Affiliation:
1Laboratory of Immunopathology and Immunosenescence, Department of Biomedicine, Neuroscience and Advanced Diagnostics, University of Palermo, 90134 Palermo, Italy
ORCID: https://orcid.org/0000-0002-6075-0845
Affiliation:
1Laboratory of Immunopathology and Immunosenescence, Department of Biomedicine, Neuroscience and Advanced Diagnostics, University of Palermo, 90134 Palermo, Italy
ORCID: https://orcid.org/0000-0003-2593-3221
Affiliation:
2Department of Molecular and Developmental Medicine, University of Siena, 53100 Siena, Italy
3VisMederi srl, 53100 Siena, Italy
4VaepiX, Joint Research Laboratory, University of Siena, 53100 Siena, Italy
ORCID: https://orcid.org/0000-0001-7595-4974
Affiliation:
5Institute of Immunology, University of Tübingen, 72072 Tübingen, Germany
6Health Sciences North Research Institute, Sudbury, ON P3E 2H3, Canada
ORCID: https://orcid.org/0000-0002-3600-0163
Affiliation:
2Department of Molecular and Developmental Medicine, University of Siena, 53100 Siena, Italy
4VaepiX, Joint Research Laboratory, University of Siena, 53100 Siena, Italy
†These authors contributed equally to this work.
Email: trombetta@unisi.it
ORCID: https://orcid.org/0000-0001-7229-9919
Affiliation:
1Laboratory of Immunopathology and Immunosenescence, Department of Biomedicine, Neuroscience and Advanced Diagnostics, University of Palermo, 90134 Palermo, Italy
†These authors contributed equally to this work.
ORCID: https://orcid.org/0000-0001-8004-2363
Explor Immunol. 2026;6:1003244 DOI: https://doi.org/10.37349/ei.2026.1003244
Received: July 16, 2025 Accepted: March 17, 2026 Published: April 13, 2026
Academic Editor: Pierre-Antoine Gourraud, Public Health Université de Nantes, France
This review highlights the significant sex-based differences in immune responses to influenza infection and vaccination. Men are generally more susceptible to severe influenza outcomes, while women often mount stronger immune responses but experience more adverse effects. These disparities are influenced by biological factors, including sex hormones and genes, as well as gender-related social and environmental conditions. Evidence from both human and animal studies reveals sex-specific variations in antibody production, vaccine effectiveness, and clinical outcomes. Age, hormonal status, and stress further modulate these differences. Understanding these complex interactions is essential for developing tailored and equitable vaccination and treatment strategies.
The coronavirus disease 2019 (COVID-19) pandemic has heightened awareness of sex-specific differences in immune responses to severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). Indeed, substantial evidence supports a sex-biased susceptibility to viral infections, with males generally exhibiting increased incidence and severity, whereas females more frequently develop immunopathology-driven complications [1]. Influenza also represents a global public health problem that affects human populations through seasonal epidemics and pandemics; infections in humans are caused by influenza A viruses (IAVs) (H1N1 and H3N2 subtypes) and influenza B viruses (IBVs) (Victoria and Yamagata lineages) [2, 3]. The incidence, severity, and case fatality rates associated with influenza vary between men and women [4]. This observation is far from unexpected, as numerous studies on infectious diseases in males and females consistently demonstrate a clear pattern of sex-related disparities. Such variations are primarily driven by inherent biological elements, notably genetic material situated on the sex chromosomes and hormonal influences [5–8]. Nonetheless, as stated in this review, socially constructed gender roles and environmental conditions also exert a considerable impact. An overview of the fundamental pathways by which sex hormones modulate immune function is provided in references [6, 7, 9] and in Table 1, whereas the effects of the X chromosome are discussed in the next paragraph.
| Hormone | Mechanism of action | General immunological effect |
|---|---|---|
| Oestrogen | Increases B cell proliferation, class switching to immunoglobulin (Ig)G, T helper (Th)2, and T regulatory (Treg) cell responses; modulates Treg transcriptional programs | Immune-enhancing; promotes humoral responses |
| Progesterone | Induces anti-inflammatory molecules; inhibits Th1 and Th17 pathways; modulates antigen presenting cells (APCs) activation | Immunosuppressive; anti-inflammatory |
| Testosterone | Reduces pro-inflammatory cytokines [interleukin (IL)-1β, IL-6, tumour necrosis factor (TNF)]; increases IL-10; inhibits T cell proliferation; suppresses B cells and natural killer cytotoxicity | Immunosuppressive; anti-inflammatory |
| Prolactin | Promotes B cell activation and antibody production; upregulates costimulatory molecules on APCs; modulates Th1/Th2 cytokines | Immune-enhancing |
Oestrogens are potent immunomodulators that exert context-, dose-, and receptor-dependent effects on the immune system. At low to moderate concentrations, typical of reproductive age and specific phases of the menstrual cycle, oestrogens enhance both innate and adaptive immune responses. Under these conditions, they promote increased production of pro-inflammatory cytokines, including interleukin (IL)-1β, IL-6, tumour necrosis factor (TNF)-α, and type I interferon (IFN). These effects are mediated, at least in part, by the upregulation of toll-like receptor (TLR)7 and TLR9, heightened activation of plasmacytoid dendritic cells (DCs), and stimulation of B-cell survival and differentiation, as well as T helper (Th)17 polarization [12]. In contrast, during pregnancy, oestrogen levels rise markedly, fostering a shift toward Th2- and T regulatory (Treg)-dominant immune responses, enhancing FOXP3 expression, and favouring the production of anti-inflammatory cytokines such as IL-10 and transforming growth factor-β, thereby contributing to immune tolerance at the maternal-foetal interface [13]. An additional layer of complexity arises from differential oestrogen receptor (ER) signalling. Activation of ERα is more frequently associated with pro-inflammatory pathways, whereas ERβ predominantly mediates anti-inflammatory effects. The ERα/ERβ expression ratio varies across immune cell subsets, tissues, and across the life course, thereby shaping both the qualitative and quantitative features of immune responses [14, 15]. Collectively, these mechanisms suggest that oestrogens enhance immune activation during the early phases of immune responses to facilitate pathogen clearance, while subsequently promoting regulatory pathways that restrain excessive or chronic inflammation. Disruption of this finely tuned balance may contribute to the heightened susceptibility of females to inflammatory and immune-mediated diseases.
