Affiliation:
Department of Translational Research, College of Osteopathic Medicine of the Pacific, Western University of Health Sciences, Pomona, CA 91766, USA
Affiliation:
Department of Translational Research, College of Osteopathic Medicine of the Pacific, Western University of Health Sciences, Pomona, CA 91766, USA
Affiliation:
Department of Translational Research, College of Osteopathic Medicine of the Pacific, Western University of Health Sciences, Pomona, CA 91766, USA
Affiliation:
Department of Translational Research, College of Osteopathic Medicine of the Pacific, Western University of Health Sciences, Pomona, CA 91766, USA
Affiliation:
Department of Translational Research, College of Osteopathic Medicine of the Pacific, Western University of Health Sciences, Pomona, CA 91766, USA
Email: vrai@westernu.edu
ORCID: https://orcid.org/0000-0001-6286-2341
Explor Immunol. 2026;6:1003246 DOI: https://doi.org/10.37349/ei.2026.1003246
Received: November 17, 2025 Accepted: March 10, 2026 Published: April 13, 2026
Academic Editor: Sofia Kossida, The International ImMunoGeneTics Information System, France
The article belongs to the special issue Advances in Cellular and Molecular Treatment of Autoimmune Diseases
Myasthenia gravis (MG) is an autoimmune disorder of the neuromuscular junction marked by fluctuating muscle weakness, driven by pathogenic antibodies targeting acetylcholine receptors, muscle-specific kinase, or lipoprotein receptor-related protein 4. Traditional treatments such as corticosteroids and immunosuppressants provide symptomatic relief but are limited by systemic toxicity and variable efficacy. Advances in immunology have led to targeted biologics that address specific disease mechanisms, such as B-cell depleting antibodies like rituximab and inebilizumab, complement inhibitors such as eculizumab, ravulizumab, and zilucoplan, and neonatal Fc receptor antagonists like efgartigimod and rozanolixizumab that lower pathogenic IgG levels. Emerging molecular and cellular therapies, including T-regulatory cell therapy, RNA-based modulation, and chimeric antigen receptor T-cell therapy approaches, seek to achieve durable immune tolerance rather than transient suppression. However, therapeutic response varies due to immunologic heterogeneity, long-term safety concerns, and limited global access. Future directions focus on precision medicine, biomarker-guided treatment, and AI-assisted disease monitoring, alongside next-generation therapeutics such as long-acting FcRn inhibitors (IMVT-1402), bispecific antibodies (gefurulimab), and oral small molecules (remibrutinib, iptacopan). Collectively, these developments mark a shift from generalized immunosuppression toward mechanism-based, patient-centered care aimed at restoring lasting immune balance and improving quality of life in MG. This narrative review discusses these aspects to comprehend the existing understanding and challenges.
Myasthenia gravis (MG) is a rare neurological disorder, but it is, in fact, the most common autoimmune disorder that affects the neuromuscular junction (NMJ) [1]. MG is classically thought of as an antibody-mediated autoimmune disease that commonly develops autoantibodies to acetylcholine receptor (AChR) and less commonly to targets such as muscle-specific kinase (MuSK) and low-density lipoprotein receptor-related protein 4 (LRP4) [1]. Patients with MG tend to present clinically with muscle fatigue and weakness, but can be further divided into two clinical subtypes based on muscle involvement. The first subtype is the ocular MG subtype, which is limited to the ocular and extraocular muscles, leading to symptoms such as drooping eyelids, double vision, and trouble keeping the eyes open [2, 3]. The second subtype is generalized MG (gMG), which includes limbs, respiratory, and bulbar muscles in addition to the muscles involved in the ocular subtype, creating symptoms such as difficulty speaking, swallowing, general weakness, standing from seated positions, and climbing stairs [2, 3] (Figure 1).
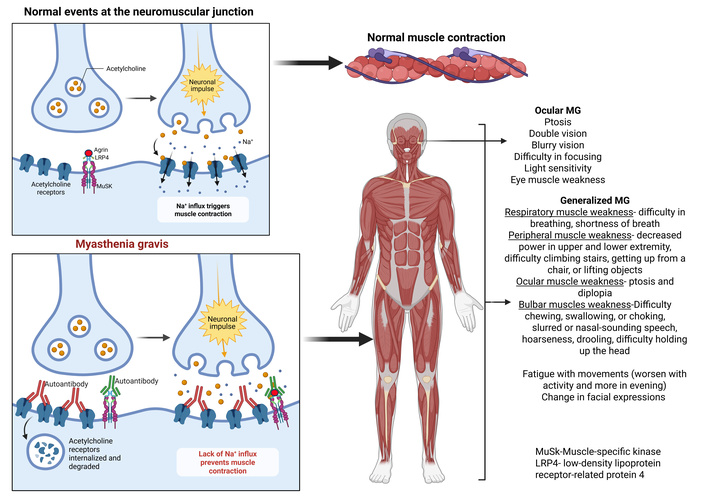
Clinical symptoms and pathophysiology of myasthenia gravis (MG). Normally, acetylcholine (ACh) attaches to its receptor (AChR) and causes muscle contraction. In MG, autoantibodies to AChR, MuSK, and LRP4 cause severe muscle weakness and fatigue by disrupting acetylcholine (ACh) signaling through complement-mediated destruction, cross-linking/degradation of receptors. Created in BioRender. Rai, V. (2026) https://BioRender.com/9h1mf65.
Regarding the prevalence of MG, most neurologists tend to see one MG patient every 3 to 4 years; however, studies have shown that the incidence of MG is rising and has doubled in the last 20 years [4]. This can be partly attributed to better diagnosis and treatment methods and the increasing longevity of the population, leading to more diagnosed cases since MG tends to be more prevalent in the elderly population [4]. Additionally, studies have shown that MG with an earlier onset (third decade of life) has a higher incidence rate in females, whereas MG in later stages of life (sixth decade and beyond) tends to be higher in males [3]. It is important to recognize the severity of MG because MG is becoming a growing concern.
