Affiliation:
1Clinical Academic Center of Trás-os-Montes and Alto Douro (CACTMAD), University of Trás-os-Montes and Alto Douro, 5000-801 Vila Real, Portugal
Email: sgoncalves@utad.pt
ORCID: https://orcid.org/0000-0002-8287-1357
Affiliation:
2Serviço de Medicina Dentária, Hospital da Luz de Vila Real, 5000-657 Vila Real, Portugal
Affiliation:
3Animal and Veterinary Research Center (CECAV), Associate Laboratory for Animal and Veterinary Sciences (AL4AnimalS), UTAD, 5000-801 Vila Real, Portugal
4Department of Genetics and Biotechnology (DGB–ECVA), UTAD, 5001-801 Vila Real, Portugal
ORCID: https://orcid.org/0000-0002-5463-0038
Affiliation:
1Clinical Academic Center of Trás-os-Montes and Alto Douro (CACTMAD), University of Trás-os-Montes and Alto Douro, 5000-801 Vila Real, Portugal
5RISE-Health, School of Health, University of Trás-os-Montes and Alto Douro, 5000-801 Vila Real, Portugal
ORCID: https://orcid.org/0000-0002-2540-682X
Affiliation:
6Unidade de Cuidados na Comunidade de Alijó, Unidade Local de Saúde de Trás-os-Montes e Alto Douro, 5070-021 Alijó, Portugal
Email: veroenf@gmail.com
ORCID: https://orcid.org/0009-0001-5266-9334
Explor Neuroprot Ther. 2026;6:1004149 DOI: https://doi.org/10.37349/ent.2026.1004149
Received: February 09, 2026 Accepted: April 07, 2026 Published: April 28, 2026
Academic Editor: Guoku Hu, Zhejiang Provincial People’s Hospital, China
The article belongs to the special issue Breakthroughs in Mechanisms and Treatments for Neurodegenerative Diseases
Neurodegenerative disorders represent a major and growing global health challenge, characterized by progressive neuronal loss, multifactorial pathophysiology, and limited disease-modifying pharmacological options. Increasing attention has therefore been directed toward non-pharmacological and integrative interventions as complementary strategies for neuroprotection and symptom management. These approaches target key mechanisms implicated in neurodegeneration, including oxidative stress, neuroinflammation, mitochondrial dysfunction, synaptic impairment, and dysregulated neuroplasticity. This narrative integrative review synthesizes current preclinical and clinical evidence on non-pharmacological interventions with demonstrated or emerging neuroprotective potential across major neurodegenerative disorders, including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and related conditions. The review focuses on four principal domains: physical activity and exercise, nutritional and dietary strategies, mind–body and psychosocial interventions, and sensory or neuromodulatory therapies. Collectively, these interventions influence convergent neurobiological pathways, including neurotrophic signaling, immune modulation, autonomic regulation, and gut–brain communication. Studies indicate that structured physical exercise enhances neurotrophic factor expression and mitochondrial resilience; dietary patterns rich in antioxidants and anti-inflammatory components mitigate oxidative damage and neuroinflammation; mind–body practices modulate stress-related neuroendocrine pathways and promote functional connectivity; and sensory or neuromodulatory interventions engage limbic and cortical networks relevant to cognition, mood, and motor control. Importantly, multimodal and integrative approaches appear to exert synergistic effects, aligning with the complex and systemic nature of neurodegenerative processes. Despite promising findings, challenges related to methodological heterogeneity, biomarker validation, and translational implementation persist. Future research should prioritize standardized protocols, objective neuroprotective endpoints, and personalized intervention frameworks supported by digital health technologies. Overall, non-pharmacological and integrative therapies represent a valuable, increasingly evidence-based component of comprehensive neuroprotective strategies, with significant potential to enhance quality of life and complement pharmacological treatments in the care of neurodegenerative diseases.
Neurodegenerative disorders constitute a major and escalating public health challenge worldwide, driven by aging populations and the growing prevalence of chronic neurological conditions [1]. Diseases such as Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), and amyotrophic lateral sclerosis (ALS) are characterized by progressive neuronal dysfunction and loss, leading to cognitive decline, motor impairment, behavioral disturbances, and reduced quality of life [2]. Despite decades of intensive research, current pharmacological treatments remain largely symptomatic, with limited capacity to halt or reverse disease progression. This therapeutic gap has intensified interest in complementary strategies that can modulate disease mechanisms, enhance neuronal resilience, and support long-term functional outcomes.
Neurodegeneration arises from complex and interacting pathological processes, including oxidative stress, mitochondrial dysfunction, neuroinflammation, excitotoxicity, impaired protein homeostasis, and synaptic failure [3]. These mechanisms evolve over extended periods and are influenced by genetic susceptibility, environmental exposures, metabolic status, and psychosocial factors. Consequently, single-target pharmacological approaches have shown limited efficacy in addressing the multifactorial nature of neurodegenerative diseases [4]. In contrast, non-pharmacological and integrative interventions offer a systems-level perspective, targeting multiple biological pathways simultaneously while also addressing behavioral, emotional, and functional dimensions of disease [5].
Growing evidence from epidemiological studies, experimental models, and clinical trials suggests that lifestyle factors, including physical activity, nutrition, stress regulation, and sensory stimulation, play a critical role in maintaining brain health and modulating neurodegenerative risk. Regular exercise has been associated with enhanced neurotrophic signaling, improved mitochondrial function, and reduced neuroinflammatory burden [6]. Dietary patterns rich in antioxidants and anti-inflammatory compounds have been linked to improved cognitive trajectories and reduced neurodegenerative risk, partly by modulating oxidative stress and the gut–brain axis [7]. Mind–body and psychosocial interventions, including mindfulness-based practices and movement therapies, influence autonomic regulation, neuroendocrine balance, and emotional resilience, all of which are increasingly recognized as contributors to the progression of neurodegenerative disease. Sensory and neuromodulatory interventions further engage central and peripheral neural circuits involved in cognition, mood, and motor control [8].
