Affiliation:
1Histology Area and Neurobiology Research Group, Faculty of Health Sciences, University of Valladolid, 42004 Soria, Spain
Email: diego.fernandez.lazaro@uva.es
ORCID: https://orcid.org/0000-0002-6522-8896
Affiliation:
2Doctoral School, University of León, Vegazana Campus, 24071 León, Spain
ORCID: https://orcid.org/0009-0003-6099-2021
Affiliation:
3Research Group “Pharmacogenetics, Cancer Genetics, Genetic Polymorphisms and Pharmacoepidemiology”, Faculty of Health Sciences, University of Valladolid, 42004 Soria, Spain
ORCID: https://orcid.org/0000-0002-5324-1014
Affiliation:
4Department of Spanish Language, Faculty of Translation and Interpreting, University of Valladolid, 42004 Soria, Spain
ORCID: https://orcid.org/0000-0001-6408-4176
Affiliation:
5Department of Anatomy and Radiology, Faculty of Health Sciences, University of Valladolid, 42004 Soria, Spain
ORCID: https://orcid.org/0000-0001-6521-2053
Affiliation:
6Department of Health Sciences, Faculty of Health Sciences, University of Burgos, 09001 Burgos, Spain
ORCID: https://orcid.org/0000-0002-6554-4602
Explor Neuroprot Ther. 2026;6:1004140 DOI: https://doi.org/10.37349/ent.2026.1004140
Received: December 13, 2025 Accepted: February 10, 2026 Published: March 16, 2026
Academic Editor: Antonio Ibarra, Anahuac University, Mexico
The article belongs to the special issue Role of Microbiota in Neurological Diseases
Background: Alzheimer’s disease (AD) is a prevalent neurodegenerative disorder in older adults, and growing evidence suggests that alterations in the gut-brain axis may contribute to its pathophysiology. Probiotics have been proposed as a potential adjunctive strategy to modulate gut microbiota and related systemic pathways; however, clinical evidence in AD remains limited and heterogeneous. This systematic review evaluated current evidence on the effects of probiotic supplementation on cognitive outcomes and health-related biomarkers in patients with AD.
Methods: Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, PubMed, Web of Science, and Scopus were systematically searched for double-blind randomized controlled trials published within the last 10 years (up to November 30, 2025). Studies assessing probiotic supplementation in clinically diagnosed AD patients were included. Methodological quality and risk of bias were evaluated using the Physiotherapy Evidence Database (PEDro) scale, the McMaster assessment tool, and the Cochrane Risk of Bias 2.0 tool.
Results: Of 253 records identified, five randomized controlled trials met the inclusion criteria, comprising 328 participants. The studies evaluated different probiotic formulations, mainly involving Lactobacillus and Bifidobacterium species, administered over short-term interventions (12 weeks). Some trials reported statistically significant improvements in selected cognitive outcomes, inflammatory and oxidative stress markers, metabolic parameters, brain-derived neurotrophic factor levels, anxiety, and instrumental activities of daily living. However, results were heterogeneous and several outcomes were non-significant. No serious adverse events related to probiotic supplementation were reported.
Discussion: Probiotic supplementation may be associated with strain-specific and short-term effects on selected cognitive and biological outcomes in patients with AD. Nevertheless, limited trial numbers, small sample sizes, and substantial heterogeneity preclude definitive conclusions. Further well-designed randomized controlled trials with standardized probiotic formulations and longer follow-up periods are required.
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder responsible for approximately 80% of dementia cases. The disease affects individuals of both sexes over 60 years of age [1]. In the world, over 55 million people live with dementia, and projections estimate more than 130 million cases by 2050, posing a major public health challenge [2]. Described for the first time in 1906 by Alois Alzheimer [3], the disease is marked by a major cognitive decline, including memory loss, apraxia, impaired judgment, and reduced attention. Altogether, these symptoms severely impact daily functioning and social relationships [4]. The histopathology of AD is characterized by extracellular β-amyloid plaques and intracellular neurofibrillary tangles of hyperphosphorylated tau protein. These lesions trigger neuroinflammation, synaptic dysfunction, neuron death, together with a marked reduction in the neurotransmitter acetylcholine [5].
Nevertheless, the strongest genetic risk factor for late onset of AD is the apolipoprotein E4 allele (ApoE4). This increases 3× the risk of AD, lowering the age of onset [6]. ApoE4 seems to promote β-amyloid and tau protein accumulation, disrupting calcium signaling, mitochondrial function, and activating neuronal apoptosis [7]. Neuroinflammation is a central event in AD progression. β-Amyloid plaques activate microglia and astrocytes, which release proinflammatory cytokines: interleukin (IL)-1β, IL-6 and tumor necrosis factor-α (TNF-α). These chemokines exacerbate neuronal damage facilitating disease progression [8]. In this context, peripheral immune cells may cross a weak blood-brain barrier, increasing inflammation and impairing hippocampal-dependent learning [9]. In addition to central nervous system (CNS) pathology, increasing evidence suggests that peripheral systemic factors may play a relevant role in AD progression, highlighting the importance of bidirectional communication between the gut and the brain. The gut microbiota is a dense and varied ecosystem located in the human intestine, including bacteria, fungi, viruses, and protists [10]. The gut microbiota comprises over 100 billion microorganisms and up to 1,000 species (ssp.), with Firmicutes and Bacteroidetes as dominant phyla [11]. Microbial composition is shaped by diet, age, genetics, birth mode, and lifestyle (i.e., physical activity) [12]. A balanced microbiota (eubiosis) supports immune system regulation, nutrient synthesis, metabolic control, and gut-brain communication. In contrast, the opposite situation is known as dysbiosis, characterized by overgrowth of pathogenic bacteria like Enterobacteriaceae. Dysbiosis is linked to inflammation and multi-organ diseases [13]. The gut-brain axis connects the gastrointestinal tract and the CNS via neural, endocrine, and immune pathways [14]. Microbial metabolites modulate this axis by influencing neurotransmitter release, stress responses, and barrier integrity. Dysbiosis favors dysregulation of the CNS, having implications for neuronal health and dysbiosis seems to be involved in neurodegenerative diseases, like AD [15], through altered neuroimmune signaling and impaired homeostatic responses through mechanisms such as increased neuroinflammation [16].
Altogether, therapeutic strategies targeting the gut-brain axis include immunomodulation, neurotransmitter modulation, and microbiota restoration through prebiotics, probiotics, and fecal transplantation [17]. Prebiotics stimulate beneficial bacteria and short-chain fatty acids (SCFAs) production. On the other hand, probiotics (mainly Lactobacillus and Bifidobacterium strains) support barrier integrity and immune modulation [18]. Specific bacteria phyla such as Lactobacillus, Bifidobacterium, and Bacillus can produce or modulate the activity of GABA, serotonin, dopamine, and acetylcholine [19]. However, probiotic effects may be transient, and in some cases (i.e., cancer immunotherapy), supplementation may impair treatment efficiency, highlighting the need for personalized protocols [20]. To date, no systematic review has been conducted to evaluate the potential health benefits of probiotics on AD patients. Therefore, we aimed to systematically review current evidence on the effects of probiotics on health biomarkers and determine whether the supplementation of probiotics improves cognitive function, oxidative stress, inflammation parameters, biomarkers related to AD, physical condition and state of microbiota in adults with AD.
A systematic review of scientific articles in English or Spanish published in the last ten years was conducted, following the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [21].
The research question for the study was formulated using the Population, Intervention, Comparison, Outcomes, and Study design (PICOS) model in accordance with evidence-based medicine (EBM) [22]. The research question for this systematic review was: Does probiotic supplementation have any health benefits for Alzheimer’s patients? To answer this question, inclusion criteria were established based on the PICOS criteria [23] (Table 1).
PICOS-based eligibility criteria for study inclusion and exclusion.
| Criteria | Inclusion criteria | Exclusion criteria |
|---|---|---|
| Population ‘P’ | Adult men and/or women over the age of 18 who are Alzheimer’s patients. | Participants diagnosed with any other neurodegenerative disease (Parkinson’s), psychiatric disorders, depression, or alcoholism other than Alzheimer’s. Patients were diagnosed with chronic gastrointestinal diseases.Participants who received any type of medication or treatment: immunosuppressants, steroids, antibiotics, chemotherapy, and probiotics in at least the last 4 weeks. Participants with a history of recreational drug abuse. |
| Intervention ‘I’ | To include a supplementation intervention that involves the use of probiotics as monotherapy, with clear information on the dose and duration of supplementation. | Administration together with other nutritional supplements. Studies in which probiotics were administered in combination with other nutritional interventions (e.g., specific diets or non-probiotic supplements) were excluded. This decision was made to isolate the effects attributable specifically to probiotic supplementation and to avoid confounding effects that could obscure the independent contribution of probiotics to the observed outcomes. |
| Comparison ‘C’ | To include a placebo, control, or sham treatment group (parallel group study design). | Patients receiving other doses of probiotics or other nutritional supplements. |
| Outcomes ‘O’ | Any biomarker related to biological, cognitive, or physical health in Alzheimer’s patients. | None. |
| Study design ‘S’ | Randomized controlled trials. | Observational studies and studies using a specific analytical approach. |
PICOS: Population, Intervention, Comparison, Outcomes, and Study design.
