotective effects in experimental ischemia. Studies by Marín-Prida and colleagues [21] showed that CPC (at cumulative doses of 2.5, 5, or 10 mg/kg, administered intraperitoneally at 30 min, 1, 3, and 6 h after the surgery) in rats subjected to permanent ischemia protected against neuronal degeneration, reduced glial activation, and enhanced antioxidant defenses. These outcomes suggest that CPC, like PCB, can modulate redox homeostasis and inflammation, but its protective profile may also involve protein-based antioxidant mechanisms inherent to the holoenzyme structure.
To date, PCB has not been directly tested in animal models of AD. However, its structural and mechanistic similarity to bilirubin, together with its proven effects in other neurodegenerative contexts, provides a strong rationale for future testing in AD [9, 10, 22]. PCB’s antioxidant, anti-inflammatory, and mitochondrial-supporting actions may counteract early pathogenic events such as oxidative stress and neuroinflammation.
Evidence from CPC and Spirulina-derived extracts offers indirect support for PCB-related neuroprotection in AD. In scopolamine-induced amnesia models, Spirulina extracts (at cumulative doses of 50, 100, 200, or 400 mg/kg) improved cognitive performance and diminished oxidative stress [23, 24]. In AD-like models, CPC at 50 and 100 mg/kg alleviated cognitive deficits induced by intracerebroventricular administration of streptozotocin, restored antioxidant balance, and modulated pathways dependent on phosphoinositide 3-kinase (PI3K), thereby contributing to neuroprotection [25]. Furthermore, CPC reduced apoptosis and inflammatory cytokines in amyloid-beta (Aβ)-challenged rodents [26]. Mechanistically, CPC activates the nuclear factor erythroid 2-related factor 2 (Nrf2) antioxidant pathway and inhibits the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB) inflammatory pathway, thereby promoting neuronal survival and synaptic function.
Overall, although direct data for free PCB in AD models are lacking, studies involving CPC suggest overlapping neuroprotective mechanisms and reinforce the hypothesis that its covalently bound prosthetic group, PCB, contributes to CPC’s antioxidant activity.
Across preclinical models of MS, ischemia, and AD, both free PCB and CPC exert convergent neuroprotective actions through complementary, yet interconnected, mechanisms. Free PCB is recognized as the primary redox-active linear tetrapyrrole molecule, mediating antioxidant and anti-inflammatory modulation, whereas CPC functions as the holoprotein containing PCB as a prosthetic group, providing additional structural and immunoregulatory functions that may enhance remyelination and cellular recovery. However, in in vivo studies, their effects cannot be completely dissociated, since part of CPC’s activity may derive from the redox reactivity of its covalently bound PCB.
Key findings from PCB and CPC preclinical studies are summarized in Table 1, which provides an integrated view of the models and their main outcomes.
Summary of preclinical evidence on PCB and CPC in models of MS, ischemic stroke, and AD.
| Model/Disease | Compound | Key findings | References |
|---|---|---|---|
| EAE-MS model | PCB | Oral PCB reduced clinical scores, suppressed IL-6, IFN-γ, TNF-α; ↓ NOX2 (CYBB); ↑ HO-1, SOD2; improved redox and inflammatory balance. | [12, 15, 16] |
| CPC | Reduced disease severity, preserved myelin, induced regulatory T cells; ↓ inflammatory cytokines; created a permissive environment for remyelination. | [17, 18] | |
| Cerebral ischemia models | PCB | ↓ Oxidative stress (malondialdehyde, protein carbonyls); ↑ HO-1, SOD2, CAT; ↓ IL-6, TNF-α; improved neurological outcomes; reduced infarct volume. | [19, 20] |
| CPC | Preserved mitochondrial function, reduced reactive oxygen species, improved survival after ischemia/reperfusion. | [21] | |
| AD models | CPC | Improved memory in scopolamine and Aβ models; ↓ Aβ aggregation, ↓ glial activation, ↑ BDNF and synaptic markers. | [23, 24] |
The data include the principal molecular mechanisms and functional outcomes. Aβ: amyloid-beta; AD: Alzheimer’s disease; BDNF: brain-derived neurotrophic factor; CAT: catalase; CPC: C-Phycocyanin; CYBB: cytochrome b-245 beta chain; EAE: experimental autoimmune encephalomyelitis; HO-1: heme oxygenase-1; IFN-γ: interferon-γ; IL-6: interleukin-6; MS: multiple sclerosis; NOX2: nicotinamide adenine dinucleotide phosphate oxidase 2; PCB: phycocyanobilin; SOD2: superoxide dismutase 2; TNF-α: tumor necrosis factor-alpha. The arrow pointing upwards (↑) represents an increase, and the arrow pointing downwards (↓) represents a decrease.
