Affiliation:
School of Food and Liquor Engineering, Sichuan University of Science and Engineering, Yibin 644000, Sichuan, China
Affiliation:
School of Food and Liquor Engineering, Sichuan University of Science and Engineering, Yibin 644000, Sichuan, China
Affiliation:
School of Food and Liquor Engineering, Sichuan University of Science and Engineering, Yibin 644000, Sichuan, China
Affiliation:
School of Food and Liquor Engineering, Sichuan University of Science and Engineering, Yibin 644000, Sichuan, China
Affiliation:
School of Food and Liquor Engineering, Sichuan University of Science and Engineering, Yibin 644000, Sichuan, China
Email: zhaoxingxiu@suse.edu.cn
ORCID: https://orcid.org/0000-0001-6983-8635
Explor Foods Foodomics. 2026;4:1010125 DOI: https://doi.org/10.37349/eff.2026.1010125
Received: June 09, 2025 Accepted: February 25, 2026 Published: March 19, 2026
Academic Editor: José Manuel Lorenzo, Centro Tecnológico de la Carne de Galicia, Spain
Aim: To establish a quantified, practical shelf-life extension protocol for boneless chicken claw (a high-collagen poultry snack for which preservation data are scarce) by combining ozone-water sterilisation with a compound preservative system and predicting shelf life through accelerated shelf-life testing (ASLT).
Methods: Initial ozone-water treatment: 4 mg L–1, 15 min to reduce indigenous microflora. Preservative optimisation: single-factor and orthogonal experiments against a defined mixed spoilage consortium. Kinetic measurements: quality changes monitored at 27°C and 37°C. Shelf-life extrapolation: ASLT-Q10 models used to predict shelf life at 4°C, 25°C, and 30°C.
Results: Ozone alone achieved 87.63% reduction in initial microbial counts and significantly delayed the total volatile basic nitrogen (TVB-N) accumulation and sensory deterioration. Optimum preservative blend (0.20 g kg–1 sodium dehydroacetate, 0.60 g kg–1 sodium diacetate, 0.03 g kg–1 sodium nitrite) inhibited the spoilage cocktail by 99.72% (p < 0.05). ASLT-derived shelf lives: 36 d at 27°C and 24 d at 37°C; Q10 extrapolation gave 91 d at 4°C, 39 d at 25°C, and 31 d at 30°C.
Conclusions: The combined ozone-compound preservative strategy effectively controls spoilage flora and quality deterioration in boneless chicken claw, providing a practical and quantified shelf-life extension tool for the high-collagen poultry-snack sector.
With the change of food culture, boneless chicken claw products, which are convenient to eat, have appeared on the market, and boneless chicken claw products have no restrictions on eating places or environments, and are rich in collagen, vitamins, calcium, iron, and other substances [1], which are deeply loved by consumers. However, at present, boneless chicken claw products have problems such as easily breeding miscellaneous bacteria, short shelf life, and inconvenient storage and transportation, resulting in a waste of chicken claw resources [2].
Ozone sterilization has been widely used in the meat industry as a high-efficiency, green, residue-free, low-cost cold sterilization technology, Karamah and Wajdi [3] found that under low temperature conditions, 0.38 mg/L ozone water treatment of chicken breast for 120 min can effectively reduce the number of aerobic mesophilic bacteria, no effect on chicken pH and water content, and the protein content only decreases by 1%; Kalchayanand et al. [4] used ozone water spray cooling technology to treat fresh beef, which proved that it can effectively inhibit the growth of Escherichia coli and aerobic bacteria on the surface of beef. Ozone sterilization, as an efficient primary sterilization technology, has been widely used in the field of meat processing. However, there are still limitations in its practical application, such as insufficient bactericidal persistence, limited permeability, and a narrow antibacterial spectrum. At present, relevant research mainly focuses on meat products with dense muscle tissue, and there is a lack of systematic research on the applicability of ozone sterilization technology for special ingredients, such as chicken claw that have high collagen content, loose tissue structure, and are easily damaged. Given that the unique tissue structure of boneless chicken claw may have a significant impact on their bactericidal efficiency, this study aims to systematically investigate the synergistic effect of ozone water concentration and treatment time, and further explore its synergistic effect with food preservatives, in order to provide optimized solutions for microbial control of boneless chicken claw.
The application of food preservatives in meat processing is an important technical means to ensure food safety and extend shelf life. The mechanism of nisin and ε-polylysine hydrochloride is to destroy the structure of the cell membrane. Sodium diacetate and sodium dehydroacetate mainly interfere with energy metabolism. The mechanism of sodium nitrite is to inhibit a specific enzyme system, while natamycin acts on cell wall components. Considering the characteristics of boneless chicken claw, such as high protein and high-water activity, preservatives with different mechanisms (nisin, sodium diacetate, sodium nitrite, natamycin, sodium dehydroacetate, and ε-polylysine hydrochloride) can achieve a synergistic antibacterial effect through functional complementarity [5–7].
Numerous studies have shown that the microorganisms that cause spoilage of meat products are mainly Pseudomonas, Enterobacteriaceae, Staphylococcus, Bacillus, and yeasts in the environment [8–10]. Escherichia coli and Staphylococcus aureus are common in poultry spoilage [11, 12]. Pseudomonas is closely related to the increase of the total volatile basic nitrogen (TVB-N) [13]. Bacillus amyloliquefaciens is common in canned and vacuum-packed meat products, while Pichia pastoris and Enterobacteriaceae have a synergistic spoilage effect [14]. Therefore, according to the preservative characteristics of boneless chicken claw products, Bacillus amyloliquefaciens (G+) and Staphylococcus aureus (G+), Escherichia coli (G–) and Pseudomonas (G–), and Pichia pastoris were selected as indicator bacteria for antibacterial testing, and the best combination of preservatives was screened and combined with ozone sterilization technology to prolong the shelf life of boneless chicken claw and reduce resource waste.