Traditional medical treatment for MG includes acetylcholinesterase inhibitors, corticosteroids, and immunosuppressants. Acetylcholinesterase inhibitors are typically the first-line treatment given their ability to increase the amount of ACh at the NMJ synaptic cleft [5]. Generally, pyridostigmine is the preferred drug of choice, with ambenonium chloride as the typical second-line option. With pyridostigmine, it is important to carefully manage the dosage as doses that are too high may lead to increased muscle weakness [5]. Prednisone is the most common corticosteroid. Corticosteroids are used because they are widely available, are inexpensive, and have a fast action, but should be clinically monitored closely due to the possibility of serious side effects. The doses and duration should be carefully administered and monitored because of associated side effects with hyperglycemia, as the most common side effect. Corticosteroids are used early on post-diagnosis to control symptoms, but should be tapered down over time to prevent side effects with chronic usage, such as insomnia, mood changes, osteoporosis, and impaired glucose tolerance [6]. Lastly, immunosuppressants have also been used in the treatment of MG; however, these medications are less effective compared to the other options. Methotrexate is the most effective immunosuppressant compared to other immunosuppressants such as tacrolimus and mycophenolate mofetil. These medications are also associated with severe side effects such as hypertension, diabetes, osteoporosis, and obesity [7]. Often, a combination of these strategies is used to treat MG.
Thus, it is apparent that there is a need for better treatment methods, and immunotherapy seems promising. One of the needs for immunotherapy is due to the significant side effects associated with existing treatment methods. Additionally, 70–80% of patients experience some form of remission either from disease burden or from discontinuation of treatment, not intended to be used long-term [8]. Moreover, the need for immunotherapy can provide benefits in long-term stability of MG, better tolerance, and better long-term safety [9]. Through this narrative review, we aim to first describe the molecular markers at play when treating MG and then discuss the current treatment measures for MG and the challenges they pose. We will also demonstrate the therapeutic advances made over recent years and how they may be beneficial in overall treatment. Lastly, through this discussion, we want to illustrate the exciting potential for more personalized MG treatment with some of the more novel therapeutic approaches being developed.
A literature search was conducted using PubMed and Google Scholar to identify the background, mechanism, and treatment for MG. The keywords, alone or in combination, such as MG, pathophysiology, diagnosis, treatment, and clinical trial, were used to search articles. A total of 262 related articles were retrieved. The articles were reviewed, and duplicate articles, only abstracts, and articles not in the English language were excluded. A total of 77 articles were used for this review, with a focus on including articles describing the treatment strategies (Figure 2). Clinical trials were looked at https://clinicaltrials.gov/.
MG is an immune-mediated autoimmune disorder where autoantibodies attack cholinergic receptors in NMJs that prevent signal transmission (Figure 1). MG is generally classified in two ways: based on the age of onset, leading to early or late onset, or depending on the muscles affected. This review focuses primarily on the muscles affected by MG, which, to reiterate, can be ocular (oculomotor and extraocular muscles) or generalized (limb and respiratory muscles), resulting in generalized muscle weakness (Figure 1). However, a further subdivision can be based on the autoantibody that is formed and the receptors that are affected. This is important to be aware of as the autoantibodies change the pathogenicity of MG on the NMJ and can lead to upstream effects that are currently poorly understood [10]. The primary categories that MG gets classified as based on autoantibody status include: AChR (85%), MuSK (6%), and LRP4 (2%) [10]. One of the primary receptor dysfunctions is with the AChR found in 80–90% of MG patients [11]. Anti-AChR antibodies contribute to muscle weakness by blocking the AChR, forming cross-links with the AChR, or by accelerated degradation of the AChR [11] (Figure 1). This prevents signal transmission by preventing ACh from binding to its receptor. The pathogenicity of AChR-MG subtype specifically primarily comes from complement activation involving the IgG1 and IgG3 subclasses [12]. Complement activation can lead to the formation of the membrane attack complex (MAC), which eventually leads to the lysis of the post-synaptic membrane, disrupting the NMJ, specifically the post-junctional folds [12].
Another receptor-based subtype, MG, is associated with the involvement of MuSK receptor, a transmembrane receptor that interacts with LRP4, which eventually leads to the formation of high-density AChR clusters necessary for signal transmission [13]. In healthy individuals, the protein agrin activates the LRP4 receptor, which in turn activates MuSK, a receptor tyrosine kinase that clusters AChRs at the NMJ. In MG, the signaling pathway involving MuSK and LRP4 is disrupted, leading to the disease. In MuSK-positive MG, autoantibodies target MuSK, often blocking the interaction between LRP4 and MuSK, which impairs AChR clustering and causes muscle weakness (Figure 3).
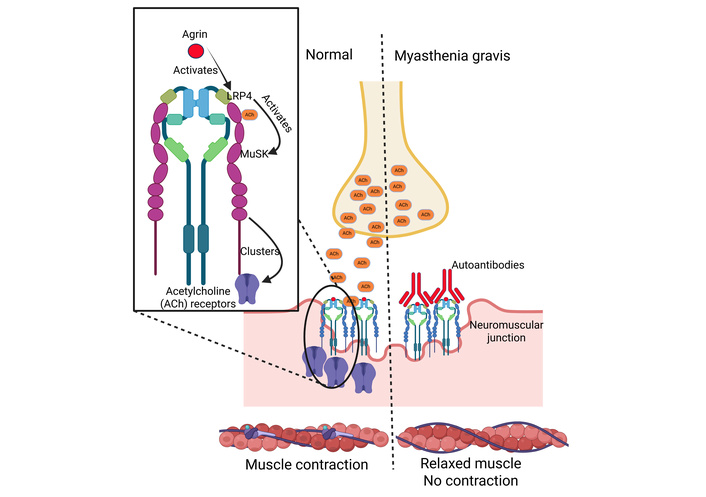
MuSK and LRP4 signaling in muscles and myasthenia gravis. Autoantibodies targeting MuSK often block the interaction between LRP4 and MuSK, which impairs AChR clustering and causes muscle weakness. MuSK and LRP4 signaling in muscles and myasthenia gravis. Created in BioRender. Rai, V. (2026) https://BioRender.com/qiddr7u. MuSK: muscle-specific kinase; LRP4: lipoprotein receptor-related protein 4.
