Affiliation:
Department of Microbial Biotechnology, Rajiv Gandhi Institute of Information Technology and Biotechnology, Bharati Vidyapeeth (Deemed to be University), Pune 411045, Maharashtra, India
ORCID: https://orcid.org/0009-0005-0208-6017
Affiliation:
Department of Microbial Biotechnology, Rajiv Gandhi Institute of Information Technology and Biotechnology, Bharati Vidyapeeth (Deemed to be University), Pune 411045, Maharashtra, India
Email: rama.bhadekar@bharatividyapeeth.edu
ORCID: https://orcid.org/0000-0002-4452-9462
Explor Foods Foodomics. 2026;4:1010126 DOI: https://doi.org/10.37349/eff.2026.1010126
Received: December 23, 2025 Accepted: February 14, 2026 Published: March 20, 2026
Academic Editor: Filomena Nazzaro, CNR, Italy
Aim: This study aimed to evaluate the potential of a lactic acid (LA) bacteria (LAB) co-culture during solid-state fermentation (SSF) of yellow peas and soybeans, with an emphasis on the production of health-promoting metabolites from legumes.
Methods: A synergistic probiotic consortium comprising L. acidophilus, L. plantarum, and L. rhamnosus was employed for SSF across 11 different legume substrates at three legume-to-water ratios over 48 h to identify optimal fermentation conditions. Based on microbial growth outcomes, yellow peas and soybeans were selected for further SSF studies at an optimized substrate-to-water ratio (1:3). Fermentation performance was assessed by monitoring microbial growth, pH changes, production of LA, short-chain fatty acids, B-vitamins, and antimicrobial activity. Data were statistically analysed using two-way ANOVA. Structural modifications of fermented substrates and bacterial colonization were examined using scanning electron microscopy (SEM).
Results: SSF exhibited distinct substrate-specific biochemical patterns. Yellow peas supported rapid LAB proliferation (16.04 log10 CFU/g) and pronounced acidification (pH 7.33→4.66), reflecting their high fermentable carbohydrate content. In contrast, soybean yielded higher LA production (7.28 g/L) despite lower viable counts, indicating enhanced per-cell metabolic activity. Short-chain fatty acid synthesis was also influenced by substrate composition and pH, with soybean showing maximum accumulation of acetic acid (11.73 g/L) and propionic acid (15.95 g/L). Butyric acid was detected at lower levels in both substrates. B-vitamin biosynthesis was also substrate-dependent: yellow peas produced higher levels of vitamin B2 (7.097 µg/mL) and B9 (3.69 µg/mL), whereas soybeans favoured vitamin B12 synthesis (2.187 µg/mL). Fermented extracts exhibited strong antimicrobial activity against Escherichia coli, Salmonella typhi, and Staphylococcus aureus, with inhibition zones reaching up to 28, 26, and 18.6 mm, respectively. SEM analysis revealed matrix erosion and aggregation of LAB colonization.
Conclusions: SSF is an efficient and sustainable strategy for producing multifunctional probiotic-enriched fermented legumes with enhanced nutritional and antimicrobial properties.
Environmentally, legumes are highly sustainable, requiring lower water and nitrogen inputs compared to animal or cereal-based protein sources, making them key candidates for addressing both nutritional and ecological concerns [1, 2]. Legumes offer a rich nutritional profile, comprising high-quality proteins (20–45%), dietary fibre (5–37%), complex carbohydrates (65–70%), 85% of which is resistant starch, polyunsaturated fatty acids (66.1–85.3% of total lipids), and polyphenolic antioxidants [3–5]. However, the nutritional potential of legumes is frequently compromised by the presence of anti-nutritional factors (ANFs), such as lectins, phytates, tannins, trypsin inhibitors, and oligosaccharides of the raffinose family. These compounds reduce protein digestibility, hinder mineral absorption, and cause gastrointestinal discomfort [6]. Moreover, the characteristic “beany” flavour of legumes may limit consumer acceptance, necessitating processing techniques that enhance both nutritional bioavailability and sensory appeal [7]. Fermentation, particularly with lactic acid (LA) bacteria (LAB) such as Leuconostoc, Lactiplantibacillus, Lacticaseibacillus, and Limosilactobacillus, has emerged as an effective strategy to overcome these limitations, facilitating the enzymatic hydrolysis of complex carbohydrates, degrading ANFs, and synthesizing bioactive peptides [7–9]. LAB can further synthesize essential B-complex vitamins, especially riboflavin (B2), folate (B9), and cobalamin (B12) [10], addressing key micronutrient limitations often associated with plant-based substrates.
Among fermentation modalities, solid-state fermentation (SSF) is particularly promising for legume processing. SSF enables microbial growth on moistened solid substrates with minimal water content, reducing energy and downstream processing demands while promoting nutrient preservation [11]. Previous studies have shown that LAB-mediated SSF can enhance the formation of functional metabolites across a range of substrates. In Spirulina, LAB-SSF increases protein-derived bioactive compounds such as GABA, L-glutamic acid, and biogenic amines [12]. Similarly, the SSF of A. argyi leaves elevated flavonoid, polyphenol, and related antioxidant activities [13]. Building upon these findings, the present study explored the application of LAB-mediated SSF to legume-based substrates. Legumes are widely consumed worldwide. Crops such as soybeans (SBs), peas, chickpeas, and beans are staple components of Mediterranean and other traditional plant-based dietary patterns, which are widely associated with improved metabolic and cardiovascular health outcomes [14] and represent promising fermentation matrices due to their favourable nutritional composition, including protein (e.g., 36.5 g/100 g in soybean), dietary fibre (e.g., 4.2 g/100 g in black beans), and carbohydrate content (e.g., 60.4 g/100 g in chickpeas) [15]. The global shift toward sustainable plant-based diets has created a strong demand for bioprocess strategies that enhance the nutritional and functional properties of legumes [16].
Short-chain fatty acids (SCFAs), such as acetate, propionate, and butyrate, produced through LAB-mediated fermentation, play vital roles in gut health by serving as energy sources for colonocytes, reducing inflammation, and improving epithelial integrity [17, 18]. The prebiotic carbohydrates present in legumes can stimulate SCFA production during gut fermentation, and this effect is increased when legumes undergo LAB-mediated pre-fermentation [19, 20]. Concurrently, LA synthesis ensures inhibition of common foodborne pathogens and spoilage-causing microorganisms. In addition to acidification, LAB fermentation yields bioactive antimicrobials such as hydrogen peroxide and bacteriocins [21–23].
