Affiliation:
Department of Ichthyology and Aquatic Environment, School of Agricultural Sciences, University of Thessaly, 38445 Volos, Greece
Email: mxatzi@uth.gr
ORCID: https://orcid.org/0000-0003-1372-1467
Affiliation:
Department of Ichthyology and Aquatic Environment, School of Agricultural Sciences, University of Thessaly, 38445 Volos, Greece
ORCID: https://orcid.org/0000-0002-3667-2947
Affiliation:
Department of Ichthyology and Aquatic Environment, School of Agricultural Sciences, University of Thessaly, 38445 Volos, Greece
ORCID: https://orcid.org/0000-0001-7536-8959
Affiliation:
Department of Ichthyology and Aquatic Environment, School of Agricultural Sciences, University of Thessaly, 38445 Volos, Greece
ORCID: https://orcid.org/0009-0003-5152-0356
Affiliation:
Department of Ichthyology and Aquatic Environment, School of Agricultural Sciences, University of Thessaly, 38445 Volos, Greece
ORCID: https://orcid.org/0000-0003-2782-8820
Affiliation:
Department of Ichthyology and Aquatic Environment, School of Agricultural Sciences, University of Thessaly, 38445 Volos, Greece
ORCID: https://orcid.org/0000-0002-5036-8706
Affiliation:
Department of Ichthyology and Aquatic Environment, School of Agricultural Sciences, University of Thessaly, 38445 Volos, Greece
ORCID: https://orcid.org/0000-0002-8454-6937
Explor Foods Foodomics. 2026;4:1010134 DOI: https://doi.org/10.37349/eff.2026.1010134
Received: August 28, 2025 Accepted: March 12, 2026 Published: April 15, 2026
Academic Editor: Gabriele Rocchetti, Food and Nutrition (Diana); Università Cattolica del Sacro Cuore (Piacenza and Cremona Campus), Italy
Aim: This study aimed to evaluate the effects of essential oil nanoemulsion-enriched diets on feed acceptability, growth performance, proximate composition, and digestive gland histology of farmed Cornu aspersum maximum.
Methods: A total of 2,000 juvenile snails were divided into four dietary treatments for a two-month feeding experiment. The control group received a commercial poultry feed, while the experimental groups were fed the same diet supplemented with an essential oil nanoemulsion at concentrations of 1 mL/kg (T1), 3 mL/kg (T2), and 5 mL/kg (T3). Eight samplings were conducted to measure snail diameter, weight, mortality, and feed residues. At the end of the experiment, fillet proximate composition and histopathological alterations of the digestive gland tissue were evaluated.
Results: A tendency toward increased snail weight was observed across all treatments. Diet enrichment led to significantly higher fillet protein content across all treatment groups (T1, T2, and T3) compared to the control, while lipid content was highest in T1. Histopathological examination revealed enlargement of hepatic ducts in T1, apoptosis of digestive cells in T2, and necrosis of digestive and calcium cells along with thinned epithelium lining in T3.
Conclusions: Dietary supplementation with essential oil nanoemulsion did not negatively affect snail growth and resulted in heavier snails with increased fillet protein content. However, histopathological evidence of toxicity rendered higher supplementation levels (T2 and T3) unsuitable. Enrichment at 1 mL/kg appears to be suitable for use in commercial snail farming.
Invertebrates constitute an important component of the global diet, and the culinary value of molluscan species is no exception [1]. Edible gastropods are known for their nutritional value, mainly for their high protein content and essential minerals [2, 3]. In addition to extensive studies on the histology, physiology, and breeding of helicine species [4–8], their meat quality has also been investigated due to their widespread consumption throughout Europe [9, 10]. Moreover, edible snail tissue is considered a premium food product, with its high protein content and low fat highlighted as key aspects of its nutritional value [9].
Apostolou et al. [11] recorded a total of 61 operational snail farms in Greece sharing a common gene pool for broodstock [12]. Notably, the highest values in snail growth were observed in open field combined with low density, while the heaviest snail fillet was achieved through a combination of net-covered greenhouses and low density [13]. It is important to highlight that nearly all snail farmers utilize a compound diet as a feed for their snails. The nutritional requirements of terrestrial snails, and in particular their macronutrient requirements, are poorly known. In nature, terrestrial snails feed on a variety of organic materials, such as plants or decaying plant material [14]. Moreover, farmed snail diets have been either empirically formulated or adapted from those designed for other farmed animals, such as poultry, partly due to the limited scientific understanding of snail nutrition [15–17].
