Affiliation:
1Centro de Investigación en Ciencias de la Salud (CICSA), Facultad de Ciencias de la Salud, Universidad Anáhuac México Campus Norte, Lomas Anáhuac, Huixquilucan, Mex 52786, México
Email: ericka.lozalo@anahuac.mx
ORCID: https://orcid.org/0000-0003-2839-0963
Affiliation:
2Escuela de Ciencias de la Salud, Universidad Anáhuac Puebla, San Andrés Cholula, Pue 72810, México
ORCID: https://orcid.org/0009-0006-1334-0399
Affiliation:
1Centro de Investigación en Ciencias de la Salud (CICSA), Facultad de Ciencias de la Salud, Universidad Anáhuac México Campus Norte, Lomas Anáhuac, Huixquilucan, Mex 52786, México
ORCID: https://orcid.org/0009-0003-2929-0742
Affiliation:
3Facultad de Ciencias de la Salud, Universidad Panamericana, CDMX 03920, México
ORCID: https://orcid.org/0009-0007-5008-6606
Affiliation:
1Centro de Investigación en Ciencias de la Salud (CICSA), Facultad de Ciencias de la Salud, Universidad Anáhuac México Campus Norte, Lomas Anáhuac, Huixquilucan, Mex 52786, México
ORCID: https://orcid.org/0000-0003-1127-9548
Affiliation:
4Medical Department, Centro de Rehabilitación e Inclusión Infantil Teletón Estado de México, Tlalnepantla, Mex 54010, México
ORCID: https://orcid.org/0000-0001-8504-8457
Affiliation:
5Área Académica de Medicina, Instituto de Ciencias de la Salud, Universidad Autónoma del Estado de Hidalgo, Pachuca de Soto, Hgo 42039, México
ORCID: https://orcid.org/0009-0007-0585-1129
Affiliation:
1Centro de Investigación en Ciencias de la Salud (CICSA), Facultad de Ciencias de la Salud, Universidad Anáhuac México Campus Norte, Lomas Anáhuac, Huixquilucan, Mex 52786, México
ORCID: https://orcid.org/0000-0003-2134-1843
Affiliation:
6Facultad de Medicina, Universidad Nacional Autónoma de México, CDMX 04510, México
ORCID: https://orcid.org/0009-0005-6014-3067
Affiliation:
1Centro de Investigación en Ciencias de la Salud (CICSA), Facultad de Ciencias de la Salud, Universidad Anáhuac México Campus Norte, Lomas Anáhuac, Huixquilucan, Mex 52786, México
ORCID: https://orcid.org/0009-0004-7282-9472
Affiliation:
3Facultad de Ciencias de la Salud, Universidad Panamericana, CDMX 03920, México
ORCID: https://orcid.org/0009-0005-3378-4897
Affiliation:
1Centro de Investigación en Ciencias de la Salud (CICSA), Facultad de Ciencias de la Salud, Universidad Anáhuac México Campus Norte, Lomas Anáhuac, Huixquilucan, Mex 52786, México
ORCID: https://orcid.org/0009-0002-2925-2896
Affiliation:
1Centro de Investigación en Ciencias de la Salud (CICSA), Facultad de Ciencias de la Salud, Universidad Anáhuac México Campus Norte, Lomas Anáhuac, Huixquilucan, Mex 52786, México
7Department of Neurosurgery and Brain Repair, Morsani College of Medicine, University of South Florida, Tampa, FL 33602, USA
ORCID: https://orcid.org/0000-0003-1884-5389
Explor Neuroprot Ther. 2026;6:1004146 DOI: https://doi.org/10.37349/ent.2026.1004146
Received: December 05, 2025 Accepted: March 05, 2026 Published: April 12, 2026
Academic Editor: Weidong Le, Sichuan Academy of Medical Sciences-Sichuan Provincial Hospital, the 1st Affiliated Hospital of Dalian Medical University, China; Rafael Franco, Universidad de Barcelona, Spain
The article belongs to the special issue Role of Microbiota in Neurological Diseases
The relationship between the gut microbiota and the central nervous system has gained attention as a biological axis that may influence the development of several neurological disorders. Recent evidence integrating genomic, neurobiological, and microbiota research shows how bacterial composition, host genetic variability, and the mechanisms of the microbiota-gut-brain axis interact in conditions such as autism spectrum disorder, epilepsy, and schizophrenia. These interactions function through neural, metabolic, and immunological related pathways involving intestinal and blood-brain barrier permeability. Genome-wide association studies (GWAS) and Mendelian randomization analyses highlight shared immunogenetic pathways that may shape both microbial profiles and neurological susceptibility. Consistent patterns of dysbiosis and alterations in neuroactive metabolites have also been reported, linking microbiota changes to neuroinflammation and disrupted neuronal signaling. This review synthesizes the current evidence supporting the integration of the microbiota-gut-brain axis and its underlying communication pathways. It also outlines the present therapeutic strategies for neurological disorders such as autism spectrum disorder, epilepsy, and schizophrenia, highlighting their potential to modulate neurological function. Additionally, it discusses the existing limitations in the field and offers insights into future research directions within this rapidly evolving area.
The incidence and Global Burden of Disease (GBD) of autism spectrum disorder (ASD), epilepsy, and schizophrenia (SCZ) have shown significant changes in recent decades, which has aroused interest in clarifying their etiopathogenic mechanisms [1–3]. In the case of ASD, a sustained increase in its overall incidence has been documented [3]. For epilepsy, a significant increase in its incidence and GBD has been reported between 1990 and 2021, with relevant regional variations [1]. On the other hand, the prevalence of SCZ remains stable, but its GBD has grown due to demographic changes and persistent environmental factors [2].
These conditions represent a substantial burden on public health due to their impact on quality of life, disability, and the associated social and health costs [4, 5]. In this context, traditional neurobiology, focused on the central nervous system (CNS), has been complemented by new approaches that integrate systemic models and host-microbiome interaction through the gut-brain axis (GBA) [6].
Normal brain function depends on regulated neurodevelopment, synaptic plasticity, balanced excitatory-inhibitory neurotransmission, and continuous metabolic, immune, and neuroendocrine signaling. Although the CNS lacks a resident microbiome, peripheral systems modulate its function, particularly the gastrointestinal (GI) tract and its microbiota [7–11]. Disruption of these regulatory processes can alter neural excitability, neuroinflammatory responses, and brain connectivity, contributing to the pathophysiology of neurodevelopmental and psychiatric disorders [12–14]. ASD, epilepsy, and SCZ share overlapping alterations in brain physiology, including disrupted neurodevelopment, altered neuronal excitability, neuroimmune dysregulation, and neurotransmitter imbalance, with varying prominence across disorders [15–27].
In this context, research increasingly focuses on the microbiota-gut-brain axis (MGBA), a bidirectional communication network linking the CNS, the enteric nervous system (ENS), the immune system, the endocrine system, and the gut microbiota (GM) [15]. The GM, composed of bacteria, viruses, and fungi, constitutes a diverse and dynamic ecosystem that supports digestion, substrate metabolism, and immune development from early stages of life [28, 29]. Beyond these functions, the GM modulates neurophysiological processes, including synaptic plasticity, neurogenic inflammation, blood-brain barrier (BBB) permeability, and hypothalamic-pituitary-adrenal (HPA) axis regulation, while contributing to nutrient metabolism, short-chain fatty acid (SCFA) production, epithelial barrier integrity, and local and systemic immune regulation [30–32].
MGBA involves multiple signaling pathways: the nervous pathway, mediated by the vagus nerve (VN); the immunological pathway, through cytokines and activated microglia; the metabolic pathway, involving SCFAs, tryptophan/serotonin (5-HT), and γ-aminobutyric acid (GABA); and the epithelial pathway, which regulates intestinal permeability and the BBB [33–36]. While the MGBA supports physiological homeostasis, its dysregulation increases vulnerability by amplifying immune activation, epithelial permeability, and aberrant neuronal signaling [15, 32, 37–40]. Gut dysbiosis is associated with immunological alterations, disrupted microbial metabolites, neurotoxic cytokine release, altered tryptophan metabolism, and neurotransmitter imbalance, which together contribute to the pathogenesis of neurological and psychiatric disorders, including ASD, epilepsy, and SCZ. Collectively, these mechanisms highlight the multifactorial nature of MGBA dysregulation [33–36, 41–44]. In parallel, human genetic studies identify variants that predispose individuals to GI and psychiatric diseases, suggesting a shared biological basis between gut and brain functions [45]. Genome-wide association studies (GWAS) have shown that individual genetic variability modulates the composition of the GM, at least partly independent of environmental factors [28, 29]. Notably, genetic loci involved in the immune system or intestinal metabolism are simultaneously associated with microbial composition and psychiatric risk [45]. This genetic-microbiota interdependence suggests a pathway by which genetic predisposition modulates the GM, which in turn influences brain development and neuronal excitability [18, 19, 46]. However, most studies have focused on GI or metabolic disorders, while a systematic integration of host genetics, GM, and neurodevelopmental or brain-excitability disorders is still lacking evidence [47, 48].
Current treatments for ASD, epilepsy, and SCZ focus on symptom management through pharmacological and behavioral interventions, which often show variable efficacy, treatment resistance, and limited control of core disease features [49–52]. As a result, interest has grown in microbiome-based strategies, including prebiotics, probiotics, psychobiotics, postbiotics, synbiotics, and fecal microbiota transplantation (FMT), as well as neuromodulatory approaches such as VN stimulation that target MGBA pathways involved in immune regulation, microbial metabolism, and neural signaling [16, 52–64]. Despite promising preliminary findings, clinical translation faces major barriers, including methodological heterogeneity, small sample sizes, lack of standardized protocols, and limited understanding of causal mechanisms [65–67]. Key challenges include defining disorder-specific efficacy, identifying reliable response biomarkers, and translating experimental findings into reproducible and scalable clinical applications [67–69]. Given the emerging nature of this area, it is essential to critically review evidence that evaluates the interaction between host genetics and GM in relation to the development of ASD, epilepsy, and SCZ, while analyzing the mechanisms of the MGBA. Therefore, the aim of this article is to review and synthesize the evidence regarding the influence of host genetics on the GM and its implications in the development and treatment of ASD, epilepsy, and SCZ, with special emphasis on the mechanisms of the MGBA.