Biological sex is defined by chromosomal composition: females possess two X chromosomes (XX), whereas males carry one X and one Y chromosome (XY). The X chromosome contains several hundred protein-coding genes, in stark contrast to the much smaller Y chromosome, which harbours only a limited set of functional genes. Whereas the Y chromosome is not required for survival, at least one X chromosome is indispensable. However, the presence of two X chromosomes in females creates a potential risk of excessive expression of X-linked genes unless stringent regulatory mechanisms are in place [6, 7, 9, 16]. Most genes involved in immune processes are located on autosomes, yet a considerable proportion of immune-related genes reside on the X chromosome, where they play key roles in pathogen sensing, antigen presentation, lymphocyte activation, and immune regulation. To balance gene dosage between the sexes, female cells initiate X-chromosome inactivation (XCI) early during embryonic development, transcriptionally silencing one X chromosome through epigenetic mechanisms [16]. These processes compact the inactive X (Xi) into heterochromatin, thereby repressing the expression of most genes. Importantly, XCI is not complete. Approximately 15–30% of genes on the Xi can escape silencing to varying extents, influenced by cell type, age, and individual genetic background. While certain genes consistently evade inactivation, others do so in a variable and context-dependent manner. This partial escape from XCI has been proposed to contribute to enhanced immune reactivity in females. Indeed, growing evidence indicates that in specific immune cell subsets, particularly activated B and T lymphocytes, the regulatory machinery governing XCI may be loosened or disrupted, allowing selective reactivation of X-linked genes. Among the most functionally relevant immune genes that escape XCI are TLR7 and TLR8, which are highly expressed in human plasmacytoid DCs and critically influence CD8+ T-cell responses, Th1 and Th17 differentiation, and B-cell activation. Another key X-linked gene is FOXP3, the master transcriptional regulator of Treg cell development and function, which is essential for the maintenance of immune tolerance. Consequently, variations in X-chromosome dosage and inactivation patterns may further shape Treg-cell biology in females [17, 18].
Nevertheless, an exclusive focus on X-linked genes and their epigenetic regulation does not fully account for sex-related differences in immune responses. This limitation is highlighted by evidence showing that the expression and functional impact of many X-linked immune genes are themselves influenced by circulating sex hormones [9]. Rather than acting independently, sex hormones and sex chromosomes interact to generate immune dimorphism. Table 1 summarizes the main effects of sex hormones on immune responses.
A central unresolved issue is whether chromosomal sex, through gene dosage and X-linked effects, or hormonal milieu, exerts the dominant influence, or whether their contributions are intrinsically interdependent and context-specific. In this regard, a recent study [19] provides compelling in vivo evidence for a functional interaction between sex chromosomes and oestradiol in the regulation of memory B cells. After puberty, cisgender women (female at birth) exhibit higher frequencies of memory B cells, critical mediators of long-term immunological memory and antibody production, than age-matched cisgender men. This advantage disappears after menopause, underscoring the importance of female sex hormones. Consistently, postmenopausal women receiving hormone replacement therapy show higher memory B-cell levels than untreated women. In transgender men with an XX chromosomal complement, pharmacological suppression of endogenous oestrogens leads to a marked reduction in memory B-cell frequencies. In contrast, oestrogen supplementation in transgender women with an XY background does not increase memory B-cell numbers. By integrating comparisons across prepubertal, post-pubertal, postmenopausal, cisgender, and transgender individuals undergoing gender-affirming hormone therapy, this work disentangles the relative in vivo contributions of hormonal exposure and chromosomal complement. The observed synergy between sex hormones and sex chromosomes offers a mechanistic explanation for the female bias in antibody-mediated immune responses.
Figure 1 illustrates some of these mechanisms in the context of influenza infection.
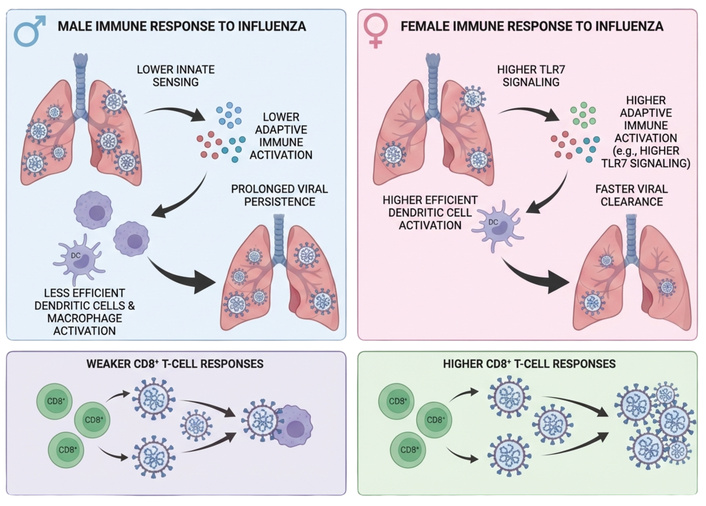
Immunological mechanisms underlying enhanced female responses to influenza infection with respect to male responses. The figure illustrates innate viral recognition and the activation of antiviral programs that are enhanced in females due to a higher TLR7 gene dosage, an X-linked receptor with biallelic expression. This leads to increased interferon production by DCs in response to TLR7 stimulation. DC responses are regulated in a sex-dependent manner and subsequently promote a more efficient adaptive immune response [20], exemplified in the figure by enhanced CD8+ T cell activity [21]. TLR: toll-like receptor; DCs: dendritic cells. Created in BioRender. Calabrò, A. (2026) https://BioRender.com/eqazsoy.