Essential oils are complex mixtures of volatile compounds produced by different plant species as secondary metabolites [18]. Currently, more than 3,000 types of essential oils are known, but only about a tenth of them are suitable for use [19]. Phytochemicals are widely recognized as safe for animals, consumers, and the environment. Regarding usage in aquaculture, these products have great potential due to the beneficial biological effects reported in aquatic species, such as growth enhancement, appetite stimulation, immunomodulatory, and antioxidant activity [20]. They contribute to the maintenance of gastrointestinal tract health and functionality through regulation of the native microbiota, which plays a crucial role in host physiology, including protection against pathogenic microorganisms, efficient nutrient absorption, and immune regulation [21].
These properties are attributed to the ability of essential oils to act as prebiotics within the intestinal environment, regulating the composition of the gut microbiota. In addition, essential oils may affect both the quantity and the composition of secretions produced by the intestinal mucosa and alter the physical and chemical characteristics of the intestinal environment, resulting in enhanced disease resistance. Stimulation of intestinal secretions facilitates improved regulation of digestion and nutrient absorption by the gut microbiome. This process may increase the availability of amino acids for protein synthesis and, consequently, lead to increased body protein content in reared animals [22].
When essential oils are included in poultry diets, feed intake has been reported to remain unchanged or slightly decreased. In contrast, improvements in weight gain and feed conversion ratio are consistently observed. These positive effects are attributed to two commonly accepted mechanisms: stimulation of digestive enzyme secretion and stabilization of the gut microbial community [23]. In swine, however, the effects of essential oils on feed intake remain inconsistent. Reported increases in intake are probably related to improved growth rate, a response commonly observed with growth-promoting feed additives. Enhanced feed digestibility following diet enrichment with essential oils has been recorded. The improvement may be attributed to increased bile and enzyme secretion and to a lesser extent, to reduced endogenous nutrient losses [23].
Several studies have investigated the use of essential oils in livestock nutrition, including poultry [24], rabbits [25], pigs [26], and fish [27]. In addition, several studies have examined the proximate composition of poultry, bovine, and pork meat following enrichment of the diet with essential oils such as oregano and rosemary [28–30]. Despite the potential benefits, relatively few studies have examined the effects of vegetable oil supplementation on the lipid composition of farmed snails (Helix aspersa maxima) fillet [15].
Studies on the use of essential oils in snails have mainly focused on their application for snail control [31–34], while only a limited number have evaluated their inclusion in snail diets and the associated effects on growth performance [35, 36]. Linhart and Thompson [35] used 6 chemotypes of Thymus vulgaris in a feeding experiment with adult and juvenile Cornu aspersum aspersum (C. a. aspersum), whereas Lemjallad et al. [37] enriched snail diets with rosemary powder. The only study conducted in Greece by Vokou et al. [36] used three terrestrial snail species [C. a. aspersum, Eobania vermiculata (E. vermiculata), Helix lucorum] fed with diets enriched with oregano leaves or oregano essential oil. To date, no studies have evaluated the effects of essential oil-enriched diets on snail fillet composition. This study aimed to investigate the effects of nanoemulsion containing various essential oils on feed acceptability, growth performance, chemical composition of snail fillet, and digestive glands’ tissue morphology in Cornu aspersum maximum (C. a. maximum). Digestive gland morphology was used as a rapid and accessible indicator of animal welfare. By evaluating these quality factors, this study seeks to establish acceptable inclusion rates of essential oils in the diets of farmed C. a. maximum.
The four experimental diets were formulated to be isoproteic (14%), isolipidic (1.1%), and isoenergetic (21.11 kJ/g) (Table 1). The control diet (C) was a commercial poultry feed commonly used in snail farming that was well-grounded and mixed with 300 g/kg of calcium carbonate.
Proximate composition of diets (% as fed).
| Proximate composition | C | T1 | T2 | T3 |
|---|---|---|---|---|
| Moisture | 7.80 ± 0.08 | 7.33 ± 0.47 | 7.33 ± 0.37 | 7.12 ± 0.19 |
| Crude protein | 14.31 ± 0.74 | 13.72 ± 1.15 | 13.80 ± 1.07 | 13.10 ± 0.40 |
| Crude fat | 1.04 ± 0.04 | 1.10 ± 0.20 | 1.17 ± 0.11 | 1.12 ± 0.27 |
| Total carbohydrates | 53.64 ± 3.14 | 51.97 ± 3.56 | 54.92 ± 1.70 | 53.46 ± 2.21 |
| Ash | 31.59 ± 1.60 | 32.27 ± 1.35 | 31.47 ± 2.88 | 31.36 ± 0.36 |
| Gross energy (kJ/g) | 21.11 ± 0.12 | 21.09 ± 0.11 | 21.15 ± 0.08 | 21.10 ± 0.11 |
Mean ± standard deviation (n = 3). Where C: control diet, T1: diet with 1 mL/kg inclusion level of the essential oil nanoemulsion, T2: diet with 3 mL/kg inclusion level of the essential oil nanoemulsion, T3: diet with 5 mL/kg inclusion level of the essential oil nanoemulsion.