In preclinical studies investigating the neuroprotective potential of CPC and PCB, different routes of administration and dosing regimens have been employed. CPC has frequently been administered intraperitoneally or orally, with doses ranging from low milligram quantities to higher repeated administrations depending on the experimental design [15, 17, 18, 21, 23]. PCB, often studied as a purified chromophore or derived from Spirulina extracts, has been delivered via oral gavage, intraperitoneal injection, and in some cases incorporated into dietary formulations [12, 15, 16, 19, 20]. Despite evidence of beneficial effects across various animal models of neurodegenerative disorders, the heterogeneity in administration procedures and dosing schedules complicates direct comparisons between studies. Future research should aim to standardize delivery methods and optimize dose ranges to better define therapeutic windows and facilitate translation into clinical applications.
The beneficial effects of PCB on the CNS are explained through several interrelated molecular mechanisms that converge on the control of oxidative stress and neuroinflammation (Figure 2) [6, 12, 17].
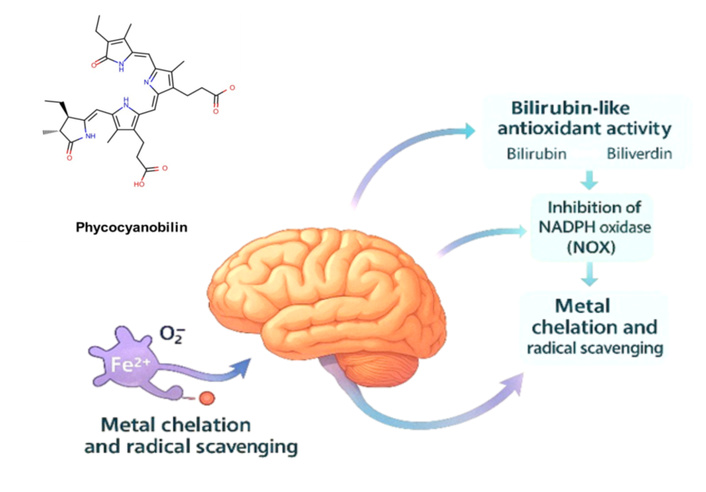
Molecular mechanisms of PCB protection in the CNS. PCB exerts its neuroprotective effects through three complementary molecular pathways: (a) Bilirubin-like redox cycling, in which PCB undergoes reversible redox interconversions analogous to the biliverdin/bilirubin pair, thereby neutralizing ROS and maintaining cellular redox balance; (b) inhibition of NOX, where PCB suppresses NOX-dependent superoxide generation and downstream inflammatory signaling; and (c) metal chelation and radical scavenging, whereby the conjugated π-electron system of PCB facilitates binding to redox-active metals (Fe2+, Cu2+) and scavenging of hydroxyl and peroxyl radicals, thereby preventing lipid peroxidation and preserving mitochondrial and neuronal integrity. CNS: central nervous system; NOX: nicotinamide adenine dinucleotide phosphate oxidase; PCB: phycocyanobilin; ROS: reactive oxygen species.
