Affiliation:
1Department of Translational Biomedicine and Neuroscience – DiBraiN, University of Bari “Aldo Moro”, 70124 Bari, Italy
2MIRROR-Medical Institute of Regeneration and Repairing and Organ Replacement, Interdepartmental Center, University of Bari “Aldo Moro”, 70124 Bari, Italy
3School of Dentistry, University of Dundee, DD1 4HN Dundee, United Kingdom
4Department of Health and Life Sciences, De Montfort University, LE1 9BH Leicester, United Kingdom
Email: marco.tatullo@uniba.it
ORCID: https://orcid.org/0000-0001-7340-0708
Affiliation:
5Department of Oral and Maxillo-Facial Science, University of Rome “Sapienza”, 00195 Rome, Italy
ORCID: https://orcid.org/0000-0001-9234-5636
Affiliation:
6Stem Cells and Medical Genetics Units, Biomedical Section, Tecnologica Research Institute and Marrelli Health, 88900 Crotone, Italy
ORCID: https://orcid.org/0009-0005-3009-9237
Affiliation:
7Department of Diagnostics and Public Health, University of Verona, 37100 Verona, Italy
ORCID: https://orcid.org/0009-0006-0015-3922
Affiliation:
8Department of Mechanics, Mathematics and Management, Polytechnic University of Bari, 70125 Bari, Italy
ORCID: https://orcid.org/0000-0003-0442-1964
Affiliation:
6Stem Cells and Medical Genetics Units, Biomedical Section, Tecnologica Research Institute and Marrelli Health, 88900 Crotone, Italy
ORCID: https://orcid.org/0009-0003-5652-2502
Affiliation:
6Stem Cells and Medical Genetics Units, Biomedical Section, Tecnologica Research Institute and Marrelli Health, 88900 Crotone, Italy
ORCID: https://orcid.org/0000-0003-0916-8101
Explor Immunol. 2026;6:1003242 DOI: https://doi.org/10.37349/ei.2026.1003242
Received: November 19, 2025 Accepted: March 18, 2026 Published: March 29, 2026
Academic Editor: Wantong Song, Chinese Academy of Sciences, China
The article belongs to the special issue Immuno-Materials: at the interdisciplinary of immunology and materials
Immuno-materials, which represent a confluence of immunology and materials science, are dramatically transforming the fields of healthcare. Through the targeted modulation of immune responses, these innovative materials offer promising avenues for advancements in vaccine development, immunotherapy, tissue engineering, and diagnostics. This review examines the fundamental principles of immuno-materials, elucidating key concepts and methodologies pertinent to their development. We investigate strategies for engineering materials that can elicit customized immune responses, capable of both stimulation and suppression, and we discuss their potential roles as adjuvants, carriers, or scaffolds. Furthermore, we address the challenges and opportunities associated with the clinical translation of these materials, emphasizing the critical importance of biocompatibility, safety, and efficacy. This review aims to critically evaluate and contextualize the transformative potential of immuno-materials, mapping their journey from laboratory design to proven clinical applications in enhancing human health.
The host immune system serves as a sophisticated surveillance network, evolved to distinguish “self” from “non-self” through the recognition of highly conserved molecular motifs. Central to this process is the detection of pathogen-associated molecular patterns (PAMPs)—derived from exogenous microorganisms—and damage-associated molecular patterns (DAMPs), which are endogenous signals released during cellular stress or tissue injury [1]. These motifs are recognized by a specialized repertoire of pattern recognition receptors (PRRs), such as toll-like receptors (TLRs), primarily expressed on innate immune cells, including macrophages and dendritic cells. This system is crucial for maintaining human health and combating diseases, as it is closely linked to the pathogenesis of various conditions, including cancer, autoimmune diseases, and inflammatory disorders [2].
There are two major arms of the human immune system: the innate immune system and the adaptive immune system. The innate immune system serves as the primary line of defence, responding rapidly and non-specifically. This initial response involves inflammation and the recruitment of innate effector cells, including macrophages and neutrophils; the latter act to eliminate pathogens and foreign bodies through specialized mechanisms such as phagocytosis, degranulation, and the release of antimicrobial enzymes. The adaptive immune system comes into play when the innate response is insufficient, responding specifically to antigens and developing long-term immune memory. Upon implantation, biomaterials are not inert bystanders; they are immediately recognized as foreign entities, often triggering a cascade of DAMP-mediated signaling that shapes the subsequent foreign body response (FBR). Extensive research has demonstrated that these interactions can be modulated through engineered strategies designed to mitigate chronic inflammation and promote integration [3]. The activation of specific PRRs is highly sensitive to the material’s physicochemical profile. For instance, hydrophobic surfaces often lead to the adsorption of proteins in denatured states, exposing hydrophobic patches that act as DAMPs, subsequently triggering TLR4 signaling. In contrast, the NLRP3 inflammasome is primarily sensitive to the internalization of particulate or crystalline immuno-materials, where lysosomal rupture and the release of cathepsins serve as the secondary signal for activation [4]. Emerging evidence also suggests that substrate stiffness influences the cGAS-STING pathway; extreme mechanical strain or confined migration on stiff matrices can compromise nuclear integrity, leading to the leakage of genomic DNA into the cytoplasm and the initiation of a type I interferon response [5].
In the rapidly evolving landscape of biomedical research, the field of immuno-materials has emerged as a highly promising area of research, merging materials science with immunology to develop novel strategies for immune modulation [6, 7]. These materials represent a sophisticated convergence of design and functionality, enabling the controlled regulation of immune cell behavior in both in vitro and in vivo contexts, with transformative potential for cancer immunotherapy [8], vaccine development, and regenerative medicine [9].
