375 results in Exploration of Targeted Anti-tumor Therapy
Latest
Sort by :
- Latest
- Most Viewed
- Most Downloaded
- Most Cited
Open Access
Review
ESR1 mutations in ER-positive breast cancer: from endocrine resistance to ctDNA-guided therapeutic interception
Thais Martinez ... Hisham F. Bahmad
Published: May 26, 2026 Explor Target Antitumor Ther. 2026;7:1002375
This article belongs to the special issue Breaking Boundaries in Breast Cancer Care: Emerging Controversies and Innovation in Surgical and Medical Approaches
Open Access
Review
The evolution of chemotherapy in brain tumors: from historical milestones to precision medicine in glioblastoma
Alessia Ciafarone, Paola Palumbo
Published: May 21, 2026 Explor Target Antitumor Ther. 2026;7:1002374
This article belongs to the special issue Novel Precision Medicine Approaches to Brain Tumors (Primary and Metastatic)

Open Access
Review
RNA therapeutic targeting of recalcitrant and rare cancers
Afeez Adekunle Ishola ... Bashir Lawal
Published: May 21, 2026 Explor Target Antitumor Ther. 2026;7:1002373
Open Access
Review
Triple-negative breast cancer: current understanding and future perspectives
Alhasan Alobaidi, Vikrant Rai
Published: May 14, 2026 Explor Target Antitumor Ther. 2026;7:1002372
Open Access
Original Article
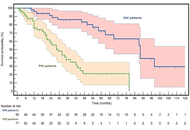
Oligometastatic versus polymetastatic colon cancer: functional and genomic determinants of divergent metastatic trajectories
Roberto Sirica ... Alessandro Ottaiano
Published: May 13, 2026 Explor Target Antitumor Ther. 2026;7:1002371
Open Access
Mini Review
The evolving landscape of first-line and subsequent therapies in EGFR-mutated NSCLC: efficacy, resistance, and tolerability
Carminia Maria Della Corte ... Floriana Morgillo
Published: April 27, 2026 Explor Target Antitumor Ther. 2026;7:1002370
Open Access
Commentary
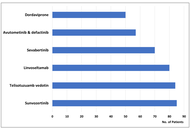
FDA 2025 Cancer Drug Approvals: targeted therapy dominates
Jan Trøst Jørgensen
Published: April 27, 2026 Explor Target Antitumor Ther. 2026;7:1002369
This article belongs to the special issue Precision Oncology: Molecular Classification, Efficacy Prediction, and Treatment Decision-Making
Open Access
Systematic Review
The evolving role of targeted radioligand therapy in small cell and non-small cell lung cancer: a systematic review
Serin Moghrabi ... Akram Al-Ibraheem
Published: April 27, 2026 Explor Target Antitumor Ther. 2026;7:1002368

Open Access
Case Report
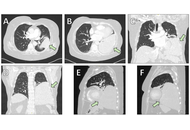
Solitary fibrous tumor of the lung: diagnostic challenges and surgical management
Vasileios Leivaditis ... Manfred Dahm
Published: April 21, 2026 Explor Target Antitumor Ther. 2026;7:1002367
Open Access
Original Article
Lorlatinib in advanced ALK-positive NSCLC after prior progression on ALK inhibitors: real-world experience in Russia
Sergey V. Orlov ... Evgeny N. Imyanitov
Published: April 16, 2026 Explor Target Antitumor Ther. 2026;7:1002366

Open Access
Original Article
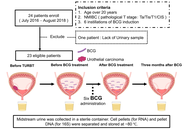
Changes of urinary immunity and microbiome after intravesical BCG therapy and their association with outcomes in NMIBC
Yuki Oda ... Kiyohide Fujimoto
Published: April 13, 2026 Explor Target Antitumor Ther. 2026;7:1002365
Open Access
Original Article
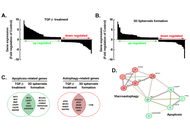
Disrupting the TGF-β-regulated epithelial-mesenchymal transition, apoptotic and autophagic phenotypes of 3D glioblastoma spheroids via glycolytic inhibition
Maellis Payet-Desruisseaux ... Borhane Annabi
Published: March 30, 2026 Explor Target Antitumor Ther. 2026;7:1002364
This article belongs to the special issue Novel Precision Medicine Approaches to Brain Tumors (Primary and Metastatic)
Open Access
Original Article
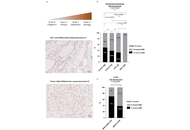
CHD4 and NOX4 expression in thyroid tumor tissues
Salma Fenniche ... Rabii Ameziane El Hassani
Published: March 25, 2026 Explor Target Antitumor Ther. 2026;7:1002363
Open Access
Original Article
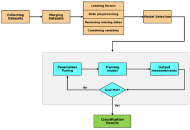
Survival prediction in triple-negative breast cancer: a Cox model with fairness assessment using ISO/IEC TR 24027:2021 in a MENA cohort
Mehrshad Alirezaei Farahani ... Marjan Mansourian
Published: March 20, 2026 Explor Target Antitumor Ther. 2026;7:1002362
Open Access
Review
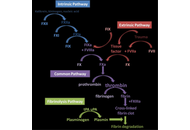
Emerging roles of haemostatic proteins as markers of disease progression and prognosis in breast cancer
Ogochukwu O. Izuegbuna
Published: March 09, 2026 Explor Target Antitumor Ther. 2026;7:1002361
This article belongs to the special issue Breaking Boundaries in Breast Cancer Care: Emerging Controversies and Innovation in Surgical and Medical Approaches
Open Access
Systematic Review
Comparison of weekly docetaxel regimens in prostate cancer: a systematic review and frequentist network meta-analysis
Shree Rath ... Amar Lal
Published: February 27, 2026 Explor Target Antitumor Ther. 2026;7:1002360
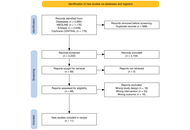
Open Access
Mini Review
Benign, persistent, and invasive: mechanistic and translational approaches to middle‑ear cholesteatoma
Pinelopi Samara ... Ioannis Athanasopoulos
Published: February 24, 2026 Explor Target Antitumor Ther. 2026;7:1002359
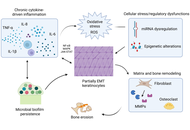
Open Access
Review
Glioblastoma pathophysiology: roles of aging driven changes in STAT3 interactions with NF-κB dimer components in the modulation of the mitochondrial melatonergic pathway and night-time inflammation resolution
George Anderson
Published: February 13, 2026 Explor Target Antitumor Ther. 2026;7:1002358

Open Access
Perspective
Application of explainable artificial intelligence integrating with electronic health record in oncology
Yuhan Yang, Xici Liu
Published: February 04, 2026 Explor Target Antitumor Ther. 2026;7:1002357

Open Access
Editorial
The critical need for robust decision support in the era of precision cancer therapeutics
Maurie Markman
Published: January 22, 2026 Explor Target Antitumor Ther. 2026;7:1002356
 Previous
Previous