Affiliation:
1Environmental Parasitology Laboratory, Department of Basic Pathology, Biological Sciences Sector, Federal University of Paraná (UFPR), Curitiba, PR 81531-980, Brazil
ORCID: https://orcid.org/0009-0000-3462-3642
Affiliation:
2Adolfo Lutz Institute, Center for Chemical and Bromatological Sciences, São Paulo 09040-160, Brazil
ORCID: https://orcid.org/0000-0002-1052-8883
Affiliation:
3Morphology and Microscopy Center – Food Center, Adolfo Lutz Institute (IAL), São Paulo 01246-902, Brazil
ORCID: https://orcid.org/0000-0002-6720-736X
Affiliation:
1Environmental Parasitology Laboratory, Department of Basic Pathology, Biological Sciences Sector, Federal University of Paraná (UFPR), Curitiba, PR 81531-980, Brazil
ORCID: https://orcid.org/0009-0007-7789-7599
Affiliation:
1Environmental Parasitology Laboratory, Department of Basic Pathology, Biological Sciences Sector, Federal University of Paraná (UFPR), Curitiba, PR 81531-980, Brazil
ORCID: https://orcid.org/0009-0003-9445-0730
Affiliation:
4Food Hygiene Laboratory, Federal University of Paraná (UFPR), Curitiba, PR 81531-980, Brazil
ORCID: https://orcid.org/0000-0002-6174-392X
Affiliation:
4Food Hygiene Laboratory, Federal University of Paraná (UFPR), Curitiba, PR 81531-980, Brazil
ORCID: https://orcid.org/0000-0001-7904-904X
Affiliation:
1Environmental Parasitology Laboratory, Department of Basic Pathology, Biological Sciences Sector, Federal University of Paraná (UFPR), Curitiba, PR 81531-980, Brazil
Email: diego.leal@ufpr.br
ORCID: https://orcid.org/0000-0001-7838-4006
Explor Foods Foodomics. 2026;4:1010133 DOI: https://doi.org/10.37349/eff.2026.1010133
Received: December 05, 2025 Accepted: March 22, 2026 Published: April 13, 2026
Academic Editor: Zuhaib F Bhat, SKUAST-Jammu, India
Aim: Enteral nutrition (EN) improves patient health. However, the use of fresh produce may increase the risk of parasitic contamination. Recovery of parasites from enteral formulations is challenging and no studies have yet addressed this issue. The primary goal of this study was to standardize methodologies for detecting helminth eggs in different enteral formulations prepared with fresh produce, aiming to establish a reproducible protocol for food safety assessments.
Methods: Two homemade enteral preparations (HEP) with mixed raw fresh fruits or vegetables were produced and artificially contaminated with two doses of Ascaris suum eggs (1 = 207 and 2 = 76 eggs). HEP 1 contained cabbage, orange juice, lettuce, watercress, and filtered water, while HEP 2 consisted of strawberries and filtered water. To estimate the egg recovery rate, four protocols per preparation/dose were analyzed in triplicate (48 trials total). The following variables were evaluated: homogenization (manual or using a magnetic stirrer) and dispersion solution (1 M glycine, pH 5.5 or 0.1% Alconox®). All protocols shared the following steps: sedimentation, centrifugation, and total sediment analysis.
Results: The highest recovery efficiency for HEP 1 was achieved with Protocol 2 (glycine + magnetic stirrer), with averages of 66% (Dose 1) and 55% (Dose 2). For HEP 2, Protocol 4 (Alconox® + magnetic stirrer) performed best, yielding 66% (Dose 1) and 52% (Dose 2). Viable eggs of Toxocara sp., and hookworm were naturally detected in HEP 1 and 2, respectively.
Conclusions: This is the first study to standardize and measure the detection sensitivity of a methodology for detecting parasites in enteral formulations. Since most patients receiving these types of food require intensive care, strict quality control is essential, including evaluation of the parasitological quality of EN to avoid exacerbating their already compromised health.
Enteral nutritional therapy (ENT) is typically administered to malnourished patients or those at nutritional risk, aiding in the recovery or maintenance of nutritional status for individuals unable to meet their needs through oral feeding [1]. The use of home enteral nutrition (HEN) is encouraged, as it improves patients’ health outcomes, reduces clinical and nutritional complications, and transitions care from hospitals to households while lowering healthcare costs [2, 3].