Despite the growing interest in bioprocessing, the majority of studies have emphasized submerged fermentation or the enhancement of a single metabolite [10, 21]. There remains a significant knowledge gap regarding the comparative evolution of multiple legume matrices under SSF, particularly using LAB consortia for multifunctional transformation. Specifically, the synergistic enhancement of SCFAs, vitamin biosynthesis, antimicrobial activity, and probiotic viability within a single SSF system has rarely been addressed. Furthermore, while fermentation is inherently dynamic, most research relies on endpoint analysis, leaving the systematic, time-dependent evolution of metabolites in SSF systems insufficiently explored. In view of this, eleven legumes, including SB, yellow pea (YP), white kidney bean (WKB), mung bean or green gram (GM), cowpea (BEB), black kidney bean (BKB), lima bean (LB), black gram (BG), black chickpea (KC), field bean (FB), and horse gram (HG), were initially screened in the present study to evaluate their suitability as substrates for LAB-mediated SSF. Selected matrices demonstrating favourable fermentation performance were further investigated in detail. The consortium comprising L. acidophilus (Lactobacillus acidophilus), L. plantarum [Lactobacillus plantarum (Lactiplantibacillus plantarum)], and L. rhamnosus [Lactobacillus casei var. rhamnosus (Lacticaseibacillus rhamnosus)], selected here, is based on prior evidence of its superior synergistic antimicrobial activity against a wide range of pathogens compared to individual strains [24]. This consortium also demonstrates enhanced efficacy in the presence of prebiotic co-substrates [25] and exhibits multi-enzyme activities, including anti-proteolytic, anti-collagenase, and anti-elastase effects, and significant anti-angiogenic properties [26]. The results would contribute to the development of sustainable, functional legume-based fermentations with applications in both food biotechnology and health-oriented biomanufacturing.
Analytical-grade LA, acetic acid (AA), and propionic acid (PA), vitamins: B2 and B9, HPLC-grade acetonitrile, trifluoroacetic acid, and orthophosphoric acid, were purchased from SRL Chemicals (India). The analytical grade butyric acid (BA) was obtained from Sigma-Aldrich (St. Louis, MO, USA), and B12 was sourced from HiMedia Laboratories (India). Analytical-grade sulfuric acid was obtained from Merck (India). Ascorbic acid, ethylenediaminetetraacetic acid (EDTA), and sodium dihydrogen phosphate were obtained from HiMedia Laboratories (India).
L. acidophilus NCIM (National Collection of Industrial Microorganisms) 2660 (ATCC 11975), L. plantarum NCIM 2374 (NCIB 6376), and L. rhamnosus NCIM 2364 (ATCC 7469) were procured from the NCIM, National Chemical Laboratory (NCL), Pune, India. Each strain was individually revived in de Man, Rogosa, and Sharpe (MRS) broth (HiMedia, India) and incubated at 37°C for 24 h. Equal volumes of the active cultures were mixed in a 1:1:1 ratio to form a consortium, as described by Bhola and Bhadekar (2019) [24]. The inoculum was standardised to ~1010 CFU/mL by serial dilution and plate counting and stored at 4°C. A 0.2% (v/v) inoculum of this consortium was used in all fermentation experiments. Uninoculated media were used as controls in all experiments. Clinical isolates of E. coli, S. aureus, and S. typhi were obtained from Bharati Hospital, Pune, India, and maintained on Mueller–Hinton (MH) medium (HiMedia, India).
Eleven legumes, including SB, YP, WKB, GM, BEB, BKB, LB, BG, KC, FB, and HG, representing the botanical species Glycine max, Pisum sativum L., Phaseolus vulgaris, Vigna radiata, Vigna unguiculata, Phaseolus vulgaris L., Phaseolus lunatus, Vigna mungo, Cicer arietinum, Lablab purpureus, and Macrotyloma uniflorum, respectively, were procured from the local market and washed thoroughly with sterile distilled water. Coarse flour was prepared from each legume for further analysis. The legume flours were hydrated in distilled water at three different ratios (1:1, 1:2, and 1:3 w/v) [27], and supplemented with 5% (w/w) sucrose (SRL, India) [28] to support the bacterial growth. The mixtures were autoclaved at 121°C for 15 min, cooled to room temperature, and inoculated with the LAB consortium. SSF was conducted in sterile Petri plates at 37°C for 48 h. Total viable counts (TVCs) were determined post-fermentation, and legumes were selected for further study based on their microbial growth performance.
Fermentation experiments were conducted using selected formulations inoculated with the LAB consortium under static conditions. Samples were collected at 24-hour intervals over a 5-day period and analysed for viable cell counts, pH, LA concentration, SCFA profile, B-complex vitamins (B2, B9, and B12), and antimicrobial activity as described below.
Bacterial growth was assessed by plating serial dilutions of homogenised fermented samples on MRS agar, followed by incubation at 37°C for 48 h [29]. Results were expressed as log10 CFU/g.
LA and SCFAs: AA, PA, and BA were quantified using a Shimadzu LC-20AT HPLC system equipped with a photodiode array (PDA) detector. Separation was achieved on a reversed-phase C18 column (Shim-Pack Solar C18, 5 μm, 4.6 mm I.D. × 250 mm) at room temperature. Cell-free supernatants (CFSs) were acidified to pH 1.8–2.0 using 0.1 M H2SO4 prior to injection. The mobile phase consisted of Solvent A (1% acetonitrile in 20 mM sodium dihydrogen phosphate, pH 2.2) and Solvent B (acetonitrile). The gradient elution program was adapted from Kulshreshtha et al. (2012) [30]. The flow rate was 1.5 mL/min, and detection was performed at 210 nm. Quantification was based on external calibration using standard solutions (50–200 mg/L) of AA, LA, PA, and BA.
Cell lysate (CL) fractions were used for B-vitamin quantification. A 500 μL aliquot of each lysate was mixed with 0.1% ascorbic acid and 1 mM EDTA (HiMedia, India) and heated at 70°C for 10 min. After centrifugation at 10,000× g for 10 min, the supernatant was filtered using 0.22 μm syringe filters. Quantification was performed using the HPLC system described above, with detection at 318 nm. The mobile phase consisted of Solvent A (0.1% trifluoroacetic acid in water, pH 2.6) and Solvent B (acetonitrile), following a gradient elution program adapted from Albawarshi et al. (2022) [31]. The flow rate was set at 0.9 mL/min, and the column oven was maintained at 30°C. Standard solutions of B2, B9, and B12 were used for calibration.