By leveraging the intricate understanding of how PRRs detect precisely patterned structures, researchers are developing materials that selectively target and modulate immune pathways (Figure 1). Notable initiatives, such as those at the Wyss Institute, focus on scaffolds designed to attract and retain condition-specific T cells, demonstrating the clinical potential of this interdisciplinary field [10]. This perspective provides a comprehensive analysis of the current state of research on immuno-materials, elucidating their diverse applications across medicine, biotechnology, and materials science. By synthesizing primary findings and identifying critical literature gaps, the present review establishes a strategic roadmap for the development of next-generation immunomodulatory platforms, offering a foundational framework for optimizing these materials for both therapeutic and diagnostic purposes.
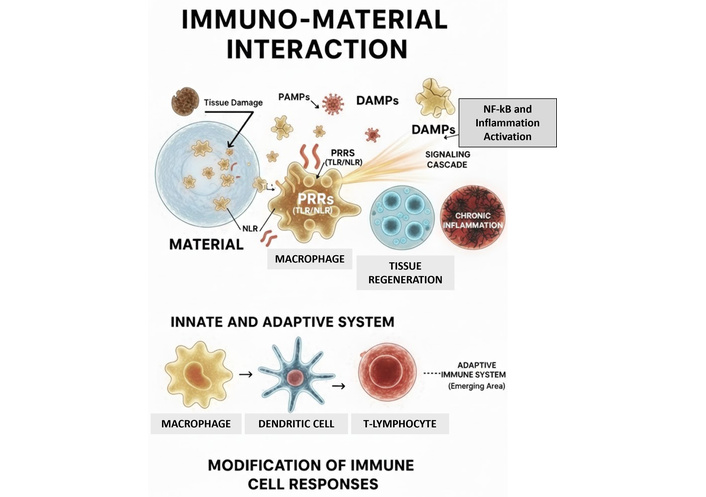
Conceptual schematic of the immuno-material interface. The interaction begins with the adsorption of proteins and the release of damage-associated molecular patterns (DAMPs) from damaged tissue and cell stress. These signals, along with potential pathogen-associated molecular patterns (PAMPs), are recognized by pattern recognition receptors (PRRs) such as toll-like receptors (TLRs) on innate cells (macrophages, dendritic cells). This triggers a signaling cascade leading to either tissue regeneration or chronic inflammation [foreign body response (FBR)]. The involvement of the adaptive system (T-lymphocytes) is highlighted as an emerging area of research in biomaterial science.
Advancements in biomaterials for targeted immune modulation represent a rapidly evolving field with profound implications for regenerative medicine and immunotherapy [11].
Among key physicochemical parameters, surface wettability emerges as a critical property that dictates the biological performance of the bio-interface. Rather than mere hydrophilicity, wettability governs the thermodynamics of the protein corona formation—the spontaneous adsorption of host proteins onto the material surface upon implantation. The configuration and density of this protein layer are essential in modulating the initial immune recognition. For instance, surfaces with optimized wettability promote the adsorption of specific proteins that steer macrophage polarization toward an anti-inflammatory (M2) phenotype, which is vital for minimizing fibrosis and supporting constructive tissue remodeling. Conversely, extreme hydrophobic states can lead to protein denaturation, triggering a pro-inflammatory response and compromising the scaffold’s long-term integration. Therefore, engineering surface wettability is not merely a structural choice but a fundamental strategy to control targeted chemoattraction, cytokine release, and overall functional tissue integration.
In this context, the bio-interface is not merely a physical boundary but a dynamic regulator of immune cell behavior. Upon implantation, surface properties dictate the initial protein adsorption patterns—often referred to as the “Vroman effect”—which subsequently govern cell adhesion. Specifically, surface topography and stiffness serve as critical mechanical cues. As demonstrated by Sridharan et al. (2019) [12], high substrate stiffness promotes a pro-inflammatory M1-like polarization by enhancing focal adhesion formation and activating mechanosensitive signaling pathways. Conversely, McWhorter et al. (2013) [13] established that specific surface geometries, such as micro-grooves that induce cell elongation, can actively drive an anti-inflammatory M2 phenotype, even in the absence of biochemical polarization factors. These mechanical signals are integrated and relayed to the nucleus via the translocation of the YAP/TAZ transcriptional co-activators, as characterized by Dupont et al. (2011) [14], thereby modulating the expression of immuno-responsive genes.
An innovative strategy in the development of immuno-materials involves the design of biomimetic materials that replicate the properties of natural immune cells and tissues. Biomimetic materials are engineered to replicate the intricate tissue structure and the native microenvironment (Figure 2). By mimicking the topographical cues and mechanical stiffness of the extracellular matrix (ECM), these scaffolds provide the necessary spatial organization to guide multicellular behavior and facilitate functional tissue regeneration. These engineered biomaterials can be designed to interact with the immune system in highly specific manners, such as selectively activating or inhibiting immune cells. By meticulously tailoring both the physical and chemical properties of these materials, researchers can create platforms for precise immune modulation, leading to more effective therapeutic outcomes [15]. A variety of advanced techniques have been employed to achieve this tailored immune response. For instance, strategies such as surface functionalization, controlled release mechanisms, and the incorporation of bioactive molecules enable researchers to finely tune the interaction between biomaterials and immune cells.