Frozen boneless chicken claw (Each boneless chicken claw used in the experiment weighs approximately 60–70 g, and all are from the same batch), factory direct (Bahang Food Development Co., Ltd.), stored in –20°C freezer, thaw it under running water when using. Wasabi-flavored boneless chicken claw product is prepared in the laboratory.
Escherichia coli, Staphylococcus aureus, Pseudomonas, Bacillus amyloliquefaciens, and Pichia pastoris used in the experiment are kept by this laboratory.
Nisin, sodium diacetate, sodium nitrite, natamycin, sodium dehydroacetate, ε-polylysine hydrochloride, all are food-grade additives, Zhejiang Yinuo Biotechnology Co., Ltd. Yeast paste, peptone, glucose, tryptone, yeast extract powder, plate counting agar, all are of analytical grade, Beijing Oboxing Biotechnology Co., Ltd. Sulfuric acid, hydrochloric acid, boric acid, magnesium oxide, sodium chloride, potassium iodide, sodium thiosulfate, all are of analytical grade, Sinopharm Chemical Reagent Co., Ltd.
Purified water, white granulated sugar, monosodium glutamate, salt, lactic acid, oil consumption, pickled pepper, chopped pepper, soybean oil, rattan pepper oil, mustard seasoning oil, Rongrong supermarket. Vacuum packaging bags (food-grade), manufactured by Zhuoan Packaging, Hebei, China.
Electronic balance, HJ-50002, Hangzhou Youheng Double Co., Ltd., China. Vacuum refrigerated centrifuge, RTI6000C, Shanghai Roewe Instrument Co., Ltd., China. Ultra-clean workbench, SW-CJ-IF, Suzhou Antai Air Technology Co., Ltd., China. Ozone generator, GN-S10X, Anhui Jieyu Electronic Technology Co., Ltd., China. Vertical pressure steam sterilizer, LDZX-50FBS, Shanghai Shenshen Medical Device Factory, China. pH meter, desktop PHS-3C, Shanghai Leici, China. Biochemical incubator, LRH-250, Shanghai Qixin Scientific Instrument Co., Ltd., China. Kjeldahl nitrogen determination instrument, K9840, Shandong Haineng Scientific Instrument Co., Ltd., China.
Ozone water with concentrations of 2 mg/L, 3 mg/L, and 4 mg/L was prepared, 0.05% glacial acetic acid was added, respectively, and then boneless chicken claw were sterilized for 10 min, 15 min, and 20 min, respectively, and the sterilization rate of ozone water under different processes was determined. At the same time, the treated boneless chicken claw were grouped according to different treatments and stored at room temperature, and the effects of ozone water treatment on the quality of boneless chicken claw were explored through the sensory evaluation of boneless chicken claw on the 0th and 7th days and the changes of the TVB-N content. The CK group served as the control group without ozone water sterilization treatment. The CY1, CY2, and CY3 groups were treated with ozone water at a concentration of 2 mg/L, while the CY4, CY5, and CY6 groups were treated with ozone water at a concentration of 3 mg/L. The CY7, CY8, and CY9 groups were treated with ozone water at a concentration of 4 mg/L, with treatment durations of 10, 15, and 20 minutes, respectively. The sterilization conditions of ozone water are shown in Table 1.
The different ozone sterilization process conditions.
| Conditions | CK | CY1 | CY2 | CY3 | CY4 | CY5 | CY6 | CY7 | CY8 | CY9 |
|---|---|---|---|---|---|---|---|---|---|---|
| Concentration/mg/L | - | 2 | 2 | 2 | 3 | 3 | 3 | 4 | 4 | 4 |
| Time/min | - | 10 | 15 | 20 | 10 | 15 | 20 | 10 | 15 | 20 |
CK is the control group without ozone water sterilization; CY is the ozone water sterilization treatment group.
The determination of the sterilization rate of ozone water is calculated using the following formula:
Where:
Sr: Sterilizing rate.
Tc: Total number of colonies in the control group.
Te: Total number of colonies in the experimental group.
According to the standardization GB/T 22210-2008 of China [15] and the effect of ozone on the quality of meat products, the sensory evaluation index of boneless chicken claw after ozone water treatment is used to comprehensively evaluate the color, taste, smell and tissue state of boneless chicken claw after sterilization, and the sensory score adopts a 1–5 point system, dissatisfied (1 point), average (2 points), medium (3 points), good (4 points), excellent (5 points), and the scoring criteria are shown in Table 2.
Sensory score standard of boneless chicken claw after ozone treatment.
| Assessment items | Description of the highest score |
|---|---|
| Color | The color is white and uniform, and it is shiny. |
| Palate | The taste is crisp and tender, and it is chewy. |
| Smell | Chicken feet have a unique fragrance and no ozone smell. |
| Organizational status | The organization is tight, the shape is complete, elastic, and not soft. |
TVB-N refers to the total amount of volatile basic nitrogen compounds produced by the decomposition of proteins in food under the action of microorganisms or enzymes. The higher the TVB-N content, the more severe the protein spoilage [16].
Considering the regulatory compliance, broad-spectrum antimicrobial activity, process applicability and safety [17], according to the standardization GB2760-2014 of China “National Food Safety Standard for the Use of Food Additives” on the allowable addition of preservatives and limits for marinated meat products, natamycin (0.30 g/kg), ε-polylysine hydrochloride (0.3 g/kg), nisin (0.5 g/kg), sodium dehydroacetate (0.5 g/kg), sodium nitrite (0.15 g/kg) and sodium diacetate (3 g/kg) were selected to inhibit mixed spoilage bacteria.
