Affiliation:
1Department of Physiology and Allied Sciences, Amity Institute of Health Allied Sciences, Amity University, Noida 201301, Uttar Pradesh, India
Email: stripathy@amity.edu
ORCID: https://orcid.org/0000-0003-0549-2086
Affiliation:
2African Medicines Innovations and Technology Development Platform, Department of Pharmacology, School of Clinical Medicines, Faculty of Health Sciences, University of the Free State, Bloemfontein 9300, South Africa
ORCID: https://orcid.org/0000-0003-2921-8882
Explor Immunol. 2026;6:1003243 DOI: https://doi.org/10.37349/ei.2026.1003243
Received: October 17, 2025 Accepted: February 11, 2026 Published: April 09, 2026
Academic Editor: Calogero Caruso, University of Palermo, Italy
Genistein, a soy-derived isoflavone, has attracted considerable interest for its anti-inflammatory and anticancer properties. Chronic inflammation is a major driver of tumour initiation, progression, metastasis, and therapeutic resistance, creating a need for multi-target agents capable of modulating both inflammatory and oncogenic pathways. This review critically evaluates the molecular mechanisms, pharmacological basis, and translational prospects of genistein in inflammation-related cancers, incorporating recent advances in omics technologies and nanotechnology-based delivery systems. A comprehensive survey of the literature from major scientific databases was conducted, and evidence was assessed based on mechanistic depth, strength of data, and relevance to clinical translation. Genistein modulates multiple interconnected signalling pathways, including NF-κB, MAPK, PI3K/Akt, JAK/STAT, and apoptotic cascades, leading to reduced pro-inflammatory cytokine production, oxidative stress, and tumour cell proliferation. Epigenetic regulation, growth-factor pathway inhibition, and non-coding RNA modulation further contribute to its pleiotropic effects. Nanotechnology-based delivery platforms are being explored to improve their poor oral bioavailability and therapeutic performance in preclinical models. However, clinical translation remains limited by low bioavailability, context-dependent estrogenic activity, inter-individual metabolic variability, and the lack of large-scale, well-controlled clinical trials, standardized dosing, and validated biomarkers. Genistein represents a promising multi-target lead compound at the intersection of inflammation, immunology, and oncology, with future progress dependent on integrating multi-omics data and advanced delivery technologies for personalised clinical applications.
Isoflavones represent a group of phytochemicals, derived and found naturally in plants, composed chiefly of legumes, especially soybeans. Due to their similarity to 17β-estradiol, they can act as phytoestrogens within mammalian physiology. Among these phytoestrogens, the most studied and biologically active molecule is genistein [5,7-dihydroxy-3-(4-hydroxyphenyl)-4H-1-benzopyran-4-one]. These phytochemicals have received popular attention because of their diverse biological activities and are predominantly present in soybeans (Glycine max) and soy products. The two forms present within the soy diet are the glycosylated and aglycone forms, referred to as genistin and genistein, respectively, and it is the aglycone form that is biologically active [1].
Production of soybeans has experienced a significant boost globally. Regions such as Latin America, specifically Brazil and Argentina, have emerged as quite viable players in the production and subsequent exportation of soybean products. Such widespread production has assisted in enhancing the global food chain and the availability of genistein-enriched foods [2]. Countries where soy is traditionally consumed in large amounts, specifically in Asia (Japan, China, Korea), have shown a lower incidence of hormone-related cancers, like breast and prostate cancer, as well as lower incidences of chronic metabolic diseases like cardiovascular diseases and type 2 diabetes [3–5], which have attracted researchers to focus on genistein, one of the active compounds of soy, as having a pivotal role in producing these desirable results [6]. Available data suggest that only up to 10% of the Asian population takes in daily amounts of soy protein up to 25 g or isoflavones up to 100 mg. The significance of these data for the formulation of soy intake recommendations for non-Asian populations is the subject of much discussion [7].
The availability of soy in Latin America and the incorporation of soy into functional foods, nutraceuticals, and dietary supplements have ensured the availability of genistein on a global platform. The study and analysis of genistein have progressed from nutritional epidemiology to molecular pharmacology and have entered the clinical realm. This transition has occurred owing to the adoption of integrated approaches such as metabolomics, transcriptomics, and proteomics studies in molecular science [8]. Further, innovations in nanotechnology-based formulations are also improving the availability and metabolism rate of this compound and thus improving its therapeutic utility [9].
In the context of the increasing prevalence of chronic inflammatory diseases and cancer worldwide, the search for bioactive substances that are not only efficacious but also non-toxic and of natural origin assumes great importance. Genistein, a compound found abundantly in soybeans that are grown in large quantities in Latin America, has a unique distinction to offer. This highlights the relevance of genistein as a compound that could be used in a preventive or therapeutic capacity.
Accordingly, the scope of the present review includes a discussion of the biological functions of genistein, its mechanisms of action, and its applications in an effort to provide an update of its developments in the area of inflammation and cancer studies, in addition to reviewing the latest advances in the area of omics and nanotechnology in improving its efficacy and applications.
A comprehensive search was conducted across multiple databases, including PubMed, ScienceDirect, Scopus, and Web of Science. The search was designed to identify relevant published literature, using specific keywords. The primary keywords included genistein, anti-inflammation, cancer, anticancer, molecular mechanisms, prevention, treatment, in vivo, in vitro, and clinical studies. Additionally, the reference lists of selected articles were manually screened to identify further relevant studies.