A comprehensive literature search was conducted using PubMed, EBSCO, Cochrane, Google Scholar, and ScienceDirect databases. The search strategy employed a combination of Medical Subject Headings (MeSH) including “Gut-Brain Axis”, “Microbiota”, “Autism Spectrum Disorder”, “Epilepsy”, “Refractory Epilepsy”, “Schizophrenia”, “Gut Microbiota”, “Dysbiosis”, “Microbiota-Brain-Immune System Axis”, “GWAS”, “Mendelian Randomization”, “Gut microbiota variations”, “Short Chain Fatty Acids”, “Probiotics”, “Psychobiotics”, “Postbiotics”, “Synbiotics”, “Fecal Matter Transplantation”, “Vagus Nerve Stimulation”, “Encapsulation of fecal biomaterials”, “Polymeric Drug Delivery”.
The initial database research allowed for a preliminary section of significant articles, which were able to help in the identification of more specific studies through the snowball method. This technique encompassed reviewing all the first reference lists of the selected articles, which helped us to track more citations of key sources to identify more literature important to the topic.
This review includes all types of scientific articles, as determined by the author’s criteria, conveying the relationship between the GM and the brain, its principal communication pathways, actual treatment, participant microorganisms, and its relation with genetic studies. This research included cohort studies, observational studies, randomized controlled trials, meta-analysis, case control studies, systematic reviews, experimental animal studies, book chapters, and narrative reviews. Moreover, there was no language restriction. No restrictions on publication year were applied; however, studies published between 2016 and 2025 were prioritized to capture the most recent evidence.
Moreover, articles were screened based on their title, abstract, and full text. The relevant information, including the main topics of the articles, was selected for a qualitative synthesis. Finally, a total of 206 studies were included in the present article.
The human microbiome is distributed across various organ systems, including the GI tract, skin, respiratory system, oral cavity, and urogenital tract. Each site contains a distinct microbial community that interacts with the host’s physiology at both organ and systemic dimensions [8, 70]. In the GI tract, the microbiome plays a central role in nutrient metabolism, production of SCFAs, preservation of epithelial barrier integrity, and regulation of local and systemic immune responses.
SCFAs, mainly acetate, propionate, and butyrate, are produced through anaerobic fermentation of dietary fibers by GM, primarily within the Firmicutes and Bacteroidetes phyla [31, 71]. Their production depends on the intake of non-digestible carbohydrates and can be influenced by diet, prebiotics, antibiotics, and environmental factors [37, 71].
Moreover, SCFAs have diverse effects on the host’s physiology. In the gut, they provide energy to colonocytes, strengthen epithelial barrier integrity, and regulate mucosal immunity. Butyrate promotes epithelial and regulatory T cell differentiation and induces anti-inflammatory cytokines such as IL-10, supporting immune balance and reducing inflammation [32, 37]. SCFAs also act as signaling molecules through G protein-coupled receptors (GPR41/FFAR3, GPR43/FFAR2), influencing metabolic, immune, and neuroendocrine pathways [32, 37].
Altered SCFAs production and microbiota composition are linked to inflammatory bowel disease (IBD), colorectal cancer, and neurodegeneration [31, 37, 71, 72]. On the other hand, reduced SCFAs levels are associated with impaired barrier function, inflammation, and metabolic dysregulation, whereas restoring SCFAs producing bacteria or increasing dietary fiber intake may have therapeutic benefits [37, 71]. Moreover, microbial metabolites and components can circulate systemically, affecting the function of remote organs and regulating processes including neuroinflammation, cardiometabolic homeostasis, and kidney function [8–10].
In this way, the GM can modulate CNS activity through immune, endocrine, and neural mechanisms, even though under normal conditions the brain itself is not directly colonized by microbes [11, 70]. In the context of the GBA, converging evidence suggests that alterations in microbial neural communication may contribute to several neurodevelopmental and neuropsychiatric disorders. Moreover, growing evidence indicates that disruptions in gut-brain signaling are implicated in the pathophysiology of conditions such as ASD, SCZ, and epilepsy by processes such as neurotoxic cytokine release, redirection of tryptophan metabolism toward the kynurenine pathway, glutamatergic dysfunction, neuroinflammation, and dopaminergic disturbances [42–44, 73].
In addition, SCFAs also influence immune and epigenetic mechanisms, both of which are altered in the present disorders [44, 74].
During intrauterine life and the first years after birth, the GM plays an active role in neurological development. The gut and the brain communicate through a set of signals mediated by the MGBA, with the fundamental components of this system being the VN, the HPA axis, microbiota-derived metabolites, immune mediators, and enteroendocrine signaling mechanisms [75, 76].
The CNS development during gestation is particularly susceptible to both internal and external environmental factors. During this period, the fetus is exposed to molecules derived from the maternal microbiota, whose metabolites are able to cross the placenta and circulate within the fetal bloodstream [75]. Maternal GM-derived metabolites have been shown to exert early influences on the fetal metabolome, modulating key processes involved in neurodevelopment. This microbial signaling participates in neuronal differentiation from approximately the 12th week of gestation onward [77], and also contributes to the formation of the BBB, myelination, and other essential aspects of CNS maturation [76]. Moreover, metabolites such as trimethylamine-N-oxide (TMAO), N, N, N-trimethyl-5-aminovalerate, and hippurate are associated with neurite growth processes and with the pathophysiology of various neurological disorders [75].
The BBB begins to form during early stages of gestation, establishing a highly selective barrier between the systemic circulation and the brain [76]. Likewise, the formation and proper functioning of the BBB rely on metabolic signals originating in the gut. Both microbiota-produced metabolites, such as SCFAs, and certain dietary components modulate essential homeostatic processes, participating in the direct regulation of the BBB [].
The influence of the GM on postnatal neurodevelopment is fundamental, particularly during the first three years of life, which are considered a critical period for CNS maturation []. From birth onward, the composition of the microbiome is progressively shaped by multiple factors, including the route of delivery and type of feeding, antibiotic exposure, and early contact with maternal and environmental microorganisms, all of which contribute to initial microbial colonization and its evolution throughout the early years of life [76, 78, 79].
During vaginal birth, initial microbial colonization of the neonate occurs through direct exposure to the maternal vaginal and fecal microbiota, such as Lactobacillus and Prevotella. In contrast, infants born by cesarean section develop a microbiota resembling the maternal skin flora, with a predominance of Staphylococcus [80, 81], along with a lower abundance of Bifidobacterium and an almost complete absence of Bacteroides [82]. Early-life nutrition is a major determinant of infant microbiome composition, human breast milk being one of its most important sources, containing an estimated microbial load of approximately 3 log₁₀ CFU/mL with more than 700 diverse microorganisms [82–84].
Therefore, the GM plays a decisive role in neurodevelopment from the early stages of gestation and continues to exert influence throughout the first years of life [82]. Moreover, maternal microbiota-derived metabolites modulate essential neurodevelopmental processes, while after birth, microbial colonization is shaped by multiple environmental factors [75, 78]. This dynamic interaction underscores the importance of a well-balanced intestinal ecosystem beginning in the prenatal period, as it represents a critical modulator of brain development and long-term neurological health [75].
Accumulating evidence over recent years indicates that the GBA plays a critical role in the regulation of CNS function, notwithstanding the anatomical distance between the gut and the brain [85]. The MGBA is formalized as a bidirectional contact where neuronal, neuroendocrine, metabolic, and immunological pathways, along with the substantial participation of both the VN and the HPA axis, facilitate communication between the gut, the brain, and immune system [86].
The neural pathway represents the main and fastest communication route between the gut and the brain, between the ENS and the VN, as well as in the modulation of neuronal activity by microbial metabolites [15].
As the tenth cranial nerve, the VN supplies intestinal innervation through its celiac and hepatic branches. It transmits afferent signals from the GI tract to the vagal nuclei in the brainstem, modulating both limbic and cortical centers [40]. The branches of the VN have sensory properties in their afferent area, representing 80% of the fibers originating in the nodosum ganglion. In contrast, motor properties in 20% of its fibers originate from the dorsal motor nucleus of the VN [40, 87]. Furthermore, it influences neural pathways that modulate emotions, mood, and cognitive functions [88].
Currently, the VN has been distinguished as a mechanosensor that not only detects mechanical stimuli through ion channels such as ASIC, TRP, and PIEZO, but also differentiates pharmacological and hormonal changes at enteric levels [40, 88]. Additionally, its nerve endings and sensory cells, called “neuropods”, have the capacity to detect metabolites and GM that significantly influence VN actions [88]. The GM has essential functions at the intestinal level, such as maintaining pH, controlling peristalsis, nourishing epithelial cells, providing digestive enzymes and vitamins, and regulating the excitability of nerve endings that affect immune and endocrine pathways related to the CNS [40, 88]. It is worth mentioning that the VN does not have direct contact with the GM. It communicates through epithelial cells that transmit luminal signals, diffusion of compounds, and bacterial metabolites; so neuroendocrine cells are a primary component for the chemosensitivity interface [89].
The immunological pathway actively participates in the intestinal barrier, not only as a static barrier but also as a dynamic one that responds to both internal and external stimuli [90]. It is composed of the intestinal mucosal epithelium, an outer mucosal layer with commensal microbiota, antimicrobial proteins, secretory immunoglobulin A molecules, a middle layer with specialized epithelial cells, and an inner lamina propria where the immune system is located, populated by innate cells such as dendritic cells and macrophages, as well as cells of the adaptive response such as T and B cells [90, 91]. Moreover, dysbiosis causes an exacerbated increase in harmful lipopolysaccharide (LPS)-forming bacteria, and because it directly affects the intestinal barrier, the gut becomes significantly more permeable, facilitating the translocation of pathogens into the bloodstream [15, 40]. This leads to a systemic inflammatory response mediated by the intestines’ lymphoid tissue in relation to GM, integrating neuro-inflammatory processes influenced by inflammatory cytokines such as IL-1β, IL-6, TNF-α and IFN-γ that cross both the intestinal barrier and the BBB, generating damage to resident microglia of the CNS and promoting neuroinflammation by activating TLR4 and the nuclear factor kappa B pathway [88].
The interaction between hormonal pathways and neuronal signals represented by the VN is fundamental for bidirectional communication of the MGBA [73].
Although its nerve endings do not maintain direct contact with the GM or the luminal contents, the VN detects intestinal chemical changes indirectly through enteroendocrine cells (EECs). These cells represent approximately 1% of the intestinal epithelium and function as chemo sensors capable of identifying GM-derived metabolites, particularly SCFAs [21, 74, 89].
Upon activation, EECs release mediators such as cholecystokinin, glucagon-like peptide-1, and 5-HT, whose intestinal synthesis depends on microbial metabolites [21, 89]. Likewise, 5-HT acts on 5-HT3 receptors present on vagal afferent fibers, allowing rapid communication with brain circuits related to autonomic, emotional, and cognitive regulation [89].