The amount of preservatives added: the concentration of preservatives is tested according to the limits of 20%, 40%, 60%, 80%, and 100%, respectively. The limited and added amounts of different preservatives are shown in Table 3.
Limit and addition amount of different preservatives.
| Preservative type | Limit (g/kg) | 20% (g/kg) | 40% (g/kg) | 60% (g/kg) | 80% (g/kg) | 100% (g/kg) |
|---|---|---|---|---|---|---|
| Sodium nitrite | 0.15 | 0.03 | 0.06 | 0.09 | 0.12 | 0.15 |
| Natamycin | 0.30 | 0.06 | 0.12 | 0.18 | 0.24 | 0.30 |
| ε-Polylysine hydrochloride | 0.30 | 0.06 | 0.12 | 0.18 | 0.24 | 0.30 |
| Nisin | 0.50 | 0.10 | 0.20 | 0.30 | 0.40 | 0.50 |
| Sodium dehydroacetate | 0.50 | 0.10 | 0.20 | 0.30 | 0.40 | 0.50 |
| Sodium diacetate | 3.00 | 0.60 | 1.20 | 1.80 | 2.40 | 3.00 |
Compared with a single microorganism, using mixed microorganisms is a convenient and rapid method for detecting the overall sensitivity of microorganisms to preservatives. The mixed microbial suspension was prepared following a previously described protocol with minor modifications; the detailed steps are given below [18]. Take 10 mL of bacterial solution and centrifuge at 8,000 r/min for 10 minutes, wash with sterile physiological saline, and then resuspend and precipitate with 5 mL of physiological saline to obtain a bacterial and yeast suspension of 107 CFU/mL. Mix bacterial suspensions of different strains in equal volumes to obtain a mixture of bacteria and yeast microorganisms.
Inhibition circle: Using the double-layer agar method [19], pour water agar medium into the sterile plate, put the Oxford cup smoothly after cooling, then add the semi-solid medium with the inoculation amount of the indicator fungus suspension of 2%, take out the Oxford cup after cooling, add 200 μL of preservative solution to the hole, take sterile distilled water as the blank control, diffuse at 4°C for 4 h, incubate at 30°C to observe whether the plate has a transparent circle, and measure the diameter of the inhibition circle with vernier caliper.
OD value of bacterial solution: 2% indicator bacterial suspension was inoculated in 10 mL liquid medium, 1 mL of preservative solution of different concentrations was added, 1 mL of sterile water was added to the control group, no indicator bacteria were added as blank, placed at 30°C, incubated at 180 r/min for 24 h, the absorbance value was measured at 600 nm, the number of microorganisms was expressed by OD value, and the inhibition rate was calculated.
The inhibition rate is calculated by the following formula:
Where:
IR: Inhibition rate.
ODc: OD value of bacterial solution in the control group.
ODe: The OD value of the bacterial solution in the experimental group.
According to the results of the single-factor test, three preservatives with good antibacterial effect were selected, and the orthogonal test of three factors and three levels was carried out with the antibacterial rate as the index, and the preservatives were compounded. The orthogonal test factors and levels are shown in Table 4.
Orthogonal test table of compound preservatives.
| Level | Factor | |||
|---|---|---|---|---|
| A (Sodium dehydroacetate/%) | B (Sodium diacetate/%) | C (Sodium nitrite/%) | D (Empty columns) | |
| 1 | 20 | 20 | 10 | 1 |
| 2 | 30 | 30 | 15 | 2 |
| 3 | 40 | 40 | 20 | 3 |
Based on 100 g of sauce, the formula was purified water 29%, white granulated sugar 17.4%, monosodium glutamate 2.9%, salt 2.3%, lactic acid 1.5%, oil consumption 3.4%, pickled pepper 17.4%, chopped pepper 17.4%, soybean oil 8%, rattan pepper oil 0.2%, mustard seasoning oil 0.5%.
Boneless chicken claw were sterilized with ozone water (Based on the optimal conditions determined in Ozone water sterilization process, the samples were treated with 4 mg/L ozonated water for 15 min), mixed with the sauce (Formula of sauce for boneless chicken claw) with compound preservatives at 1:1 (w/v), vacuum-packed, stored at 27°C and 37°C respectively, and the relevant indexes were measured every 3 days.
Accelerated shelf-life testing (ASLT) is widely adopted by the food industry as the standard protocol for predicting the shelf life of ambient-stable products [20, 21]. ASLT shortens the analytical timeframe required for shelf-life determination, and its widespread adoption is largely attributed to readily available temperature-based kinetic models such as the Q10 approach [22]. The ASLT method and Q10 model were used to predict the shelf life of boneless chicken claw. Q10 is a key kinetic parameter used to quantify the effect of temperature on the rate of food quality deterioration. The Q10 method refers to the sensitivity of temperature to the reaction. In general, the rate of food deterioration reaction doubles for every 10°C increase in temperature. Therefore, the boneless chicken claw made of sterilization and preservatives is stored at 27°C and 37°C, respectively (Note: Simulate normal temperature at 27°C and accelerated shelf life test at 37°C), and the ratio of the shelf life of the two temperatures is calculated by the following formula:
Where:
Q10: Temperature coefficient.
T°C: Here is the shelf life at 27°C.
(T+10)°C: Here is the shelf life at 37°C.
Subsequently, the sensory score, microbial index, pH value and TVB-N content of boneless chicken claw stored at 27°C and 37°C were detected, the end point of the shelf life of each group was determined, Q10 was calculated, and then the shelf life of boneless chicken claw at the specified temperature was inferred, and the shelf life was determined by the following formula:
Where:
Qs(T1): Specified shelf life at temperature T1.