Various enteral formulations are used in HEN, including food-based or homemade enteral preparations (HEP), made exclusively from fresh foods; commercial enteral formulas (CEF), composed of isolated nutrients and available ready-to-use or in powder form; and blended enteral preparations (BEP), combining natural foods with commercial formulas [4, 5].
Although CEFs are considered safer in terms of nutritional and hygienic standards, HEPs and BEPs are often preferred due to their customizable nutritional composition, adjustable volume, and lower cost [5, 6].
However, the extensive handling and inclusion of fresh produce in HEPs/BEPs increase the risk of pathogen contamination [7], posing a hidden threat to immunocompromised patients.
While bacterial contamination in HEN formulations has been extensively studied [8–13], no data exist on foodborne parasite contamination, representing a critical gap in HEN safety knowledge. Foodborne pathogens cause 600 million illnesses and 420,000 deaths annually [14], with parasitic diseases affecting 407 million people, including 91.1 million foodborne cases and 52,000 deaths [15]. Notably, helminths rank among the most prevalent global foodborne parasitic hazards [16].
Parasites exhibit high resistance to environmental stressors and disinfection, with common contamination sources including fresh juices, fruits, and vegetables [17–19], frequent ingredients in enteral formulations. Beyond acute diarrhea, intestinal parasites can lead to chronic malnutrition, iron-deficiency anemia, and physical/cognitive impairments, further compromising HEN patients’ already fragile health [20, 21].
Despite the benefits of HEN, no standardized methods or International Organization for Standardization (ISO) norms exist for detecting parasites in enteral formulations. Given this gap and the potential health risks, this study aimed to standardize methodologies for the detection of helminth eggs in HEPs containing fresh produce, enabling future monitoring of HEN safety for vulnerable patients.
To standardize methodologies for the detection of helminth eggs in HEN, we prepared two types of HEPs containing mixed raw fruits or vegetables. These were artificially contaminated with two different doses of a known quantity of Ascaris suum eggs, which served as our model organism. Following contamination, we evaluated multiple protocols to determine the most efficient method for recovering helminth eggs.
Both HEP formulations were developed using standard recipes obtained from services that assist patients using feeding tubes [22]. The base ingredients included cabbage, strawberries, and orange juice as fresh produce. To better replicate real-world conditions, where caregivers frequently add other vegetables or fruits to enteral formulations, we incorporated additional leafy vegetables into some HEPs. This approach also created a more analytically challenging matrix for contamination detection.
The preparations consisted of the following ingredients: HEP 1—two cabbage leaves, four oranges, two lettuce leaves, two watercress branches, two tablespoons of sugar, and 400 mL of filtered water; HEP 2—eight medium strawberries and 400 mL of filtered water.
All the ingredients were previously cleaned under running water and then blended. Both preparations were sieved through a plastic fine-mesh sieve, and 200 mL were placed in first-use plastic bags, making a total of four plastic bags. The bags were sealed and kept refrigerated (below 4ºC) until the artificial contamination trials.
Egg suspension (stock solution) was prepared for protocol standardization [23]. Briefly, fragments of the uterus of the female of A. suum were removed from the final third of the uterine bifurcation, where most fertile eggs are found. The suspension was washed and homogenized in a Petri dish with 0.1% Tween 80 surfactant solution to prevent egg clumps. The solution was then sieved, transferred into two centrifuge tubes, and concentrated by centrifugation at 1,250 × g for 5 minutes. After centrifugation, the supernatants were discarded, and the sediment was transferred into two separate microtubes and stored under refrigeration until use.
To determine the average egg concentration in both purified suspension doses, ten aliquots per dose were counted under an optical microscope [23]. The mean egg counts for the A. suum inoculum were Dose 1 = 207 eggs and Dose 2 = 76 eggs. These doses were used in all artificial contamination trials to evaluate the egg recovery efficiency of each protocol.
A known quantity of A. suum eggs (Dose 1 or Dose 2) was added to each HEP sample. To standardize the method, four protocols were tested in triplicate for each dose/HEP combination, totaling 48 artificial contamination trials (Figure 1).
