Affiliation:
1Department of Nutrition, Faculty of Health Sciences, Universidad Anáhuac, Huixquilucan, Estado de México 52786, Mexico
Email: ferman5@hotmail.com
ORCID: https://orcid.org/0000-0001-6649-5884
Affiliation:
2Independent Researcher, Mexico City, Mexico
ORCID: https://orcid.org/0009-0006-5713-4415
Explor Neuroprot Ther. 2026;6:1004150 DOI: https://doi.org/10.37349/ent.2026.1004150
Received: November 24, 2025 Accepted: March 18, 2026 Published: April 29, 2026
Academic Editor: Rodrigo Pacheco, Universidad San Sebastián, Chile
The article belongs to the special issue Role of Microbiota in Neurological Diseases
Growing evidence has directly linked the gastrointestinal tract, gut microbiota, and central nervous system, forming the gut-brain axis, a process that has been described as a key mechanism in regulating neurological processes. However, the presence of alterations in the composition of microorganisms in the digestive tract and dysbiosis has been linked to the activation of microglia, increased oxidative stress, alterations in the production of neurotransmitters, and exacerbation of neuroinflammation. These mechanisms have been associated with multiple pathologies and neurological conditions, and regulating them is key to the control of these diseases. In this context, various bacterial species play a neuroprotective role by promoting the integrity of the intestinal barrier, stimulating the synthesis of beneficial metabolites such as short-chain fatty acids (SCFAs), neurotransmitters, and modulating the inflammatory response. In addition, the characterization of these microbial profiles provides a broad perspective on understanding how changes in the microbiota contribute to the progression of neurological diseases. On the other hand, these new updates open up the possibility of designing personalised targeted therapeutic interventions that can regulate the gut microbiota and promote a neuroprotective and neuroregenerative environment. Another key point is that greater emphasis is placed on the need for more controlled clinical studies to validate efficacy and safety in humans, as well as knowledge of the mechanisms of action that make them possible. Finally, the modulation of the gut microbiota using probiotics, prebiotics, and postbiotics represents an innovative and effective opportunity to intervene in neuroimmune processes such as microglial activation, regulation of synaptic pruning, and neuroinflammatory pathways—processes implicated in various neurological diseases. In this context, this review integrates and analyzes the available evidence, highlighting potential interventions as treatments for these pathologies, as well as current limitations, to provide an updated framework to guide future research.
Neurological diseases are currently positioned as one of the main health burdens globally [1]. Recent estimates indicated by the Global Burden of Disease Study 2021, published in The Lancet Neurology with the participation of the World Health Organization, more than 3 billion people in the world were living with a neurological condition in that year, making this group of pathologies the leading cause of disability and poor health in the world. It has been estimated that since 1990, the global impact of these diseases has increased by 18% in premature mortality and disability-adjusted life years [2]. In addition, it is important to note that more than 80% of cases occur in low- and middle-income countries [3, 4]. Thus, the availability of neurological specialists is up to 70 times lower per 100,000 inhabitants compared to developed nations, and even if specialists are found, it is difficult for them to also understand the relationship and relevance of the intestine and its microbiota to these problems [5]. This panorama has created greater awareness and has encouraged the search for new strategies that can open new therapeutic and preventive opportunities, among which the gut microbiota and its direct interaction with the central nervous system (CNS) stand out.
The gut microbiota plays essential roles in the body’s metabolism, immunity, and homeostasis. Unquestionably, its role in digestive health is fundamental; however, evidence has shown its direct influence on neurological health through the so-called two-way communication between the gut and the brain [6]. This axis is supported by multiple pathways of interaction, ranging from immune system modulation and neuroinflammation, regulation of neurotransmitters and neurohormones, production of metabolites such as short-chain fatty acids (SCFAs), and other mechanisms [7].
In this way, intestinal dysbiosis emerges as a trigger point that can facilitate the onset and progression of neurological diseases [8]. Building on this understanding, it has recently been known that the loss of microbiota balance favors the pathological activation of microglia, the release of pro-inflammatory cytokines, the alteration of the integrity of the blood-brain barrier (BBB), and the modification in the availability of neurotransmitters directly related to neurodegeneration and synaptic dysfunction. In addition, specific bacterial strains capable of regulating these mechanisms have been examined, presenting beneficial effects in preclinical and clinical models [9]. Furthermore, another emerging strategy under study is fecal microbiota transplantation (FMT), as it seeks to restore a healthy microbial ecosystem, modulating neuroactive metabolites such as SCFAs, reducing microglia-mediated neuroinflammation, and favoring neuronal plasticity [10].
A comprehensive understanding of these mechanisms broadens our view of neurological disorders and expands the possibility of innovative interventions that could lessen the growing global burden of these diseases.
Consequently, numerous reviews addressing the relationship between the gut microbiota and neurological disorders have been published in recent years; however, most have focused on descriptive associations, taxonomic profiles, or specific neurological diseases. Significant gaps remain in the integrated understanding of shared etiological mechanisms, particularly those involving microglial activation, synaptic pruning regulation, neuroinflammation, and signaling mediated by microbial metabolites. In this context, the present review stands out by integrating experimental and clinical evidence to analyze the neuroimmune mechanisms associated with neurological pathologies, highlighting key areas that require further investigation.
The gut microbiota represents a highly diverse community of microorganisms found throughout the gastrointestinal tract. Its composition is mainly based on bacteria, although it has been specified that it also includes viruses, archaea, eukaryotic microbes, parasites, and fungi in smaller quantities [11]. This community of microorganisms is dynamic and has a genetic load that substantially exceeds the human genome, making it one of the most complex biological systems in the human body. Currently, it has been estimated that the metagenome of the human microbiome contains about 150 times more than the human genome itself [12]. For these reasons, the function of the microbiota and its symbiosis with humans and its balance in health is fundamental, as it is involved in various processes such as digestion, vitamin synthesis, regulation of the immune system, biological adaptation, and communication with the CNS [13].