Qs(T2): Specified shelf life at temperature T2.
ΔT: The temperature difference between T1 and T2.
Using the above formula, the shelf life of boneless chicken claw at the specified storage temperature can be obtained.
The detection methods for the total number of colonies, Pseudomonas, Enterobacteriaceae, Staphylococcus, Bacillus amyloliquefaciens, and yeasts strictly follow the Chinese national standards (GB 4789 series).
Statistical analysis was performed by using SPSS 21 software. Differences between means were determined by one-way analysis of variance (ANOVA). Differences with p < 0.05 were considered significant. All the experiments were conducted with three replicates.
As shown in Figure 1, ozone water treatment has a significant bactericidal effect on boneless chicken claw. When the concentration of ozone in water is 2 mg/L, the sterilization rate exhibits a significant increase with time. The sterilization rates at 10, 15, and 20 minutes are 67.66%, 74.68%, and 81.59%, respectively, indicating that under the same concentration conditions, extending the treatment time can significantly enhance the sterilization effect. When the sterilization time is 10 minutes, and the concentration of ozone water increases from 2 mg/L to 4 mg/L, the sterilization rate significantly increases from 67.66% to 86.36%, showing a clear concentration dependence.
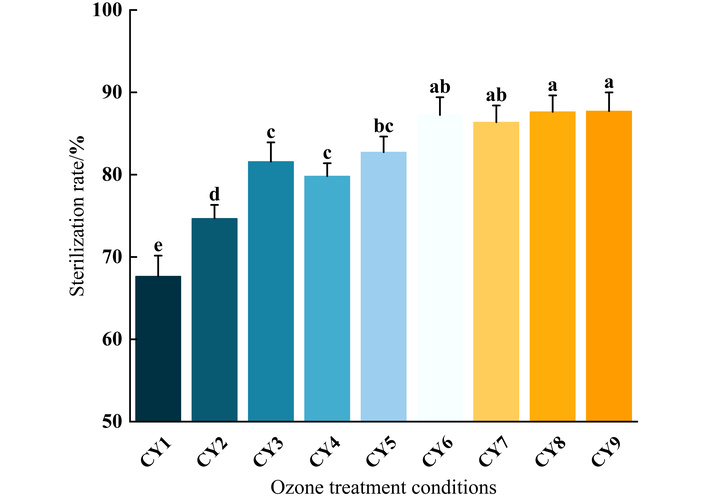
Sterilization rate of different ozone treatment conditions on boneless chicken claws. Different lowercase letters indicate significant differences (p < 0.05) among the treatment groups (CY1–CY9). Repeated superscript letters indicate that the means were not significantly different (p > 0.05). CY is the ozone water sterilization treatment group.
Therefore, when the sterilization time is the same, the higher the concentration of ozone water, the better the sterilization effect. Comparing different treatment groups, it was found that CY7 (4 mg/L, 10 min) and CY8 (4 mg/L, 15 min) groups exhibited the best sterilization efficiency, with sterilization rates of 86.36% and 87.63%, respectively. The results indicate that 4 mg/L ozone water treatment for 10–15 min may be the optimal combination for efficient sterilization.
TVB-N can reflect the degree of protein oxidation, which is an important indicator used to evaluate the freshness of meat products [23, 24]. As can be seen from Figure 2, the TVB-N content of the boneless chicken claw on day 0 was not significantly different from that of the control group. After storing at room temperature for 7 d, the amount of TVB-N content in each group increased to varying degrees, and the TVB-N value in the control group was significantly higher than that of the treatment group, reaching 13.79 mg/100 g, because the boneless chicken foot protein was degraded under the action of microorganisms and enzymes to produce a large number of amines, resulting in the increase of TVB-N value. In the boneless chicken claw treated by ozone water sterilization, most of the microorganisms were killed, and the endogenous enzymes were inactivated, and the TVB-N content increased more slowly. The TVB-N values of CY6, CY7, and CY8 groups were 8.46 mg/100 g, 8.91 mg/100 g, and 8.8 mg/100 g, respectively, which were significantly lower than those of the other treatment groups, indicating that CY6, CY7, and CY8 can effectively delay the increase of TVB-N content in boneless chicken claw.
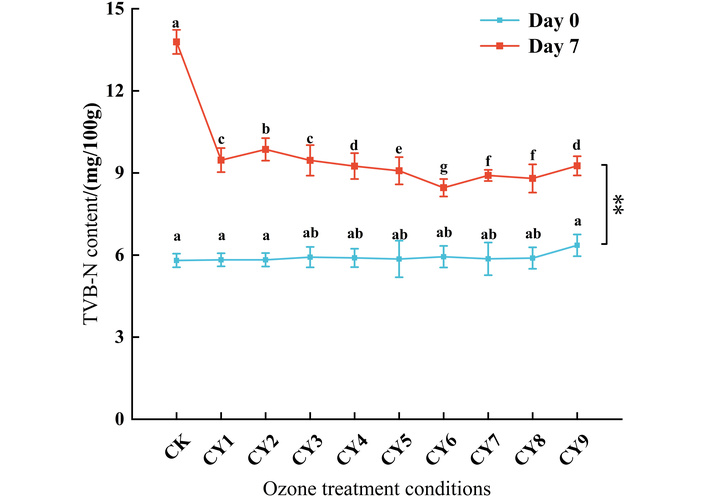
Effects of ozone water with different processes on TVB-N content in boneless chicken claw. Different lowercase letters (e.g., a, b, c, d, e, f) indicate statistically significant differences (p < 0.05) among different treatment groups at various time points (day 0 and day 7). ** indicates that there is a highly significant difference between day 0 and day 7 (p < 0.01). Repeated superscript letters indicate that the means were not significantly different (p > 0.05).