Importantly, these non-pharmacological strategies are not confined to symptom management but may exert genuine neuroprotective and neurorestorative effects by enhancing neuroplasticity, preserving synaptic integrity, and promoting adaptive immune and metabolic responses. Advances in neuroimaging, molecular biology, and biomarker research have strengthened the biological plausibility of these effects, revealing measurable changes in brain structure, connectivity, and inflammatory and oxidative profiles following integrative interventions [9]. At the same time, translational challenges remain, including heterogeneity in intervention protocols, variability in outcome measures, and limited standardization across clinical studies.
Within this evolving landscape, there is a need for integrative syntheses that bridge mechanistic insights with clinical relevance, highlighting both the opportunities and limitations of non-pharmacological neuroprotective strategies. This narrative integrative review examines the current evidence supporting non-pharmacological approaches to neuroprotection and symptom management in neurodegenerative disorders, with a focus on physical activity, nutritional interventions, mind–body and psychosocial therapies, and sensory or neuromodulatory techniques. By contextualizing these interventions within established neurobiological mechanisms and translational frameworks, this review seeks to contribute to a more comprehensive, patient-centered understanding of neuroprotective therapy.
While AD, PD, HD, and multiple sclerosis (MS) are discussed collectively in this review due to overlapping mechanisms such as neuroinflammation, oxidative stress, and synaptic dysfunction, it is important to recognize that these disorders differ substantially in their primary pathophysiology, clinical progression, and affected neural systems. Where relevant, distinctions in disease-specific evidence are highlighted to provide a more nuanced interpretation of the findings.
Within this evolving landscape, there is also a growing need to conceptualize how non-pharmacological interventions can be meaningfully integrated into patients’ daily lives. The emerging framework of “vitactions” conceptualizes structured, health-promoting “life-actions” as intrinsically meaningful and sustainable behaviors embedded within individual routines and preferences [10]. Rather than viewing interventions such as exercise, diet, or mindfulness as isolated prescriptions, this perspective emphasizes their role as lived, patient-centered practices that align with motivational and psychosocial determinants of adherence [11]. In this context, the present review not only synthesizes mechanistic and clinical evidence but also considers how these interventions may be operationalized within a more integrative and patient-centered framework.
To improve clarity, this review distinguishes, where possible, between preventive and disease-modifying effects, between symptomatic and neuroprotective outcomes, and between preclinical and clinical evidence.
This narrative integrative review aimed to synthesize current evidence on non-pharmacological approaches for neuroprotection and symptom management in neurodegenerative disorders. A structured literature search was conducted in PubMed, Scopus, and Web of Science to identify relevant studies. Search terms included combinations of keywords such as “neurodegenerative diseases”, “neuroprotection”, “non-pharmacological interventions”, “exercise”, “nutrition”, “mind–body therapies”, and “neuromodulation”.
Eligible studies included preclinical studies, clinical trials, and relevant review articles that addressed the mechanisms or clinical effects of non-pharmacological interventions in conditions such as AD, PD, and HD. Articles published in any language were considered. Priority was given to recent studies, although seminal papers were also included when relevant.
Studies were selected based on their relevance to the objectives of the review and their contribution to understanding underlying neurobiological mechanisms and translational applications. Given the narrative nature of this review, no formal quality assessment or meta-analysis was performed; instead, evidence was critically synthesized to provide an integrative and clinically meaningful overview.
Neurodegenerative disorders such as AD, PD, ALS, and HD share overlapping pathological mechanisms that culminate in progressive neuronal dysfunction and death [12]. Despite distinct clinical phenotypes, converging molecular events—including oxidative stress, mitochondrial impairment, protein misfolding and aggregation, excitotoxicity, neuroinflammation, and synaptic dysfunction—play pivotal roles in their onset and progression. These processes often form self-perpetuating cycles that exacerbate cellular damage, impair neuroplasticity, and ultimately lead to neuronal loss in both the central and peripheral nervous systems.
Among the most prominent drivers of neurodegeneration is oxidative stress, arising from an imbalance between reactive oxygen species (ROS) production and antioxidant defense systems [13]. Chronic oxidative stress damages lipids, proteins, and nucleic acids, contributing to mitochondrial dysfunction and impaired energy metabolism [14]. Mitochondria, in turn, are both sources and targets of ROS, linking metabolic failure to neuronal vulnerability. In parallel, neuroinflammation, primarily mediated by activated microglia and astrocytes, contributes to the sustained release of pro-inflammatory cytokines [e.g., tumor necrosis factor-α (TNF-α), interleukin-1 beta (IL-1β)] and reactive nitrogen species, further amplifying oxidative and excitotoxic cascades [15, 16].
Another hallmark is the accumulation of misfolded or aggregated proteins, such as amyloid-β, tau, α-synuclein, and transactive response DNA binding protein 43kDa (TDP-43), which disrupt intracellular trafficking and synaptic communication [17]. Excitotoxicity, resulting from excessive glutamate receptor activation, leads to calcium overload and activation of apoptotic pathways. Together, these processes impair neuronal communication, plasticity, and survival [17, 18].
Non-pharmacological interventions can modulate many of these molecular and cellular mechanisms, offering a multifaceted neuroprotective potential. Regular physical activity enhances mitochondrial biogenesis, upregulates neurotrophic factors such as brain-derived neurotrophic factor (BDNF), and promotes antioxidant enzyme expression [6]. Nutritional strategies rich in polyphenols, omega-3 fatty acids, and vitamins C and E can counteract oxidative stress and reduce neuroinflammatory signaling by modulating the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and nuclear factor erythroid 2-related factor 2 (Nrf2) pathways [6]. Mind–body interventions, including meditation and mindfulness, have been associated with downregulation of pro-inflammatory markers and enhanced synaptic plasticity, potentially via modulation of the hypothalamic–pituitary–adrenal (HPA) axis [19, 20].
