Affiliation:
1School of Agriculture and Biology, Engineering Research Center of Therapeutic Antibody (Ministry of Education), and School of Life Sciences and Biotechnology, Shanghai Jiao Tong University, Shanghai 200240, People’s Republic of China
ORCID: https://orcid.org/0000-0002-9805-2161
Affiliation:
1School of Agriculture and Biology, Engineering Research Center of Therapeutic Antibody (Ministry of Education), and School of Life Sciences and Biotechnology, Shanghai Jiao Tong University, Shanghai 200240, People’s Republic of China
ORCID: https://orcid.org/0000-0002-5207-2458
Affiliation:
2Tibetan Institute for Food and Drug Control, NMPA key Laboratory of Quality Control of Traditional Chinese Medicine, Tibetan Medicine, Lhasa 850000, Tibet Autonomous Region, People’s Republic of China
ORCID: https://orcid.org/0009-0001-4279-289X
Affiliation:
3Anran Zhimei (Shanghai) Biotechnology Co., Ltd., Shanghai 201403, People’s Republic of China
ORCID: https://orcid.org/0000-0002-8608-9370
Affiliation:
2Tibetan Institute for Food and Drug Control, NMPA key Laboratory of Quality Control of Traditional Chinese Medicine, Tibetan Medicine, Lhasa 850000, Tibet Autonomous Region, People’s Republic of China
Email: dazhuo1108@126.com
ORCID: https://orcid.org/0009-0003-3087-9650
Affiliation:
1School of Agriculture and Biology, Engineering Research Center of Therapeutic Antibody (Ministry of Education), and School of Life Sciences and Biotechnology, Shanghai Jiao Tong University, Shanghai 200240, People’s Republic of China
Email: xuanweizhou@sjtu.edu.cn
ORCID: https://orcid.org/0000-0002-3993-6346
Explor Foods Foodomics. 2026;4:1010124 DOI: https://doi.org/10.37349/eff.2026.1010124
Received: September 06, 2025 Accepted: February 13, 2026 Published: March 18, 2026
Academic Editor: Marcello Iriti, Milan State University, Italy
Aim: This study aimed to analyze the non-volatile chemical components in wild Ophiocordyceps sinensis (O. sinensis) from four distinct production areas in Xizang, and its fermented mycelia, with the goal of chemically evaluating the feasibility of substituting the wild samples with their mycelial preparation, and providing references for their application in daily life.
Methods: An untargeted metabolomics approach using ultra-performance liquid chromatography quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS) was employed for comprehensive analysis.
Results: A total of 89 non-volatile components were identified, mainly covering short peptides, nucleotides/derivatives, glycerophospholipids, glycosides, and amino acids. Multivariate statistical analysis revealed significant regional variation in the content of key metabolites, particularly the short peptide profile. Di- and tri-peptides were confirmed as the dominant bioactive constituents and chemometric markers distinguishing geographical origins. Compared to the wild samples, the fermented mycelia exhibited a statistically significant reduction in both the diversity and relative abundance of these signature short peptides.
Conclusions: Wild O. sinensis and its fermented mycelia both contain bioactive compounds, with the latter sometimes surpassing the wild samples in specific components. However, the fermented version doesn’t match the natural samples’ synergistic effects. The wild sample’s efficacy is heavily influenced by its environment and growth conditions. This study provides a basis for using wild O. sinensis or its mycelial products in daily diets or disease prevention.
Ophiocordyceps sinensis (O. sinensis), a unique Qinghai-Tibet Plateau resource, is distributed in China, Bhutan, India, and Nepal, with China contributing 96.4% to the global output, mainly from Tibetan, Qinghai, Sichuan, Gansu, and Yunnan [1]. It is a valuable Tibetan and Chinese medicinal material with over 20 active components, including proteins, nucleosides, sterols, and polysaccharides, used for quality control and identification. Wild O. sinensis has various pharmacological effects, such as immune regulation, antioxidant, and anti-tumor properties [2, 3]. Chemical composition varies by production area and between wild and mycelial forms, complicating market supervision [4]. Previous studies focused on specific components or categories, limiting the reflection of metabolite diversity and market application [5, 6]. Modern biotechnology offers molecular identification methods, but their application in market supervision is limited. A comprehensive and systematic analysis of wild O. sinensis from different areas, cultivated forms, and cultured mycelia is necessary.