PCB is a linear tetrapyrrole, structurally similar to biliverdin and bilirubin, which allows reversible redox interconversion between its oxidized and reduced forms [11, 27]. This bilirubin-like behavior enables PCB to neutralize reactive oxygen species (ROS) and peroxyl radicals, while maintaining intracellular glutathione levels and thiol homeostasis. Such a redox cycle represents a key antioxidant defense, analogous to the well-characterized biliverdin/bilirubin pair, thereby preventing oxidative damage to lipids, proteins, and nucleic acids [27].
PCB modulates the activity of the NOX complex (particularly NOX2/CYBB), a major enzymatic source of ROS in microglia and endothelial cells of the CNS. Transcriptomic and biochemical studies indicate that PCB downregulates CYBB and cytochrome b-245 alpha chain (CYBA) gene expression, while enhancing antioxidant enzymes such as HMOX1, SOD2, and CAT. This regulation results in a marked reduction in superoxide and hydrogen peroxide generation [16]. Such modulation contributes to anti-inflammatory effects by limiting the activation of NF-kB and the release of cytokines (e.g., IL-6, TNF-α) [12].
The conjugated π-electron system of PCB enables interactions with and chelates transition metals such as Fe2+ and Cu2+, which are key mediators of Fenton-type reactions that generate hydroxyl radicals. By sequestering these redox-active metals and directly scavenging hydroxyl and peroxyl radicals, PCB interrupts lipid peroxidation chains and preserves mitochondrial integrity [28, 29].
Collectively, these complementary mechanisms define PCB as a multitarget antioxidant and anti-inflammatory molecule, capable of mitigating oxidative stress, protecting neurons and oligodendrocytes, and fostering an environment favorable to remyelination [12, 18].
Natural extraction of PCB from cyanobacterial biomass traditionally involves isolating CPC holoproteins containing covalently bound PCB, followed by chromatographic enrichment and, when free PCB is required, controlled cleavage and bilin release [30, 31]. Although feasible, this route is limited by batch-to-batch variability in pigment content, isoforms, and purity, as well as by the presence of proteinaceous impurities and other bilins, which complicate downstream analytics [30].
Recent advances have shifted attention toward heterologous biosynthesis in E. coli, where the cyanobacterial heme-to-bilin pathway is reconstituted by co-expression of HO-1 and PCB: ferredoxin oxidoreductase (PcyA). Optimized ribosome binding sites, codon usage, and metabolic engineering strategies have enabled intracellular PCB accumulation at higher yields and purity [7, 32, 33]. Importantly, recombinant production provides a scalable source of free PCB, thereby overcoming the variability inherent to natural extracts.
For translational applications, two technical aspects are particularly critical and must be rigorously addressed to ensure safety and reproducibility. Endotoxin control is indispensable, as the removal of lipopolysaccharides derived from Gram-negative expression hosts is essential for parenteral administration; residual endotoxins can elicit strong immunogenic responses and compromise therapeutic applicability. Equally important is the assurance of analytical identity and purity, which requires comprehensive characterization through combined high-performance liquid chromatography (HPLC), ultraviolet-visible spectroscopy, and mass spectrometry criteria. This integrated approach confirms the correct bilin identity while excluding oxidized or degraded by-products, thereby safeguarding molecular integrity and functional consistency [7, 33].
In addition, PCB stability and formulation strategies remain an active area of research. Like other tetrapyrroles, PCB is both photo and redox-sensitive, requiring protective measures such as opaque containers, antioxidants, and controlled pH conditions [30]. Preclinical work has explored cyclodextrin complexes, liposomal carriers, polymeric nanoparticles, and intranasal delivery routes to enhance solubility, stability, and potential brain exposure. These approaches highlight the growing integration of biotechnological production with pharmaceutical formulation science [30, 31].