Recent studies also highlight the gut–brain axis as a key mediator of neuroprotection, in which dietary patterns, probiotics, and stress-reduction strategies can influence neuroinflammatory tone and neurotransmitter balance via microbiota-derived metabolites, such as short-chain fatty acids. In addition, sensory and neuromodulatory interventions—such as music therapy, acupuncture, or aromatherapy—appear to engage autonomic and limbic circuits, thereby contributing to improved homeostasis and emotional regulation [21–24].
Collectively, these findings suggest that non-pharmacological approaches act on multiple converging targets, offering both symptomatic relief and disease-modifying potential. By enhancing intrinsic repair mechanisms, reducing oxidative and inflammatory stress, and fostering neuroplasticity, integrative interventions may complement pharmacological agents and represent promising avenues for translational neuroprotective therapy.
Given the multifactorial nature of neurodegenerative disorders and the convergence of oxidative, inflammatory, metabolic, and synaptic mechanisms, non-pharmacological interventions may exert their effects through overlapping and interdependent biological pathways. Although categorized into distinct domains—physical activity, nutritional strategies, mind–body therapies, and sensory or neuromodulatory approaches—these interventions converge on shared molecular targets, including neurotrophic signaling, mitochondrial resilience, immune modulation, autonomic regulation, and synaptic plasticity. To synthesize these interrelationships, a conceptual framework of converging neuroprotective mechanisms is presented in Figure 1. This model illustrates how diverse integrative therapies act at multiple biological levels yet ultimately influence common functional outcomes such as cognitive preservation, motor performance, emotional regulation, and disease trajectory.
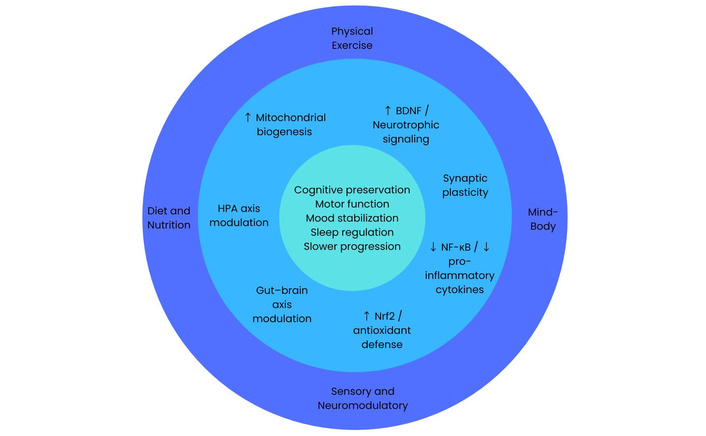
Conceptual framework of converging neuroprotective mechanisms targeted by non-pharmacological interventions in neurodegenerative disorders. Distinct intervention domains act through overlapping molecular and systems-level pathways, ultimately influencing cognitive, motor, and affective outcomes. BDNF: brain-derived neurotrophic factor; HPA: hypothalamic–pituitary–adrenal; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; Nrf2: nuclear factor erythroid 2-related factor 2.
Physical activity is among the most consistently supported non-pharmacological strategies for neuroprotection [25]. A growing body of experimental and clinical evidence demonstrates that regular exercise exerts multidimensional benefits on brain structure and function, mitigating many of the molecular mechanisms underlying neurodegenerative disorders. These effects include enhanced neurotrophic signaling, modulation of oxidative and inflammatory pathways, improved cerebral perfusion, and stimulation of synaptic plasticity [26, 27].
At the cellular level, exercise stimulates mitochondrial biogenesis and bioenergetic efficiency, increasing neuronal resilience to metabolic stress [28]. It also promotes the expression of neurotrophic factors such as BDNF, nerve growth factor (NGF), and insulin-like growth factor-1 (IGF-1), which are essential for neuronal survival, synaptic maintenance, and neurogenesis—particularly within the hippocampus and motor circuits [26]. Aerobic and resistance training have been shown to attenuate β-amyloid accumulation, reduce α-synuclein aggregation, and modulate microglial neuroinflammatory phenotypes from pro-inflammatory (M1) to neuroprotective (M2) states [28, 29].
Exercise also has systemic effects that contribute to neuroprotection. Enhanced vascular function and angiogenesis improve cerebral blood flow and oxygen delivery, while reductions in insulin resistance and systemic inflammation indirectly support neuronal health [29]. The activation of antioxidant pathways, including the upregulation of superoxide dismutase (SOD) and catalase, helps neutralize ROS and preserve mitochondrial integrity. Moreover, exercise modulates the HPA axis and reduces glucocorticoid-induced neurotoxicity, thereby stabilizing the neuroendocrine environment [30].
From a behavioral perspective, different modalities of physical activity yield distinct but complementary benefits. Aerobic training (e.g., walking, cycling, swimming) is most strongly associated with cognitive enhancement and hippocampal volume preservation, while resistance exercise improves motor control and balance, particularly relevant in PD [6, 26]. Mind–body forms of exercise, such as yoga, Tai Chi, and Qigong, integrate movement with controlled breathing and mindfulness, contributing additional autonomic and psychosocial regulation that enhances overall neuroprotective outcomes [31].
Although beneficial effects have been observed across multiple neurodegenerative conditions, the magnitude and specific outcomes may vary depending on disease-specific mechanisms and clinical features. In clinical settings, structured exercise interventions have shown measurable benefits in patients with mild cognitive impairment (MCI), AD, PD, and MS [32, 33]. Randomized controlled trials report improvements in executive function, gait performance, and mood, as well as reductions in inflammatory biomarkers [34]. Neuroimaging studies further reveal increased hippocampal connectivity and gray matter density following regular exercise programs, reinforcing the translational potential of movement-based therapies in MS [35].
Despite robust evidence, challenges remain regarding the standardization of exercise protocols, optimal frequency and intensity, and long-term adherence in patients with advanced neurodegenerative disease. Future translational research should prioritize precision exercise prescriptions, integrating physiological biomarkers and digital monitoring tools to tailor programs to individual capacities and disease stages. Combining physical activity with dietary, cognitive, or sensory interventions may also yield synergistic neuroprotective effects, representing a promising direction for integrative neurotherapeutics.