Metabolomics, utilizing mass spectrometry, enables the qualitative and quantitative analysis of endogenous metabolites in biological samples, offering a comprehensive chemical profile through multivariate data analysis and high-throughput detection. Ultra-performance liquid chromatography quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS) technology ionizes molecules, charges them, and measures their mass-to-charge ratios to identify and analyze compounds, known for its separation power, sensitivity, and specificity. This study applies UPLC-Q-TOF-MS-based metabolomics to analyze non-volatile components of wild O. sinensis and its fermented mycelia from four Xizang regions, comparing their chemical profiles to assess the potential of fermented mycelia as a supplement to wild O. sinensis. The analysis of metabolite differences aids in identifying chemical markers, laying a foundation for chemical fingerprinting and informed market regulation.
Wild O. sinensis samples, especially from its immature stages known as caterpillar fungus, were collected from four primary distribution areas within the Xizang Autonomous Region of China (Figure 1). Among these locations, the average altitude is approximately 4,150 meters above sea level in Baqing County, Nagqu City (Nagqu, NQ), 3,240 meters in Riwoqe County, Qamdo City (Qamdo, QD), 3,420 meters in Gongbujiangda County, Nyingchi City (Nyingchi, NC), and 3,260 meters in Gyaca County, Shannan City (Shannan, SN).
O. sinensis strain was isolated from wild specimens in Xizang and authenticated by the Plant Biotechnology Research Center, Shanghai Jiao Tong University. The preparation of O. sinensis mycelia, known as Jun Si Ti (JST) in Chinese pinyin, was conducted using conventional methods described in the previous publications (Figure 2) [7].
All analyses were conducted using a UPLC-IM-Q-TOF-MS system from Waters Corporation (MA, USA). The solvents used, formic acid and methanol, were of the highest chromatographic grade, sourced from Fisher Scientific (MA, USA). Ultrapure water, essential for maintaining the integrity of the analysis, was procured through the Millipore water purification system (MA, USA). All additional reagents employed in the study were of analytical grade, ensuring the accuracy and reliability of the experimental results.
Four wild samples of O. sinensis (NQ, QD, NC, SN) were dried at 60°C, then at least 3 samples from different sampling sites of wild O. sinensis were pooled together for the extraction of metabolites. After being thoroughly ground using a grinder, 0.100 g of each sample of the NQ, QD, NC, SN, and JST powders was accurately weighed. Each powder should be dissolved in 1 mL of methanol solution (75%, v/v). Carefully vortex each sample for a precise 1-minute interval, and then proceed to sonicate the samples in an ultrasonic cell disruptor for a duration of 60 to 120 min to ensure thorough cell disruption. After removal, place the samples at 4°C for overnight extraction. The extracts are then centrifuged at 13,000 × g for 10 min. Subsequently, the supernatant is filtered through a 0.22 μm microporous membrane to obtain the test solution, which is stored at 4°C for subsequent use.
Agilent SB-C18 column (50 mm × 4.6 mm, 1.8 μm), mobile phase A is 0.1% aqueous formic acid solution, mobile phase B is acetonitrile, with the following gradient profile: 0–12 min, 10%–95% B; 12–16 min, 95% B; 16–16.5 min, 95%–10% B; 16.5–20 min, 10% B. The column temperature is set at 35°C, and the injection volume is 5 μL.
LC-Q-TOF-MS equipped with a photodiode array detector (PAD) and an electrospray ionization (ESI) source, operating in both positive and negative ion modes. The parameters are as follows: negative ionization mode voltage –3.0 kV, nitrogen as the carrier gas, gas temperature 350°C, flow rate 8 L·min–1, positive ionization mode voltage 3.8 kV; nebulizer pressure 45 psi; fragmentor voltage 100 V; skimmer voltage 65 V. The eluent is detected by PAD scanning from 200 to 600 nm. Data acquisition is conducted within an m/z range of 50 to 1,000 Da.