The review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [10]. The inclusion criteria comprised full-length articles published in peer-reviewed journals that reported original findings on the anticancer properties of genistein, including mechanistic insights, preclinical studies, and clinical investigations. Only articles published in the English language were included. Exclusion criteria included published conference abstracts, unpublished data, and non-English language publications. From each eligible study, data were extracted on publication year, study design (in vitro, in vivo, or clinical), cancer type, inflammation of different diseases, intervention or treatment details, molecular mechanisms studied, and major findings. Extracted data were organized systematically to summarize the current evidence base regarding the anticancer effects of genistein.
Dietary intake of soy products contributes significantly to genistein levels in populations with soy-rich diets, such as those in East Asian countries, where daily isoflavone consumption averages 25–50 mg, compared to less than 3 mg per day in Western diets [7]. This difference is thought to contribute to the lower prevalence of hormone-dependent cancers, cardiovascular diseases, and metabolic disorders in Asian populations. Genistein exerts a wide range of biological and pharmacological activities that extend beyond its estrogenic role. Its pleiotropic mechanisms include modulation of intracellular signalling pathways, regulation of oxidative stress responses, and control of inflammatory mediators. Functionally, genistein has been shown to interact with estrogen receptors (ERα and ERβ), with higher affinity for ERβ, leading to tissue-specific effects that differ from endogenous estrogens [11]. It is also reported that genistein exerts antioxidant properties by scavenging reactive oxygen species (ROS) and activating cytoprotective pathways such as the Nrf2/ARE axis [12]. Inhibiting nuclear factor-kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) pathways, genistein modulates inflammation [2, 6]. Additionally, it influences cell proliferation and apoptosis by inducing cell cycle arrest and apoptosis in malignant cells, while protecting normal tissues from stress-induced injury [9].
Evidence from preclinical, clinical, and epidemiological studies has linked genistein to a wide array of health benefits (Table 1), such as genistein interferes with multiple hallmarks of cancer, including uncontrolled proliferation, resistance to apoptosis, angiogenesis, and metastasis [11]. Regular intake of soy isoflavones has been associated with improved vascular function, reduced atherosclerosis, and regulation of lipid metabolism [12] and improved insulin sensitivity, modulation of adipogenesis, and prevention of obesity and type 2 diabetes [13]. By enhancing osteoblast differentiation and inhibiting osteoclast activity, genistein contributes to bone preservation and has potential applications in osteoporosis [14]. Genistein also exhibits protective effects against neuroinflammation and oxidative stress in models of neurodegenerative disorders [14].
Different health benefits from a legume-based diet, such as soybeans and soy products.
| Nutritional source | Health benefit | Mechanistic basis | References |
|---|---|---|---|
| Chickpeas, lentils, fava beans | Hormone-dependent cancer prevention (e.g., breast, prostate) | Binds ERβ, modulates estrogen signalling, induces apoptosis, inhibits angiogenesis | [15–17] |
| Soy protein-rich diets (East Asia) | Cardiovascular protection | Improves endothelial function, lowers low-density lipoprotein (LDL), and enhances nitric oxide synthesis | [18–20] |
| Isoflavone-enriched foods | Bone health and osteoporosis prevention | Stimulates osteoblast activity, inhibits osteoclast differentiation | [21–23] |
| Legume-based diets | Metabolic regulation (type 2 diabetes, obesity) | Enhances insulin sensitivity, reduces adipogenesis, and regulates lipid metabolism | [24, 25] |
| Soy isoflavones | Anti-inflammatory effects | Downregulates NF-κB, MAPK, JAK/STAT pathways; reduces cytokines (IL-6, TNF-α) | [26–28] |
| Soy-rich nutrition | Neuroprotection and cognitive benefits | Antioxidant defence, ERβ modulation, reduces neuroinflammation | [29, 30] |
| Plant-derived isoflavones | General antioxidant support | Scavenges reactive oxygen species (ROS), upregulates endogenous antioxidant enzymes [superoxide dismutase (SOD), catalase] | [31, 32] |
ER: estrogen receptor; MAPK: mitogen-activated protein kinase; NF-κB: nuclear factor-kappa B.
Genistein (4′,5,7-trihydroxyisoflavone) is a naturally occurring isoflavone, classified under the polyphenolic family of secondary metabolites. Structurally, it consists of a C15 skeleton with two aromatic rings (A and B) connected by a heterocyclic pyran ring (C) (Figure 1). This chemical arrangement confers estrogenic activity on genistein due to its similarity to 17β-estradiol, allowing it to bind to ERs [21, 33]. The hydroxyl groups at the 5, 7, and 4′ positions are critical for hydrogen bonding, radical scavenging, and receptor interaction. The molecule exhibits poor water solubility, moderate lipophilicity, and is predominantly found in soy as glycosides (e.g., genistin), which require enzymatic hydrolysis in the intestine to release the biologically active aglycone [8, 11].