From a vitactions perspective, physical activity may be most effective when integrated as a meaningful and enjoyable component of daily life, rather than a prescribed task, thereby enhancing long-term adherence and neuroprotective impact.
Despite strong support from experimental and clinical studies, the strength of evidence varies across populations and outcomes. While multiple randomized controlled trials demonstrate benefits in cognitive and motor domains, some studies report modest or non-significant effects, particularly in advanced disease stages. Furthermore, heterogeneity in intervention protocols and outcome measures limits comparability. Overall, the evidence base is strongest for aerobic exercise in MCI and early-stage disease, whereas data in advanced neurodegeneration remain less consistent.
Overall, evidence for exercise is strongest for symptomatic clinical benefits, while disease-modifying and neuroprotective effects are primarily supported by preclinical and emerging clinical data.
Nutrition plays a central role in maintaining neuronal health and modulating the biological processes that underlie neurodegeneration. Accumulating evidence from epidemiological, experimental, and clinical studies demonstrates that specific dietary patterns and bioactive nutrients exert neuroprotective effects through antioxidant, anti-inflammatory, and metabolic regulatory mechanisms [36]. These nutritional strategies not only mitigate neuronal injury but also influence synaptic plasticity, neurogenesis, and brain connectivity—key determinants of resilience against the progression of neurodegenerative disease.
At the molecular level, many dietary compounds modulate oxidative stress and inflammatory signaling, two critical pathways involved in neurodegeneration. Polyphenols—including resveratrol, curcumin, and flavonoids from fruits, tea, and cocoa—scavenge ROS and activate Nrf2, thereby enhancing endogenous antioxidant defense [37]. Concurrently, these compounds suppress NF-κB activation, leading to reduced expression of pro-inflammatory cytokines (IL-1β, TNF-α, IL-6) and inducible nitric oxide synthase (iNOS) [38, 39].
Omega-3 polyunsaturated fatty acids, particularly docosahexaenoic acid and eicosapentaenoic acid, play pivotal roles in membrane fluidity, synaptic transmission, and the resolution of inflammation via specialized pro-resolving mediators such as resolvins and protectins [40]. Omega-3 supplementation attenuates β-amyloid deposition, improves dopaminergic signaling, and modulates microglial polarization toward neuroprotective phenotypes [41].
Vitamins and minerals—notably vitamins E, C, D, and B-complex, as well as zinc and selenium—also contribute to neuroprotection through antioxidant mechanisms, homocysteine metabolism regulation, and modulation of neurotransmitter synthesis [11, 42]. Deficiencies in these micronutrients have been associated with increased oxidative stress and accelerated cognitive decline [43].
Beyond individual nutrients, dietary patterns provide a more holistic framework for understanding nutritional neuroprotection. The Mediterranean diet, characterized by a high intake of fruits, vegetables, whole grains, olive oil, legumes, and fish, has consistently been linked to a reduced risk of AD, PD, and HD [44, 45]. Its neuroprotective benefits are attributed to the synergistic effects of antioxidants, healthy fats, and anti-inflammatory components, which collectively improve vascular function and reduce neuroinflammation [46].
The ketogenic diet (KD), high in fats and low in carbohydrates, promotes the production of ketone bodies, which serve as alternative energy substrates for neurons and exhibit antioxidant and anti-apoptotic properties [47, 48]. Emerging evidence suggests that KD may improve mitochondrial function, stabilize neuronal networks, and attenuate seizure activity and neuroinflammatory processes [49].
Similarly, the MIND [Mediterranean-Dietary Approaches to Stop Hypertension (DASH) Intervention for Neurodegenerative Delay] diet, explicitly designed to support cognitive function, emphasizes green leafy vegetables, berries, and whole grains. Prospective studies indicate that greater adherence to MIND diet recommendations is associated with slower cognitive decline and improved memory performance [50].
Recent research highlights the gut–brain axis as a crucial mediator of diet-induced neuroprotection. The gut microbiota communicates with the central nervous system via immune, endocrine, and neural pathways, influencing neuroinflammation, stress reactivity, and neurotransmitter synthesis [51]. Diets rich in prebiotics and probiotics enhance microbial diversity and promote the production of short-chain fatty acids such as butyrate, which possess anti-inflammatory and epigenetic regulatory properties. Dysbiosis, in contrast, has been linked with neuroinflammatory priming and blood–brain barrier disruption in neurodegenerative diseases [52, 53].
Clinical trials and longitudinal cohort studies reinforce the potential of dietary modulation for the prevention and management of neurodegenerative diseases. In AD, adherence to the Mediterranean or MIND diet is associated with delayed onset and slower progression of cognitive impairment [54, 55]. In PD and HD, diets enriched with antioxidants and omega-3 fatty acids correlate with improved motor and non-motor symptoms [56, 57]. Despite these promising findings, heterogeneity in dietary assessment methods, small sample sizes, and short intervention durations limit the generalizability of current results.
Future research should integrate nutrigenomic and metabolomic approaches to elucidate individualized responses to diet and optimize personalized nutritional interventions. Combining dietary modification with other lifestyle strategies—such as physical activity or mindfulness—may yield synergistic effects on neuroprotection, providing a foundation for integrative therapeutic models in neurodegenerative disease care. Within a vitactions framework, dietary patterns are not merely nutritional prescriptions but represent culturally and personally meaningful practices that can support sustained behavioral change and metabolic resilience.
Although epidemiological and longitudinal studies support nutritional strategies such as the Mediterranean and MIND diets, the level of clinical evidence remains heterogeneous. While some interventional studies suggest beneficial effects on cognitive decline and disease progression, others report limited or inconsistent findings, partly due to differences in dietary assessment methods, adherence, and population characteristics. Evidence for specific dietary interventions, such as the KD or micronutrient supplementation, is still emerging and often derived from small-scale or short-term studies. Furthermore, isolating the effects of individual nutrients from broader lifestyle factors remains challenging. These limitations highlight the need for well-controlled clinical trials and standardized dietary intervention protocols.