Previously, it was said that the establishment of the intestinal microbiota began from birth to extrauterine contact. However, several studies have now shown the presence of bacteria or bacterial DNA in amniotic fluid and meconium [14–16]. Thus, by knowing that embryos and fetuses are already exposed to microbial signals during gestation, it is possible to understand the positive and negative effects of alterations in the maternal microbiota on embryonic and fetal development and growth. Likewise, it is associated with the maturation or deterioration of the immune, intestinal, metabolic, and neurological systems [17].
It is important to consider that the microbiota adapts and modifies over the years and responds to the adaptation and needs of the person, generating an individual fingerprint that responds to the physiological changes of each stage of life. Studies in animal models have shown that transplanting microbiota from older mice to young mice triggers inflammation in the CNS, damage to the integrity of the intestinal barrier, and microglial activation, generating adverse neurological and metabolic consequences [18]. Evidence has also been found indicating that when microbiota is transplanted from healthy young mice to older models with impaired microbiota, it may improve immune, cognitive, and metabolic function [19]. These findings indicate that the organism needs to be adapted through evolutionary correction of microorganisms, diet, and general habits to reach the optimal point of health at each stage of life.
Once the gut microbiota is established in the adult, it is mainly composed of bacteria belonging to the phyla Firmicutes and Bacteroidetes, which make up about 90% of the total. Groups such as Proteobacteria, Actinobacteria, Fusobacteria, and Verrucomicrobia are found in smaller numbers [13, 20]. This composition can be considered characteristic of a state of equilibrium or eubiosis, although its percentages change according to each person and are still under study. In addition, it serves as a starting point to understand the alterations that occur in pathological situations, including those associated with neurological disorders.
One of the important mechanisms of the microbial community is the ability to metabolize non-digestible polysaccharides such as fiber, contributing to the generation of SCFAs such as butyrate, propionate, and acetate, which are critical for gut and metabolic health [20]. In addition to participating in the synthesis of vitamins such as K, B1, B2, B5, B7, and B9, contributing to protection against pathogens, maintenance of the epithelial barrier of the intestine, regulation of behavior and mood, repair of the nervous system, memory, and immune maturation of the host [21, 22].
The distribution of the gut microbiota is not homogeneous; it varies in quantity and composition throughout the digestive tract. In proximal regions such as the stomach, duodenum, and jejunum, where more oxygenated conditions predominate, the microbial load is relatively low. On the other hand, a high concentration of strict anaerobic bacteria is observed in the colon, responsible for key processes such as the fermentation of complex carbohydrates and the production of SCFAs [13]. These compositional patterns by segment are summarized in Figure 1.
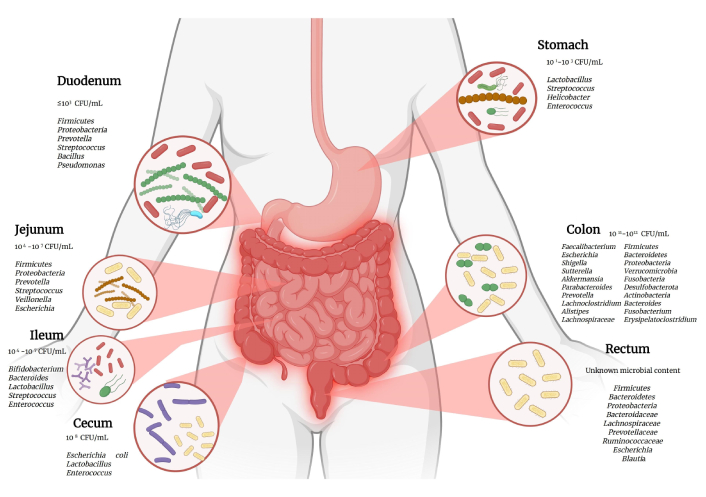
Segmental distribution of human gut microbiota (CFU/mL) [13, 23]. The estimated microbial load (CFU/mL) and the predominant bacterial genera in each segment of the intestine, from the duodenum to the rectum, are shown. CFU: colony-forming unit. Created in BioRender. (2026) https://BioRender.com/9rv9dro.
The human gut contains approximately 500 million neurons that, although smaller than the 100 billion present in the brain, constitute a highly complex and functional enteric nervous system. Through this system, it can communicate directly with the CNS through neuronal, immunological, metabolic, and endocrine pathways. All the connections that make up this network are called the microbiota-gut-brain axis, a bidirectional signaling system that favors the reciprocal modulation between the state of the gut microbiota and brain function [24].
One of the fastest and most direct bidirectional pathways of this communication is the vagus nerve, a key component of the parasympathetic system [25, 26]. The afferent pathway transmits information from the intestine to the CNS and accounts for approximately 80% of the fibers, while the efferent pathway that carries signals from the CNS to the periphery makes up 20% of the vagal fibers [27]. Thus, the main communication route in the microbiota-gut-brain axis is established from signals from the gut to the CNS, supporting the idea that the gut exerts significant control over brain function and neurological repair. At the same time, it is important to consider that the intestine coordinates the functioning of multiple systems through the different neuronal, inflammatory, and immunological communication pathways throughout the body [25–27]. In this way, clinically it translates into the regulation of functions such as heart rate and intestinal motility, in addition to transporting signals generated by the microbiota through the enteric nervous system, especially from the myenteric plexuses. Due to their conditions and complexity, bacteria do not interact directly with vagal fibers; they only indirectly modulate their signaling. This mechanism is key to understanding that it is not necessary to make direct effects, so it is necessary to understand all the postbiotics or mechanisms of action in these pathways [25, 26].