The strong oxidation of ozone makes ozone water bactericidal. At the same time, high-dose ozone treatment may adversely affect the smell and texture of boneless chicken claw [25, 26]. Therefore, we evaluated the sensory score of ozonated samples immediately after treatment (day 0) and after 7 days of ambient storage. As shown in Figure 3, the sensory score of the treatment group (CY1–CY8) on day 0 was not significantly different from that of the control group (CK), while the CY9 group (4 mg/L, 20 min) had obvious ozone residual smell due to the long treatment time, and the sensory score was significantly lower than that of other groups. After 7 days of storage at room temperature, the sensory score of boneless chicken claw decreased in varying degrees, and there were significant differences among the treatment groups. The sensory score of the control group (CK) without sterilization treatment decreased to (3.13 ± 0.06) after storage at room temperature for 7 days. Although there was no significant change in appearance, its elasticity and chewiness decreased under the action of microorganisms and enzymes. Part of the sauce in the boneless chicken claw dissolved, and the sensory score decreased. Groups CY1–CY7 showed compromised sensory scores due to insufficient ozone concentration or exposure time, permitting microbial spoilage. Conversely, the CY9 group suffered accelerated oxidative deterioration from excessive ozone treatment. The sensory score of the CY8 group (4 mg/L, 15 min) on the 7th day was (4.33 ± 0.05), indicating that this process can effectively delay the decline of the sensory score of boneless chicken claw during storage.
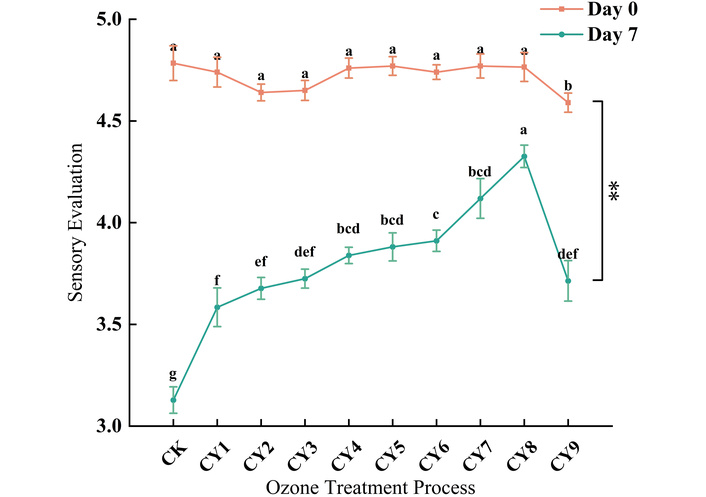
Effects of different process ozone water on the sensory quality of boneless chicken claw. Different lowercase letters (e.g., a, b, c, d, e, f) indicate statistically significant differences (p < 0.05) among different treatment groups at various time points (day 0 and day 7). ** indicates that there is a highly significant difference between day 0 and day 7 (p < 0.01). Repeated superscript letters indicate that the means were not significantly different (p > 0.05).
The effects of ozone water treatment under different processing conditions on the sterilization efficiency, TVB-N value, and sensory score of boneless chicken claw revealed the following findings: The CY7 and CY8 treatment groups achieved a shorter sterilization time while maintaining a higher microbial inactivation rate. Additionally, the CY6, CY7, and CY8 groups exhibited lower TVB-N content in the treated samples. After 7 days of storage at ambient temperature, the CY8-treated group demonstrated the highest sensory score, indicating its effectiveness in retarding protein oxidation in boneless chicken claw. Therefore, based on the comprehensive analysis of sterilization efficiency, TVB-N inhibition, and sensory score, the CY8 treatment (4 mg/L, 15 min) was selected for subsequent experiments.
Sodium diacetate is a new, safe, efficient, and low-toxic preservative that can participate in the normal metabolism of the human body. Its acetic acid molecules can penetrate the cell wall and cause intracellular protein degeneration [27]. Meanwhile, sodium diacetate can improve the taste of food, which has been widely used in meat products. According to Figure 4a, both the antibacterial circle diameter and the antibacterial rate of sodium diacetate for mixed spoilage bacteria increased significantly with the increase of the addition amount. In this test, the antibacterial rate of sodium diacetate against the mixed spoilage bacteria was more than 50%. At 20%, the inhibition rate is 59.39%, the diameter of the antibacterial coil is 12.38 mm; at 40%, the inhibition rate is 61.36%, the diameter of the antibacterial coil is 17.7 mm; when the addition rate is 100%, the inhibition rate is 75.76%, the diameter is 28 mm. This indicates that sodium diacetate was effective in inhibiting mixed spoilage bacteria. ε-Polysine hydrochloride mainly destroys the cell membrane structure of microorganisms, interferes with the intracellular material transfer, redox, and information transfer processes, and eventually leads to cell death [28]. According to Figure 4b, with the increase of ε-polylysine hydrochloride addition, the antibacterial circle diameter and antibacterial rate of mixed spoilage bacteria increased significantly, and the OD value of bacterial fluid showed a significant decrease. When the amount of ε-polylysine hydrochloride was 100%, the diameter of the antibacterial sphere was 13.63 mm, and the OD value of the bacterial solution was 