Another key group of metabolites that modulate EECs and VN activity is derived from tryptophan metabolism. Reducing circulating tryptophan levels in the GM, it affects serotonergic neurotransmission, which impacts the functioning of both the CNS and the ENS [38, 92].
As outlined above, GBA is recognized as a bidirectional system through which genetic, immunological, and microbiological factors influence both GI function and mental health. The coexistence of diseases such as irritable bowel syndrome (IBS), celiac disease, or IBD with psychiatric disorders such as depression, anxiety, or SCZ suggests the existence of shared genetic and pathophysiological mechanisms [78, 93, 94].
IBS affects 5–10% of the world’s population, and it’s more common in women than in men. It is characterized by a disorder of the GBA interaction in which people experience recurrent symptoms due to altered bowel habits, abdominal pain, and in some cases bloating. This syndrome is associated with mental health disorders such as depression and anxiety. The literature reports that people with IBS are three times more likely to develop depression and anxiety than people without the disease [95]. A meta-analysis conducted by Zamani et al. (2019) [96] reported that people with IBS have symptoms of depression in 39% and anxiety in 29%.
Physiopathologically, stress causes the autonomic nervous system to secrete corticotropin-releasing factor, which affects intestinal function and consequently causes GI symptoms, as it affects the microbiome by altering GI function [95]. The literature has reported that psychiatric disorders are the most common comorbidity of IBS, and that patients who have it experience more debilitating and severe GI symptoms [97].
Psychiatric disorders represent significant diseases because their etiology is complex, involving neurobiological dysfunctions, genetic vulnerability, and environmental factors. Currently, research has been conducted on the relationship between the GI tract and neuropsychiatric processes. Hence, the MGBA has been linked to the pathophysiology of various neurodevelopmental, psychotic, and affective disorders, as the literature reports that alterations in the composition of the GM, such as changes in bacterial metabolites or the presence of dysbiosis, directly influence neuroplasticity, neurotransmission, and the integrity of the BBB, favoring the pathophysiology of psychiatric disorders [15, 98]. The GM regulates fundamental brain processes related to microglia activity, neurogenesis, and myelination, all of which are altered in depression, ASD, epilepsy, and SCZ [93, 99]. Disruption of the GM has been linked to SCZ, ASD, and epilepsy by altering microbial diversity, composition, and metabolite production. In SCZ, dysbiosis is associated with pro-inflammatory metabolites, oxidative stress, and neurotransmitter imbalances that may exacerbate the disease pathology [100, 101]. On the other hand, ASD, microbial imbalances affect neurodevelopment, immune function, and gut barrier integrity, potentially influencing social and cognitive behaviors [15–17]. Similarly, in epilepsy, altered GM profiles can impact neuronal excitability, neurotransmitter levels, and systemic inflammation, contributing to seizure susceptibility and severity [16, 102, 103]. Lebwohl et al. (2021) [104] conducted a cohort study in Sweden, which included 19,186 children diagnosed with celiac disease, who were followed up for an average of 12.3 years. At the end of the study, they reported that children diagnosed with celiac disease were at a higher risk of developing psychiatric disorders.
At the same time, the integration of microbial genetics and psychiatric genetics has led to new advances in understanding the pathophysiological mechanisms of their relationship in psychiatric disorders. Genomic studies report that autoinflammatory and immune-mediated diseases share genetic characteristics with other disorders such as epilepsy and SCZ [105]. The literature on GWAS and their relationship with the microbiome has identified causal relationships with psychiatric disorders [46]. Knowing that there is a relationship between GM, immunology, and neurobiology with psychiatric disorders, the role of GWAS has been investigated, as they are crucial for identifying the genetic architecture shared with neurodevelopmental disorders such as ASD, epilepsy, and SCZ [106–108]. Therefore, we will investigate the associations of these disorders with GWAS (Figure 1).
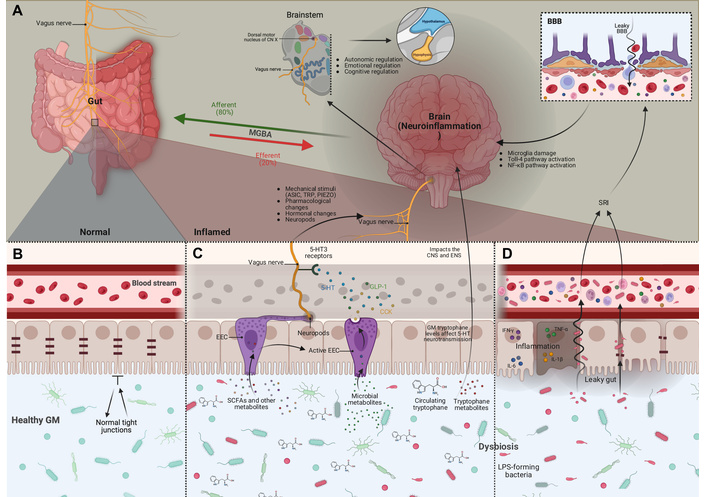
Schematic overview of the pathways of the MGBA. A. The VN is the bidirectional pathway of the MGBA, composed of afferent fibers (80%) that transmit intestinal signals to the CNS, and efferent fibers (20%) that regulate gastrointestinal functions. The VN detects mechanical stimuli through ion channels (ASIC, TRP, and PIEZO), as well as hormonal and pharmacological signals from the intestine through its sensory branches and neuropod cells. These inputs are transmitted to the dorsal motor nucleus of VN, from where vagal projections influence hypothalamic and pituitary activity, along with other brain regions related to autonomic, emotional, and cognitive functions. B. The intestinal barrier consists of epithelial cells with tight junctions. In the lumen, a balanced commensal microbiota interacts with this barrier; under normal conditions, it prevents the passage of luminal products into the circulation and supports the signals involved in MGBA. C. The VN detects intestinal changes indirectly by EECs that are chemosensors; these cells release mediators such as CCK, GLP-1, and 5-HT in response to microbial metabolites, particularly SCFAs, mechanisms that define the neuroendocrine/metabolic pathway. Once activated, EECs transmit these signals to vagal afferents through neuropods, while 5-HT stimulates these fibers via 5-HT3 receptors, sending information to brain regions. SCFAs also modulate EEC activity and neuronal plasticity, while tryptophan and its metabolites modify serotonergic neurotransmission in both CNS and ENS. D. When dysbiosis increases intestinal permeability, the MGBA immune pathway is activated. The loss of tight junctions allows microbial products such as LPS to enter the circulation, inducing pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, and IFN-γ). These mediators can cross the BBB, damage microglia, and promote neuroinflammation via TLR4 and NF-κB pathways. MGBA: microbiota-gut-brain axis; VN: vagus nerve; CNS: central nervous system; EECs: enteroendocrine cells; SCFAs: short-chain fatty acids; CCK: cholecystokinin; GLP-1: glucagon-like peptide-1; 5-HT: serotonin; ENS: enteric nervous system; LPS: lipopolysaccharides; BBB: blood-brain-barrier; NF-κB: nuclear factor kappa B.
ASD refers to a group of early-onset difficulties in social communication accompanied by repetitive sensory and motor patterns of behavior, which stem from a substantial genetic contribution along with additional contributing factors [109]. In ASD, GWAS have identified genes involved in synaptic signaling, neuronal development, and intercellular communication [110]. Interestingly, several of these genes, such as CNTNAP2, SHANK3, and NLGN4, also regulate intestinal functions and interaction with the microbiota [111, 112].
It is well known that patients diagnosed with ASD have alterations in their GM, characterized by dysbiosis, reduced microbial diversity, and elevated markers such as calprotectin and lactoferrin. These findings suggest the presence of persistent intestinal inflammation and altered immune pathways that simultaneously impact the GI system and neurodevelopment [18, 19]. Several systematic reviews and meta-analyses have evaluated the association between IBD and ASD, finding a higher prevalence of Crohn’s disease and ulcerative colitis in people with ASD, reinforcing the possibility of shared immunogenetic susceptibility [113, 114].
Previous data have served as the basis for establishing a bidirectional relationship between the microbiome and ASD, which has enabled the development of subsequent studies using Mendelian randomization (MR) to investigate genetic causality. These analyses have highlighted the role of intestinal permeability and interaction with immune genes, positioning dysbiosis as a modulator of genetic pathways relevant to neurodevelopment in ASD [115]. Along these lines, Yang et al. (2025) [116] combined GWAS and MR analyses, identifying a genetic overlap between loci associated with microbiota and immunity and loci at risk for ASD, demonstrating a shared genetic-microbial architecture. Complementarily, Hetta et al. (2025) [115] reviewed evidence of the GBA by integrating genetic data from ASD, highlighting genes linked to immunity, metabolism, and intestinal barrier function, suggesting common mediating mechanisms. This approach has been corroborated by Li et al. (2023) [117], who, using single nucleotide polymorphisms (SNPs) derived from GWAS consortia in MR analyses, demonstrated a bidirectional relationship between microbiota and ASD.
Epilepsy is characterized as a chronic neurological disorder that affects more than 70 million people worldwide. Its pathophysiology is complex, and 60% of cases are idiopathic [118]. Moreover, it has been demonstrated that patients diagnosed with epilepsy have GI symptoms, and patients with IBD are more susceptible to developing epilepsy, indicating a relationship between epilepsy and the gut. Ding et al. (2021) [119] reported that patients with epilepsy have an altered microbiota composition that promotes neuroimmunity and neuroinflammation.
A GWAS meta-analysis conducted by the International League Against Epilepsy reported that 26 risk loci for the development of epilepsy were found. The results of the analysis revealed that the most relevant variants are located in genes involved in neuronal synapses, and enriched genes expressed in inhibitory and excitatory neurons were found. The study, therefore, highlighted that the CACNA2D2 gene, which encodes the calcium channel subunit, is related to the risk of presenting epilepsy [106]. Likewise, a systematic review conducted by Jacobs et al. (2025) [120] aimed at identifying risk variants for epilepsy reported that 79 SNPs located in 64 genes were found to be associated with epilepsy. The most associated genes were SCN1A, which encodes the sodium channel subunit in neurons, which is essential for action potential. Other variants observed in the Asian population were rs2292096 and rs149212747, which were associated with epileptic status and focal epilepsy [120]. Moreover, Zhang et al. (2024) [121] identified genes that are related to the risk of developing epilepsy and reported 1,506 genes, classifying them according to their potential association with the development of the disease, highlighting SCN1A. Another associated gene is STXBP1, which is even related to encephalopathy, and the BCOR and APC2 genes [121]. Another gene associated with the development of focal epilepsy is PROX1, which encodes transcription factors and plays a role in nerve cell maturation [122]. Finally, a study conducted by Thakran demonstrated 30 variants in seven loci associated with the risk of developing epilepsy. The related loci were rs17031055/4q31.3 (DCHS2), rs9322462/6q25.2 (CNKSR3), rs73182224/3q27.2 (DGKG), rs2938010/10q26.13 (CTBP2), rs11652575/17p11.2 (SLC5A10), and rs75328617/8q24.23 (RNU1-35P) [123, 124].