Table 1 summarizes the predefined eligibility criteria used to select studies for the systematic review, structured according to the PICOS framework. These criteria were applied consistently during the screening and selection process to identify relevant randomized controlled trials evaluating probiotic supplementation in patients with AD.
The search for the different studies analyzed in this systematic review was carried out between July 1 and November 30, 2025, in the electronic databases Medline (PubMed), Web of Science (WOS), and Scopus, including all results published in the last ten years. The search strategy accepted articles published in Spanish and English, excluding manuscripts published in other languages and based on the PRISMA guidelines [21]. All high-quality databases guarantee good bibliographic support.
To identify the relevant studies for this paper, searches were conducted using Medical Subject Headings (MeSH) together with the Boolean operators “AND” and “OR” to combine them and the search truncator “*” to refine the study. The search algorithms used to carry out this study were as follows: “Alzheimer disease” AND “gastrointestinal microbiome” AND “neuroinflammation”; “Alzheimer disease” AND “probiotics” OR “dietary supplements”; “Alzheimer disease” AND “probiotics” AND “microbiota”; “Alzheimer Disease” AND “probiotics” AND “inflammation”; “Alzheimer” OR “Alzheimer Disease” AND “probiotic*” OR “probiotics” AND “cognition” OR “memory” OR “cognition disorders”; and “Alzheimer disease” AND “probiotics” AND “dietary supplements” AND “clinical trial”. The complete electronic search strategies and records identified for each database are provided in Table S1. Two reviewers (AMCSM and AMFA) independently screened titles and abstracts, excluding those lacking either full text or final analysis of results. Full texts were sourced for relevant articles. Inclusion criteria were independently assessed by these two reviewers, and a third reviewer (DFL) participated in cases of disagreement. Additional records were obtained through a snowball search, checking the reference lists of publications eligible for full-text review and using ResearchGate® to identify possible articles not included in the databases mentioned above.
According to the CONSORT Statement for Controlled Trials 2010 [24], the following data were collected: name of the first author, year of publication, country where the study was conducted, study design, sample size, sex and age of participants, duration of intervention, dose, pharmaceutical form, and route of administration. All data included were extracted independently using the systematic review software Covidence.
The McMaster University Occupational Therapy Evidence-Based (McMaster) methodological assessment questionnaire [25] and the Physiotherapy Evidence Database (PEDro) methodological assessment scale [26] were used to assess the studies previously selected for this systematic review.
The tool used to assess the risk of bias was the Revised Cochrane Risk of Bias 2.0 [27]. This scale, included in the Covidence tool, was used to assess the potential risk of bias in each study, examining five possible sources of bias: (i) bias arising from the randomization process; (ii) deviations from the planned intervention; (iii) missing outcome data; (iv) outcome measurement; and (v) selective reporting of results. The latter also examines bias arising from period or carryover effects. This tool generates an overall risk rating (i.e., “low risk”, “uncertain risk”, “high risk”).
The main result was changes in cognitive function parameters (Alzheimer’s Disease Assessment Scale-Cognitive [ADAS-Cog], Clinical Dementia Rating [CDR], Categorical Verbal Fluency Test [CFT], Mini-Mental State Examination [MMSE], test your memory [TYM]); oxidative stress (ultrasensitive 8-hydroxy-2’-deoxyguanosine [8-OHdG] in serum , total glutathione [GSH], malondialdehyde [MDA], nitric oxide [NO], protein carbonyl content [PCC], total antioxidant capacity [TAC], superoxide dismutase antioxidant [SOD]); inflammatory status (high-sensitivity C-reactive protein [hs-CRP], IL-6, IL-8, IL-10, IL-1β, transforming growth factor-β [TGF-β], TNF-α); glycemic profile (Homeostasis Model Assessment of pancreatic β cells [HOMA-β], Homeostatic Model Assessment of Insulin Resistance [HOMA-IR], fasting plasma glucose [FPG], Quantitative Insulin Sensitivity Check Index [QUICKI], insulin [INS]); lipid profile (total cholesterol [TC], high-density lipoproteins [HDL], low-density lipoprotein [LDL], low-density lipoprotein receptor [LDLR], triglycerides [TG], very-low-density lipoprotein [VLDL]); metabolic/biochemical profile (peroxisome proliferator-activated receptor gamma [PPAR-γ]); anthropometric (body mass index [BMI]); psychological status (Generalized Anxiety Disorder-7 [GAD-7]); physical condition (basic activities of daily living [BADL], instrumental activities of daily living [IADL]); AD biomarkers (brain-derived neurotrophic factor [BDNF]); gut microbiota status; hormonal (cortisol).
A quantitative meta-analysis was considered but ultimately deemed inappropriate due to substantial clinical and methodological heterogeneity across included studies. Sources of heterogeneity included differences in probiotic strains and formulations, supplementation doses, duration of intervention, outcome measures, and baseline disease severity. Additionally, the limited number of trials and small sample sizes further reduced the statistical robustness of pooled estimates.
Although cognitive function was assessed in several trials using similar instruments, including the MMSE, meaningful quantitative pooling was not feasible. Studies differed in baseline cognitive severity, comparator conditions, probiotic composition, and reporting of outcome data, precluding standardized effect size calculation.
The literature search yielded 259 records. Of these, 253 were initially obtained from Medline (PubMed) (n = 87), Scopus (n = 73), and WOS (n = 93), and 6 came from additional sources, such as ResearchGate and reference lists of relevant studies. After excluding 182 duplicates, a total of 71 articles were examined. Once the title and abstract had been evaluated, 15 articles were considered as possible records. Records identified through additional sources (reference lists and ResearchGate) were included in the same duplicate removal and screening process as database-derived records. All remaining records were assessed using identical eligibility criteria, as illustrated in the PRISMA flow diagram. After a complete review and evaluation of the possible records from databases and other sources, 5 original studies were included in the systematic review [28–32] (Figure 1).
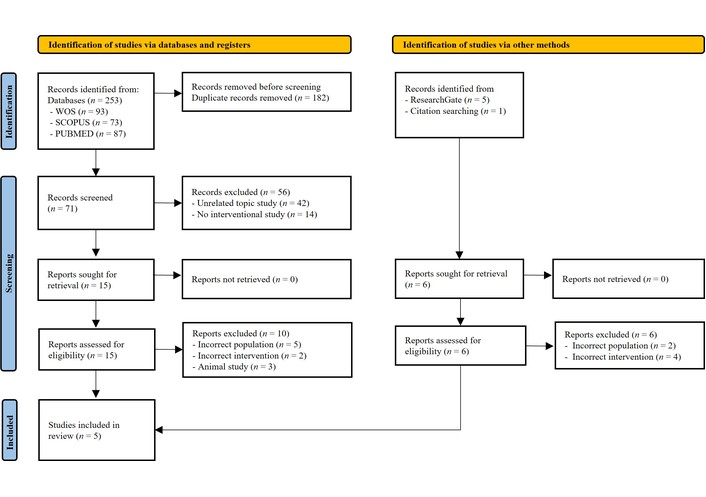
PRISMA flow diagram of study selection. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Adapted from [21]. © Author(s) (or their employer(s)) 2019. CC BY 4.0.
The flow diagram (Figure 1) illustrates the identification, screening, eligibility assessment, and inclusion of studies in the systematic review, in accordance with the PRISMA guidelines. Records were identified through database searches (PubMed, WOS, and Scopus) and additional sources. After duplicate removal, titles and abstracts were screened, followed by full-text assessment for eligibility, resulting in the final inclusion of randomized controlled trials.
The five studies [28–32] included were rated “Excellent”, with a score of 16 [28–30, 32], and 15 [31], on the McMaster methodological scale [25] obtained a score (Table 2).
Methodological quality assessment of included studies using the McMaster Critical Review Form [25].
| Study | Item | Total | % | Quality | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | ||||
| Agahi et al. [28] 2018 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 16 | 100 | E |
| Akbari et al. [29] 2016 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 16 | 100 | E |
| Akhgarjand et al. [30] 2022 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 16 | 100 | E |
| Hsu et al. [31] 2023 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 15 | 93.8 | E |
| Tamtaji et al. [32] 2019 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 16 | 100 | E |
E: Excellent; 0: criterion not met; 1: criterion met; item 1: purpose of the study; item 2: literature review; item 3: study design; item 4: blinded; item 5: sample description; item 6: sample size; item 7: ethics and consent; item 8: validity of results; item 9: reliability of results; item 10: description of intervention; item 11: statistical significance; item 12: statistical analysis; item 13: clinical significance; item 14: conclusions; item 15: clinical implications; item 16: study limitations.