Current evidence suggests a stronger role in prevention and risk reduction, with more limited and heterogeneous data supporting disease-modifying effects in clinical populations.
Mind–body, and psychosocial therapies represent a rapidly expanding area of interest in neuroprotection and neurorestoration. These interventions, which include mindfulness meditation, yoga, Tai Chi, Qigong, music therapy, and cognitive-behavioral strategies, act on both psychological and physiological domains to enhance brain resilience [5, 58]. Increasing evidence from neuroimaging, biomarker, and clinical studies suggests that these approaches modulate neural circuitry, neurochemical balance, and inflammatory tone, ultimately promoting neuroplasticity and cognitive stability in patients with neurodegenerative disorders.
Mind–body practices exert neuroprotective effects primarily by regulating the HPA axis and balancing the autonomic nervous system [59]. Chronic stress and hyperactivation of the HPA axis contribute to hippocampal atrophy, neuroinflammation, and glucocorticoid-mediated neurotoxicity—processes that exacerbate neurodegenerative pathology. Mindfulness and meditative practices reduce cortisol secretion, enhance parasympathetic activity (as reflected by increased heart rate variability), and promote functional connectivity within fronto-limbic networks involved in emotion regulation and cognitive control [60, 61].
Functional magnetic resonance imaging (MRI) studies reveal that long-term meditation is associated with increased gray matter density in the hippocampus, prefrontal cortex, and anterior cingulate cortex—regions critical for memory, attention, and executive function [62]. These structural adaptations are accompanied by improved synaptic efficiency and upregulation of neurotrophic factors, such as BDNF, which support neuronal survival and synaptic plasticity. Furthermore, psychosocial interventions can attenuate systemic inflammation by downregulating NF-κB-related pathways and decreasing circulating levels of IL-6, TNF-α, and C-reactive protein [63, 64].
In AD, HD and MCI, mindfulness-based stress reduction (MBSR) and yoga interventions have demonstrated improvements in working memory, processing speed, and emotional well-being. These effects may be mediated by reduced stress reactivity and improved attentional control [65, 66]. In PD, Tai Chi and Qigong enhance balance, motor coordination, and postural stability while reducing fatigue and depressive symptoms. Such multimodal benefits reflect the integration of motor training with proprioceptive feedback and cognitive engagement, which collectively strengthen cortico-striatal connectivity and motor planning circuits [67, 68].
Music therapy has also shown promise as a non-invasive neuromodulatory tool. Listening to or producing music activates dopaminergic reward pathways and sensorimotor networks, leading to improvements in gait rhythm, mood, and motivation in PD, HD, and post-stroke rehabilitation [69, 70]. These effects underscore the role of audiovisual and rhythmic stimulation in enhancing neuroplasticity and functional recovery.
Psychosocial stress, social isolation, and mood disturbances are recognized risk factors for neurodegenerative disease onset and progression [71]. Interventions fostering social engagement, emotional expression, and cognitive stimulation can mitigate these risks and improve quality of life. Group-based mindfulness or movement programs, for example, not only provide cognitive and physical benefits but also enhance perceived social support, reducing loneliness and anxiety—factors closely associated with neuroinflammation and accelerated cognitive decline [72, 73].
Recent studies suggest that positive psychosocial experiences influence epigenetic mechanisms, such as histone acetylation and DNA methylation patterns in genes regulating inflammation and neurotrophic signaling [74]. These findings highlight the potential for mind–body interventions to exert long-term genomic and immunomodulatory effects relevant to neuroprotection.
Studies provide growing evidence that mind–body and psychosocial interventions yield clinically meaningful benefits in patients with AD, PD, and MS. Reported outcomes include improved mood, cognition, motor performance, and sleep quality. However, heterogeneity in study designs, intervention duration, and outcome measures limits direct comparisons across trials [75–77].
Future research should emphasize multimodal approaches that combine mind–body practices with physical exercise, nutritional modification, or sensory therapies to harness synergistic mechanisms. Incorporating digital tools, such as mobile mindfulness applications or virtual reality-based movement therapy, may enhance accessibility and adherence, facilitating the translation of psychosocial interventions into standard neurorehabilitative care. This aligns closely with the vitactions perspective, in which mind–body practices are embedded as intrinsically rewarding activities that reinforce emotional regulation and long-term engagement.
Mind–body interventions demonstrate promising effects on cognitive function, emotional regulation, and quality of life; however, the overall strength of evidence is variable. Many studies are based on small sample sizes, short intervention periods, and heterogeneous methodologies, limiting generalizability. While randomized controlled trials exist for interventions such as MBSR and Tai Chi, findings are not always consistent, and effect sizes are often modest. Additionally, outcomes are frequently based on subjective measures, such as self-reported well-being or mood, which may introduce bias. The lack of standardized intervention protocols and long-term follow-up data further constrains the interpretation of their sustained neuroprotective effects.
These interventions are primarily supported for their symptomatic benefits, with neuroprotective effects inferred largely from mechanistic and preclinical findings.
Sensory and neuromodulatory therapies encompass a diverse range of interventions—such as acupuncture, aromatherapy, massage therapy, music and light stimulation, and non-invasive brain modulation—that engage peripheral and central sensory pathways to promote neuroplasticity, homeostatic regulation, and symptom relief [78]. Increasing evidence suggests that these approaches can influence autonomic balance, neurotransmitter release, neuroinflammatory tone, and limbic system activity, thereby offering potential neuroprotective and restorative effects across neurodegenerative disorders [79, 80].
Sensory stimulation interventions act through multimodal mechanisms involving both bottom-up sensory inputs and top-down cortical modulation. Peripheral sensory activation—via tactile, olfactory, auditory, or visual pathways—elicits widespread neural responses in limbic, hypothalamic, and cortical regions [81, 82]. These responses modulate stress signaling, enhance dopaminergic and serotonergic transmission, and support neurovascular coupling.