Import the precise mass data results of NQ, QD, NC, SN, and JST corresponding ions into the Peak View software (version 2.2), and analyze them using Peak View Software’s Formula Finder and other functions, online databases (Human Metabolome Database, Pub Chem, Chemical Book, etc.), and secondary fragment cleavage patterns. In addition to the primary metabolites, minor metabolites are also present. However, the peak intensity in the ion chromatogram is restricted to 1 × 105, and peaks exhibiting intensities below this threshold are excluded from the analysis. Ions that meet the criteria of having a mass error less than 5 ppm, correct isotope distribution, and containing secondary fragments are selected and confirmed as target ions. Formula Finder, Mass Calculators, and others are used as reference indicators for qualitative analysis. Compounds with reference ppm values less than 5 ppm in the software, correct isotope distribution, and correct secondary fragment analysis can be identified as target compounds. Establish a known component identification table according to the major categories of compounds. The heatmap analysis of active component metabolites is implemented using the R language.
LC-Q-TOF-MS analysis was conducted on non-volatile components in wild O. sinensis from four production areas in Tibet (NQ, QD, NC, SN) and JST, resulting in total ion current (TIC) chromatograms for positive (ESI+) and negative ion (ESI–) modes for NQ, QD, NC, SN, and JST (Figure S1).
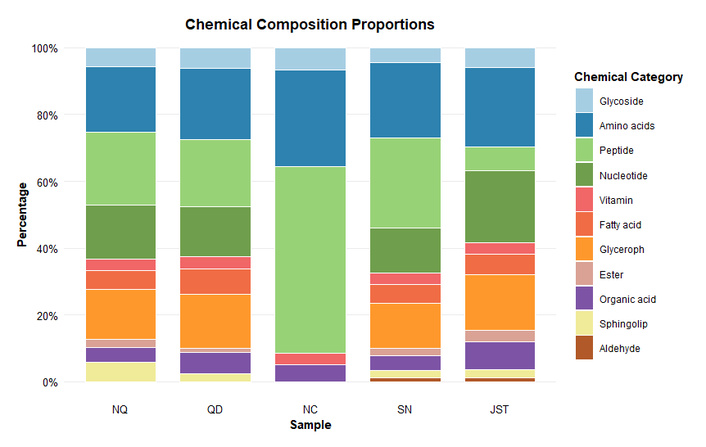
A total of 87, 80, 59, 89, and 84 non-volatile chemical components were identified in NQ, QD, NC, SN, and JST. Respectively, these components can be broadly classified into glycosides, amino acids and their derivatives, peptides (including dipeptides and tripeptides), nucleotides and their derivatives, vitamins, fatty acids, glycerophospholipid metabolites, esters, organic acids, sphingolipids, and aldehydes (Table 1).
Category of non-volatile components in wild O. sinensis and its fermented mycelia.
| Category | NQ | QD | NC | SN | Mean of O. sinensis | JST | Ratio (O. sinensis/JST) |
|---|---|---|---|---|---|---|---|
| Glycoside | 5 | 5 | 4 | 4 | 4.5 | 5 | 0.90 |
| Amino acids and derivatives | 17 | 17 | 17 | 20 | 17.75 | 20 | 0.89 |
| Peptide | 19 | 16 | 33 | 24 | 23 | 6 | 3.83 |
| Nucleotides | 14 | 12 | 0 | 12 | 9.5 | 18 | 0.53 |
| Vitamin | 3 | 3 | 2 | 3 | 2.75 | 3 | 0.92 |
| Fatty acid | 5 | 6 | 0 | 5 | 4 | 5 | 0.80 |
| Glycerophospholipid metabolites | 13 | 13 | 0 | 12 | 9.5 | 14 | 0.68 |
| Ester | 2 | 1 | 0 | 2 | 1.25 | 3 | 0.42 |
| Organic acid | 4 | 5 | 3 | 4 | 4 | 7 | 0.57 |
| Sphingolipid | 5 | 2 | 0 | 2 | 2.25 | 2 | 1.13 |
| Aldehyde | 0 | 0 | 0 | 1 | 0.25 | 1 | 0.25 |
| Total | 87 | 80 | 59 | 89 | 78.75 | 84 | 0.94 |
O. sinensis: Ophiocordyceps sinensis.
Further intuitive analysis has revealed that in JST, the primary components include amino acids and their derivatives, nucleosides, glycerophospholipid metabolites, and organic acids. In wild Cordyceps, the main components are amino acids and their derivatives, short peptides, nucleosides, and glycerophospholipid metabolites.