A big difference in pathogenesis is that the antibodies present are part of the IgG4 class and do not activate the complement pathway. They act by preventing AChR clustering by interrupting the interaction between MuSK and LRP4, leading to diminished signal transduction [14]. The interplay between MuSK and LRP4 is evident in the formation of a complex necessary for the NMJ, and as such, it is important to also discuss the less common LRP4 dysfunction. LRP4 interacts with a glycoprotein called agrin prior to it activating MuSK. Mutations in LRP4 prevent the interaction with agrin, which creates the downstream effect of being unable to activate MuSK, ultimately preventing proper formation of the NMJ [14].
Going beyond the subtypes of MG based on the autoantibodies relating to receptor subtypes is the integral role of the thymus in T cell production. MG can be perpetuated by the thymus dysfunction with regard to central tolerance. Normally, the thymus is responsible for the elimination of autoreactive T cells. However, with dysfunction, MG patients lose this key aspect of central tolerance, leading to high levels of autoreactive T cells, which worsen the autoimmune response of MG [15]. About 80% of MG with AChR involvement had thymic involvement that contributed to autoimmunity [16]. Delving deeper, about 60% of patients with AChR-MG were found to have thymic hyperplasia, and most of these patients were females with a relatively early onset (before the age of 50). Thymic abnormalities were associated with higher levels of immature T cells being differentiated into CD4 helper T cells and CD8 cytotoxic T cells, which further perpetuates the autoimmune response present in MG [16]. Overall, it is apparent that there are numerous mechanisms by which MG can manifest, as well as the key role that the thymus plays in the development of the disease.
As discussed above, AChR, MuSK, and LRP4 are cellular and molecular targets in MG, which are primarily affected by autoantibodies produced by immune cells like B cells and plasma cells. T-cells become reactive to self-antigens like AChR and prime B cells to produce autoantibodies. Antigen-presenting cells present antigens like AChR or MuSK to naive CD4+ T cells, initiating the autoimmune response. AChR is the primary target in about 85% of MG cases. Autoantibodies bind to and destroy AChRs on the muscle membrane, preventing the neurotransmitter ACh from binding and triggering muscle contraction. In about 6% of patients, autoantibodies target MuSK, a protein critical for maintaining neuromuscular synapses. In approximately 2% of patients, autoantibodies attack LRP4, which is an important binding partner for MuSK. Autoantibodies may also target other proteins at the muscle endplate, though their pathogenicity can be uncertain [17, 18]. Further, autoantibodies binding to the AChR can trigger the complement system, a cascade of proteins that can further damage the NMJ.
By understanding the immunopathology of MG, one identifies the exploitable leverage points available for therapy. Pathogenic IgG1 and IgG3 in AChR-positive MG cause the activation of complement with resultant postsynaptic injury by the effect of MAC production [1]. By contrast, MuSK antibodies, the IgG4-type, are of a nature that does not cause fixation of complement, but by implication, interference with the agrin-LRP4-MuSK signalling axis, which is specific to the clustering of AChR [18]. The mechanistic details explain the differential responses to therapy (Figure 4). AChR-positive disease is susceptible to blockade techniques against complement, but MuSK-MG is often more responsive to approaches that are B-cell directed [19]. In AChR-positive MG, complement blockade is a directed answer. Eculizumab and ravulizumab both inhibit cleavage of C5, thus preventing formation of MAC and so preserving neuromuscular transmission but without a concomitant global attenuation of the immune response [19]. In MuSK-MG, in which antibody production is the axial impetus for the activity of the disease, the targeting of B cells becomes a rational consideration. Rituximab causes depletive phenomena of CD20+ B cells, sparing the long-lived plasma cells, with the result that autoantibody production is decreased in a partial but clinically significant manner [20]. Data shows improved response in the initiation of the treatment if started earlier in the evolutionary process of the disease [21]. Studies support the clinical benefit of rituximab in terms of functional responses, diminished consumption of steroidal treatment, and an overall better durability of response in MuSK-MG [22, 23]. However, with the requirement of more profound immunosuppression, CD19 therapies, e.g., inebilizumab, which acts not just on mature B cells but on plasmablasts and some plasma cells, allow broader B-cell/plasmablast depletion. The Phase 3 trials of inebilizumab in immunological gMG show clinical advantage, which is commensurate with a more thorough antibody production blockage [24] (Figure 4).
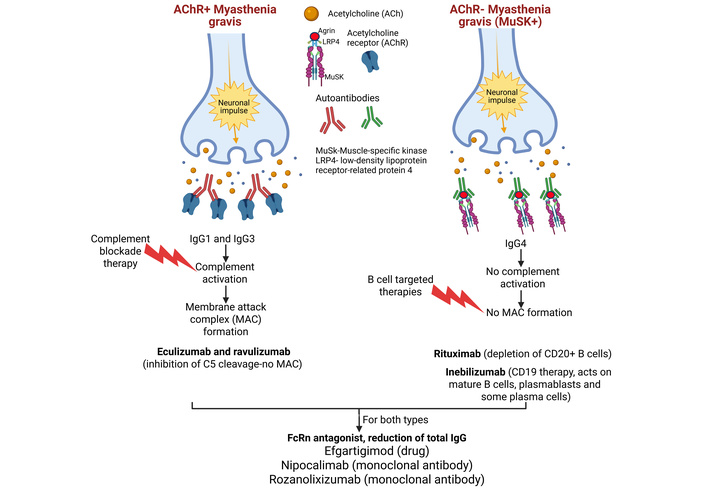
Therapeutic targets in AChR-positive and AChR-negative myasthenia gravis (MG). Therapeutic targets for AChR-positive (AChR+) and MuSK-positive (AChR–) MG include immunosuppressants and complement inhibitors for AChR+ MG, while AChR– MG is treated with other approaches. For AChR+ MG, targets include the complement system (e.g., using eculizumab), B-cell depletion, and IgG reduction. For AChR– MG, targets often focus on proteins like MuSK and LRP4, which are involved in AChR clustering, and potential therapeutic approaches include B-cell depletion. Therapeutic targets in AChR-positive and AChR-negative MG. Created in BioRender. Rai, V. (2026) https://BioRender.com/54m99ii.
