Affiliation:
1Nutritional Biochemistry Research Group, Faculty of Food Science and Nutrition, Universiti Malaysia Sabah, Kota Kinabalu, Sabah 88400, Malaysia
Email: odjohn@ums.edu.my
ORCID: https://orcid.org/0000-0001-5035-1878
Affiliation:
2School of Applied Sciences (FCA), University of Campinas (UNICAMP), Limeira, SP 13483-350, Brazil
ORCID: https://orcid.org/0000-0002-1780-2160
Affiliation:
3Department of Biosystems and Agricultural Engineering, Food and Health Engineering Laboratory, Michigan State University, East Lansing, MI 48824, USA
ORCID: https://orcid.org/0000-0001-9041-2743
Affiliation:
4Department of Bioscience, Faculty of Applied Sciences, Tunku Abdul Rahman University of Management and Technology, Setapak, Kuala Lumpur 53300, Malaysia
ORCID: https://orcid.org/0000-0002-2637-9202
Affiliation:
4Department of Bioscience, Faculty of Applied Sciences, Tunku Abdul Rahman University of Management and Technology, Setapak, Kuala Lumpur 53300, Malaysia
ORCID: https://orcid.org/0000-0001-5869-2132
Affiliation:
5Faculty of Science, Asia-Pacific International University, Muak Lek, Saraburi 18180, Thailand
Affiliation:
5Faculty of Science, Asia-Pacific International University, Muak Lek, Saraburi 18180, Thailand
ORCID: https://orcid.org/0009-0003-8752-4586
Affiliation:
5Faculty of Science, Asia-Pacific International University, Muak Lek, Saraburi 18180, Thailand
Email: tatenda@apiu.edu
ORCID: https://orcid.org/0009-0006-5079-4242
Explor Foods Foodomics. 2026;4:1010118 DOI: https://doi.org/10.37349/eff.2026.1010118
Received: August 29, 2025 Accepted: February 05, 2026 Published: March 01, 2026
Academic Editor: Cristina Garcia Viguera, CEBAS-CSIC, Spain
Betacyanins are bioactive compounds found in Caryophyllales, including red beetroots (Beta vulgaris), amaranths (Amaranthus sp.), and red dragon fruits (Hylocereus polyrhizus). Their biosynthesis requires several enzymes, including tyrosinase, 4,5-DOPA-extradiol-dioxygenase (DOD), and 5-O-glucosyl transferase (5GT). Environmentally friendly extraction techniques, such as ultrasound-assisted, microwave-assisted, supercritical fluid, and pulsed electric field extraction, improve the recovery of betacyanins from natural resources. Betacyanins have commercial value as food coloring agents, for smart food packaging, and in the nutraceutical, pharmaceutical, and cosmetic industries. Industrial application is expanding as more innovative methods are discovered. Several factors affect the absorption of betacyanins, including gastrointestinal degradation, the nature of the ingested betacyanins, and the food matrix. Betanin, the main betacyanin, is excreted through urine when given intravenously. However, orally administered betanin showed limited urinary excretion, suggesting extensive modification or digestion in the gastrointestinal tract. Biologically, betacyanins are shown to have antioxidant, anti-inflammatory, antidiabetic, hypolipidemic, gut-microbiome-modulatory, antiproliferative, and antimicrobial properties. Recent molecular docking developments showed this compound group’s potential in modulating key target enzymes and proteins.
Natural color pigments are found in plant tissues, animal cells (e.g., carminic acid and kermesic acid), the metabolism of microorganisms, or mineral sources (e.g., calcium carbonate, titanium dioxide) [1]. Betalains are a group of naturally occurring water-soluble pigments and specialized (secondary) metabolites that contain nitrogen (chromoalkaloids) [2]. Compared to anthocyanins, these pigments are water-soluble and have three times the tinctorial strength and stability at lower pH levels [3]. Plants in the order Caryophyllales are the main producers of betalains [4], which are found in the leaves, roots, and fruits of certain species within the order, including the genera Beta, Amaranthus, Opuntia, and Hylocereus [5]. Additionally, betalains are reported to be potent antioxidants [6, 7].
Betalain pigments can be categorized into two main structural groups: betaxanthins and betacyanins, which are distinguished by their yellow-orange and reddish-violet colorations, respectively [2, 8, 9]. The amounts of betalain pigments, such as betaxanthins and betacyanins, differ from plant to plant [10]. Both betacyanins and betaxanthins have betalamic acid as the core compound. Specifically, betacyanins are conjugated with cyclo-dihydroxyphenylalanine (DOPA) and undergo O-glycosylation or acetylation [11], whereas betaxanthins are formed by the condensation of betalamic acid with various amino acids, such as proline in indicaxanthin and glutamine in vulgaxanthin I [12] (Figure 1).
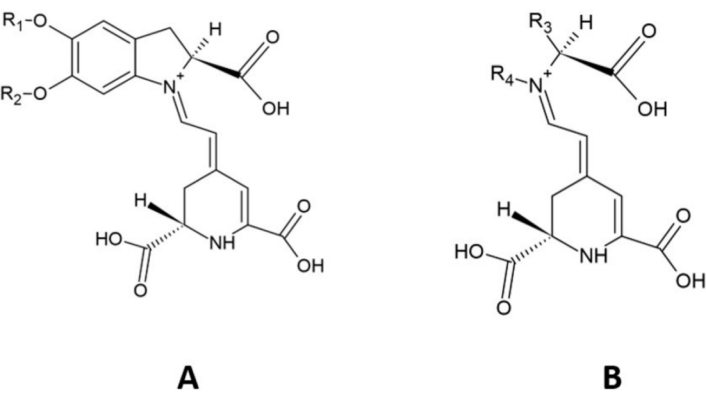
Structure of (A) betacyanin parent molecule and (B) betaxanthin parent molecule. R1 and R2: hydrogen or sugar moieties; R3 and R4: amino acids, amines, or their derivatives. Adapted from [13]. Copyright © 2023 Ornelas García, Guerrero Barrera, Avelar González, Chávez Vela and Gutiérrez Montiel. CC BY.
Betanidin-5-O-β-glucoside (betanidin) is the primary structure of all betacyanins, by which acylation and glycosylation of the 5-O- or 6-O-glucosides yields various betacyanin structures [14]. Examples of betacyanins include betanin, isobetanin, probetanin, neobetanin, amaranthin, and isoamaranthin [5, 15]. Betacyanins are classified into four distinct types based on attachment of glucosyl groups to oxygen atoms in the O-position on the cyclo-dopa moiety [14], including (i) betanin-type, (ii) gomphrenin-type, (iii) amaranthine-type, and (iv) bougainvillein-type pigments [16]. The betanin-type group has a glucosyl or derivative linked to the C5 carbon and a hydroxyl attached to C6; the amaranthin-type group contains a glucuronyl-glucosyl moiety or derivative linked to C5; the gomphrenin-type group has a hydroxyl at C5, and glucosyl or derivative at C6; and the bougainvillein-type group may harbor a diglucosyl moiety or derivative at C5 or C6 of carboxylated or decarboxylated betacyanins [14, 16].
This review discusses the natural sources, biosynthesis, bioavailability, extraction methods, and industrial applications of betacyanins. Building on this, we examine their health benefits, particularly in relation to cardiometabolic disease, and consider their potential roles as anticancer and antimicrobial agents. Furthermore, we expand on previous publications by discussing their molecular docking properties and exploring their emerging applications in food, natural products, and health.
Betanin (betanidin-5-O-β-glucoside) is the most prevalent betacyanin found in plants [17]. In particular, betanin is mainly found in red beetroots (Beta vulgaris L.), one of the best sources of betacyanins [5, 18]. Furthermore, betacyanin traces are also visible in all tested varieties of B. vulgaris [19]. The natural red food colorant betanin, approved under food additive regulations (E162), is also permitted in small quantities and used in pharmaceuticals and cosmetics [20].
The fruits of Opuntia species contain several betacyanins, including betanidin, isobetanin, isobetanidine, and neobetanin [21], with examples like Opuntia stricta, O. joconostle, and O. dillenii [22–24]. Betacyanins, mainly betanidin 5-O-β-sophoroside, were also recovered from the edible fruits of four Melocactus species: M. violaceus, M. bahiensis, M. amoenus, and M. curvispinus [25]. Betacyanin levels are generally higher than betaxanthin levels in pear cactus and bougainvillea [10]. The peel of red dragon fruit or pitaya (Hylocereus polyrhizus) contains high betacyanin concentration, giving the peel its reddish-violet hue [26, 27]. Higher concentrations of betacyanins are recovered in the peel, which is usually discarded as food waste [28]. Interestingly, although earlier reports suggested that betacyanins and anthocyanins are mutually exclusive in plants [29, 30], studies have found both in red pitaya [27, 31], a finding that is still under discussion [32, 33]. Despite this, distinctions exist between these two compounds; for instance, anthocyanins are more stable in acidic conditions [34], whereas betacyanins are more stable from pH 3–7 [11]. The different types of plants and their betacyanin contents, such as B. vulgaris [19, 35, 36], Opuntia ficus-indica [37], Amaranth [37], H. polyrhizus [38], and Melocactus spp. [25] are summarized in Table 1.
Plant sources and their betacyanin concentration.
| Plant source (scientific name) | Plant part/variety | Betacyanin concentration | Units | Citation |
|---|---|---|---|---|
| Red Beet (Beta vulgaris L.) | ‘Monorubra’ cultivar (hypocotyl extract) | 9.699 | mg/100 mL | Skalicky et al. [19] |
| Red Beet (Beta vulgaris L.) | ‘Libero’ cultivar (hypocotyl extract) | 8.42 | mg/100 mL | Skalicky et al. [19] |
| Red Beet (Beta vulgaris L.) | Red beetroot (total betalains) | 650–800 | µg/g fw | Koss-Mikołajczyk et al. [35] |
| Red Beet (Beta vulgaris L.) | Red beetroot | 218.80 | mg/100 g | Sokolova et al. [36] |
| Prickly Pear (Opuntia ficus-indica) | Purple fruit peel extract | 77.47 | mg/g of source | Sigwela et al. [37] |
| Amaranth (Amaranthus sp.) | Leaf (amaranth) | 5.07 | mg/g of source | Sigwela et al. [37] |
| Red Dragon Fruit (Hylocereus polyrhizus) | Peel extract | 36.67 | mg/100 g | Halimfanezi and Asra [38] |
| Melocactus (Melocactus amoenus) | Fruit extract | ~0.08 | mg/g | Sutor and Wybraniec [25] |
fw: fresh weight.
Plants of the Amaranthaceae family contain betacyanins, consisting of amaranthine-type, gomphrenin-type, and betanin-type pigments [39]. For instance, Alternanthera, Amaranthus, Beta, Chenopodium, Celosia, and Gomphrena are several genera in Amaranthaceae containing betalains [40]. Amaranthus gangeticus is rich in betacyanins, including amaranthine, iso-amaranthine, betanin, and iso-betanin [41]. Gomphrenin-I, a specific betacyanin, has been identified in the flowers of Gomphrena globosa [39, 42, 43]. Betanin monoglucoside, and its 4-coumaroyl and feruloyl derivatives, are the main betacyanins present in the fresh juice of Basella rubra fruits [44]. The fruits of nine Mammillaria species contain betanidin 5-O-(6′-O-malonyl)-β-sophoroside, identified via spectroscopic techniques [45]. The fruits of Basella alba L. and Basella alba L. var. “Rubra”, and mushrooms of genera Amanita, Hygrocybe, and Hygrophorus also contain betalains [11]. Additional sources of betacyanins include Talinum triangulare [46], some Bougainvillea species [47], and wild pokeweed (Phytolacca americana) [48].
Identifying new natural sources of betacyanin can increase available pigment alternatives beyond Caryophyllales plants. For example, Garcinia, with over 250 species, should be explored for its betacyanin content due to its variety and colorful nature [49]. In addition, extracting pigments from underutilized sources or food waste like fruit peel is promising for its environmental and practical benefits [50, 51].
Several steps are needed for the biosynthesis of betacyanins (Figure 2). Betanin goes through additional glycosylation and acylation reactions, leading to more complex betacyanins [52]. The formation of beta cyclins is a complex process involving many enzymes. Studies in plants have shown a positive correlation between betacyanin levels and a bifunctional tyrosinase enzyme (cytochrome P450 enzyme CYP76AD1). This enzyme is necessary for the hydroxylation and oxidation activities needed for the biosynthesis of betalains, which are further modified to give betacyanins. This correlation was discovered by Wang et al. [53] when they observed tyrosinase synthesis together with betacyanin production in the cotyledons of Suaeda salsa seedlings grown in the dark. When exposed to light, the levels of tyrosinase and betacyanin decreased, suggesting a link between exposure to light and the degradation of both molecules. Other studies also show an inverse relationship between betacyanin synthesis and temperature. Lower temperatures favor synthesis, and higher temperatures reduce synthesis in B. vulgaris root samples [54, 55]. Apart from temperature, other ecological factors play roles in the synthesis and accumulation of betacyanin. These include salinity, pH (4–6), and the presence of metals in the environment [56].