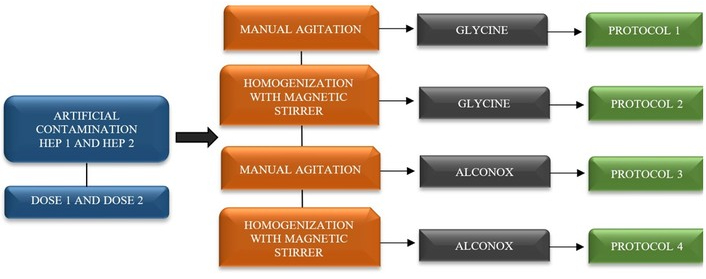
Protocols and variables were analyzed for the standardization of a methodology for detecting helminth eggs in homemade enteral preparations (HEP) containing vegetables or fruits, artificially contaminated with two doses of Ascaris suum.
The protocols compared recovery efficiency using the following variables: 1) Dissociation solution—use of 1 M glycine (Synth, Brasil), pH 5.5 or 0.1% Alconox (Alconox Inc., USA); 2) Homogenization—by manual stirring or magnetic stirrer. All protocols shared the following steps: sedimentation time, centrifugation, and reading the entire sediment.
Initial tests were conducted to determine the optimal sample dilution. A 1:3 ratio of (50 mL of each HEP + 150 mL of 1 M glycine or 0.1% Alconox) was tested first, but the sample was too opaque for proper microscopic analysis. A second test used a 1:7 ratio (25 mL of HEP + 175 mL of glycine or Alconox), which improved clarity.
After this, 25 mL of HEP 1 and 2 were measured and transferred to a first-use plastic bag or to a beaker. Then, 175 mL of 1 M glycine, pH 5.5 or 0.1% Alconox was added. Both inocula were vortexed, and 10 μL of each dose of egg suspension was added at various points of HEP. The contaminated sample in the plastic bag was homogenized using smooth, pendulum-like manual agitation for 3 minutes, while the contaminated sample in the beaker was homogenized using a magnetic stirrer for 10 minutes.
Contaminated samples were transferred to a clean sedimentation cup pre-treated with 0.1% Tween 80 elution solution. To rinse residual material, 50 mL of dissociation solution was used to wash the bag/beaker, and the rinse was added to the sedimentation cup (final volume: 250 mL/sample). Samples were left to sediment at room temperature for 2 hours.
After sedimentation, the supernatant was discarded, and 25 mL of the sample was centrifuged (1,250 × g, 5 minutes). After centrifugation, the supernatant was discarded again, and all the remaining sediment from each artificial contamination test was examined under an optical microscope to enumerate helminth eggs. Recovery efficiency was calculated considering the ratio between the number of A. suum eggs recovered and the number of eggs seeded into each HEP [24].
The best protocol was defined by calculating the average of each triplicate based on the highest egg recovery efficiency rate obtained in HEP 1 and 2 artificially contaminated, considering both doses.
Concomitantly, during the standardization of the methodology, a sample without artificial contamination (negative/blank control) of each HEP was processed to confirm the absence of Ascaris sp. eggs under natural conditions.
During standardization, all HEP samples were screened for naturally occurring helminth eggs. When detected, viability was assessed using the Trypan Blue exclusion test [25]: in an aliquot of 50 μL of sample, an aliquot of 6 μL of 0.1% Trypan Blue (Inlab, São Paulo, Brazil) was added to the slide. The microscopic analysis was performed within 3–5 minutes after preparation [26]. Eggs were deemed viable if no dye uptake occurred.
Descriptive statistics (mean, standard deviation, and percentages) were calculated to summarize the data. The normality assumption was assessed using the Shapiro-Wilk test. Since the variables violated the normality assumption, non-parametric tests were employed for further analysis.
The Kruskal-Wallis test was used to compare protocol performance in standardizing helminth egg detection in HEP and the recovery rates (mean and percentage of eggs recovered) across doses and protocols. A p-value < 0.05 was considered statistically significant. All analyses were performed using Statistica Statsoft version 7® software.
The average egg recovery efficiency rates across the four protocols and two doses (48 total trials) ranged from 47% to 66% for HEP 1 (Table 1) and 40% to 66% for HEP 2 (Table 2).