In this context, vagal signaling represents a pathway through which intestinal dysbiosis can translate into functional changes in the CNS, directly intervening in etiopathogenic processes and offering a possible therapeutic target directed at the gut-brain axis.
The gut microbiota has a major influence on the chemical interaction of the microbiota-gut-brain axis, not only through the vagus nerve, but also through the biosynthesis and regulation of essential neurotransmitters such as serotonin, dopamine, catecholamines, and gamma-aminobutyric acid (GABA). In the case of serotonin, it is estimated that approximately 90% is synthesized in the gastrointestinal tract, and although it does not directly cross the BBB [26], it can regulate breathing, cardiovascular response, blood flow, and indirectly stimulate the CNS to modify brain neurotransmission and thus modify human behavior [28]. In addition, it has inflammation-regulating functions by stimulating and inhibiting interleukins (ILs), which allows modulating the synthesis of tumor necrosis factor alpha (TNF-α) and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), among other ILs [29]. It is important to consider that the microbiota modulates tryptophan bioavailability and serotonin transporter (SERT) expression, participating in the reuptake of serotonin from the CNS and in the gut. These mechanisms indirectly influence the central synthesis of serotonin [30]. On the other hand, recent studies have shown that some gut bacteria can synthesize serotonin or its precursors, and this could influence host signaling [30, 31].
The biotransformation of serotonin into melatonin is a process of great relevance. Since melatonin not only contributes to the regulation of sleep, but it also acts as a powerful repair agent at the cellular level, associated with its antioxidant properties. It has been associated with enzymes such as glutathione peroxidase and superoxide dismutase, which could mitigate oxidative stress and protect the nervous system and other organs from premature aging as well as the degeneration of their functions [32].
On the other hand, some intestinal bacterial species express glutamate decarboxylase, which favors the conversion of glutamate into GABA. Microbial GABA has been proposed as a postbiotic measurer since changes in the microbiota are related to variations in both circulating GABA and brain levels that could have an impact on states of anxiety and stress, among others. In addition, some research has focused on the fact that specific transporters of serotonin and GABA allow their transit to the CNS, which could explain the effect of the microbiota and the intestine on cognitive functions and human behavior, as well as indirect pathways that send electrochemical impulses and may justify enteric control over the CNS [33].
In addition, it has been associated that several strains of intestinal bacteria, such as Lactobacillus, Bifidobacterium, Prevotella, and Clostridium, have participated in the biosynthesis, regulation, and catabolism of dopaminergic and serotonergic neurotransmitters, directly implicating cognitive and motivational functions and neuropsychiatric disorders such as depression and anxiety [34, 35].
Taken together, this evidence points to how the microbiota acts as a first-line chemical modulator within the gut-brain axis, establishing a functional bridge that takes signals from the gut environment and transforms them into central neuronal, hormonal, and immune responses [26].
In other words, microbial modulation of neurotransmitters and neurohormones not only has implications for central neurotransmission but also influences synaptic plasticity processes and neuroimmune regulation; these aspects are essential for the etiological understanding of the development of neurological pathology strategies.
The production of SCFAs in the digestive tract, such as acetate (60%), propionate (25%), and butyrate (15%), is involved in regulating the function of the nervous system and modulating neurophysiological processes [36]. These are resulting products derived from the fermentation of polysaccharides by gut bacteria, and several studies and research protocols have increased their relevance in the context of neurological diseases. These metabolites can play local roles in the regulation of intestinal homeostasis. However, they have also been associated with acting as systemic signalers with the ability to cross the BBB, modulating neuroinflammatory, brain plasticity, and epigenetic responses [26, 37].
For example, butyrate has been associated with the process of inhibiting histone deacetylases (HDAC), favoring gene expression that activates synaptic plasticity and memory processes. Its participation in G-protein-coupled receptors (GPRs; GPR41/GPR43) has been implicated, promoting anti-inflammatory responses and neurotransmitter regulation [36–38].
On the other hand, propionate has shown the ability to act on GPRs, reduce oxidative stress and inflammation, and modulate neuronal functions in serum and brain models of neurodegeneration [36]. This recent evidence supports that SCFAs are fundamental mediators of the microbiota-gut-brain axis, with therapeutic potential through dietary interventions, the use of prebiotics such as fiber or microbiota modulators that favor their restorative production. This evidence positions SCFAs as important key mediators in the communication between the gut microbiota and the brain, reinforcing the need to consider dietary and microbiomodulatory strategies as a fundamental part of the approach to neurological diseases. In addition to this, it has recently been observed that SCFAs have been associated with neurogenesis, since they promote the proliferation of neural progenitor cells. And in turn, SCFAs regulate microglia activity, modulating synaptic pruning and reducing neuroinflammation. This is relevant because neuroinflammation can cause excessive synapse deletion, compromising neuronal connections, and SCFAs can contribute to maintaining synaptic balance by regulating microglial signaling mechanisms [39, 40].
Consequently, the role of SCFAs in modulating microglia, BBB integrity, and epigenetic regulation represents a functional link between the gut microbiota and neuroinflammatory and neurodegenerative processes, with effective potential for diagnosis and intervention.
The gut microbiota plays an indispensable role in the control of the HPA axis, as it is responsible for managing the reaction to stress. Several studies that have been carried out in animal models, free of germs, have indicated that the alteration of the microbiota causes levels of hyperactivity in the HPA axis in situations that are considered stressful, and this has been evidenced by the increase in the levels of corticotropin [adrenocorticotropic hormone (ACTH)] and corticosterone [41].