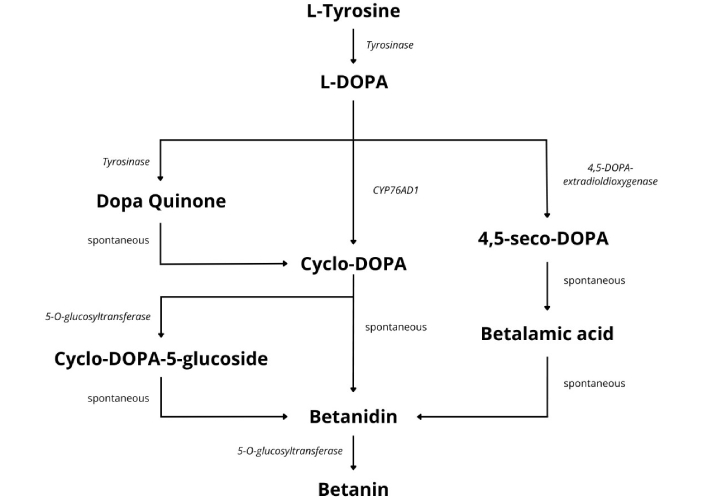
The biosynthesis of betacyanin pigments. In a series of reactions, tyrosine is converted to L-DOPA by tyrosine hydroxylase, which is then used by 4,5-DOPA-extradiol-dioxygenase (DOD) to produce 4,5-seco-DOPA, which is transformed into betalamic acid, which serves as a building block for either betacyanins or betaxanthins synthesis. This figure was drawn following pathway information synthesized from other papers referenced in this review [52–68]. L-DOPA: L-dihydroxyphenylalanine.
Apart from the tyrosinase enzyme, other enzymes are instrumental in the biosynthesis of betacyanins. These include 4,5-DOPA-extradiol-dioxygenase (DOD) and 5-O-glucosyl transferase (5GT) [57]. Their heterologous expression has been shown to lead to de novo production of betacyanin pigments in transgenic Eustoma plants [58]. DOD is a critical enzyme in this pathway. It catalyzes the formation of a betalamic acid, a structural intermediate for all betalains [59]. Researchers can make either beta-cyclins or betaxanthins using betalamic acid as a starting point [60]. The reaction begins with a tyrosine amino acid converted to L-DOPA by tyrosine hydroxylase. From DOPA, several pathways can branch off to give betanidin. In one pathway, DOD uses DOPA as a substrate for this reaction and cleaves it via an extradiol cleavage reaction, producing 4,5-seco-DOPA, which spontaneously undergoes a recyclization reaction, forming betalamic acid, which spontaneously becomes betanidin.
In another pathway, L-DOPA is metabolized by various enzymes like tyrosinase to give DOPA-quinone. DOPA-quinone undergoes an intramolecular cyclization, forming a molecule called aminochrome, which spontaneously converts to cyclo-DOPA. Cyclo-DOPA is glycosylated at the 5-O position by cyclo-DOPA 5-O-glucosyltransferase to form cyclo-DOPA 5-O-glucoside. Finally, cyclo-DOPA 5-O-glucoside spontaneously condenses with betalamic acid to form betanidin [11, 61–63]. Betanidin is further glycosylated by betanidin 5-O-glucosyl-transferase encoded by B5GT to form betanin, the most common betacyanin [64, 65].
Recent studies show that betalamic acid can be engineered chemically [60, 66]. Other researchers are looking for ways to increase yield by optimizing the pathway from tyrosine to the final betalain product by selecting the best-performing isozymes from betalain-producing species and the optimal combination of regulatory elements that drive their expression [64]. Timoneda et al. [64] reported a seven-fold increase in betalain production in Nicotiana benthamiana. Other studies are exploring whether betalain production could be engineered in various organisms to have new potential sources for betalains. These developments and other current biochemical metabolic engineering studies have been discussed by Polturak and Aharoni [52].
Further, metabolic engineering for betalain production in crops like vegetables, fruits, and cereals may offer new food resources beneficial for healthcare, since betalains are primarily found naturally in plants of the order Caryophyllales. Several advancements have been made, for example, with the help of appropriate specific promoters, the transgenic tomato fruits and potato tubers with co-expression of the genes involved in betacyanin biosynthesis CYP76AD1 from B. vulgaris, DOD (DOPA 4,5-dioxygenase), and 5GT (cyclo-DOPA 5-O-glucosyltransferase) from Mirabilis Jalapa, have dark red tissues with an enriched accumulation of betacyanins (betanin and isobetanin) [67].
Current developments in betanin production include combinatorial engineering of plant P450 and UDP-glycosyltransferases (UGTs) enzymes and precursor metabolisms to enhance the de novo betanin production in Saccharomyces cerevisiae [68]. Li et al. [68] employed a multifaceted strategy focusing on improving the activity of the key cytochrome P450 enzyme, CYP76AD. Targeted mutagenesis of the CYP76AD enzyme led to the discovery of a mutant that exhibited a ~7-fold increase in betanin titer compared to the wild-type enzyme, underscoring the effectiveness of targeted mutagenesis in fine-tuning P450 activity. Notably, upregulation of the bio-synthetic pathway and optimizing the UDP-glucose levels supported efficient glycosylation, a critical step in betanin formation [68]. Glitz et al. [69], critically assessed which of the UGTs increased the production of betanin in yeast.
The authors’ comprehensive in vivo screening of 27 plant-derived UGTs in Saccharomyces cerevisiae revealed two previously uncharacterized UGTs—CqGT2 (UGT73A37) from Chenopodium quinoa and BgGT2 (UGT92X1) from Bougainvillea glabra—which the authors suggest are likely involved in betanin synthesis in their native plants [69]. Functional validation of these UGTs in Yarrowia lipolytica revealed that CqGT2 was the most effective UGT for betanin production in this non-conventional yeast host. Li et al. [68] and Glitz et al. [69] highlight how protein engineering and pathway optimization can enable high-level production of complex plant-derived natural products in microbial hosts.
Extraction processes for obtaining phytochemicals such as betacyanins from plant sources need to be carefully chosen [70]. Conventional methods, such as Soxhlet and decoction, are often inefficient and environmentally unfavorable. This occurs due to the use of large amounts of organic solvents and the need for continuous heating, sometimes lasting several hours, raising environmental and safety concerns [71, 72]. This has driven the development of green extraction techniques, aiming to minimize solvent use, reduce labor, and improve sustainability. This section explores various green extraction methods for betacyanin extraction, including ultrasound-assisted extraction (UAE), microwave-assisted extraction (MAE), supercritical fluid extraction (SFE), enzyme-assisted extraction (EAE), and pulsed electric field (PEF). These methods offer improved automation, enhanced selectivity, higher extraction efficiency, and reduced solvent consumption [70, 71].
The UAE principle utilizes acoustic cavitation, which induces microjetting, resulting in surface peeling and particle breakdown, thereby increasing the extraction yield [70, 71]. This method has shown promising results when used to recover betacyanins from red dragon fruit. Studies have investigated the effects of various UAE parameters, including temperature, solvent composition, and power, on betacyanin yield [73, 74] (Table 2). Optimal conditions have been identified, resulting in yields of 0.84–1.31 mg/g dry weight (dw) [73]. Furthermore, combining the UAE with enzymatic treatment has demonstrated significant improvements in betacyanin extraction [75]. Response surface methodology was used to optimize this combined approach, achieving a high betacyanin concentration of 154.24 mg/L. Silva et al. [76] investigated the effect of water as a solvent for UAE and found that 83 W of ultrasonic power and a solvent/sample ratio of 75 ml/g yielded optimal results from beetroot. These studies collectively demonstrate the potential of UAE and its combinations for efficient and effective extraction of betacyanins from red dragon fruit, enhancing the recovery of these valuable bioactive compounds. In conclusion, after undergoing ultrasonic and enzymatic treatments, the bioactive compounds in the tested samples were enhanced, contributing greatly to their antioxidant properties, according to the study.
MAE is a technique that involves using electromagnetic waves, usually ranging from 0.3 to 300 GHz, to generate heat by interacting directly with molecules through ionic conduction and dipole rotation, particularly with polar molecules [77]. By applying electromagnetic waves, MAE can selectively heat polar molecules within plant matrices, leading to increased extraction yields. Studies have shown that MAE can significantly enhance betacyanin extraction from various sources, including red beetroot [78] and Amaranthus tricolour leaves [79]. For example, applying MAE to extract betalains from red beetroot resulted in a significant increase in betacyanin content, reaching 130 mg/L compared to 75 mg/L [78] (Table 2). Similarly, optimizing MAE parameters for A. tricolour leaves led to a high betacyanin recovery of 71.95 mg/g of dw [79]. Furthermore, MAE has been successfully employed for extracting betacyanins from beet root peel using water as a solvent [80]. These findings demonstrate the potential of MAE as a promising green extraction method for obtaining betacyanins from various plant sources, offering advantages such as reduced processing time and energy consumption.
Supercritical fluid can be produced by subjecting a gas to certain pressure and temperature conditions. This substance exhibits properties between those of a gas and a liquid, allowing for the dissolution and extraction of substances [81]. Betacyanin extraction through supercritical carbon dioxide (SCCD) and high hydrostatic pressure (HHP) in beetroot (B. vulgaris L.) was tested [82] (Table 2). The study involved the application of treatments using SCCD and HPP methods. For SCCD, the treatments were administered at 10, 30, and 60 MPa for 10 minutes at a temperature of 45°C. On the other hand, for HPP, treatments were applied at 200, 400, and 500 MPa for 5 minutes at a temperature of 20°C. It was observed that the use of SCCD treatment at 30 MPa was particularly advantageous as it improved the bioaccessibility of total betacyanins by 58%. Moreover, when HHP was applied, higher bioaccessibility of betacyanins was noted in HHP200 MPa and HHP400 MPa, by 35% and 32%, respectively [82]. Demonstrating that applying these techniques has increased the extraction of this compound, improving bioaccessibility.
Another type of extraction is SFE. Fathordoobady et al. [83] used SFE with CO2 as solvent plus conventional extraction using 10% of EtOH/water as co-solvent (EtOH/water, 10/90, v/v) to extract betacyanin contents of the peel and fresh extract from the red pitaya (H. polyrhizus) (Table 2) [83]. The extraction process was conducted at 25 MPa and 50°C. The flesh of H. polyrhizus provided higher betacyanins than its peel, regardless of the applied extraction method. Moreover, EtOH/water solvent mixtures of 50/50 (v/v) and 70/30 (v/v) resulted in the highest desirable betacyanins from the peel and flesh of H. polyrhizus, in the order of 28.44 mg/100 mL and 120.28 mg/100 mL, respectively [83]. In a different study by the same team, they optimized the extraction conditions for betacyanins from H. polyrhizus peel, resulting in a 25.49 mg/100 mL of total betacyanins. The optimised conditions were 25 MPa, 50°C, and 15% co-solvent [84].
The application of enzymes such as pectinases or cellulases offers an alternative form of betacyanin extraction from plant tissues through the hydrolysis of cell walls [81]. Lombardelli et al. [85] suggest in their research a need for a fitted protocol based on a targeted substrate enzymatic extraction that avoids using organic solvents to recover betalains from unsold red beets. The enzymatic mix was developed based on the composition of red beet cell walls. It contained 37% cellulase, 28% polygalacturonase and pectin lyase, and 35% xylanase. The mixture was applied at pH 5.5, at 45°C and 25°C. The enzymatic mix’s total dose and processing time were optimized (10–50 U/g, 20−300 min) to enhance betacyanin recovery yield. The authors demonstrated that the minimal suitable total dosage was 25 U/g at 45°C and at 25°C temperatures. Also, at 45°C, the extraction yield of betacyanin ranged from 6.03 to 12.15 mg/mL per unit at 20 min and 120 min, respectively [85].
Therefore, Van et al. [86] utilized the EAE of betacyanins from red dragon fruit peel (Table 2). The source was obtained from the Pectinex Ultra SP-L obtained from Aspergillus aculeatus. The authors aimed to consider the enzyme concentration and the extraction time, in which the enzyme concentrations ranged between 0.25–2.75% of the v/w dw of the fruit peel, with the extraction time varied from 5 min to 30 min. The betacyanin content increased by 31.59% (from 0.21 to 0.30 mg/g) when the concentration increased from 0.25% to 1.75% v/w dw. However, after 1.75%, the concentration did not increase in an exponential model. Hence, the authors determined 1.75% as the optimum extraction concentration. When comparing extraction time, the concentration of betacyanin increased up to 20 min, reaching a maximum of 0.30 mg/g, and remained constant [86].
This method induces electroporation, in which an intense electric field permeabilizes the cell membrane through dielectric breakdown [87]. Here, two electrodes are used to apply short-duration pulses of moderate electric voltage (0.5–20 kV/cm) to a substrate [88]. The PEF method has been utilized to extract betacyanin. Koubaa et al. [89] tested PEF to extract betacyanins from purple Opuntia fruits. They adjusted the electric fields and number of pulses using water as the extraction solvent, with constant agitation at room temperature for 1 h [89]. They found that applying 50 pulses at 20 kV/cm produced a yield of 50 mg betacyanins per 100 g fresh weight (fw), which was a significant two-fold improvement compared to the conventional method of constant agitation with water at room temperature for 1 h, which yielded only 20 mg per 100 g fw [89].