On the other hand, when assessed using the PEDro scale [26], four of those studies obtained the maximum score of 10 points [28, 29, 31, 32], and one scored 9 points [30]. For all studies included [28–32], scores are considered “excellent” quality in the PEDro scale assessment [26] (Table 3).
Methodological quality assessment of included studies using the PEDro scale [26].
| Study | Item | Total | % | Quality | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | ||||
| Agahi et al. [28] 2018 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 | 100 | E |
| Akbari et al. [29] 2016 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 | 100 | E |
| Akhgarjand et al. [30] 2022 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 9 | 90 | E |
| Hsu et al. [31] 2023 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 9 | 90 | E |
| Tamtaji et al. [32] 2019 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 10 | 100 | E |
E: Excellent; 0: criterion not met; 1: criterion met; item 1: specified eligibility criteria; item 2: random assignment of subjects to groups; item 3: concealment of assignment; item 4: groups were similar at baseline in relation to the most important prognostic indicators; item 5: blinding of participants; item 6: blinding of personnel; item 7: blinding of the evaluator; item 8: outcome measures in at least 85% of participants; item 9: whether results were reported for all subjects who received treatment or were assigned to the control group, or if this was not possible, whether at least one key outcome was analyzed by “intention to treat”; item 10: statistical comparisons between groups for at least one key outcome; item 11: measures of variability and precision. PEDro: Physiotherapy Evidence Database.
Table 2 presents the methodological quality assessment of the randomized controlled trials included in the systematic review, evaluated using the McMaster Critical Review Form. Each item reflects a specific methodological criterion, and the total score represents overall methodological quality according to the tool’s guidelines. Scores are reported for transparency and should be interpreted in conjunction with the risk of bias assessment.
Table 3 summarizes the methodological quality of the included randomized controlled trials as assessed using the PEDro scale. Individual item scores and total scores are reported to provide a structured overview of study quality. These results are presented for descriptive purposes and should be interpreted alongside the risk of bias evaluation.
Although high scores were obtained on the McMaster and PEDro scales, these tools primarily assess reporting quality and selected methodological features. Therefore, high scores should not be interpreted as the absence of potential bias.
Table 4 shows the results of the RoB risk of bias assessment tool [27] applied to the studies in this review [28–32]. Figure 2 presents a summary of the authors’ judgments on each RoB item in each included study. None of the studies selected for this systematic review [28–32] were assessed as having a high risk of bias, but all of them were found to have an uncertain risk of bias in outcome assessor blinding and selective reporting.
Risk of bias assessment of included studies using the Cochrane Risk of Bias 2.0 tool [27].
| Study | Random sequence generation (selection bias) | Allocation concealment (selection bias) | Blinding of participants and personnel (performance bias) | Blinding of outcome assessors (detection bias) | Incomplete outcome data (attrition bias) | Selective reporting (reporting bias) | Other biases |
|---|---|---|---|---|---|---|---|
| Agahi et al. [28] 2018 |  |  |  |  |  |  |  |
| Akbari et al. [29] 2016 |  |  |  |  |  |  |  |
| Akhgarjand et al. [30] 2022 |  |  |  |  |  |  |  |
| Hsu et al. [31] 2023 |  |  |  |  |  |  |  |
| Tamtaji et al. [32] 2019 |  |  |  |  |  |  |  |
+: Low risk of bias; ?: uncertain risk of bias.
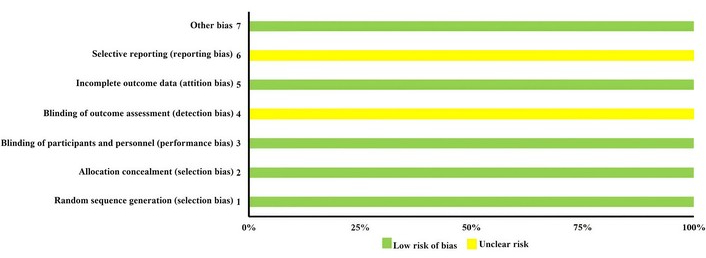
Risk of bias assessment of included studies using the Cochrane Risk of Bias 2.0 tool [27].
Table 4 presents the risk of bias assessment of the randomized controlled trials included in the systematic review, evaluated using the Cochrane Risk of Bias 2.0 tool. Bias was assessed across predefined domains, including the randomization process, deviations from intended interventions, missing outcome data, outcome measurement, and selective reporting. Overall judgments are reported to provide a transparent overview of potential sources of bias and should be considered when interpreting study findings.
Figure 2 summarizes the risk of bias assessment for the randomized controlled trials included in the systematic review, evaluated using the Cochrane Risk of Bias 2.0 tool. Risk of bias judgments are presented across the predefined domains: randomization process, deviations from intended interventions, missing outcome data, outcome measurement, and selection of the reported result. Overall risk of bias judgments reflect domain-level assessments and highlight areas of uncertainty that should be considered when interpreting the findings.
Importantly, although no study was judged to be at high risk of bias overall, all included trials presented uncertainties in at least one domain, particularly regarding blinding of outcome assessors and selective reporting. These uncertainties may have influenced effect estimates and contributed to variability across reported outcomes.
The total number of participants at the start of the studies was 328 (89 men, 149 women, and 90 unspecified subjects) in five randomized, double-blind, placebo-controlled clinical trials in humans [28–32]. All participants were AD patients diagnosed according to at least the criteria of the National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association (NINDS-ADRDA) and the National Institute on Aging and Alzheimer’s Association (NI-AAA) [33, 34]. The included studies were conducted in AD patients from Iran [28–32] and Taiwan, China [31].
The composition of the probiotics used for supplementation was of the ssp. Lactobacillus and Bifidobacterium from commercial pharmaceutical companies in the five studies included [28–32], specifically Lactobacillus fermentum [28, 29], Lactobacillus plantarum [28, 31], Lactobacillus acidophilus [28–32], Lactobacillus casei [29] and Lactobacillus rhamnosus [30], Bifidobacterium lactis [28], Bifidobacterium bifidum [28, 29, 31, 32], Bifidobacterium longum [28, 29, 31, 32], Bifidobacterium breve [31] and Bifidobacterium animalis [31]. Probiotic supplementation used ssp. in combination [28, 29, 31, 32] or individually [30] with doses in colony-forming units (CFU) of 1–5 × 109 CFU/day [28, 29, 32], 6–10 × 109 CFU/day [31] and > 11 × 109 CFU/day [30] administered orally in capsules [28, 30–32] or solution [29], once a day [29, 31, 32], twice a day [30] or once every two days [28] for 12 weeks [28–32]. Probiotic supplementation was administered in gastro-resistant capsules [28, 30–32] or in solution (mixed in milk) [29]. Four studies used a placebo as the control group (CG) [28–30, 32], and the study conducted by Hsu et al. [31] used a sham treatment (active control) with a dose of 5 × 107 CFU/day. This dose is considered insufficient to achieve benefits as described by the International Scientific Association for Probiotics and Prebiotics in 2025 [35], so it can be considered an adequate comparator (Table 5).
Characteristics of participants and probiotic supplementation protocols in the included studies.
| Characteristics | Types | Studies |
|---|---|---|
| Alzheimer’s diagnosis | NINS-ARDA | [28–32] |
| NI-AAA | [28–32] | |
| Other | [28, 30–32] | |
| Formulation of the strain | Single | [30] |
| Combined | [28, 29, 31, 32] | |
| Supplement products | Registered Product® | [28–32] |
| Manufactured product | - | |
| 1–5 | [28, 29, 32] | |
| Total dose (109 CFU/day) | 5–10 | [31] |
| > 11 | [30] | |
| ssp. | Lactobacillus | [28–32] |
| Bifidobacterium | [28–32] | |
| Control group | Placebo | [28–30, 32] |
| Simulated treatment1 | [31] | |
| Duration | 12 weeks | [28–32] |
| Dosing schedule | Once daily | [29, 31, 32] |
| Twice daily | [30] | |
| Every other day | [28] | |
| Pharmaceutical form | Capsules | [28, 30–32] |
| Solution | [29] |
CFU: colony-forming units; NI-AAA: National Institute on Aging and Alzheimer’s Association; NINDS-ADRDA: National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association; ssp.: species. 1 Intake of probiotics 5 × 107 CFU/day is considered insufficient to achieve the benefits described by the International Scientific Association for Probiotics and Prebiotics (2025).
Table 5 summarizes the main characteristics of participants and probiotic supplementation protocols across the randomized controlled trials included in the systematic review. Information includes diagnostic criteria for AD, sample size, probiotic strains and formulations, dosage, duration of intervention, comparator groups, and mode of administration. Table 5 is intended to facilitate comparison of study designs and intervention characteristics across trials.
Table 6 summarizes the information from the studies included in the present review.