Additionally, research indicates that circadian glucocorticoid patterns are altered in animal models without microbiota or treated with antibiotics, generating an alteration in synchronization with the body’s active cycle. With the above, studies indicate that microbiota not only modulate the magnitude of the stress response, but also the relationship with biological rhythms [42].
It is known that the continuous activation of the HPA axis not only affects brain function due to the prolonged increase in glucocorticoids, but it can also increase intestinal permeability and produce dysbiosis, facilitating the translocation of endotoxins such as lipopolysaccharides (LPS) and promoting levels of inflammation at the systemic level and neuroinflammation [43]. On the other hand, alterations in the microbiota that are produced by prolonged stress have been associated with variations in their composition and reduction of beneficial metabolites such as SCFAs, which act by regulating the immune and neuroendocrine response. Therefore, evidence suggests that the regulation of gut microbiota may be a protective factor against mood disorders and neurological diseases.
About 70% of the immune cells present in the body reside in the gut, making it a key immune focus. When there is an overgrowth of gram-negative bacteria in the gut microbiota, LPS are released that tend to activate innate cells such as macrophages, neutrophils, and dendritic cells. With this process, these cells generate pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, TNF-α) that can cross the BBB and alter the activation of microglia and glial cells, favoring neuronal dysfunction. In addition, these pro-inflammatory cytokines modulate afferent nerves and promote changes in signaling from the gut to the brain [9, 13, 44].
Under balanced conditions, the microbiota modulates the expression and function of Toll-like receptors (TLRs) in the intestine, especially in epithelial and immune cells, which is essential for proper immune maturation [13, 45, 46]. The alterations generated in TLR signaling derived from intestinal dysbiosis are likely to generate induction of chronic intestinal inflammation and cause an impact on inflammatory signaling pathways that, secondarily, tend to affect the CNS [46, 47].
On the other hand, it is important to consider immuno-neurological signaling pathways, which have identified at least 3 mechanisms in which the microbiota can affect brain function via immunomodulation. The first is the cytokines induced in the intestine that participate in circulating and crossing the BBB, directly affecting brain activity. The second is TLRs expressed in peripheral brain structures, which respond to circulating microbe-associated molecular patterns (MAMPs) and generate cytokines that feed back into the brain [47]. And, finally, the direct binding of circulating IL-1 to IL-1 receptors (IL-1Rs) in perivascular macrophages, which favors the triggering of prostaglandin E2 production and functional regulation of the brain [25].
Additionally, intestinal immune activation and its impact on neuroinflammation constitute a central axis in microglial dysfunction and processes such as synaptic pruning, highlighting the role of the gut-brain axis in the pathophysiology of neurological diseases.
On the other hand, it has been hypothesized in recent research that the gut microbiota regulates CNS homeostasis by shaping the phenotype of CD4+ T lymphocytes in the intestinal mucosa through the synthesis of specific molecular mediators. That is why, as long as the microbiota is under conditions of equilibrium and mediators such as GABA and SCFAs such as acetate, propionate and butyrate promote anti-inflammatory stimuli favoring the regulation of regulatory T cells (Tregs), a neuroprotective effect can be maintained, however, when there is the presence of dysbiosis, this composition of signals is altered, increasing mediators such as glutamate and dopamine that, by interacting with high-affinity receptors, they induce the differentiation of T cells into Th1 and Th17 pro-inflammatory phenotypes. It has been proposed that these autoreactive cells could be initially activated in the lymphoid tissues associated with the intestine either by molecular mimicry with microbial components or by the encounter with antigens in an inflammatory state to then infiltrate the brain, enhance neuroinflammation, and trigger neurodegeneration processes [48].
As a result of this complex network of interactions, intestinal dysbiosis represents a fragmentation in the equilibrium of the projections described above. This concept has been established as a mismatch in the composition of the microbiota, which contributes to the proliferation of pathogenic species and, therefore, reduces beneficial bacterial diversity. This condition generates a chronic inflammatory state, altering the production of neurotransmitters and compromising the integrity of the intestinal barrier and the BBB, intensifying the stress response through the HPA axis [24].
This is how dysbiosis is configured as a key point that favors the appearance or aggravation of neurological disorders.
The gut microbiota makes an important contribution to microglia homeostasis, regulating its capacity for surveillance, phagocytosis, and inflammatory response. In a state of equilibrium, microbial metabolites such as acetate, propionate, and butyrate maintain microglia as a modulatory phenotype, favoring a controlled expression of pattern recognition receptors (PRRs), thus limiting inflammatory activation. It significantly regulates the homeostasis of microglia, modulating the capacity for vigilance, phagocytosis, and inflammatory response [49]. However, the dysbiosis that occurs due to the reduction of bacteria that produce SCFAs and the increase in pro-inflammatory species favors the increase in LPS and metabolites such as N6-carboxymethyllysine (CML), which activate TLR4 and specific signaling pathways such as NF-κB, inciting changes towards a pro-inflammatory M1 microglial phenotype [50]. This M1 state is characterized by the overproduction of pro-inflammatory cytokines (IL-1β, TNF-α, IL-6), reactive oxygen species (ROS), and nitric oxide (NO), which intensifies the neuroinflammation process and causes synaptic dysfunction and axonal damage.
On the other hand, dysbiosis can increase the risk of intestinal permeability, authorizing the systemic passage of pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs), which in turn break the BBB and facilitate the passage of inflammatory signals to the CNS. Recently, it has been identified that the control of the intestinal microbiome can contribute to the reduction of microglial activation produced by a pathology, which is considered a fundamental part of neurological therapy.
Microglia are innate immune cells that are housed in the CNS, making up approximately 10% of the cells in the brain. Previously considered passive cells of brain pathology, they are now known to fulfill essential functions in the development, homeostasis, and disease processes. These roles include neurogenesis, angiogenesis, maintaining BBB integrity, pruning and remodeling synapses, supporting neuronal transmission, preserving myelin, and preventing the removal of cellular debris from apoptotic cells. The functions described above make microglia active guardians of brain health capable of responding swiftly and adaptively to environmental disturbances in the CNS.