Among the methods discussed, UAE is generally the most effective for preserving bioactivity, as its cavitation mechanism enhances mass transfer and compound release while operating at relatively low temperatures. Compared with MAE, UAE better maintains pigment color and antioxidant potential under mild conditions, especially when optimized or combined with enzymatic treatments [73, 75]. MAE provides rapid extraction and high yields of betalains and phenolics, often resulting in elevated antioxidant activity in assays such as DPPH, ABTS, and FRAP. However, because MAE is a thermal technique, prolonged exposure or high power can degrade pigments and reduce color stability, making it less suitable for bioactivity preservation unless parameters are carefully optimized [79].
SFE, which uses CO2 with a polar co-solvent, also excels at retaining pigment integrity and antioxidant activity because it operates at low temperatures and limits oxidative degradation. Nevertheless, its total pigment recovery is typically lower than that of MAE or UAE, meaning it better preserves bioactivity per compound extracted but yields less extract overall [83, 84]. Low-temperature EAE has demonstrated superior pigment integrity and color stability compared to conventional solvent extraction, owing to enzymatic hydrolysis under mild conditions that prevent thermal degradation [85]. Combining EAE with UAE further enhances both pigment release and antioxidant capacity while maintaining gentle extraction conditions [87]. Finally, PEF is recognized as one of the most promising non-thermal pretreatments for preserving bioactivity, as it disrupts cell membranes through electroporation and enables subsequent extractions under mild conditions. When applied before UAE or solid-liquid extraction, PEF improves both yield and the retention of heat-sensitive compounds [87].
Selecting the optimal betacyanin extraction method requires careful consideration of factors like sustainability, yield, cost, and future applications. While methods like microwave or ultrasound excel in resource efficiency, factors like extraction time might favor techniques such as supercritical fluid or MAE. The green extraction methods are illustrated in Figure 3 and Table 2.
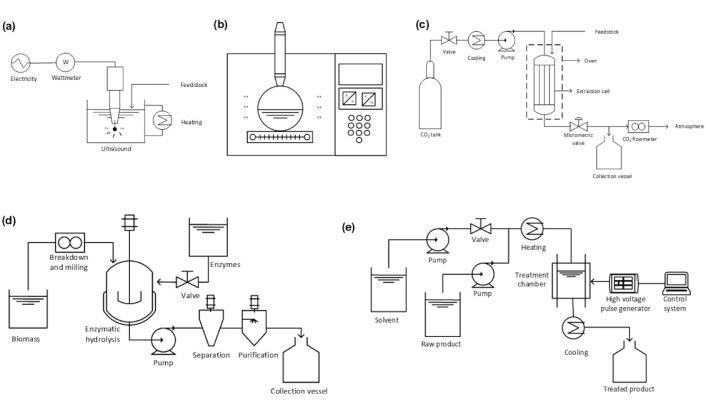
Green methods used in betacyanin extractions. (a) Ultrasound-assisted extraction (UAE); (b) microwave-assisted extraction (MAE); (c) supercritical fluid extraction (SFE); (d) enzyme-assisted extraction (EAE); and (e) pulsed electric field (PEF). Adapted with permission from [90]. © 2023 Elsevier B.V.
Green extraction methods for betacyanin extraction.
| Methods | ||||||
|---|---|---|---|---|---|---|
| Ultrasound-assisted extraction | ||||||
| Source | Solvent | Temperature (°C) | Time duration (min); ultrasound frequency (kHz/W) | Solid:liquid ratio (g/mL) | Betacyanin content | References |
| Hylocereus polyrhizus (F.A.C.Weber) Britton & Rose | Methanol in water | 10–60 | 2–30; 200/24 | 0.1:10–0.3:10 | 0.84–1.31 mg/g dw | [73] |
| Hylocereus polyrhizus | n.d. | 30–70 | 2–32; n.d. | n.d. | 134.87–154.86 mg/L | [75] |
| Beta vulgaris L. | Water | 30–60 | 30–60; 25 | 25–75 mg/mL | 3.36–4.48 mg/g | [76] |
| Microwave-assisted extraction | ||||||
| Source | Solvent | Power (W) | Time; cycles (s) | Solid:liquid ratio (g/mL) | Betacyanin content | References |
| Beta vulgaris L. | Ethanol | 360–900 | 2–6 s; 140–420 s | 1:20 | 130 mg/L | [78] |
| Amaranthus tricolour | Water | 200–700 | 5–15 min; 0.5–1 s | 1:80 | 71.95 mg/g dw | [79] |
| Beta vulgaris L. | Water | 100–800 | 30–50 s; n.d. | 0.1–0.2 w/v | 115.89 mg/100 g fw | [80] |
| Supercritical fluid extraction | ||||||
| Source | Solvent | Temperature (°C); pressure (psi/MPa) | Time (min) | Solid:liquid ratio (g/mL) | Betacyanin content | References |
| Beta vulgaris L. | n.d. | 45–85; 10–60 and 200–500 | 5–10 | n.d. | 10–23% | [82] |
| Hylocereus polyrhizus | Ethanol in water | 50; 25 | 90 | 1:10 | 28.44–120.28 mg/100 mL | [83] |
| Hylocereus polyrhizus | Ethanol in water | 40–60; 20–30 | 90 | 1:10 | 25.49 mg/100 mL | [84] |
| Enzyme-assisted extraction | ||||||
| Source | Solvent | Enzyme (%) | Temperature (°C); time (min) | Solid:liquid ratio (g/mL) | Betacyanin content | References |
| Beta vulgaris ssp. | Acetate buffer | CL: 37, PG and PL: 28, and XL: 35 | 25–45; 20–300 | 1:15 | 6.03–12.15 mg/L/U | [85] |
| Hylocereus polyrhizus | Water | AA: 0.25–2.75 | 20–50; 5–30 | 1:10 | 0.21–0.30 mg/g | [86] |
| Beta vulgaris ssp. | Water | AN: 0.1 | 30–70; 30–180 | n.d. | 135.23–154.42 mg/L | [75] |
CL: cellulase; PG: polygalacturonase; PL: pectin-liasic; XL: xylanase; AA: Aspergillus aculeatus; AN: Aspergillus niger; n.d.: not described.
The demand for natural food colorings has increased in recent years, driven by growing consumer awareness of health and environmental concerns. The drive for healthier living has pushed for the replacement of artificial food colorants with natural food coloring. The Natural Food Colours Association (NATCOL) estimates the food color market to grow from $1.6 billion to $2.5 billion between 2020 and 2025 [91, 92]. The market share of betacyanins is small due to extraction challenges, limited availability, and stability, but it has potential to grow [93, 94]. According to the Food and Drug Administration (FDA), under the E-162 code in the European Union, beetroot is permitted as a natural red food colorant [95]. Betacyanins present unique properties that make them suitable for various food applications, being an attractive alternative to synthetic food colorants [96]. However, a critical factor in the industrial application of betacyanins as natural food colorants is the stability under different processing and storage conditions [97, 98]. Recent research focused on understanding the stability of betacyanins in a variety of food matrices, including beverages (juices, smoothies, and herbal teas), confectionery (candies, gummies, and jellies), and dairy products (yogurt and ice cream) [99–101].
For instance, Roriz et al. [100] assessed the application and stability of betacyanin obtained from an enriched extract of red-fleshed pitaya peels (Hylocereus costaricensis) and the flowers of Amaranthus caudatus. The betacyanin-rich extracts were applied as a natural food colorant in tagliatelle pasta and meringue cookies. The main results obtained demonstrated that the recovered natural extracts showed promising stability, maintaining a deep pink color over 14 days of storage time [100]. Moreover, the consumer acceptance of betacyanin-based natural food colorings delves into the technical aspects of formulation and sensory perception [102, 103]. Recently, studies demonstrated that there is a positive acceptance of betacyanin-based food colorings, especially when considering factors such as naturalness, color intensity, sensory attributes, transparency in labeling, and perceived health benefits. Consumers associate betacyanin-based food colorings with potential health benefits, attributing antioxidant properties to these natural pigments [104, 105]. Furthermore, foods rich in betalains (including betacyanin) are part of many traditional cuisines. Beetroot is widely consumed in Mediterranean and Eastern European dishes [106], amaranth in South and Southeast Asia [107], cactus pear in Mexico and the Southwestern United States [4], and pitaya in Central America [108] and Southeast Asia [109]. This should make it easier to accept their inclusion in foods as natural pigments.
Driven by the growing demand for eco-friendly alternatives in the food industry, biodegradable films are the future of food packaging. The incorporation of betacyanins has emerged as a promising candidate for imparting coloration and additional functionalities to food packaging [110]. Recent research has shown that the incorporation of betacyanins in biodegradable films goes beyond coloration, and these pigments have demonstrated antioxidant properties, potentially enhancing the shelf life of perishable food products [111, 112]. The films incorporated with betacyanins also exhibited better flexibility and tensile strength, contributing to their overall performance in food packaging [113]. In addition, one notable property of betacyanins is their strong antimicrobial activity against pathogenic microorganisms, such as Staphylococcus aureus, Listeria monocytogenes, Escherichia coli, and Salmonella [114, 115].
Moreover, betacyanins exhibit a color change in response to variations in pH [116, 117]. In acidic environments, betacyanins display a vibrant red color, while the color shifts towards violet and blue in more alkaline or basic conditions [93, 118]. The pH sensitivity property of betacyanins makes them suitable for incorporation into food packaging materials as natural pH indicators. The food packaging based on betacyanins can act as a natural food indicator, especially in perishable food where pH changes may signify the onset of microbial activity or spoilage, specifically for this study, the usage of time-temperature bioindicator film using betacyanin was able to determine the shelf-life of fresh goat milk at room temperature (5 h) and freezing temperature (31 days) [112]. pH-sensitive betacyanin packaging aligns with the concept of smart packaging, where the packaging material interacts with the product or its environment to provide real-time information [119]. In conclusion, betacyanins offer a sustainable and multifunctional solution for enhancing the properties of biodegradable films [120]. Their ability to provide color, antioxidant benefits, and antimicrobial action positions them as valuable additives in the pursuit of eco-friendly and high-performance packaging materials, being a promising alternative for the production and application of betacyanins in the food industry.
Betacyanins can offer a natural and sustainable alternative for the cosmetic industry. The unique properties of betacyanins, including their antioxidant effects, make them a valuable addition to cosmetic formulations [121, 122]. The vibrant red to violet color can improve the development of a spectrum of shades in lipsticks, blushes, and eyeshadows. Betacyanins, as a natural pigment for the cosmetic industry, can align with the trends in the growing consumer preference for clean, plant-based ingredients in cosmetics [123, 124]. Betacyanins offer potential antioxidant benefits for the cosmetic products [11]. Studies suggested that these natural pigments may help neutralize free radicals, contributing to the prevention of oxidative stress and promoting health benefits [5, 125]. Incorporating betacyanins into cosmetic formulations aligns with the industry’s pursuit of ingredients that offer both cosmetic and therapeutic properties, contributing directly to skin aging and eye degenerative diseases [126]. However, the effective industrial production of cosmetic products based on betacyanins is scarce and should be better developed to consolidate the application of betacyanins extracted from natural resources.
Betacyanins have been extensively studied for their beneficial health properties and are gaining more exposure as nutraceuticals and in the pharmaceutical industry [127]. Betacyanins exhibit strong antioxidant properties through their radical scavenging activities, which are important to combat oxidative stress-related disorders such as cardiovascular diseases, diabetes and neurodegenerative disorders [127]. They are also reported to have anti-obesity and metabolic-regulating effects. For instance, the consumption of betacyanin-rich formulation (200 mg/kg/day for 14 days) improved lipid profiles, insulin sensitivity, body weight gain, hepatic steatosis, and reduced adipose tissue size, making them suitable candidates as nutraceutical products for targeting metabolic syndrome [128].
The anti-inflammatory activities of betacyanins are also well-documented. Highlighting their effects in modulating inflammatory pathways and reducing the production of pro-inflammatory cytokines [IL-6, tumor necrosis factor alpha (TNF-α), IL-1β, or nuclear factor-kappa B (NF-κB)] and enzymes such as cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) in various biological models [129]. These anti-inflammatory properties are beneficial to manage chronic inflammatory disorders such as arthritis, inflammatory bowel disease, and metabolic syndrome, and have been demonstrated to have multi-organ protection, such as heart, liver, kidney, gut, lungs, and neuroprotection [130]. Additionally, numerous in vivo and in vitro studies have demonstrated the anticancer activities of betacyanins, mediated through mechanisms such as apoptosis induction, cell cycle arrest, and inhibition of cell proliferation and angiogenesis [4]. Overall, the ability to modulate oxidative stress, inflammation, and cancer-related pathways positions betacyanins as attractive and strong candidates as adjuncts or lead compounds in drug or nutraceutical development.