Most of the studies about the sex-related response to influenza infection have been conducted in mouse models. Although mice are not natural hosts for human influenza viruses, they represent an indispensable and cost-effective experimental system for influenza research. Mouse models enable detailed investigation of disease mechanisms, evaluation of vaccines, and analysis of host immune responses, particularly through the use of powerful genetic and molecular tools. The practical advantages of mice, including their small body size, low maintenance costs, ease of breeding, and the availability of extensive genetic resources, make them especially suitable for large-scale studies of influenza pathogenesis and immune regulation. At the same time, researchers must remain mindful of important species-specific differences from human influenza infection [22].
In this regard, findings of sex-based differences in immune response and protective effects following infection with or vaccination against the A(H1N1)pdm2009 influenza strain indicated that female mice generated higher levels and more effective virus-specific antibodies than males, a response seemingly driven by higher TLR7 expression in female B cells (see previous paragraph) [23]. Furthermore, after infection, female mice displayed greater numbers of antibody-producing B cells in the lungs and germinal centre B cells in lymph nodes than their male counterparts. However, the total B cell and follicular Th cell populations in the lungs and associated lymph nodes were similar between sexes, indicating that the key differences lie in the B cells actively producing the antibodies. Females also had larger germinal centres than males. Since germinal centre B cells are essential for the production of high-affinity antibodies, these findings suggest that larger germinal centres in females contribute to their heightened antibody responses.
In contrast with these findings, a study by Avitsur et al. [24] investigated the role of sex and stress in modulating the response to influenza A/PR8 viral infection, considering that males and females respond differently to stress, with a greater response in men compared to women. In that study, male and female mice were subjected to repeated cycles of restraint (RST) (i.e., alternating periods of restriction/stress and release/reward), followed by A/PR8 viral infection. The results demonstrated that RST altered the response to infection in a sex-dependent manner. Specifically, RST increased the expression of pro-inflammatory cytokines such as IL-6 in the lungs of both infected males and females, but with a more pronounced effect in the latter. Additionally, RST resulted in elevated lung IL-1α and IL-1β expression in infected females, but not in males. Interestingly, RST did not affect circulating corticosterone levels in influenza-infected animals. These findings suggest that males exhibit stronger immune and behavioural responses to influenza viral infection than females, and that stress amplifies these responses in both sexes. Other studies have demonstrated that chronic stress significantly alters the pathophysiology of influenza viral infection, resulting in enhanced viral replication in the lungs of infected mice [24].
Interestingly, reports indicated that men and women also differ in their physiological response to stress [25]. This raises the possibility that sex-related differences in the nature of the physiological response to stress may modulate sex-specific differences in responses to infection.
In human studies, researchers have also compared male and female responses after deliberate exposure of healthy volunteers to the A/California/04/2009 (H1N1) virus, focusing on antibody responses to haemagglutinin (HA) and neuraminidase (NA), the virus’s primary surface glycoproteins. The study found that women were more likely to develop symptoms and experience a more severe clinical course, even though HA antibody titres were similar between sexes. Notably, men exhibited significantly higher NA antibody titres post-challenge, despite similar levels of viral shedding, and were 16% less likely to report symptoms. These findings suggest that NA antibodies may serve as indicators of clinical outcomes following influenza infection, highlighting their important role in symptom mitigation and disease attenuation. This also implies that anti-NA responses may contribute to sex-based differences in influenza outcomes [26]. Nonetheless, caution is warranted, as experimental infection in volunteers does not fully replicate the complexity of natural infections in real-world settings.
In the context of seasonal outbreaks, males across all age groups consistently exhibit higher hospitalization rates compared to females, with this difference being particularly evident worldwide among young children and older people, as shown in studies conducted, for example, in Canada [27]. Evidence from Denmark indicates that the likelihood of hospitalization shifts with puberty: boys are more frequently hospitalized before puberty, whereas girls are more often affected after puberty [28]. Advancing age is the most prominent risk factor for mortality linked to seasonal influenza, with approximately 90% of deaths occurring in individuals older than 65 years [29]. Additionally, age influences influenza infection outcomes, as noted above.
The 2009 influenza pandemic offered a rare opportunity to analyse morbidity patterns across sex and age groups without the confounding effects of herd immunity, since the vast majority of the population lacked prior immunity to the novel virus strain. A retrospective study analysing 2,024,367 A(H1N1)pdm2009 cases in Japan revealed that males under the age of 20 and those over 80 were more frequently affected than females, whereas in the 20 to 79 age brackets, the trend was reversed [30]. A possible explanation for the higher incidence in younger boys could be their relatively immature immune systems compared to girls [31]. In contrast, the trend among adults may reflect a greater number of asymptomatic or mild cases in men, or potentially lower healthcare-seeking behaviour in males relative to females of similar ages [30]. Two other studies on the 2009 influenza pandemic reported a higher proportion of severely infected women compared to men [32, 33], finding that infected females were more likely to be hospitalized with severe respiratory disease than their male counterparts. As discussed in the section addressing the role of gender, these differences may also be attributed to gender-related patterns of exposure and health-seeking behaviour. Moreover, increased morbidity and mortality among pregnant women were documented during the 2009 H1N1 pandemic, with pregnant women representing a disproportionately high percentage of severe cases and exhibiting a four- to ten-fold increased risk compared with the general population [34]. Age-specific mortality during the 1918 influenza pandemic followed a W-shaped curve characterized by high mortality in infants and young children, as well as in older people, with a third peak of mortality in individuals aged 15 to 30 years. Those over the age of thirty could have been protected by pre-existing cross-immunity, likely due to an H1 flu virus that was in circulation in 1889. However, the 1918 Spanish Flu disproportionately harmed pregnant women, leading to significantly higher risks of severe disease, hospitalization, premature birth, stillbirth, and maternal death, often due to secondary pneumonia [35, 36].