Persistence of the antibody is not merely a matter of production but also of recycling. The neonatal Fc receptor (FcRn) holds IgG from degradation, increasing its half-life. Inhibition of FcRn enhances the catabolism of IgG and incidence of pathogenic load in the system, but without suppression of the pan-leucocyte pool [25, 26]. This allows an area of autoantibody therapeutic availability over the area of improvement in circulating autoantibodies, irrespective of the specificity of the clone. Efgartigimod, an FcRn antagonist, also obtains a reduction of total IgG in a dose-dependent manner, affecting MG-activities of daily living (MG-ADL) and quantitative MG (QMG) grading scores, which are improved [27]. Nipocalimab has obtained analogous results in improvement in functionality in Phase 3 trials [28]. Rozanolixizumab has shown its useful effect in both instances of AChR-positive and MuSK-positive, and provides evidence of the flexibility of FCRn blockade as a disease modification strategy [29] (Figure 4).
Cell-based assays (CBAs) for MG, particularly those using immunofluorescence (IF), are emerging as highly sensitive methods for detecting anti-AChR and anti-MuSK antibodies, often outperforming traditional radioimmunoassays (RIAs) by detecting low-affinity pathogenic antibodies. They offer superior detection of conformation-dependent, conformational antigens in their natural membrane state, making them ideal for diagnosing seronegative MG (SNMG). The current and routinely used assays, enzyme-linked immunosorbent assay (ELISA), radioimmunoprecipitation assay (RIA), and indirect IF test (IIFT), have limited sensitivity. This might be one of the explanations for the lack of serological antibody detection in SNMG patients. Contrary to this, fixed CBA (F-CBA) and live CBA (L-CBA) have a higher sensitivity compared to routinely used assays. The reported sensitivity for RIPA, ELISA, and F‐CBA was 82.8%, 81.0%, and 70.7%, respectively. F‐CBA exhibited the highest specificity overall (95.0%). The sensitivity and specificity for detecting AChR of F‐CBA were 73.6% and 95.0%; ELISA were 81.1% and 85.0%, and RIPA were 81.1% and 95.0%, respectively [30–33]. CBAs have various advantages over routine assays. CBAs can identify antibodies in 60–70% of patients who are “double seronegative” (negative for both AChR and MuSK) via traditional tests. Because antigens are expressed in their native membrane environment, they are more likely to detect pathogenic antibodies that rely on the structural conformation of the AChR complex, particularly when clustered by proteins like rapsyn. Unlike RIA, CBA does not require radioactive reagents and can be automated, making it suitable for routine lab use [30, 34]. There are limitations associated with CBAs. L-CBAs require specialized cell culture facilities, making them difficult to implement outside of research centers. While F-CBA is more convenient, it may be less sensitive than L-CBA in some cases, and its sensitivity can be influenced by sample dilution. IF, especially with L-CBA, requires skilled personnel for accurate visual interpretation. As a newer technology, there is a lack of fully standardized, universal protocols compared to older methods [30, 31, 34–36]. While F-CBA is becoming a, if not the, new gold standard, it is sometimes recommended to reserve L-CBA for patients with strongly suspected MG who test negative on F-CBA and RIA.
Recent advances in molecular immunology and biotechnology have transformed the therapeutic landscape of gMG. Traditional broad immunosuppressive approaches are being replaced by targeted monoclonal antibodies (mAbs), FcRn inhibitors, and emerging cell- and gene-based strategies that directly address the underlying autoimmune mechanisms [19].
Therapies using mAbs have redefined the management of MG by selectively targeting immune components implicated in disease pathogenesis [37]. Rituximab is a mAb that depletes CD20+ B cells responsible for producing pathogenic autoantibodies that were initially developed for B-cell lymphomas [20]. However, some meta-analysis studies have identified the benefits of rituximab in MG, particularly MuSK-MG [22, 23]. Yang et al. [22] reported improved clinical status in 91% of MuSK-MG patients following low-dose rituximab treatment, with significant reductions in QMG scores, measured before and after treatment. Additionally, a cohort study found initiation of rituximab within 12 months of onset was associated with shorter time to remission (7 months vs. 16 months) compared to later use [21]. But the authors acknowledged that a placebo-controlled randomized clinical trial is warranted to corroborate their findings. An emerging mAb for treatment of MG includes inebilizumab, which is an anti-CD19 mAb that is being investigated for its broader B-cell/plasmablast depletion potential [38]. In Phase 3, a double-blind, randomized, placebo-controlled trial, inebilizumab had significant reductions in QMG scores and was found to improve function and reduce disease severity in gMG [24]. mAb therapies targeting B-cell lineages represent a major advancement in the precision management of MG, with accumulating evidence supporting their efficacy and early use in disease modulation.
FcRn inhibitors represent a mechanistically novel therapeutic class that modulates IgG recycling [25]. By blocking FcRn-mediated IgG rescue from lysosomal degradation, these agents lower circulating pathogenic IgG autoantibody titers without broadly suppressing immune cells [26]. Efgartigimod, an FcRn inhibitor, has demonstrated strong clinical efficacy in patients with AChR-positive MG. A meta-analysis reported a treatment response rate of approximately 78%, highlighting its substantial therapeutic benefit in this population [27]. Similarly, nipocalimab, another FcRn inhibitor, has been shown to significantly improve both QMG and MG-ADL scores [39]. When used alongside standard-of-care therapies, it has exhibited a favorable safety profile and provided sustained disease control over a 6-month treatment period [28]. Rozanolixizumab, an anti-FcRn antibody approved in 2023, has demonstrated clinically significant improvements in patients with AChR- or MuSK-positive MG. It exerts its therapeutic effect by promoting IgG degradation through FcRn blockade [29]. Overall, the clinical evidence supports FcRn inhibition as a targeted and well-tolerated strategy that effectively mitigates pathogenic autoantibody activity in gMG.