Recovery efficiency of Ascaris suum eggs in artificially contaminated HEP 1 (leafy vegetables and orange juice) across four processing protocols comparing homogenization methods and dissociation solutions.
| Nº protocol | Nº of eggs recovered trial 1 (%) | Nº of eggs recovered trial 2 (%) | Nº of eggs recovered trial 3 (%) | Final average of recovered eggs (%) | Standard deviation | p1 | p2 |
|---|---|---|---|---|---|---|---|
| Dose 1—207 eggs | |||||||
| 1 | 157 (76) | 129 (62) | 117 (57) | 134 (65) | 20 | 0.01 | 0.110 |
| 2 | 134 (65) | 164 (79) | 112 (54) | 137 (66) | 26 | ||
| 3 | 109 (53) | 106 (51) | 104 (50) | 106 (51) | 2.5 | ||
| 4 | 148 (71) | 117 (57) | 109 (53) | 125 (60) | 21 | ||
| Dose 2—76 eggs | |||||||
| 1 | 38 (50) | 48 (63) | 32 (42) | 39 (52) | 8 | 0.74 | 0.204 |
| 2 | 39 (51) | 42 (55) | 45 (59) | 42 (55) | 3 | ||
| 3 | 39 (51) | 44 (58) | 41 (54) | 41 (54) | 2.5 | ||
| 4 | 38 (50) | 36 (47) | 34 (45) | 36 (47) | 2 | ||
1: Manual agitation with glycine; 2: homogenization with magnetic stirrer and glycine; 3: manual agitation with Alconox®; 4: homogenization with magnetic stirrer and Alconox®; p1: Kruskal-Wallis analysis for dose and protocol used; p2: Kruskal-Wallis analysis for average and % of eggs recovered.
Recovery efficiency of helminth eggs in artificially contaminated HEP 2 (strawberry-based) across four processing protocols comparing homogenization methods and dissociation solutions.
| Nº protocol | Nº of eggs recovered trial 1 (%) | Nº of eggs recovered trial 2 (%) | Nº of eggs recovered trial 3 (%) | Final average of recovered eggs (%) | Standard deviation | p1 | p2 |
|---|---|---|---|---|---|---|---|
| Dose 1—207 eggs | |||||||
| 1 | 124 (60) | 103 (50) | 107 (52) | 111 (54) | 11 | 0.00001 | 0.103 |
| 2 | 114 (55) | 87 (42) | 119 (57) | 107 (51) | 17 | ||
| 3 | 92 (44) | 77 (37) | 80 (39) | 83 (40) | 8 | ||
| 4 | 135 (65) | 99 (48) | 177 (86) | 137 (66) | 39 | ||
| Dose 2—76 eggs | |||||||
| 1 | 40 (53) | 41 (54) | 32 (42) | 38 (50) | 5 | 0.70 | 0.502 |
| 2 | 32 (42) | 40 (53) | 28 (37) | 33 (44) | 6 | ||
| 3 | 28 (37) | 34 (45) | 39 (51) | 34 (44) | 5 | ||
| 4 | 38 (50) | 47 (62) | 34 (45) | 40 (52) | 7 | ||
1: Manual agitation with glycine; 2: homogenization with magnetic stirrer and glycine; 3: manual agitation with Alconox®; 4: homogenization with magnetic stirrer and Alconox®; p1: Kruskal-Wallis analysis for dose and protocol used; p2: Kruskal-Wallis analysis for average and % of eggs recovered.
For HEP 1, Protocol 2 demonstrated the highest recovery rates, using glycine (1 M, pH 5.5) with magnetic stirrer homogenization, achieving 66% for Dose 1 and 55% for Dose 2. Although this method produced a more turbid sample (Figure 2A) compared to the clearer Alconox® solutions (Figure 2B), it was selected as the optimal method for leafy green vegetable HEPs due to its superior recovery performance.

Recovery of A. suum eggs from home enteral preparation 1 (HEP 1) using distinct dissociation solutions. (A) A. suum egg (red arrow), visualized at 40× magnification in HEP 1 (1 M glycine, pH 5.5). (B) A. suum egg (red arrow), visualized at 40× magnification in HEP 1 (0.1% Alconox). Scale: 50 μm.
For HEP 2, Protocol 4 using 0.1% Alconox® with magnetic stirrer homogenization showed the best results, with recovery rates of 66% for Dose 1 and 52% for Dose 2. This protocol was consequently chosen for berry fruit-containing HEPs.
Ascaris sp. eggs were not detected in any blank/negative controls analyzed.