Glycosides are natural compounds formed by the connection of a sugar moiety (such as glucose, galactose) and a non-sugar moiety (referred to as the aglycone or the glycone) through a glycosidic bond (acetal bond). In this experiment, four common saccharides were detected in NQ, QD, NC, SN, and JST under both positive and negative ion modes (shown in Table 1), which are sucrose, D-mannitol, D-fucose, and Blumeoside A (Figure 3b). D-mannitol, also known as cordycepin, is an important active substance or marker in O. sinensis, with a wide range of biological functions [8]. Blumeoside A was also identified in JST, which is a new type of iridoid glycoside. Iridoid compounds are widely distributed and have neuroprotective effects, anti-tumor effects, and hepatoprotective effects [9]. Therefore, this newly discovered component in JST is worth further in-depth study.
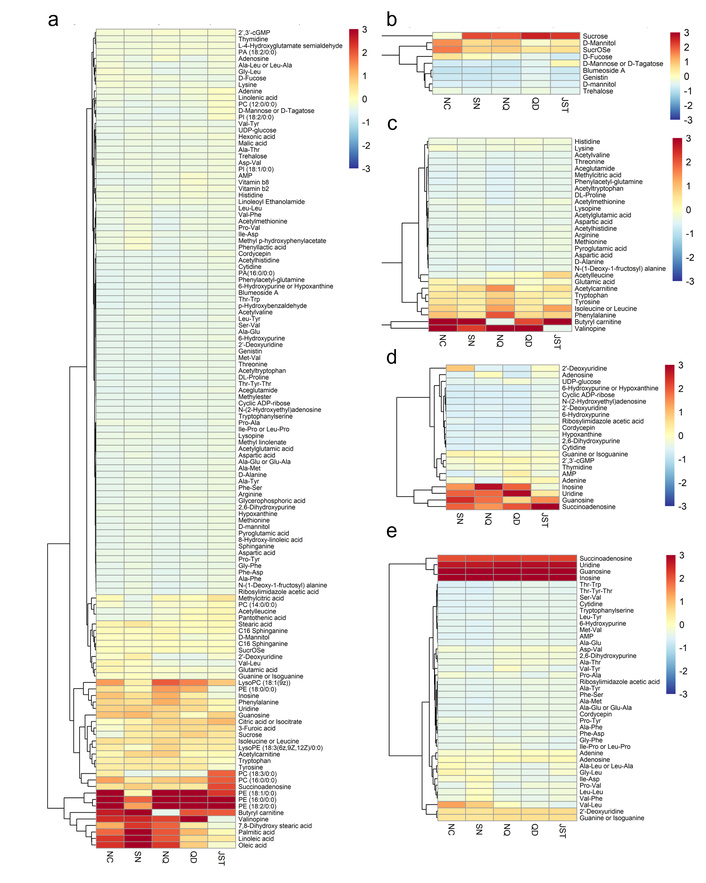
Presents the cluster analysis of metabolites in wild O. sinensis and its mycelia. (a) cluster analysis of total components in wild O. sinensis and its mycelia; (b) cluster analysis of glycosides; (c) cluster analysis of amino acids and their derivatives; (d) cluster analysis of nucleosides; (e) cluster analysis of peptides. O. sinensis: Ophiocordyceps sinensis.
Amino acids are the basic units that make up proteins, with amino (-NH2) and carboxyl (-COOH) functional groups, both connected to the same α-carbon atom. Their structural formula is H2N-CH(R)-COOH, where R is the variable side chain that determines the physicochemical properties and classification of amino acids. Amino acid derivatives are biologically active substances formed by chemical modification of amino acids (such as substitution, decarboxylation, hydroxylation, etc.). Amino acids and their derivatives have a wide range of biological activities and have been widely used in the fields of medicine, food, and cosmetics [10]. All samples tested in this survey contained a rich variety of amino acids and their derivatives. Particularly, NC had the richest variety of amino acids and their derivatives, followed by JST and SN, then with the least in NQ and QD (Figure 4). Detailed analysis reveals that all samples contained these common components: N-(1-deoxy-1-fructosyl) alanine, tyrosine, acetylcarnitine, acetyl leucine, phenylalanine, etc. For adults, there are 8 essential amino acids, namely phenylalanine, lysine, threonine, methionine, tryptophan, leucine, isoleucine, and valine; for infants and young children, histidine, arginine, tyrosine, and cysteine are also included. NC contained 4 of them: lysine, phenylalanine, threonine, either leucine or isoleucine. JST contained 4 of them: lysine, phenylalanine, tryptophan, and threonine. NQ and QD contained 6 of these essential amino acids: lysine, phenylalanine, histidine, arginine, tyrosine, and tryptophan. SN contained 4 of them: phenylalanine, tyrosine, tryptophan, and methionine (Table S1E).