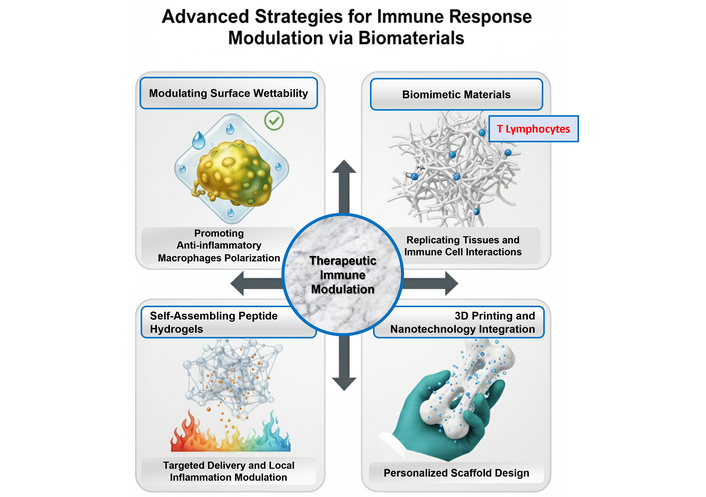
Immune response modulating strategies through biomaterial. This figure illustrates current integrated techniques for modulating the host immune response through tailored biomaterial design. Modulating Surface Wettability (top left): Manipulation of surface energy and hydration layers to influence the initial protein corona formation. Optimizing wettability promotes the polarization of macrophages toward an anti-inflammatory M2 phenotype. Biomimetic Materials (top right): Scaffolds engineered to replicate complex 3D tissue structures and mechanical cues. These architectures facilitate coordinated interactions between the material and immune cell populations, including the involvement of T-lymphocytes in the long-term integration process. Self-Assembling Peptide Hydrogels (bottom left): Nanostructured systems designed for targeted delivery and local modulation of the inflammatory environment, minimizing systemic exposure. 3D Printing and Nanotechnology Integration (bottom right): Convergence of additive manufacturing and nanoscale precision to create personalized, custom-designed scaffolds for optimized clinical outcomes.
Emerging technologies, including 3D printing and nanotechnology, further facilitate the design of complex structures that improve the efficacy of immuno-materials. These technologies enable the creation of highly customized biomaterial scaffolds that can mimic the complex architecture of native tissue, providing not only mechanical support but also biochemical cues that promote favorable immune responses.
Moreover, the integration of biomaterials with cellular therapies, such as stem cells or engineered immune cells, offers an innovative avenue for modulating immune responses. This combination transforms biomaterials from a simple structure into dynamic platforms capable of actively engaging with the immune system [16].
The properties, applications, and current translational status of the main types of these immuno-materials are summarized in Table 1.
Comparison of key properties, applications, and clinical translation status of major classes of immuno-materials.
| Type of immuno-materials | Keyproperties | Main applications | Advantages | Current limits | Clinical translation stage |
|---|---|---|---|---|---|
| Peptide-based hydrogel | Self-assembly, biocompatibility, controlled release | Immunotherapy, tissue engineering | High specificity, localized release, biocompatibility | Understanding long-term mechanisms | Mainly pre-clinical (some in Phase I/II trials) |
| Biomimetic biomaterials | Replicate structure, specific interaction | Immune modulators, scaffolds | Precise cell-immune targeting | Complexity of synthesis and control | Pre-clinical/Early clinical research |
| Functionalized nanoparticles | Modified surfaces, molecular targeting | Vaccines, therapeutic delivery | Modulatory, stability, and immune efficacy | Potential toxicities and rapid clearance | Clinical use (e.g., mRNA vaccines) & Phase II/III |
Hydrogels represent a promising category of biomaterials, demonstrating significant potential for immunomodulatory applications in tissue engineering and regenerative medicine. Their unique physicochemical properties render them particularly suitable for these fields. Notably, hydrogels are characterized by their high-water content, which is crucial for creating a conducive microenvironment that supports cell survival, proliferation, and function [17]. This hydration facilitates nutrient diffusion and waste removal, critical factors for maintaining cellular homeostasis [18]. Additionally, the biocompatibility of hydrogels ensures seamless integration with surrounding tissues, minimizing the risk of adverse immune reactions. Their inherent biodegradability allows for gradual resorption, enabling the replacement of the hydrogel matrix with natural tissue over time. This property is crucial for long-term tissue engineering applications, as it promotes natural healing processes without leaving permanent foreign materials. Moreover, hydrogels possess remarkable drug-loading capacity, which enables the controlled release of therapeutic agents, growth factors (GFs), or anti-inflammatory molecules. This capability enhances their utility in modulating immune responses and promoting tissue regeneration by allowing for sustained delivery of bioactive substances, tailored to meet the specific temporal and spatial needs of the target tissue [19].
The ease of fabrication and manipulation of hydrogels offers significant advantages in customizing their composition, structure, and functionality. This versatility allows researchers and clinicians to tailor hydrogels to specific therapeutic requirements, optimizing their performance for particular applications. Another notable advantage of hydrogels is their potential for minimally invasive delivery. They can be injected into the body, significantly reducing the need for traditional surgical procedures. This feature not only minimizes the risk of complications but also mitigates unwanted inflammatory responses from the host [20]. Such characteristics are especially beneficial in delicate applications, such as joint repair or wound healing, where conventional surgical interventions might exacerbate inflammation or damage surrounding tissues [21]. Recent breakthroughs have demonstrated the potential of integrating bioactive cues with sophisticated delivery systems. For instance, MOF-modified multifunctional scaffolds incorporating exosomes have shown promise in accelerating vascularized bone regeneration [22]. Furthermore, the development of microenvironment self-adaptive hydrogels represents a significant step forward in post-surgical therapy, allowing for on-demand immunomodulation and tissue repair [23, 24]. The precision offered by 3D-bioprinting of anisotropic bicellular hydrogels further enables the complex reconstruction of biological interfaces, such as the osteochondral boundary [20].