For the preparation of the remaining three diets, the control feed was supplemented with a commercial nanoemulsion containing a mix of 5 medicinal and aromatic plants (MAPs) plant essential oils at three different concentrations: 1 mL/kg (T1 diet), 3 mL/kg (T2 diet), and 5 mL/kg (T3 diet). The inclusion levels of the essential oil nanoemulsion are expressed as mL per kg of feed and refer to the volume of nanoemulsion added per kilogram of feed during preparation. Premixture of feed was kindly provided by Dioscurides P.C., a MAPs processing company located in Anarrachi (Ptolemaida, Greece). The company uses strictly Greek MAPs as raw materials, cultivated by trained farmers, according to the standards of integrated crop management, in selected areas mainly of Central and Northern Greece. The studied plant essential oils were oregano oil (50%), lavender oil (12.5%), clove oil (12.5%), rosemary oil (12.5%), and absinthe oil (12.5%). The chosen concentration levels of nanoemulsion in the diets were based on the supplier’s proposal without exceeding the levels mentioned by Linhart and Thompson [35]. The nanoemulsion was incorporated into each diet by carefully spraying the corresponding concentration and then was allowed to be absorbed by the feed for at least 3 hours under a fume cupboard.
A total number of 2,000 juvenile snails, C. a. maximum mean weight of 2.75 g and an 18.54 mm mean shell diameter, were supplied by a local heliciculture farm and transferred to the experimental farming facilities (net-covered greenhouse) of the University of Thessaly (Volos, Greece). The facilities consisted of a net-covered greenhouse that spans an area of 300 m2 with a galvanized metal frame. It is covered with 80% polyethylene shade net. In the net-covered greenhouse, four compartments were established (2 m by 10 m), around the perimeter of which a low-voltage power fence was installed. In these compartments, broadleaf plants (cabbage, lettuce) were planted. Temperature and humidity were monitored daily in both farming systems. The maintenance of the required humidity was achieved by a low-pressure water spray cooling system. During the experimental period, the system operated daily for 30 min each day except on rainy days. Both air temperature and relative humidity remained at a favorable level throughout the experiment, with mean values of 18.4°C and 80%, respectively.
The snails were equally allocated to four experimental treatments (C, T1, T2, and T3; 500 snails per treatment). Each treatment was conducted in two separate breeding units, which served as experimental replicates. Prior to the initiation of the feeding, snails were acclimated for 10 days and fed a commercial diet suitable for the species. After the initiation of the trial, snails were fed every second day (2 kg per treatment). Any feed residue was weighed before providing the next meal to calculate feed consumption.
The trial lasted 56 days in total. Sampling (90 snails per treatment) was conducted weekly in the morning before feeding. Snail weight (g) was recorded using a precision balance (EMB 200-2, Kern & Sohn, Balingen, Germany), and snail size as depicted by shell diameter (mm) was measured with digital calipers. Mortality was recorded, and dead snails were removed. Finally, feed residues were weighed every two days to calculate feed consumption.
At the end of the experimental period, all remaining live snails were collected, weighed individually, and their diameter was measured. Snails were dissected after anesthesia using twenty drops of Eugenia caryophyllus oil (clove oil, CHEMCO, Malsch, Germany) diluted in 50 mL of water [9], organs were separated, and soft body weight, fillet mass, and digestive gland weight were recorded separately. Whole snails and tissue samples were collected from each treatment group for analyses. 120 snail fillets (30 of each group) were analyzed for proximate composition according to AOAC [38], and 5 digestive gland tissues per group were kept for histological analysis.
Snail growth, feed utilization, and snail production characteristics were assessed and evaluated:
Growth rate (g/day/snail) = (Wf–Wi)/fattening period (days), where Wf and Wi are the mean final and initial weight, respectively.
Weight gain (g) = Wf (g)–Wi (g), where Wf and Wi are the mean final and initial weight, respectively.
Soft body weight (%) = soft body weight × 100/snail weight (refers to snails’ visceral mass and its percentage is calculated relative to the total weight, including shell, soft body, and fillet).
Shell weight (%) = shell weight × 100/snail weight.
Fillet mass (%) = fillet mass × 100/snail weight.
Feed consumption (g/snail/day) = total feed consumption/total duration of the experiment in days.
Feed consumed (FC g/snail) = total amount of feed consumed (g)/snail.
Feed conversion ratio (FCR) = total feed consumed (g)/total weight gain (g).
Mortality rate (%) = number of dead snails × 100/initial number of live snails.