Betacyanins present a promising potential for industrial application [131]. The application of betacyanin as a natural food colorant and food packaging is well-established, offering a safe and sustainable alternative to synthetic dyes and as a smart material to indicate the food is fresh during storage [132]. Another potential application of betacyanin is in fluorescent microscopy, which is being marketed as a natural histology stain replacing dyes like eosin [56]. However, the large-scale application of betacyanin in the cosmetic and pharmaceutical industry should be better explored in the future. Extracts containing betacyanins have garnered attention for their vibrant hues, making them a desirable choice in the food industry. Recent studies demonstrated the extraction of betacyanins from plant sources. The most promising, sustainable, and modern extraction techniques of betacyanins from plant sources are UAE, PEF extraction, pressurized liquid extraction, and subcritical water extraction [133]. However, efforts are necessary to develop new extraction methodologies that not only enhance the yield of betacyanins but also minimize the need for extensive downstream purification, which might be a cost-effective operation for the industrial application of betacyanins.
The development of cost-effective extraction and downstream purification processes aims to optimize the overall process, reducing the economic costs associated with purification steps [134]. For instance, the application of a process based on simultaneous extraction, in-line purification, and on-line analysis could be an alternative to obtain real-time information and a high yield and purity of betacyanins [135, 136]. Strieder et al. [137] described that coupling of advanced analytical techniques can be used to efficiently extract, separate, and analyze fractions of bioactive compounds, supplying an integrated method to produce high-added value ingredients for several industrial applications. In addition, emerging technologies are also increasingly applied, such as the use of nanoencapsulation of various bioactive compounds, extracting emulsions using SFE, to develop functional foods and nutraceuticals. This method aims to produce bioactive compound-loaded nanocapsules with higher encapsulation efficiency, smaller size, and reduced organic residual solvents [138].
Therefore, to ensure the industrial application of purified betacyanins, these innovative extraction and purification processes are crucial for eliminating impurities and refining the raw plant extracts into a purified extract suitable for industrial application [139]. In addition, the economic constraint has prompted a need for innovative approaches to facilitate the effective industrial utilization of betacyanins. The purification process should balance between cost-effectiveness and maintaining the integrity of betacyanin-rich extracts. Finally, betacyanins exhibit immense potential for industrial applications. However, future studies should address the cost of downstream purification processes for the effective industrial application of betacyanin-rich extracts, ensuring the profitable and sustainable industrial application of betacyanins in the food, pharmaceutical, and cosmetic industries.
Bioavailability studies are important to measure the potential bioactive effects of phytocompounds in living subjects; this can be further translated into humans for health assessments and applications [140]. Currently, the mechanisms of how betacyanins are absorbed, metabolized, and excreted are yet to be fully described. Betacyanins begin to degrade immediately upon ingestion in the stomach. A study administering betacyanin solution into Wistar rat stomachs demonstrated that while continuous exposure to gastric secretions degrades these pigments, a portion is still absorbed directly through the gastric wall. The dose and exposure time significantly influence the rate of degradation and the intensity of absorption [141]. This is supported by earlier work calculating that 24–29% of betalain pigments are degraded in the stomach [142], a figure consistent with in vitro models showing a 21–35% loss in the gastric phase [143, 144].
The intestinal phase is where the most significant degradation occurs, drastically reducing bioaccessibility. Bioaccessibility refers to the proportion of a compound that is released from the food matrix in the gastrointestinal tract and becomes available for intestinal absorption. In vitro models show losses of 46–55% in the intestinal phase [143, 144], leaving only 29–38% of ingested betalains remaining [145]. The food matrix plays a critical role; liquid matrices like juices show better intestinal absorption and bioavailability than solid (fiber-rich) foods [146]. This is due to several factors. For example, solid foods must first undergo mechanical and enzymatic breakdown before pigments can be solubilized. These extra steps tend to delay release so that molecules face more extensive intestinal/microbial degradation before release. Pigments in liquid matrices are already solubilized, less trapped by fiber, and more exposed to the absorptive environment [145]. Fermentation can increase bioaccessibility, as shown by fermented red dragon fruit juice having higher betanin levels (46.22%) than raw juice (43.76%) after digestion [26]. This is because the fermentation process partially breaks down the plant cell walls, increasing the release of betacyanin pigments into the solution phase and increasing the bioaccessible fraction during digestion.
Despite significant digestion, betacyanins and their metabolites are absorbed into the systemic circulation. They have been detected in human and animal blood plasma and urine, both in their native form [147, 148] and as deglucosylated, decarboxylated, and dehydrogenated metabolites [149, 150]. Absorption occurs via paracellular transport and can be hindered by specific gut transporters like multidrug resistance-associated protein 2 (MRP2) and competition with glucose transporters due to its glycosylated structure [151, 152]. The liver contributes to metabolism, with first-pass extraction reducing systemic concentrations by approximately 23% [141].
Elimination occurs primarily through renal and fecal excretion, with overall bioavailability being very low. Human studies show a renal recovery of only 0.13% and rapid elimination [145]. Fecal excretion shows a higher degree of decomposition, indicating extensive fragmentation and interactions with gut microbiota [145]. A strong positive correlation between urinary and fecal metabolite profiles underscores the gut’s profound impact on systemic bioavailability and is a key source of inter-individual variability. Excretion rates are also influenced by the food matrix, with betacyanins from beet juice being excreted faster than those from solid beet slices [150].
Several critical mechanisms remain unresolved. The specific role of the liver is not fully explained; for example, biliary excretion of betacyanins appears limited [153–155]. This suggests that the pigments may be rapidly metabolized into uncharacterized or polar secondary compounds that are not detected by current analytical methods or that the presence of alternative hepatic clearance pathways. The precise nature of the interaction with gut microbiota and the identity of all resulting metabolites require further elucidation [141, 145]. As mentioned earlier, decarboxylation and dehydrogenation reactions convert betacyanins into multiple decarboxylated and oxidized pigments (e.g., C‑2/C‑17 decarboxylates, 2,15‑bidecarboxy forms, neobetanin derivatives) [27], but there is a dearth of data on their direct bioactivity. Other similar types of pigments show that metabolites have some activity. Decarboxylated gomphrenin-type metabolites, a subset of betacyanins, have been shown to have strong anti-inflammatory effects in vitro, modulating cytokine release and immune cell activity more effectively than native or parent molecules [156, 157]. Hence, a major future direction is to characterize these unknown metabolites to fully understand the pharmacokinetic pathway and the compounds responsible for the observed health benefits.
The absorption and bioavailability of betacyanins are affected by a complex interplay of numerous factors. These include dosage, gastric conditions, metabolic processes in different organs, the presence of aglycones, specific transport mechanisms (e.g., MRP2), biotransformation pathways, and excretion processes. Crucially, the food matrix (liquid vs. solid, fermented vs. raw) and the structural form of the betacyanin (e.g., glycosylated) are significant determinants of its absorption efficiency [26, 150–152]. Ultimately, intestinal uptake and systemic metabolism are closely related to biotransformation by gut microbiota [141, 145].
Together, these studies show that the absorption of betacyanins is affected by numerous factors and potentially is degraded mainly by the gastrointestinal tract. Several factors that influence the absorption of betacyanins have been identified, among them are dosage, gastric conditions, metabolic processes in different organs, the presence of aglycones, transport mechanisms, biotransformation pathways, food matrices, fermentation status, excretion processes, intestinal biotransformation, and gut microbiota interaction. The bioavailability of betacyanins is summarized in Figure 4.
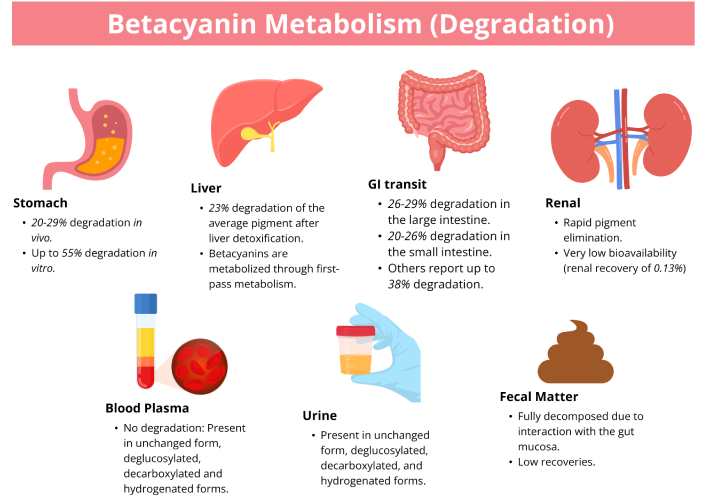
The absorption and excretion of betacyanin pigments in various body systems and excretions. Orally given betacyanins showed low urinary recoveries, suggesting rapid metabolism or enhanced bodily elimination. Betacyanins are highly degraded in the stomach and intestines. Absorption of betacyanins is affected by the food matrix.
Metabolic syndrome is a constellation of conditions, including central obesity, hypertension, type 2 diabetes or impaired glucose metabolism, and dyslipidemia. This syndrome is associated with an increased risk of cardiovascular disease and other complications such as chronic inflammation, chronic kidney disease, and non-alcoholic fatty liver disease [158, 159]. Among natural compounds showing promise for metabolic syndrome management, betacyanins—vibrant plant pigments found in red beetroot, prickly pear, and dragon fruit—have emerged as particularly interesting candidates due to their multi-target biological activities. This section aims to discuss recent reports on the effects of betacyanins on cardiometabolic diseases and proposes their mechanisms of action on both animal and human models.
The findings from preclinical data on betacyanins show consistent demonstration of their dual capacity to combat both oxidative stress and inflammation, two processes intricately linked in metabolic syndrome. Reactive oxygen species (ROS) can activate pro-inflammatory signaling pathways, while inflammatory cells themselves generate further ROS. Betacyanins appear to disrupt this vicious cycle at multiple points. In animal models of diet-induced obesity, streptozotocin-induced diabetes, and diclofenac (DF)-induced acute liver injury model, administration of betanin (the most studied betacyanin) at doses of 10–40 mg/kg consistently acts as a direct redox regulator. It significantly reduces the concentrations of toxic lipid peroxidation end-products like malondialdehyde (MDA) and protein carbonyls (PC), key markers of oxidative damage to cellular structures [160–162].
Apart from acting as free-radical scavengers, betacyanins also enhance the body’s endogenous antioxidant defense system. Studies report a significant upregulation in the activity and gene expression of fundamental antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) [160–163]. The master regulator of this coordinated defense is the transcription factor nuclear factor erythroid 2-related factor 2 (Nrf2) [164]. Research by Mousavi et al. [161] and Villa-Jaimes et al. [162] demonstrates that betacyanins activate the Nrf2 pathway, leading to the increased transcription of a battery of cytoprotective genes, including hepatic enzymes. This Nrf2 activation represents a sophisticated, indirect mechanism by which betacyanins boost the cell’s long-term resilience to oxidative attack.
The reduction in oxidative stress translates into the corresponding anti-inflammatory effects. A reduced oxidative load inherently dampens the activation of redox-sensitive inflammatory pathways. The most critical of these is the NF-κB pathway, a primary regulator of the inflammatory response. Abedimanesh et al. [163] showed that betanin treatment in diabetic rats directly reduced the mRNA expression of NF-κB in liver and blood tissues. Further qPCR analysis increased the expression level of adenosine monophosphate-activated protein kinase (AMPK) and Sirtuin-1 (SIRT1) in blood and liver [163]. Another study also noted a decrease in inflammatory markers (IL-6, IL-1β, and TNF-α) while increasing anti-inflammatory markers (IL-10) in diet-induced obese mice [128]. These findings showed that betanin can reduce inflammation and oxidative stress symptoms associated with diabetes and may produce the same effects in humans.
This molecular finding is corroborated by functional outcomes. In vitro, betanin demonstrated a marked ability to inhibit the expression of key NF-κB-driven pro-inflammatory mediators in macrophage cells, including cytokines like IL-6 and IL-1β, as well as enzymes like iNOS and COX-2 [165]. Simultaneously, it promoted the expression of the anti-inflammatory cytokine IL-10. In vivo, these mechanisms manifest as reduced hepatic inflammation and pancreatic lesions, as observed in histological analyses [128, 163]. The study by Fernando et al. [165] further illustrates the synergy between antioxidant and anti-inflammatory actions by showing that betanin’s radical-scavenging capacity counteracted hydrogen peroxide-induced ROS generation, thereby proactively suppressing the trigger for inflammation.
The convergence of these antioxidant and anti-inflammatory mechanisms may explain the significant improvements seen in key organs associated with metabolic syndrome pathogenesis. Betacyanins demonstrate remarkable hepatoprotective effects across multiple models. In obese Wistar rats, betanin administration reversed hepatic tissue damage, evidenced by reduced aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels, decreased lipid accumulation, and improved liver structure [160]. Similarly, in DF-induced acute liver injury, Opuntia robusta fruit extract rich in betacyanins reduced oxidative biomarkers while restoring glutathione levels and activating the Nrf2 pathway [162]. The administration of betacyanins from red and white pitaya also improved liver steatosis in high-fat diet-induced obesity in mice [8, 128]. These findings suggest betacyanins protect against both metabolic and toxic liver injury through complementary antioxidant and anti-inflammatory mechanisms.