In this context, peptide-based hydrogel biomaterials have emerged as a promising strategy for modulating the immune system. These supramolecular materials exhibit the ability to self-assemble into various nanostructures through an intricate network of interactive forces, including hydrogen bonding, van der Waals interactions, and hydrophobic interactions. This dynamic self-assembly not only allows for the formation of stable structures but also enables these materials to respond to microenvironmental stimuli, such as pH and temperature. Such responsiveness can be strategically harnessed to achieve targeted therapeutic effects, particularly in the realm of immunotherapy. Recent research has highlighted the versatility of peptide-based hydrogels across numerous biomedical applications, with a particular focus on enhancing the efficacy of immunotherapy and cancer treatment. Their distinctive characteristics enable them to serve as effective delivery systems for immunomodulatory agents, ensuring sustained release and localized action at tumor sites or areas of inflammation. This localized delivery is critical in minimizing systemic side effects and maximizing therapeutic impact [25]. By incorporating specific peptides designed to interact with immune cells, these hydrogels can facilitate the recruitment and activation of T cells, dendritic cells, and other crucial components of the immune system. This targeted engagement amplifies the body’s natural immune response, potentially leading to improved outcomes in cancer immunotherapy [26]. To bridge the gap between material design and clinical reality, Table 1 highlights several platforms that have successfully transitioned to the bedside. For instance, PuraStat®, a self-assembling peptide hydrogel based on RADA16, is now widely utilized in surgical procedures to provide immediate hemostasis and a pro-regenerative environment without the risk of thermal damage to surrounding tissues [27]. In the realm of chronic immune modulation, glatiramer acetate serves as a hallmark of biomimetic success; by mimicking myelin basic protein, it effectively re-educates the host immune system to mitigate the progression of multiple sclerosis [28, 29]. Finally, the rapid global deployment of mRNA-carrying lipid nanoparticles has underscored the transformative power of functionalized nanoparticles, though it has also emphasized the need to monitor and mitigate side effects such as PEG-induced hypersensitivity to ensure long-term clinical safety [30].
Vaccines have historically been a pivotal strategy in the prevention and management of infectious diseases. Traditional vaccine development techniques, primarily based on weakened or inactivated forms of pathogens, often face significant challenges related to efficacy, safety, and adaptability to emerging infectious threats. In recent years, there has been a growing interest in the utilization of immune materials for vaccine development, offering innovative strategies that have the potential to enhance both the effectiveness and safety profiles of vaccines. One of the most promising aspects of immuno-materials is their ability to mimic the structural and biochemical properties of pathogens. By engineering materials that closely resemble the morphology, surface characteristics, and molecular signatures of viruses and bacteria, researchers can induce a more robust and targeted immune response [31]. This biomimetic approach enhances the recognition of the vaccine by the immune system and is particularly advantageous for addressing pathogens that are challenging to detect or that have developed resistance to conventional vaccines. By activating the innate features of these pathogens, immuno-materials can effectively prime the immune system, leading to enhanced and more durable protection. This novel approach not only addresses the current limitations in vaccine efficacy but also paves the way for more adaptable and resilient vaccination strategies in the face of emerging infectious threats.
Additionally, immuno-materials can function as adjuvants, substances that enhance the body’s immune response to an antigen. The incorporation of specific immuno-materials into vaccine formulations has the potential to stimulate more vigorous antibody production. This could result in a notable reduction in the number of doses required for effective immunization, while also providing longer-lasting protection [32]. Such adaptability is crucial in the context of emerging infectious diseases, where rapid and robust immune responses are essential. Moreover, the versatility of immuno-materials is also reflected in the methods of vaccine administration. By tailoring delivery systems to align with the specific requirements of each vaccine and its target population, researchers can optimize vaccination strategies. For example, immuno-materials can facilitate the creation of vaccines suitable for mucosal administration, such as intranasal and oral routes. This approach not only increases convenience for patients but may also enhance efficacy for specific diseases by eliciting local immune responses [33].
Furthermore, the utilization of immuno-materials can effectively address logistical challenges associated with vaccine storage and distribution. By engineering materials that maintain stability under a wide range of environmental conditions, the accessibility of vaccines can be significantly improved, particularly in remote or resource-limited regions. This stability ensures the continued efficacy of vaccines while extending their reach to underserved populations [34].
In summary, the incorporation of immuno-materials into vaccine development represents a transformative shift in the field of immunization. By leveraging the distinctive properties of these materials, researchers can develop more efficacious, safer, and adaptable vaccines that meet the challenges posed by modern infectious diseases.
The field of tissue engineering and regenerative medicine is experiencing a transformative shift, largely propelled by the advent of immuno-materials. These cutting-edge biomaterials possess unique immunomodulatory properties that enable them to interact dynamically with the immune system at the site of injury or implantation. By harnessing the biological capabilities of immuno-materials, researchers are developing advanced therapeutic strategies that not only promote tissue regeneration but also mitigate inflammatory responses, thereby facilitating optimal integration of engineered tissues with the host environment [35].
The primary objective of tissue engineering is the development of scaffolds that provide structural support while simultaneously facilitating the regeneration of functional tissue. In the field of regenerative medicine, the utilization of biocompatible and biodegradable biomaterials is essential for effective tissue replacement, injury repair, and targeted drug delivery. Historically, traditional biomaterials have primarily focused on mechanical properties and biocompatibility; however, the incorporation of immunomodulatory functions represents a significant paradigm shift in this field. Immuno-materials have the potential to actively influence the immune response, a critical factor during both the acute and chronic phases of tissue regeneration. By modulating the activity of immune cells and their secreted factors, these materials can direct the healing process towards more favorable outcomes. One of the main challenges in employing these materials lies in the management of inflammation. While inflammation is an essential component of initial wound healing, its persistence can lead to detrimental effects, including delayed healing, biomaterial rejection, additional damage, and fibrosis [36]. Therefore, the careful selection of biomaterials is crucial to minimize inflammation and optimize tissue regeneration. This process necessitates a comprehensive understanding of how the physicochemical properties of biomaterials, such as surface topography, chemical composition, and mechanical properties, influence their interactions with the immune system. A significant advantage of immuno-materials is their ability to mitigate chronic inflammation, which poses a substantial barrier to effective tissue regeneration [37]. Chronic inflammation, often manifesting as a persistent FBR characterized by the sustained activation of macrophages and the formation of a fibrous capsule, represents a significant barrier to the long-term functionality of implanted immuno-materials. Immuno-materials can be strategically designed to release bioactive molecules or express surface markers that selectively recruit anti-inflammatory immune cells [38]. To move beyond theoretical frameworks, it is essential to highlight specific bioactive cues that have demonstrated efficacy in modulating the immune microenvironment. For instance, the localized delivery of anti-inflammatory cytokines such as IL-10 and TGF-β1 via heparin-based hydrogels has shown significant potential in suppressing the M1 pro-inflammatory response and promoting tissue integration. In the context of bone regeneration, the synergistic release of bone morphogenetic protein-2 (BMP-2) and vascular endothelial GF (VEGF) from biomimetic scaffolds not only promotes osteogenesis but also modulates the early immune infiltration to prevent fibrous encapsulation. Furthermore, recent studies have pivoted towards the use of extracellular vesicles (EVs) and exosomes as “bio-active packages”; these nanostructures, when tethered to collagen scaffolds, deliver a complex cocktail of miRNAs and proteins that actively resolve inflammation in chronic wound models, offering a superior safety profile compared to recombinant GFs [39, 40].