Another investigation [37] observed that the likelihood of testing positive for laboratory-confirmed A(H1N1)pdm2009 decreased with advancing age. However, from approximately age 45 to 50, the decline plateaued or even reversed slightly before dropping significantly again in the late 50s. This pattern was especially notable among hospitalized individuals and among women, independent of geographical location. These findings point to sex-specific variation in influenza susceptibility with ageing, potentially linked to immune system changes associated with menopause. This phenomenon is unlikely to be exclusive to the 2009 outbreak and could be further explored using data from regular seasonal influenza waves. Additionally, a study by Wong et al. [38] explored how age and sex interacted in laboratory-confirmed influenza notifications by subtypes across Australia between 2009 and 2015. The data showed elevated notification rates among women aged 15–69 for influenza A subtypes, and among women aged 20–74 for IBVs. In contrast, males aged 0–4 had significantly higher rates regardless of virus type or subtype. For IAVs, A(H1N1)pdm2009, and IBVs, higher notification rates were also seen in males aged 0–14 and those older than 85, confirming the data on male predominance in cases at the youngest and oldest ages, while women were more represented in adult age groups, according to hormonal phases. This highlights a potentially important age-by-sex interaction in vulnerability to infection, which appears to be influenced by the virus subtype and by reproductive or menopausal state.
Mortality differences associated with avian influenza and other respiratory infections vary by sex, age, geographic location, and social factors. Global data suggest that the influenza A(H5N1) virus is associated with higher fatality rates in women [39], while in Europe, excess mortality during seasonal influenza peaks and during the COVID-19 pandemic was higher in men (see below) [40]. Similar patterns were also observed during the initial wave of human H7N9 infections, where the incidence was generally higher in males than in females; but when mortality was evaluated relative to incidence, the female-to-male death ratio was notably elevated, reaching 1.2 among women of reproductive age (15 to 49 years). This suggests that females in this age group were more likely to die from H7N9 influenza than their male counterparts [41]. Recent research also shows elevated mortality rates in rural areas, particularly among older adults, women, and individuals identifying as white [42]. These patterns are consistent with previous findings indicating increased pneumonia-related mortality in women [43], likely due to hormonal factors, comorbidities, and disparities in healthcare access, highlighting the complex interaction of biological, social, and systemic determinants of infectious disease outcomes.
Lastly, as previously mentioned, an analysis of mortality data from 27 European countries, based on Eurostat death counts and excess mortality estimates generated using the EuroMOMO model, a European mortality monitoring activity, aiming to detect and measure excess deaths that may be related to seasonal infections, extreme weather events, and other public health threats, on time, consistently demonstrated higher excess mortality among males than females during periods of increased mortality, including those associated with seasonal influenza circulation across the 2016/17 to 2019/20 seasons. In all seasons characterized by excess mortality, incidence rates were systematically higher in males, with a linear relationship observed between overall excess mortality and sex differences, corresponding to approximately 40 additional male deaths per 100 excess deaths per 100,000 population and an overall female-to-male mortality incidence ratio of about 0.7. These findings indicate that periods of excess mortality related to the winter circulation of respiratory pathogens are associated with a disproportionately greater increase in mortality among males. Importantly, the magnitude of this sex disparity was comparable across influenza seasons and during the SARS-CoV-2 pandemic, suggesting that male excess mortality represents a general phenomenon that is largely independent of the specific respiratory pathogen involved [40].
However, as suggested by several lines of evidence discussed above, the female survival advantage is context-dependent and may, under certain conditions, translate into increased disease severity and mortality. Heightened antiviral responses in females, characterized by robust type I IFN production and enhanced pro-inflammatory cytokine release, can promote more efficient viral clearance but may also increase the risk of immune-mediated tissue damage and pulmonary immunopathology, which are major determinants of severe influenza outcomes [44]. Hormonal status further modulates these effects across the life course: oestrogens, depending on circulating levels, can amplify antiviral immunity while simultaneously enhancing inflammatory cascades. Pregnancy represents a well-established high-risk state, due to sustained high levels of oestrogens and progesterone, as documented during both seasonal and pandemic influenza outbreaks, including the 1918 Spanish influenza pandemic [45, 46]. Furthermore, as previously mentioned (and discussed in the section addressing the role of gender), beyond biological sex, gender-related factors, including caregiving roles, differential exposure to infection, delayed access to healthcare, and disparities in treatment, may further contribute to worse outcomes in women in specific socio-cultural contexts [34]. Finally, sex differences in influenza morbidity and mortality vary according to viral subtype, pandemic versus seasonal settings, and population age structure, with pregnant women and younger adult females disproportionately affected in several pandemics, underscoring the need for sex- and gender-informed analyses in influenza research and public health strategies.