SCZ is a psychiatric syndrome marked by psychotic features like hallucinations, delirium, and deorganized speech, as well as cognitive impairments affecting executive function, memory, and processing speed, which are common in this pathology [125]. Heritable factors could explain almost 80% of the risk of SCZ in the population; however, only a small part of this component has been demonstrated to be attributable to common disease-associated single-nucleotide variants [126].
GWAS and gene expression profiling have identified multiple genes that implicate immune-related pathways, inflammatory processes, cytoskeletal development, and synaptic function; a few of them have also been reported in IBD, particularly in regions that encode genes of the major histocompatibility complex and modulators of the intestinal immune response [127].
Thanks to large-scale GWAS studies, it has been possible to identify more than 270 loci associated with SCZ, including GRIN2A, DRD2, and MHC genes. These findings have served as the basis for identifying overlaps with GI disorders [93, 128]. Key findings include genes related to neurodevelopment and immune regulation, with common SNPs highlighting immune functions associated with GI disorders [129].
Nikolova et al. (2021) [19] reviewed the evidence on microbiota alterations in psychiatric disorders, including SCZ, noting that microbial changes may influence immune pathways and intestinal permeability linked to genetic risk factors.
There is currently evidence of a shared genetic etiology, mediated by immune mechanisms, between GI diseases and SCZ. This has been demonstrated through cross-GWAS and MR analyses, identifying overlaps of SNPs in the major histocompatibility complex, IL23R, and Th17 pathways [45]. For their part, Xie et al. (2024) [20] performed integrated GWAS analyses showing that SCZ shares multiple loci with GI disorders, identifying immune and dopaminergic genes that link GI and psychiatric phenotypes. Shared transcriptional mechanisms between inflammation and neuropsychiatric vulnerability have also been proposed. This was demonstrated by TWAS by the Uellendahl-Werth et al. (2022) [130] group, who identified genes such as PTPN22, ATG16L1, and HLA-DQA1, expressed in both the intestines and the brain. Similarly, Wang et al. (2024) [127] showed through genetic cross-analysis that SCZ and IBD share loci such as IL23R, STAT3, and TNFSF15, suggesting pleiotropy and possible bidirectional causal effects. This bidirectional causality between IBD and SCZ was also demonstrated by Qian et al. (2022) [131], who identified the involvement of IL23/Th17 and MHC genes through MR, proposing that immune dysregulation may affect both systems [131] (Table 1).
GWAS studies and their associations with neurological disorders.
| GWAS and its associations with epilepsy. | ||||
|---|---|---|---|---|
| Study | Type of study | SNPs and loci identified | Associated genes and immunological pathways | Main implications |
| International League Against Epilepsy Consortium on Complex Epilepsies, 2023 [106] | Meta-analysis | 26 Risk loci | CACNA2D2, SCN1A, and GABRA2 | Synaptic transmission, the balance between excitatory and inhibitory neurons |
| Jacobs et al., 2025 [120] | Systematic review | 79 SNPs associated with 64 genes | BRAP, SCN1A, and ALDH2 | Neuroinflammation and ion channel regulation |
| Malekpour et al., 2023 [129] | Literature review | 90 SNPs associated | APOE, IL1B, TNF, and SCN2A | Immune modulation and neuroinflammation |
| Zhang et al., 2024 [121] | Systematic review | 1,506 associated genes | APC2, SCN1A, BCOR, and STXBP1 | Neurodevelopment, vesicle transport, and ion channels |
| Thakran et al., 2025 [123] | Literature review | 30 SNPs in 7 loci | DCHS2, DGKG, CNKSR3, SLC5A10, CTBP2, and RNU1-35P | Cell adhesion and transcription regulation |
| Kim et al., 2025 [122] | Cohort | PROX1 | PROX1 | Neural maturation and transcriptional regulation |
| GWAS and their association with SCZ. | ||||
| Study | Type of study | SNPs and loci identified | Associated genes and immunological pathways | Main implications |
| Nikolova et al., 2021 [19] | Systematic review | None | Immune/barrier pathways | Microbiota influences SCZ immune biology |
| Gong et al., 2023 [45] | Cross-trait GWAS + MR | Shared SNPs | MHC, IL23R, TNF genes | Shared immune genetic etiology |
| Wang et al., 2024 [127] | Cross-trait GWAS + MR | Shared loci | IL23R, STAT3, TNFSF15 | Bidirectional IBD-SCZ links |
| Uellendahl-Werth et al., 2022 [130] | TWAS | TWAS loci | PTPN22, ATG16L1, HLA-DQA1 | Shared gut-brain transcriptional genes |
| Xie et al., 2024 [20] | Integrated GWAS | Multiple shared loci | Immune + dopaminergic genes | Broad polygenic overlap GI-SCZ |
| Owen et al., 2023 [126] | Cross-trait GWAS + MR | None highlighted | Inflammatory/neuromodulatory genes | IBS shares genetic architecture with SCZ |
| Qian et al., 2022 [131] | MR study | Existing SNP instruments | MHC, IL23/Th17 genes | Causal IBD-SCZ pathways |
| Trubetskoy et al., 2022 [128] | Large GWAS | > 270 loci | GRIN2A, DRD2, MHC genes | Defines SCZ polygenic architecture |
| GWAS and their association with ASD. | ||||
| Study | Type of study | SNPs and loci identified | Associated genes and immunological pathways | Main implications |
| Zhou et al., 2025 [132] | Narrative review (GWAS + MR) | Overlapping loci | Immune/metabolic genes | Shared microbiome-genetic |
| Hetta et al., 2025 [115] | Narrative review | Indirect | Barrier/immune genes | Genetic modulation of GBA |
| Malekpour et al., 2025 [133] | MR study | Microbiota SNPs | Microbial/immune genes | Bidirectional microbiota-ASD causality |
| Chen et al., 2022 [36] | MR study | Microbiota SNPs | Immune genes | Causal microbial effects on ASD |
| Chen et al., 2025 [134] | Cohort study | Metabolite SNPs | Metabolic genes | GI metabolites linked to ASD |
We describe GWAS studies related to epilepsy, SCZ, and ASD, where we describe the type of study, the SNPs and loci identified, and the associated genes and their pathophysiological involvement with the GBA. SNPs: single nucleotide polymorphisms; MR: mendelian randomization, GWAS: genome wide association studies; ASD: autism spectrum disorder; IBD-SCZ: inflammatory bowel disease-schizophrenia.
Although this section summarizes evidence linking alterations in the gut microbiome with GI disorders and psychiatric phenotypes, these associations should be interpreted with caution. The reported relationships are primarily correlational and do not necessarily indicate direct causal mechanisms, as they may be influenced by multiple confounding factors, including dietary patterns, medication exposure, disease chronicity, psychosocial context, and population heterogeneity across the studied cohorts.
Moreover, GWAS and MR analyses offer valuable insights into potential causal pathways; however, they are subject to important limitations, such as the risk of horizontal pleiotropy and the limited generalizability of findings, given that a substantial proportion of studies have been conducted in predominantly European or Asian populations. Consequently, these results should be regarded as hypothesis-generating rather than conclusive evidence, underscoring the need for replication in more diverse populations and for longitudinal study designs to better elucidate the complexity of these interactions.
As it has been described before, in recent years, there has been a growing interest in the role of the GM in neurodevelopmental and psychiatric disorders, due to the increasing understanding of the GBA over the years, contributing to conditions such as ASD, SZD, and epilepsy [112].
Over the past few years, multiple studies have demonstrated reproducible alterations in the composition of the GM in patients with ASD compared to neurotypical subjects. In a systematic review, Ding et al. (2017) [135] reported that most studies agree on a reduction of microorganisms such as Bifidobacterium and Prevotella, but an increase in Clostridium spp., Desulfovibrio, and Bacteroides in children with ASD. This suggests a pattern of intestinal dysbiosis and the activation of low-grade inflammatory processes, mechanisms that may contribute to alterations in the GBA. It was also noted that SCFAs, particularly propionic acid, play a key role in modulating neuronal gene expression [135].
In a study conducted by Wang et al. (2012) [136], 23 children with ASD were compared with 31 controls. Total fecal SCFA levels were significantly higher in ASD patients (136.6 ± 8.7 mmol/kg) than in controls (111.1 ± 6.6 mmol/kg; p < 0.05). Furthermore, propionic acid has been shown to modulate the expression of the tyrosine hydroxylase gene via the transcription factor CREB, suggesting an epigenetic mechanism through which SCFAs may influence monoaminergic neurotransmitter pathways such as dopamine (DA) and 5-HT [136].
Similarly, Kang et al. (2017) [137] conducted a clinical trial involving 18 children with ASD who received FMT for eight weeks, followed by an additional eight-week follow-up period. Using 16S rRNA sequencing, a reduced microbial diversity was initially observed in ASD patients compared to neurotypical children. The results revealed an abundance of Desulfovibrio and Sutterella and a significant decrease in Bifidobacterium. However, after FMT intervention, normalization of the microbial profile was reported, with an increase in Bifidobacterium and Prevotella, and a reduction in Desulfovibrio and Sutterella. Moreover, microbial changes correlated with significant clinical improvements in GI symptoms, including a reduction in constipation, diarrhea, and abdominal pain. These modifications were also associated with improvements in ASD behavioral scales, specifically the Childhood Autism Rating Scale and the Social Responsiveness Scale, both of which decreased by approximately 10% at the end of treatment. This suggests that changes in the GM occurred concomitantly with reductions in behavioral symptoms [137].