The statistical analysis revealed significant differences between protocols for Dose 1 (207 eggs) in both HEP types (HEP 1: p = 0.01; HEP 2: p = 0.00001), while no significant differences were found for Dose 2 (76 eggs) in either HEP 1 (p = 0.74) or HEP 2 (p = 0.70). Comparison of recovery efficiency between doses within each protocol showed no statistically significant differences for either HEP type: HEP 1 showed p = 0.110 for Dose 1 and p = 0.204 for Dose 2, while HEP 2 showed p = 0.103 for Dose 1 and p = 0.502 for Dose 2.
During the standardization of the methodology, natural contamination by Toxocara sp. eggs were detected in HEP 1, and hookworm eggs in HEP 2. The viability test using Trypan Blue dye revealed that both eggs were viable, as they did not take up the dye.
The presence of pathogens in fresh produce and ingredients commonly used in blenderized formulations in HEN, can pose a threat to human health since this type of food does not undergo thermal treatment [11, 27]. Among them, parasites remain a great challenge because they are difficult to remove from fresh produce, are highly resistant to sanitization processes, and exhibit a low infective dose, amplifying both the likelihood of transmission and the associated public health risks [28, 29]. This is particularly concerning for immunocompromised patients (e.g., immunodeficient or immunosuppressed individuals), since the use of parasite-exposed HEN may exacerbate their clinical condition.
However, the detection of foodborne parasites is usually underreported [30, 31] and faces challenges due to methodological complexity and, in some cases, high isolation costs [32, 33], which can represent a prohibitive cost for regions more vulnerable and susceptible to intestinal parasitosis, such as Latin America.
Moreover, there are currently no regulations assessing parasitic contaminants in complex food matrices like fresh-food enteral formulations. These nutrient-dense mixtures, designed for medically vulnerable patients, combine multiple ingredients that complicate parasite detection.
To the best of our knowledge, this is the first study to standardize and measure the detection sensitivity of two methodologies for detecting parasites in mixed fresh produce used in enteral formulations intended for patients with special healthcare needs.
The standardized protocols for helminth egg detection presented here offer a practical tool that can be readily implemented by both public and private food safety agencies. The protocols would enhance food safety monitoring and reinforce public health protection. Furthermore, a validated method would enable rapid response during foodborne outbreak investigations involving these products.
Effective helminth parasite detection in food involves critical steps, as it requires optimal elution and concentration from the food matrix [34, 35]. While various solutions exist for this purpose, we selected two specific approaches for testing in HEP: a surfactant-based solution (0.1% Alconox®); and an amino acid solution (1 M glycine, pH 5.5).
Glycine is synthesized through alkaline hydrolysis of meat/gelatin using potassium hydroxide and chemically produced from monochloroacetic acid and ammonia [36]. Due to its surfactant properties, glycine effectively interacts with cysts, oocysts, and helminth eggs that are tightly bound to plant matrices, enhancing their detachment [23, 24, 32].
The composition of Alconox® contains a surfactant called sodium dodecylbenzenesulfonate (C12H25C6H4SO3Na) and tetrasodium pyrophosphate (Na4P2O7). The mixture of these two compounds has been shown to effectively remove protozoa from fresh produce [37, 38].
Given that enteral formulations can be made up of different ingredients, and that caregivers of bedridden patients add fresh produce, standardizing and measuring the sensitivity of the methodology by applying different eluent solutions demonstrates the need for adaptation and choice depending on the type of food used in the enteral formulation (whether composed of fruits or vegetables). Our results from artificial contamination experiments show that for HEPs composed mostly of vegetables, the 1 M glycine (pH 5.5) solution should be used for helminth egg identification, as it achieved the highest recovery rates with both doses of A. suum eggs. Similarly, for HEPs composed of berry fruits, the best protocol performance was observed when using the 0.1% Alconox® dissociation solution, regardless of the dose applied.
Previous studies evaluating helminth egg recovery from leafy vegetables have typically employed only one homogenization method [23, 24]. In contrast, our study directly compared two approaches: manual agitation versus magnetic stirring. For both HEP formulations, magnetic stirring yielded superior recovery rates across all doses and dissociation solutions. This advantage likely stems from reduced sample handling and the use of glass beakers, which minimize egg adhesion compared to other materials [39].
In our standardization using HEP 1 (leafy vegetables) and HEP 2 (fruits), we evaluated two contamination levels: a higher dose (207 eggs) and a lower dose (76 eggs). This dual-dose approach strengthens the methodology by demonstrating consistent egg recovery efficiency across different potential contamination scenarios.