Antimicrobial properties of the fermented extracts were assessed using the agar well diffusion method. Whole cell biomass (WCB) was prepared by suspending the fermented samples individually in sterile distilled water at 1:1 w/v. The CFS was obtained by centrifuging samples at 10,000× g for 15 min, while the CL was prepared by sonicating the pellets in 0.1 M phosphate buffer using a probe sonicator (LABMAN Scientific). Agar plates were prepared with a 1:1 mixture of MRS agar and MH agar overlaid on 2% agar base and then pre-seeded individually with standardised (0.5 McFarland) cultures of E. coli, S. aureus, and S. typhi. Wells were prepared using a sterile cork borer. Fifty microliters of each test fraction (CFS, CL, and WCB) were dispensed into wells, and the plates were incubated at 37°C for 24 h [24].
LAB samples were further analysed via SEM to assess cellular morphology and colonisation when cultivated under stress conditions. Three sample treatments were prepared: (i) unfermented control, (ii) 48-hour fermented sample, and (iii) fermented sample exposed to 0.5% bile salts at pH 2.5 for 3 h. Cells were fixed following the protocol described by Ammar (2017) [32]. Initially, the cells were fixed in 2.5% glutaraldehyde, dehydrated in a graded ethanol series (30% to 100%), and sputter-coated with gold. SEM imaging was performed using an FEI Nova NanoSEM 450 system.
Statistical analysis was performed using GraphPad Prism (10) with two-way ANOVA followed by Tukey’s post hoc test. Significance was set at p < 0.05. All experiments were conducted in biological triplicate (n = 3). Graphs were generated using Microsoft Excel (Microsoft Corporation, USA), with error bars indicating standard deviation.
The growth performance of the LAB consortium was evaluated at the end of 48 h across eleven different legume substrates at three legume-to-water ratios: 1:1, 1:2, and 1:3 (w/v) (Figure 1). In all cases, an increase in water content from 1:1 to 1:2 led to a progressive enhancement in bacterial growth. Statistical analysis (two-way ANOVA followed by Tukey’s test) revealed significant differences between the 1:2 and 1:3 ratios in eight legumes (p < 0.05), while three legumes (WBG, HG, and BEB) showed no significant difference (ns) among the tested legumes, as shown in Figure 1. YP demonstrated the highest LAB proliferation, increasing from 10.12 log10 CFU/g at the 1:1 ratio to 16.04 log10 CFU/g at 1:3, representing a 58.5% increase. SB also showed substantial support for LAB growth, rising from 10.72 log10 CFU/g to 13.01 log10 CFU/g, a 21.36% increase. Other legumes, including HG, KC, and BKB, exhibited a moderate increase in bacterial growth, ranging from 14 to 33%. Thus, at a 1:3 legume-to-water ratio, both YP and SB demonstrated maximum microbial growth. Both these legumes possess distinct nutritional compositions. YP contains higher levels of carbohydrates and fibre, while SB offers significantly more protein but fewer fermentable carbohydrates (Table 1) [15]. Hence, both the legumes were selected for further experiments.
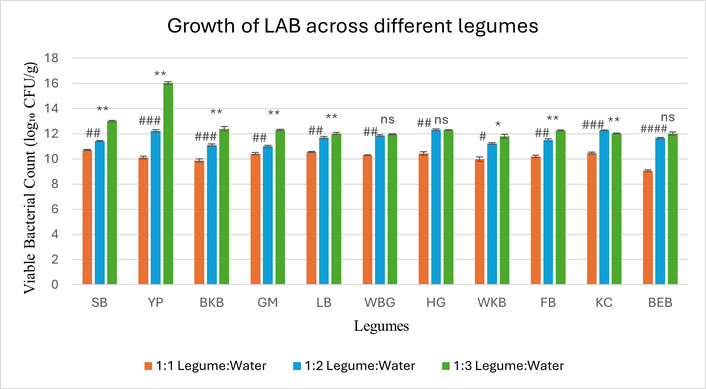
Viable counts of LAB (log10 CFU/g) in eleven legumes after 48 h of fermentation at substrate-to-water ratios of 1:1, 1:2, and 1:3 (w/v). Statistical differences between 1:1 and 1:2 are indicated by (#), and those between 1:2 and 1:3 are indicated by (*), as assessed by Tukey’s test. Significance levels: # or * p < 0.05; ## or ** p < 0.01; ### or *** p < 0.001; #### or **** p < 0.0001; ns: not significant. Legume codes: SB (soybean), YP (yellow pea), WKB (white kidney bean), GM (mung bean), BEB (cowpea), BKB (black kidney bean), LB (lima bean), BG (black gram), KC (black chickpea), FB (field bean), HG (horse gram). Error bars represent standard deviation (SD), n = 3. LAB: lactic acid bacteria.
| Composition | Soybeans | Yellow peas |
|---|---|---|
| Protein | 36.5% | 23.1% |
| Carbohydrate | 30.2% | 61.6% |
| Fat | 19.9% | 3.89% |
| Dietary fibre | 9.3% | 22.2% |
| Sucrose | 6.35% | 1.85% |
| Raffinose | 1.15% | 0.6% |
| Stachyose | 2.85% | 1.71% |
| Verbascose | - | 2.3% |
| Starch | 10.9–11.7% | 39.44–46.23% |
| Total sugars | 7.33% | 3.14% |
Following inoculation with 0.2% (v/v) of a 1010 CFU/mL bacterial suspension, the population dynamics of LAB in YP and SB matrices were monitored over 120 h (Figure 2). In YP, bacterial counts increased rapidly, reaching a maximum of 16.04 log10 CFU/g at 48 h, corresponding to an approximate 108-fold increase relative to the initial inoculum. Between 72 and 120 h, bacterial populations remained relatively stable, ranging from 15.08 to 15.51 log10 CFU/g. In SB, bacterial counts increased over time, peaking at 14.60 log10 CFU/g at 72 h, indicating an approximate 106-fold increase. This was followed by a plateau phase, with values ranging from 14.34 to 14.54 log10 CFU/g between 96 and 120 h. These results indicate that both legume matrices supported substantial bacterial growth; however, YPs facilitated more rapid proliferation and achieved higher maximum population densities compared to SBs under identical conditions.