In the same line, Lewandowska-Pietruszka et al. (2023) [138] analyzed 44 studies on the composition and function of the GM in children with ASD. Specifically, Adams et al. (2011) [139], De Angelis et al. (2013) [140], Wang et al. (2012) [40], Liu et al. (2019) [141], and He et al. (2023) [142] reported an increase in Firmicutes ranging from approximately 36–81%, as well as Pseudomonadota around 78%, and a decrease in Bacteroidetes to about 56%. Consequently, the Bacteroidetes:Firmicutes (B:F) ratio tends to be around 56% compared with controls [40, 138–142]. These findings were associated with GI symptoms and more severe behavioral scores in several studies [138]. Moreover, the review highlighted that up to 70% of children with ASD reported GI symptoms, supporting a close clinical interaction between dysbiosis and somatic manifestations of the disorder. Additionally, the review presents heterogeneous but consistent evidence regarding alterations in microbial metabolites relevant to the GBA. Several primary studies, including Kang et al. (2018) [143], De Angelis et al. (2013) [140], and He et al. (2023) [142], showed changes in SCFAs profiles and tryptophan metabolites, with reported variations in propionate, butyrate, and acetate that correlated with behavioral parameters in some cohorts.
Collectively, these studies support that ASD is associated with a reproducible intestinal dysbiosis characterized by an increase in Firmicutes, particularly Clostridium spp., Desulfovibrio, and Sutterella, along with a decrease in Bacteroidetes, Bifidobacterium, and Prevotella [135, 138, 141–142]. These microbial shifts correlate with alterations in microbial metabolites, particularly propionate and butyrate, that modulate epigenetic and neurotransmitter pathways such as DA and 5-HT, in addition to influencing intestinal inflammation and epithelial permeability [136, 138, 143, 144]. In summary, the evidence supports a functional role of the GM in the GBA of ASD, although longitudinal studies are required to confirm causality and therapeutic potential [137, 138].
Unlike predominantly psychiatric disorders such as ASD, epilepsy is a neurological condition in which alterations of the GM have gained increasing relevance due to their potential influence on neuronal excitability and therapeutic response [49]. In recent years, interest in understanding this interaction has grown in both pediatric and adult populations [102]. Key lines of research include the role of the microbiota in drug-resistant epilepsy (DRE) and the microbial changes induced by the ketogenic diet (KD), which are considered potential mediators because of their anticonvulsant effects [102].
Although intestinal dysbiosis has been proposed to contribute to the pathophysiology of epilepsy through mechanisms of the GBA, the exact processes remain incompletely understood [49, 102, 145]. This microbial ecosystem plays an active role in immune modulation and inflammatory processes and produces multiple neurotransmitters involved in bidirectional communication with the CNS [21, 22]. These functions suggest that alterations in bacterial composition may influence neuronal excitability and, consequently, seizure threshold.
Various gut bacteria are capable of synthesizing neurotransmitters relevant to brain activity [21, 22, 102, 119]. Certain Bacillus species produce DA and norepinephrine (NE); Bifidobacterium generates GABA; Enterococcus and Streptococcus synthesize 5-HT; and Escherichia coli can produce both NE and 5-HT [49]. Given that epilepsy is characterized by an imbalance between GABAergic inhibition and glutamatergic excitation, these bacterial metabolites may play a key modulatory role in its pathophysiology [102].
De Caro et al. (2019) [146] explored the relationship between intestinal inflammation and epilepsy in a murine model. The authors induced colitis using dextran sodium sulfate in CD1 mice and observed that GI inflammation increased susceptibility to picrotoxin-induced seizures and reduced the anticonvulsant efficacy of drugs such as phenobarbital, valproate, carbamazepine, and phenytoin. Anti-inflammatory treatment partially reversed these effects, suggesting that intestinal inflammation enhances neuronal excitability and alters the response to antiepileptic drugs, reinforcing the functional link between GM and epilepsy [146].
Reproducible differences in the composition of the GM of patients with epilepsy have been described when compared with healthy controls [119]. Likewise, variations have been reported between the microbiota of pediatric and adult patients, as well as between those with DRE, leading to the proposal of differentiated dietary strategies [147]. In children with DRE, Xie et al. (2017) [148] reported reduced microbial diversity and a predominance of Firmicutes and Actinobacteria, along with a decrease in Bacteroidetes and Proteobacteria, suggesting a characteristic dysbiotic profile associated with the disease.
Several studies have identified alterations in the GM in epilepsy, generally characterized by reduced microbial diversity and dysbiosis compared with healthy controls [119, 149, 150]. Both studies reported decreased alpha diversity in patients with epilepsy, as well as increases in Actinobacteria and shifts in genera such as Blautia, Subdoligranulum, and Bifidobacterium, particularly associated with DRE [149, 150]. However, heterogeneous results were observed in other taxonomic variations: Gong et al. (2020) [150] reported an increase in Verrucomicrobia and a reduction in Proteobacteria, whereas Cui et al. (2022) [149] found higher abundance of Proteobacteria and Escherichia-Shigella in patients. Although these findings do not define a unique microbial signature, they collectively support the presence of an altered microbiota profile associated with epilepsy, particularly in DRE.
Conventional therapy for epilepsy relies on the use of antiepileptic drugs; however, around 30% of patients do not achieve adequate seizure control, a condition known as DRE [49, 50]. In these cases, it becomes essential to explore alternative or complementary treatments that may improve therapeutic response. Several studies suggest that DRE may be associated with alterations in the GM. Peng et al. (2018) [51] observed that patients with DRE who experienced four or fewer seizures per year showed a greater abundance of Bifidobacterium and Lactobacillus compared with those with more frequent seizures, suggesting that dysbiosis may contribute to the maintenance of neuronal hyperexcitability and pharmacoresistance.
Among the most promising non-pharmacological approaches is the KD, characterized by a high proportion of lipids, low carbohydrate content, and adequate protein intake [151–153]. This diet induces a state of ketosis with low glucose levels and elevated fatty acids and ketone bodies, which have been linked to antiepileptic and neuroprotective effects by enhancing neuronal energy efficiency and modulating inhibitory neurotransmission [152, 153]. Traditionally, the KD has been used in pediatric patients with refractory epilepsy, where it has shown significant clinical benefits. Martin-McGill et al. (2020) [151] reported that children treated with the KD exhibited up to three times less epileptic activity and were six times more likely to achieve ≥ 50% seizure reduction compared with those on conventional diets.
The therapeutic effect of the KD may be mediated, at least in part, by changes in the GM. Xie et al. (2017) [148] demonstrated that after one week of KD intervention, there was a relative increase in Bacteroides, Prevotella, and Bifidobacterium, along with a decrease in Firmicutes and partial normalization of the B:F ratio. These changes correlated with a ≥ 50% reduction in seizure frequency in 64% of patients [148].
Taken together, current evidence indicates that epilepsy, particularly DRE, is associated with gut dysbiosis characterized by reduced bacterial diversity and a B:F imbalance, which may contribute to both epileptogenesis and pharmacoresistance [102, 119, 146, 148]. This imbalance promotes a pro-inflammatory state and alterations in neurotransmitters such as GABA and 5-HT, fostering neuronal hyperexcitability [21, 22]. In this context, the KD has shown beneficial effects by remodeling the microbiota, increasing Bacteroides, Prevotella, and Bifidobacterium, and normalizing the B:F ratio, changes associated with significant seizure reduction [148, 151]. These findings strengthen the role of the GBA as a key component in the pathophysiology and management of epilepsy and DRE.
Analogous to ASD and epilepsy, SCZ has emerged as another neuropsychiatric condition in which the GM plays a relevant modulatory role. Over the past few years, metagenomic and metabolomic studies have demonstrated consistent alterations in the bacterial composition and microbial diversity of patients with SCZ, including a reduction of butyrate-producing bacteria and an increase in pro-inflammatory genera, suggesting a functionally active dysbiosis within the GBA [154].
In this context, a recent meta-analysis by Murray et al. (2023) [155] examined 10 studies including more than 1,200 participants and found that patients with SCZ exhibited a significant reduction in microbial alpha diversity (p < 0.001) and an altered B:F ratio, together with an increase in the phyla Proteobacteria and Actinobacteria. These changes reflect a pro-inflammatory and metabolically dysfunctional microbial state. The study highlighted the decrease of microbial metabolic pathways associated with butyrate synthesis, along with an increase in pathways producing propionic acid and succinate. At the genus level, Lactobacillus, Prevotella, Akkermansia, Clostridium, and Bacteroides were increased, while Faecalibacterium, Roseburia, and Coprococcus (butyrate-producing bacteria) were markedly decreased [155].
Complementarily, in a metagenomic study including 171 individuals (90 unmedicated patients and 81 controls), Zhu et al. (2020) [156] identified a significant increase in Akkermansia muciniphila, Bacteroides plebeius, and Clostridium symbiosum (p < 0.01) in SCZ patients. These species, which are involved in intestinal mucin degradation, were associated with increased epithelial permeability and LPS translocation-processes capable of activating microglia and inducing neuroinflammation. Moreover, the authors reported a reduction of Faecalibacterium prausnitzii, negatively correlated with plasma IL-6 levels, supporting the link between loss of butyrate-producing bacteria and systemic inflammation [156].
In another study, Li et al. (2020) [157] analyzed the GM of 90 patients with SCZ and 69 controls using 16S rRNA sequencing. They observed an increase of Prevotella copri and Lactobacillus fermentum in individuals with greater clinical severity according to the Positive and Negative Syndrome Scale. Correlations between these genera and negative and cognitive symptoms were r = 0.42 and r = 0.39, respectively (p < 0.05). The authors concluded that Prevotella, through the production of succinate and other intermediates of the tricarboxylic acid cycle, may contribute to neuronal mitochondrial dysfunction and altered cerebral energy metabolism described in SCZ [157].
Consistently, Deng et al. (2022) [23] in a cohort of 92 patients and 81 controls, confirmed a significant decrease of Faecalibacterium and Roseburia (p < 0.001), along with elevated Escherichia-Shigella and Ruminococcus gnavus. The loss of butyrate-producing bacteria was associated with higher levels of C-reactive protein and IL-6, and showed a negative correlation with total Negative Syndrome Scale score (r = −0.41, p < 0.001). This pattern supports the hypothesis that intestinal dysbiosis contributes to systemic inflammation and neuroimmune dysfunction in SCZ [23].
Similarly, Li et al. (2024) [24] integrated metagenomic and metabolomic analyses in 53 patients with SCZ and 57 controls, identifying an increase in Bacteroides and Alistipes (propionate-producing species) and a reduction in Coprococcus and Ruminococcus bromii (butyrate-producing species). Fecal levels of butyrate and isobutyrate were 35% lower in patients (p < 0.001), correlating with greater clinical severity (r = −0.46) and higher plasma IL-1β levels [24].