For moisture mass fraction determination (%), 3 g were dried at 105°C in an oven (TS 8056; Termaks, Bergen, Norway) until constant mass, and the water content was determined gravimetrically (AOAC, 2020) [38]. Dry matter of each group was pooled, and 10 g of dry matter was used for the following analyses. Crude protein mass fraction (%) was tested by the Kjeldahl method (N × 6.25; Behr Labor-Technik, Düsseldorf, Germany) using 0.2 g dry matter and then expressed on a net basis, and crude fat by the Soxhlet method (SOXTHERM® SOX416 macro; Gerhardt, Königswinter, Germany). For ash mass fractions (%) estimation, a water-free sample was combusted in a muffle furnace (Nabertherm L9/12/C6; Lilienthal, Germany) by heating at 600°C for 3 h, and the ash content was measured gravimetrically. Gross energy (kJ/g) was evaluated using an adiabatic oxygen bomb calorimeter (C7000; IKA Werke, Staufen, Germany). Crude protein, crude fat, and ash mass fractions of each group were expressed on a net basis. Carbohydrate mass fraction (%) was calculated by the difference between 100 and the sum of the crude protein, crude fat, and ash content on a net basis. Crude protein, crude fat, total carbohydrates, and ash mass fractions of each group were expressed on a dry basis. All measurements were carried out in triplicate, and the values were averaged.
Digestive gland tissues were immersed in a fixative solution and left undisturbed for 24 hours at 4°C. The fixed tissues were then rinsed in running water for an additional 24-hour period before paraffin embedding. During processing, tissue fragments were sequentially immersed in graded ethanol solutions of increasing concentrations (50%, 70%, 95%, and two changes of 100%), each for one hour. Subsequently, the samples were cleared in xylene for two hours and transferred to an oven at 65°C in a specialized container filled with liquid paraffin, ensuring thorough impregnation over a two-hour period. The impregnated tissues were then positioned in dedicated metal molds containing liquid paraffin, which solidified within a few minutes at room temperature. Using a paraffin microtome (MICROM HM315), longitudinal sections of the hepatopancreas, 5 µm thick, were obtained. Selected sections were stained with Harris hematoxylin solution (Scharlab S.L., Sentmenat, Barcelona, Spain) and eosin Y (VWR International, Leuven, Belgium) [39].
Following staining, the tissues were examined under a light microscope equipped with a digital camera (ProgRes Plus 2, Jenoptik Optical Systems GmbH, Jena, Germany). Histological images were captured at appropriate magnification levels using Progress Capture 2.1 software.
Data for statistical analysis was evaluated for normal distribution by employing the Shapiro-Wilk test for normality and homogeneity of variance by employing Bartlett’s and Levene’s tests. One-way ANOVA was used to compare means. In cases where ANOVA showed statistically significant differences, the data were subjected to Tukey’s test to identify differences between treatments. The significance level was set at 0.05. Statistical analysis was performed using SPSS 20® software (SPSS Inc., Chicago, IL, USA).
All groups of snails promptly accepted the respective diets, and feed consumption was similar (P > 0.05) among all groups (Table 2). While weight values remained similar throughout most of the experiment, a statistically significantly lower value was observed in the T3 group compared to the control group on day 42. At the end of the experiment, the mean final body weight of snails from all treatments ranged from 14.70–15.14 g (Table 2), and there were not found significant differences among them. The same result also applies to weight gain and growth rate.
Snail growth indicators and production characteristics for each experimental treatment.
| Growth indicators and production characteristics | Experimental treatments | |||
|---|---|---|---|---|
| C | T1 | T2 | T3 | |
| Initial weight (g) | 2.92 ± 1.41a | 2.65 ± 1.87a | 2.61 ± 1.63a | 2.81 ± 1.85a |
| 14 days | 5.77 ± 2.14a | 5.93 ± 2.65a | 5.57 ± 2.40a | 5.35 ± 2.20a |
| 28 days | 8.35 ± 4.30 a | 9.22 ± 3.67 a | 8.51 ± 2.83a | 8.53 ± 3.75a |
| 42 days | 14.42 ± 5.13b | 11.6 ± 4.06a,b | 10.32 ± 4.33a,b | 10.18 ± 3.25a |
| Final weight (g) (56 days) | 15.14 ± 2.31a | 14.70 ± 2.19a | 14.97 ± 3.38a | 14.78 ± 2.70a |
| Weight gain (g) | 12.22a | 12.05a | 12.36a | 11.97a |
| Growth rate (g/d/snail) | 0.22a | 0.22a | 0.22a | 0.21a |
| Feed quantity (kg) | 50.55a | 46.63a | 49.73a | 46.95a |
| FCR | 8.25 | 7.76 | 8.01 | 7.86 |
| FC (g/snail) | 100.80a | 93.52a | 99.68a | 94.08a |
Mean ± standard deviation (n = 90). a,b: Values within each row not sharing a common superscript letter are significantly different (P < 0.05). Where C: experimental treatment fed the control diet, T1: experimental treatment fed the T1 diet, T2: experimental treatment fed the T2 diet, T3: experimental treatment fed the T3 diet. FCR: feed conversion ratio; FC: feed consumed.