Vaccination remains the most reliable approach for managing seasonal influenza outbreaks and is a cornerstone strategy in pandemic prevention. The murine model remains the primary choice for initial influenza vaccine evaluation due to its cost-effectiveness, scalability, and the availability of specialized genetic and immunological tools. Different formulations of the influenza vaccine are currently in use, including adjuvanted, high-dose, and trivalent types, though direct comparative data on their relative vaccine effectiveness (VE) remain limited. One study investigated the influence of biological sex on the development of humoral immune responses to seasonal trivalent whole inactivated virus (WIV) and split virus (SV) influenza vaccines using a Swiss mouse model [47]. The results showed that sex differences in total IgG and neutralizing antibody titres are not only influenza strain-specific but also depend on the type of vaccine administered. Furthermore, the study revealed that the IgG subclass profile is specific to the vaccine type. Specifically, the WIV vaccine promoted IgG2a responses in both male and female mice, with a shift in the IgG2a/IgG1 titre ratio due to a weaker IgG1 response in females. In contrast, mice immunized with the SV vaccine exhibited nearly equal levels of IgG2a and IgG1 isotypes across both sexes.
The VE is influenced by numerous variables. It tends to be reduced in older adults compared to younger individuals, likely due to age-associated immune decline and poorer general health, as commonly reported [48]. Sex-based differences in vaccine responses are a growing area of research focus. Given the historical underrepresentation of women in clinical studies [10], investigations that stratify data by both sex and age are necessary [49].
However, existing findings do reveal distinctions in immune responses to seasonal influenza vaccines between men and women. Some double-blind, randomized trials across diverse populations have shown consistently stronger antibody responses post-vaccination in both adult and older women when compared to men of similar ages [50, 51]. A Canadian study spanning 7 influenza seasons (from 2010–2011 to 2016–2017) examined the interaction between sex and VE against medically attended, laboratory-confirmed influenza disease [52]. The study also explored whether sex-related differences in VE varied by influenza subtype/lineage, age group, or season. Results showed a modest sex-related effect on VE across most outcomes and seasons, with females typically demonstrating higher VE estimates. These sex-based effects were age-dependent, being more pronounced in older adults aged 50 years and above, compared to younger groups (< 20 years and 20–49 years), though variation existed depending on the subtype or lineage. The largest sex differences were observed for influenza A(H3N2) and B(Victoria) viruses, compared to A(H1N1)pdm2009 and B(Yamagata).
One retrospective analysis, involving 2,243 participants stratified by sex and age (15–64 and ≥ 65 years), examined influenza vaccine responses across 13 consecutive flu seasons (2006–2018) [53]. The authors observed no significant sex-based differences in baseline immunity or seroprotection rate (SPR; antibody titres ≥ 40) before vaccination in either age cohort. Interestingly, a higher SPR was reported in younger men (57.7%) versus younger women (42.9%) for classical A(H1N1) strains [53]. This discrepancy was hypothesized to stem from a higher median age within the male cohort, possibly increasing the frequency of prior exposure to A(H1N1) strains (pre-1957 and post-1977), compared to women who may have only encountered post-1977 strains. Post-vaccination, older women exhibited significantly stronger antibody responses than older men to classical A(H1N1), A(H1N1)pdm2009 viruses, and B/Victoria lineage, as measured by seroconversion rates (defined as a fourfold rise in antibody titres). No sex-based difference was noted for A(H3N2) [53]. Notably, older women in this study were of a higher median age than men. These results indicate a more robust vaccine-induced humoral response in older women.
Other reports confirm enhanced immune responses in older women [54, 55], though this does not appear to extend to antibody avidity, the strength of antibody-antigen binding, which reflects immune response quality and VE [56]. In one study, an inactivated 2009 H1N1 pandemic vaccine triggered tenfold higher antibody levels in older than younger adults [57]. Despite elevated antibody titres in older women, avidity was significantly greater in older men, implying superior antibody quality in males. Another investigation evaluated neutralizing antibody responses to a trivalent inactivated influenza vaccine across young and older adults and explored potential peripheral blood biomarkers underlying sex-based immune differences [58]. Women exhibited stronger responses to H3N2 and, to a lesser degree, to influenza B, while no sex-related differences were seen for H1N1, though age played a significant role. A cluster of lipid metabolism-related genes, likely regulated by testosterone, was associated with lower vaccine responsiveness in men. Those with higher testosterone levels and elevated expression of these genes had markedly lower vaccine-induced neutralizing antibody production. These findings further support the immunosuppressive role of testosterone, which likely acts by downregulating immune activation transcription factors, creating a feedback loop through repression of lipid metabolism genes. In another study [59], testosterone levels were found to be significantly lower in older than in younger men 42 days post-vaccination. A negative relationship was found between testosterone and seroconversion in adult, but not older, men. Oestrogen levels were also significantly lower in older versus younger women, and higher oestrogen concentrations correlated with stronger neutralizing antibody responses, especially in older women. These patterns suggest that oestrogen enhances, while testosterone suppresses, vaccine-induced antibody generation. While sex hormone levels influence immune responses in both sexes, the immunological effects of ageing appear more pronounced in women [59].
Real-world evidence concerning the effects of co-administration of influenza vaccines together with pneumococcal and/or COVID-19 vaccines is still sparse. During the 2023–2024 influenza season, a retrospective cohort analysis was performed encompassing the entire older population of an Italian province (43.9% of the population aged ≥ 60 years received an influenza vaccine, i.e., n = 46,355/105,527). The objective was to assess the VE of the quadrivalent influenza vaccine, administered either on its own or in combination with other recommended immunizations. Among the older adult population in this province, all administered inactivated quadrivalent influenza vaccines were associated with a marked and statistically significant reduction in the risk of hospitalization or death linked to influenza and/or pneumonia. Moreover, the simultaneous administration of pneumococcal and/or COVID-19 vaccines conferred additional protection against both outcomes. No significant differences in VE were detected across sex, age, or between enhanced versus standard formulations [60].