Summary of included studies evaluating the effects of probiotic supplementation on health-related outcomes in patients with AD.
| First author, year of publication, and country | Study design | Participants (baseline sample size and characteristics, withdrawals, and final sample size) | Intervention | Outcomes | Results |
|---|---|---|---|---|---|
| Agahi et al. [28] 2018 Iran | Randomized, double-blind, placebo-controlled clinical trial | n = 60 enrolledAD (NINDS-ADRDA, NI-AAA TYM < 45 out of 50)n = 48 (17 ♂ and 31 ♀) includedIG n = 25 (7 ♂ and 18 ♀)Age (mean ± SD): 79.70 ± 1.72 yearsWeight (mean ± SD): 60.12 ± 1.12 kgHeight (mean ± SD): 156.77 ± 1.23 cmBMI (mean ± SD): 24.05 ± 1.07 kg/m2CG n = 23 (10 ♂ and 13 ♀)Age (mean ± SD): 80.57 ± 1.79 yearsWeight (mean ± SD): 60.63 ± 1.26 kgHeight (mean ± SD): 156.43 ± 1.86 cmBMI (mean ± SD): 24.44 ± 1.33 kg/m2Withdrawals due to death from aging (n = 12) (CG n = 7, IG n = 5)25 participants IG23 participants CG | 3 × 109 CFU1× capsule A + 1× capsule B every other day orallyA: Lactobacillus fermentum, Lactobacillus plantarum, Bifidobacterium lactis (Zist Takhmir Company; Tehran, Iran)B: Lactobacillus acidophilus, Bifidobacterium bifidum, Bifidobacterium longum (Milad Farmed Company; Tehran, Iran)Placebo 500 mgMaltodextrin12 weeks | Anthropometry:BMICognitive function:TYM (over 50 points)Oxidative stress:TACGSHMDANO8-OHdGInflammation:IL-6IL-10TNF-α | IG vs. GC↔ BMI↑ TYM↔ TAC↑ GSH↓ MDA↓ NO↓ 8-OHdG* ↑ IL-6↓ IL-10↓ TNF-α |
| Akbari et al. [29] 2016 Iran | Randomized, double-blind, placebo-controlled clinical trial | n = 60 (12 ♂ and 48 ♀)AD (NINDS-ADRDA, NI-AAA)IG n = 30 (6 ♂ and 24 ♀)Age (mean ± SD): 77.67 ± 2.62 yearsHeight (mean ± SD): 157.77 ± 2.03 cmWeight (mean ± SD): 59.03 ± 1.99 kgBMI (mean ± SD): 23.77 ± 0.73 kg/m2CG n = 30 (6 ♂ and 24 ♀)Age (mean ± SD): 82.00 ± 1.69 yearsHeight (mean ± SD): 157.43 ± 1.86 cmWeight (mean ± SD): 56.63 ± 2.21 kgBMI (mean ± SD): 22.73 ± 0.68 kg/m2Withdrawals due to death from aging (n = 8) (CG n = 4, IG n = 4)26 participants IG26 participants CG | 2 × 109 CFU/g strain:Lactobacillus acidophilusLactobacillus caseiBifidobacterium bifidumLactobacillus fermentum200 mL milk + probiotics day orally (Tak Gen Zist Pharmaceutical Company; Tehran, Iran)Placebo: 200 mL milk per day orally12 weeks | Anthropometry:BMICognitive function:MMSEOxidative stress:MDACTGSHNOInflammation:hs-CRPBiochemicals:HOMA-βQUICKIFPGLDLHDLTGVLDLCT | IG vs. CG↔ BMI* ↑ MMSE↓ MDA↔ CT↔ GSH↔ NO* ↓ hs-CRP↓ HOMA-β↑ QUICKI↔ FPG↔ LDL↔ HDL* ↓ TG* ↓ VLDL↔ CT |
| Akhgarjand et al. [30] 2022 Iran | Randomized, double-blind, placebo-controlled clinical trial | n = 90 (48 ♂ and 42 ♀)AD (NINDS-ADRDA, NI-AAA, mild or moderate FAST)IG1: n = 30 (16 ♂ and 14 ♀)Age (mean ± SD): 67.93 ± 7.8 yearsWeight (mean ± SD): 67.07 ± 6.6 kgBMI (mean ± SD): 23.71 ± 1.4 kg/m2IG2: n = 30 (16 ♂ and 14 ♀)Age (mean ± SD): 67.90 ± 7.9 yearsWeight (mean + SD): 65.97 ± 7.1 kgBMI (mean ± SD): 79 ± 2.4 kg/m²CG: n = 30 (16 ♂ and 14 ♀)Age (mean ± SD): 67.77 ± 7.9 yearsWeight (mean ± SD): 62.27 ± 6.8 kgBMI (mean ± SD): 23.93 ± 2.2 kg/m2No withdrawals | 1 × 1015 CFU2 capsules orallyGroup 1: Lactobacillus rhamnosusGroup 2: Bifidobacterium longum R0175 (Lallemand Company; Canada)Placebo 2 capsules/day of xylitol, maltodextrin, and malic acid orally (Lallemand Company; Canada)12 weeks | Cognitive function:MMSECFTPsychological state:GAD-7Physical condition:BADL → BarthelIADL→ Lawton | IG1 vs. CG* ↑ MMSE* ↑ CFT* ↓ GAD-7↔ ABVD* ↑ AIVDIG2 vs. CG* ↑ MMSE* ↑ CFT* ↓ GAD-7↔ ABVD* ↑ AIVD |
| Hsu et al. [31] 2023 Taiwan, China | Randomized, double-blind, active-controlled clinical trial | n = 40 (12 ♂ and 28 ♀)AD (DSM-V, NINCDS-ADRDA, NI-AAA, MMSE 10–25; CDR 0.5–2) (Treatment with donepezil or rivastigmine)IG: n = 20 (4 ♂ and 16 ♀)Age (mean ± SD): 75.4 ± 8.0 yearsCG: n = 20 (8 ♂ and 12 ♀)Age (mean ± SD): 75.8 ± 7.3 yearsWithdrawals (n = 8) (CG n = 4, IG n = 4)16 participants IG16 participants CG | IG: 1 × 1010 CFU/dayCG1: 5 × 107 CFU/day1 capsule daily orally with 5 probiotic strains in equal proportions:Bifidobacterium longum subsp. infantis BLI-02, Bifidobacterium breve Bv-889, Bifidobacterium animalis subsp. lactis CP-9, Bifidobacterium bifidum VDD088, and Lactobacillus plantarum PL-02 (Glac Biotech Co., Ltd.; Tainan, Taiwan, China)12 weeks | Cognitive function:MMSEADAS-CogCDROxidative stress:MDAPCCSODInflammatory markers:IL-1βIL-10Physical condition:BADLAD biomarker:BDNFIntestinal microbiota:StrainsHormonal:Cortisol | IG vs. CG↔ MMSE↔ ADAS-Cog↔ CDR* ↓ MDA* ↓ PCC* ↑ SOD* ↓ IL-1 β↑ IL-10↔ ABVD* ↑ BDNF↑ Bifidobacterium↑ Lactobacillus↑ Ruminococcus↑ Clostridium↑ Akkermansia↓ Megamonas* ↓ Cortisol |
| Tamtaji et al. [32] 2019 Iran | Randomized, double-blind, placebo-controlled clinical trial | n = 90 (gender not specified)AD (NINDS-ADRDA, NI-AAA, Golabchi Welfare Organization, and Madar, Shayestegan, Amin Welfare Organizations)IG probiotics + selenium: n = 27Age (mean ± SD): 76.2 ± 8.1 yearsBMI (mean ± SD): 20.7 ± 3.2 kg/m2CG1 selenium: n = 26Age (mean ± SD): 78.8 ± 10.2 yearsBMI (mean ± SD): 21.2 ± 1.2 kg/m2CG2 placebo: n = 26Age (mean ± SD):78.5 ± 8.0 yearsBMI (mean + SD): 21.5 ± 2.4 kg/m2Withdrawals (n = 11), personal reasons: CG1 n = 4, CG2 n = 4, IG n = 327 participants IG26 participants CG126 participants in CG2 | 2 × 109 CFU/day of each strain + 200 µg/day selenium 1 capsule orallyLactobacillus acidophilus, Bifidobacterium bifidum, Bifidobacterium longum (Tak Gen Zist Pharmaceutical Company; Tehran, Iran)Selenium 200 µg/day 1 capsule orally (Naturals Pharmaceutical Company; Coquitlam, Canada)Placebo: starch 1 capsule orally (Tak Gen Zist Pharmaceutical Company; Tehran, Iran)12 weeks | Cognitive function:MMSEOxidative stress:CTGSHMDANOInflammation:IL-8TNF-αTGF-βhs-CRPBiochemicals:INSHOMA-IRLDLHDLTGVLDLQUICKILDLRPPAR-γ | IG vs. CG1* ↑ MMSE↓ TNF-α↔ IL-8↔ TGF-β↓ hs-CRP↑ TAC↑ GSH↔ MDA↔ NO↓ INS↓ HOMA-IR↓ LDL↔ HDL↓ TG↔ VLDL↔ QUICKI↑ LDLR↑ PPAR-γ |
♂: male; ♀: female; ↑: non-significant increase; ↓: non-significant decrease; ↔: no significant change; * ↑: significant increase; * ↓: significant decrease; 8-OHdG: ultrasensitive 8-hydroxy-2’-deoxyguanosine; AD: Alzheimer’s disease; ADAS-Cog: Alzheimer’s Disease Assessment Scale-Cognitive; BADL: basic activities of daily living; BDNF: brain-derived neurotrophic factor; BMI: body mass index; CDR: Clinical Dementia Rating; CFT: Categorical Verbal Fluency Test; CFU: colony-forming units; CG: control group; DASS-21: anxiety, depression, and stress scale; DSM-V: Diagnostic and Statistical Manual of Mental Disorders, 5th Edition; FAST: Functional Assessment Staging Tool; FPG: fasting plasma glucose; GAD-7: Generalized Anxiety Disorder-7; GSH: total glutathione; HOMA-β: Homeostasis Model Assessment of pancreatic β cells; HDL: high-density lipoproteins; HOMA-IR: Homeostatic Model Assessment of Insulin Resistance; hs-CRP: high-sensitivity C-reactive protein; IADL: instrumental activities of daily living; IG: intervention group; IL: interleukin; INS: insulin; kg: kilograms; LDL: low-density lipoproteins; LDLR: low-density lipoprotein receptor; m2: square meters; MDA: malondialdehyde; MMSE: Mini-Mental State Examination; NI-AAA: National Institute on Aging and Alzheimer’s Association; NIH TCB: NIH (US National Institutes of Health) cognitive battery; NINDS-ADRDA: National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association; NO: nitric oxide; PCC: protein carbonyl content; PPAR-γ: peroxisome proliferator-activated receptor gamma; QUICKI: Quantitative Insulin Sensitivity Check Index; SD: standard deviation; SOD: superoxide dismutase antioxidant; TAC: total antioxidant capacity; TC: total cholesterol; TG: triglycerides; TGF-β: transforming growth factor-β; TNF-α: tumor necrosis factor-α; TYM: test your memory; VLDL: very-low-density lipoprotein. 1 Probiotic intake of 5 × 107 CFU is considered insufficient to achieve the benefits described by the International Scientific Association for Probiotics and Prebiotics (2025).