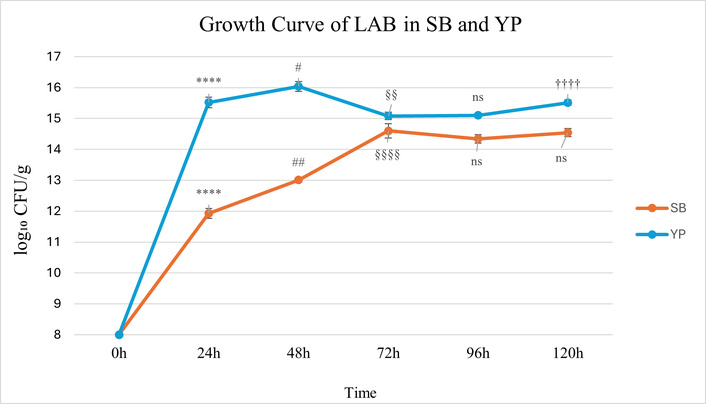
Growth of LAB in yellow pea (YP) and soybean (SB) during 120 h of fermentation. Statistical analysis was performed using two-way ANOVA followed by Tukey’s post hoc test. 0–24 h: p < 0.0001 (****); 24–48 h: p < 0.05 (#), p < 0.01 (##); 48–72 h: p < 0.01 (§§), p < 0.0001 (§§§§); 72–96 h: ns (not significant); 96–120 h: p < 0.0001 (††††); ns (not significant). Error bars represent standard deviation (SD) (n = 3). LAB: lactic acid bacteria.
Progressive acid accumulation was accompanied by a decline in pH for both matrices. The pH of the YP matrix decreased to 4.66 at 96 h (Figure 3), whereas SB showed a moderate reduction to 5.16 at 48 h (Figure 4). These pH profiles closely mirrored changes in organic acid concentrations, indicating active microbial metabolism and substrate-specific buffering capacities.
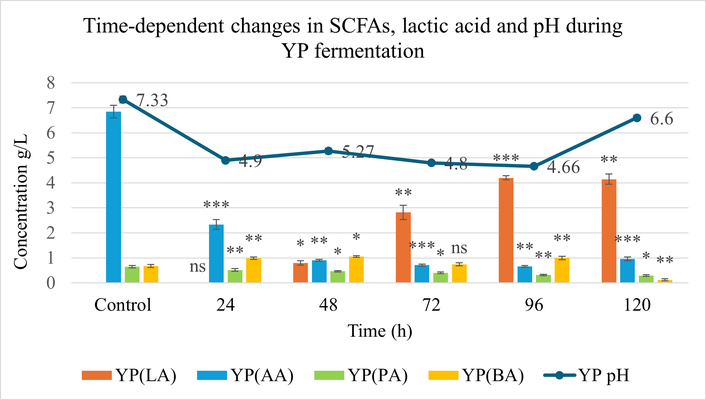
Production of lactic acid (LA), acetic acid (AA), propionic acid (PA), and butyric acid (BA) during solid-state fermentation (SSF) of yellow peas (YP) over 120 h, compared to the unfermented control. Statistical analysis was performed using two-way ANOVA followed by Tukey’s post hoc test. Significance: p < 0.05 (*), p < 0.01 (**), p < 0.001 (***); ns (not significant) vs. control. Error bars represent standard deviation (SD), (n = 3). SCFAs: short-chain fatty acids.
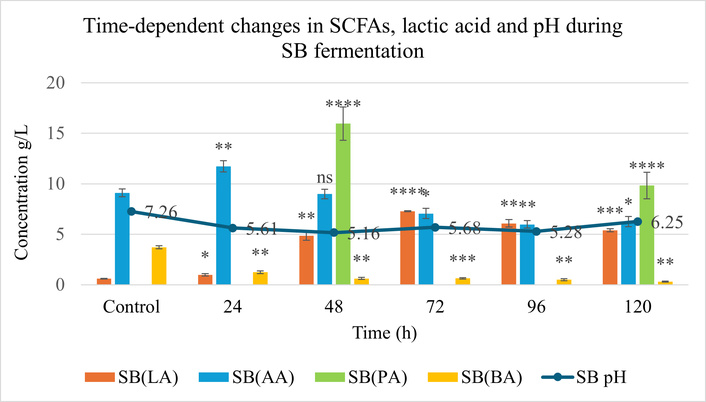
Production of lactic acid (LA), acetic acid (AA), propionic acid (PA), and butyric acid (BA) during solid-state fermentation (SSF) of soybean (SB) over 120 h, compared to the unfermented control. Statistical analysis was performed using two-way ANOVA followed by Tukey’s post hoc test. Significance: p < 0.05 (*), p < 0.01 (**), p < 0.001 (***), p < 0.0001 (****); ns (not significant) vs. control. Error bars represent standard deviation (SD), (n = 3). SCFAs: short-chain fatty acids.
LA production showed the most pronounced temporal variation. In YP, maximum LA production reached 4.2 g/L at 96 h, while in SB, 7.28 g/L at 72 h. SB exhibited a sharper rise in LA, corresponding to a 12.1-fold increase as compared to its control.
In YP, BA levels increased steadily in the mid-phase of fermentation (24–72 h), peaking at 1.05 g/L at 48 h, representing a 1.5-fold rise compared to the control. In contrast, in SB, maximum accumulation of AA was observed at 24 h, with a concentration of 11.73 g/L, and PA, which was absent in the control, peaked later at 48 h, reaching 15.95 g/L.
B2 production showed the most substantial increase among the B-group vitamins. At the end of 96 h in YP, vitamin B2 reached a maximum concentration at 7.097 µg/mL, corresponding to a 4.9-fold increase relative to the control (Figure 5), while in SB, attaining 0.97 µg/mL, striking a 35.9-fold enhancement, at 96 h from an initial level of 0.027 µg/mL (Figure 6).