One of the major limitations of genistein as a therapeutic agent is its low oral bioavailability. After ingestion, genistein glycosides are hydrolyzed in the gut to release the aglycone form, which undergoes absorption in the small intestine. However, genistein is subjected to first-pass metabolism in the liver, leading to glucuronidation, sulfation, and rapid systemic clearance, resulting in reduced plasma concentrations of the active compound [8, 11]. Inter-individual variability, influenced by gut microbiota composition, dietary factors, and genetic polymorphisms, further modulates its absorption and metabolism [8, 11].
To overcome these challenges, different formulation strategies have been explored, including nanoparticle encapsulation (nano-genistein or hybrid nanocarriers) to improve solubility and stability, lipid-based carriers (liposomes, phytosomes) to enhance intestinal absorption [9], and co-administration with bioenhancers such as piperine to slow metabolism [34].
The plasma half-life of genistein ranges between 6 and 8 hours, depending on dose and formulation [8]. It tends to accumulate in estrogen-responsive tissues (breast, prostate, uterus) as well as bone, liver, and brain, consistent with its pleiotropic biological activities. The balance between aglycone and conjugated metabolites determines its bioactivity at the cellular level [11].
Genistein exerts its diverse biological and therapeutic effects through a complex network of molecular interactions. Its ability to act both as a phytoestrogen and a pleiotropic signalling modulator allows it to influence cell proliferation, inflammation, oxidative stress, and survival pathways [1, 9].
Genistein structurally mimics 17β-estradiol, enabling it to bind to ERs. It displays a higher affinity for ERβ than for ERα, producing tissue-selective effects distinct from endogenous estrogens [1]. ERβ activation is associated with anti-proliferative, pro-apoptotic, and anti-inflammatory functions [1, 31]. Through ER signalling, genistein regulates the expression of genes involved in cell cycle control, apoptosis, and differentiation [15, 17].
This selective ER modulator (SERM)-like activity positions genistein as a potential alternative to hormone replacement therapy with reduced risk of carcinogenesis compared to synthetic estrogens [1, 15, 17].
Oxidative stress contributes significantly to the pathogenesis of inflammatory disorders, neurodegeneration, and cancer. Genistein enhances cellular defence via direct scavenging of ROS and reactive nitrogen species (RNS) [1]. It activates the Nrf2/ARE signalling axis, leading to the transcription of antioxidant genes such as heme oxygenase-1 (HO-1), glutathione peroxidase, and superoxide dismutase [6, 32]. Genistein also preserves mitochondrial function and prevents lipid peroxidation in stressed tissues [32]. By reinforcing redox homeostasis, it contributes to long-term cytoprotection [1, 6, 32].
Chronic inflammation is a central driver of multiple pathologies, including cancer. Genistein inhibits NF-κB signalling, resulting in reduced transcription of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) [1, 2, 9]. It also downregulates MAPK pathways [extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), p38], thereby modulating stress responses and cytokine production [1, 2], and suppresses the JAK/STAT pathway, relevant in cancer and autoimmune diseases [1, 9]. Genistein reduces inducible enzymes such as cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), limiting prostaglandin and nitric oxide production [1, 2, 9]. Collectively, these mechanisms prevent uncontrolled inflammation, which otherwise promotes tissue damage, fibrosis, and tumour progression (Figure 2).
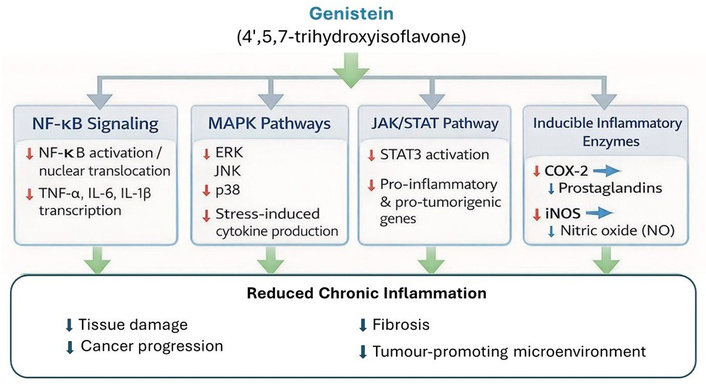
Anti-inflammatory mechanisms of genistein. It inhibits nuclear factor-kappa B (NF-κB) activation and nuclear translocation, suppresses mitogen-activated protein kinase (MAPK) signalling pathways (ERK, JNK, and p38), downregulates the JAK/STAT pathway, inhibits inducible inflammatory enzymes, including cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), resulting in reduced transcription of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β, attenuating stress-induced cytokine production, and reduces the expression of pro-inflammatory and pro-tumorigenic genes, decreasing prostaglandin and nitric oxide production. Collectively, these mechanisms contribute to reduced chronic inflammation, limiting tissue damage, fibrosis, and inflammation-driven tumour progression. ERK: extracellular signal-regulated kinase; JNK: c-Jun N-terminal kinase.