Shell diameter at the end of the experiment was smaller in snails fed with the control diet compared to the other treatments, but this was not statistically significant (Table 3). Overall, the diameter values did not show any statistically significant difference at any point in the feeding experiment.
Snail diameter (mm) for each experimental treatment.
| Diameter (mm) | Experimental treatments | |||
|---|---|---|---|---|
| C | T1 | T2 | T3 | |
| Initial | 18.95 ± 3.72a | 18.25 ± 4.37a | 18.12 ± 4.01a | 18.82 ± 5.06a |
| 14 days | 24.59 ± 3.87a | 23.98 ± 4.27a | 23.71 ± 3.59a | 23.57 ± 3.86a |
| 28 days | 28.85 ± 5.13a | 29.61 ± 5.07a | 28.67 ± 3.96a | 28.94 ± 4.71a |
| 42 days | 30.99 ± 6.23a | 31.61 ± 4.50a | 30.48 ± 4.97a | 30.23 ± 3.94a |
| Final (56 days) | 32.09 ± 4.40a | 36.42 ± 2.44a | 35.83 ± 5.62a | 36.38 ± 4.97a |
Mean ± standard deviation (n = 90). a: Values within each row not sharing a common superscript letter are significantly different (P < 0.05). Where C: experimental treatment fed the control diet, T1: experimental treatment fed the T1 diet, T2: experimental treatment fed the T2 diet, T3: experimental treatment fed the T3 diet.
Mortality was observed in all experimental groups during the trial. When the mortality rate was calculated as mean values over the entire experimental period, the control group showed the highest overall mortality (6.2%), while the lowest was recorded in T2 (3.69%). Mortality rate during the experiment in T1 and T3 was similar, at approximately 5%.
Table 4 shows the snail production characteristics, such as shell weight, soft body weight, and fillet mass, at the end of the experimental period. All snail groups exhibited similar (P > 0.05) shell weights and did not exhibit significant differences among treatments (Table 4). There was a trend of higher soft body weights in snails fed the nanoemulsion-treated diets compared to the control group, but this was not significant (P > 0.05).
Snail production characteristics for each experimental treatment.
| Production characteristics | Experimental treatments | |||
|---|---|---|---|---|
| C | T1 | T2 | T3 | |
| Shell weight (g) | 3.20 ± 1.85a | 2.86 ± 0.66a | 3.28 ± 1.17a | 2.89 ± 0.63a |
| Shell weight (%) | 21.13 ± 13.48a | 19.45 ± 2.81a | 21.91 ± 5.76a | 19.55 ± 3.21a |
| Whole soft body weight (g) | 9.56 ± 2.13a | 9.92 ± 1.53b | 10.35 ± 2.40b | 10.44 ± 2.08b |
| Whole soft body weight (%) | 62.03 ± 11.23a | 67.48 ± 7.57a | 69.14 ± 10.35a | 70.63 ± 10.19a |
| Fillet mass (g) | 2.60 ± 0.37a | 2.58 ± 0.34a | 2.68 ± 0.38a | 2.79 ± 0.49a |
| Fillet mass (%) | 17.17 ± 2.26a | 17.55 ± 2.36a | 17.9 ± 2.72a | 18.87 ± 3.13a |
Mean ± standard deviation (n = 15). a,b: Values within each row not sharing a common superscript letter are significantly different (P < 0.05). Where C: experimental treatment fed the control diet, T1: experimental treatment fed the T1 diet, T2: experimental treatment fed the T2 diet, T3: experimental treatment fed the T3 diet.
Finally, fillet mass despite being highest in the experimental treatment T3 and lowest in the control group, did not exhibit significant differences among treatments. According to Table 4, the fillet mass of the snails remained unchanged despite the increasing nanoemulsion concentration.
Fillet moisture and ash contents did not exhibit significant differences among the groups (Table 5). Fillets of experimental treatment T1, T2, and T3 exhibited significantly higher protein content (60.12%, 60.20%, and 59.53%, respectively) compared to the control diet (55.20%). Fillet fat of snails fed with the enriched diets T1 and T2 presented significantly (P < 0.05) higher content (2.79% and 2.42%) than that of the control (1.18%) and the T3 group (1.93%). According to Table 5, diet enrichment affected carbohydrate content of snail fillet while gross energy remained stable.