Despite promising preclinical data, several challenges must still be addressed before PCB can advance to clinical use. The first challenge relates to pharmacokinetics and BBB permeability, as direct evidence of PCB penetration into the CNS remains limited [6]. Clarifying whether PCB acts centrally or through peripheral immunometabolic modulation will require standardized biodistribution assays, including liquid chromatography-tandem mass spectrometry (LC-MS/MS), plasma-to-brain ratio measurements, and radiolabeled tracing. Furthermore, toxicological and clinical data on PCB represent a significant gap, given the lack of formal toxicological evaluation of the molecule. Although CPC and Spirulina are generally recognized as safe (GRAS) [34] for dietary use, intravenous or chronic administration of PCB will require rigorous safety studies and, ultimately, clinical trials.
From a regulatory standpoint, PCB may follow a nutraceutical pathway, leveraging its existing GRAS status but limiting therapeutic claims, or a drug development pathway, which requires comprehensive pharmaceutical evaluation [35] but allows for specific disease indications. Additionally, the potential for combinatorial use of the molecule should be considered. Given its multitarget profile, PCB may complement DMTs in MS [6] or be combined with antioxidants in AD and stroke [36], potentially enhancing neurorepair and remyelination.
Finally, the industrial and therapeutic value of recombinant PCB is considerable. Heterologous expression systems provide consistent, high-purity PCB suitable for pharmaceutical development, ensuring reproducibility while enabling innovation in formulations such as nanoparticles, cyclodextrins, and liposomes, as well as supporting intellectual property development.
PCB is a multitarget molecule with antioxidant, anti-inflammatory, mitochondrial-protective, and remyelination-supporting potential. Preclinical models of MS, ischemic stroke, and neurodegeneration have demonstrated its ability to modulate redox-sensitive pathways, dampen neuroinflammation, and improve functional outcomes. The heterologous production of PCB in engineered systems provides reproducible, high-purity, and scalable preparations for pharmaceutical development.
Key challenges include characterizing its pharmacokinetics, establishing BBB permeability, and defining a good laboratory practice-compliant toxicological profile. The regulatory pathway must clarify whether PCB will advance as a nutraceutical, a drug candidate, or potentially both.
Hence, PCB offers a unique opportunity to bridge natural product pharmacology with modern biotechnology, positioning it as a promising candidate for future neuroprotective and neurorepair strategies. In this regard, PCB may evolve from a nutraceutical curiosity into a clinically relevant neurotherapeutic.
AD: Alzheimer’s disease
BBB: blood-brain barrier
CAT: catalase
CNS: central nervous system
CPC: C-phycocyanin
CYBB: cytochrome b-245 beta chain
DMTs: disease-modifying therapies
EAE: experimental autoimmune encephalomyelitis
GRAS: generally recognized as safe
HO-1: heme oxygenase-1
IL-6: interleukin-6
MS: multiple sclerosis
NADPH: nicotinamide adenine dinucleotide phosphate
NF-kB: nuclear factor kappa-light-chain-enhancer of activated B cells
NOX2: nicotinamide adenine dinucleotide phosphate oxidase 2
PCB: phycocyanobilin
ROS: reactive oxygen species
SOD2: superoxide dismutase 2
TNF-α: tumor necrosis factor-alpha
We thank Dr. Ania Cabrales-Rico and Osmany Guirola-Cruz for their technical assistance with the images, which was essential for the successful completion of this manuscript. The authors acknowledge the use of ChatGPT (OpenAI, San Francisco, CA, USA) to assist in improving the readability of the manuscript. All content was critically reviewed and approved by the authors, who take full responsibility for the publication.
SDL and YRÁ: Methodology, Writing—original draft, Formal analysis. GPR: Supervision, Methodology, Writing—review & editing. All authors read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1183
Download: 30
Times Cited: 0
Leonel Pereira, Ana Valado
Sharon Smith ... David Heal
Prerna Sarup ... Sonia Pahuja
Ekaterina P. Krutskikh ... Artem P. Gureev
Susmita Das ... Shylaja Hanumanthappa
Sneha Bagle ... Sadhana Sathaye
Luis Antonio Ramirez-Contreras ... Andrés Frausto de Alba
Agustina Lulustyaningati Nurul Aminin ... Muhammad Ajmal Shah