Human studies provide promising evidence for cardiovascular benefits. A pilot randomized crossover trial by Rahimi et al. [166] demonstrated that two-week supplementation with betalain and betacyanin-rich (50 mg betacyanins/betalains daily) extracts (O. stricta and red beetroot) improved several cardiovascular markers in male patients, with clinically meaningful reductions in homocysteine, LDL, and non-HDL cholesterol concentrations. Additionally, Cheok et al. [167] reported that red dragon fruit powder (24 g) consumption (providing 33 mg betalains daily for 14 days) improved endothelial function and arterial stiffness in healthy adults, though it did not significantly reduce blood pressure as shown by Rahimi et al. [166]. These findings suggest betacyanins may primarily benefit vascular function and atherogenic lipid profiles. However, the short intervention period, small sample size, and specific age group limit the application of this study to the wider population. Hence, further studies with a longer intervention period, a wider age group, and a larger sample size are suggested to increase the validity of this study.
The renal protective effects of betacyanins are particularly relevant given the high risk of diabetic nephropathy in metabolic syndrome. Sutariya and Saraf [168] demonstrated that betanin treatment from Opuntia elatior (50–100 mg/kg/day) in diabetic rats inhibited proteinuria, reduced urine output, reduced blood glucose concentration, and restored antioxidant enzyme activities in kidney tissue. Importantly, betanin modulated the expression of genes involved in epithelial-mesenchymal transition [transforming growth factor beta (TGF-β), type IV collagen, alpha-smooth muscle actin (α-SMA), E-cadherin] which are the markers of myofibroblasts, and cell-cell adhesion molecule, suggesting it can counteract diabetic nephropathy progression by inhibiting the TGF-β signaling pathway and increasing antioxidant defense systems [168].
Betacyanins demonstrate significant effects on glucose homeostasis and insulin sensitivity, one of the major defects in metabolic syndrome. In streptozotocin-induced diabetic rats, betanin treatment (10–40 mg/kg) produced comprehensive anti-diabetic effects: reduced fasting blood glucose, improved glucose tolerance, and increased serum insulin levels [163]. Histological analyses revealed pancreatic regeneration, suggesting potential for β-cell protection and restoration [163]. The mechanisms underlying these improvements extend beyond antioxidant and anti-inflammatory effects to include modulation of key metabolic sensors. The upregulation of AMPK and SIRT1 [163] is particularly significant, as these enzymes enhance insulin sensitivity, promote glucose uptake in muscles, and improve mitochondrial function. Additionally, betacyanins decrease fibroblast growth factor 21 (FGF21) resistance, further contributing to improved metabolic regulation [8]. These multi-target effects on glucose metabolism make betacyanins particularly promising for addressing the complex pathophysiology of insulin resistance in metabolic syndrome.
Additionally, betacyanins demonstrate significant effects on adipose tissue biology and lipid metabolism. In obesity models, betacyanin supplementation reduced body weight gain, visceral adipose tissue size, and serum lipid levels [8, 128]. In a study published by Khoo et al. [27], the addition of red pitaya extracts containing betacyanins and anthocyanins for 48 h effectively reduced the H2O2-induced oxidative stress in 3T3-L1 cells. Additionally, the peel extract showed better antioxidant properties than the pulp due to the higher concentration of betacyanin and anthocyanin in the peel. The peel extract also showed better lipid reduction effects on 3T3-L1 cells compared to the pulp extract [27]. However, as both betacyanins and anthocyanins were reported in this study, it is difficult to elucidate the exact effects of these two phytochemicals, as both may have synergistic effects on cellular physiology. Thus, further studies should aim to assess the effects of combined phytochemicals, e.g., betacyanins and anthocyanins starting from in vitro to in vivo studies.
Mechanistically, in vitro betacyanin treatment in 3T3L adipocytes inhibits adipogenesis through downregulation of key transcriptional regulators, peroxisome proliferator-activated receptor gamma (PPARγ), CCAAT/enhancer-binding protein alpha (C/EBPα), and sterol regulatory element-binding protein 1c (SREBP-1c), and reduction in PPARγ protein expression involved in adipogenesis [169]. While simultaneously promoting fatty acid oxidation in vivo, through increased expression of lipid metabolism-related genes (AdipoR2, Cpt1a, Cpt1b, Acox1, PPARγ, Insig1, and Insig2) [8]. These dual actions on lipid storage and utilization make betacyanins particularly promising for addressing adipose tissue dysfunction in metabolic syndrome.
Recent evidence suggests betacyanin’s metabolic benefits may be partially mediated through gut microbiota modulation. The consumption of red beetroot juice containing betacyanins in humans correlated with increased beneficial bacteria, including Akkermansia muciniphila, Bifidobacterium, and Coprococcus, while decreasing less desirable species [170]. This microbial shift was associated with increased production of short-chain fatty acids (SCFAs), particularly butyric and isobutyric acid, with specific betacyanin metabolites showing strong correlations with SCFA production [170]. The results showed the ability of betacyanin-containing juice to increase beneficial gut bacteria populations. For instance, the increase in Akkermansia population has been correlated with a healthy diet and decreased risk of obesity and other metabolic disorders [170]. Although the increase of E. coli is thought to be harmful, a recent report showed that commensal E. coli may be beneficial for iron transport [171]. Highlighting the influence of strain specificity of this bacterium and its functions in the human gut [172].
In an animal study, supplementation of 200 mg/kg of betacyanins for 14 days also improved (Firmicutes:Bacteroides) F/B ratio and increased Anaerotruncus, Mucispirillum, and Akkermansia populations at the genus level [128]. An increase in Akkermansia population was also seen after supplementation of Davidson’s plums in Wistar rats’ diet, which is associated with better metabolic and physiological parameters after being given a high-carbohydrate, high-fat diet [173]. The consistent increase in Akkermansia muciniphila across studies [128, 170] is particularly noteworthy given its association with improved metabolic parameters. The ability of betacyanins to modulate the gut-microbiota-liver axis represents a novel mechanism through which they may influence systemic metabolism in metabolic syndrome.
Overall, betacyanins were shown to have beneficial effects against cardiometabolic symptoms in various models (Table 3). Their effects are mediated through several mechanisms such as antioxidant [27, 160–162, 165], anti-inflammatory [163, 165], hepatoprotective [160, 162, 163], pancreatoprotective [163], renal-protective [168], antidiabetic [8, 128, 160, 163], antilipidemic [8, 128, 160, 163], antiobesity [8, 27, 128] and cardioprotective [166, 167] mechanisms, potentially through modulation of gut microbiota population [128, 170] with correlative changes at gene expression levels (Figure 5).
Betacyanins and their effects on cardiometabolic diseases.
| Source | Model: in vitro/in vivo | Dosage/Duration of treatment | Biological effects and mechanisms of action | References |
|---|---|---|---|---|
| Betacyanin (red beetroot, Beta vulgaris) | Human subjects | 3 days, 14 days114.5 mg/day betacyanins, 15.6 mg/day polyphenols, nitrate 228.5 mg/day | ↑ Akkermansia muciniphila, Escherichia coli↓ Bacteroides fragilisPositive correlations of betacyanins and Bifidobacterium and CoprococcusmInverse correlation of betacyanins with Ruminococcus and Peptoniphilus↑ SCFAs (butyric and isobutyric)Total excreted amount of betacyanins positively correlated with contents of (iso)butyric acid, (iso)valeric acid and total SCFAs; (iso)butyric acid and total SCFA contents showed a positive relationship with genera Blautia, Collinsella, Lachnobacterium, and Lactobacillus, but inverse association with Ruminococcus | Wang et al. [170], 2023 |
| Betanin (fresh beetroot juice, Beta vulgaris) | In vivoWistar rats, high-fat food | 20 mg/kg20 days | ↓ Blood glucose levels, insulin and insulin resistance↓ Liver damage (AST and ALT levels) and hepatic lipid accumulation↓ Oxidative stress [decreased hepatic MDA, increased superoxidase dismutase, catalase (CAT) and glutathione peroxidase (GPx) activities]↓ Plasma triglycerides | da Silva et al. [160], 2019 |
| Betanin (pure compound) | In vivoStreptozotocin-induced diabetic animalSprague-Dawley rats | 10, 20 and 40 mg/kg body weight28 days | ↓ Fasting blood glucose concentration↑ Serum insulin levelImproved glucose tolerance testImproved lipid profile and liver enzymes concentrationsImproved liver and pancreas histology↑ AMPK and SIRT1↓ NF-κB mRNA expressions | Abedimanesh et al. [163], 2021 |
| Betanin (pure compound) | In vivoStreptozotocin-induced diabetic animalSprague-Dawley rats | 10, 20 and 40 mg/kg body weight28 days | ↑ Total antioxidant capacity level, protein carbonyl and malondialdehyde (MDA)↑ Activity of hepatic antioxidant enzymes, superoxide dismutase (SOD), CAT, and GPx↑ Expression of Nrf2, SOD, GPx, and CAT genes | Mousavi et al. [161], 2022 |
| Betanin and neobetanin (red beetroot and yellow prickly pear) | In vitroRAW 264.7 macrophages | 10, 50, 100 μM | ↓ Inflammatory cytokines mRNA expressions (IL-6, IL-1β, iNOS, and COX-2)↓ ROS formation↓ NOX-2 mRNA levels↑ HO-1 and γGCS mRNA expression↑ IL-10 mRNA expression | Fernando et al. [165], 2023 |
| Hylocereus undatus peel (white pitaya) | In vivoC57BL/6 mice | 50, 100 and 200 mg/kg14 weeks | ↓ High-fat diet induced body weight gain↓ Adipose tissue hypertrophy↓ Hepatosteatosis, insulin resistanceImproved glucose tolerance↑ Expression of lipid metabolism related genes (AdipoR2, Cpt1a, Cpt1b, Acox1, PPARγ, Insig1, and Insig2)↑ Expression of FGF21-related genes (β-Klotho and FGFR1/2)Effects might be due to improved fatty acid oxidation, ↓ fatty acid biosynthesis, ↓ FGF21 resistance | Song et al. [8], 2016 |
| Hylocereus polyrhizus (red pitaya) | In vivoC57BL/6 mice | 200 mg/kg14 weeks | ↓ High-fat diet induced body weight gain↓ Visceral adipose tissue weight↓ Serum levels of blood lipids↓ Hepatic steatosis↓ Size of both white and brown adipocyte↓ Glucose intolerance and insulin resistance↓ Inflammatory cytokines (IL6, IL-1β, TNF-α) but ↑ anti-inflammatory cytokine (IL-10)↓ Firmicutes proportion↑ Bacteriodetes and Proteobacteria proportions | Song et al. [128], 2016 |
| Hylocereus polyrhizus cv. Jindu (betacyanin and anthocyanins) | In vitro3T3-L1 cells | 0.0625–1.0 mg/mL24 and 48 h | Both peel and pulp extract had similar total antioxidant activities against H2O2-induced oxidative stress on 3T3-L1 cell linePeel extract had inhibitory effects on 3T3-L1 cell lipid accumulation | Khoo et al. [27], 2022 |
| Betanin | In vitro3T3-L1 cells | 10–50 μM of betanin for 72 h (lipid accumulation)24 h for protein and 6 h for gene expressions | ↓ Number of lipid dropletsBetanin levels at 10, 25 and 50 μM ↓ lipid accumulation by 19.6%, 26.1% and 32.6%, respectivelyBetanin levels at 10, 25 and 50 μM ↓ intracellular triglyceride levels in 3T3-L1 adipocytes by 10.5%, 16.6% and 18.1%Suppressed expression of adipogenic genes PPARγ, C/EBPα and SREBP-1c↓ PPARγ protein expression | Chen et al. [169], 2019 |
| Hylocereus polyrhizus (betalains including betacyanins) | Human subjects (healthy men and women) | Double-blind, randomized controlled crossover trial24 g of dragon fruit powder containing 33 mg of betalains, 14 days | Both acute and sustained consumption of dragon fruit improved flow-mediated dilationAcute consumption ↓ pulse-wave velocity but not sustained consumption↓ Heart-rate augmentation index after 14 daysNo significant changes in blood pressure (central systolic and peripheral diastolic)Only two metabolites were significantly increased in the plasma after consumption of dragon fruit powder | Cheok et al. [167], 2022 |
| Opuntia elatior Mill (red flower prickly pear)Betanin | In vivoSprague-Dawley rats (STZ-induced) | 25, 50, and 100 mg/kg/day8 weeks | Inhibiting proteinuria↓ Blood glucose and nephrotoxicity markers (serum creatinine and BUN levels)Restored antioxidant enzymes activities in kidney tissues (↓ TBARS, ↑ SOD, CAT)↓ Glomerular surface area, glomerulosclerosis, tubulointerstitial fibrosisModulated mRNA and protein expression of TGF-β, type IV collagen, α-SMA and E-cadherin | Sutariya and Saraf [168], 2017 |
| Opuntia robustaBetacyanin/Betanin | In vivoWistar rats [diclofenac (DF)-induced acute liver injury model] | Opuntia robusta (OR) fruit extract (800 mg/kg/5 days, orally) (4.45 mg of betanin equivalents)Betanin (25 mg/kg/5 days, orally) | Pretreatment with OR extract and betanin ↓ MDA levels and maintained GSH levelOR extract increased Cat, Sod1 expressionBetanin ↑ Cat expressionOR extract or betanin only ↓ Sod2 expression but DF treatment ↑ Sod2 expression with corresponding treatmentsOR or betanin ↑ Nrf2 gene expressionOR or betanin ↓ Hmox1, Gclc expressionsOR or betanin ↓ active caspase-3-positive cells | Villa-Jaimes et al. [162], 2023 |
| Opuntia stricta (Prickly pear cactus)Betalain/Betacyanin | Human subjects (men with coronary artery disease)Randomised, double-blind, crossover trial: 2 weeks each (total duration 10 weeks) | 50 mg betacyanin/betalains per day (blood collection 12 h post fasting, 0, 14, 28, 42, 56 and 70 days) | ↓ Homocysteine level↓ Systolic blood pressure↓ Fasting blood glucose↓ Non-HDL cholesterol and LDL cholesterol | Rahimi et al. [166], 2019 |
↑: increase; ↓: decrease; CAT: catalase; COX-2: cyclooxygenase-2; iNOS: inducible nitric oxide synthase; NF-κB: nuclear factor-kappa B; Nrf2: nuclear factor erythroid 2-related factor 2; ROS: reactive oxygen species; SCFAs: short-chain fatty acids; SIRT1: Sirtuin-1; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BUN: blood urea nitrogen; C/EBPα: CCAAT/enhancer-binding protein alpha; FGF21: fibroblast growth factor 21; FGFR1/2: FGF receptor 1/2; GSH: reduced glutathione; HO-1: heme oxygenase 1; PPARγ: peroxisome proliferator-activated receptor gamma; SREBP-1c: sterol regulatory element-binding protein 1c; TGF-β: transforming growth factor beta; TNF-α: tumor necrosis factor alpha; α-SMA: alpha-smooth muscle actin.