Cell-based immunomodulation represents a rapidly advancing frontier in the treatment of MG, offering the potential to restore long-term immune tolerance rather than merely suppress disease activity [19]. These approaches stem from the recognition that MG is driven by a loss of self-tolerance within both the B-cell and T-cell compartments, leading to the production of pathogenic autoantibodies targeting AChR and MuSK [40]. By directly manipulating or reprogramming immune cell populations, cell-based therapies aim to re-establish immune homeostasis. Hematopoietic stem cell transplantation (HSCT) is performed to reconstitute the immune system, which can induce sustained remission in severe, treatment-refractory MG [41]. Clinical use of HSCT is expanding with durable remission and reduced autoantibody titer in select MG patients, which suggests the efficacy of this treatment [41]. However, significant risks of infection, organ toxicity, and treatment-related mortality currently confine HSCT to highly refractory cases under strict clinical protocols [42]. T-regulatory (Treg) cell therapy has emerged as a more targeted and potentially safer approach to re-establish immune tolerance [43]. In MG, defects in Treg number and function contribute to unchecked autoreactive B- and T-cell activity. Ex vivo expansion of autologous Tregs followed by reinfusion has shown early signs of immunologic benefit in preclinical MG through attenuating disease severity by suppressing pathogenic T- and B-cell responses [44]. Furthermore, it has been found that ex vivo administration of Tregs with anti-inflammatory markers such as CD25, CTLA-4, and Foxp3 can suppress in vitro AChR-induced proliferation of T cells [45]. In addition to cell-based approaches, strategies that shift the Th17/Treg balance toward regulation are under investigation, as thymic and peripheral immune imbalances contribute to disease severity [46]. This allows for a more precise immunomodulatory strategy rather than global immunosuppression. These findings highlight the potential of cell-based immunomodulatory interventions to achieve long-term remission in MG.
Building on advances in cancer immunotherapy, chimeric antigen receptor T-cell (CAR-T) therapy is also being explored as a transformative option for autoimmune neuromuscular diseases. CAR-T constructs can be engineered to selectively target and eliminate autoreactive B cells or plasma cells responsible for producing MG-associated autoantibodies [47]. For instance, Descartes-08, an mRNA-based anti-B-cell CAR-T product in Phase 3 of clinical trials, has demonstrated clinically meaningful decreases in MG-ADL and QMG scores at up to 9 months [48]. KYV-101 (Phase 1) builds upon this as a next-generation CD19-directed CAR-T therapy that acts through deep B-cell depletion and immune reset with a single infusion in patients [49]. However, there have been some concerns regarding long-term immunosuppression and the potential to induce cytokine release syndrome [10]. To overcome these challenges, newer strategies, including the chimeric autoantibody receptor T-cell (CAAR-T) therapy, have been developed [50]. Unlike conventional CAR-T therapies that broadly deplete the entire B-cell population, CAAR-T therapy selectively removes only the autoreactive B-cell clones involved in disease pathogenesis, thereby preserving normal immune function [10]. These therapies have the potential to induce deep, durable remission with a single intervention, while new strategies have addressed complications including cytokine release syndrome, neurotoxicity, and long-term immune reconstitution [49].
Clinical research on cell therapies for MG has advanced rapidly, with early-stage trials showing significant, durable improvements in symptoms. mRNA CAR-T therapy (Descartes-08), a Phase 2b trial for gMG, demonstrated that 57% of participants achieved minimal symptom expression by 6 months, with effects lasting up to 12 months. This therapy is highlighted as a potential first-line, outpatient, long-lasting, and precise immunotherapy [48]. KYV-101 (CAR-T agent), an interim Phase 2 KYSA-6 data indicated that 100% of patients experienced rapid, robust improvements in MG-ADL and QMG scores, with 100% of patients free of nonsteroidal immunosuppressants, high-dose steroids, and FcRn/complement inhibitors (reported by NeurologyLive), potentially offering a “one and done” approach (NCT06193889). Beyond traditional T-cell suppression, research is exploring organ-specific CAR Treg cells and mRNA-based approaches that offer more control, reducing risks compared to traditional CAR-T therapy. Early trials have shown that these cell-based approaches can produce long-term remission in various autoimmune diseases, reducing the need for continuous medication.
Advances in gene editing and RNA-based modulation offer the possibility for precisely correcting or silencing the molecular drivers of MG [51]. Technologies such as CRISPR/Cas9 are under investigation to re-engineer immune tolerance by eliminating autoreactive B-cell or plasma cell clones, restoring thymic tolerance, or reprogramming immune progenitors toward self-tolerance [52]. Currently, most therapies remain in a preclinical phase due to challenges that include delivery specificity, off-target risk, and long-term safety [53]. In contrast, therapies involving RNA interference (RNAi) enable silencing of mRNA encoding complement components, cytokines, or enzymes implicated in disease activity [54]. These RNA-targeting therapeutics include small interfering RNAs, miRNA modulators, and antisense oligonucleotides that induce degradation of mRNA without altering genomic DNA [55]. In the pathogenesis of MG, long non-coding RNA (LncRNA)/miRNA/mRNA axes, such as SNHG16/let-7c-5p/IL-10 and MALAT-1/miR-338-3p/MSL2, affect nuclear factor kappa beta (NF-κB) and tumor necrosis factor (TNF) signaling pathways, and regulation of Treg cell differentiation [56]. The ability to specifically target these pathways underscores the future potential of RNA-based platforms to regulate key immune pathways implicated in MG pathogenesis. Continued progress in vector design, high-fidelity editing, and RNA stabilization is likely to move gene editing and RNA-based modulation toward clinical translation.