Recently, a comprehensive meta-analysis was conducted using phase 3 randomized controlled trial data collected between 2010 and 2018 to investigate sex-specific differences in responses to seasonal influenza vaccination [61]. Haemagglutination inhibition antibody titres were used to quantify strain-specific immune responses, and sex-based differences in geometric mean ratios (GMRs) were estimated. In addition, pooled risk ratios for seroconversion were calculated for females and males using random-effects models, and VE against laboratory-confirmed influenza was evaluated. Analyses were stratified by age group (18–64 years and ≥ 65 years). Overall, immunogenicity data from 33,092 healthy adults across 19 trials were included, while VE estimates were derived from 6,740 participants in a single study. No significant sex-related differences in immunogenicity were observed among adults younger than 65 years; however, among older adults, females exhibited a significantly higher likelihood of seroconversion for all influenza strains compared with males. Consistently, GMRs were higher in older females across all strains, and vaccine efficacy was also greater in older females than in older males. This investigation represents the first meta-analysis integrating both individual participant data and aggregate trial data to specifically evaluate sex differences in the immunogenicity and effectiveness of seasonal influenza vaccines. Although evidence on vaccine efficacy remains limited, the findings provide robust support for sex-based disparities in vaccine-induced immune responses. By leveraging data from phase 3 randomized trials, the study addressed critical gaps in understanding the influence of sex on post-vaccination immunity. While the use of randomized trial data minimizes bias relative to observational designs, the absence of sex-stratified analyses in the original trials limits the inferential strength of randomization [61]. Nevertheless, these results challenge the adequacy of a uniform vaccination strategy and reinforce the growing recognition that vaccine dosing and evaluation should incorporate sex-specific considerations. Accordingly, the systematic collection and reporting of sex-disaggregated vaccination outcomes should be regarded as a research and public health priority (see below) [62].
Thus, growing evidence shows that there are sex-based variations in the immune response to influenza vaccines. However, the molecular mechanisms contributing to these variations remain unclear. Using bioinformatics approaches, Wen et al. [63] analysed the genes that contribute to sex-specific differences in response to influenza vaccination. Their findings revealed that, on day 1 post-vaccination, females exhibited higher expression of genes related to the immune response compared to males, whereas males showed higher expression of genes involved in calmodulin binding, a calcium-dependent regulatory process that controls intracellular signalling pathways essential for immune cell activation. This pattern suggests that females may mount a stronger immediate immune response to seasonal influenza vaccines within the first 24 hours, while males may preferentially engage calcium-mediated signalling mechanisms early after vaccination. However, most of these sex-different genes displayed opposite expression patterns by day 3 or day 14, suggesting that adult males might sustain longer-lasting immunity from the influenza vaccine compared to adult females.
Multiple prospective, randomized, double-blind clinical trials conducted across different countries have demonstrated that adult and older women experience more intense and frequent localized reactions, such as inflammation and pain at the injection site, than their male counterparts [4, 50, 64, 65]. In older populations, the method of vaccine delivery can influence the magnitude of sex-based variations in antibody production [66]. Influenza vaccines are typically given via intramuscular injection, although subcutaneous delivery is also used. In older women, intramuscular administration of the influenza vaccine produced significantly stronger antibody responses against influenza A strains compared to subcutaneous delivery, though no such difference was observed for B strains [50]. This finding underscores the importance of using the intramuscular route in this demographic. Additionally, intramuscular injections were associated with fewer local side effects in women compared to subcutaneous injections, an important consideration, given that influenza vaccines are generally linked to a higher incidence of adverse reactions in females [49].
Hence, adverse effects following vaccination may deter uptake across populations, with women showing higher levels of vaccine hesitancy (see below) [62, 66]. This may be partly due to their typically stronger immune responses, which are associated with a greater frequency of side effects [62]. Biological factors, such as smaller deltoid muscles and thicker subcutaneous fat, may also contribute to the higher incidence of side effects [67]. Moreover, gendered differences in symptom reporting could influence data accuracy: women may be more likely to report discomfort, while men may underreport symptoms [67, 68]. A meta-analysis confirmed that local reactions were more common in females across all ages [65]. In one study, women were three times more likely to report adverse events after influenza vaccination, with age-related declines more pronounced in females [67]. Interestingly, in men, elevated oestrogen levels were found to correlate with IL-6 and manifestations of adverse reactions, an effect not seen in women, highlighting possible sex-specific immune regulation [67].