In the five studies included in this review [28–32], validated tests were used to measure cognitive function. To enhance comparability across heterogeneous studies, results were synthesized narratively and summarized in a structured table (Table 6) indicating the direction of effects and statistical significance for each outcome. Probiotic supplementation showed a significant increase (P < 0.05) in the MMSE [36] in three [29, 30, 32] of the four studies [29–32] that used it compared to the CG. In addition, Akhgarjand et al. [30] evaluated verbal fluency using the CFT, which showed a significant improvement (P < 0.05) in both supplemented groups, group 1 and group 2, with 2 capsules daily of 1 × 1015 CFU of Lactobacillus rhamnosus and Bifidobacterium longum, respectively, versus the CG. The study conducted by Agahi et al. [28] reported a non-significant increase (P > 0.05) in cognitive function improvement after supplementation with a mixed probiotic in the TYM memory test versus the placebo group that received maltodextrin. In the ADAS-Cog test, no changes were reported in the intervention group (IG) compared to the active CG [31].
The antioxidant capacity of AD patients benefited compared to the CG, with significant increases (P < 0.05) in SOD activity [31] and a non-significant trend toward increase (P > 0.05) in GSH [28, 32] and TAC [32]. In addition, the biomarker of structural lipid damage, MDA, decreased significantly (P < 0.05) [31], and MDA and 8-OHdG showed a non-significant trend toward reduction (P > 0.05) [28, 29]. Regarding the other oxidative stress mediators, there was a significant decrease (P < 0.05) in PCC [31] and a non-significant decrease (P > 0.05) in NO [28].
However, other studies [28, 29, 32] showed no changes after 12 weeks of probiotic supplementation between the two study groups, IG and CG, for oxidative stress biomarkers: TAC [28, 29], MDA [32], NO [29, 32].
Akbari et al. [29] reported a significant reduction (P < 0.05) in hs-CRP in AD patients supplemented with 200 mL of probiotic milk (2 × 109 CFU/g Lactobacillus acidophilus, Lactobacillus casei, Bifidobacterium bifidum, and Lactobacillus fermentum) compared to the group that received milk without probiotics. In addition, Tamaji et al. [32] reported a non-significant reduction (P > 0.05) in hs-CRP.
About inflammatory cytokines, only a significant reduction (P < 0.05) in IL-1β [31] and a non-significant reduction (P > 0.05) in TNF-α [28, 32] were observed in the IG compared to the CG. However, a significant increase (P < 0.05) in IL-6 was observed in the probiotic-supplemented group compared to the placebo group. IL-8 levels were similar in IG and CG in AD patients at the end of the 12-week clinical trial conducted by Tamtaji et al. [32].
The effect of probiotic activity on anti-inflammatory ILs was very limited. IL-10 increased [31] or decreased [28] without statistical significance (P > 0.05) after supplementation with probiotic mixtures of Lactobacillus and Bifidobacterium ssp. versus CG. Furthermore, the probiotic intervention had no different effect on TGF-β levels [32] compared to the group that received a placebo treatment of starch.
Lipid metabolism benefited from probiotic supplementation in AD patients after significant reductions (P < 0.05) in TG [29] and VLDL [29]. In addition, Tamtaji et al. [32] demonstrated a downward trend (P > 0.05) in LDL and TG. These researchers also described that LDLR expression showed no statistically significant differences (P > 0.05), allowing for lower LDL levels [32] in the IG compared to the CG. However, probiotic supplementation did not report variations in LDL [29], HDL [29, 32], and CT [29] levels compared to the non-supplemented group at the end of the studies [29, 32].
A non-significant decrease (P > 0.05) in HOMA-β production [28] and in INS concentration [32], with a tendency toward an increase (P > 0.05) in INS sensitivity, QUICKI [28], and a substantial reduction in INS resistance HOMA-IR [32] in IG versus CG. Although no statistically significant differences were observed, non-significant trends toward changes in INS-related parameters were reported. This would mean that the body needs less INS to maintain normal blood glucose levels, which can improve metabolic health.
In the study conducted by Tamtaji et al. [32], PPAR-γ increased without statistical significance (P > 0.05), which could potentially lead to a notable improvement in carbohydrate and lipid metabolism.
The physical condition of AD patients was assessed using BADL and IADL [30, 31]. For BADL assessed in two of the included studies [30, 31], no changes were observed between the two study groups, IG and CG. However, IADL experienced a significant improvement (P < 0.05) in the study conducted by Akhgarjand et al. [30], evaluated using the Lawton and Brody scale [37], in the IG (Lactobacillus rhamnosus and Bifidobacterium longum) compared to the CG.
In the study conducted by Hsu et al. [31], they reported that the IG intestinal microbiota showed slight changes with Bifidobacterium, Lactobacillus, Ruminococcus, Clostridium, and Akkermansia at the genus level, all of which increased, while Megamonas decreased. However, these changes at the phylum and genus levels were not significantly different after 12 weeks of probiotic supplementation compared to the CG.
Akhgarjand et al. [30] assessed anxiety using the GAD-7 scale [38], demonstrating a significant reduction (P < 0.05) in anxiety in patients supplemented with 1 × 1015 CFU of Lactobacillus rhamnosus (IG1) and Bifidobacterium longum (IG2) compared to the group that received a placebo.
BNDF showed a significant increase (P < 0.05) in plasma levels after probiotic supplementation with a combined supplementation of 5 probiotic strains [31] versus the CG.
The decrease in IG was significantly (P < 0.05) greater than in the active CG in cortisol levels [31].
No differences were observed between IG and CG in BMI in two of the included studies [28, 29].
Table 6 provides a structured overview of the randomized controlled trials included in the systematic review, detailing study design, participant characteristics, probiotic interventions, outcome measures, and main findings. Reported results indicate the direction of change and statistical significance for each outcome as described in the original studies. Findings should be interpreted in the context of study heterogeneity, sample size, and risk of bias.
The purpose of this systematic review was to critically investigate the effect of probiotic supplementation in people diagnosed with AD. A total of five studies [28–32] met the established inclusion criteria. Probiotics were administered in gastro-resistant capsules [28, 30–32] or mixed in milk [29], which act as protective vehicles for Lactobacillus and Bifidobacterium ssp. against stomach acidity and other unfavorable conditions in the digestive system, thus ensuring that they reach the intestine alive and active. Therefore, we assume that they could exert their potential beneficial effects. Participants who received probiotic supplements of Lactobacillus and Bifidobacterium ssp. showed significant benefits in cognitive function, BDNF, cortisol, GAD-7, and IADL. In addition, probiotic supplementation showed certain protective antioxidant and/or anti-inflammatory effects and effects on the metabolic profile. The interpretation of the observed effects should consider the methodological limitations identified in the risk of bias assessment. Uncertainties related to outcome assessor blinding and selective reporting may lead to overestimation of intervention effects, particularly in small trials with multiple outcome measures. Consequently, the consistency and magnitude of reported benefits should be interpreted cautiously. To provide a clearer analysis, this section has been divided into the following sections.