Complement activation plays a central role in the pathogenesis of AChR-positive MG, driving immune-mediated damage through MAC formation [57]. Therapeutic inhibition of complement component C5 has therefore emerged as an effective strategy to interrupt this downstream effector pathway. Eculizumab is a humanized mAb that blocks cleavage of C5 and was the first targeted biologic approved for AChR-positive MG [58]. In a Phase 3, randomized, double-blind, placebo-controlled, multicentre study (REGAIN trial), eculizumab was found to improve the MG-ADL and QMG scores, exacerbation frequency, and corticosteroid burden, while also proving to be safe long-term [59]. Ravulizumab was built on these results as a next-generation C5 inhibitor that was engineered for extended half-life via targeted amino acid substitutions [60]. Ravulizumab demonstrated sustained efficacy and durability in trials where 88.1% of patients achieved a clinically meaningful improvement in MG-ADL scores [60]. Most recently, zilucoplan was approved to treat AChR-positive MG as a synthetic macrocyclic peptide C5 inhibitor [61]. Its favorable efficacy and tolerability balance are present as an option to treat a comprehensive patient population. Together, these complement inhibitors have redefined the therapeutic landscape of AChR-positive MG by offering targeted, mechanism-based control of disease activity, improving functional outcomes, and reducing treatment burden for patients with refractory or moderate-to-severe disease.
Cytokine-targeted therapies are emerging as a promising strategy in MG, aiming to modulate the upstream immune pathways that initiate autoimmunity rather than addressing downstream antibody effects. Elevated levels of pro-inflammatory cytokines, such as IL-6, IL-17, IL-21, IFN-γ, and TNF-α, contribute to disease activity by promoting germinal center formation, activating T-helper (Th17 and Tfh) cells, and stimulating B-cell-mediated autoantibody production [62]. IL-6 plays a central role by driving differentiation of naive CD4+ T cells into Th17 cells while inhibiting Treg development. In experimental autoimmune MG (EAMG) models, IL-6 blockades have been shown to reduce disease severity, lower anti-AChR antibody titers and B-cell counts, and down-regulate Th17-associated genes such as IL-17, IL-21, and IL-23R [63, 64]. Anti-IL-6 mAb drugs, such as tocilizumab or satralizumab, have been associated with improvements in muscle strength, reduced steroid requirements, and stabilization of disease activity, particularly in patients with concomitant inflammatory MG disease [65].
IL-17 and IL-22 further exacerbate symptoms by promoting inflammation and B-cell differentiation, whereas IL-21 amplifies B-cell activation through CD40–CD40L signaling [64]. These insights suggest that targeting cytokine networks, particularly IL-6, IL-17, and IL-21, may disrupt the inflammatory T-cell–B-cell interaction that sustains the autoimmune cascade. Although clinical experience with anti-IL-17 mAbs (such as secukinumab or ixekizumab) in MG remains limited, emerging data suggest that IL-17 blockade may be particularly effective in Th17-driven disease phenotypes and could synergize with IL-6 inhibition by more fully restoring immune tolerance [66]. By restoring immune balance and limiting autoreactive signaling, cytokine-based therapies may complement existing biologic and cellular treatments, paving the way for more precise and durable disease control.
Various active or recruiting clinical trials for MG are currently investigating novel therapies, including mAbs (inebilizumab, tocilizumab, satralizumab), complement system inhibitors, FcRn inhibitors, and CAR-T cell therapies (Table 1). Key trials include Phase 3 evaluations of inebilizumab (MINT study) and batoclimab, along with head-to-head comparisons of nipocalimab and efgartigimod.
Ongoing clinical trial to improve the treatment of myasthenia gravis.
| Trial ID | Phase/Status | Aim of the trial |
|---|---|---|
| NCT06463587MyClad | Phase 3, randomized, double-blind, placebo-controlled clinical studyRecruiting | To investigate the sustained efficacy, the need for retreatment, and the long-term safety of a new formulation of oral cladribine as compared with placebo in participants with gMG. |
| NCT06626919 | Phase 1 studyRecruiting | To evaluate the safety, tolerability, and preliminary efficacy of anitocabtagene autoleucel, a BCMA-directed CAR-T therapy, in subjects with gMG. |
| NCT06799247AURORA | Phase 3, randomized, double blind, placebo-controlled study.Recruiting | To evaluate the efficacy, safety, and tolerability of Descartes-08, mRNA CAR-T therapy, in patients with gMG. |
| NCT06359041RESET-MG | Phase 1/2, open-label, multicenter studyActive, not recruiting | To evaluate the effects of CABA-201, a CAR-T therapy, in patients with gMG. |
| NCT06704269 | A Phase I/II studyRecruiting | To assess the safety, efficacy, and cellular kinetics of YTB323, a biological CAR-T therapy, in participants with treatment-resistant gMG. |
| NCT06456580UPSTREAM MG | A randomized, double-blind, placebo-controlled Phase 3 studyRecruiting | To evaluate the efficacy and safety of telitacicept in the treatment of gMG. |
| NCT06193889KYSA-6 | A Phase 2/3, open-label, randomized, controlled, multicenter studyRecruiting | To investigate the effects of KYV-101, an autologous fully human anti-CD19 CAR-T therapy, vs. ongoing standard-of-care immunosuppressive therapy in patients with gMG. |
| NCT06414954SYNAPSE-MG | Phase 2 proof-of-concept, dose range finding studyRecruiting | To evaluate the safety and efficacy of 3 dose levels of NMD670 vs. placebo in adult patients with MG with antibodies against AChR or MuSK. |
| NCT06558279ADAPT oculus | Randomized, double-blinded, placebo-controlled, Phase 3, parallel-group design studyActive, not recruiting | To evaluate the efficacy and safety of efgartigimod PH20 SC in adult patients with ocular MG. |
| NCT06106672 | A Phase 1b/2a First-in-Human (FIH) clinical trialRecruiting | To assess the safety, tolerability, pharmacodynamics, and efficacy of multiple ascending doses of CNP-106 in MG patients. |
| NCT06055959ziMyG | Phase 2/3, open-label studyRecruiting | To assess the pharmacokinetics, pharmacodynamics, safety, tolerability, immunogenicity, and activity of zilucoplan in pediatric study participants with gMG. |