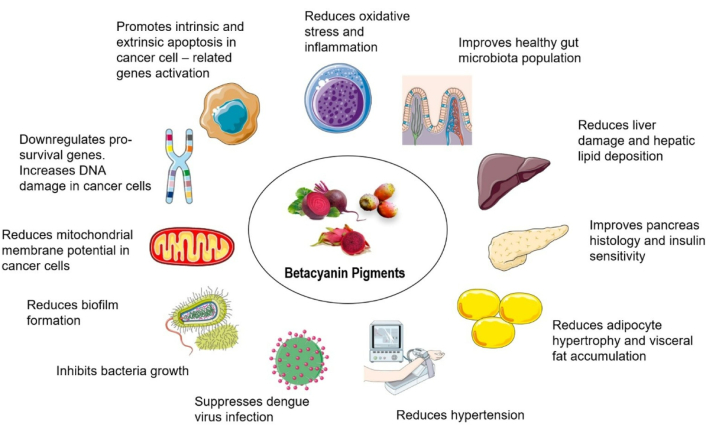
The potential health-promoting effects of betacyanin pigments in cardiometabolic disorders, cancer, and as antimicrobial agents. Betacyanin acts on these disorders through various mechanisms, including physiological, biochemical, and gene alterations.
Despite promising evidence, several limitations must be acknowledged. First, effective animal doses (10–100 mg/kg) may not be easily achieved through human dietary intake. Second, human studies remain limited and often use combination extracts, making it difficult to attribute effects solely to betacyanins. The bioavailability issue is particularly important, as human studies show minimal increases in plasma polyphenol levels despite functional benefits [167], suggesting metabolites rather than parent compounds may mediate effects.
Future research should focus on well-designed human randomized controlled trials in populations using standardized betacyanin preparations. Research identifying the most bioactive metabolites and understanding how inter-individual variability in gut microbiota affects response to betacyanin intervention will be crucial for developing personalized nutrition approaches. Additionally, more studies are needed to elucidate potential synergistic effects between betacyanins and other phytochemicals, as well as their interactions with conventional pharmacological treatments for metabolic syndrome.
However, translation to clinical practice requires addressing significant challenges, including bioavailability issues, standardization of effective doses, and understanding individual variability in response. While betacyanin-rich foods can be recommended as part of a healthy diet for metabolic syndrome management, their specific therapeutic application requires further rigorous clinical investigation to establish efficacy, optimal dosing, and potential for personalized nutrition approaches based on individual metabolic and microbial profiles.
Numerous studies were conducted over the years to investigate the anticancer properties of betacyanins. Betacyanin-rich plants such as B. vulgaris (red beetroot), Basella rubra (Malabar spinach), Amaranth cruentus (sprouts), Opuntia ficus-indica (cactus pear), and H. polyrhizus (red pitaya) demonstrated anticancer potentials against various human cancer cell lines such as HepG2, HeLa, MCF-7, and Caco-2, elucidating several fundamental mechanisms through which betacyanins exert their anticancer effects.
Sreekanth et al. [174] (2007) investigated the anticancer potential of betanin isolated from cactus pear. There was a 50% decrease in K562 cell proliferation after treatment with 40 µM betanin for 24 h. Betanin caused apoptotic cell death, as morphological and structural changes such as a condensed nucleus and membrane blebbing were observed in K562 cells. DNA fragmentation in K562 cells was detected in a progressively increasing manner after treatment with betanin at different concentrations for 24 h [174]. This corresponded to inter-nucleosomal cleavage, which is one of the characteristics of apoptosis. Furthermore, decreased mitochondrial membrane potential was also observed with leakage of cytochrome c into the cytosolic fractions of the K562 cells after treatment with 40 µM betanin at different time points [174], showing the effect of betanin in intrinsic mitochondrial apoptosis.
The anticancer potentials of Pachycereus weberi (Chico) and Escontria chiotilla (Jiotilla) showed that both Chico and Jiotilla possessed high phytochemical contents of betalains and other phenolic compounds. Chico fruit juice was able to inhibit the growth of Caco-2 and MCF-7 cell lines, whereas Jiotilla fruit extract decreased the cell viability of HepG2 and PC-3 cell lines [175]. Extracted betanin/isobetanin concentrate from fresh beetroot was found to upregulate apoptosis-related proteins, in MCF-7 cells treated with 30 µM betanin/isobetanin concentrate for 24 h, such as B-cell lymphoma 2 (Bcl-2)-associated agonist of cell death (Bad), TNF-related apoptosis-inducing ligand receptor 4 (TRAILR4), FAS, and phosphorylated p53, besides altering the mitochondrial membrane. Hence, it indicated involvement of this extract in both mitochondrial and death-receptor pathways. Furthermore, autophagosome vesicles were observed in the MCF-7 cells, suggesting that there was autophagic cell death as well when cells were treated with betanin/isobetanin [176]. This effect was also demonstrated in T21 bladder cancer cells, where a combination of purified betacyanin and vitexin-2-O-xyloside (XVX) from B. vulgaris var. cicla L. inhibits the proliferation of T24 cancer cells at 24 h and 48 h of incubation. Apoptosis was induced through activation of caspase 8 activity [177].
The anticancer properties of the beetroot hydro-alcoholic extract (BHE) of the red beetroots (B. vulgaris) were investigated previously [178]. Both BHE and its constituent betanin inhibited the growth of Caco-2 and HT-29 cell lines in a time and dose-dependent manner. The IC50 values of betanin were 64 μg/mL (HT-29), 90 μg/mL (Caco-2), and the IC50 values of BHE were 92 μg/mL (HT-29), 107 μg/mL (Caco-2) after 48 h treatment. Apoptosis was confirmed by 4’,6-diamidino-2-phenylindole (DAPI) staining, where both cell lines showed apoptotic characteristics. Flow cytometry indicated an increase in the proportion of both Caco-2 and HT-29 cells in early and late apoptosis after treatment with BHE and betanin for 48 h. This pro-apoptotic effect was further supported by the upregulation of genes including Bad, Fas-R, caspases-3, -8, and -9. Notably, the expression level of the Bad gene was found to be higher than the positive control group, 5-fluorouracil (5-FU), after both BHE and betanin treatment. Conversely, BHE treatment downregulated the expression of anti-apoptotic genes such as Bcl-2 [178]. Looking at an alternative view, as Polygonum minus phytochemicals enhance 5-FU efficacy, betacyanins could similarly synergize with 5-FU by targeting complementary pathways [179]. Additionally, given that combinations of phytochemicals have been shown to enhance colorectal cancer treatment efficacy, future research could explore the co-administration of betacyanins with complementary agents such as isothiocyanates and carotenoids or drugs to modulate intersecting signaling pathways [e.g., phosphoinositide 3-kinase (PI3K)-Akt and NF-κB] [180].
Chenopodium formosanum (djulis) was investigated for its anticancer effects on hepatoma cells, both in vitro and in vivo. Betanin was one of the bioactive compounds identified in the extract using high-performance liquid chromatography (HPLC)/electrospray ionization (ESI)-MS analysis, and it was selected to undergo further evaluation. Betanin was able to induce apoptosis in HepG2 cells by increasing apoptotic bodies, decreasing mitochondrial membrane potential, enhancing the ratio of Bcl-2-associated X protein (Bax)/Bcl-2, and activating caspase-3 and cleaved Poly (ADP-ribose) polymerase (PARP) levels. At the concentrations between 50 and 200 μM, betanin increased ROS generation in the HepG2 cells, which may contribute to the cellular antiproliferation due to oxidative stress and eventually apoptosis [181].
In parallel, nanotechnology approaches have leveraged betacyanins for enhanced efficacy using betacyanin-modified selenium nanoparticles for anticancer treatment [182]. The presence of betacyanin on the surface of these nanoparticles enhanced their anticancer properties compared to the non-modified selenium nanoparticles. These modified nanoparticles inhibited the growth of HepG2 cell lines in a dose-dependent manner [182]. Another research group synthesized Ag nanoparticle-integrated ZnO nanoflakes (ZnO/Ag nanocomposite) using red beetroot juice. The HeLa and SKOV-3 cell lines showed a significant decrease in viability after treatment with ZnO/Ag nanocomposites in a dose-dependent manner [183]. There was a high ROS generation in both HeLa and SKOV-3 cells after treatment with the nanocomposites at 2.0 mg/mL, leading to oxidative stress and ultimately cell death. In addition to this, a cell migration assay was performed, which reported a significant reduction in migration for both cell lines after 48 h incubation with the nanocomposites [183].
The anticancer effect of betanin towards human osteosarcoma (MG-63) cells was recently reported [184]. This phytocompound induced apoptosis and inhibited migration as well as adhesion of MG-63 cells in a dose-dependent manner. Morphological changes were also observed, such as DNA strand breaks and a large nucleoid appearance. Besides, apoptosis was also triggered by betanin as ROS generation was induced and subsequently decreased the mitochondrial membrane potential. This study also revealed that betanin inhibited osteosarcoma via the PI3K/Akt/mechanistic target of rapamycin (mTOR) signaling pathway [184], indicating betacyanin’s effects in modulating the oncogenic signaling pathway.
Basella rubra, containing betalain, demonstrated good antioxidant activity besides exhibiting cytotoxicity towards human cervical cancer cells (SiHa). Cellular viability of SiHa cells decreased significantly by approximately 80% after treatment with 50 mg/mL of the fruit extract for 24 h [185]. The treated cells showed morphological changes, such as cell shrinkage, blebbing, and detachment of the cells from the substratum, exhibiting classical features of apoptotic cell death. These changes became visible after 24 h of extract treatment but were absent in control cells, and the changes became more remarkable with the increasing extract concentrations. A dose-dependent decrease in growth kinetics was observed [185].
It was reported that the red pitaya peel extract possessed higher total betacyanin content (35.12 ± 0.01 mg/g sample) than the pulp extract (30.15 ± 0.03 mg/g sample) [27]. The phytochemical results also revealed that the red pitaya peel extract had a higher betanin content (9.44 ± 0.01 mg/g sample) than the pulp extract (7.44 ± 0.03 mg/g sample). While both extracts showed very low cancer-killing potential, IC50 against liver cancer cells (HepG2), the peel was slightly more effective, likely due to its higher content of these compounds. The study found that pure betanin was much more potent at killing cancer cells than either the pulp or peel extracts, as evidenced by the lower IC50 value [27].
Wang and Wang [186] (2023) fermented red beetroot juices using water kefir grains, and it was found that the fermentation process increased the betacyanin content in the juices. The juices were then tested on various cancer cell lines, such as HepG2 cells. Results clearly indicated that the red beetroot juice possessed stronger cellular inhibitory potentials after fermentation by water kefir grains, as the inhibition rate of the fermented red beetroot juice increased by 32.08% after 24 h of fermentation as compared with the unfermented red beetroot juice [186]. The fermented juices exerted growth-inhibitory effects towards HepG2 cells by increasing cellular ROS production, leading to cell apoptosis [186].