Cognitive impairment in AD patients is a progressive and irreversible process, beginning with short-term memory loss along with impaired attention, language, or executive functions [39]. Modulating this impairment is essential to mitigate the effects on AD patients [40]. In this regard, the results of this review showed significant benefits (P < 0.05) in MMSE [29, 30, 32] and CFT [31] and substantial benefits (P > 0.05) in TYM [28]. These results are superior to those described by Zhu et al. [41] and Tripathi et al. [42] for patients with AD, who reported that probiotic supplementation alone caused only modest cognitive improvement. The differences could be due to the different specific mechanisms of action of the probiotic strains administered, the cognitive status of AD patients, the bias of cognitive scales such as MMSE, and/or the study methods used. All these factors are essential for understanding the exact impact of probiotics on various cognitive domains. Importantly, probiotic effects cannot be extrapolated across formulations, as different strains and combinations may exert distinct biological actions within the gut-brain axis.
Probiotics, from the Lactobacillus and Bifidobacterium ssp., used [28–30, 32] in the studies included in this systematic review, could influence brain health, improving cognitive function by increasing the endogenous production of neurotransmitters such as serotonin, dopamine, GABA, and acetylcholine. These neurotransmitters improve cognition by participating in key processes such as attention, mood, concentration, memory, and learning, especially in AD [43]. In this sense, the gut-brain axis, where the gut microbiota communicates with the brain, plays a crucial role in the cognitive process with a beneficial impact on brain health [16]. Other mechanisms that could explain the improvements in the cognitive assessment scales, MMSE, CFT, and TYM, could be the ability of probiotics to reduce neuroinflammation [44], combat oxidative damage in the brain [45], and reduce INS resistance [32]. In addition, a significant decrease (P < 0.05) in cortisol in the IG compared to the CG [31] may be associated with slower cognitive decline in AD. High cortisol levels have been linked to faster memory loss and hippocampal shrinkage [46].
Oxidative stress, characterized by excessive production of free radicals such as reactive oxygen species (ROS) and reactive nitrogen species (RNS), hydroxyl radicals, and superoxide anions, can damage DNA, proteins, and lipids, contributing to neuronal death and neurodegeneration, which are common in AD. Oxidative stress even precedes and influences the formation of the two histopathological events, amyloid plaques and neurofibrillary tangles, suggesting an early stage of AD. In addition, oxidative stress contributes to mitochondrial dysfunction, neuroinflammation, synaptic function damage, and cognitive impairment [47].
Probiotic strains can exert antioxidant action by directly eliminating free radicals, increasing levels of antioxidant enzymes (SOD, catalase, and glutathione peroxidase), and stimulating the synthesis of antioxidants in the body [folate, GSH, vitamin K2, and B vitamins such as B2 (riboflavin), B5 (pantothenic acid), and B1 (thiamine)] and negatively influence the activity of enzymes that produce free radicals (NADPH oxidase and cyclooxygenase), potentially reducing their production of ROS and RNS [48]. In addition, probiotics can regulate the gut microbiota, attenuating dysbiosis, which may indirectly contribute to the host’s antioxidant capacity [49]. Lactobacillus rhamnosus IMC 501 and Lactobacillus paracasei IMC 502 reduced oxidative stress in athletes during 4 weeks of intense training [50]. Shen et al. [51] have shown that Bifidobacterium animalis neutralizes free radicals, reduces MDA concentration and lipofuscin levels, and improves antioxidant enzyme activity in a murine model. In addition, Lactobacillus plantarum P-8, administered to rats on high-fat diets, increases antioxidant capacity and protects liver function [52].
For all the above reasons, probiotic supplements of Lactobacillus and Bifidobacterium ssp. have substantial antioxidant potential [45], which is reflected in a significant reduction in oxidative stress (P < 0.05) for 8-OHdG [31], MDA [31], PCC [31] and a non-significant reduction (P > 0.05) in MDA [28, 29] and NO [28]. In addition, indirect antioxidant power through SOD was significantly increased (P < 0.05), and notable increases in TAC were reported (P > 0.05) [32]. However, the results of studies included in this systematic review [28, 29, 32] were less clear, as they did not report differences between IG and CG in TAC [28, 29], MDA [32], and NO [29, 32]. This could suggest that the effect of probiotics is not universal but depends on the individual characteristics of the supplemented subject, such as age, health status, lifestyle, and diet. In addition, the response to probiotics may depend on the predominant pathology, and not all probiotic strains are equally effective for all symptoms [53], especially complex ones in AD patients.
Inflammatory processes, whether neuroinflammatory or systemic, contribute to the development and progression of AD, contributing to neuronal damage, synaptic loss, and, therefore, cognitive impairment. Sustained neuroinflammation stimulates the accelerated formation of amyloid plaques and tau tangles and associated comorbidities such as sleep disturbances, depression, and anxiety, among others [54]. In this sense, the Aβ protein could disproportionately stimulate microglia or astrocytes, which would be responsible for producing large amounts of inflammatory cytokines IL-1, IL-6, and TNF-α [55].
Understanding these pathophysiological inflammatory mechanisms is crucial for developing innovative therapeutic strategies. In this regard, probiotic supplementation could act on the inflammatory status of AD by increasing the production of SCFAs such as butyric acid, stimulating antioxidant protection, inhibiting the activation of the NF-κB pathway, stimulating the production of anti-inflammatory cytokines, and suppressing the synthesis of inflammatory cytokines that modulate inflammation [44]. This would give probiotics the ability to influence neuroinflammation or systemic inflammation beyond the intestine with a potential reduction in inflammation markers [56]. In this regard, Fernández-Lázaro et al. [57] have described possible benefits after supplementation with several strains of Lactobacillus and/or Bifidobacterium for the inflammatory response compared to non-supplemented athletes after exercise, specifically based on significant increases in IL-10 and significant decreases in TNF-α, IL-6, and IL-8. Additionally, De Marco et al. [58] found that the supernatant of Lactobacillus acidophilus and Lactobacillus casei can reduce the release of TNF-α and stimulate the secretion of IL-10. However, the effects of supplementation with probiotics, Lactobacillus ssp. and Bifidobacterium, on cytokine levels may be inconsistent because the studies included [28, 31, 32] have shown mixed or contradictory results for proinflammatory cytokines [28, 31]. In this regard, Hsu et al. [31] reported a significant reduction (P < 0.05) in IL-1β, while Agahi et al. [28] showed a significant increase in IL-6. Although two included studies [28, 32] showed a notable downward trend (P > 0.05) in TNF-α. The same is true for anti-inflammatory cytokines, showing opposite results with non-significant decreases or increases (P > 0.05) for IL-1 [28] and IL-4 [31], respectively. This variability could be due to factors such as the specific probiotic ssp. used, the dose and/or duration of supplementation, the health status of the AD patient (pre-existing inflammation and neuroinflammation), and the timing of cytokine measurement [59, 60]. The observed increase in IL-6 in one included trial [28] should be interpreted with caution, as elevated IL-6 may reflect a pro-inflammatory response rather than a therapeutic benefit. This finding may be strain-specific, related to baseline inflammatory status, or represent a transient immune activation [28]. Therefore, probiotic supplementation cannot be considered to exert a uniform anti-inflammatory effect across all cytokine pathways in AD. Therefore, more well-designed randomized controlled trials are needed to better understand the effects of probiotics on cytokine profiles.
The observed ability of probiotics to significantly (P < 0.05) [29] or markedly (P > 0.05) [32] reduce hs-CRP, a key inflammatory biomarker in a complex disorder such as AD, represents a highly relevant finding. These results are similar to those described in two meta-analyses [61, 62] in patients with diabetic nephropathy [62] and schizophrenia [61] that have shown a significant reduction (P < 0.05) in hs-CRP levels, or in a trial in older adults [63] with low-grade systemic inflammation that showed modest effects without reaching statistical significance (P > 0.05) for hs-CRP reduction. Perhaps the reduction in serum hs-CRP levels observed in patients with AD or other pathologies receiving probiotic supplementation treatment is due to the beneficial effects demonstrated by these supplements through their anti-inflammatory, antioxidant, and restoration of the microbiome, which help to decrease the activation of inflammatory pathways, attenuating systemic inflammation, including intestinal inflammation and neuroinflammation [31, 44, 56, 58].