0.683, while the antibacterial rate was only 54.18%, which had a poor inhibitory effect on mixed spoilage bacteria. Sodium dehydroacetate is a new generation of food preservative, showing good inhibitory effect on bacteria, mold, and yeast [29]. According to Figure 4c, the diameter and antibacterial rate of the bacteriostatic sphere of sodium dehydroacetate against mixed spoilage bacteria increased significantly with the increase of the amount added to 100%, the diameter of the bacteriostatic sphere increased significantly to 23.30 mm, and the antibacterial rate was as high as 89.97%. As a new broad-spectrum bacteriostatic agent, sodium dehydroacetate can penetrate into the cell body and inhibit the respiration of microorganisms, so as to achieve the effect of antibacterial preservation. Therefore, dehydroacetate sodium has a significant inhibitory effect on mixed spoilage bacteria [30]. According to Figure 4d, with the increase of sodium nitrite, the inhibitory circle diameter and antibacterial rate of sodium nitrite to mixed spoilage bacteria increased significantly, and the OD value of bacterial fluid showed a significant decrease. When the amount of sodium nitrite is 20% or more, the antibacterial rate of mixed spoilage bacteria is above 60%, when the amount of sodium nitrite is 40% or more, the diameter of antibacterial coil is greater than 20 mm; when the amount is 100%, the antibacterial rate is 79.35% and the diameter is 28.05 mm, which proves that sodium nitrite has significant effect of inhibition on mixed spoilage bacteria. According to Figure 4e, with the increase of natamycin concentration, the inhibitory circle diameter and inhibitory rate of natamycin against mixed spoilage bacteria increased significantly, and the OD value of bacterial fluid decreased significantly. When the added amount reaches 60%, the inhibition rate of mixed spoilage bacteria reached more than 30%; when the addition volume continued to increase to 80% and 100%, 14.14 mm and 14.57 mm, no significant change. At this time, the antibacterial rate was 42.02% and 48.67%, respectively. None have reached more than 50%, because natamycin relies mainly on its lactone ring structure, acting with sterol compounds on the fungal cell membrane. Disrupt the cytoplasmic membrane structure of fungal cells, thus damaging the cell membrane permeability. The inhibitory effect on fungi, such as mold and yeast, and the poor antibacterial effect against bacteria. Therefore, the inhibitory effect of natamycin against both bacteria and yeast is not ideal [31, 32]. It can be seen from Figure 4f that the antibacterial effect of nisin on mixed spoilage bacteria has significant limitations. It was found that the nisin treatment group did not form an obvious inhibition zone. Quantitative analysis showed that there was no significant dose-dependent change in the inhibitory rate of nisin on mixed spoilage bacteria in the range of experimental concentration. Although nisin, as a new biological preservative, showed a strong inhibitory effect on Gram-positive bacteria (G+), its antibacterial activity against Gram-negative bacteria (G–) and yeast was weak. This result is consistent with the existing research conclusion [33], indicating that nisin as a single preservative has no significant inhibitory effect on complex microorganisms.
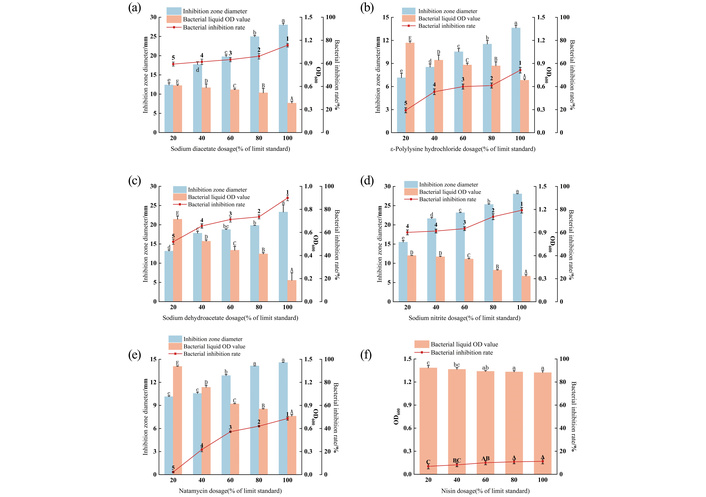
The antibacterial effect of a single preservative. (a) Antibacterial effect of sodium diacetate; (b) antibacterial effect of ε-polylysine hydrochloride; (c) antibacterial effect of sodium dehydroacetate; (d) antibacterial effect of sodium nitrite; (e) antibacterial effect of natamycin; (f) antibacterial effect of nisin.
Different preservatives have different sources, antibacterial mechanisms, and antibacterial spectrum ranges, and the use of a single preservative often does not achieve a good antibacterial effect [25]. In the food industry, two or more preservatives are usually added to the food in a certain proportion to extend the shelf life. According to the results of a single preservative antibacterial experiment, sodium nitrite, sodium dehydroacetate, and sodium diacetate have an obvious inhibitory effect on the mixed spoilage bacteria that easily appear in boneless chicken claw products. Therefore, sodium dehydroacetate (A), sodium diacetate (B), and sodium nitrite (C) were selected as independent variables, and a three-factor three-level orthogonal experiment was conducted with antibacterial rate as the evaluation index. The results of the orthogonal experiments are presented in Table 5, while the ANOVA results are shown in Table 6.