Genistein inhibits cell proliferation by inducing cell cycle arrest at the G1/S and G2/M transitions. It upregulates CDK inhibitors such as p21Cip1 and p27Kip1, suppresses cyclin D1 and cyclin E expression, and blocks receptor tyrosine kinase signalling [epidermal growth factor receptor (EGFR), platelet-derived growth factor receptor (PDGFR)], reducing MAPK/ERK and PI3K/Akt pathway activity [35]. In hormone-sensitive cancers, it also modulates ER-dependent signalling to reduce estrogen-driven proliferation [2, 36]. For apoptosis, genistein activates both intrinsic (mitochondrial) and extrinsic (death receptor) pathways. It upregulates pro-apoptotic proteins (Bax, Bad) while downregulating anti-apoptotic Bcl-2 and Bcl-xL, leading to cytochrome c release, caspase-9, and caspase-3 activation [37, 38]. It also enhances Fas/FasL signalling, promoting caspase-8 activation [38], and stabilizes p53, facilitating apoptosis under stress conditions [38]. Genistein suppresses NF-κB and activates Nrf2 responses, thereby reinforcing apoptosis while reducing oxidative damage [39].
These dual effects on proliferation and apoptosis highlight its promise as a chemopreventive and therapeutic agent, effective in preclinical models of breast, prostate, colon, and pancreatic cancers [40]. It may also enhance the sensitivity of tumours to chemotherapy and radiotherapy [1]. Beyond oncology, modulation of cell turnover and apoptosis may support tissue regeneration, aging, and inflammatory disorders [40, 41].
Emerging evidence identifies genistein as a potent epigenetic modulator. It influences DNA methylation by regulating DNA methyltransferase (DNMT) activity, leading to demethylation and reactivation of tumour suppressor gene promoters [28, 34, 42]. This reactivation restores critical regulatory pathways governing cell cycle arrest and apoptosis in cancer cells. Genistein also induces selective changes in global DNA methylation patterns in a context-dependent manner, modulating the transcriptional landscape through effects on both tumour suppressor genes and oncogenes [43, 44].
In addition to DNA methylation, genistein modulates histone post-translational modifications. It inhibits histone deacetylases (HDACs), resulting in increased histone acetylation and chromatin relaxation, thereby facilitating transcription of growth-inhibitory and pro-apoptotic genes [45]. Genistein further influences histone methyltransferases and demethylases, altering histone methylation marks that favour tumour suppressor gene expression and suppress oncogenic transcriptional programs [46, 47].
Genistein also regulates epigenetic networks through non-coding RNAs. It upregulates tumour-suppressive microRNAs (miRNAs), including miR-34a and the miR-200 family, while downregulating oncogenic miRNAs such as miR-21 and miR-155, thereby inhibiting proliferation, invasion, and chemoresistance [28, 48, 49]. Additionally, genistein modulates the expression of specific long non-coding RNAs involved in chromatin remodelling and transcriptional regulation, contributing to apoptosis induction and growth inhibition [50, 51].
Collectively, this epigenetic plasticity highlights genistein’s potential for long-term disease prevention and therapeutic intervention.
Genistein inhibits protein tyrosine kinase activity, particularly targeting EGFR and PDGFR [52–55]. Its interaction with growth factor signalling is concentration-dependent and context-specific, primarily investigated in cancer chemoprevention and therapeutic settings [56–58]. As a protein tyrosine kinase inhibitor, genistein suppresses phosphorylation and activation of EGFR and other ErbB family members, including HER2/ErbB2, thereby attenuating ligand-driven proliferative and survival signalling [52, 53, 59]. Genistein also inhibits insulin-like growth factor-1 receptor (IGF-1R) signalling and its crosstalk with ER pathways. At higher concentrations, tyrosine kinase inhibition predominates, resulting in anti-proliferative effects through concurrent blockade of ER- and GF-mediated signalling, whereas at lower concentrations, estrogenic activity may dominate. In ER+/HER2+ cells, low-dose genistein can paradoxically enhance cell proliferation by activating ER signalling and amplifying ER-HER2 crosstalk [60].
Genistein has been extensively studied for its diverse pharmacological and therapeutic effects. Its role as a diet-derived bioactive compound spans across multiple organ systems, with mounting evidence highlighting its ability to regulate inflammation and carcinogenesis [1, 2, 6, 9, 14].
Genistein exerts potent anti-inflammatory effects through multifaceted molecular mechanisms, including suppression of NF-κB and MAPK pathways, activation of Nrf2 antioxidant defences, and regulation of Toll-like receptor (TLR) and inflammasome activity [1, 2, 6]. Genistein inhibits activation of NF-κB, a central transcription factor that regulates pro-inflammatory cytokines (TNF-α, IL-1β, IL-6). It prevents phosphorylation and degradation of IκBα, thereby reducing NF-κB nuclear translocation. This downregulation leads to decreased production of adhesion molecules (ICAM-1, VCAM-1) and inflammatory mediators [2, 6, 9, 15].
Genistein suppresses the MAPK pathways (ERK, JNK, and p38), which are crucial in cytokine production and immune cell activation [2, 6]. By attenuating MAPK signalling, genistein reduces inflammatory gene transcription and tissue damage. Genistein modulates TLR signalling, reducing the downstream inflammatory cascade triggered by pathogen-associated molecular patterns (PAMPs). It also inhibits activation of the NLRP3 inflammasome, thereby reducing caspase-1 activation and IL-1β/IL-18 secretion [12, 29]. In obesity and diabetes models, genistein reduces adipose tissue inflammation by lowering TNF-α and IL-6. It improves endothelial function by reducing vascular inflammation and oxidative stress [2, 6]. Genistein suppresses tumour-promoting inflammation in the tumour microenvironment, downregulates COX-2, iNOS, and pro-inflammatory cytokines, thereby limiting inflammation and disease progression (Table 2). In neurodegenerative disease models, genistein reduces microglial activation and cytokine release, protecting neurons from inflammation-induced apoptosis. Genistein decreases airway hyperresponsiveness and inflammatory cytokines in models of asthma and COPD. In tuberculosis and other infectious conditions, it modulates macrophage-mediated inflammatory responses [2, 6, 29].