In summary, recent evidence confirms that SCZ is associated with intestinal dysbiosis characterized by reduced microbial diversity and a marked decrease in butyrate-producing bacteria such as Faecalibacterium, Roseburia, and Coprococcus [154, 155]. These alterations are accompanied by an increase in pro-inflammatory genera such as Prevotella, Lactobacillus, and Akkermansia, promoting a systemic inflammatory and metabolically dysfunctional state []. This state is reflected in elevated IL-6 and IL-1β levels, correlating with greater clinical severity and negative or cognitive symptoms [24, 157]. Finally, the decrease in SCFAs, especially butyrate, contributes to the dysfunction of the GBA and the neuroimmune impairment observed in this disorder [154, 155] (Table 2).
Summary of gut microbiota variations in ASD, epilepsy/DRE, and SCZ.
| Condition | Increased abundance | Reduced abundance | Related findings | References |
|---|---|---|---|---|
| ASD | -Firmicutes-Bacteroides-Pseudomonadota-Clostridium spp.-Desulfovibrio-Sutterella | -Bacteroidetes-Bifidobacterium-Prevotella | -Dysbiosis with low-grade inflammation.-Increased propionic acid and altered SCFAs profiles.-SCFAs modulation of DA and 5-HT in neuronal gene expression and monoaminergic pathways.-Changes in tryptophan metabolites.-Metabolite alterations correlate with GI symptoms and behavioral scores. | [135, 136, 141–144]. |
| Epilepsy and DRE | -Firmicutes (↑ in DRE)-Actinobacteria-Blautia -Subdoligranulum-Bifidobacterium (↑ in DRE and after KD)-Clostridiales-Bacteroides (↑ after KD)-Prevotella (↑ after KD) | -Bacteroidetes-Proteobacteria (variable)-Lactobacillales-Bifidobacterium (variable) | -Overall reduced alpha diversity.-Dysbiosis with altered B:F ratio.-Altered GABA, 5-HT, NE, and DA-producing bacteria.-Pro-inflammatory gut environment.-Stress-induced “leaky gut”.-DRE-associated dysbiosis linked to pharmacoresistance and seizure frequency.-KD remodels the microbiota (↑ Bacteroides, Prevotella, Bifidobacterium; ↓ Firmicutes; partial normalization of F:B ratio).-KD-associated ≥ 50% seizure reduction. | [21, 22, 49–51, 102, 119, 145–153]. |
| SCZ | -Lactobacillus-Prevotella-Akkermansia-Clostridium-Bacteroides-Escherichia/Shigella-Ruminococcus gnavus-Alistipes (propionate-producing) | -Faecalibacterium-Roseburia-Coprococcus-Ruminococcus bromii(All associated with reduced butyrate production, higher IL-6 and greater clinical severity) | -Altered B:F ratio.-Higher abundance of taxa linked to immune activation.-Reduced microbial pathways for butyrate synthesis.-Increased microbial pathways for propionate and succinate production.-Reduced alpha diversity.-Increased mucin degradation leading to “leaky gut”.-Greater translocation of LPS.-Prevotella-derived succinate and TCA-cycle intermediates.-Elevated pro-inflammatory cytokines (IL-6, IL-1β).-Decreased SCFAs. | [23, 24, 154–157]. |
The table outlines bacterial taxa with increased or reduced abundance in each condition, the associated metabolic or immunological findings, and key references supporting these observations. Notable patterns include shifts in the F:B ratio, alterations in SCFA-producing species, increased pro-inflammatory taxa, and changes linked to gut permeability, neurotransmitter pathways, and systemic neuroinflammation. ASD: autism spectrum disorder; DRE: drug-resistant epilepsy; SCZ: schizophrenia; KD: ketogenic diet; SCFAs: short-chain fatty acids; DA: dopamine; 5-HT: serotonin; GABA: γ-aminobutyric acid; NE: norepinephrine; LPS: lipopolysaccharide; TCA: tricarboxylic acid cycle; B:F ratio: Bacteroidetes:Firmicutes ratio.
Recent evidence suggests that alterations of the MGBA in psychiatric and neurodevelopmental disorders are neither uniform nor specific to a single diagnostic entity, but rather depend on multiple biological, environmental, and clinical conditions. The factors that may contribute to this variability are outlined below.
During childhood and adolescence, microbial alterations appear to exert a more profound impact on intestinal permeability, immune activation, and the production of neuroactive metabolites (such as SCFAs) [158, 159]. Pediatric patients with ASD or epilepsy exhibit a more unstable microbiota due to reduced bacterial diversity. In contrast, in SCZ, MGBA alterations appear to be influenced predominantly by chronic inflammation, metabolic status, lifestyle factors, and disease duration [160].
Sex-related differences have been demonstrated and are mediated by sex hormones, which modulate Th1, Th2, and Th17 lymphocyte expression as well as the composition of the GM. These effects alter intestinal permeability, SCFA production, and systemic inflammatory signaling toward the CNS [158, 161].
Low fiber intake and diets high in saturated fats or ultra-processed foods are associated with reduced microbial diversity, decreased abundance of butyrate-producing bacteria, and a pro-inflammatory state. In contrast, fiber-rich diets promote SCFA production, which has been associated with neuroprotective effects [162, 163].
Sustained activation of the HPA axis alters intestinal permeability through cortisol-mediated mechanisms, reinforcing a bidirectional cycle between dysbiosis, inflammation, and symptom exacerbation [158].
Psychotropic medications act as direct modulators of the MGBA by altering the composition and function of the GM. Antipsychotics, antiepileptics, and antidepressants reduce alpha diversity, decrease butyrate-producing bacteria, and promote the expansion of pro-inflammatory taxa (e.g., Proteobacteria) [160, 164, 165].
We concluded that MGBA alterations in ASD, epilepsy, and SCZ do not represent static or uniform biological states, but rather dynamic phenomena profoundly modulated by age, sex, diet, stress, comorbidities, genetic burden, pharmacological exposure, and the clinical course of the disease. This heterogeneity likely explains the variability observed in clinical presentation, therapeutic response, and prognosis across these disorders.
Current evidence suggests that prebiotics, probiotics, psychobiotics, postbiotics, synbiotics, FMT, and other microbiome-targeted interventions are under active investigation for their capacity to modulate the GBA in neuropsychiatric and neurodevelopmental disorders, including ASD, epilepsy, and SCZ. Nevertheless, the strength, consistency, and disorder-specific relevance of the available evidence vary substantially across both clinical conditions and intervention classes.
Probiotics are defined as live microorganisms that have a beneficial impact on the host. They are mainly composed of Bifidobacterium and lactic acid-producing bacteria. The metabolites synthesised by probiotics are key mediators in diet-induced host-microbe interactions, and can exert anti-oxidant and anti-inflammatory effects [16]. Prebiotics are termed as nonviable food components that can also regulate GM as an alternative to probiotic supplementation. They are mainly composed of oligosaccharides and fermented fibers, and have also shown positive effects on certain neurological disorders [53]. The stimulation of probiotic bacteria by prebiotics increases the production of can reverse the effects of dysbiosis and chronic stress; thus, they may be an effective treatment for neurological conditions [166].
A similar principle applies to ASD, in which it has been demonstrated that there is an increase in the content of Clostridium hystoliticum, a known toxin-producing bacterium, that leads to inflammation [57]. However, it is unclear whether these changes in the microbiota are responsible for the symptoms of ASD or are simply correlated with restricted dietary preferences [56]. In this regard, a study conducted by Mazzone et al. (2024) [56] showed that the use of probiotic L. reuteri had a significant positive effect on the reduction of social impairments and improvement of adaptive social skills, such as social communications, social motivation, and mental state understanding. Given that social functioning is complex and multifaceted, future studies should focus on specific clinical subdomains of social functioning.
DRE has forced scientists to search for alternative therapeutic alternatives. It has been shown that SCFAs have some effects on neuroinflammation in epileptic settings. These effects involve a decrease in oxidants and inflammatory cytokines with the subsequent restoration of mitochondrial function, as well as protection of BBB integrity and reduction of responses to psychosocial stress that contribute to epileptogenesis. SCFAs administration can be done through probiotics and prebiotics. Currently, there are several preclinical and clinical trials being conducted to properly assess the effect that this pathway of SCFAs administration may have on epilepsy patients [55].
In the case of SCZ, several studies in which probiotics have reported positive outcomes. These results include a decrease in circulating C-Protein levels and enhanced antioxidant capacity of plasma, indicating a reduction in inflammation [54]. Also, considering that an important side effect of antipsychotic medications is impairment of cognitive functions, a study carried out by Gronier et al. (2018) [52] reported that the ingestion of prebiotics improved cognitive flexibility in animal models when compared to placebo. While this requires replication with a larger study and in humans, cognitive decline is not unique to SCZ treatment, so this represents an important window of opportunity for further research.
Psychobiotics, defined as live microorganisms that, when administered in adequate amounts, confer mental health benefits through modulation of the GBA, have demonstrated potential in preclinical and early clinical studies in ASD and SCZ. These agents may influence neurotransmitter synthesis, immune signaling, and regulation of the HPA axis, with reported effects on clinical symptoms and inflammatory markers in these populations [58, 59, 167]. In ASD, psychobiotic interventions have been associated with favorable shifts in GM composition and increased production of neuroactive metabolites, including SCFAs and serotonin, both of which play key roles in neurodevelopmental processes. In addition, patients with ASD have been associated with altered levels of Bacteroides, Firmicutes, Prevoltella and Clostridium. These alterations can all be managed with the administration of psychobiotics [59, 60, 168]. In SCZ, psychobiotics and related probiotic formulations have shown potential to mitigate neuroinflammation and cognitive dysfunction; however, the clinical evidence remains constrained by small sample sizes and substantial methodological heterogeneity [169].
Synbiotics, combinations of probiotics and prebiotics, have been specifically studied in ASD. In vitro data suggest that synbiotics containing Limosilactobacillus fermentum K73 can shift the GM toward increased abundance of beneficial genera and enhance production of butyric acid and microbial serotonin, while reducing propionic acid, changes considered favorable in ASD pathophysiology [60]. These findings support the rationale for symbiotic interventions as adjunctive strategies in ASD and SCZ. However, the current evidence on the use of synbiotics is inconsistent, given that the formulations of the administered compound, the dosages, the lengths of treatment, the evaluation tools, and the method of administration are variable [170]. More research is warranted to fully uncover the benefits of this therapeutic option in all neuropsychiatric conditions.
Postbiotics, non-viable microbial products or metabolites, are emerging as a safer and more stable alternative to live probiotics, with anti-inflammatory and antioxidant properties. In SCZ, postbiotics such as SCFAs have been proposed to modulate neuroinflammation and BBB integrity, but clinical data are preliminary and largely extrapolated from mechanistic studies [59, 169].