| NCT06517758 | A randomized, double-blind, placebo-controlled, multicenter, Phase 3 studyRecruiting | To evaluate efficacy, safety, and tolerability of iptacopan in patients with AChR+ gMG who are on stable standard of care (SOC) treatment with symptom management and immunosuppression in participants aged 18 to 75 years with gMG. |
| NCT04833894 | Phase 2/3, open-label, uncontrolled trialRecruiting | To investigate the pharmacokinetics, pharmacodynamics, safety, and activity of efgartigimod IV in children and adolescents aged from 2 to less than 18 years of age with gMG. |
| NCT06607627 | An open-label, single-arm, Phase 3 studyRecruiting | To assess the pharmacokinetics and pharmacodynamics of gefurulimab in pediatric participants ages 6 to less than 18 with AChR+ gMG. |
| NCT05265273 | An open-label, uncontrolled multicenter Phase 2/3 studyRecruiting | To evaluate the safety and efficacy of an investigational medication, nipocalimab, for the treatment of gMG in children aged 2 to less than 18 years old. |
| NCT05644561 | A Phase 3, open-label, single-arm, multicenter studyActive, not recruiting | To characterize the pharmacokinetics and pharmacodynamics of treatment with ravulizumab intravenous infusion in pediatric participants with gMG. |
| NCT06392386 | A Phase 2/3, open-label, uncontrolled studyRecruiting | To measure the pharmacokinetics, pharmacodynamics, safety, tolerability, and immunogenicity of efgartigimod PH20 SC in pediatric participants with gMG aged 2 to < 18 years. |
| NCT04524273 | A Phase 3, multi-center, randomized, quadruple-blind, placebo-controlled studyActive, not recruiting | To assess the efficacy and safety of batoclimab as induction and maintenance therapy in adult participants with gMG. |
| NCT06436742 | Phase 1b studyRecruiting | To assess the safety and tolerability of ARGX-119 in adult participants with DOK7-congenital myasthenic syndromes. |
| NCT04951622 | Phase 3, multicenter, randomized, double-blind, placebo-controlled studyRecruiting | To evaluate the efficacy and safety of nipocalimab compared to placebo in participants with gMG. |
| NCT05403541 | A Phase 3, multi-center, randomized, quadruple-blind, placebo-controlled studyActive, not recruiting | To assess the efficacy and safety of batoclimab as induction and maintenance therapy in adult participants with gMG. |
| NCT05070858 | Phase 3Recruiting | To test how safe the pozelimab and cemdisiran combination therapy and cemdisiran alone are and how well they work in adult patients with gMG. |
| NCT06298552 | A randomized, double-blinded, placebo-controlled, Phase 3, parallel-group design studyActive, not recruiting | To measure the efficacy and safety of efgartigimod compared to placebo in participants with AChR binding antibody (AChR-Ab) seronegative gMG. |
gMG: generalized myasthenia gravis; BCMA: B-cell maturation antigen; CAR-T: chimeric antigen receptor T-cell; AChR: acetylcholine receptor; MuSK: muscle-specific kinase.
MG exhibits marked immunologic and clinical heterogeneity driven by variability in autoantibody specificity. Approximately 80% of patients produce antibodies against the AChR, 4–8% against MuSK, and a smaller proportion against low-density LRP4, while the remainder are classified as SNMG [18, 67]. Each subtype demonstrates distinct pathogenic mechanisms and therapeutic responses. AChR antibodies, primarily IgG1 and IgG3, induce complement-mediated destruction of the postsynaptic membrane, whereas MuSK antibodies, largely IgG4, interfere with NMJ formation through inhibition of the agrin–LRP4–MuSK pathway without complement activation [67]. These immunologic differences manifest clinically—MuSK-MG patients often experience more severe bulbar and respiratory weakness, respond poorly to acetylcholinesterase inhibitors, and show greater benefit from B-cell depletion therapies such as rituximab, while thymectomy is less effective [5, 67]. Conversely, seronegative patients lack identifiable antibodies yet display heterogeneous clinical features, suggesting unrecognized pathogenic targets or non-antibody-mediated mechanisms [18, 67]. This immunologic diversity underlies patient-to-patient variability in disease severity and therapeutic response, posing a major challenge to developing standardized molecular and cellular interventions.
Long-term immunotherapy in MG is effective but associated with treatment-specific systemic risks that vary by therapeutic class. Over two years, 60% of patients had achieved the treatment goal of minimal manifestation (MM) status with an oral prednisolone dose of ≤ 5 mg/day (5 mg MM), while more frequent plasmapheresis and higher doses of prednisolone within 3 months were linked to difficulty achieving this status [68]. Long-term conventional immunosuppressants may be associated with the risk of intolerance, delayed onset of action, and systemic toxicity [69]. Targeted biologic therapies demonstrate improved disease control but introduce distinct safety considerations. Complement inhibitors such as eculizumab, ravulizumab, and zilucoplan represent promising options for refractory MG; however, they are associated with an increased risk of infection by encapsulated bacteria, particularly Neisseria meningitidis, necessitating vaccination and long-term infection monitoring [69]. B-cell depleting therapies, including rituximab and inebilizumab, carry risks of serious infections, hypogammaglobulinemia, herpes zoster reactivation, and rare cases of progressive multifocal leukoencephalopathy (PML) due to sustained B-cell suppression [69]. FcRn antagonists, while not causing broad immune cell depletion, reduce total IgG levels and may increase susceptibility to infections, although long-term immune surveillance data remain limited [69]. Cellular therapies, such as CAR-T therapy and HSCT, introduce additional safety concerns related to immune reconstitution. Reported adverse effects include cytokine release syndrome, neurotoxicity, prolonged immunosuppression, and infection risk, restricting their use to highly refractory cases within specialized centers [10, 42]. These findings emphasize that while immunotherapy is central to MG management, its prolonged use may lead to systemic toxicity and infection risk, requiring careful long-term monitoring [68, 69].