The geographical distribution patterns of wild O. sinensis and its mycelia metabolic lineages. The heatmap was done by R (–3 < z score < 3). O. sinensis: Ophiocordyceps sinensis.
Short polypeptides, also referred to as small peptides or oligopeptides, are formed through the connection of 2 to 10 amino acids via peptide bonds. They possess a molecular weight ranging from approximately 200 to 1,000 Da and are characterized by their direct and complete absorption by the human body. This survey found that wild O. sinensis samples contained a variety of short peptides (dipeptides and tripeptides), while JST did not contain unique short peptides, only 6 of them, namely Ile-Asp, Pro-Tyr, Val-Tyr, Gly-Phe, Ala-Phe (Figure 3e). The distribution pattern of short peptides in wild O. sinensis samples exhibits similarity to that of amino acids and their derivatives, with the number of short peptides in each sample ordered as follows: NC, SN, NQ, QD. A tripeptide, Thr-Tyr-Thr, was detected in NQ, QD, and JST, but not in SN and NC. Dipeptides and tripeptides are more easily absorbed than free amino acids, with an absorption rate 2 to 2.5 times that of free amino acids. Peptide fragments can be absorbed intact from the intestinal epithelium into the bloodstream, thereby promoting potential biological effects at the tissue level [11]. Dipeptides and tripeptides containing Trp, Tyr, Cys, and Met residues are described as having high free radical scavenging activity, such as Val-Tyr with antioxidant activity [12]. Short peptides also impart flavor to food, such as Ala-Glu and Gly-Leu, giving bitterness to dry-cured ham [13]; however, short peptides isolated from O. sinensis have not been reported to date.
Nucleosides, biologically active compounds derived from nucleotides, play crucial roles in physiological functions. The 2020 Chinese Pharmacopoeia identifies adenosine as the main effective component in O. sinensis (Chinese Pharmacopoeia [S], Part I. 2020:119). This study detected 12–14 nucleoside types in wild O. sinensis and 18 types in a more diverse sample (Figure 3d). All samples shared 9 common nucleosides, while NQ and JST shared five additional nucleosides not found in others. JST contained three unique nucleosides such as cyclic adenosine 5'-diphosphate-ribose (ADP-ribose), 2'-deoxyuridine, and thymidine (Table S1A–E). Cordyceps nucleosides are key active components in O. sinensis with multiple biological functions [14, 15]. The roles of unique compounds in JST, such as cyclic ADP-ribose and thymidine, warrant further investigation in O. sinensis.
In addition to the components mentioned above, this survey also discovered other active ingredients, including vitamins, fatty acids, glycerophospholipid metabolites, esters, organic acids, sphingolipids, aldehydes, etc. (Table 1). Some of these substances and their metabolites are important structural molecules that maintain normal human life activities and constitute cell membranes, while others participate in important signaling processes such as cell growth, differentiation, aging, and programmed cell death [16].
To better illustrate the differences in the distribution of components in O. sinensis from different regions, we conducted a cluster analysis of the metabolic species in wild O. sinensis and its mycelia. The results show that wild O. sinensis from different geographical sources exhibit significant enrichment characteristics in key metabolic categories such as amino acids and their derivatives, short peptides, nucleosides, and glycerophospholipid metabolites. Despite geographical differences, the afore-mentioned metabolic categories still dominate the overall chemical component profile. It is noteworthy that compared to the fruiting bodies of O. sinensis, the variety of short peptide metabolites in the mycelia is significantly reduced, while the abundance of amino acid derivatives (such as N-acetylglutamate, γ-aminobutyric acid) and nucleosides (such as adenosine, guanosine) is comparable to that of the fruiting bodies (Figure 3).