Regarding adverse events, Guillain-Barré Syndrome (GBS), an acute, immune-mediated polyradiculoneuropathy, has been identified as one of the serious neurological complications after vaccination. Evidence of a significantly increased incidence of GBS following the 1976 swine influenza vaccination campaign in the USA led to a long-standing debate about a possible association between influenza vaccination and GBS. Prior to the COVID-19 pandemic, vaccine-associated GBS was most frequently discussed in relation to influenza vaccines. Other vaccines, aside from influenza vaccines, that were subjects of debate included those for meningococcus, measles/mumps/rubella, and human papillomavirus; however, reported GBS cases following these vaccinations have been very limited, and the speculative nature of this association persists, contributing to ongoing controversy. A recent study using data from the World Health Organization global database of adverse event reports of medicines and vaccines (from 1967 to 2023; total reports n = 131,255,418) investigated vaccine-associated GBS. Among 22,616 reports of all drug-associated GBS identified between 1978 and 2023, 15,377 cases were attributed to vaccines, with 8,072 occurring in males (52.49%). Cumulative reports of vaccine-associated GBS increased steadily over time, with a marked rise following the introduction of COVID-19 vaccines in 2020. Importantly, age- and sex-stratified analyses revealed similar associations between males and females, while the risk increased with advancing age. COVID-19 vaccines showed the lowest association with GBS compared with other vaccines [69]. In a large nationwide retrospective epidemiological study conducted in Denmark between 2002 and 2016, influenza vaccination preceded only 1.5% of all GBS cases, and vaccination was associated with a 1.9-fold transient increase in GBS risk, confined to the first month following immunization [70]. A case-control study conducted between October 2010 and May 2011 across seven Italian Regions estimated an attributable risk from two to five GBS cases per 1,000,000 vaccinations in adults [71]. Overall, evidence indicated that GBS occurs more frequently in males than females and that the incidence increases with age. A systematic review of 63 studies estimated an annual GBS incidence ranging from 1.1 to 1.8 per 100,000 per year, with a clear age-dependent increase [72]. However, the biological basis for the higher incidence observed in males is still unknown. Taken together, data on the association between GBS and seasonal flu vaccination are heterogeneous and vary across influenza seasons. If there is an increased GBS risk following influenza vaccination, it is very small, estimated at approximately one to two additional GBS cases per million doses of influenza vaccine administered [73]. The benefits of influenza vaccination in preventing influenza-related morbidity and mortality far outweigh the potential risk of GBS. These findings underscore the need for interdisciplinary research to improve understanding of sex-based differences in vaccine response and to inform more equitable public health strategies.
Figure 2 illustrates the role of immunological mechanisms, particularly B-cell-mediated processes, in the enhanced antibody response [19] to anti-influenza vaccination (and presumably to influenza infection).
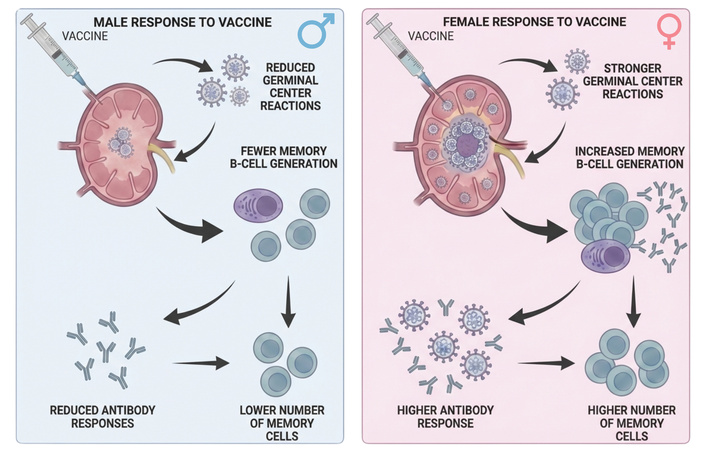
Immunological mechanisms underlying enhanced antibody responses to influenza vaccination. B cell activation, differentiation into antibody-secreting plasma cells, and the generation of memory B cells contribute to female robust humoral responses following anti-influenza vaccination and are likely to play a similar role during natural influenza infection. Created in BioRender. Calabrò, A. (2026) https://BioRender.com/eqazsoy.
Gender, shaped by sociocultural norms and assigned roles, also plays a part in modulating immune responses and intersects with the exposome, the cumulative record of environmental influences from early life onward [74]. Gender influences behaviours and living conditions, including diet quality, educational attainment, and healthcare accessibility, all of which are critical determinants of immune health. For instance, in certain cultural or regional settings, women may encounter limitations in accessing nutrient-rich foods or medical services due to economic disparities or traditional gender expectations. Poor nutritional status, particularly deficiencies in key micronutrients such as zinc and selenium, can impair immune defences and increase susceptibility to infections. Nevertheless, beyond these biological consequences, the most influential gender-linked determinants of anti-influenza immunity are social, occupational, and behavioural in nature. Female household contacts were more likely to become infected, consistent with caregiving roles that increase exposure risk. In addition, women constitute a substantial proportion of healthcare personnel, further increasing gender-linked occupational exposure during the influenza epidemics. All these factors shape patterns of viral exposure, tendencies to seek medical help, utilization of healthcare resources, including vaccination services, and disease reporting behaviours [44, 75]. In conclusion, gendered responses to environmental factors are multifactorial and result in distinct immune modulation and health outcomes, with women generally being more affected, often adversely, by such influences.
Concerning vaccination, gender shapes the ways in which individuals engage with vaccination, and these behaviours are in part influenced by underlying biological sex-related differences in immune responses to vaccines. Worries about potential side effects, or prior negative experiences following immunization, are closely associated with vaccine hesitancy [76], since, as previously discussed, females tend to report a higher frequency and intensity of vaccine-related adverse events, a pattern that aligns with the generally higher levels of vaccine hesitancy observed among women and girls relative to men and boys [77]. In fact, vaccine side effects also appear to interfere more substantially with women’s everyday lives. For example, studies examining adverse reactions to vaccines have shown that women are more likely than men to report disruptions in daily functioning, including sleep disturbances and difficulties in caregiving responsibilities. Women additionally report greater use of analgesics after vaccination and more often plan their vaccination appointments around rest days, anticipating possible side effects [78]. Importantly, higher levels of vaccine hesitancy among women do not necessarily translate into lower vaccination rates, since in many settings, women demonstrate higher vaccine acceptance than men, although this pattern varies across populations. For instance, lower uptake has been reported among women belonging to certain racial and ethnic minority groups. Reduced vaccination in specific groups may reflect disparities in access to healthcare, missed vaccination opportunities, attitudes and beliefs about vaccines, limited trust in institutions, and broader socioeconomic constraints. These factors may also differ between high-income and low- and middle-income countries, where variations in healthcare systems, educational attainment, poverty, cultural norms, and historical as well as current public health policies can differentially shape vaccination behaviours [79].