Acupuncture, one of the most extensively studied sensory interventions, exerts neuroprotective effects by activating the vagal–adrenal anti-inflammatory axis and modulating microglial polarization [83]. Experimental studies demonstrate that electroacupuncture reduces neuroinflammation, oxidative stress, and neuronal apoptosis by regulating the phosphoinositide 3-kinase Akt signaling (PI3K/Akt) and the Nrf2/HO-1 (heme oxygenase-1) pathways. Moreover, acupuncture enhances cerebral blood flow and synaptic plasticity, thereby improving motor and cognitive performance in animal models of PD and AD [84–86].
Aromatherapy and olfactory stimulation influence neurocognitive and emotional processes via direct projections from the olfactory bulb to limbic structures such as the amygdala and hippocampus [24, 87]. Essential oils rich in monoterpenes (e.g., linalool, 1,8-cineole, limonene) have demonstrated antioxidant, anti-inflammatory, and cholinergic-modulating properties. Studies suggest that inhalation of lavender, rosemary, or bergamot essential oils can reduce anxiety, improve sleep, and enhance cognitive performance in individuals with MCI, effects that may reflect modulation of GABAergic and serotonergic systems [88–91].
Massage therapy and tactile stimulation are associated with reduced stress, anxiety, and cortisol levels, contributing to improved relaxation and overall well-being in clinical populations [92, 93]. These effects can enhance sleep quality, relieve anxiety, and attenuate pain perception—symptoms that commonly worsen neurodegenerative disease burden. Mechanistically, tactile input activates C-tactile afferents, triggering oxytocin release and promoting neuroendocrine stability and emotional well-being [94].
In addition to traditional sensory therapies, non-invasive brain stimulation (NIBS) techniques such as transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS) are gaining attention as potential neuroprotective tools [95]. TMS induces electric currents that modulate cortical excitability and promote synaptic plasticity, particularly in motor and prefrontal regions. Repetitive TMS has demonstrated improvements in motor function and mood in PD and HD, and may enhance cortical connectivity in AD [96, 97].
The therapeutic effects of these techniques are highly dependent on stimulation parameters. In repetitive TMS (rTMS), low-frequency stimulation (≤ 1 Hz) is generally associated with inhibitory effects on cortical excitability, whereas high-frequency stimulation (≥ 5 Hz) enhances excitability [98]. Stimulation intensity is typically defined relative to the individual motor threshold, ensuring both safety and reproducibility. In contrast, tDCS modulates neuronal membrane potentials via weak electrical currents, with anodal stimulation increasing cortical excitability and cathodal stimulation decreasing it [99]. Treatment protocols vary widely in terms of session duration, number of sessions, and cumulative exposure, contributing to heterogeneity in clinical outcomes.
Targeted brain regions play a crucial role in determining therapeutic outcomes. In neurodegenerative disorders, the dorsolateral prefrontal cortex (DLPFC) is frequently targeted to improve cognitive function and mood regulation, particularly in AD and MCI [100]. Stimulation of the primary motor cortex and supplementary motor area has been explored in PD to enhance motor performance and reduce bradykinesia [101]. These region-specific approaches reflect attempts to modulate dysfunctional neural circuits, although optimal targeting strategies remain under investigation.
Similarly, tDCS, by applying weak electrical currents to targeted cortical areas, facilitates long-term potentiation-like mechanisms and may enhance cognitive performance [102]. The neuroprotective potential of these modalities is supported by increased BDNF expression, reduced levels of neuroinflammatory cytokines, and restoration of disrupted neural networks. When combined with behavioral or rehabilitative therapies, NIBS may potentiate functional recovery through activity-dependent synaptic reinforcement [103].
Despite promising findings, several limitations constrain the clinical translation of these techniques. Considerable heterogeneity exists in stimulation protocols, including differences in frequency, intensity, electrode placement, and treatment duration, making comparisons across studies challenging [104]. Small sample sizes, short follow-up periods, and variability in outcome measures limit many clinical trials. Furthermore, inter-individual differences in brain anatomy and disease progression may influence responsiveness to stimulation. As a result, the long-term efficacy and optimal therapeutic parameters of TMS and tDCS remain to be fully established, highlighting the need for standardized, large-scale clinical trials [105].
Emerging research supports the use of multisensory stimulation, which combines visual, auditory, tactile, and olfactory cues to engage distributed brain networks simultaneously. In dementia care, multisensory environments (Snoezelen therapy) have been associated with improved mood, attention, and communication. Similarly, light therapy targeting circadian rhythms can improve sleep–wake regulation and cognitive alertness, particularly in AD and PD [106].
Combining sensory and neuromodulatory strategies with exercise, mindfulness, or nutritional interventions may amplify their effects by engaging overlapping neuroimmune and neuroendocrine pathways. These multimodal approaches can facilitate neuroplastic remodeling, stabilize affective states, and improve quality of life—aligning with a patient-centered model of integrative neuroprotection [8, 42].
While evidence from preclinical and clinical studies supports the neuroprotective potential of sensory and neuromodulatory interventions, methodological heterogeneity and limited sample sizes remain challenges. Standardization of protocols, identification of optimal stimulation parameters, and the use of objective biomarkers (e.g., neuroimaging, electrophysiology, and inflammatory markers) are essential to advancing translational validity.
Future directions should prioritize neurophenotyping approaches to identify responders to specific sensory modalities, leveraging wearable and digital technologies for real-time monitoring. Integration of these interventions into conventional neurorehabilitation could bridge mechanistic understanding and clinical application, positioning sensory and neuromodulatory therapies as valuable complements within comprehensive neuroprotective frameworks. From a vitactions standpoint, these interventions may be particularly valuable when tailored to individual sensory preferences, enhancing both adherence and therapeutic responsiveness.