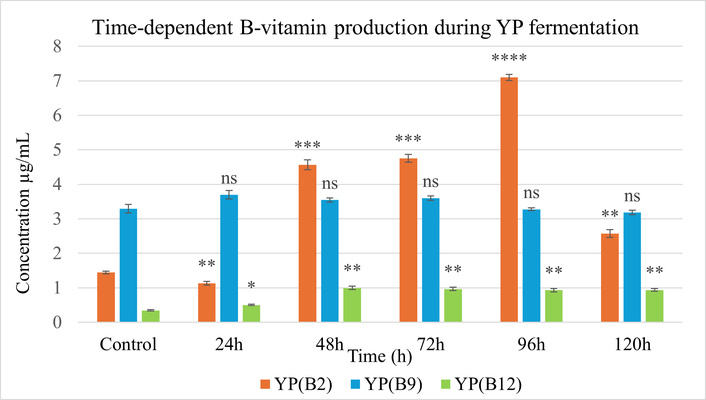
Vitamin B2, B9, and B12 levels during solid-state fermentation (SSF) of yellow pea (YP) for 120 h compared to the unfermented control. Statistical analysis was performed using two-way ANOVA followed by Tukey’s test; p < 0.05 (*), p < 0.01 (**), p < 0.001 (***), p < 0.0001 (****); ns (not significant) vs. control. Error bars indicate standard deviation (SD), (n = 3).
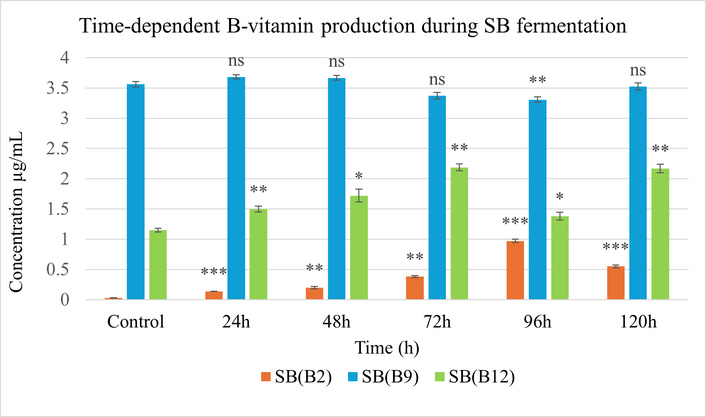
Vitamin B2, B9, and B12 levels during solid-state fermentation (SSF) of soybean (SB) over 120 h compared to the unfermented control. Statistical analysis was performed using two-way ANOVA followed by Tukey’s test; p < 0.05 (*), p < 0.01 (**), p < 0.001 (***), ns (not significant) vs. control. Error bars indicate standard deviation (SD), (n = 3).
At the end of 24 h in both substrates, B9 concentrations exhibited moderate variation. In YP, vitamin B9 concentration was 3.69 µg/mL; similarly, SB showed a comparable peak of 3.683 µg/mL, with minimal change thereafter, indicating limited B9 biotransformation during fermentation.
B12 concentration was enhanced in both legume matrices, though with differing kinetics. In YP, the maximum level was 0.99 µg/mL at 48 h. SB demonstrated a higher final concentration of 2.187 µg/mL at 72 h.
These observations suggest that YP favours B2 and early B12 production, whereas SB demonstrates higher overall yields of vitamins B2 and B12.
The antimicrobial potential of WCB, CL, and CFS obtained from fermented YP and SB matrices was evaluated against S. aureus, E. coli, and S. typhi (Figure 7). Antimicrobial activity was not observed in non-fermented controls when processed identically to fermented samples across all tested fractions at various fermentation times.
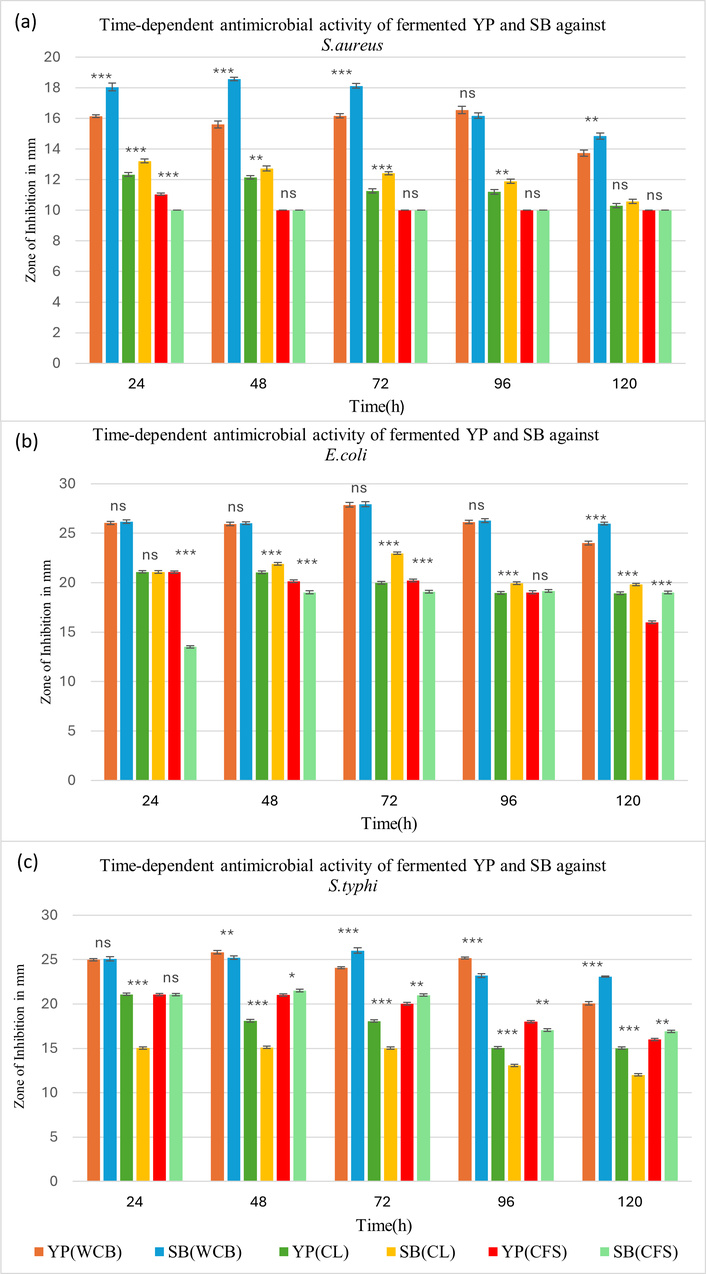
Antibacterial activity of fermented samples against (a) S. aureus, (b) E. coli, and (c) S. typhi using yellow pea (YP) and soybean (SB) substrates across 120 h of fermentation. WCB (whole cell biomass), CL (cell lysate), and CFS (cell-free supernatant). Statistical analysis was performed using two-way ANOVA followed by Tukey’s post hoc test. Significance: p < 0.05 (*), p < 0.01 (**), p < 0.001 (***); ns (not significant) between YP and SB. Error bars represent standard deviation (SD), (n = 3).