It is currently known that the absence of microbiota produces immature and hypofunctional microglia, mainly characterized by changes in their morphology and metabolic dysfunctions that lead to a degenerative impact on cognitive and memory regions [9]. These findings confirm a primary link between the microbiota and its direct and indirect influence on the nervous system.
Additionally, intestinal dysbiosis acts as a critical factor in the overactivation of microglia, the brain’s innate immune cells, and the subsequent neuroinflammation—processes that undoubtedly contribute to the development and progression of various neurodegenerative diseases. The mechanism behind this complex interaction involves the disruption of the intestinal barrier caused by dysbiosis, which allows bacterial products and pro-inflammatory metabolites to translocate into systemic circulation. Once in the bloodstream, these stimuli tend to cross the compromised BBB [9, 49–53].
Within the brain, such metabolites and immune signals with altered profiles, such as bile acids like tauro-β-muricholic acid (TβMCA) and bacterial peptidoglycans, interact directly with microglia. This interaction results in microglia overactivation, evidenced by increased expression of markers such as ionized calcium-binding adapter molecule 1 (Iba-1), CD68, and CD16/32, along with characteristic morphological changes, such as increased cell size, roundness and shortening of prolongations, or the incorporation of amoeboid forms.
Microglial overactivation is characterized by the release of a cascade of pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β. Changes are also visualized at the molecular level, such as the activation of signaling pathways like TLR4/MyD88/NF-κB, the NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome, and p38 mitogen-activated protein kinase (p38 MAPK), which intervene in the neuroinflammatory state. An important aspect of this microglial dysfunction to consider is excessive synaptic pruning, a physiologically normal process that, when dysregulated, leads to synapse clearance, as seen in the placement of synaptic markers with activated microglia. Finally, this chronic neuroinflammation and microglia dysfunction directly contribute to neuronal loss, synaptic deficits, and impaired cognitive function, elements that are essential in the development and exacerbation of neurodegenerative diseases [9, 53, 54]. Figure 2 summarizes the central role of gut dysbiosis in neurological diseases.
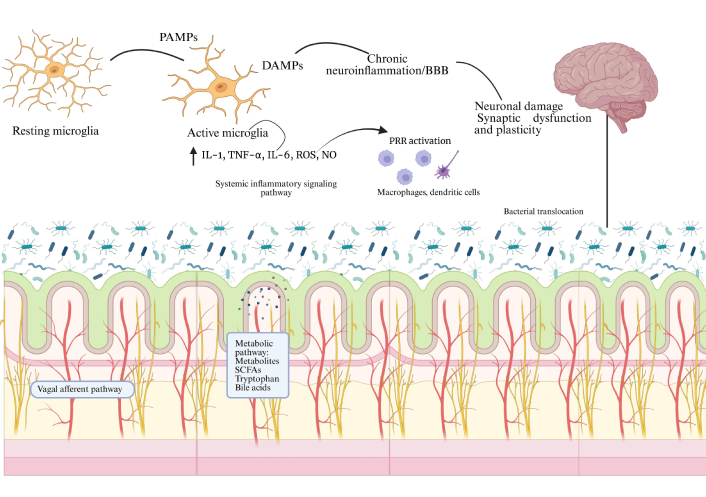
Mechanisms of dysbiosis-induced neuroinflammation. It is established that an imbalance in the gut microbiota compromises the integrity of the epithelial barrier, increasing intestinal permeability and contributing to the translocation of bacterial components such as LPS, PAMPs/DAMPs. These signals activate PRRs expressed by gut immune cells, both macrophages and dendritic cells, leading to the release of pro-inflammatory cytokines. Systemic inflammation and BBB dysfunction favor microglial activation and the transition from a pro-inflammatory state, resulting in chronic neuroinflammation, synaptic and neuronal dysfunction [49–55]. ↑ indicates increased levels. PAMPs: pathogen-associated molecular patterns; DAMPs: damage-associated molecular patterns; IL-1: interleukin-1; TNF-α: tumor necrosis factor alpha; ROS: reactive oxygen species; NO: nitric oxide; BBB: blood-brain barrier; PRR: pattern recognition receptor; SCFAs: short-chain fatty acids; LPS: lipopolysaccharides. Created in BioRender. (2026) https://BioRender.com/b96nbs0.
There are many bacterial strains and each has different effects on different signaling pathways, and even among them, they can self-regulate in their local and distant effects, all dependent on their balance in the digestive tract. Table 1 refers to the main bacterial species that have been linked to and have had a positive effect on different neurological disorders. In this visualization, those that contribute as a protective factor for the progression of the disease stand out. Increasing updates establish possible strategies for microbiome modulation.
Bacterial strains of interest in neurological diseases.