The evidence supporting sensory and neuromodulatory interventions is evolving but remains less robust than that in other domains. While preclinical studies and early-phase clinical trials suggest beneficial effects on neuroinflammation, neurotransmission, and functional outcomes, high-quality randomized controlled trials are still limited. Techniques such as TMS and tDCS show promising but variable results, with outcomes highly dependent on stimulation parameters and target regions. Similarly, interventions such as aromatherapy, acupuncture, and multisensory stimulation are often supported by small-scale studies with heterogeneous designs and subjective endpoints. This variability underscores the need for standardized protocols, larger clinical trials, and objective biomarkers to establish efficacy and reproducibility better.
Evidence in this domain remains largely preclinical or based on small clinical studies, with symptomatic effects more consistently reported than disease-modifying outcomes.
A summary of the main non-pharmacological interventions, their mechanisms, target populations, and clinical outcomes is presented in Table 1.
Summary of non-pharmacological interventions and clinical evidence.
| Intervention type | Mechanisms | Target population | Key outcomes | References |
|---|---|---|---|---|
| Exercise | ↑ BDNF, ↑ mitochondrial function, ↓ inflammation | AD, PD, MCI, HD | Cognitive function, motor control, mood | [6, 25–28, 31–34] |
| Diet (Mediterranean, MIND) | Antioxidant, anti-inflammatory, gut–brain modulation | AD, PD, HD | Cognitive performance, disease progression | [11, 38, 46, 48, 50, 52] |
| Mind–Body (MBSR, Tai Chi, Yoga) | ↓ HPA axis, ↑ neuroplasticity, ↓ cytokines | AD, PD, MS, HD | Cognition, balance, mood, QOL | [59, 62, 65, 70, 74] |
| Sensory and neuromodulatory | ↑ Neurotransmitter modulation, ↓ inflammation, ↑ cortical connectivity | AD, PD, HD; dementia | Mood, cognition, sleep, motor function | [80, 81, 84, 87, 93, 94] |
↑: increases; ↓: decreases; AD: Alzheimer’s disease; HD: Huntington disease; MBSR: mindfulness-based stress reduction; MCI: mild cognitive impairment; MIND: Mediterranean-Dietary Approaches to Stop Hypertension Intervention for Neurodegenerative Delay; MS: multiple sclerosis; PD: Parkinson’s disease; QOL: quality of life; BDNF: brain-derived neurotrophic factor; HPA: hypothalamic–pituitary–adrenal.
Despite growing recognition of the neuroprotective potential of non-pharmacological and integrative interventions, significant challenges remain in establishing their efficacy, mechanistic clarity, and translational validity. Addressing these gaps is essential to advance non-drug strategies from supportive care to evidence-based, mechanistically informed components of neuroprotective therapy.
One important limitation across the reviewed literature is the substantial heterogeneity in intervention protocols, including differences in duration, frequency, intensity, and delivery formats. This variability is particularly evident in non-pharmacological approaches such as exercise, dietary interventions, mind–body therapies, and sensory or neuromodulatory techniques. As a result, direct comparisons across studies are challenging, and the identification of optimal therapeutic parameters remains limited. This heterogeneity also complicates the inclusion of standardized dosage or protocol-specific data within integrative summaries. Future research should prioritize the development of harmonized intervention frameworks and reporting standards to enable more precise characterization of dose–response relationships and facilitate clinical translation.
Current studies are characterized by wide variability in intervention protocols, intensity, duration, and outcome measures. This heterogeneity limits the ability to compare results and draw firm conclusions regarding dose–response relationships or long-term efficacy [107]. Many trials rely on small samples, short follow-up periods, and subjective endpoints. The absence of standardized biomarkers for neuroprotection—such as validated imaging, electrophysiological, or molecular measures—further complicates interpretation [108]. Future research should adopt multicenter randomized designs, harmonized outcome measures, and biomarker-driven endpoints to enhance reproducibility and clinical relevance.
Another important consideration is the variability in the strength and type of evidence supporting different non-pharmacological interventions. While some approaches—such as physical exercise and certain dietary patterns—are supported by randomized controlled trials and longitudinal studies, others remain primarily grounded in preclinical research or small-scale clinical investigations. In addition, inconsistent findings and occasional negative or neutral results have been reported, often reflecting differences in study design, patient populations, and intervention protocols. This heterogeneity underscores the need for careful interpretation of current evidence and highlights the importance of distinguishing between well-established, emerging, and exploratory interventions in future research.
A critical challenge lies in translating promising preclinical data into clinical practice. Animal models rarely replicate the full complexity of human neurodegenerative disease, and the physiological effects of lifestyle interventions often differ across species. Furthermore, non-pharmacological approaches do not fit neatly into traditional regulatory pathways for drug approval, posing barriers to standardization, quality control, and reimbursement [109, 110]. Collaboration between neuroscientists, clinicians, and regulatory authorities will be necessary to define evaluation frameworks that capture both functional and mechanistic outcomes of these interventions.
The integration of non-pharmacological interventions into standard neurological care remains inconsistent. Clinical implementation requires interdisciplinary models that combine neurology, rehabilitation, nutrition, psychology, and complementary medicine. Effective delivery also depends on training health professionals in evidence-based protocols and ensuring patient adherence through accessible, culturally adapted programs [111]. The incorporation of these interventions into multimodal care pathways could enhance treatment personalization and reduce reliance on pharmacological polytherapy.
Beyond mechanistic alignment and biomarker-driven precision, a patient-centered conceptual lens may further strengthen the translational integration of non-pharmacological therapies. The framework of “vitactions”, recently proposed in the context of neuroprotective therapy, conceptualizes structured, essential “life-actions” as foundational components of prevention and treatment. Rather than viewing non-pharmacological strategies merely as adjunctive prescriptions (e.g., exercise sessions or dietary plans), the vitactions perspective reframes them as inherently meaningful, intrinsically motivating activities embedded within the individual’s daily life context. This approach emphasizes alignment with patient preferences, emotional readiness, and existing routines—factors that critically influence adherence and long-term sustainability [10, 112].