Orthogonal test results of compound preservatives.
| Order number | Factor | ||||
|---|---|---|---|---|---|
| A (Sodium dehydroacetate) | B (Sodium diacetate) | C (Sodium nitrite) | D (Empty column) | Inhibition rate/% | |
| 1 | 20% | 20% | 10% | 1 | 75.40 |
| 2 | 20% | 30% | 15% | 2 | 86.71 |
| 3 | 20% | 40% | 20% | 3 | 93.24 |
| 4 | 30% | 20% | 20% | 2 | 82.09 |
| 5 | 30% | 30% | 10% | 3 | 80.84 |
| 6 | 30% | 40% | 15% | 1 | 94.90 |
| 7 | 40% | 20% | 15% | 3 | 80.42 |
| 8 | 40% | 30% | 20% | 1 | 90.41 |
| 9 | 40% | 40% | 10% | 2 | 98.53 |
| K1 | 84.70 | 79.30 | 84.92 | 86.90 | |
| K2 | 85.94 | 85.57 | 86.92 | 88.69 | |
| K3 | 89.78 | 95.56 | 88.85 | 84.83 | |
| R | 5.08 | 16.26 | 3.93 | 3.86 | |
| Level of factor | B > A > C | ||||
| Optimal level | A3B3C3 | ||||
Results of variance analysis.
| Factor | Sum of squares of deviations | Free degree | Mean square | F | Conspicuousness (p) |
|---|---|---|---|---|---|
| Sodium dehydroacetate | 111.717 | 2 | 55.859 | 12.504 | < 0.0001 |
| Sodium diacetate | 1,201.612 | 2 | 600.806 | 134.492 | < 0.0001 |
| Sodium nitrite | 62.336 | 2 | 31.168 | 6.977 | 0.005 |
Note: 0.01 < p < 0.05 is significant, and p < 0.01 is highly significant.
As shown in Table 5, the order of influence of different preservatives on the antibacterial rate against mixed spoilage bacteria was: B (sodium diacetate) > A (sodium dehydroacetate) > C (sodium nitrite). Table 6 compares the F-values and reveals that the order of influence of different preservatives on the antibacterial rate against mixed spoilage bacteria was: sodium diacetate > sodium dehydroacetate > sodium nitrite. This result is consistent with the range analysis from the orthogonal experiments. The significance analysis indicates that sodium dehydroacetate, sodium diacetate, and sodium nitrite all have extremely significant effects (p < 0.01) on the antibacterial rate against mixed spoilage bacteria. By comparing the K value in Table 5, the best compound preservative combination is A3B3C3, which consists of: sodium dehydroacetate: 0.20 g/kg (40% of the maximum permitted level), sodium diacetate: 0.60 g/kg (40% of the maximum permitted level), sodium nitrite: 0.03 g/kg (20% of the maximum permitted level). This combination complies with the regulatory requirement that when multiple food additives with the same function are used together, the sum of their individual usage ratios relative to their maximum permitted levels should not exceed 1. However, since this specific combination did not appear in the orthogonal experimental results, further verification tests are required to confirm its efficacy.
Validation tests conducted under the A3B3C3 optimal conditions demonstrated that the composite preservative combination achieved an antibacterial rate of 99.72% against mixed spoilage bacteria, surpassing the maximum antibacterial rate (98.53%) observed in the orthogonal experimental results. Therefore, the optimal composite preservative formulation was determined to be: sodium dehydroacetate 0.20 g/kg, sodium diacetate 0.60 g/kg, and sodium nitrite 0.03 g/kg.
According to Table 7, at the same storage temperature, the quality of boneless chicken claw undergoes significant changes with prolonged storage time. With the extension of storage time, the color of the product deepens, collagen dissolution occurs, and changes in taste and flavor occur, resulting in a significant decrease in sensory score. The pH range of commercially available boneless chicken claw products is 3.5–4.5, which is consistent with the pH range of the mustard-flavored boneless chicken claw product developed in this experiment. The acidic substances, such as lactic acid and pickled peppers, added to the product give it an initial pH value of (4.58 ± 0.005). The storage experiment results showed that the pH value of the product showed a significant downward trend with prolonged storage time (p < 0.05), mainly due to the growth and reproduction of acid-producing microorganisms and their metabolic acid production during storage. Meanwhile, the TVB-N content showed a significant upward trend (p < 0.05) during storage, mainly due to the growth and reproduction of microorganisms and the degradation of proteins by extracellular proteases secreted by them. Under storage conditions of 27°C, the total number of colonies in the sample exceeded the standard on the 39th day. At this time, the pH value of the product was (3.70 ± 0.012), and the TVB-N content was (14.55 ± 1.14) mg/100 g. Sensory scores showed obvious signs of spoilage. Based on a comprehensive evaluation of microbial indicators and sensory score, the shelf life of the product at 27°C is determined to be 36 days.
The changes in quality indexes of boneless chicken claws at 27°C.
| Storage time/d | Total number of colonies/(CFU/g) | Escherichia coli | Pathogenic bacteria | pH | TVB-N/(mg/100 g) | Sensory score |
|---|---|---|---|---|---|---|
| 0 | < 1 | - | - | 4.58 ± 0.005 | 4.57 ± 0.54 | 96.4 ± 0.8 |
| 3 | 8 ± 3 | - | - | 4.48 ± 0.014 | 5.59 ± 0.11 | 92.4 ± 1.4 |
| 6 | 60 ± 6 | - | - | 4.33 ± 0.005 | 5.78 ± 0.15 | 90.6 ± 1.6 |
| 9 | 120 ± 9 | - | - | 4.28 ± 0.005 | 7.74 ± 1.08 | 85.7 ± 1.8 |
| 12 | 390 ± 29 | - | - | 4.27 ± 0.005 | 8.84 ± 0.25 | 81.4 ± 1.6 |
| 15 | 640 ± 35 | - | - | 4.26 ± 0.005 | 9.53 ± 0.25 | 78.6 ± 0.9 |
| 18 | 930 ± 46 | - | - | 4.25 ± 0.009 | 10.75 ± 1.22 | 76.0 ± 1.6 |
| 21 | 1,190 ± 61 | - | - | 4.16 ± 0.005 | 11.62 ± 1.10 | 71.5 ± 0.7 |
| 24 | 2,230 ± 95 | - | - | 4.18 ± 0.005 | 12.27 ± 1.07 | 63.1 ± 2.5 |
| 27 | 3,820 ± 112 | - | - | 4.03 ± 0.008 | 13.68 ± 1.05 | 60.2 ± 2.5 |
| 30 | 5,500 ± 137 | - | - | 4.06 ± 0.009 | 13.9 ± 1.06 | 55.0 ± 1.7 |
| 33 | 7,350 ± 219 | - | - | 3.99 ± 0.008 | 14.11 ± 1.06 | 49.0 ± 1.5 |
| 36 | 9,600 ± 268 | - | - | 3.81 ± 0.008 | 14.16 ± 1.04 | 44.2 ± 1.4 |
| 39 | Super | - | - | 3.70 ± 0.012 | 14.55 ± 1.14 | Metamorphic |
Pathogenic bacteria (Staphylococcus aureus, Pseudomonas, Bacillus amyloliquefaciens). “Super” indicates that the total number of colonies exceeds 10,000 CFU/g. “-” indicates that the bacteria are not detected. “< 1” indicates that all dilutions (including liquid sample stock) plates are sterile, and colony growth is then calculated as less than 1.0 multiplied by the lowest dilution.