The WCB fraction exhibited the strongest antimicrobial activity among all tested fractions, with inhibition zones ranging from 13 to 28 mm. Samples derived from SB showed higher activity as compared to YP against the selected clinical isolates. Among the tested pathogens, E. coli was the most sensitive organism, followed by S. typhi. These findings indicate that the antimicrobial potential of fermented legumes is predominantly associated with intact or surface-bound bacterial components.
The CL fraction of SB also demonstrated a higher antimicrobial effect than that of YP, showing an inhibition zone of 22.98 mm against E. coli. The CFS fraction from SB also exhibited notable inhibition, with a maximum zone of 21.51 mm against S. typhi. Both CL and CFS fractions of YP showed comparable activity against S. typhi and E. coli.
Collectively, these results confirm that fermented YP and SB matrices exhibit broad-spectrum antimicrobial potential, primarily mediated through cell-associated mechanisms.
SEM analysis was conducted on YP samples, as they exhibited the highest microbial growth during fermentation. After 48 h of fermentation, the surface of the YP exhibited noticeable roughening and localized structural changes, likely due to microbial enzymatic activity. Dense populations of rod-shaped LAB were observed adhering to the legume matrix, often embedded within spherical extracellular deposits, presumed to be exopolysaccharides (EPS) (Figure 8).

SEM images of yellow pea (YP) samples: (a) unfermented control with rough surface; (b) 48 h fermented YP with dense lactic acid bacteria (LAB) in extracellular polymeric substances (EPS); (c) fermented YP after stress conditions (0.5% bile salts, pH 2.5, 3 h) showing adherent, intact LAB cells. SEM: scanning electron microscopy.
In the non-fermented control, no microbial structures were detected, confirming substrate sterility and validating that subsequent observations were due to the inoculated LAB. The surface of the unfermented legume appeared smooth and structurally intact, with no evidence of microbial adherence or surface disruption.
In this study, legumes were selected as fermentation substrates as they are low-cost, protein-rich matrices containing intrinsic prebiotic carbohydrates (resistant starches, oligosaccharides) that facilitate microbial colonization and metabolic flux toward SCFA and vitamin biosynthesis. Organic acid production is often monitored over extended fermentation periods, typically up to five days. The temporal dynamics of vitamin biosynthesis have been less frequently investigated, with most studies reporting vitamin levels only at selected time points, such as 24, 48, or 72 h [36–38]. Therefore, a comprehensive time-dependent evaluation is essential to better understand metabolite progression and to determine optimal fermentation intervals for simultaneous enhancement of multiple functional compounds. SSF of legume flours with Lactobacillus consortium represents a scalable bioprocessing platform that integrates microbial growth support up to 7 log10 CFU/g with substrate valorisation, improving protein accessibility, reducing antinutrient load, and enhancing metabolite recovery yields [39, 40]. Multiple additional legumes, including HG, FB, KC, WKB, GM, BEB, BKB, LB, and BG, were also screened, demonstrating that the LAB consortium maintains functional robustness across diverse legume matrices. Maximum LAB viability was observed at a legume-to-water ratio of 1:3 (~67% moisture), consistent with previous findings that 60–65% moisture is ideal for nutrient diffusion and microbial metabolic activity [41]. Viable counts ranged from 11.8 to 12.4 log10 CFU/g across most of the screened legumes at 1:3 moisture, demonstrating strong microbial adaptability and substrate compatibility. Although YP exhibited the highest viability (16.04 log10 CFU/g), several other legumes, such as BKB, GM, FB, and HG, also supported substantial LAB proliferation. Among the legumes studied, YP and SB were selected for detailed analysis because of their contrasting macronutrient compositions, as mentioned in Table 1.
Fermentation kinetics revealed that YP supported more rapid LAB proliferation, 15.52 log10 CFU/g and pH drop to 4.9, reaching within the first 24 h, whereas SB exhibited delayed growth peaking at 14.6 log CFU/g by 72 h with a more gradual pH decrease. Previous studies on LAB reported 8–9 log10 CFU/g growth of L. casei and L. plantarum on lentils [42], while Śliżewska et al. (2020) [27] observed proliferation of Lactobacillus strain up to 10.28 log10 CFU/g using wheat, barley, rye, and maize flours. These findings indicate that the higher fermentable carbohydrate content of YPs promotes rapid and efficient fermentation. Conversely, SBs support slower and more gradual microbial growth and acidification, likely due to their limited availability of fermentable sugars and high concentrations of ANFs such as trypsin inhibitors and phytic acid, which may hinder microbial metabolism by chelating essential nutrients or inhibiting enzymatic pathways critical for LAB activity [43].
LA production also exhibited clear substrate-dependent variability. In the present study, LA accumulation reached 4.2 g/L in YP at the end of 96 h and 7.28 g/L in SB at 72 h, with SB producing higher LA despite lower LAB counts, suggesting enhanced per-cell metabolic activity. This may result from acid stress-induced amino acid catabolism, which buffers the intracellular pH while sustaining LA synthesis [44]. In YP, a rapid decline in pH was observed within the first 24 h, which may be due to its distinct carbohydrate composition (Table 1), with a high content of complex carbohydrates. Oligosaccharides such as verbascose, raffinose, and stachyose are utilized in mixed-acid pathways that generate organic acids like formic and succinic, along with carbon dioxide, lowering the pH during early fermentation [45]. In contrast, SB contains a higher proportion of readily fermentable soluble sugars, particularly hexoses (Table 1), supports rapid homolactic fermentation, resulting in earlier and higher accumulation of LA. Thus, the substrate-specific sugar profile strongly governs acid production patterns and explains the contrasting pH–LA dynamics observed between YP and SB during the early fermentation. At later fermentation stages (≥ 120 h), a modest increase in pH was observed for both substrates. This apparent decoupling of pH and LA levels can be attributed to the shift in metabolism of microorganisms to consume organic acids after readily available sugars are depleted, leading to the production of alkaline metabolites, such as ammonia, that partially neutralize the accumulated organic acids in the medium [46]. Previous studies have also demonstrated the influence of substrate composition on LA yield. For example, SB yielded 136.2 mg/kg of LA using L. sakei in 24 h [21], while oil palm empty fruit bunches (EFBs) produced 6.49 g/L at 120 h via L. acidophilus [47]. In the present study, LA accumulation in YP and SB reached 4.14 and 5.38 g/L at the end of 120 h respectively. Moreover, LAB strains, particularly L. plantarum, have been shown to increase LA output by up to 94.9%, correlating with enhanced protease and cellulase activity that promotes the synergistic degradation of complex biomass, such as SB meal [48].