| Strain studied | Pathology or disorder studied | Intervention | Mechanism of action | References |
|---|---|---|---|---|
| Bifidobacterium breve | MPTP-induced Parkinson’s disease (PD) in mouse models | 1 × 109 CFU/200 μL of saline | Neuroprotection induced by reduction of neuroinflammation and oxidative stress, maintenance of barriers, modulation of microbiota, increase of SCFAs and neurotrophic factors, improving neurotransmitters and motor function. | Li et al. [56], 2022 |
| Bifidobacterium breve | Mild cognitive impairment in older patients with suspected condition | 2 × 1010 CFU for 24 weeks | It improves cognitive function, especially orientation, possible modulation of inflammation and microglia, decreased brain atrophy, and indirect effects via metabolites and cellular components despite minimal changes in the microbiota. | Asaoka et al. [57], 2022 |
| Bifidobacterium breve | Mild cognitive impairment in older adults | 2.0 × 1010 CFU for 24 weeks | Potential to improve cognitive function through the suppression of inflammation-related genes. | Kobayashi et al. [58], 2019 |
| Bifidobacterium breve | Mild cognitive impairment in older adults | 2 × 1010 CFU/day for 16 weeks | The cognitive benefits may be associated with anti-inflammatory effects, modulation of the inflammatory response, and promotion of hippocampal function. | Xiao et al. [59], 2020 |
| Lactobacillus rhamnosus GG | Noise-induced cognitive deficits and systemic inflammation in rats | 1 × 108 CFU/mL for 56 days | It improves cognitive deficits and reduces inflammation by modulating the gut-brain axis, strengthening the intestinal and blood-brain barrier. In addition, it increases SCFAs and regulates pro-inflammatory cytokines. | Li et al. [60], 2023 |
| Lacticaseibacillus rhamnosus E9 | MPTP-induced PD in a mouse model | 1 × 108 CFU/mouse/day for 15 days | It exerts neuroprotection by preserving dopaminergic neurons, reducing oxidative stress, and strengthening the intestinal barrier. | Aktas et al. [61], 2024 |
| Lactobacillus rhamnosus GG | Cognitive impairment in mice with sepsis | 5 × 109 CFU/mL for 1–3 weeks | It mitigates sepsis-induced cognitive decline by preserving brain-derived neurotrophic factor (BDNF) and p-TrkB in the hippocampus, protecting neuronal survival, and possibly modulating the microbiota-gut-brain axis. | Wang et al. [62], 2024 |
| Lactobacillus rhamnosus GG | Middle-aged and older adults with cognitive impairment | 20 × 109 CFU/day | Modulation of the gut-brain-microbiome axis through immune signaling, vagal pathways, enteroendocrine cells, and microbial metabolites; reduction of inflammatory cytokines and direct influence on neurotransmission and hippocampal pathways with indirect benefits. | Sanborn et al. [63], 2020 |
| Lactobacillus rhamnosus and Bifidobacterium lactis | Healthy older adults, with or without suspected mild cognitive impairment, experiencing age-related cognitive and emotional decline | 3.3 × 109 CFU/day for 10 weeks | Modulation of the gut-brain axis, including immune regulation and neuroprotection, reduction of inflammation, modulation of the hypothalamic-pituitary-adrenal (HPA) axis, improvement of stress and mood, as well as direct effects on neurotransmission, neurogenesis, and synaptic plasticity. | Ruiz-Gonzalez et al. [64], 2025 |
| Lactiplantibacillus plantarum HEAL9 | Cognitive decline and progression of Alzheimer’s disease | 1 × 109 CFU for two months | It improves cognition and intestinal motility, reduces astrogliosis, microgliosis, accumulation of Aβ1–42, and inflammation via inhibition of the NLRP3 inflammasome, as well as the increase of butyrate. | Di Salvo et al. [65], 2024 |
| Lactiplantibacillus plantarum | PD | 6 × 1010 CFU for 2 weeks | It improves motor deficits, modulates neurotransmitters, protects dopaminergic neurons, and reduces oxidative stress. | Lu et al. [66], 2021 |
| Lactiplantibacillus plantarum | PD, specifically in mouse models | 1 × 109 CFU in 200 μL of saline for 28 days | It protects the nigrostriatal pathway by preserving neurons and dopamine, decreases glial activation, neuroinflammation, and oxidative stress, as well as the increase in norepinephrine and neurotrophic factors. | Liao et al. [67], 2020 |
| Lactiplantibacillus plantarum | Patients with Rett syndrome (RTT) carrying MECP2 gene mutations | 6 × 1010 CFU/day | It likely regulates neurotransmitters and increases levels of BDNF, as well as reducing neuroinflammation and systemic oxidative stress. | Wong et al. [68], 2024 |
| Saccharomyces boulardii | Hemiplegic spastic cerebral palsy in murine models | 1 × 107 CFU/day for 9 days | Improvement of behavior and emotional conditions. In addition, reduction in inflammatory processes and modulation of the HPA axis. | Tao et al. [69], 2021 |
| Saccharomyces boulardii | Rotenone-induced PD in a rat model | 1 × 109 CFU/day for 60 days | Protection of dopaminergic neurons. As well as improvement of motor function, modulation of the intestinal immune response, increase in butyrate, and reduction of glial activation, neuroinflammation. | Ipek and Basaloglu [70], 2026 |
| Saccharomyces boulardii | Cognitive impairment associated with antibiotic-induced intestinal dysbiosis in a murine model | 90 mg/kg once a day for 21 days | Prevention of dysbiosis due to antibiotics, normalization of intestinal permeability, reduction of inflammation and oxidative stress, generating protection to hippocampal neurons, reversing cognitive deterioration. | Roy Sarkar et al. [71], 2021 |
| Akkermansia muciniphila | Liver disease and cognitive impairment in a murine model | 1 × 109 CFU/mL in 200 μL | Reduction of inflammation and intestinal permeability, normalizes serotonin levels and increases BDNF, and improves neuroplasticity processes. | Kang et al. [72], 2024 |
| Akkermansia muciniphila | Alzheimer’s disease and cognitive impairment in a murine model | 5 × 109 CFU in 200 μL for 6 months | Optimizes glucose and lipid metabolism, as well as the intestinal barrier. It reduces cortical Aβ, modulates inflammation and insulin resistance, favoring neuroprotection, and improving cognitive function. | Ou et al. [73], 2020 |
| Akkermansia muciniphila | Alzheimer’s disease in a murine model | 1 × 109 CFU for 7 months | Promotes the reduction of microglia activation and inflammation. They improve intestinal transit, memory, and anxiety. | Kunevičius et al. [74], 2025 |
MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; CFU: colony-forming unit; SCFAs: short-chain fatty acids; p-TrkB: phosphorylated tropomyosin receptor kinase B; Aβ: beta-amyloid; NLRP3: NOD-like receptor family pyrin domain containing 3; MECP2: methyl-CpG-binding protein 2.