Moreover, the design of immuno-materials can be tailored to enhance their interaction with the immune system over time. By meticulously regulating the timing and dosage of bioactive molecule release, these materials can effectively mimic the natural healing process, providing sustained support for tissue regeneration. This temporal regulation facilitates a gradual transition from the initial inflammatory phase to a reparative phase that encourages tissue formation. The capacity of immuno-materials to reduce chronic inflammation and foster a favorable immune milieu not only optimizes the integration of engineered tissues with the host but also enhances the overall effectiveness of regenerative therapies. The incorporation of immunological cues into biomaterials promotes host cell attraction and proliferation, thereby facilitating improved vascularization and better incorporation of the engineered tissue into the surrounding environment [25]. This capability is particularly relevant in applications such as orthopaedic implants, where seamless integration with the surrounding bone and soft tissue is essential for both functionality and longevity. Successful integration minimizes the risk of implant rejection and ensures that the surrounding tissue can provide adequate support for the mechanical loads placed upon the implant. The interplay between the implant and the biological environment is critical; thus, enhancing this interaction through advanced biomaterials is a key focus in orthopaedic research. Recent advances in biomaterials science have also highlighted the potential of combining multiple immunomodulatory strategies within a single scaffold. For example, the simultaneous incorporation of anti-inflammatory cytokines, chemokines, and ECM components can lead to the development of a multifunctional platform. This innovative approach not only provides a robust substrate for cell attachment and proliferation but also actively orchestrates the immune response, thereby promoting regeneration [25]. This multidimensional strategy reflects the intrinsic complexity of biological systems and encourages the implementation of a more holistic strategy in the field of tissue engineering.
In summary, the incorporation of immuno-materials in tissue engineering represents a significant advancement in the field of regenerative medicine. By enhancing scaffolds and implants with immunomodulatory properties, researchers are addressing the limitations of traditional biomaterials while simultaneously identifying new avenues for enhanced tissue regeneration. This innovative approach has the potential to facilitate the development of more effective and biocompatible solutions to a wide range of clinical challenges, paving the way for future breakthroughs in restoring tissue function and improving patient outcomes [41]. As research in this area advances, the development of immuno-materials may emerge as a cornerstone of regenerative medicine, potentially transforming therapeutic practices and enhancing the efficacy of tissue repair and regeneration interventions.
In situ tissue engineering using bioresorbable scaffolds is emerging as a transformative therapeutic approach aimed at generating functional human tissues. By harnessing the patient’s intrinsic immune response, these scaffolds provide not only structural integrity but also actively guide de novo tissue formation. This strategy holds particular promise for cardiovascular applications, such as the development of bio-synthetic blood vessels and heart valves, and is progressively advancing toward clinical implementation (Table 2).
Representative clinical applications of immuno-materials.
| Clinical trial ID | Study title | Material category | Medical condition | Study status | Results available |
|---|---|---|---|---|---|
| NCT02718625 | Study of SANTYL® vs Hydrogel (SoloSite®) for Pressure Ulcers | Peptide Based Hydrogel | Pressure Ulcers | Terminated | Yes |
| NCT01350102 | The Relationship of Hemoglobin A1c and Diabetic Wound Healing | Peptide Based Hydrogel | Diabetic Wound Healing | Terminated | Yes |
| NCT06561789 | Remineralization Effect of βTricalcium Phosphate Nanoparticles Versus FunctionalizedβTricalcium Phosphate Nanoparticles | Functionalized Nanoparticles | Remineralization | Completed | No |
| NCT04277377 | Nanoparticle for DSA Removal | Functionalized Nanoparticles | DSA Removal | Active, not recruiting | No |
| NCT04892173 | JNJ-90301900 (NBTXR3) Activated by Radiotherapy With or Without Cetuximab in LA-HNSCC | Functionalized Nanoparticles | LA-HNSCC | Recruiting | No |
| NCT01246336 | Exploratory Study Using Nanotechnology to Detect Biomarkers of Parkinson’s Disease From Exhaled Breath | Functionalized Nanoparticles | Parkinson’s Disease | Completed | No |
| NCT05001282 | A Study to Evaluate ELU001 in Patients with Solid Tumors That Overexpress Folate Receptor Alpha (FRα) | Functionalized Nanoparticles | Solid Tumors (FR overexpressing) | Terminated | No |
| NCT05940701 | The Effectiveness of Biomimetic Materials in Preventing Enamel Demineralization During Fixed Orthodontic Treatment | Biomimetic Materials | Enamel Demineralization | Unknown status | No |
| NCT06717230 | Digital, Conventional, And Biomimetic Approaches for Restoring Endodontically Treated Teeth | Biomimetic Materials | Endodontically Treated Teeth | Recruiting | No |
| NCT06473545 | Retrospective Evaluation of a Novel Biomimetic Synthetic Bone Substitute (Creos® Syntogain) | Biomimetic Materials | Bone Substitute | Completed | No |
| NCT01305356 | Augment® Injectable Bone Graft Compared to Autologous Bone Graft as a Bone Regeneration Device in Hindfoot Fusions | Biomimetic Materials | Hindfoot Fusions | Completed | Yes |
| NCT04328051 | Influence of the Implant-prosthetic Connection in the Marginal Bone Loss and Bacterial Leakage | Biomimetic Materials | Marginal Bone Loss and Bacterial Leakage | Unknown status | No |
| NCT03328884 | Evaluation of the Efficacy and Safety of Nal-IRI for Progressing Brain Metastases in Breast Cancer Patients (Phenomenal) | Biomimetic Materials | Brain Metastases in Breast Cancer Patients | Completed | No |