Probiotic supplementation modestly improved the lipid profile in AD patients. Akabari et al. [29] studied that 200 mL/day of milk with a probiotic mixture (2 × 109 CFU/g Lactobacillus acidophilus, Lactobacillus casei, Lactobacillus fermetum, and Bifidobacterium bifidum) helped to significantly reduce (P < 0.05) VLDL and TG. In addition, Tamtaji et al. [32] reported a notable (P > 0.05) decrease in LDL and TG, with a trend (P > 0.05) toward an increase in LDLR and PPAR-γ. In fact, Lactobacillus and Bifidobacterium have been widely shown to have beneficial effects in reducing cholesterol levels in vitro, in animal studies, and in human clinical trials [64]. Jones et al. [65] in their double-blind, randomized, parallel-controlled study demonstrated that consumption of yogurt with Lactobacillus reuteri (NCIMB 30242) produced reductions in TG (4.8%), HDL (8.9%), and LDL (6.0%). In addition, a meta-analysis reported the effectiveness of probiotics in reducing TG, TC, and LDL [66]. Probiotics modulate cholesterol metabolism by suppressing cholesterol synthesis and improving cholesterol transformation [64].
Some of the mechanisms described by Jia et al. [67] for improving the lipid profile involve the adsorption and absorption of cholesterol by bacteria, additional hydrolysis of conjugated secondary bile acids by microbial bile salt hydrolases, and reduction of cholesterol through the production of cholesterol-lowering metabolites, such as SCFAs and secondary bile acids. Probiotics accelerate the hepatic transformation of cholesterol into bile acids through the secretion of bile salt hydrolase or cholesterol reductase, thereby promoting the entry of cholesterol from the blood into the liver and reducing serum cholesterol levels [68]. In this regard, a recent study in fish (C. nasus) reported that supplementation with 1 × 108 CFU/g with Lactobacillus plantarum and Saccharomyces cerevisiae (9:1) in high-fat diets promoted cholesterol metabolism and fatty acid β-oxidation and inhibited fatty acid biosynthesis in the liver of C. nasus, reducing lipid accumulation [69]. In addition, Lactobacillus rhamnosus negatively regulated the expression of hepatic 3-hydroxy-3-methylglutaryl-CoA reductase mRNA in mice fed a high-fat diet, demonstrating a decrease in serum TC levels [70, 71], although the included study evaluating TC did not describe differences between the probiotic-supplemented group and the CG [29]. New-generation probiotics, such as Bacterioides, offer different mechanisms compared to traditional probiotics, such as the transformation of cholesterol into coprostanol and sulfonated cholesterol, which would be more specific and effective ways of reducing TC [67].
The reductions in TG [29, 32] described in this review could be related to the substantial increase (P > 0.05) in PPAR-δ signaling [32], which would promote β-oxidation of fatty acids [72]. In this regard, probiotic supplementation significantly reduces serum TG levels in mice fed high-fat diets, linked to increased hepatic expression of PPAR-δ and decreased expression of sterol regulatory element binding protein 1c (SREBP-1c) [73].
Improvements or avoidance of fluctuations in lipid profile, cholesterol, or TG through probiotic supplementation could contribute to an improvement in anthropometric parameters, although BMI did not show any changes compared to the CG after probiotic supplementation [28, 29] and a reduction in the risk of developing AD or other related dementias and cardiovascular diseases [6]. Cholesterol participates in the formation of lipid rafts, which are small areas in the cell membrane where APP is processed into Aβ protein, a key component of amyloid plaques in AD. Cholesterol synthesis and the transport of ApoE, a protein involved in cholesterol metabolism, can affect Aβ production, especially for the ApoE4 isoform [7], or alter the insulation of nerve cells in the brain [74].
Cerebral and peripheral INS resistance could accelerate cognitive dysfunction through hyperinsulinemia and altered INS signaling. In AD patients, INS levels are low in the brain and cerebrospinal fluid but high in plasma, and this may be related to altered signal transduction [75, 76]. In this regard, in AD, both the hippocampal formation and the cerebral cortex show reduced responses to INS signaling, with a notable impairment of the INS-IR-IRS1-PI3K and IGF-1-IR-IRS2-PI3K signaling pathways. It has been suggested that cerebral INS resistance, closely associated with IRS1 dysfunction, possibly triggered by Aβ oligomer activity, could promote cognitive impairment, independently of the classic pathology of AD, with altered hippocampal plasticity due to INS resistance [77].
In the two studies included in the systematic review [29, 32], which evaluate glucose metabolism, probiotic supplementation had no significant effect, although there was a notable tendency toward reduction in HOMA-β [29], HOMA-IR [32], and INS [32] with moderate increases in PPAR-γ [32] and QUICKI [32]. These results are like those reported in a meta-analysis conducted by Kasińska and Drzewoski [78], who reported a non-significant reduction in HOMA-IR, FPG, and INS levels in patients with type 2 diabetes mellitus supplemented with probiotics. In addition, Mazloom et al. [79] also reported that probiotic supplementation had a non-significant decrease in fasting blood glucose and substantial non-significant benefits on INS metabolism markers.
Although overall, probiotic supplementation would slightly improve sensitivity and decrease INS resistance, which would mean that the body’s cells would respond better to INS, requiring less INS to achieve the same reduction in blood glucose levels. The inconclusive results of studies included in the systematic review [29, 32] could be related to their methodology, such as dosages, administration of combined or individual nutrients, duration of intervention, and other possible confounding factors. Perhaps greater correction of dysbiosis could suggest restoration of neuroendocrine system dysfunction [49] or greater stimulation of GLP-1 [80], directly or indirectly mediated by SCFAs, produced by probiotics, would improve carbohydrate metabolism and INS sensitivity.
The improvement in the physical condition of AD patients is reflected in a significant improvement in IADL (P < 0.05) [30] after 12 weeks of supplementation with Lactobacillus rhamnosus and Bifidobacterium longum compared to the CG. Athletes have used probiotic supplementation to increase physical performance [81]. The possible mechanisms underlying the ergogenic benefits of probiotics are that probiotics can potentially improve oxygen uptake, reduce inflammation, and help maintain energy levels by improving nutrient absorption. In addition, probiotics may improve the bioavailability of NO, which can enhance vasodilation by intensifying blood flow and oxygen supply to muscles, help modulate and correct this inflammation, and exercise-induced muscle damage [82, 83]. Even the significant reduction (P < 0.05) in cortisol described by Hsu et al. [31] may contribute to faster recovery from IADL in AD patients. Taken together, probiotics would reduce recovery time and improve overall physical performance [82, 83].
However, extrapolating this directly to AD patients requires careful consideration, given that in two of the included studies [30, 31], no differences were reported for BADL between IG and CG at the end of the study. Therefore, although probiotics may have beneficial effects on cognitive function and other aspects of health relevant to patients with AD, such as modulating the gut microbiota by restoring balance and reducing dysbiosis, reducing inflammation, improving INS sensitivity or other metabolic parameters, cognitive function, and antioxidant action [41, 44, 56, 57], the mechanisms and results may differ from those observed in athletes.
The health and balance of the gut microbiota is key in various neurodegenerative diseases due to its bidirectional communication with the CNS through the gut-brain axis [84]. In the study conducted by Hsu et al. [31], they reported that the gut microbiota of IG showed slight changes with increases in Lactobacillus, Bifidobacterium, Ruminococcus, Clostridium, and Akkermansia ssp., and a decrease in Megamonas. In general, probiotics would modulate intestinal dysbiosis through various mechanisms, such as competition with pathogens, stimulation of the immune system (increased production and release of IgA, IL-10, and TGF-β), and improvement of intestinal barrier function (increasing mucin glycoprotein production, stimulating goblet cells, and strengthening tight junctions), inducing the production of substances that inhibit the growth of pathogens, such as SCFAs and bacteriocins [53]. An increase in bacteria such as Lactobacillus and Bifidobacterium is associated with an improvement in the intestinal barrier, the immune response, and a slowing of the progression of cognitive decline in AD [85]. Clostridium butyricum induces butyrate (which is an SCFA), with antioxidant and anti-inflammatory effects, and is a vital nutrient for intestinal health, as it serves as the main source of energy for colon cells [86]. Akkermansia reduces systemic inflammation (IL-6) and has a positive effect on metabolic imbalances by positively influencing lipid and glucose metabolism [87]. Furthermore, in AD models implemented in animals, an increase in Akkermansia could stimulate better and greater cognitive performance [87]. A reduction in the Megamonas population is associated with an improvement in the neuroinflammatory process [88], and an increase serves as a predictive biomarker of rapidly progressing cognitive decline [89].
Serum BDNF is considered a biomarker of cognitive function in older adults [90]. The physiological role of BDNF includes neuroprotection and synaptogenesis, among other processes [91]. Given that the formation and proper modification of memories are essential for cognitive processes and mental health, an imbalance in BDNF levels is associated with various psychological and neurodegenerative disorders such as AD. Consequently, the decrease in BNDF associated with AD can rapidly impair synaptic plasticity and lead to accelerated neurodegeneration [92]. In fact, it has been shown that serum BDNF levels in patients with AD were lower than in healthy individuals [92].