It can be seen from Table 8 that the total number of colonies of boneless chicken claw stored at 37°C exceeded the standard on the 27th day, the pH value was (3.82 ± 0.011), and the TVB-N content was (12.34 ± 1.07) mg/100 g. The sensory score showed that they had no edible value. The comprehensive evaluation results determined that the shelf life of the product at 37°C was 24 days. The Q10 value of boneless chicken claw was calculated according to the formula as follows:
The changes in quality indexes of boneless chicken claws at 37°C.
| Storage time/d | Total number of colonies/(CFU/g) | Escherichia coli | Pathogenic bacteria | pH | TVB-N/(mg/100 g) | Sensory score |
|---|---|---|---|---|---|---|
| 0 | < 1 | - | - | 4.58 ± 0.005 | 4.57 ± 0.54 | 96.4 ± 0.8 |
| 3 | 13 ± 2 | - | - | 4.43 ± 0.005 | 5.67 ± 0.19 | 91.4 ± 1.5 |
| 6 | 200 ± 36 | - | - | 4.33 ± 0.009 | 6.55 ± 0.25 | 86.8 ± 0.9 |
| 9 | 300 ± 10 | - | - | 4.22 ± 0.008 | 8.91 ± 1.09 | 78.7 ± 1.6 |
| 12 | 660 ± 18 | - | - | 4.17 ± 0.009 | 9.67 ± 0.16 | 72.5 ± 0.6 |
| 15 | 1,000 ± 52 | - | - | 4.15 ± 0.012 | 10.16 ± 1.09 | 69.6 ± 1.0 |
| 18 | 3,980 ± 142 | - | - | 4.12 ± 0.008 | 10.85 ± 1.05 | 63.5 ± 1.1 |
| 21 | 6,410 ± 369 | - | - | 4.08 ± 0.008 | 11.26 ± 1.04 | 59.4 ± 2.3 |
| 24 | 8,770 ± 129 | - | - | 3.92 ± 0.005 | 11.27 ± 1.11 | 46.3 ± 2.3 |
| 27 | Super | - | - | 3.82 ± 0.011 | 12.34 ± 1.07 | Metamorphic |
Pathogenic bacteria (Staphylococcus aureus, Pseudomonas, Bacillus amyloliquefaciens). “Super” indicates that the total number of colonies exceeds 10,000 CFU/g. “-” indicates that the bacteria are not detected. “< 1” indicates that all dilutions (including liquid sample stock) plates are sterile, and colony growth is then calculated as less than 1.0 multiplied by the lowest dilution.
The shelf life of boneless chicken claw at 4°C, 25°C, and 30°C was subsequently predicted through formula calculation as follows:
4°C:
25°C:
30°C:
In summary, the shelf life of boneless chicken claw was predicted to be 91 days at 4°C, 39 days at 25°C, and 31 days at 30°C through the ASLT method combined with the Q10 model.
This study optimized the preservation process of boneless chicken claw by using ozone water to kill bacteria and compound preservatives, and evaluated the quality changes and shelf life at different storage temperatures. The results showed that treating with 4 mg/L ozone water for 15 minutes can achieve a sterilization rate of 87.63%, and the inhibition rate of composite preservatives (sodium dehydroacetate 0.20 g/kg, sodium diacetate 0.60 g/kg, sodium nitrite 0.03 g/kg) on mixed spoilage bacteria reached 99.72%. The shelf-life prediction based on the ASLT method and Q10 model shows that the shelf life of boneless chicken claw is 36 days and 24 days at 27°C and 37°C, respectively; while the shelf life at 4°C, 25°C, and 30°C is 91 days, 39 days, and 31 days, respectively. This study provides a feasible technical solution for the preservation of boneless chicken claw, which is conducive to extending the shelf life of products and ensuring food safety. Future research can further optimize processing parameters and combine them with intelligent packaging or modified atmosphere preservation technology, which may achieve the effect of improving the storage stability of boneless chicken claw.
ASLT: accelerated shelf-life testing
OD: optical density
TVB-N: total volatile basic nitrogen
YW: Methodology, Investigation, Conceptualization, Validation, Formal analysis, Data curation, Writing—original draft, Writing—review & editing. YH: Conceptualization, Resources, Supervision, Project administration, Writing—review & editing. JZ: Investigation, Supervision, Writing—review & editing, Methodology. ZZ: Investigation, Conceptualization, Methodology. XZ: Funding acquisition, Investigation, Visualization. All authors read and approved the submitted version.
The authors have declared no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
Supported by the Zigong concept verification project [No.: 2024-GNYZ-05] and the Scientific Research and Innovation Team Program of Sichuan University of Science and Engineering [SUSE652A009]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 683
Download: 51
Times Cited: 0