Figure 3 illustrates the role of gender in shaping immune responses to influenza infection and anti-influenza vaccination.
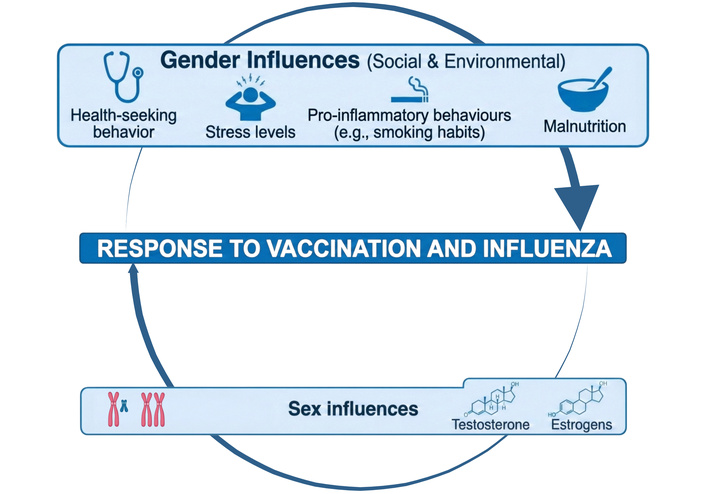
Role of gender-related determinants in shaping immune responses to influenza infection and vaccination. Gender-associated factors modulate susceptibility to influenza, disease outcomes, and vaccine responsiveness. For example, women represent a large proportion of the healthcare workforce and the vast majority of informal caregivers, which may increase exposure and susceptibility to infection; however, female greater attention to personal health and preventive practices, which can facilitate disease control and promote vaccine uptake [6, 7]. Other gender-related determinants, such as malnutrition and pro-inflammatory lifestyle patterns, act more directly on the immune system, impairing responses to both influenza infection and vaccination [80]. Together, these determinants, along with immunological pathways, contribute to inter-individual variability in host responses to natural infection and anti-influenza vaccination. Created in BioRender. Calabrò, A. (2026) https://BioRender.com/n3oo0y4.
This review provides compelling evidence that males and females display markedly different immunological responses to both influenza infection and vaccination. Recognizing such sex-specific disparities is highly significant, as they may affect the trajectory of disease, the VE, and the frequency or severity of side effects. Gaining deeper insight into these variations is crucial for informing the design of more personalized, efficient, and fair approaches to prevention and therapy, ultimately contributing to improved health outcomes across diverse demographic groups. Importantly, sex-specific immune and inflammatory profiles also suggest that therapeutic responses to antiviral and immunomodulatory treatments may differ between men and women, raising the possibility that sex-informed treatment strategies could optimize clinical efficacy while minimizing adverse effects.
Future research should prioritize a more detailed characterization of sex-specific immune mechanisms and address current limitations of influenza vaccines, including suboptimal immunogenicity in males and a higher burden of adverse reactions in females. One major obstacle to accurately defining the relationship between vaccination and side effects in women stems from persistent methodological biases. Women have historically been underrepresented in drug and vaccine trials, often due to concerns regarding hormonal fluctuations or potential risks during pregnancy. Consequently, much of the available evidence is derived from studies conducted largely in male populations, limiting its generalizability. In addition to biological and genetic determinants, gender-related factors, such as behavioural, social, and environmental influences, should therefore be systematically incorporated into the evaluation of immune-based interventions. There is a clear need for transparent reporting, explicit recognition of sex- and gender-specific effects, and the integration of basic, clinical, and behavioural research to guide optimal vaccine development and deployment. Incorporating sex and gender considerations into vaccine design, clinical recommendations, and public health implementation represents a concrete step toward precision medicine. While vaccines have traditionally been developed under a “one-size-fits-all” paradigm, accumulating evidence supports the feasibility and necessity of a more nuanced approach. Accounting for sex and gender in both vaccination and treatment strategies has strong potential to enhance therapeutic effectiveness, reduce adverse events, and improve overall health outcomes.
COVID-19: coronavirus disease 2019
DCs: dendritic cells
ER: oestrogen receptor
GBS: Guillain-Barré Syndrome
GMRs: geometric mean ratios
HA: haemagglutinin
IAVs: influenza A viruses
IBVs: influenza B viruses
IFN: interferon
IL: interleukin
NA: neuraminidase
RST: repeated cycles of restraint
SARS-CoV-2: severe acute respiratory syndrome coronavirus-2
SPR: seroprotection rate
SV: split virus
Th: T helper
TLR: toll-like receptor
Treg: T regulatory
VE: vaccine effectiveness
WIV: whole inactivated virus
XCI: X chromosome inactivation
Xi: inactive X
AC: Investigation, Writing—original draft, Writing—review & editing. AA: Writing—review & editing. EM: Writing—review & editing. GP: Writing—review & editing. CMT: Conceptualization, Investigation, Writing—original draft, Writing—review & editing, Supervision. CC: Conceptualization, Investigation, Writing—original draft, Writing—review & editing, Supervision. All authors read and approved the submitted version.
Calogero Caruso, who is the Editor-in-Chief of Exploration of Immunology, had no involvement in the decision-making or review process of this manuscript. Emanuele Montomoli is the founder and Chief Scientific Officer of VisMederi srl. The other authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
This research received no external funding.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1633
Download: 29
Times Cited: 0