As reported by Kapadia et al. [187] (2011), B. vulgaris extract showed cytotoxicity towards human prostate (PC-3) and breast (MCF-7) cancer cell lines. However, the beetroot extract was less potent than doxorubicin. Its cytotoxic effect was most likely due to its high betacyanin content, specifically betanin [187]. Lee et al. [188] (2014) also reported that betacyanins like betanin and betaine decreased the HepG2 cell viability in a dose-dependent manner, and betanin was shown to be more effective than betaine. A study conducted by Krajka-Kuźniak et al. [189] (2013) reported that the growth of HepG2 cells showed a dose-dependent decrease after betanin treatment, with a decrease of 50% in cell proliferation at the concentration of 200 µM betanin. A notable contradiction exists in colon cancer models, where Caco-2 cells remained viable after 24 h treatment with the highest dose of betanin [190], while others reported significant apoptosis at lower concentrations [178], potentially reflecting differences in experimental conditions or cell characteristics.
In terms of cell cycle arrest, the treatment of betanin in K562 cells was found to have increased hypodiploid apoptotic DNA content and a reduced number of cells in the S and G2 phases, as shown through flow cytometry analysis [174]. Fermented juice containing betacyanins was also shown to arrest the growth of cancer cells at the G1 phase, and the population of cells entering the next cell cycle was also significantly reduced [186].
Based on the studies conducted so far, we conclude that betacyanins, specifically betanin and betalains, indeed possess promising potential as anti-cancer agents since they inhibit growth in various cancer cell lines in vitro. These mechanisms demonstrate consistent dose-dependent and time-dependent responses across multiple cancer cell lines, while revealing important structure-activity relationships and cell-type-specific variations. The findings are summarized in Table 4.
Betacyanins and their anticancer properties.
| Source | Model: in vitro | Dosage/Duration of treatment | Biological effects and mechanisms of action | References |
|---|---|---|---|---|
| Beta vulgaris L. (red beetroot) | Human breast (MCF-7) | Proliferative index (ratio between viable cell counts 48 h post-seeding and number of seeded cells) was used to estimate the cell viability after treatment of betanin/isobetanin concentrate. Cells were treated using concentrations from 10, 20, 30, 40 µM and were incubated for 24 h to 48 h. | Extrinsic and intrinsic apoptotic pathways—morphological changes and DNA fragmentation observed; ↑ expression levels of apoptosis-related proteins such as Bad, TRAILR4, FAS, phosphorylated p53; ↓ mitochondrial membrane potential; lower percentage of cells arrested at G1 phase (for 2D culture), higher percentage of cells in the S phase (for 3D culture); ↑ autophagic activity. | Nowacki et al. [176], 2015 |
| Human bladder (T24) | Sulforhodamine B (SRB) assay was used to measure cell viability. Cells were treated at different concentrations (25, 50, 100 µg/mL), and then incubated for 24, 48, and 72 h. | Extrinsic apoptotic pathway—betacyanin downregulated the pro-survival gene CTNNB1 (β-Catenin), resulting in the reduction of cell proliferation via induction of caspase-8 activity. | Scarpa et al. [177], 2016 | |
| Human colon (Caco-2) | MTT assay was used to evaluate the cytotoxicity of betanin, cells were treated with various concentrations (100, 200, 300, 400, 500 µM) and incubated for 24 h at 37°C. | Intrinsic apoptotic pathway—betanin ↑ DNA damage in a concentration-dependent manner and ↓ the mitochondrial transmembrane potential, as well as induced procaspase-3 cleavage and caspase-3 activity. | Zielińska-Przyjemska et al. [190], 2016 | |
| Human colon (Caco-2, HT-29) | MTT assay was used to test the beetroot hydro-alcoholic extract (BHE) at various concentrations (20, 40, 60, 80, 100, 120, 140 µg/mL) and then incubated for 24 h and 48 h. | Extrinsic and intrinsic apoptotic pathways—with the treatment of BHE, higher percentage of cells found to be undergoing early and late apoptosis stage as morphological changes (DNA fragmentation, cell shrinkage) were observed. After treated with BHE for 48 h, pro-apoptotic genes such as Bad, Fas-R, caspase-3, caspase-8, and caspase-9, were upregulated and the expression levels of anti-apoptotic gene Bcl-2 was greatly reduced. | Saber et al. [178], 2023 | |
| Human liver (HepG2) | MTT assay was used to evaluate the antiproliferative effects of betacyanin-modified selenium nanoparticles (Bc@SeNPs). Cells were treated with various concentrations (10, 20, 30, 40 µg/mL) and incubated for 24, 48, and 72 h. | Extrinsic and intrinsic apoptotic pathway—appearance of cells became round and vacuolated after treated with Bc@SeNPs. Cellular ROS levels ↑ greatly, followed by ↓ in mitochondrial membrane potential, resulting in upregulation of p53 gene, activation of caspase-3 and caspase-9 cleavage, as well as downregulation of Bcl-2 gene. | Tang et al. [182], 2021 | |
| Human liver (HepG2) | Cell Counting Kit-8 (CCK-8) was used to determine the cell viability after being treated and incubated for 24 h with red beetroot juices fermented by water kefir grains. | Apoptotic cell death—fermented beetroot juices found to have induced ROS production, ↑ both early and late apoptosis rates, and arrested cells at G1 phase, thus ↓ the number of cells entering next cell cycle. | Wang and Wang [186], 2023 | |
| Basella rubra L. (Basellaceae) | Human cervical (SiHa) | MTT assay was used to determine the cytotoxic effects of betalains. Different concentrations were used (2.5, 5, 10, 15, 20, 25, 37.5, 50, 62.5, 75, and 100 mg/mL) and then cells were incubated for 24 h. | Apoptotic cell death—morphological changes of the cells were observed after 24 h of treatment, including cell shrinkage, detachment of cells from substratum, and blebbing. | Kumar et al. [185], 2015 |
| Opuntia ficus-indica (cactus pear) | Human chronic myeloid leukemia (K562) | MTT assay was used to assess the cell proliferation and treated with betanin (10, 20, 40 and 80 µM) and then incubated for 24 h. | Extrinsic and intrinsic apoptotic pathways—DNA fragmentation and morphological changes such as blebbing and chromatin condensation were seen after treated with betanin. Higher percentage of cells were at sub G0/G1 phase, leading to ↓ number of cells that enter the S and G2 phase of cell cycle. The mitochondrial membrane potential has also been changed, hence there is an ↑ in the cytosolic levels of cytochrome c which was released by the mitochondria. Poly (ADP-ribose) polymerase (PARP) cleavage is also being induced for apoptotic cell death. | Sreekanth et al. [174], 2007 |
| Chenopodium formosanum (Djulis) | Human liver (HepG2) | MTT assay was used to test for the cell viability, various concentrations of ethanolic extracts (50, 250, 500 µg/mL) were used and the cells were incubated for 24, 48, and 72 h. | Extrinsic and intrinsic apoptotic pathways—after treatment with betanin, morphological changes such as chromatin condensation and nuclear fragmentation were seen, ↑ percentage of cells arrested in the sub-G0 phase, as well as ↑ percentage of cells in early or late apoptosis/necrosis. ROS generation was induced, and mitochondrial membrane potential was significantly ↓, resulting in upregulation of caspase-3 activity and cleavage of PARP protein. | Chu et al. [181], 2020 |
↑: increase; ↓: decrease; ROS: reactive oxygen species; Bcl-2: B-cell lymphoma 2; Bad: Bcl-2-associated agonist of cell death; TRAILR4: TNF-related apoptosis-inducing ligand receptor 4.
They were found to be effective against human breast (MCF-7), human bladder (T24), human colon (Caco-2, HT-29), human liver (HepG2), human cervical (SiHa), and human chronic myeloid leukemia (K562) cell lines. These phytocompounds were able to induce apoptotic cell death towards cancer cells via both intrinsic and extrinsic pathways [174, 176–178, 181, 184–186], and also affect cell migration [184]. Nevertheless, more in-depth investigations using in vivo model systems are vital to validate betacyanins’ therapeutic abilities against cancer for clinical application. Furthermore, employing proteomics and metabolomics analyses can facilitate clarifying the specific pathways (e.g., 2-oxocarbodylic acid metabolism) that are potentially affected by betacyanin, as demonstrated by the 5-FU nanoparticle combination for colon cancer [191].
Antimicrobial resistance (AMR) is of significant concern worldwide and impacts human, animal, and food health and safety [192]. AMR is projected to worsen due to the lack of innovation, a decline in funding, and an emphasis on antimicrobial research over the last several decades, antibiotic overuse, inappropriate prescribing, extensive use in agricultural production, and the lack of new antibiotics [192–195]. As such, it is imperative that new, effective, and accessible antimicrobials are developed.
Many new antibiotics are synthetic derivatives of older drugs such as tetracycline, fluoroquinolones, and aminoglycosides, for example [196], but these may not meet the needs for clinical practice, especially in hard-to-reach and low-resource communities. In fact, many micro-organisms are resistant to these drugs. Alternative and supplementary treatments will help bridge this gap in treating infectious diseases and recent trends in research and a surge in interest in natural therapies is driving a re-evaluation of the health benefits of natural products. For example, drimenol, a sesquiterpene alcohol from mainly Polygonum spp. and Drimys spp. has been reported to have significant antimicrobial activities [197]. The antimicrobial activity of betalains is well documented, and recent studies show antimicrobial activity of betacyanins and betanin.
Biofilms are of significant concern due to their impact on healthcare, water systems, and the food industry [198]. These bacterial communities are difficult to remove, and where removal is possible, harsh physical or chemical methods may be required [198]. Preventing biofilm formation is a legitimate and preferable goal [199]. A study by Yong et al. [200], showed that extracts from Amaranthus dubius and H. polyrhizus inhibited biofilm formation by approximately 30–50% when in combined action. Yong et al. [200], also separately showed that H. polyrhizus alone had antibacterial activity against 10 pathogenic Gram-positive and 6 Gram-negative bacteria and showed that refrigerating the extract at 4 degrees for 6 days increased the antibacterial activity by reducing the MIC by 8–16 fold.
Vulić et al. [201] reported that commercial beetroot pomace had antibacterial activity against 6 Gram-negative bacteria and 5 Gram-positive bacteria, comparing quite favorably to conventional controls (ceforaxime/claulanic acid), with the pomace showing reduced growth in an area that was at least half to as large as the control. This study showed that the pomace was not as effective as the conventional drugs they were compared with [201]. Gong et al. [202] reported that beetroot extract could inhibit the growth of L. monocytogenes and maintain cooked pork for a longer period of time without spoiling. Using a C. elegans model, Choo et al. [203] showed that beetroot extract could completely inhibit the growth of MRSA at 76 h. Furthermore, at 18 h post-infection, beetroot extract completely inhibited further MRSA growth [203]. Betacyanins from H. polyrhizus and A. dubius were shown to decrease the proliferation of Dengue virus type two in a Vero cell culture. The A. dubius extract exhibited significantly greater inhibition of dengue virus [204].
The extraction method has been shown to significantly impact the concentration and antimicrobial efficacy of the betacyanins from fruits and vegetables [205]. Tenore et al. [205] showed that different extract fractions from peel and fruit flesh of red pitaya fruits had differing effects on the antimicrobial properties. Betacyanins extracted from the flesh of red pitaya showed higher efficacy than whole flesh or peel extract [205]. The antibacterial activity of the flesh and peel extracts of betacyanin-rich extracts is dependent on the quantity and quality of betacyanin in the extract fraction. The fraction extracted from the flesh was more efficacious with a lower minimum inhibitory concentration [205]. Tenore et al. [205] study and others indicate the important considerations affecting the antimicrobial capacity include the extraction method, method and length of storage [101, 206, 207], environmental growth conditions [53], and the state of ripeness [19]. These considerations affect the test results reported, but can also affect the efficacy of any prescribed usage for therapeutic or preventative purposes.
The review by Wijesinghe and Choo [208] provides a broad look at the betalains and addresses some of the problems with comparing studies. There are different standards for reporting concentrations, the different sources of the active betacyanins, and the extraction, storage, and application methods. A review of studies indicates that the minimum inhibitory concentration of conventional drugs is much lower than that of the betacyanin extracts from different sources when comparing broth dilution and agar well diffusion methods [208].
Sutor and Wybraniec [25] report the quantitation of betacyanins from M. violaceus, M. bahiensis, M. amoenus, and M. curvispinus using chromatographic and mass spectrometric methods. The wide range in quality and quantity of betacyanin suggests that more work needs to be done to describe and understand the possibility of using all plants that produce betacyanins for antimicrobial purposes [25]. The available research suggests that betacyanins or betanin may play an important role in the control of infectious diseases as research provides important information on the best extraction, characterization, and concentration of the active phytochemicals, location of the most efficacious betacyanins or betanin fractions (i.e., pulp, peel, etc.), shelf-life, and other information necessary for use in therapeutics. Some of the antimicrobial effects of betacyanins are summarized in Table 5.