Anti-inflammatory effects of genistein.
| Mechanism/Pathway | Inflammatory condition/model | Study type | References |
|---|---|---|---|
| NF-κB inhibition results in a reduction of TNF-α, IL-6, and IL-1β | Rheumatoid arthritis, lipopolysaccharide (LPS)-stimulated macrophages | In vitro, in vivo | [1, 2] |
| MAPK pathway suppression (ERK, JNK, p38) | Colitis, LPS-induced inflammation | In vitro, in vivo | [1, 6] |
| JAK/STAT signalling modulation | Autoimmune disorders, cancer-related inflammation | In vitro | [59] |
| COX-2 and iNOS downregulation results in a reduction of prostaglandin and NO | Osteoarthritis, inflammatory bowel disease | In vitro, in vivo | [2, 14] |
| Inhibition of TLR4 signalling results in a reduction in MyD88/NF-κB pathway activation | Sepsis, LPS-challenged models | In vitro, in vivo | [61] |
| Antioxidant-mediated inflammation reduction (ROS scavenging, Nrf2 activation) | Neuroinflammation, cardiovascular inflammation | In vitro, in vivo | [1, 12] |
| Modulation of inflammasome activation (NLRP3) | Liver inflammation, metabolic syndrome | In vitro | [62] |
COX-2: cyclooxygenase-2; ERK: extracellular signal-regulated kinase; iNOS: inducible nitric oxide synthase; JNK: c-Jun N-terminal kinase; MAPK: mitogen-activated protein kinase; NF-κB: nuclear factor-kappa B; ROS: reactive oxygen species; TLR4: Toll-like receptor 4.
Chronic inflammation is a key factor in the progression of autoimmune diseases, metabolic disorders, and cancer. Genistein demonstrates broad-spectrum anti-inflammatory activity. These mechanisms suggest that genistein could be developed as a nutraceutical or adjuvant therapy for chronic inflammatory diseases such as rheumatoid arthritis, inflammatory bowel disease, asthma, and COPD [6, 24].
The most extensively studied therapeutic application of genistein is its role in cancer prevention and therapy, due to its ability to modulate multiple cellular signalling pathways. It exhibits multiple effects (Figure 3), including regulation of cell proliferation, apoptosis, angiogenesis, metastasis, and epigenetic modifications [1, 2]. Unlike cytotoxic chemotherapy, genistein demonstrates a chemopreventive and chemosensitizing profile with relatively low toxicity, making it a promising candidate for integrative cancer therapy (Table 3) [3].
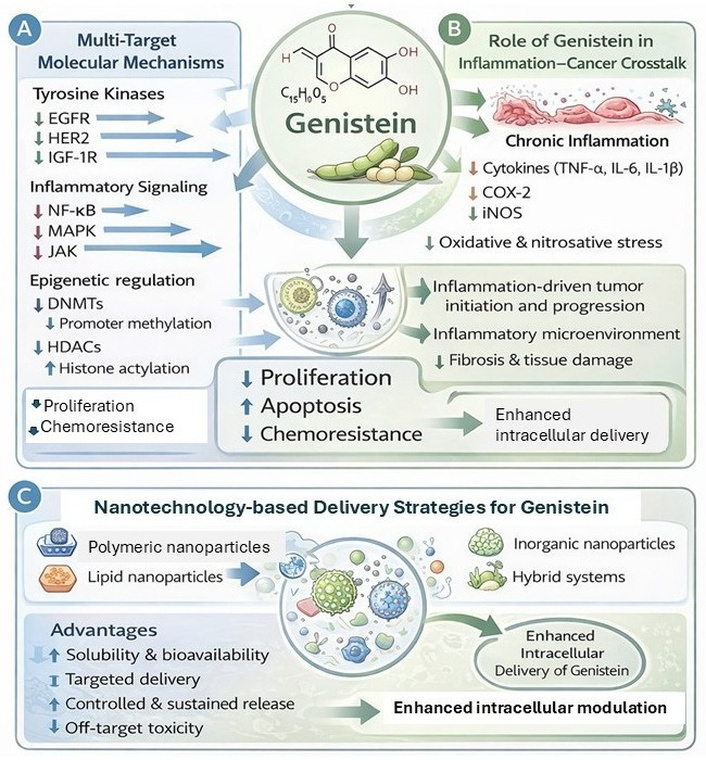
Conceptual schematic illustrating the (A) multi-target molecular mechanisms of genistein, (B) role of genistein in inflammation-cancer crosstalk, and (C) nanotechnology-based delivery strategies of genistein. The figure integrates evidence from published preclinical studies and is author-generated for illustrative purposes. This figure was created using AI-assisted visualization tools and refined by the authors. No original experimental data are presented. COX-2: cyclooxygenase-2; DNMTs: DNA methyltransferases; EGFR: epidermal growth factor receptor; HDACs: histone deacetylases; IGF-1R: insulin-like growth factor-1 receptor; iNOS: inducible nitric oxide synthase; MAPK: mitogen-activated protein kinase; NF-κB: nuclear factor-kappa B.