The results of a previous meta-analysis suggest that a combination of several Lactobacillus and Bifidobacterium ssp. significantly increases BDNF levels in people with neurological disorders with depression and neurological disorders [93]. These results are consistent with the study conducted by Hsu et al. [31], included in this review, which reported a significant increase (P < 0.05) in serum BDNF in the IG compared to the non-supplemented group after 12 weeks of supplementation with Lactobacillus rhamnosus and Bifidobacterium longum R0175 in patients with AD. Increases in BNDF would suggest an improvement in memory and neuroplasticity (the nervous system’s ability to change and adapt at a functional and structural level). In other words, BDNF could offer greater brain protection and expansion of neural networks, increasing cognitive-behavioral functions [91]. This would be supported by significant improvements in MMSE cognitive scores [31] in AD patients supplemented compared to a CG. It has been suggested that combined supplementation with Lactobacillus and Bifidobacterium could have a synergistic effect (greater than that of each genus separately) on increasing serum BDNF levels, which would provide benefits for brain health [94] through various mechanisms involving the gut-brain axis, such as modulation of the composition of the gut microbiota, production of SCFAs, and anti-inflammatory action [95].
Probiotic supplementation can induce anxiolytic effects in healthy older adults [96] and in athletes. Therefore, probiotic supplementation could represent a cost-effective and easily accessible intervention to support mental health and well-being in patients with AD. It should be noted that people with AD may experience anxiety as an emotional reaction to their cognitive decline and its impact on their lives [97]. In this regard, the study included in the systematic review conducted by Akhgarjand et al. [30] demonstrated a significant reduction (P < 0.05) in anxiety in patients supplemented with 1 × 1015 CFU of Lactobacillus rhamnosus (IG1) and Bifidobacterium longum (IG2) for 12 weeks versus CG. The gut-brain axis plays a role in regulating mood and anxiety, and probiotics may influence this connection through brain chemistry, such as the production of neurotransmitters like serotonin, which is crucial for mood regulation [98].
Although several trials reported statistically significant changes in selected cognitive and biological outcomes, these findings should not be interpreted as evidence of consistent or generalized clinical benefit. The observed effects were heterogeneous, strain-specific, and derived from small, short-term studies. Consequently, current evidence supports a preliminary and exploratory role for probiotics in AD rather than a definitive therapeutic application.
First, only a limited number of randomized controlled trials met the inclusion criteria, which restricts the overall strength of the available evidence. Excluding studies that combine probiotics with other nutritional interventions may limit the generalizability of the findings, particularly given that multimodal dietary approaches are common in clinical practice. Second, substantial heterogeneity was observed across studies with respect to outcome measures, probiotic strains and formulations, supplementation dose, and duration of intervention. The absence of quantitative synthesis limits the ability to estimate pooled effect sizes and should be considered when interpreting the findings.
Additional limitations should be acknowledged. Most included trials involved relatively small sample sizes and short intervention periods (12 weeks), limiting the ability to assess long-term clinical effects and sustainability of outcomes. Moreover, the included studies were conducted in a limited number of geographic regions, which may constrain the generalizability of the results to broader and more diverse populations. The absence of standardized probiotic formulations and dosing protocols further complicates comparisons across studies and hinders the identification of strain-specific effects. In addition, this systematic review was not prospectively registered in an international database, which may represent a potential source of methodological limitation, despite adherence to a predefined PICOS framework [22] and PRISMA guidelines [21].
Despite these limitations, this systematic review also has notable strengths. It was conducted in accordance with PRISMA guidelines [21] and incorporated a comprehensive search strategy across three major databases, complemented by grey literature screening. Furthermore, methodological quality and risk of bias were rigorously assessed using validated tools, including the McMaster questionnaire [25], the PEDro scale [26], and the Cochrane Risk of Bias 2.0 tool [27]. Importantly, all included studies were double-blind, randomized controlled trials in clinically diagnosed patients with AD, ensuring a minimum standard of internal validity.
Taken together, while current evidence suggests that probiotic supplementation may influence selected cognitive and biological outcomes in AD, the limitations highlighted underscore the need for future well-designed, adequately powered randomized controlled trials with standardized probiotic interventions and longer follow-up periods. Such studies will be essential to clarify clinical relevance, strain-specific effects, and translational applicability.
Probiotic supplementation has emerged as a promising complementary approach for modulating selected health-related outcomes in patients with AD. Evidence from a limited number of randomized controlled trials suggests that short-term supplementation with specific Lactobacillus and Bifidobacterium strains may be associated with improvements in certain cognitive measures, IADL, anxiety, and selected biological markers, including oxidative stress, inflammatory parameters, metabolic indicators, and BDNF levels. Importantly, no serious adverse events related to probiotic supplementation were reported in the included studies, supporting an acceptable short-term safety profile.
Nevertheless, the available evidence remains preliminary and heterogeneous. Reported effects varied across studies depending on probiotic strain, formulation, dosage, outcome assessed, and participant characteristics. In addition, small sample sizes, short intervention durations, and limited geographic representation constrain the generalizability and clinical interpretation of the findings. Consequently, probiotic supplementation cannot yet be considered an established therapeutic strategy for AD, and observed benefits should be interpreted with caution.
Future research should prioritize well-designed, adequately powered randomized controlled trials with standardized probiotic formulations, clearly defined clinical outcomes, and longer follow-up periods. Such studies are essential to clarify strain-specific effects, elucidate underlying mechanisms within the gut-brain axis, and determine whether probiotic supplementation can offer sustained clinical benefits as an adjunctive intervention in the management of AD.
8-OHdG: ultrasensitive 8-hydroxy-2’-deoxyguanosine
AD: Alzheimer’s disease
ADAS-Cog: Alzheimer’s Disease Assessment Scale-Cognitive
ApoE4: apolipoprotein E4 allele
BADL: basic activities of daily living
BDNF: brain-derived neurotrophic factor
BMI: body mass index
CDR: Clinical Dementia Rating
CFT: Categorical Verbal Fluency Test
CFU: colony-forming units
CG: control group
CNS: central nervous system
FPG: fasting plasma glucose
GAD-7: Generalized Anxiety Disorder-7
GSH: total glutathione
HDL: high-density lipoproteins
HOMA-IR: Homeostatic Model Assessment of Insulin Resistance
HOMA-β: Homeostasis Model Assessment of pancreatic β cells
hs-CRP: high-sensitivity C-reactive protein
IADL: instrumental activities of daily living
IG: intervention group
IL: interleukin
INS: insulin
LDL: low-density lipoprotein
LDLR: low-density lipoprotein receptor
MDA: malondialdehyde
MMSE: Mini-Mental State Examination
NI-AAA: National Institute on Aging and Alzheimer’s Association
NINDS-ADRDA: National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association
NO: nitric oxide
PCC: protein carbonyl content
PEDro: Physiotherapy Evidence Database
PICOS: Population, Intervention, Comparison, Outcomes, and Study design
PPAR-γ: peroxisome proliferator-activated receptor gamma
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
QUICKI: Quantitative Insulin Sensitivity Check Index
RNS: reactive nitrogen species
ROS: reactive oxygen species
SCFAs: short-chain fatty acids
SOD: superoxide dismutase antioxidant
TAC: total antioxidant capacity
TC: total cholesterol
TG: triglycerides
TGF-β: transforming growth factor-β
TNF-α: tumor necrosis factor-α
TYM: test your memory
VLDL: very-low-density lipoprotein
WOS: Web of Science
The supplementary table for this article is available at: https://www.explorationpub.com/uploads/Article/file/1004140_sup_1.pdf.
The authors would like to thank the Neurobiology Research Group at the University of Valladolid and the Department of Cell Biology, Genetics, Histology, and Pharmacology at the University of Valladolid School of Medicine for their collaboration in conducting this review and providing infrastructure.
DFL: Conceptualization, Investigation, Writing—original draft, Writing—review & editing, Visualization, Supervision. AMCSM: Data curation, Investigation, Writing—original draft. AMFA: Investigation, Writing—review & editing. BVO: Investigation, Writing—review & editing. JMA: Investigation, Writing—review & editing. GS: Conceptualization, Writing—review & editing, Visualization. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The datasets supporting the findings of this study are available from the corresponding author upon reasonable request.
The research is funded by the TCUE 2024-2027 Plan, approved by agreement on September 16, 2024, and has been selected within the framework of an operational program co-financed by the European Regional Development Fund (ERDF) and the Regional Government of Castile and León (067/230003 POC). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 2875
Download: 24
Times Cited: 0
Adnan Akhtar Shaikh ... Niveditha Nair
Salomón Páez-García ... Miguel Germán Borda
Zhengrui Li ... Jing Li
Ericka C. Loza López ... Felipe Esparza Salazar
Fernando Leal-Martínez, Irene Alejandra Berumen Coronado
Alejandro Borrego-Ruiz, Juan J. Borrego
Natalia Rojas-Sánchez ... Antonio Ibarra