Paraprobiotics, inactivated microbial cells, are not specifically addressed in the current medical literature relevant to ASD, epilepsy, or SCZ, and their role remains speculative.
Bacteriocins, antimicrobial peptides produced by bacteria, are not recognized as established therapeutic agents for GBA modulation in ASD, epilepsy, or SCZ in the current literature. Their mention in this context is not supported by clinical or translational studies in the referenced sources [59, 169].
For epilepsy, the reviewed medical literature does not provide direct evidence regarding the use of psychobiotics, postbiotics, synbiotics, paraprobiotics, or bacteriocins for GBA modulation or symptom management. In summary, psychobiotics and synbiotics have the most evidence for potential benefit in ASD and SCZ, primarily through modulation of GM, neuroactive metabolites, and immune signaling [58–60, 167–169]. Postbiotics represent a promising area for future research, particularly in SCZ. Paraprobiotics and bacteriocins are not established interventions for these indications. Across all categories, the field is limited by small sample sizes, inconsistent methodologies, and a lack of large-scale, long term clinical trials, highlighting the need for further research to clarify efficacy, safety, and mechanistic pathways [58, 59, 169, 171].
FMT is defined as a procedure involving the transfer of minimally handled fecal material from healthy donors to the gut of a recipient, and can be administered by a colonoscope, as an enema, as a nasogastric or nasoduodenal tube [172]. Also, for preoperative preparation, patients generally receive high-volume bowel cleansing regardless of the administration route. Moreover, some protocols additionally administer GI motility inhibitors to improve the retention of fecal microbiota in the receptor [173]. This process is intended to correct microbiota-related imbalances and associated diseases [174]. In today’s medicine, FMT has been investigated as a therapeutic approach for a range of communicable and noncommunicable diseases, with growing interest in recent years in its potential application to neurological and psychiatric disorders such as SCZ, epilepsy and ASD [61–63].
As previously mentioned, patients with ASD often present with GI disorders. This correlation was demonstrated by Hsiao et al. (2013) [175] using a maternal immune activation model. The offspring exposed to this maternal condition showed alterations in GM, including increased gut permeability observed in 3-week-old mice, as well as changes in social behavior similar to those seen in ASD patients.
As it has been mentioned before, Kang et al. (2017) [137] conducted a clinical trial with 18 children who received FMT for eight weeks with a similar follow-up period, where the microbiota changes significantly enhanced GI recovery, including constipation reduction, diarrhea, and abdominal pain. Moreover, these modifications were associated with a decrease of a 10% in conductual scales for ASD, such as the Childhood Autism Rating Scale and Social Responsiveness Scale, after the end of the treatment, which indicates that the changes of the microbiota occurred at the same time as the reduction of conductual symptoms in the patients [137].
As it has been previously mentioned, alterations of GM may have a close relation to neuronal hyperexcitability and neuroinflammation present in epilepsy [25–27]. Hence, it has been recently demonstrated that in genetically modified animal models of epilepsy, alterations in the GM occur at early stages, even before the onset of seizures, and persist as the condition progresses, and their manipulation is able to reduce the seizure recurrence [176]. Also, several studies have reported positive outcomes of microbiota transplantation. One of those cases reports a 22-year-old female diagnosed with Crohn’s disease and resistant epilepsy, who underwent three microbiota transplantation sessions with a subsequent 20-month follow-up, where it was evidenced that the patient had a full remission of seizures without the need for antiepileptic drugs [177].
For years, data have demonstrated that patients living with SCZ frequently suffer from concomitant GI disorders characterized by variations in microbiota communities in comparison to healthy patients [178]. Although the relationship between GBA and SCZ is not fully understood, some studies that have only been conducted in mouse models have shown a possible relation between the two. For example, in past years, Babulas et al. (2006) [179] showed that prenatal exposure to a microbial infection increased the risk of developing SCZ by 10–20 times later in life. Moreover, in a mouse study, researchers showed that mice that were colonized with microbiota of SCZ models produced behavioral alterations similar to glutamatergic SCZ profiles. On the other hand, such changes did not occur in mice colonized with microbiota from healthy controls. Correspondingly, SCZ microbiota recipients showed disruptions in microbial and host metabolic pathways related to amino acid and lipid metabolism, notably involving glutamate, a major player in SCZ pathology. Remarkably, post-transplantation abundances of Aerococcaceae and Rikenellaceae accurately discriminated SCZ model mice from controls in 100% of the population [180].
As it has been previously mentioned, VN is the main neural pathway of the MGBA. Its endings can detect gut microorganisms and their metabolites, subsequently conveying this information to the CNS [88]. For decades, vagal activity has been linked to complex behaviors such as attention, motion, emotion, and communication due to its connection to the prefrontal cortex and amygdala [64]. Vagus nerve stimulation (VNS) is a technique that uses electrical impulses to stimulate the VN, it requires surgical implantation of a bipolar electrode around the left cervical nerve and a pulse generator under the skin of the left chest. In this context, Steenbergen et al. (2021) [64] reported that VNS, applied via a mild electrical stimulus to the auricular branch of the VN, improved the recognition of specific emotions. These results strengthen the notion that VNS may serve as a therapeutic strategy to enhance social functioning in neurological disorders [64]. Its main side effects include surgical complications, dyspnea, paresthesias, headache, pharyngitis, pain, cough, or hoarseness [181].
As for ASD, this condition often presents with characteristics of low vagal activity, such as flat expression and intonation, difficulties with vocalizing, decreased response to social auditory information, and a low baseline cardiac parasympathetic activity [182]. Moreover, VNS has emerged as a possible adjuvant therapy for ASD [183]. A study conducted by Hull et al. (2015) [184] showed that VNS in children with ASD is associated with behavioral improvement, as well as seizure control in children with refractory epilepsy. Also, it has been proven that VNS considerably elevates the BDNF levels, which are associated with an increased neural plasticity, which is often dysregulated in patients with ASD [185]. While this represents a promising therapeutic alternative due to its neuroinflammation benefits, further investigations are necessary to validate these effects and thoroughly understand the underlying mechanisms [183].
VNS is approved for adults and children over four years old with partial-onset seizures, and it can be used off-label in younger patients. The VN, composed of 80% afferent fibers, projects to brainstem centers and cortical regions such as the thalamus, anterior cingulate, somatosensory cortex, and prefrontal cortex. It is through these pathways that VNS can desynchronize epileptiform activity, resulting in the regulation of thalamocortical circuits and lowering the frequency of epileptic seizures [186, 187]. Hence, VNS provides anti-inflammatory properties that also affect the GI system through the HPA axis activation and vasovagal reflex-induced cortisol release, which involves an anti-TNF effect [188]. In this way, VNS is able to treat resistant epilepsy and also treat inflammatory gut disorders like IBD, which has been shown to have a close correlation with the incidence of epilepsy. It has been demonstrated in meta-analyses that patients, independent of age, have shown considerable advantages after being treated with VNS [189]. Still, more studies that compare GI symptoms and disorders correlated to epilepsy are needed to demonstrate the possible dual benefits VNS can provide.
Considering the role VNS plays between inflammation and neuropsychiatric disorders, SCZ has also been involved in this category. It has been demonstrated that VNS induces changes in cerebral blood flow, with special reductions in the hippocampus and amygdala. Given that the hippocampus regulates the DA system in SCZ, Perez et al. (2014) [190] conducted a study in rats to determine whether VNS can be beneficial in the management of this psychiatric condition. They found that chronic VNS normalizes ventral tegmental area and hippocampal hyperactivity, and reverses hyperresponsivity to psychomotor stimulants. This leads to the conclusion that VNS may be a beneficial nonpharmacological treatment for SCZ. Despite its reported benefits, some studies have shown inconsistent results, likely due to the lack of standardized methods. Further research is therefore needed before it can be established as a robust therapeutic option for patients with SCZ or patients with schizophrenic-like symptoms [191, 192].
As previously mentioned, microbiome-based therapies are currently being actively investigated. A major challenge associated with these strategies is maintaining their viability under adverse conditions, such as gastric acidity and the action of digestive enzymes, to effectively reach their site of action. Various drug delivery systems have been developed to ensure the efficacy and safety of these therapies [193, 194].
Among these strategies, biopolymer-based encapsulation has been a promising approach, as it enables the formation of a protective barrier that enhances the survival of microbiome-based therapeutics during transit through the GI tract [194, 195]. The key properties of biopolymers employed in these encapsulation systems include biocompatibility, biodegradability, structural stability, elasticity, and sustainability, among others [194].
Other microbiome-based therapeutic strategies integrated into polymeric drug delivery systems include microbe-derived nanoparticles, microbiota-targeted probiotic formulations, microbiota-modulating hydrogels, microbiota-responsive nanoparticles, and oral FMT capsules [193].
Hydrogels are an example of polymeric platforms integrated into microbiome-based drug delivery systems. They are distinguished by a unique combination of properties that confer high versatility, including the ability to finely tune their fluidity and stiffness, as well as to recover their structural integrity after damage [196]. These features support their application in advanced drug delivery systems by enabling the controlled and localized release of therapeutic agents [193, 196].
Encapsulation of fecal biomaterials represents a significant translational step in reframing fecal microbiota transfer from a procedural intervention into a reproducible, orally deliverable therapeutic platform. Oral FMT capsules are designed to protect complex microbial communities from gastric degradation while enabling targeted intestinal release, improving patient acceptability, and facilitating standardization. Reflecting this transition toward a pharmacological framework, a double-blind, randomized, placebo-controlled trial has been designed to evaluate fecal microbiome transfer for GI symptoms in autistic adolescents and adults [197]. This trial exemplifies the shift from exploratory microbiome manipulation to controlled therapeutic development.
In summary, innovative drug delivery systems contribute to improving the precision and effectiveness of microbiome-based interventions, representing a key technological advance that enhances the stability, safety, and targeted delivery of these therapies, although more research is needed to truly demonstrate the long-term benefits of these therapies.
In recent years, interest in developing therapies aimed at modulating the GM has increased. However, the complexity of the biological, biochemical, immunological, and genetic interactions within the gut ecosystem poses a considerable challenge in designing robust experimental models capable of accurately assessing the efficacy of these interventions. The available evidence remains methodologically heterogeneous due to the wide range of strategies employed, such as FMT, probiotics, targeted dietary interventions, or neural stimulation, which vary significantly in dosage, treatment duration, and participant selection criteria. This methodological diversity hinders the ability to draw clear and reproducible conclusions regarding their clinical utility [65, 66]. Additionally, much of the current understanding of the gut–brain axis is derived from animal studies, which only partially reproduce human physiology, thereby limiting the extrapolation of these findings to real-world clinical contexts [198].