Snail fillet proximate composition (% of dry weight) for each diet group.
| Proximate composition | C | T1 | T2 | T3 |
|---|---|---|---|---|
| Moisture (% of wet weight) | 79.91 ± 1.27a | 81.18 ± 0.80a | 80.79 ± 1.15a | 80.89 ± 1.71a |
| Crude protein | 55.20 ± 0.21a | 60.12 ± 0.67b | 60.20 ± 0.60b | 59.53 ± 0.63b |
| Crude fat | 1.18 ± 0.20a | 2.79 ± 0.11c | 2.42 ± 0.53b,c | 1.93 ± 0.22a,b |
| Total carbohydrates | 38.37 ± 0.40a | 31.46 ± 1.16b | 31.57 ± 0.78b | 33.08 ± 0.89b |
| Ash | 5.25 ± 0.26a | 5.62 ± 0.55a | 5.80 ± 0.40a | 5.46 ± 0.25a |
| Gross energy (kJ/g) | 20.05 ± 0.15a | 20.49 ± 0.19a | 20.52 ± 0.10a | 20.48 ± 0.17a |
Mean ± standard deviation (n = 3). a,b,c: Values within each row not sharing a common superscript letter are significantly different (P < 0.05). Protein, fat, and ash content on a dry basis. Where C: experimental treatment fed the control diet, T1: experimental treatment fed the T1 diet, T2: experimental treatment fed the T2 diet, T3: experimental treatment fed the T3 diet.
The digestive tubules of snails fed with the control diet are shown in Figure 1A. The digestive cells appeared morphologically normative. Embedded in the epithelium of the digestive tubules, calcium cells that possess spherules were observed. The spherules’ appearance was moderate. The presence of excretory cells could be identified. The haemolymphatic sinus was surrounded by connective tissue mainly consisting of supporting adipose cells. Due to intracellular activity of digestive cells of snails fed the T1 diet (Figure 1B), the hepatic duct appeared slightly dilated, while there were few calcium cells. In Figure 1C (T2 diet), digestive cell apoptosis was apparent, with living cells pushing dying neighboring cells towards the lumen. The rearrangement of the remaining digestive cells and the generation of new ones filled the gaps left by the dead ones, thereby maintaining the integrity of the epithelium of the digestive tubule. Αpoptotic bodies could be seen within the dilated hepatic ducts. There appeared to be very few calcium cells present, which did not contain well-formed spherules. There was a vacuole containing an undigested residue granule. There was necrosis of digestive and calcium cells of snails fed the T3 diet (Figure 1D). Hepatic duct borders could not be differentiated easily from the digestive tubule’s matrix since the digestive cells were fragmented within the lumens. Furthermore, calcium cells appeared to have dissolved cytoplasm, while a significantly thinned epithelium lining was observed.
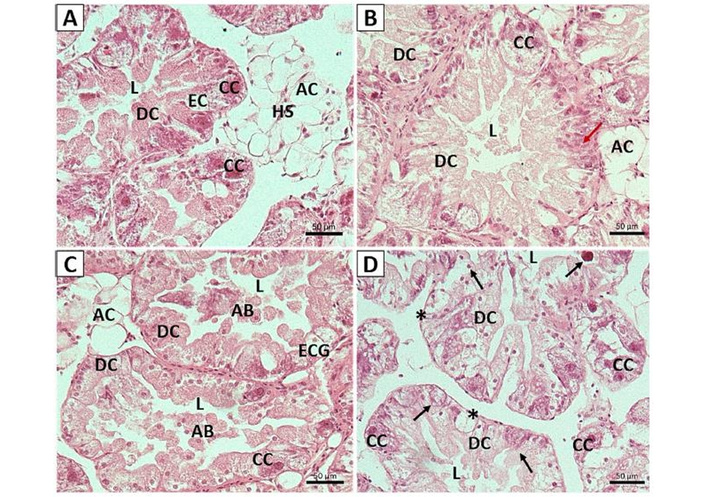
Digestive gland micrographs of snails fed with a) control diet, b) T1 diet, c) T2 diet, d) T3 diet. Transverse sections were used for Ηematoxylin and Εosin staining. AB: apoptotic bodies; AC: adipose cell; CC: calcium cell; DC: digestive cell; EC: excretory cell; ECG: excretory cell with granule; HS: haemolymphatic sinus; L: lumen; *: thinned epithelium lining; black arrow: necrotic cells; red arrow: multinucleated digestive cells. Scale bar: 50 μm.