From a translational standpoint, this framework complements mechanistic models by introducing an adherence-centered dimension. Conventional non-pharmacological interventions are often protocol-driven and condition-specific, potentially overlooking psychological engagement and reward pathways that modulate neuroplasticity and stress physiology. By integrating biologically informed selection (as outlined in Figure 2) with patient-valued, rewarding activities, vitaction-oriented strategies may enhance sustained behavioral change and amplify neuroprotective effects through reinforcement learning mechanisms and dopaminergic engagement [113].
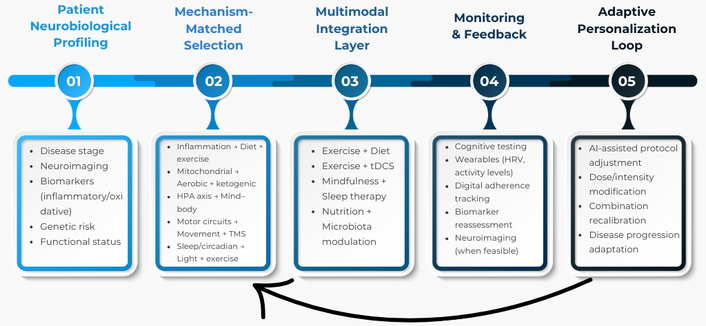
Translational precision pipeline for integrative neuroprotection. Patients are first stratified using clinical, neuroimaging, and molecular biomarkers. Interventions are then selected based on dominant pathophysiological mechanisms and combined in multimodal protocols. Continuous monitoring using functional, digital, and biological markers enables adaptive personalization through a feedback-loop model aligned with precision medicine principles. HRV: heart rate variability; tDCS: transcranial direct current stimulation; TMS: transcranial magnetic stimulation.
The future of non-pharmacological neuroprotection lies in personalized and technology-supported interventions. Advances in digital health—such as wearable sensors, virtual reality rehabilitation, and mobile applications for mindfulness and exercise tracking—enable real-time monitoring of behavioral adherence and physiological responses. Integration of artificial intelligence and machine-learning algorithms can support individualized adaptation of interventions based on biomarkers, genetics, and digital phenotyping data [114, 115]. Moreover, combining multiple approaches (e.g., physical activity, dietary modulation, and stress-reduction practices) may achieve synergistic effects by targeting convergent neurobiological pathways.
To operationalize the integration of non-pharmacological strategies within clinical practice, a translational precision framework is proposed (Figure 2). This pipeline model emphasizes patient stratification based on clinical phenotype, disease stage, neuroimaging findings, and molecular biomarkers, followed by mechanism-matched selection of targeted interventions. Rather than applying uniform lifestyle recommendations, this approach aligns specific therapies—such as exercise, dietary modulation, mind–body practices, or neuromodulatory techniques—with dominant pathophysiological drivers, including neuroinflammation, mitochondrial dysfunction, or HPA axis dysregulation. The model further incorporates multimodal integration, objective monitoring through functional and biological markers, and adaptive personalization using digital tools and iterative feedback. By structuring integrative neuroprotection within a closed-loop, precision-based system, this framework bridges mechanistic insights and clinical implementation, advancing the field toward individualized, biomarker-informed therapeutic strategies. Within the proposed precision pipeline (Figure 2), the vitactions perspective may operate primarily at the levels of intervention selection and adaptive personalization, ensuring that mechanistically matched therapies are also intrinsically meaningful and behaviorally sustainable.
As integrative neuroprotective strategies become more widely promoted, ensuring equity of access and scientific integrity is paramount. Socioeconomic disparities, digital literacy gaps, and cultural factors may affect participation and adherence. Ethical frameworks must emphasize informed consent, realistic expectations, and transparency in communication of benefits and limitations.
Non-pharmacological and integrative interventions offer a promising, multidimensional approach to neuroprotection in neurodegenerative disorders, targeting key mechanisms such as oxidative stress, neuroinflammation, synaptic dysfunction, and impaired neuroplasticity. Evidence from preclinical and clinical studies demonstrates that structured physical activity, dietary modulation, mind–body practices, and sensory or neuromodulatory therapies can improve cognitive, motor, and emotional outcomes, often exerting synergistic effects when combined. While methodological heterogeneity and translational challenges remain, the integration of biomarkers, personalized protocols, and digital monitoring tools provides a path toward optimized, patient-centered neuroprotective strategies. Ultimately, these approaches should complement pharmacological therapy, forming a holistic framework to slow disease progression, enhance quality of life, and advance the field of translational neurotherapeutics.
AD: Alzheimer’s disease
ALS: amyotrophic lateral sclerosis
BDNF: brain-derived neurotrophic factor
HD: Huntington’s disease
HPA: hypothalamic–pituitary–adrenal
IL-1β: interleukin-1 beta
KD: ketogenic diet
MBSR: mindfulness-based stress reduction
MCI: mild cognitive impairment
MIND: Mediterranean-Dietary Approaches to Stop Hypertension Intervention for Neurodegenerative Delay
MS: multiple sclerosis
NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells
NIBS: non-invasive brain stimulation
Nrf2: nuclear factor erythroid 2-related factor 2
PD: Parkinson’s disease
ROS: reactive oxygen species
tDCS: transcranial direct current stimulation
TMS: transcranial magnetic stimulation
TNF-α: tumor necrosis factor-α
SDG: Conceptualization, Methodology, Supervision, Resources, Project administration, Visualization, Validation, Writing—original draft. SDG, RGD, NM, AC, and VE: Investigation, Writing—review & editing. All authors read and approved the submitted version.
The authors declare that they have no conflicts of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
© The Author(s) 2026.
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.
Copyright: © The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
View: 874
Download: 24
Times Cited: 0
Ningyun Hu ... Rong Ma
Afra Wasama Islam ... Saumya Rajesh Kadam