SCFA production was similarly influenced by pH dynamics. In SB, AA and PA reached maximum concentrations (AA: 11.73 g/L at 24 h; PA: 15.95 g/L at 48 h) at pH 5.16–5.61. In contrast, YP’s rapid acidification curtailed the window for secondary acid biosynthesis. When the pH dropped below 4.5, AA and PA levels declined due to the inhibition of acetyl-CoA and propionyl-CoA pathways [49]. BA levels in SB were 1.17-fold higher than in YP but remained ~60% lower than in unfermented SB. This reduction likely reflects a LAB-driven metabolic shift prioritizing LA synthesis, along with the possible enzymatic conversion of BA into volatile esters, such as ethyl butyrate. Additionally, BA’s cytotoxicity may restrict its accumulation during extended fermentation [50, 51]. Fermentations using L. plantarum on jicama extract have demonstrated that LAB can produce all three major SCFAs, including 1.58 mg/mL PA, 1.31 mg/mL AA, and 1.05 mg/mL BA [52]. L. acidophilus cultivated in MRS supplemented with 2% salt produced 4.23 g/L AA, 0.0246 g/L PA, and 0.024 g/L BA after 24 h [49], while skim milk-based fermentation after 48 h reported AA production of 2.243 g/L with 1% glucose, with BA concentrations of 0.111 g/L, and PA production up to 0.019 g/L in skim milk alone [53]. Compared to MRS broth and dairy-based fermentation systems, the elevated SCFA production observed in legume-based SSF in the present study highlights the strong influence of substrate composition, particularly complex carbohydrates and oligosaccharides, on microbial metabolic flux and secondary acid biosynthesis. This highlights the ability of LAB to biosynthesize a full spectrum of SCFAs, underscoring the potential of diverse plant substrates to support SCFA biosynthesis even without butyrate-producing co-cultures. Given the health-promoting effects of SCFAs on energy metabolism, immune modulation, and gut integrity, fermented legumes represent a sustainable dietary source compared to chemical sources and monoculture fermentation approaches [54, 55].
Legume fermentation also enriches the B-vitamin content. B2 accumulation was higher in YP (7.097 µg/mL) compared to SB (0.97 µg/mL) at the end of 96 h. Previous reports of L. plantarum isolated from rice-based foods demonstrated production of up to 0.7 µg/mL [10] and 0.481 µg/mL in soymilk fermentations [56]. B2 enrichment following fermentation has been widely reported in traditional fermented foods. Tempeh fermented by R. oryzae and natto fermented by Bacillus natto contain approximately 3.58 µg/g and 1.9 µg/g B2 [15], respectively, while idli batter fermentation has been reported to yield up to 5.9 µg/g B2 [57]. B9 production in both YP and SB reached similar levels (~3.6 μg/mL) at 24 h, indicating a strong and sustained biosynthetic capacity under these conditions. Tempeh and natto contain relatively low B9 concentrations (0.24 µg/g and 0.08 µg/g, respectively) [15]. L. fermentum was earlier reported for B9 concentration in chemically defined B9-free medium, where production peaked at 0.092 μg/mL after 24 h [10], and L. rhamnosus K4E yielded 0.24 μg/mL after 12 h [38], highlighting the high biosynthetic potential of select strains under legume-based SSF. These vitamins serve as nutraceuticals and key metabolic cofactors. B2-derived coenzymes (FMN and FAD), for instance, are crucial in redox reactions and energy metabolism, enhancing the biosynthesis of LA and SCFAs during fermentation [10]. SB supported higher yields (2.187 µg/mL) of B12 than YP (0.96 µg/mL), as observed at 72 h. This difference likely reflects strain-specific interactions with matrix components, as certain LAB (e.g., L. plantarum, L. casei, L. acidophilus) exhibit enhanced vitamin synthesis in response to precursor availability and matrix complexity. In comparison, plant-based fermented foods typically contain negligible amounts of vitamin B12, with tempeh reported to contain approximately 0.0008 µg/g and natto showing undetectable levels [15]. Acetobacter malorum in sourdough systems has been reported to produce up to 0.018 µg/mL in 48 h [58], while Das et al. (2021) [38] reported B12 production up to 0.084 µg/mL in soymilk fermented after 12 h with L. fermentum. This indicates a better yield of vitamins in legume-based SSF systems. The decrease in certain vitamin levels after an initial increase during Lactobacilli fermentation is generally caused by the consumption of these vitamins by the bacteria for their own growth and metabolism, as well as the degradation of specific vitamins due to the highly acidic environment [59].
The antimicrobial activity observed in LAB-fermented SB and YP extracts demonstrates the capacity of LAB to produce bioactive metabolites capable of inhibiting both Gram-positive and Gram-negative bacteria. Among the tested fractions, WCB exhibited the strongest inhibition (up to 27.93 mm) for E. coli, followed by CL (up to 22.98 mm) and CFS (up to 21.51 mm). This hierarchy suggests that intact cells retain greater antimicrobial potency through the combined effects of membrane-bound bacteriocins, intracellular enzymes, and sustained acid production [24, 60]. Comparatively less antagonistic effect on S. aureus may be attributed to its acid tolerance and thick peptidoglycan wall, and the damaging effect of LA on lipopolysaccharide (LPS) barrier [61, 62].
This is evidenced by earlier studies reporting effective suppression of pathogenic and spoilage organisms following LAB fermentation in legume seed sprouts, including reductions in mold counts by 1 Log10 CFU/mL, E. coli and Klebsiella spp. by 2 Log10 CFU/mL, and complete elimination of Salmonella spp., Shigella spp., and Staphylococcus epidermidis. Additionally, S. aureus and S. saprophyticus populations declined by approximately 3 Log10 CFU/mL, frequently reaching undetectable levels [63]. The superiority of plant-based substrates is further supported by SSF studies, where LAB-fermented Spirulina exhibited inhibition zones ranging from 9–16 mm against S. aureus, significantly exceeding those observed under submerged fermentation conditions [12]. Similarly, under optimized SSF conditions, L. plantarum fermenting bean by-products achieved maximum antimicrobial activity, resulting in reductions of approximately 2 and 4 Log10 CFU/g against Listeria monocytogenes and E. coli, respectively [64]. When compared with dairy-based systems, the antimicrobial activity observed in the present study was notably stronger against E. coli. LAB isolates from fermented milk have previously shown inhibition zones of up to 15 mm against E. coli and approximately 22 mm against S. dysenteriae [65]. These observations reinforce the potential of LAB, particularly under SSF conditions, to generate bioactive compounds with selective antimicrobial effects, which may be enhanced by specific substrates and strain–matrix interactions.