Currently, the greatest availability of experimental models for microbiome research is found in murine models, which have become fundamental tools for exploring biological mechanisms and generating hypotheses regarding potential causal relationships. However, these models present substantial limitations for clinical translation, as they do not fully recapitulate the complex interactions between the human microbiome and host-specific factors such as diet, neuroendocrine regulation, genetic background, immune status, and pharmacological exposure. Consequently, a central question remains regarding the clinical applicability of these findings: to what extent do the observed microbiome changes represent true causal determinants of disease, and to what extent do they reflect secondary adaptations to the biological and clinical context of the host [67]?
This uncertainty is further accentuated when considering the feasibility of personalized interventions based on genetic-microbial profiles. Although host genetics contributes to microbiome configuration, this contribution explains only a fraction of interindividual variability, whereas dynamic environmental and clinical factors exert a substantial influence on microbial composition and function. At present, there is no consensus on what constitutes a healthy or pathological microbial profile, nor on which combination of genetic variants, microbial taxa, and metabolic pathways would reliably predict therapeutic response. This raises critical questions regarding the temporal stability of such profiles, their reproducibility across populations, and their actual utility as tools for clinical stratification. Moreover, microbial profiles are highly dynamic and undergo continuous changes in response to host-determined conditions, such as diet, which represents an additional limitation for the development of predictive models capable of guiding personalized therapeutic strategies [67, 68].
In parallel, the scarcity of large-scale randomized controlled trials with standardized selection criteria limits the ability to establish direct causal relationships between specific microbiological profiles and mental or neurological disorders. In this context, it remains complex to discern whether the effects observed in experimental studies are driven by the implanted microbiome itself or instead reflect pre-existing host characteristics that condition the biological response. These methodological limitations are further compounded by bioethical considerations, particularly regarding interventions during early stages of life. Modulating the microbiome during critical periods of neurodevelopment raises significant ethical dilemmas, including the possibility of unintended long-term effects, the challenge of obtaining fully informed consent in pediatric populations, and the risk of medicalizing developmental processes in the absence of clear biomarkers of clinical benefit. Taken together, these issues underscore the need for more comprehensive research approaches, robust ethical frameworks, and longitudinal evidence before considering the widespread clinical application of microbiome-targeted therapies [199]. Furthermore, it is essential that the development of these emerging therapeutic strategies be guided by bioethical principles, ensuring equitable access to knowledge and its benefits, particularly in developing nations where socioeconomic constraints may limit implementation and must be addressed within a framework of protection and distributive justice [69].
The efficacy of these therapies is also influenced by the clinical, biological, and immunological context of each individual [65, 66]. For instance, many probiotic strains fail to achieve stable colonization of the gut, meaning that their potential benefits depend on continuous administration to maintain functionally relevant levels. Clinical trials have shown that individuals who consume probiotics for a long time exhibit greater changes and clinical benefits than those who consume them for a short time. Nevertheless, probiotics have been shown to increase microbial diversity even in the absence of stable engraftment. In animal models, prolonged antibiotic exposure followed by probiotic administration has resulted in the restoration of up to 99.8% of microbiota diversity compared to markedly reduced recovery in the absence of probiotics. These findings position probiotics as a potential adjunctive strategy for a variety of diseases [65].
In the case of the KD, although a significant reduction in seizure frequency has been observed in pediatric patients with refractory epilepsy, its mechanism of action remains unclear. However, the extent to which this therapeutic effect depends directly on microbiome-related shifts in microbial populations or on the metabolic alterations inherent to the ketogenic state remains unclear. Current evidence has not yet identified with precision which specific microorganisms are involved or which biomarkers mediate this response, limiting the ability to establish a robust causal relationship between microbial composition and the anticonvulsant effect. Therefore, further research is required to elucidate the mechanisms underlying the interaction between the KD and intestinal microbial populations [148].
FMT has shown therapeutic potential in various GI and extra-GI conditions; however, its use is limited by the challenges associated with its mode of administration. Colonoscopy, one of the most commonly used delivery routes, is an invasive procedure that carries risks such as intestinal perforation and may be unsuitable for critically ill patients [65]. Additionally, the selection of optimal donors still lacks global standardization, despite the need for stringent criteria, including clinical, serological, and microbiological evaluations, to ensure safety and prevent pathogen transmission [200].
Marcella et al. (2021) [201] conducted a review of 129 studies published between 2000 and 2020, including a total of 4,241 patients who underwent 5,688 FMT procedures, and reported that most treatment-related adverse events were mild and self-limited, such as diarrhea, abdominal distension, and nausea. However, the analysis identified that 1.39% of patients (59 cases) experienced serious adverse events directly attributable to FMT, among which five fatal outcomes were documented. Notably, these severe events occurred exclusively in individuals with pre-existing impairment of the intestinal mucosal barrier, underscoring the importance of rigorous candidate selection and careful evaluation of risk factors before the procedure. Most serious events were observed in the context of upper-GI endoscopic administration, highlighting the urgent need for standardized protocols and clear regulatory guidance regarding delivery routes and patient follow-up [201]. However, the implementation of microbiota-based therapies faces significant regulatory barriers. Depending on the region, these interventions may be classified as drugs, tissues, or procedures, leading to discrepancies in quality control, storage requirements, oversight, and authorization pathways for their use. Even when appropriate protocols are followed, the risk of transmitting potentially pathogenic microorganisms or antimicrobial resistance genes persists, necessitating robust regulatory frameworks and effective surveillance mechanisms. This lack of uniform regulation particularly affects low- and middle-income countries, where limited access to biomedical infrastructure increases patient vulnerability [202].
Despite these challenges, the future landscape of microbiome-targeted therapies remains highly promising. The development of next-generation probiotics, including those engineered using genetic technologies such as CRISPR, opens the possibility of creating microorganisms capable of producing immunomodulatory metabolites, functioning as biological sensors, or serving as delivery vehicles for targeted drug administration, with potential applications in inflammatory diseases such as cancer [203, 204]. These biotechnological advances are expected to enable increasingly precise and personalized medical approaches tailored to each individual’s microbial composition and may pave the way for novel therapeutic strategies for mental and neurological disorders.
One of these technologies is the use of bacteriophages as delivery vehicles for modified CRISPR systems without the need to introduce bacterial strains into the host, thereby facilitating the modification and selection of specific genes. For instance, experimental models have described the engineering of M13 phages loaded with base editors, demonstrating the successful modification of genes in bacterial strains isolated from a mouse model. These genetically edited strains maintained the modification for up to two weeks [205]. Such engineering approaches have enabled a growing number of experiments focused on the targeted modification of specific genes, with multiple potential applications, including gene inactivation or the eradication of antibiotic-resistant strains. Therefore, the prospect of creating microbial strains capable of producing immunomodulatory compounds or functioning as biological markers is entirely feasible [206].
Finally, the integration of predictive models based on artificial intelligence will enable the analysis of complex microbial patterns, identification of key species associated with health and disease, and generation of personalized recommendations, including dietary adjustments or therapeutic strategies tailored to an individual’s microbial profile [203]. These advances, combined with improvements in metagenomics and metabolomics, outline a future in which microbiota-based interventions can be developed with greater precision, safety, and efficacy.
Current evidence shows that both ASD, epilepsy, and SCZ are complex conditions, which although they have been studied for decades, until recent years, it has been proposed that their origin can be explained beyond neurobiology focused on the nervous system. The integration of host genetics, GM, and MGBA has allowed us to understand how neurodevelopmental, immunological, metabolic, and endocrinological factors converge.
The reviewed findings suggest that individual genetic variability influences microbial composition and, in turn, modulates key neurological pathways using microbial metabolites, peripheral immunity, and neuroendocrine regulation. This knowledge has encouraged the development of new microbiota-based therapies, such as next-generation probiotics, gene editing tools, and predictive models supported by artificial intelligence. Together with omics technologies, they offer us a promising panorama to carry out personalized interventions.
However, despite studies demonstrating the relationship and treatment of the present conditions with genetics and the microbiome, questions remain about the specific biological pathways by which these factors interact within MGBA. Understanding the pathophysiology involved will allow the development of precise therapeutic strategies adjusted to individual biology.
5-HT: serotonin
ASD: autism spectrum disorder
B:F: Bacteroidetes:Firmicutes ratio
BBB: blood-brain barrier
CNS: central nervous system
DA: dopamine
DRE: drug-resistant epilepsy
EECs: enteroendocrine cells
ENS: enteric nervous system
FMT: fecal microbiota transplantation
GABA: γ-aminobutyric acid
GBA: gut-brain axis
GBD: global burden of disease
GI: gastrointestinal
GM: gut microbiota
GWAS: genome-wide association studies
HPA: hypothalamic-pituitary-adrenal
IBD: inflammatory bowel disease
IBS: irritable bowel syndrome
KD: ketogenic diet
LPS: lipopolysaccharide
MGBA: microbiota-gut-brain axis
MR: Mendelian randomization
NE: norepinephrine
SCFA: short-chain fatty acid
SCZ: schizophrenia
SNPs: single nucleotide polymorphisms
TMAO: trimethylamine-N-oxide
VN: vagus nerve
VNS: vagus nerve stimulation
ECLL: Investigation, Writing—original draft, Writing—review & editing, Validation, Supervision. RMP: Investigation, Writing—original draft, Writing—review & editing. GCH: Conceptualization, Investigation, Writing—original draft, Writing—review & editing, Visualization. MFBG: Conceptualization, Investigation, Writing—original draft. AMV: Conceptualization, Investigation, Writing—original draft. XAVT: Conceptualization, Investigation, Writing—original draft. ESA: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. EJGA: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. PCMP: Conceptualization, Writing—original draft, Writing—review & editing. DDLV: Investigation, Writing—original draft. LMG: Investigation, Writing—original draft. IVAA: Writing—review & editing, Validation. FES: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
This paper represents independent research supported by Universidad Anáhuac México. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1394
Download: 17
Times Cited: 0
Adnan Akhtar Shaikh ... Niveditha Nair
Salomón Páez-García ... Miguel Germán Borda
Zhengrui Li ... Jing Li
Diego Fernández-Lázaro ... Juan Mielgo-Ayuso
Fernando Leal-Martínez, Irene Alejandra Berumen Coronado
Alejandro Borrego-Ruiz, Juan J. Borrego