| NCT00389428 | Multicenter Study of CPX-351(Cytarabine: Daunorubicin) Liposome Injection in Patients With Advanced Hematologic Cancer | Biomimetic Materials | Advanced Hematologic Cancer | Completed | No |
| NCT07302828 | Effect of Double Nutri® Liposomal Encapsulation Technology on Human Absorption of Vitamin C and Glutathione Liquid Sachet | Biomimetic Materials | Human Absorption of Vitamin C and Glutathione | Completed | Yes |
| NCT01631110 | Re-licensing Study to Assess Inflexal V Formulated With WHO Recommended Influenza Strains | Biomimetic Materials | Influenza | Completed | Yes |
| NCT01893177 | Re-licensing Study to Assess Inflexal V Formulated With WHO Recommended Influenza Strains (2013-14) | Biomimetic Materials | Influenza | Completed | No |
| NCT03511820 | A Post Marketing Surveillance Study of Lipo-AB® (Amphotericin B) in Neutropenic Patients With Persistent Fever | Biomimetic Materials | Neutropenic Patients With Persistent Fever | Completed | No |
| NCT00697944 | Ambisome® Preemptive Treatment of Multiple Candida Colonization in Sepsis Patients (AMBIDEX) | Biomimetic Materials | Multiple Candida Colonization in Sepsis Patients | Unknown status | No |
| NCT00361842 | Multicenter Study Of CPX-1 (Irinotecan HCl: Floxuridine) Liposome Injection In Patients With Advanced Colorectal Cancer | Biomimetic Materials | Advanced Colorectal Cancer | Completed | Yes |
| NCT00507962 | Cisplatin HAI Study in Patients With Advanced Cancer and Dominant Liver Involvement | Biomimetic Materials | Advanced Cancer and Dominant Liver Involvement | Completed | No |
| NCT01083979 | Intravesical Liposomes for Ulcerative Cystitis | Biomimetic Materials | Ulcerative Cystitis | Completed | Yes |
| NCT01731470 | Intravesical Liposomes for Interstitial Cystitis/Painful Bladder Syndrome (IC/PBS) | Biomimetic Materials | Interstitial Cystitis/Painful Bladder Syndrome (IC/PBS) | Completed | Yes |
| NCT01333423 | Maximum Tolerated Dose (MTD) of Liposomal Doxorubicin in Combination With Seliciclib for Patients With Metastatic Triple Negative Breast Cancer (TNBC) | Biomimetic Materials | Metastatic Triple Negative Breast Cancer (TNBC) | Withdrawn | No |
| NCT03629171 | Liposome-encapsulated Daunorubicin-Cytarabine and Venetoclax in Treating Participants With Relapsed, Refractory or Untreated Acute Myeloid Leukemia | Biomimetic Materials | Relapsed, Refractory or Untreated Acute Myeloid Leukemia | Recruiting | No |
| NCT04526288 | CPX-351 Versus Immediate Stem Cell Transplantation for the Treatment of High-Grade Myeloid Cancers With Measurable Residual Disease | Biomimetic Materials | High-Grade Myeloid Cancers With Measurable Residual Disease | Terminated | Yes |
| NCT05627232 | Tazemetostat and Palbociclib With CPX-351 for R/R AML | Biomimetic Materials | R/R AML | Recruiting | No |
| NCT04195945 | CPX-351 or CLAG-M Regimen for the Treatment of Acute Myeloid Leukemia or Other High-Grade Myeloid Neoplasms in Medically Less-Fit Patients | Biomimetic Materials | Acute Myeloid Leukemia or Other High-Grade Myeloid Neoplasms | Active, not recruiting | No |
| NCT00002147 | Use of Stealth Liposomal Doxorubicin HCl (DOX-SL) in the Treatment of Moderate to Severe AIDS-Related Kaposi’s Sarcoma. | Biomimetic Materials | Moderate to Severe AIDS-Related Kaposi’s Sarcoma | Completed | No |
| NCT00002313 | A Study of Nystatin in HIV-Infected Patients | Biomimetic Materials | AIDS-Related Kaposi’s Sarcoma | Completed | No |
| NCT03878199 | Testing the Effect of Taking Ruxolitinib and CPX-351 in Combination for the Treatment of Advanced Phase Myeloproliferative Neoplasms | Biomimetic Materials | Advanced Phase Myeloproliferative Neoplasms | Completed | No |
| NCT00002318 | A Comparison of DOX-SL Versus Adriamycin Plus Bleomycin Plus Vincristine in the Treatment of Severe AIDS-Related Kaposi’s Sarcoma | Biomimetic Materials | Severe AIDS-Related Kaposi’s Sarcoma | Unknown status | No |
| NCT00002105 | Randomized, Comparative Trial of DOX-SL (Stealth Liposomal Doxorubicin Hydrochloride) Versus Bleomycin and Vincristine in the Treatment of AIDS-Related Kaposi’s Sarcoma | Biomimetic Materials | AIDS-Related Kaposi’s Sarcoma | Completed | No |
| NCT00001059 | Comparison of Liposomal Doxorubicin Used Alone or in Combination With Bleomycin Plus Vincristine in the Treatment of Kaposi’s Sarcoma in Patients With AIDS | Biomimetic Materials | Kaposi’s Sarcoma in Patients With AIDS | Completed | No |
| NCT04933331 | Early Caries Lesion Management Observational Study (ELMO) | Biomimetic Materials | Early Caries Lesion | Unknown status | No |
| NCT00761644 | Doxil, Bevacizumab and Temsirolimus Trial | Biomimetic Materials | Not in source | Completed | No |
A critical factor influencing the efficacy of these clinical therapies is the inter-patient variability, encompassing genetic, biological, and physiological profiles, which can significantly dictate tissue regeneration outcomes. This heterogeneity underscores the necessity for precision engineering of scaffolds, allowing the optimization of tissue formation tailored to individual immunological landscapes. To achieve this, current research prioritizes the integration of advanced biomaterials that mimic the natural ECM while modulating the immune response in a controlled and predictable manner. The incorporation of bioactive molecules and GFs into these scaffolds has been shown to enhance cellular activities and promote angiogenesis. However, the incorporation of immunological cues into biomaterials is strategically designed to facilitate targeted endogenous cell recruitment rather than a generalized host cell attraction. Specifically, the integration of tethered chemokines, such as CXCL12 (SDF-1α), or specific bioactive motifs, enables the selective recruitment of endogenous progenitor cells and pro-healing immune subsets, particularly M2-like macrophages [42]. This orchestrated chemoattraction is a fundamental requirement for accelerating in situ tissue engineering, as it ensures functional vascularization and the seamless integration of the scaffold within the host microenvironment [43].