Anti-cancer effects of genistein.
| Mechanism | Cancer type | Study type | References |
|---|---|---|---|
| Cell cycle arrest (G2/M or G1/S) via cyclin/CDK (cyclin-dependent kinases) downregulation | Breast, prostate, colon | In vitro, in vivo | [1, 53] |
| Apoptosis induction (caspase activation, Bcl-2 inhibition) | Breast, pancreatic, leukemia | In vitro, in vivo | [11, 12] |
| Anti-angiogenesis (VEGF inhibition) | Breast, lung | In vitro, in vivo | [13] |
| Suppression of metastasis (MMPs, EMT inhibition) | Prostate | In vitro | [5, 16] |
| Tyrosine kinase inhibition (EGFR, PDGFR) | Pancreatic, prostate | In vitro, in vivo | [2, 9] |
| Epigenetic regulation (DNA methylation, miRNA modulation) | Breast, prostate | In vitro | [43, 44] |
| Anti-inflammatory (NF-κB, MAPK suppression) | Colorectal, liver | In vitro | [2] |
EGFR: epidermal growth factor receptor; EMT: epithelial-to-mesenchymal transition; MAPK: mitogen-activated protein kinase; MMPs: matrix metalloproteinases; NF-κB: nuclear factor-kappa B; PDGFR: platelet-derived growth factor receptor; VEGF: vascular endothelial growth factor.
It inhibits cell proliferation by interfering with the cell cycle, particularly inducing arrest at the G2/M or G1 phase, downregulating cyclins (cyclin D1, cyclin E) and CDKs while upregulating cell cycle inhibitors (p21, p27) [53, 63]. It also suppresses growth factor receptor signalling, including EGFR and HER2, commonly overexpressed in cancers [2, 5].
Genistein induces apoptosis by activating intrinsic (mitochondrial) and extrinsic (death receptor) apoptotic pathways. It enhances pro-apoptotic proteins (Bax, Bak) and decreases anti-apoptotic proteins (Bcl-2, Bcl-xL), and activates caspase cascades, leading to programmed cell death [6].
It inhibits the expression of VEGF (vascular endothelial growth factor) and HIF-1α, key mediators of tumour angiogenesis, and shows anti-angiogenic activity. It suppresses endothelial cell proliferation and migration, reducing neovascularization in tumours [53].
Anti-metastatic properties have been identified when it downregulates matrix metalloproteinases (MMP-2, MMP-9) that degrade the extracellular matrix. It reduces epithelial-to-mesenchymal transition (EMT) by restoring E-cadherin expression and limits tumour invasion and metastatic spread [8]. It also acts as a DNA methylation modulator, reactivating silenced tumour suppressor genes, modulating histone acetylation and miRNA expression, contributing to long-term chemopreventive effects [9, 11].
As previously discussed, genistein’s modulation of key oncogenic pathways suppresses NF-κB, PI3K/Akt, and MAPK pathways, reducing pro-survival and inflammatory signalling. It enhances p53 activity, promoting apoptosis and DNA repair. In hormone-dependent cancers, genistein acts as a phytoestrogen, binding to ERβ and modulating ER signalling [12].
In breast cancer studies, it exhibits dual estrogenic/anti-estrogenic effects depending on the hormonal context. In ER-positive breast cancer, genistein competes with estradiol, potentially lowering estrogen-driven proliferation, and sensitizing breast cancer cells to tamoxifen and other endocrine therapies [58].
It inhibits androgen receptor signalling and prostate-specific antigen (PSA) expression, reducing tumour growth and angiogenesis in prostate cancer models [53].
It has also been reported that, in lung and gastrointestinal cancers, it suppresses EGFR-mediated proliferation in non-small cell lung cancer (NSCLC) and reduces COX-2 and inflammatory mediators in colorectal and gastric cancers [64].
In pancreatic cancer, it inhibits NF-κB and STAT3 signalling, reducing cancer stem cell populations and enhancing the efficacy of gemcitabine and other chemotherapeutic agents [53].
Epidemiological studies link high soy consumption with lower incidence of hormone-dependent cancers such as breast, prostate, and endometrial cancers [3, 5]. Preclinical models also support genistein’s efficacy against gastrointestinal, lung, and pancreatic cancers [2, 15].
The rapid development of high-throughput omics technologies has transformed our understanding of phytochemicals, enabling comprehensive insights into their metabolism, molecular interactions, and systemic effects. For genistein, omics tools—particularly metabolomics, transcriptomics, proteomics, and epigenomics—have provided critical evidence linking its intake to specific cellular pathways and health outcomes [1, 6, 9].
Metabolomics allows the identification and quantification of genistein and its metabolites in biological systems, offering a detailed view of its bioavailability and metabolic fate. Genistein undergoes extensive phase II metabolism, generating glucuronide and sulfate conjugates detectable in plasma and urine [8, 11]. Advanced metabolomics studies have revealed tissue-specific accumulation patterns and inter-individual variability in metabolism [11]. Comparative studies in populations with high vs. low soy intake have correlated genistein-derived metabolites with reduced cancer risk and improved cardiometabolic profiles [3]. Metabolomics thus bridges dietary intake with personalized health responses to genistein.