Although several new molecular therapies have demonstrated clinical benefit, access to these treatments remains limited in real-world practice. Belimumab, a mAb against B lymphocyte stimulator, and iscalimab, which targets the CD40 antigen, for example, have failed to demonstrate therapeutic efficacy in MG. These gaps reflect the incomplete translation of experimental success into broad clinical availability. Additionally, access to such treatment is further constrained by cost barriers as well as variations in health-care systems among different countries, which can impact people with MG globally [40]. This may leave many patients dependent on older, less targeted immunosuppressive therapies.
The future of MG treatment is moving toward more precise, durable, and patient-centered approaches. Instead of relying on broad immunosuppression, upcoming strategies aim to tailor therapy to the unique immune and genetic profile of each patient. Precision medicine strategies, such as molecular profiling, immune monitoring, and biomarker discovery, guide individualized treatment plans [70]. Also, the growing use of AI to analyze multi-omics and clinical data may soon help predict disease flares, optimize dosing intervals, and identify which patients are most likely to benefit from specific biologic or cellular therapies [71].
Among biologic agents, the next generation is focused on greater efficacy, longer duration, and easier administration. New FcRn inhibitors, such as IMVT-1402, are being designed to achieve deeper reductions in pathogenic IgG while requiring less frequent dosing and causing fewer injection-site reactions compared to earlier agents like efgartigimod and rozanolixizumab [72]. Similarly, bispecific antibodies such as gefurulimab are being engineered to bind both FcRn and albumin, extending their half-life and maintaining stable therapeutic effects with less frequent dosing [73]. These advances demonstrate a shift toward biologics that combine high efficacy with improved patient convenience.
At the same time, oral small-molecule therapies are emerging as promising alternatives to injectable treatments. Agents like remibrutinib, a selective Bruton’s tyrosine kinase (BTK) inhibitor, and iptacopan, a factor B inhibitor of the complement pathway, are in clinical development for MG and other autoimmune conditions [74, 75]. These oral drugs may offer comparable disease control with the added benefits of simpler administration, reduced treatment burden, and potentially lower cost, making them attractive options for long-term management. There have also been advances in B-cell targeted therapies. While rituximab remains the most widely used B-cell depleting agent, newer antibodies are expanding beyond CD20 to target broader or more specific populations of B cells and plasma cells [76]. Inebilizumab exemplifies this trend by targeting cells across the B-cell maturation spectrum. These next-generation therapies may produce more durable remissions and reduce the need for repeated dosing cycles [24].
Further development in the future of MG treatment involves ncRNAs. Although their role is not completely understood, there seems to be a connection between ncRNAs and mRNA expressed in MG patients, giving reason to believe that it is involved in the pathological disease process of MG [70]. Furthermore, it was found that ncRNA levels were increased in the serum, as well as having alterations in the thymus and peripheral blood cells in MG patients [70]. miRNAs have also been found to play a role in the development of MG because miRNAs can stimulate pro-inflammatory cytokine production, which contributes to the immune response present in MG [77]. As mentioned previously, the mechanism remains unclear; however, genes for miRNAs have been seen to be increased in diseased states, prompting additional investigation into this as a potential target for treatment [77]. Both ncRNAs and miRNAs, given the evidence found for their potential correlation with MG, deserve further investigation as research on MG treatment advances.
Together, these developments point toward a new era in MG treatment: one defined by integration and personalization. As biologics, small molecules, and cell-based therapies continue to evolve, combining them with AI-guided precision tools could help clinicians create personalized treatment plans for each patient. The goal of future therapies is not limited to only symptom control, but also durable immune balance and improved quality of life for people with MG.
Significant progress has been made in the treatment of MG through the development of targeted molecular and cellular therapies that move beyond nonspecific immunosuppression. Advances in complement inhibition, B-cell depletion, FcRn blockade, and emerging cell-based strategies have expanded therapeutic options and improved disease control for many patients. The central challenge moving forward lies in addressing the marked immunologic heterogeneity of MG and translating mechanistic insights into durable, individualized treatment strategies. Precision medicine approaches that integrate autoantibody profiling, immune biomarkers, and clinical phenotyping will be critical to optimizing therapy selection and minimizing unnecessary immunosuppression. At the same time, therapies aimed at reconstructing immune tolerance, rather than transiently suppressing pathogenic pathways, represent a key scientific priority for achieving sustained remission. Future research must focus on defining long-term safety profiles, identifying predictors of therapeutic response, and improving global access to advanced treatments. Continued integration of molecular immunology, cellular engineering, and data-driven clinical decision tools has the potential to fundamentally reshape MG management toward durable immune balance and improved quality of life.
AChR: acetylcholine receptor
CAAR-T: chimeric autoantibody receptor T-cell
CAR-T: chimeric antigen receptor T-cell
CBAs: cell-based assays
F-CBA: fixed cell-based assay
FcRn: neonatal Fc receptor
gMG: generalized myasthenia gravis
HSCT: hematopoietic stem cell transplantation
IF: immunofluorescence
L-CBA: live cell-based assay
LncRNA: long non-coding RNA
LRP4: lipoprotein receptor-related protein 4
MAbs: monoclonal antibodies
MAC: membrane attack complex
MG: myasthenia gravis
MG-ADL: myasthenia gravis-activities of daily living
MM: minimal manifestation
MuSK: muscle-specific kinase
NMJ: neuromuscular junction
QMG: quantitative myasthenia gravis
RIAs: radioimmunoassays
SNMG: seronegative myasthenia gravis
TNF: tumor necrosis factor
Treg: T-regulatory
SP, SN: Conceptualization, Writing—original draft. HM, TS: Writing—original draft. VR: Conceptualization, Writing—review & editing, Supervision. All authors reviewed and approved the final version of the manuscript.
The authors declare no conflict of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
The authors received no specific funding for this study.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1295
Download: 24
Times Cited: 0
Cheng Bao ... Hong-Wei Chen
María Fernanda Segovia ... Gerson D. Keppeke