Based on the cluster heat map analysis of the metabolome of O. sinensis across different regions, we observed significant geographically dependent differences in components. The four geographical groups (NC, SN, NQ, QD) showed clear clustering separation in their metabolic expression profiles, with the metabolic expressions of the NC and SN groups being the most similar, while the QD group exhibited distinct regional specificity in metabolic expression compared to other areas. The JST group formed an independent branch through hierarchical clustering, suggesting that its metabolic composition is unique compared to wild O. sinensis, which may be related to its different growth status. These data confirm from a metabolic perspective that there is a significant interactive effect between the chemical components of wild O. sinensis and its geographical habitat and growth status (Figure 4).
Nucleosides in O. sinensis, such as adenosine, cordycepin, uridine, guanosine, inosine, and purines, are crucial bioactive components. Adenosine, a quality marker in the Chinese Pharmacopoeia, offers cardioprotection, anti-inflammatory, and anti-tumor effects [17, 18]. Cordycepin, another key quality indicator, has anti-tumor, anti-inflammatory, anti-viral, anti-diabetic, anti-ischemic, and anti-leukemic properties [2, 19]. Guanosine has anticonvulsant effects [20], uridine shows antidepressant activity and nervous system regulation, inosine has anti-inflammatory effects, xanthine benefits asthma, and hypoxanthine aids iron absorption and cognitive development [21]. JST and samples from NQ have similar nucleoside profiles, with unique components like cyclic adenosine diphosphate ribose (cADPR), 2',3'-cGMP, 2',3'-deoxyuridine, and thymidine. cADPR is a potent calcium mobilization messenger, critical for cell proliferation and differentiation, while thymidine is a precursor for the antiretroviral drug zidovudine (AZT). These compounds contribute to JST’s diverse bioactivities compared to wild O. sinensis. Cordycepin acid (mannitol) and cordycepin are important active components [5]; cordycepin acid is found in all samples, but cordycepin is absent in the sample from QD, possibly due to its low content affecting detection. This suggests QD’s sample quality is lower than that of other wild O. sinensis and OS samples. Our analysis shows JST has more compounds and higher nucleoside content than samples from NQ and QD, and more nucleoside components than wild O. sinensis from four production areas (Table 1). Thus, JST could potentially replace wild O. sinensis.
Short peptides, especially dipeptides and tripeptides, are another class of non-volatile components with significant differences in variety between JST and wild O. sinensis, with the wild variety having a noticeably greater number of types than the mycelia. The primary reason for this outcome is the presence of carcass remnants from the moth larvae in wild O. sinensis. Most bioactive peptides are directly derived from traditional dietary proteins, such as milk proteins, marine organism proteins, meat proteins, and plant proteins [22]. For example, enzymatic hydrolysis has been utilized to derive potent angiotensin-converting enzyme (ACE) inhibitory peptides from the sarcoplasmic protein extract of beef gluteus medius, revealing that the active peptide is a hexapeptide capable of lowering blood pressure. Additionally, two ACE inhibitory peptides have been isolated from soybean alkaline hydrolysates [23].
O. sinensis is the result of O. sinensis parasitizing the larvae of bat moths and absorbing nutrients from them for reproduction. Consequently, the abundant proteins in these larvae are readily degraded by O. sinensis to produce a diverse range of short peptides, potentially explaining why wild O. sinensis has been identified to have more types of short peptides (dipeptides and tripeptides) than JST. Dipeptides and tripeptides are more easily absorbed than free amino acids, with an absorption rate 2–2.5 times that of free amino acids. Peptide fragments can be absorbed intact from the intestinal epithelium into the bloodstream, thereby promoting potential biological effects within the body at the tissue level [11]. Therefore, in terms of the variety of short peptides, wild O. sinensis may be nutritionally richer than the mycelium. Some studies have proven that certain dipeptides have bioactivity, such as Leu-Tyr having antidepressant effects, and Tyr-Tyr having anti-tumor effects [24, 25]. Dipeptides and tripeptides containing Trp, Tyr, Cys, and Met residues are described as having high free radical scavenging activity, such as the antioxidant activity of Val-Tyr [10]. Short peptides also impart flavor to food [26]. Currently, there are few reports on the isolation of bioactive dipeptides and tripeptides from O. sinensis. Cordymin, isolated from the fruiting bodies of C. militaris, has shown anti-inflammatory activity, and cordycepedipeptide A, cordyceamides A, and cordyceamides B, isolated from the fermentation broth of O. sinensis, have been reported to be toxic to several tumor cells, but the research is not in-depth [27, 28]. Other classes of non-volatile components, such as glycosides, amino acids and their derivatives, fatty acids, esters, sphingolipids, vitamins, and glycerophospholipid metabolites, exhibit minimal differences between wild O. sinensis and its mycelia, or the component varieties are identical, such as vitamins and sphingolipids. Alternatively, the varieties found in JST vary among the four production areas of wild O. sinensis, including amino acids and their derivatives, organic acids, glycosides, fatty acids, and glycerophospholipid metabolites.