Genistein influences global gene expression through ER-dependent and ER-independent pathways. Transcriptomic profiling (RNA-seq, microarrays) has identified downregulation of oncogenes (e.g., c-Myc, cyclin D1) in cancer cells [41], upregulation of tumour suppressor genes (e.g., p21, PTEN), modulation of immune-related genes leading to reduced pro-inflammatory cytokine expression [2], and regulation of metabolic gene networks associated with glucose homeostasis and lipid metabolism [25]. These transcriptomic signatures demonstrate how genistein exerts multi-targeted therapeutic effects.
Proteomics has uncovered genistein’s impact at the protein expression and signalling level through its tyrosine kinase inhibition activity. Genistein alters phosphorylation states of proteins downstream of EGFR, PDGFR, and VEGFR [52, 53, 55]. Proteomic data also show increased expression of pro-apoptotic proteins (Bax, caspase-3) and decreased anti-apoptotic proteins (Bcl-2, survivin) [41]. NF-κB subunits, COX-2, and iNOS are consistently suppressed following genistein exposure [2]. This proteomic evidence complements transcriptomic findings, validating the mechanistic pathways of genistein action.
Epigenomic studies highlight genistein’s ability to reprogram cellular states without altering DNA sequence. Genistein modulates promoter methylation of tumour suppressors such as p16 and BRCA1 [42–45]. It influences histone acetylation and methylation, altering chromatin accessibility for transcription [43]. Genistein has been shown to regulate oncogenic and tumour-suppressive miRNAs, including miR-21, miR-155, and let-7 family members [46, 48, 49]. These epigenetic effects support the concept of genistein as a nutritional epigenetic modulator with implications for cancer prevention and therapy.
Recent studies increasingly employ multi-omics integration, combining metabolomics, transcriptomics, and proteomics to capture a holistic view of genistein’s biological actions. Such approaches have identified network-level interactions, showing how genistein simultaneously regulates oxidative stress, inflammation, and proliferation. Systems biology modelling, supported by omics data, predicts genistein’s synergistic effects with conventional drugs, providing a framework for host-directed and precision therapies [1, 6, 9].
Despite compelling preclinical evidence supporting the therapeutic promise of genistein, several challenges limit its translation from bench to bedside. Addressing these issues is critical for harnessing its full potential in the prevention and treatment of inflammation-driven diseases and cancer.
One of the primary barriers is genistein’s poor oral bioavailability. It has low aqueous solubility and is unstable under gastrointestinal conditions, and undergoes extensive first-pass metabolism, leading to glucuronidation and sulfation [8, 11]. Additionally, inter-individual variability due to differences in gut microbiota composition further modulates its absorption and systemic levels [2]. Advances in nanotechnology-based delivery systems (nanoparticles, micelles, phytosomes) and prodrug approaches are showing promise in improving solubility, absorption, and tissue targeting (Table 4) [65–74].
Genistein nano delivery systems in cancer therapy.
| Nanocarrier type | Targeted cancer type | Key findings | References |
|---|---|---|---|
| Self-assembled genistein nanoparticles | Glioblastoma | Suppress epithelial-mesenchymal transition by targeting MMP9. | [65] |
| Genistein nanocrystals | General cancer types | Improve aqueous solubility and bioavailability of genistein. | [66] |
| Platelet membrane-coated liposomes | Lung cancer | Enhance targeting and therapeutic efficacy in lung cancer treatment. | [67] |
| TPGS-b-PCL nanoparticles | Cervical cancer | Enhance the anticancer effect of genistein-loaded nanoparticles. | [68] |
| Different formulations of genistein influence bone mineral status during chemically induced breast cancer | Bone health | Nanogenistein have a negative effect on the amount of calcium in the bone tissue, indicating its increased distribution to the bone. | [69] |
| Anti-CD44-conjugated olive oil liquid nanocapsules | Pancreatic cancer stem cells | Selective targeting and increased antitumour efficacy in pancreatic cancer stem cells. | [70] |
| Genistein-loaded transfersomes | Skin melanoma | Developed a novel topical delivery system for skin melanoma treatment. | [71] |
| TPGS-b-PCL nanoparticles | Melanoma | PCL-based nanoparticles can enhance the delivery and therapeutic efficacy of genistein in melanoma treatment. | [72] |
| Genistein-loaded biocompatible nanocarriers | Hematopoietic malignant cells | Promising biocompatible chemotherapeutic agent against hematopoietic malignant cells. | [73] |
| Genistein-chitosan derivative nanoparticles | Breast cancer | Potential against breast cancer through active targeting and synergistic treatment strategies. | [74] |
MMP-9: matrix metalloproteinase-9.
Although genistein is considered safe as a dietary component, pharmacological doses raise questions regarding potential hormone-related effects in reproductive tissues. Biphasic responses, where low doses are protective but high doses may stimulate proliferation in some cancer cell types, have been reported [75]. Moreover, genistein has limited long-term clinical safety data, especially in vulnerable populations such as children, pregnant women, and the elderly. Well-designed dose-ranging clinical trials are therefore needed to establish optimal dosing, treatment duration, and safety thresholds [1].