Betacyanins and their antimicrobial properties.
| Source | Model | Dosage/Duration of treatment | Biological effects and mechanisms of action | References |
|---|---|---|---|---|
| A. dubiusH. polyrhizus | In vitro (S. aureus) | 0.313 mg/mL to 2.5 mg/mL per 24 h | Reduced biofilm by 30–50% | Yong et al. [200], 2019 |
| B. vulgaris | In vitro (S. aureus, S. typhimurium, C. fruendii, S. sciuri, B. cereus) | 4.09 ± 0.20 mg betanin equivalents/gram of beetroot pomace extract/24 h at 37°C | Inhibited S. typhimurium, C. fruendii, S. aureus, S. sciuri, B. cereus | Vulić et al. [201], 2013 |
| B. vulgaris | In vitro (L. monocytogenes)ATCC 19116 | Beetroot extract: 20 mg/mL (1 MIC) at 37°C for 3 h, 40 mg/mL (2 MIC) at 37°C for 1 h | L. monocytogenes (108 log CFU/mL) was completely inhibited by treatment with 20 mg/mL for 3 h and 40 mg/mL for 1 h. | Gong et al. [202], 2022 |
| In vitro (L. monocytogenes) in cooked pork | Beetroot extract: 20 mg/mL (1 MIC), 40 mg/mL (2 MIC), both stored 6 days | The counts of L. monocytogenes decreased during storage, compared to the control group. | ||
| B. vulgaris | In vivo (C. elegans) | 20 mg/mL/18 h at 37°C; 76 h | Complete inhibition of MRSA growth; killed MRSA to extend the life of C. elegans | Choo et al. [203], 2020 |
| H. polyrhizus and A. dubius | In vitro (Dengue virus type 2) | 125.8 µg/mL (H. polyrhizus) vs. 14.62 µg/mL (A. dubius) (IC50) 48 h at 37°C | Increased infection inhibition | Chang et al. [204], 2019 |
A. dubius: Amaranthus dubius; H. polyrhizus: Hylocereus polyrhizus; B. vulgaris: Beta vulgaris; L. monocytogenes: Listeria monocytogenes; S. aureus: Staphylococcus aureus.
Molecular docking of natural products is increasingly used to predict phytochemical interactions with new biological targets, explaining traditional uses and uncovering novel medicinal applications [209]. This approach accelerates drug discovery by saving time and costs and enabling the introduction of new molecular scaffolds for further testing and optimization [210].
The well-documented radical scavenging activity of betacyanins is computationally rationalized by their strong predicted interactions with key enzymatic sources of oxidative stress. Enzymatically, xanthine oxidase (XO) catalyzes the conversion of xanthine and hypoxanthine into uric acid, generating ROS implicated in oxidative stress-related pathologies [211]. Molecular docking studies by Ramirez-Velasquez et al. [17] revealed that betanin, isobetanin, betanidin, and isobetanidin exhibit strong interactions with XO’s active site, with binding affinities exceeding that of febuxostat, a known XO inhibitor [212]. Betanidin, isobetanidin, and betanin formed stable complexes via electrostatic, hydrogen bonding, and hydrophobic interactions, supporting their potential as effective XO inhibitors and antioxidants [17].
In the inflammation pathway, NF-κB activation involves its nuclear translocation and induction of pro-inflammatory pathways [213]. ElSayed et al. [214] (2023) demonstrated via molecular docking and CASTp analysis that betanin binds the DNA-binding domain of NF-κB. Its central indole ring anchors via π-stacking with TYR36, stabilized by polar interactions with Arg33, Lys122, Arg124, and Lys218 [214]. This binding site overlap with DNA suggests betanin competitively inhibits NF-κB activation, corroborated by in vivo data showing reduced NF-κB expression post-treatment. Furthermore, lipoxygenase (LOX) enzymes mediate inflammation via eicosanoid production and numerous phytochemicals and natural products induce the desired anti-inflammatory effects through inhibition of these enzymes [215]. Betanidin significantly inhibits LOX activity in a dose-dependent manner. Molecular docking indicates that indoline-derived betacyanin occupies a pocket near the loop preceding helix 2. However, glucosylation of betanidin to betanin markedly diminishes its LOX-inhibitory capacity [216]. However, a high docking score is necessary but not sufficient for biological activity, as it fails to account for crucial pharmacokinetic factors like bioavailability and metabolism.
When considering its antimicrobial activity, Chaari et al. [217] (2023) investigated the interactions of betacyanin and betaxanthin—constituting 75–95% and 5–25% of betalains, respectively—with bacterial DNA/RNA polymerases and cell membranes of S. aureus and Salmonella enterica. Molecular docking revealed betacyanin formed stable complexes with DNA and RNA polymerases via van der Waals, hydrogen bonding, and various π-interactions. Both pigments also disrupted membrane integrity by inhibiting efflux pumps (AcrAB-TolC in S. enterica; MepR/MepA in S. aureus), indicating promising antibacterial activity against foodborne pathogens. Molecular docking studies have demonstrated strong interactions between betacyanin and critical severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) viral proteins, particularly the spike glycoprotein in its closed state (PDB ID: 6VXX) and the receptor-binding domain (PDB ID: 6YLA). These interactions exhibited binding affinities comparable to established antiviral agents such as nelfinavir and hydroxychloroquine sulfate, indicating potential for therapeutic intervention in COVID-19 [218].
In cancer-related targets, betanin has shown promising binding behavior with enzymes involved in DNA replication and cell cycle regulation. Notably, it formed more favorable interactions with human DNA topoisomerase I than topoisomerase II, and demonstrated robust affinity with cyclin-dependent kinase (CDK)-6, an essential cell cycle protein. Additionally, betanin exhibited moderate interactions with apoptosis-associated proteins, Bcl-2 and caspase-3, with docking scores of −4.522 and −6.108 kcal/mol, respectively. Supporting the role of betanin in inhibiting DNA replication, suppressing cell cycle progression, and promoting apoptotic activity, paralleling the effects of the chemotherapeutic agent doxorubicin, to which it bears structural similarity [219].
Molecular docking provides a valuable framework for connecting the dots between betalain chemistry and observed biology. It translates empirical findings into structural hypotheses, suggesting that betalains may act as multi-target agents by directly modulating key enzymes and receptors. However, critical interpretation is important as the consistent theme of high binding affinity across diverse targets may point to both their therapeutic potential and a limitation of docking algorithms. The true mechanism of action likely involves a combination of direct target engagement and indirect effects stemming from their potent redox activity and modulation of core signaling pathways like Nrf2 and NF-κB. Therefore, docking predictions must be viewed as the starting point for rigorous laboratory experimental validation, not as conclusive proof of mechanisms [220].
Overall, these molecular docking studies, despite being computational, reveal that betacyanins can target key enzymes and proteins involved in inflammation, antioxidant defense, antimicrobial, antiviral, and anticancer pathways. This supports their promising role as nutraceuticals with diverse therapeutic applications (Figure 6).
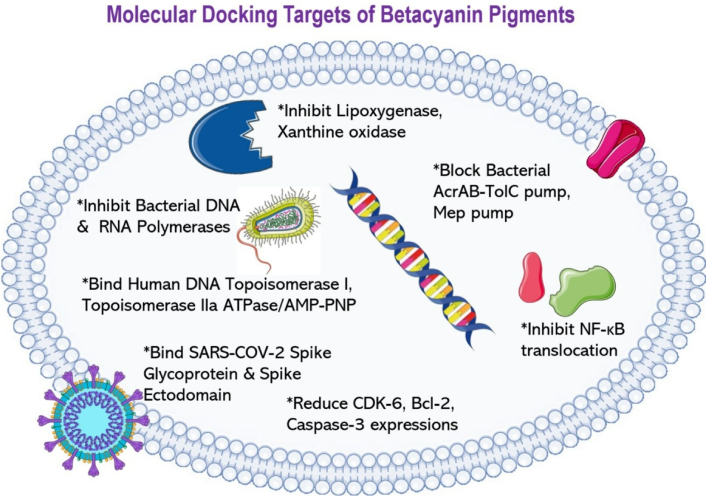
The molecular docking targets of betacyanin pigments on enzymes and/or proteins involved in various cellular pathways. Betacyanins demonstrated inhibitory activities in LOX, xanthine oxidase, in bacterial DNA and RNA polymerases, in human DNA topoisomerase I and IIa, in NF-κB protein, in genes involved in cell cycle and apoptosis, and in SARS-CoV-2 virus spike protein and receptor binding domain. These observations likely explain the molecular aspects of betacyanins’ activities observed in biological studies. LOX: lipoxygenase; NF-κB: nuclear factor-kappa B.
This review discusses the natural sources, biosynthesis, extraction methods, applications, antimicrobial, anticancer, and the positive effects of betacyanins against cardiometabolic diseases, with novel reports of the effects of this compound group on gut microbiome and molecular docking properties. Despite the favorable effects reported in the article, it is important to note that the results reported in these studies should be interpreted with some limitations. For instance, the number of studies reporting the effects of specific betacyanins from plants on disease models is still limited. Many studies used products that have a combination of different phytochemicals and pigments, thus complicating the interpretation. Betalain-rich food also comprises several other potentially relevant compounds, such as polyphenols and nitrates, and their interactions in biological systems should be considered. To answer this, further studies could consider separating individual components and comparing their effects with complete or combined betacyanins from whole fruits or plants.
To enhance bioavailability, diversifying food sources, food processing, and food matrices could potentially increase betacyanin concentration and thus enhance absorption, overcoming the challenges of their limited absorption. Moreover, more studies should be done to increase the extraction of betacyanins from plant waste such as fruit peels. This will greatly reduce the environmental impact through food waste and generate income for growers. More rigorous clinical trials should be implemented soon to provide strong evidence-based information for clinical practice. Considering the current information available in the literature, existing evidence is pointing towards the application of betacyanins as a promising natural food pigment having multiple health benefits, acting through various biological mechanisms, including their interactions with the gut microbiome and binding of key cellular components.
5-FU: 5-fluorouracil
5GT: 5-O-glucosyl transferase
ALT: alanine aminotransferase
AMPK: adenosine monophosphate-activated protein kinase
AMR: antimicrobial resistance
AST: aspartate aminotransferase
Bad: B-cell lymphoma 2-associated agonist of cell death
Bax: B-cell lymphoma 2-associated X protein
Bcl-2: B-cell lymphoma 2
BHE: beetroot hydro-alcoholic extract
BUN: blood urea nitrogen
C/EBPα: CCAAT/enhancer-binding protein alpha
CAT: catalase
CCK-8: Cell Counting Kit-8
CDK: cyclin-dependent kinase
COX-2: cyclooxygenase-2
DAPI: 4’,6-diamidino-2-phenylindole
DF: diclofenac
DOD: 4,5-dihydroxyphenylalanine-extradiol-dioxygenase
DOPA: dihydroxyphenylalanine
dw: dry weight
EAE: enzyme-assisted extraction
ESI: electrospray ionization
FGF21: fibroblast growth factor 21
FGFR1/2: fibroblast growth factor receptor 1/2
fw: fresh weight
GPx: glutathione peroxidase
GSH: reduced glutathione
HHP: high hydrostatic pressure
HO-1: heme oxygenase 1
HPLC: high-performance liquid chromatography
iNOS: inducible nitric oxide synthase
LOX: lipoxygenase
MAE: microwave-assisted extraction
MDA: malondialdehyde
MRP2: multidrug resistance-associated protein 2
mTOR: mechanistic target of rapamycin
NF-κB: nuclear factor-kappa B
Nrf2: nuclear factor erythroid 2-related factor 2
PEF: pulsed electric field
PI3K: phosphoinositide 3-kinase
PPARγ: peroxisome proliferator-activated receptor gamma
ROS: reactive oxygen species
SARS-CoV-2: severe acute respiratory syndrome coronavirus 2
SCCD: supercritical carbon dioxide
SCFAs: short-chain fatty acids
SFE: supercritical fluid extraction
SIRT1: Sirtuin-1
SOD: superoxide dismutase
SREBP-1c: sterol regulatory element-binding protein 1c
TGF-β: transforming growth factor beta
TNF-α: tumor necrosis factor alpha
TRAILR4: tumor necrosis factor-related apoptosis-inducing ligand receptor 4
UAE: ultrasound-assisted extraction
UGTs: UDP-glycosyltransferases
XO: xanthine oxidase
α-SMA: alpha-smooth muscle actin
The authors would like to thank Mr. James Wong Yung Khai and Mr. Luther Ku for their assistance in drawing some of the diagrams in this manuscript. English grammar was improved using the Grammarly application and proofread by Dr. Elvin M. Walemba.
ODJ: Supervision, Project administration, Conceptualization, Investigation, Visualization, Writing—original draft, Writing—review & editing. WGS: Investigation, Visualization, Writing—original draft. APGdS: Conceptualization, Investigation, Visualization, Writing—original draft, Writing—review & editing. SAT: Investigation, Writing—original draft, Writing—review & editing, Validation. CYB: Investigation, Writing—original draft, Writing—review & editing, Validation. RN: Investigation, Writing—original draft, Writing—review & editing, Validation. EMW: Investigation, Writing—original draft, Writing—review & editing, Validation. ATM: Conceptualization, Investigation, Visualization, Writing—original draft, Writing—review & editing, Validation. All authors have read and agreed to the published version of the manuscript.
The authors declare that there are no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