The presence of nanoemulsion in the snail diet led to increased weight without reducing feed consumption. In contrast, Linhart and Thompson [35] reported that, among six chemotypes of Thymus vulgaris tested in adult and juvenile C. a. aspersum snails, only those fed exclusively with linalool exhibited an increase in weight. Furthermore, Vokou et al. [36] in a feeding experiment on three species of terrestrial snails (C. a. aspersum, E. vermiculata, Helix lucorum) observed that Origanum vulgare hirtum oil reduced feed consumption in all three species, while C. a. aspersum and E. vermiculata snails actively avoided areas with high essential oil concentrations. The different results in the present study may be attributed to the concentration of essential oil or to the combination of aromatic plants included in the nanoemulsion.
According to the literature and by comparing the results of our feeding experiment with previous studies, we observed a similar snail growth rate (0.22 g/snail/day). In a feeding experiment in which C. a. aspersum snails were fed with an enriched diet containing rosemary powder, Lemjallad et al. [37] reported a maximum growth rate of 0.2 g/snail/day, although the average rate was considerably lower. Furthermore, Apostolou et al. [13] recorded a mean growth rate of 0.20 and 0.18 g/snail/day for C. a. maximum.
Mean weight of snail’s fillet in our dietary experiment ranged from 2.6 to 2.8 g, surpassing the value (2.1 g) reported by Kougiagka et al. [9] for the fillet of farmed C. a. maximum snails. This notable difference can be attributed to the reduced quantity of feed supplied to the snails by farmers [11]. No statistically significant differences in mean snail fillet mass were observed among treatments of our experiment, although enriched diets showed a tendency toward higher values. In contrast, Apostolou et al. [13] recorded snail fillet mass inside a net-covered greenhouse of 4.51 g, due to higher final (17.74 g) weight of snails.
Mortality rate remained low throughout the experiment, especially in the experimental treatments fed with an enriched diet. These findings demonstrated an improvement in snail in snails’ viability, reducing the mortality rate through the incorporation of nanoemulsion. Similar results were obtained by Lemjallad et al. [37] and El Khayari et al. [40], where the mortality rate was lower in groups fed with an enriched diet. The significantly increased mortality observed in the T3 group during the final two weeks of the experiment (approximately twice that of the other groups) may be associated with the higher nanoemulsion dose incorporated into the diet. A similar effect has already been demonstrated by Ali et al. [41] involving snails of E. vermiculata and Monacha obstructa species.
Diet enrichment did not affect moisture and ash content. Regarding fat content, fillet from snails fed the T1 and T3 diets exhibited increased levels, whereas Ranucci et al. [29] reported that dietary enrichment reduced fat content in pork meat.
Snail fillets from T1 and T3 groups exhibited higher protein content, a finding that may be explained by enhanced protein digestibility in enriched diets, as suggested by previous studies. Dias Junior et al. [42] reported increased crude protein digestibility in lambs fed with diets containing increasing levels of essential oils. Similarly, Cheng et al. [43] observed that supplementation with oregano essential oil in chicken diet may enhance muscle protein deposition by improving crude protein absorption. In contrast, no changes in protein content were reported in chicken and beef meat following dietary enrichment with a mixture of essential oils, including oregano and rosemary, incorporated in a nanoemulsion [28, 30].
Mc Donnell et al. [33] investigated the adverse effects of pesticides on snail welfare, noting that snails are frequently regarded as pests. The authors examined 11 essential oils to determine the optimal oil concentration for controlling C. a. aspersum. Essential oils are known to have molluscicidal properties. Similarly, Ferreira et al. [31, 32] reported that thymol negatively affects the growth, reproduction, and survival of Subulina octona and Bradybaena similaris.
Although comparisons with studies conducted in vertebrates such as poultry and pigs may provide useful indications regarding potential mechanisms, these findings should be interpreted with caution due to the physiological differences between vertebrates and gastropods.
As regards the histomorphology findings, enrichment of snail feed with the T1 diet did not result in notable alterations in digestive or calcium cells. At the end of the feeding trial, a limited number of calcium cells were observed, without a significant presence of calcium spherules. Calcium spherules are considered part of a complex intracellular detoxification mechanism [44]. According to Delakorda et al. [45], calcium spherules in digestive gland cells are intracellular membrane-bound structures, and their structure and chemical composition may naturally vary depending on environmental conditions and the snail’s life cycle. The lumen of the digestive tubules appeared slightly enlarged, consistent with observations reported by Ziegler et al. [46] in a study investigating the effects of the antidepressants citalopram and venlafaxine on the aquatic snail Planorbarius corneus.