Most current evidence comes from in vitro and animal models, which may not fully recapitulate human disease complexity. Clinical trials to date have shown modest or inconsistent outcomes, often due to small sample sizes, short treatment durations, and lack of standardized formulations and biomarkers for efficacy [3, 12]. Large-scale, multi-center randomized controlled trials (RCTs) using standardized genistein formulations and validated molecular biomarkers are necessary to establish clinical relevance.
While genistein targets multiple cancer pathways, drug resistance mechanisms (e.g., upregulation of alternative signalling routes) may limit its standalone efficacy. Investigating synergistic effects of genistein with chemotherapeutics, immunotherapies, and targeted drugs, as well as exploring host-directed therapeutic strategies, in which genistein enhances immune and metabolic resilience in inflammatory and neoplastic disorders, may help avoid resistance development [6, 9].
Beyond its well-established anti-inflammatory and anti-cancer roles, genistein shows potential in neurodegeneration (Alzheimer’s, Parkinson’s disease) via antioxidant and anti-amyloid effects [31, 32], in cardiometabolic disorders by regulating lipid metabolism and insulin sensitivity [26], and in bone health as a safer alternative to hormone replacement therapy [23]. Systematic exploration of these new therapeutic domains, supported by omics-guided mechanistic studies, will expand the clinical relevance of genistein [1].
Genistein, an isoflavonoid found in soya foods, is one of the most researched phytoestrogens, with immense uses in human biology. Various biologic uses of genistein, including selective ER modulation, oxidative protection mechanisms, inflammation regulation, epigenetic pathway regulation, and tyrosine kinase inhibition, qualify it to act as a multi-target biologic.
In vivo studies clearly reveal the efficacy of genistein in the suppression of inflammation, inhibition of tumorigenesis, promotion of apoptosis, and control of immune metabolism. Besides its role in combating cancer and inflammation, the applications of genistein have also been discovered in the areas of cardiac shielding, control of bone functions, and neuronal shielding, thus clearly underlining the importance of the compound for overall health with the help of nutraceuticals. Despite the strong efficacy of the compound in various studies, the unavailability of effective oral utilization and the differences in the metabolic rate of the compound for advanced studies in each individual have acted as an obstacle to its development.
The most prominent of these translational barriers is the hydrophobic nature of genistein due to its low aqueous solubility, very low oral availability, rapid metabolism rate, and position-dependent biological activity. Moreover, despite the encouraging results of in vitro as well as in vivo studies, the current literature reveals a remarkable paucity of well-designed large-scale clinical trials assessing genistein alone or in combination with conventional therapies for their anti-cancer as well as anti-inflammatory activities. Overcoming these barriers will help translate genistein from a bench-level molecule to a translationally relevant agent.
Nanotechnology-based delivery systems have been introduced as one of the promising approaches to address the challenges. The use of nanoformulations has shown potential in improving genistein stability, concentration-specific targeting, pharmacokinetic modulation, and minimizing off-targeting in preclinical studies. Nevertheless, there is still a need to focus on improving scalability, long-term safety, and regulatory compliance of nanocarrier platforms. Comparative analysis studies between nanocarrier systems will be useful in determining which systems are applicable in a clinical setting.
From an immunological and oncological viewpoint, the potential applications of genistein emerge as part of immune therapies. The ability to inhibit chronic inflammation, act on cytokine signalling pathways, indirectly alter the expression of immune checkpoint proteins, and interact with the tumour-immune microenvironment indicates a promising role for this compound alongside and after immunotherapies such as immune checkpoint blockers and cancer vaccines. Rationally designed combination therapies are thus one area for future research development.
In looking to the future, it can be said without any doubt that the future of genistein lies where there are connections known as nutrition, molecular medicine, and precision medicine. By integrating the ancient knowledge of nutrition with modern biological techniques such as those of metabolomics, transcriptomics, and epigenomics, it would not be impossible to give genistein a “new hat to wear” as a next-generation bioactive agent once validated in a modern biological context. However, to date, it has the promise of being a “hope of the future” within the biomedical sciences.
COX-2: cyclooxygenase-2
EGFR: epidermal growth factor receptor
ER: estrogen receptor
ERK: extracellular signal-regulated kinase
iNOS: inducible nitric oxide synthase
JNK: c-Jun N-terminal kinase
MAPK: mitogen-activated protein kinase
miRNA: microRNA
MMPs: matrix metalloproteinases
NF-κB: nuclear factor-kappa B
PDGFR: platelet-derived growth factor receptor
ROS: reactive oxygen species
TLR: Toll-like receptor
VEGF: vascular endothelial growth factor
The authors acknowledge the use of OpenAI’s ChatGPT as an assistive tool for English language improvement and for conceptual support in preparing schematic figure layouts. All scientific content, interpretations, and final figures were critically reviewed, edited, and approved by the authors.
The authors gratefully acknowledge the support from the Amity Institute of Health Allied Sciences, Amity University, Uttar Pradesh, for encouraging the study and for providing infrastructural and administrative support. The authors are also thankful to the South African Technology Innovation Agency for providing time, resources, and infrastructure to do this publication.
ST: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. MGM: Validation, Writing—review & editing, Supervision. Both authors read and approved the submitted version.
Both authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 774
Download: 19
Times Cited: 0