Furthermore, the utilization of 3D bioprinting technology enables the fabrication of highly customizable scaffolds that can accurately replicate the complex architecture of native tissues. This technology allows for precise control over the spatial distribution of various cell types, biomaterials, and bioactive agents, creating a microenvironment that closely mimics the physiological conditions necessary for optimal tissue regeneration [44]. By tailoring scaffold design to the specific tissue needs, researchers can significantly enhance host integration and improve overall therapeutic outcomes.
Mesenchymal stem cells (MSCs) have emerged as a promising tool in the field of immunotherapy due to their unique immunomodulatory properties. Isolated from diverse sources such as bone marrow, adipose tissue, umbilical cord, and dental tissues, MSCs are gaining recognition for their potential in addressing various immune-related disorders [45]. A key mechanism underlying the immunomodulatory effect of MSCs is their capacity to secrete a diverse array of bioactive molecules, including cytokines, chemokines, and GFs. These molecules modulate the function and activity of various immune cells, including T cells, B cells, natural killer (NK) cells, and dendritic cells, ultimately leading to a significant reduction in immune responses. Furthermore, MSCs promote the differentiation and expansion of regulatory T cells, which play a crucial role in maintaining immune tolerance and preventing excessive immune activation [46]. In addition to their paracrine effects, MSCs can also directly interact with immune cells through cell-to-cell contact. This direct interaction enables MSCs to inhibit T cell proliferation and activation by expressing inhibitory molecules on their surface or inducing T cell apoptosis [47]. Moreover, MSCs influence the function of antigen-presenting cells, thereby modulating the initiation and progression of immune responses.
Due to their immunomodulatory properties, MSCs have attracted significant interest in the scientific community for potential clinical applications [46]. Recent studies have identified novel approaches, such as the encapsulation of MSCs in hydrogels, as a means to enhance their therapeutic efficacy. This strategy protects the cells from a hostile inflammatory microenvironment while improving their survival and functionality following implantation. The creation of an optimal healing environment through hydrogel encapsulation of MSCs may facilitate the regeneration of damaged tissues and the resolution of inflammatory responses.
Despite promising advancements in the use of MSCs for immunomodulation, the exact mechanisms by which biomaterials influence the immunoregulatory functions of these cells remain poorly understood. A deeper understanding of these mechanisms is crucial for advancing knowledge in the fields of tissue engineering and regenerative medicine. Elucidating the interactions between biomaterials and MSCs has the potential to facilitate the development of more effective therapeutic strategies aimed at repairing damaged tissues, ultimately leading to improved patient outcomes.
In summary, MSCs represent a valuable tool for immunomodulation, with the potential to treat a wide range of immune-related disorders [48].
The application of MSC-derived EVs, particularly exosomes, is rapidly gaining traction as a cornerstone of next-generation immunomodulation and regenerative medicine [49]. These nano-sized, membrane-bound vesicles (30–150 nm) function as specialized transport vehicles for bioactive molecules, including signaling proteins, regulatory lipids, and diverse RNA species (miRNA, mRNA). By mediating horizontal gene transfer and surface receptor interactions, EVs play a pivotal role in intercellular communication, effectively reprogramming the local microenvironment to suppress chronic inflammation and orchestrate functional tissue repair.
A primary advantage of MSC-derived EVs lies in their capacity to modulate both innate and adaptive immune responses. This is particularly relevant in pathological contexts where persistent pro-inflammatory states, such as immune disorders or chronic wounds, hinder healing [39]. Unlike traditional cell-based therapies, which may present risks of mal-differentiation, tumorigenicity, or vascular occlusion (e.g., lung trapping), EVs offer a safer, acellular approach. Their reduced immunogenicity and inability to self-replicate minimize the potential for adverse host-graft interactions, positioning them as a robust alternative to whole-cell transplantation. Furthermore, the structural versatility of exosomes allows for the targeted delivery of therapeutic molecules. Their small size facilitates the penetration of biological barriers, such as the blood-brain barrier (BBB), enhancing efficacy in treating neurodegenerative diseases while limiting the systemic side effects often associated with conventional pharmacotherapy [50]. As summarized in Table 3, the transition from MSCs to EVs addresses several critical bottlenecks, including the logistical complexities of maintaining cell viability and the standardization of good manufacturing practice (GMP)-compliant batches. However, a significant functional divergence remains: while MSCs act as “living bioreactors” capable of dynamically sensing and responding to inflammatory fluctuations, EVs are “static” mediators that lack autonomous environmental sensing. Consequently, current research is shifting toward the bioengineering of EV cargo and their integration with responsive immuno-materials to create “smart” delivery systems that can mimic the adaptive behavior of whole cells.