Dysbiosis of the gut microbiota has currently been focused on as a relevant factor in the etiology and progression of various neurological pathologies, acting directly on the microbiota-gut-brain axis. Recently, evidence suggests that microbial alterations can occur in the early stages of the pathology, even before neuronal damage, neuroinflammation, pathological protein aggregation, and dysfunction of biological barriers [75].
In Alzheimer’s disease (AD), patients manifest an altered microbial composition characterized by an increase in Proteobacteria, Bifidobacterium, and Phascolarctobacterium and significantly reducing butyrate-producing bacteria belonging to Firmicutes, Clostridiaceae, and Lachnospiraceae. Consequently, these changes are associated with increased intestinal permeability, systemic and brain neuroinflammation, as well as accelerated deposition of β-amyloid peptides and phosphorylation of tau. In addition, studies of FMT from murine models with AD have been carried out, and the results have been impressive in showing the worsening of cognitive impairment in the recipient animals, further reinforcing dysbiosis as a causal role in disease progression [75, 76].
However, in Parkinson’s disease (PD), intestinal dysbiosis is considered an early risk factor and a modulator in the progression of the disease, in addition, patients who have been diagnosed with PD consistently present a decrease in SCFA-producing bacteria such as Roseburia, Faecalibacterium, Blautia, and Lachnospiraceae, and in turn, an increase in Akkermansia, Lactobacillus, and Verrucomicrobiaceae. These movements in the gut microbiota favor the loss of the integrity of both the intestinal and BBBs, the activation of neuroinflammatory responses, and the increase of circulating LPS; therefore, it has been determined that this phenomenon promotes misfolding, the aggregation of α-synuclein in the intestine, facilitating its spread to the CNS through the vagus nerve and exacerbating dopaminergic degeneration [77, 78].
Finally, another of the most common diseases at the neurological level is amyotrophic lateral sclerosis (ALS), where dysbiosis of the gut microbiota has been recognized as a relevant modulator of neuroinflammation and systemic metabolism. Patients who have been confirmed with a diagnosis of ALS have a reduction in SCFAs and an increase in potentially pathogenic genera. It has been studied that these alterations contribute to the deterioration of the intestinal barrier and the gut-liver axis, allowing access to a systemic translocation of bacterial endotoxins such as LPS, generating activation of microglia and astrocytes, promoting the release of pro-inflammatory cytokines. In addition, the decrease in SCFAs with neuroprotective properties, in conjunction with exposure to microbiome-derived neurotoxic metabolites, such as β-methylamino-L-alanine (BMAA) and bacterial amyloids, may exacerbate the misfolding of ALS-associated proteins, increasing oxidative stress and accelerating motor neuron degeneration [79].
According to the evidence, it is clear that alterations in the gut microbiota, including an increase in Bacteroidetes and Enterobacteriaceae and a reduction in Lachnospiraceae, are associated with cognitive impairment and greater clinical severity, contributing to neuroinflammatory and metabolic processes that could accelerate the progression of the disease [75, 77].
FMT is one of the emerging strategies that has gained great relevance. It is an infusion of intestinal microbiota that comes from a healthy donor and has a recipient with intestinal dysbiosis. The objective of this transplant is to restore a eubiotic microbiome. FMT can be administered by capsule, endoscopy, or nasoenteric. The central mechanism consists of the repopulation of the intestine with a functional bacterial population that can modulate the gut-brain axis, reducing LPS and pro-inflammatory cytokines. In this way, the restoration of neuroactive metabolites, especially SCFAs, is sought to improve the integrity of the intestinal and BBB [80].
Within these metabolites, acetate has been positioned as key in microglial maturation and the regulation of its energy metabolism, phagocytosis, and neuroinflammation. Studies in murine models showed that acetate can reverse immature microglial phenotypes and attenuate the neuroinflammation associated with dysbiosis. In this way, FMT is positioned not only as a gastrointestinal strategy but also as a neuromodulatory intervention capable of impacting microglial homeostasis [49, 74–80].
On the other hand, the standardized protocols that have to be established must guarantee safety, reproducibility, and scalability of the procedure. It is essential that donor screening be adjusted, which currently has demanding bases and does not have a uniform regulation by the FDA, limiting the entry of healthy donors [81]. Therefore, the supervision of these action plans must be carried out under the supervision of a specialist doctor and a stool bank coordinator, thus ensuring the quality control and traceability of the samples for at least 30 years. Likewise, from an operational approach, protocols must precisely define donor screening processes to minimize the transmission of bacterial, viral, parasitic, and multidrug-resistant pathogens. To do this, it is mandatory to integrate clinical evaluations with rigorous laboratory tests in both blood and feces. It is important to note that these evaluations should not be carried out only in initial periods; they should be repeated quarterly, keeping all products in quarantine until the negativity of the donor is officially confirmed at the end of the donation period. On the other hand, in terms of processing and logistics, standardization requires that the delivery of feces occurs within a maximum of 2 h after evacuation, with a sample processing of less than 6 h ideally, to preserve taxonomic viability. In addition, for the treatment to be effective and have clinical benefits, the use of at least 50 g of feces diluted in 0.9% NaCl with 10% glycerol as a cryoprotectant is recommended, allowing storage at –80°C for up to two years. Finally, procedures should be considered to improve clinical acceptance, prioritizing less invasive routes of administration such as oral capsules, which have been shown to have a level of efficacy comparable to colonoscopy, but with a superior safety profile and greater ease of use. The implementation of these procedures, along with mandatory reporting of data in national registries, are critical steps in transforming FMT from an emerging intervention to a regulated and accessible biological therapy [82–84].