The two optimal protocols demonstrated robustness through both high recovery rates and their ability to detect naturally occurring helminth contaminants. During artificial contamination trials using a glycine solution (1 M, pH 5.5) with HEP-containing lettuce, watercress, cabbage, and orange, we identified natural contamination by Toxocara sp. eggs.
Toxocara sp. ranks 20th in the FAO/WHO risk assessment of foodborne parasites [16]. Despite this classification, toxocariasis remains significantly underreported while posing substantial public health challenges. The highest disease burden occurs in Brazil, China, the United States, Korea, Japan, India, Austria, and France [40].
Also, viability tests applied to naturally detected eggs in this study, including Trypan Blue staining, confirmed their viability. This remains the most common viability assessment method due to its cost-effectiveness and simplicity for evaluating Toxocara sp., Ascaris sp., and Trichuris sp. [25, 41]. As a colorimetric reagent, Trypan Blue stains only dead cells (visible under low-resolution microscopy) by penetrating compromised membranes, while viable cells exclude dye [41]. Therefore, the finding of viable eggs, with the potential to infect humans, adds relevance to public health, especially for the vulnerable group that receives this type of food.
Further, the protocol using Alconox® enabled the detection of hookworm eggs in strawberry preparation. While these eggs are non-infective in this stage, their presence indicates fecal contamination [42].
Due to the growing reliance on outpatient nutritional care, HEN has become an increasingly important intervention. Recognized for its cost-effectiveness, HEN optimizes dietary transitions and improves patients’ quality of life [2]. However, epidemiological data from Europe, North America, Australia, and New Zealand reveal that a significant proportion of foodborne illnesses stem from inadequate food preparation practices in household settings [43, 44].
The detection of other genera of helminth eggs during standardization reinforces that washing with running water alone is not sufficient, demonstrating that parasite removal efficiency depends on both processing methods (washing and elution) and is closely related to food matrix characteristics. Fruits and leafy vegetables have irregular, porous, and rough surfaces, which significantly hinder the effective removal of attached parasites [28].
Moreover, the development of methods for detecting and determining the viability of infectious stages of pathogens in food and other matrices provides important tools for identifying and understanding transmission routes and estimating the risk of infection [29]. While our study established standardized methodologies, it would be relevant for future work to perform its validation and evaluate home-prepared formulations, as we could not control ingredient selection by caregivers. However, between main meals, it is common for individuals who require HEN to receive juices containing vegetables and fruits.
This finding highlights the need to establish stricter guidelines regarding the quality of home-prepared enteral formulations, including the specific consideration of the presence of parasites in enteral nutrition (EN), as well as the implementation of rigorous sanitary control measures, to reduce the risk of a wide variety of foodborne diseases.
These guidelines can support decision-making by healthcare professionals, regulatory agencies, caregivers, and public health managers involved in HEN, promoting the improvement of protocols, standards, safe practices, and policies focused on contamination control and risk reduction to health.
The developed methodologies are low-cost, reasonably easy to implement, and proven to be sensitive and efficient for detecting helminth eggs in different enteral formulations composed of fresh vegetables and fruits. The detection of other helminths under natural contamination in fresh produce reinforces the fact that analysis of the presence of parasites in these foods should be recommended, considering the frequency with which they are used, and the absence of studies carried out on this subject. Due to the lack of a standardized method for analyzing parasite contamination in this food matrix, this research could contribute to future analysis of their presence in EN, composed of fresh produce, and provide agility in investigations into possible food contamination or outbreak situations.
BEP: blended enteral preparations
CEF: commercial enteral formulas
EN: enteral nutrition
ENT: enteral nutritional therapy
HEN: home enteral nutrition
HEP: homemade enteral preparations
ISO: International Organization for Standardization
LK: Conceptualization, Data curation, Investigation, Methodology, Writing—original draft, Writing—review & editing, Formal analysis, Visualization. ECdM, MAMM, IARC, JVdA, MRB, and LSF: Data curation, Investigation, Writing—review & editing. DAGL: Conceptualization, Data curation, Investigation, Methodology, Writing—original draft, Writing—review & editing, Supervision, Formal analysis, Visualization. All authors read and approved the submitted version.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
This study did not receive external funding support.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 488
Download: 44
Times Cited: 0