Comparative analysis of MSCs vs. MSC-derived EVs in immunomodulation.
| Feature | Mesenchymal stem cells (MSCs) | MSC-derived EVs/exosomes | Key reference |
|---|---|---|---|
| Nature | Living, cellular therapy; metabolically active. | Acellular, membrane-bound vesicles (paracrine mediators). | [51] |
| Mechanism | Dynamic sensing; adapts response to microenvironmental cues via cell-cell contact and paracrine signaling. | Targeted delivery of pre-defined bioactive cargo (miRNA, proteins, lipids). | [52] |
| Safety profile | Potential risks of maldifferentiation, lung trapping (IV), or inflammatory activation. | Minimal immunogenicity; no risk of tumorigenesis or self-replication. | [53] |
| Stability | Requires cryopreservation; sensitive to handling and transport logistics. | High stability; amenable to lyophilization and easier “off-the-shelf” distribution. | [52] |
| Biological barriers | Limited by cell size; often sequestered in the lungs after systemic administration. | Small size (30–150 nm) allows better penetration of biological barriers (e.g., BBB). | [54] |
| Regulatory path | Complex; requires rigorous characterization of living cell batches. | Emerging as biologic products, potentially easier standardization and scale-up. | [55] |
EVs: extracellular vesicles; BBB: blood-brain barrier.
This field is witnessing an influx of clinical trials designed to validate the potency of MSC-EVs across diverse pathologies. The integration of these cell-free strategies into the broader framework of biomaterial science represents a promising frontier for the treatment of immune-mediated diseases and the advancement of precision regenerative medicine.
The clinical translation of immune-materials from laboratory proof-of-concept to bedside application is obstructed by several hurdles, starting with manufacturing scalability. The transition to large-scale GMP production often compromises the precise physicochemical surface properties—such as ligand nanospacing, density, and topography—that are critical for effective immune modulation. While microfluidics has improved consistency for nanoparticles, the production of hierarchical 3D-bioprinted scaffolds still faces structural variability at scale, necessitating the integration of high-throughput screening to ensure batch-to-batch consistency. Furthermore, the heavy reliance on preclinical animal models frequently fails to recapitulate the human immune system, leading to unpredicted immunogenicity; thus, the adoption of organ-on-a-chip technologies and “humanized” models is becoming a critical step for accurate screening [6]. Regulatory complexity further complicates this landscape, as multifunctional platforms classified as combination products must satisfy the distinct safety standards of both medical devices and pharmacological agents. Beyond these technical barriers, the biological complexity and individual variability (age, sex, and comorbidities) of the FBR necessitate a shift toward personalized immuno-materials. Finally, to mitigate risks of chronic inflammation or systemic toxicity, the field must focus on “dynamic resolution”—where the metabolic fate of degradation by-products is meticulously mapped—ultimately leading to self-adaptive, intelligent systems capable of leveraging cell-free cues, such as exosomes, to steer immune polarization with unprecedented real-time precision.
Despite the therapeutic potential of immuno-materials, their clinical translation is hindered by significant immune-related risks. The most prevalent is the FBR, a chronic inflammatory state where persistent macrophage activation leads to the formation of foreign body giant cells (FBGCs) and a dense fibrous capsule that can impair device function or lead to implant failure [56]. Furthermore, the degradation products of certain polymers may induce local or systemic toxicity. Of particular concern is ‘off-target’ immune activation, where materials intended for localized modulation trigger systemic inflammatory responses or, in rare cases, exacerbate underlying autoimmune conditions.
The convergence of immunology and materials science has established a transformative frontier: the field of immuno-materials. This review has demonstrated that the successful development of these platforms surpasses simple biocompatibility, requiring a rigorous multidisciplinary integration of materials science, immunology, and bioengineering. As evidenced by the transition from fundamental recognition mechanisms (PAMPs/DAMPs) to clinical applications, such as peptide-based hemostats and lipid nanoparticle-based vaccines, the synergy between these disciplines is the key to mastering the complex bio-interface.
While significant challenges remain, particularly regarding manufacturing scalability (GMP), regulatory validation of combination products, and the inherent variability of the human immune response, the field is moving toward a new era of precision immuno-modulation. The shift from retrospective literature summaries to a strategic, primary-source-driven roadmap identifies a clear path forward: the integration of “intelligent” and self-adaptive systems, such as 3D-bioprinted anisotropic scaffolds and cell-free bioactive cues like exosomes.
To fully harness the potential of immuno-materials, future efforts must prioritize “dynamic resolution” and personalized approaches that account for individual patient profiles. By bridging the gap between laboratory-scale innovation and clinical reality through advanced methodologies like organ-on-a-chip and humanized models, immuno-materials will not only address pressing global health challenges but will fundamentally redefine the future of precision medicine and regenerative therapy.
DAMPs: damage-associated molecular patterns
ECM: extracellular matrix
EVs: extracellular vesicles
FBR: foreign body response
GFs: growth factors
GMP: good manufacturing practice
MSCs: mesenchymal stem cells
PAMPs: pathogen-associated molecular patterns
PRRs: pattern recognition receptors
TLRs: toll-like receptors
MT, AF, FP: Conceptualization, Writing—original draft, Writing—review & editing. AP, RR, FI, GP: Investigation, Formal analysis, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 721
Download: 21
Times Cited: 0