FMT interventions emerge as promising strategies to modulate the gut-brain axis that is linked to microglial activation. However, the evidence in humans is still insufficient, or the results are often mixed. Research is required with homogeneous criteria in the route of administration, dose, donor/batch characterization, metabolite control, neuroinflammatory biomarkers, and other previously specified clinical outcomes.
Current evidence suggests that probiotics play a significant role in the adjunctive treatment of various neurological disorders through their interaction with the gut-brain axis. Furthermore, the ability of different strains to influence these diseases by regulating systemic inflammation, neuroinflammation, modulating the immune response, strengthening the intestinal barrier, and producing neuroactive metabolites has been studied in recent years [85].
This is why probiotics are considered one of the most interesting and scientifically supported emerging strategies. Several narrative and systematic reviews agree that probiotic supplementation can be associated with improvements in cognitive parameters and inflammatory markers linked to neurological disorders. Although clinical evidence is heterogeneous, the observed effects appear to be mediated by changes in cytokine profiles, reduction of oxidative stress, and modulation of microbial metabolites involved in neurotransmission and neuronal plasticity [86].
In summary, the advantages appear to be predominant, as systematic analyses of clinical trials have shown that probiotic administration has contributed to improved cognitive function, quality of life, and inflammatory biomarkers in various neurological contexts. However, the limitations and challenges facing the updating of this topic are that the results depend on factors such as the strain used, the dose, the duration of the intervention, bacterial overpopulation [small intestinal bacterial overgrowth (SIBO)], and the composition of the baseline microbiota, highlighting the need for more standardized clinical protocols. Treatment for neurological diseases must be interdisciplinary and include emerging therapies that contribute to the production of SCFAs, neurotransmitter regulation, and the interaction of neuroendocrine and immunological pathways. The most studied probiotic strains available in both animals and humans are compiled in Table 1.
Prebiotics are non-digestible substrates that promote the activity and growth of microorganisms in the gut, which can indirectly influence brain function by promoting the production of microbial metabolites such as SCFAs. Current evidence suggests that prebiotic supplementation improves certain cognitive aspects such as memory, attention, and neurological skills, as well as modulating the gut-brain neuroimmune response. However, the clinical effects are still varied and not as specific as those studied with probiotics, since factors such as dosage, duration, and the study population still need to be defined [87, 88].
On the other hand, postbiotics are microbial metabolites and cellular components released by the gut microbiota that can exert direct physiological effects without the need to administer microorganisms. Several recent reviews have described how these metabolites can cross the BBB via specific transporters and modulate immunological and metabolic processes in the CNS, promoting neuroprotective pathways involved in neurological diseases. Both prebiotics and postbiotics have clinical evidence demonstrating positive effects on neurological disorders by intervening in neuromodulation and the production of neuroactive metabolites. However, significant challenges remain, such as the lack of randomized clinical trials, variability in response between individuals and dosages, and identifying the most effective types of prebiotics that support their impact on neurological diseases with clinical evidence. All of the above underscores the need for larger controlled trials to solidify specific recommendations for clinical use [88].
The evidence collected confirms the central role of the gut microbiota as a regulator of brain health through associated immune, metabolic, and neuroendocrine pathways in the gut-brain axis. In addition, alterations in microbial composition and functions contribute to intestinal dysbiosis, favoring a critical pathophysiological state that promotes the process of neuroinflammation, BBB dysfunction, and microglial activation, thus increasing the risk of neurological pathologies. These challenges have had to be intervened through new strategies that allow finding treatment windows, with recent research that has shown positive effects in preclinical and clinical models, including Bifidobacterium breve, Lactobacillus rhamnosus GG, Lactiplantibacillus plantarum, Saccharomyces boulardii, and Akkermansia muciniphila, where cognitive improvements, modulation of pathological symptoms, and protection against neuroinflammation and amyloidogenesis are evidenced. Emerging interventions continue to advance and so far, FMT, prebiotics, probiotics, and postbiotics have shown promising and beneficial effects that can contribute to the treatment of neurological diseases. However, these advances still require more standardized protocols, a more robust understanding of the mechanisms, and acceptable clinical validation. In general, this review seeks to consider the gut microbiota as an etiological factor, biomarker, and therapeutic intervention, in order to favor the need to strengthen research efforts to reduce the growing global burden of neurological diseases.
AD: Alzheimer’s disease
ALS: amyotrophic lateral sclerosis
BBB: blood-brain barrier
CNS: central nervous system
FMT: fecal microbiota transplantation
GABA: gamma-aminobutyric acid
GPRs: G-protein-coupled receptors
HPA: hypothalamic-pituitary-adrenal
ILs: interleukins
LPS: lipopolysaccharides
NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells
PD: Parkinson’s disease
SCFAs: short-chain fatty acids
TLRs: Toll-like receptors
TNF-α: tumor necrosis factor alpha
FLM: Investigation, Writing—review & editing, Supervision, Formal analysis. IABC: Investigation, Writing—original draft, Writing—review & editing. Both authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 863
Download: 37
Times Cited: 0
Adnan Akhtar Shaikh ... Niveditha Nair
Salomón Páez-García ... Miguel Germán Borda
Zhengrui Li ... Jing Li
Diego Fernández-Lázaro ... Juan Mielgo-Ayuso
Ericka C. Loza López ... Felipe Esparza Salazar
Alejandro Borrego-Ruiz, Juan J. Borrego
Natalia Rojas-Sánchez ... Antonio Ibarra