In snails fed the T2 diet, apoptotic phenomena were observed in epithelial digestive cells, accompanied by a reduction in calcium cells and their content. Abdel-Azeem et al. [47], who exposed Helix aspersa snails to titanium dioxide nanoparticles at two concentrations via spraying, reported luminal dilation and the presence of cellular blebs at the lower concentration, indicative of impending cell death. Rearrangement of remaining cells and the generation of new cells to fill the gaps left by dead cells contribute to the maintenance of epithelial structural integrity [48]. In the present study, a few calcium cells were observed in the T2 group, and these did not contain well-formed spherules.
These findings are consistent with those reported by Hamed et al. [49], who observed in land snail E. vermiculata a marked reduction in both the number of calcium cells and the size of calcium spherules following administration of carbamate-treated baits, although this was accompanied by complete degeneration of the tubular epithelium. In contrast, in our study, the digestive tubules maintained their overall structural integrity.
In snails fed the T3 diet, necrosis of both digestive and calcium cells was observed. Cellular swelling and necrotic changes were evident across all cell types, and excretory cells were scarcely identifiable within the digestive tubules. Hamed et al. [49] reported that, following fourteen days of exposure to methiocarb-treated baits, the cytoplasm of calcium cells appeared dissolved, while the cytoplasm of digestive cells was completely degenerated, resulting in a severely compromised epithelial structure. Similar observations were reported by Farfar et al. [8], who noted a pronounced reduction in epithelial lining height and extensive necrosis in Cornu aspersum snails exposed to cadmium and copper. Additionally, Mohammadein et al. [50] observed that the boundaries between digestive and calcium cells in the digestive glands of E. vermiculata became indistinguishable between polluted and unpolluted sites, reflecting severe histological disruption.
The results of this study indicate that diet supplementation with essential oil nanoemulsion did not reduce feed acceptability, as snails across all treatments consumed similar amounts of feed regardless of concentration level. Growth performance was not negatively affected during the experimental period, and snails receiving enriched diets exhibited a tendency toward slightly higher fillet mass and increased protein content compared with the control group.
Histopathological evaluation revealed no adverse alterations in the digestive gland tissue of snails fed the lowest supplementation level (T1), whereas higher inclusion levels (T2 and T3) induced histological changes, suggesting that essential oils may have toxic effects when administered at elevated concentrations. These findings highlight the importance of careful dosage selection to balance potential nutritional benefits with animal welfare considerations. While the histological findings provide clear evidence of structural changes in the digestive gland, it should be noted that the analysis was limited to five samples per treatment group. Future studies with larger sample sizes could further validate the extent of these tissue modifications across the entire population. It should be emphasized that the present study evaluated the effects of dietary supplementation during the initial growth phase of snail breeding, from the juvenile stage up to sexual maturation. Reproductive performance, long-term health status, and physiological stress responses were not assessed. Therefore, the observed outcomes cannot be applied to later production stages without further investigation.
Within these limitations, supplementation at 1 mL/kg appeared to be well tolerated during the growth phase and may represent a promising inclusion level for further evaluation. The present study should be regarded as a first step toward defining optimal essential oil supplementation strategies in the diet of farmed C. a. maximum. Future research should focus on long-term trials involving reproduction and welfare indicators to determine whether this supplementation level is suitable for broader commercial application.
C. a. aspersum: Cornu aspersum aspersum
C. a. maximum: Cornu aspersum maximum
E. vermiculata: Eobania vermiculata
MAPs: medicinal and aromatic plants
We gratefully acknowledge Mr. Ioannis Agrokostas (Roi Pelle Snail Farm, Magnesia, Greece) for supplying the snails and feed used in the experiments and the undergraduate students Apostolia Tsitsaki and Ioannis Tzoutzoulis for their assistance during the experimental procedure.
MH: Conceptualization, Validation, Writing—review & editing, Supervision. KA: Investigation, Writing—original draft, Writing—review & editing. EK: Investigation, Writing—original draft, Writing—review & editing. TSK: Investigation, Writing—original draft. CA: Validation, Writing—review & editing. ITK: Conceptualization, Writing—review & editing. DV: Supervision. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
The authors confirm that the ethical policies of the journal, as noted on the journal’s author guidelines page, have been adhered to, and the appropriate ethical review committee approval has been received. The authors confirm that they have followed EU standards for the protection of animals used for scientific purposes and feed legislation. No ethical approval was required for using Cornu aspersum maximum in animal studies. However, our experiments were conducted according to the internal guidelines of the University of Thessaly, Greece.
Not applicable.
Not applicable.
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
This study was part of the project coded ΔΜΡ1-0010874 financed under the Partnership and Cooperation Agreement (NSRF) 2014–2020, the European Regional Development Fund (ERDF), and National Resources, Operational Program entitled “West Macedonia, Action 2014–2020”. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 490
Download: 52
Times Cited: 0