SEM analysis revealed significant matrix degradation and LAB colonization in YP. After 48 h, fermented samples exhibited smoother, eroded surfaces indicative of enzymatic breakdown of proteins, fibres, and lipids, consistent with previous studies [66]. Numerous bacilli-shaped LAB cells were visible adhering to the matrix, often in proximity to spherical EPS-like structures, suggestive of biofilm formation. These findings align with previous SEM reports of LAB colonization on olive surfaces during fermentation [67].
Observed clustering and EPS secretion support classic phases of biofilm development: initial adhesion, maturation, and dispersal, as supported by morphological clustering and EPS secretion [68]. The SEM images showing LAB entrapped in fibrous grooves under acid and bile stress further suggest stress-induced adaptation.
Collectively, these findings demonstrate that legume-based SSF using a LAB consortium is an effective strategy for enhancing the nutritional and functional properties of plant-derived substrates. Screening of multiple legumes confirmed the broad adaptability of the consortium, with YP and SB selected for detailed evaluation due to their contrasting compositional profiles. Fermentation influenced probiotic viability and metabolite production patterns, resulting in substrate-specific variations in LA, SCFAs, and B-complex vitamins. YP supported higher BA production, whereas SB exhibited greater LA, PA, and AA production compared to their respective controls. Additionally, fermented extracts demonstrated strong antimicrobial activity, and SEM analysis confirmed matrix degradation and microbial colonization. A consolidated summary of functional and biochemical changes in selected substrates before and after fermentation is presented in Table 2.
Summary of LAB metabolites.
| Metabolite | Yellow pea (unfermented) | Yellow pea (post fermentation) (maximum value; time) | Soybean (unfermented) | Soybean (post fermentation) (maximum value; time) |
|---|---|---|---|---|
| Lactic acid (LA) | - | 4.2 g/L; 96 h | 0.6 g/L | 7.28 g/L; 72 h |
| Acetic acid (AA) | 6.85 g/L | 2.33 g/L; 24 h | 9.1 g/L | 11.73 g/L; 24 h |
| Propionic acid (PA) | 0.65 g/L | 0.51 g/L; 24 h | - | 15.95 g/L; 48 h |
| Butyric acid (BA) | 0.68 g/L | 1.05 g/L; 48 h | 3.71 g/L | 1.23 g/L; 24 h |
| Vitamin B2 | 1.443 µg/mL | 7.097 µg/mL; 96 h | 0.02 µg/mL | 0.97 µg/mL; 96 h |
| Vitamin B12 | 0.34 µg/mL | 0.99 µg/mL; 48 h | 1.14 µg/mL | 2.187 µg/mL; 72 h |
| Vitamin B9 | 3.29 µg/mL | 3.69 µg/mL; 24 h | 3.56 µg/mL | 3.683 µg/mL; 24 h |
| LAB count | - | 16.04 Log10 CFU/g; 48 h | - | 14.6 Log10 CFU/g; 72 h |
| Antimicrobial compounds (E. coli) | - | 27.87 mm; 72 h | - | 27.93 mm; 72 h |
| Antimicrobial compounds (S. typhi) | - | 25.84 mm; 48 h | - | 26.02 mm; 72 h |
| Antimicrobial compounds (S. aureus) | - | 16.54 mm; 96 h | - | 18.57 mm; 48 h |
LAB: lactic acid bacteria.
Despite these promising outcomes, certain limitations remain, including the lack of strain-specific metabolite attribution and the observation that different substrates preferentially enhanced specific metabolites rather than simultaneously maximizing all functional compounds. This indicates that substrate selection plays a critical role in directing metabolic outputs and may require tailored optimization depending on the targeted metabolite. Future studies should therefore focus on improving metabolite yield through selection or blending of complementary legume substrates, incorporation of targeted prebiotic supplementation, and optimization of fermentation parameters. Furthermore, advanced metabolomic and molecular investigations, along with in vivo validation, will be essential to confirm the health benefits and functional efficacy of fermented legume products. Overall, these findings highlight the potential of LAB-mediated SSF as a sustainable approach for developing nutrient-enriched plant-based functional foods.
AA: acetic acid
ANFs: anti-nutritional factors
B12: cobalamin
B2: riboflavin
B9: folate
BA: butyric acid
BEB: cowpea
BG: black gram
BKB: black kidney bean
CFSs: cell-free supernatants
CL: cell lysate
EDTA: ethylenediaminetetraacetic acid
EPS: exopolysaccharides
FB: field bean
GM: mung bean or green gram
HG: horse gram
KC: black chickpea
LA: lactic acid
LAB: lactic acid bacteria
LB: lima bean
MH: Mueller–Hinton
MRS: de Man, Rogosa, and Sharpe
PA: propionic acid
SBs: soybeans
SCFAs: short-chain fatty acids
SEM: scanning electron microscopy
SSF: solid-state fermentation
WCB: whole cell biomass
WKB: white kidney bean
YP: yellow pea
Declaration of generative AI and AI-assisted technologies in the writing process: During manuscript preparation, the authors used Paperpal (Editage) to enhance language clarity and grammar. Following this assistance, all content was carefully reviewed and revised by the authors, who take full responsibility for the final published version.
The authors express their sincere gratitude to Bharati Vidyapeeth (Deemed to be) University for institutional support throughout this study. The authors also acknowledge the Department of Biotechnology, Government of India, for infrastructural support provided under the DBT-Builder Scheme (Grant No. BT/INF/22/SP41297/2021).
AR: Conceptualization, Methodology, Formal analysis, Investigation, Visualization, Writing—original draft, Writing—review & editing. RB: Conceptualization, Resources, Validation, Supervision, Writing—review & editing. Both authors read and approved the submitted version.
The authors declare no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
This work did not receive any specific grant for the conduct of the study.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 1040
Download: 69
Times Cited: 0