Non-volatile components in wild O. sinensis vary significantly by production area, with amino acids, their derivatives, short peptides, and organic acids differing notably. Amino acids and short peptides are more abundant in NC and SN, while organic acids are more prevalent in JST. Glycerophospholipid metabolites are less common in NC. Samples from lower altitudes (below 4,000 meters) in NC and SN contain a richer variety of these components, with QD, the lowest in altitude, having the fewest varieties. Geographical origin and developmental stage significantly affect wild O. sinensis metabolite quality, which is crucial for its scientific assessment, quality control, and sustainable use. However, the study’s small sample size limits generalizability, and comprehensive pharmacological validation is lacking. Our laboratory is conducting complementary research on the pharmacological activities of wild O. sinensis from various origins to address these limitations.
In summary, there are significant functional differences between wild O. sinensis and its fermented mycelium. Wild O. sinensis typically regulates immune function through the synergistic effects of polysaccharides, nucleosides, and cyclic peptides, which are suitable for systemic immune balance. On the other hand, mycelia rely on a high content of polysaccharides and nucleosides, making it more suitable for non-specific immune enhancement.
Wild O. sinensis and its fermented mycelia are both repositories of bioactive compounds, with the fermented mycelium potentially exceeding the wild variety in certain active components. However, the fermented form’s overall functionality falls short of the synergistic benefits offered by the intricate blend of compounds present in naturally occurring samples. The active components of wild O. sinensis are significantly influenced by its geographical origins and growth conditions, highlighting the importance of these factors in its efficacy. Looking ahead, it is imperative to integrate metabolomics with clinical research to craft targeted mycelial product formulations, such as those designed to assist in tumor treatment, while also safeguarding the wild resources. This study lays a theoretical foundation for the judicious incorporation of O. sinensis into daily diets or as a preventative measure against diseases by analyzing the metabolite profiles and functional disparities between wild O. sinensis and its fermented mycelia.
ACE: angiotensin-converting enzyme
ADP-ribose: adenosine 5'-diphosphate-ribose
cADPR: cyclic adenosine diphosphate ribose
ESI: electrospray ionization
O. sinensis: Ophiocordyceps sinensis
PAD: photodiode array detector
UPLC-Q-TOF-MS: ultra-performance liquid chromatography quadrupole time-of-flight mass spectrometry
The supplementary figure for this article is available at: https://www.explorationpub.com/uploads/Article/file/1010124_sup_1.pdf. The supplementary table for this article is available at: https://www.explorationpub.com/uploads/Article/file/1010124_sup_2.pdf.
We would like to acknowledge the Instrumental Analysis Center at Shanghai Jiao Tong University for their invaluable assistance in conducting sample testing analysis and expertly handling data processing.
XWZ: Conceptualization, Writing—review & editing. MYJ: Investigation, Writing—original draft. LDZ and J Lin: Validation, Writing—review & editing, Supervision. J Lan and DD: Investigation, Resources. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
The authors confirm that the data supporting the findings of this study are available within the article and supplementary materials.
This work was supported by the Science and Technology Projects of Xizang Autonomous Region, China and the NMPA Key Laboratory for Quality Control of Traditional Chinese Medicine, Tibetan Medicine [XZ202401YD0020]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 872
Download: 17
Times Cited: 0
